Abstract
Background
Physical use of the affected upper extremity can have a beneficial effect on motor recovery in people after stroke. Few studies have examined neurological mechanisms underlying the effects of forced use in non-human primates. In particular, the ventral premotor cortex (PMV) has been previously implicated in recovery after injury.
Objective
To examine changes in motor maps in PMV after a period of forced use following ischemic infarct in primary motor cortex (M1).
Methods
Intracortical microstimulation (ICMS) techniques were used to derive motor maps in PMV of four adult squirrel monkeys before and after an experimentally induced ischemic infarct in the M1 distal forelimb area (DFL) in the dominant hemisphere. Monkeys wore a sleeved jacket (generally 24 hrs/day) that forced limb use contralateral to the infarct in tasks requiring skilled digit use. No specific rehabilitative training was provided.
Results
At 3 mos post-infarct, ICMS maps revealed a significant expansion of the DFL representation in PMV relative to pre-infarct baseline (mean = +77.3%; n = 3). Regression analysis revealed that the magnitude of PMV changes was largely driven by M1 lesion size, with a modest effect of forced use. One additional monkey examined after ∼18 months of forced use demonstrated a 201.7% increase, unprecedented in non-human primate studies.
Conclusions
Functional reorganization in PMV following an ischemic infarct in the M1 DFL is primarily driven by M1 lesion size. Additional expansion occurs in PMV with extremely long periods of forced use but such extended constraint is not considered clinically feasible.
Introduction
Acquired brain injuries, such as cortical strokes, frequently result in sensorimotor deficits, particularly in fine manipulative abilities and skilled use of the upper extremities contralateral to the injury. 1,2 Several studies have shown that requiring physical use of the affected upper extremity can have a beneficial effect on recovery of motor function. 3 -5 A commonly adopted approach is Constraint-Induced Movement Therapy (CIMT), which typically involves constraint of the less impaired upper extremity by wearing a padded mitt that restricts use of the hand and is coupled with behavioral training (repetitive and adaptive task practice) of the more impaired limb for up to 6 hours per day for a period of 2 weeks. 3 Variations on the initial protocols increased the duration for up to 90% of waking hours, or provided constraint for 2–5 hours per day for over 50–60 days. 6 Clinical trials in people after stroke have shown significant functional improvements using CIMT with both early (3–9 mos) or delayed (15–21 mos) treatment. 7 Although neuroimaging and non-invasive brain stimulation studies have suggested putative mechanisms for recovery, 8 -10 universal consensus on the neurological mechanisms involved has remained elusive.
There is considerable evidence that functional recovery after cortical injury may be largely due to adaptive plasticity that occurs in intact cortical and subcortical motor areas. 11,12 Structures either adjacent to, or remote from the injured area are thought to compensate for the function of the damaged cortex and contribute to recovery of lost functions. 13 This idea has gained support due to multiple examples of neural plasticity observed after injury. 14 -16
Previous studies have demonstrated that after a focal infarct in the primary motor (M1) distal forelimb area of squirrel monkeys, movement representations in the spared M1 tissue adjacent to the injury are altered. 17 -19 In contrast, distal representations in ventral premotor cortex (PMV) expand 3 mos after a cortical infarct in M1 even in naturally recovering monkeys. 16 It is of great interest to further extend our understanding of the potential modulatory effects of behavioral interventions such as CIMT on neurophysiologic reorganization in premotor cortex. It was the goal of the present study to examine representational changes in PMV after a period of forced use following a large experimentally induced ischemic infarct in the distal forelimb representation of M1 and compare these changes to those observed in naturally recovering monkeys. The results provide insights into two important translational questions: (1) Does constant limb constraint (24 hrs/day) in the absence of intensive rehabilitative training drive greater expansions in premotor maps beyond what is observed during spontaneous recovery? and (2) Are there benefits to extremely long constraint periods than are typically considered feasible in clinical cohorts?
Methods
Intracortical microstimulation (ICMS) techniques were used to derive detailed maps of M1 and PMV distal forelimb (DFL) representations in 4 adult male squirrel monkeys (Saimiri sciureus) before and after focal ischemic infarcts in the DFL area of M1 and periods of forced use of the contralateral impaired limb (Figure 1(A)). Forelimb preference and manual dexterity were assessed using a pellet retrieval task that required the skilled use of the digits. Then, ICMS mapping techniques were used to physiologically identify the M1 DFL representation contralateral to the preferred forelimb, to be targeted for the infarct. The PMV DFL representation was examined to obtain a baseline comparison to post-infarct and post forced-use maps. After derivation of the M1 and PMV forelimb representations, a focal ischemic infarct in the electrophysiologically defined M1 DFL area was induced. Each monkey was fitted with a jacket with a long mesh sleeve sewn closed over the distal forelimb ipsilateral to the M1 lesion (less impaired forelimb). This sleeve restricted the use of the forelimb, encouraging use of the impaired forelimb for fine manipulation and normal cage behavior (Figure 2). The sleeve does not restrict climbing, postural control, or gross manipulation of objects with the jacketed limb. In 3 monkeys during a 3-month recovery period, limited periodic assessment of manual dexterity was conducted (50 trials 1–3 times per week), but no intensive training on the task was conducted. This differs from our previous studies that employed daily repetitive practice on the behavioral task for a few weeks after injury (400–600 trials daily), but no constraint device on the less impaired limb.
20,21
Thus, the novel aspect of the present design was that the monkeys wore the constraint device 24 hours/day, encouraging use of the impaired limb for daily activity. ICMS mapping before and 3 mos after M1 ischemic infarct (case 521). (A): Pre-infarct map of the M1 and PMV DFL area. The location of the intended lesion is outlined with the red contour. Examination of the lesion during post-lesion mapping revealed some sparing of the M1 DFL intended for infarct (blue contour). (B): ICMS mapping of the PMV DFL post-infarct. Each dot represents a microelectrode penetration site. For sites where multi-joint movements could be evoked, the color at the center depicts the movement at the lowest threshold and the outer ring depicts movement evoked by increasing the stimulation current within 2 μA. Most non-responsive sites (i.e., no movement evoked at 30 μA; black dots) were verified before mapping was complete. Blue dots: proximal movements; green: wrist/forearm; red: digit; yellow: orofacial. Scale bars = 1 mm. Jacketed Pellet Retrieval Task: Each monkey was fitted with a jacket with a long mesh sleeve sewn closed over the distal forelimb ipsilateral to the M1 lesion (unimpaired forelimb). This sleeve restricted the use of the unimpaired forelimb, encouraging the use of the impaired, unrestricted forelimb for any fine manipulation and normal cage behavior.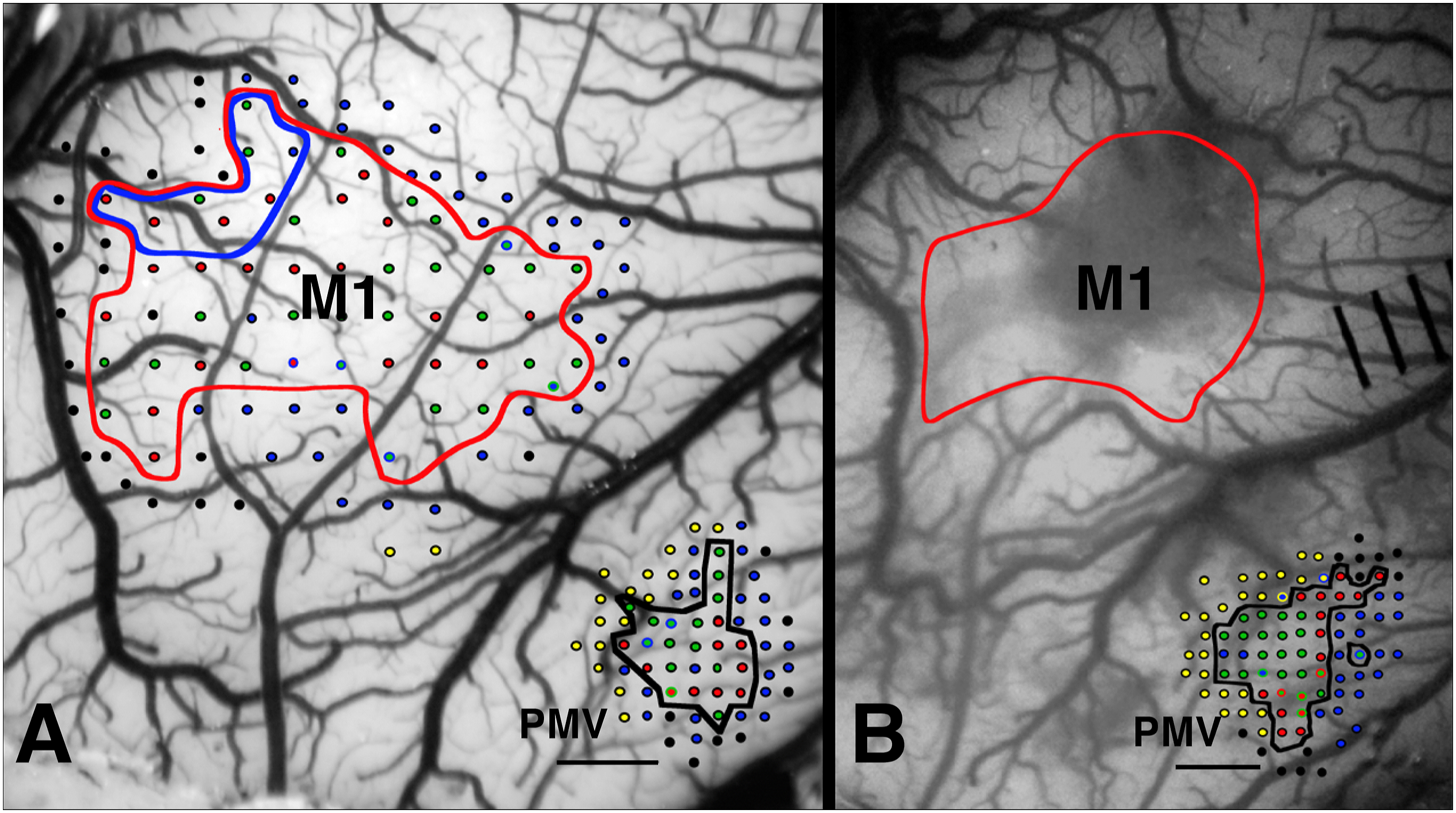

At the end of the 3-month period a second representational map of both the M1 and the PMV DFL area was derived in 3 monkeys (Figure 1(B)). Results were compared to historical controls from a previous study in monkeys that did not experience forced use via the jacket and sleeve after the infarct (i.e., natural, or spontaneous recovery; n = 5). 16 In a fourth monkey (950), jacketing was intermittent (removed for 30 days during the first 3 post-infarct months, and then placed on for 15 months). A post-lesion map was derived at 18 months post-infarct to examine changes in PMV with long-term forced use. Two of the monkeys (983 and 950) had previously been used as non-trained controls in a study examining map changes with motor training (completed 6+ months prior to the PMV baseline map and M1 infarct). 22
Behavioral Methods
To assess forelimb preference and manual dexterity, random probe trials using a modified Klüver board were conducted periodically (Figure 2). This entailed presentations of 45-mg banana flavored food pellets in wells of five different diameters ranging from 9.5 mm (well 5) to 25.0 mm (well 1). Typical retrieval of food pellets from the smallest well required the insertion of one or two digits, as well as specific movement combinations. 17 Dexterity was assessed as the number of digit flexions needed to retrieve food pellets from each well. Each trial was video recorded for later analysis. During probe trials for assessment of forelimb preference, a single food pellet was placed randomly into one of the five wells and the monkey was allowed to retrieve it with either forelimb. A reach was defined as the passing of the distal forelimb completely through the cage bars. A reach ended after the distal forelimb was withdrawn back through the cage bars into the cage. The forelimb used on the initial reach, the forelimb used for subsequent reaches and the forelimb used to dislodge or retrieve the pellet was recorded. Five probe trials for each well size were conducted with well 1 (largest well) to the monkey’s left and then again with the board repositioned with well 1 to the right, for a total of 50 trials in each forelimb preference session. Three sessions conducted during the week prior to the pre-infarct map were used to determine pre-infarct forelimb preference and dexterity performance. The baseline forelimb preference score was derived using the average of the percentage of left-forelimb initial reaches and percentage of left-forelimb retrievals. 23 Sessions consisting of unjacketed probe trials were conducted during the initial post-infarct period and at several times later to track forelimb preference. During the first four weeks, three sessions were conducted each week with the less impaired limb jacketed and one to three sessions were conducted each week during the final 8 weeks. Video records of individual trials were analyzed to determine the total number of finger flexions per retrieval, and then averaged for each day and each week. Behavioral data were normalized to pre-infarct performance and mean flexions per retrieval analyzed using the Dunnett’s test for pairwise mean comparisons to examine differences between pre-infarct performance and each weekly post-infarct performance.
Surgical and Electrophysiological Procedures
An ICMS mapping procedure was conducted in each monkey to identify the DFL representation (digits, wrist, and forearm) and its borders with the proximal forelimb (shoulder and elbow) and face representations in both M1 and PMV of the hemisphere contralateral to the preferred limb. 16,18 Briefly, under aseptic conditions, a craniotomy was performed over M1 and PMV under halothane/nitrous oxide gas anesthesia. A cylinder was affixed over the opening and filled with warm, sterile silicone oil. Gas anesthesia was then withdrawn and ketamine was administered. Throughout the experimental procedure, core temperature and vital signs were monitored. Intracortical microstimulation mapping procedures were then conducted under ketamine anesthesia. The microelectrode consisted of a platinum wire insulated by a glass micropipette filled with 3.5 M NaCl (impedance ∼600 kΩ) that was advanced perpendicular to the cortex to a depth of ∼1750 μm (targeting layer 5). Movements were evoked by a stimulus of thirteen 200 µs cathodal pulses delivered at 350 Hz repeated at 1/sec. Interpenetration distances of 250 μm and 500 μm were used in the PMV and M1 DFL areas, respectively. Movements evoked by ICMS at near-threshold levels defined movement fields (maximum current: 30 μA). From these neurophysiologic data, representational map boundaries were determined to outline different cortical efferent zones (digit, wrist/forearm, etc.). Care was taken to maintain a relatively stable anesthetic state throughout the procedure. Further details of ICMS procedures and discussion of possible sources of variation in these ICMS-derived motor maps have been presented previously. 18,23,24
Infarct Procedure
After derivation of baseline maps, surface blood vessels supplying the M1 DFL area were permanently occluded by using microforceps connected to a bipolar electrocoagulator. This technique consistently produced focal, columnar infarcts through all six layers of the cerebral cortex. This model was not designed to mimic clinical stroke per se, but to provide a reliable method for producing physiologically identified ischemic infarcts. The infarcts were predictable in size and did not affect the underlying white matter. 17,19 After surgical closing, the animal was monitored in a temperature-controlled incubator until it was awake and alert. Animals were cared for in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and with the approval of the University of Kansas Medical Center Institutional Animal Care and Use Committee.
Map Construction and Areal Measurements
Representational maps were generated by a computer algorithm that used the x-y locations of electrode penetrations to establish unbiased borders midway between adjacent sites with different response representations. The DFL representation was defined as cortical regions in which ICMS evoked movement of the digits or wrist/forearm, but not elbow or shoulder movements.
Due to necrosis and scavenging of the infarcted tissue, histological examination, although useful for verifying that all layers of the infarcted area were destroyed, could not be used to accurately define the volume of chronic lesions. A less direct method was used that takes advantage of the fact that after vascular electrocoagulation the ischemic cortex becomes blanched and easily distinguishable from non-infarcted tissue. Therefore, the post-infarct estimate of intact M1 DFL area was derived by comparison of the digital photograph of the intact vasculature taken during the post-infarct mapping procedure with reference to the pre-infarct photograph taken before the ischemic lesion. 16,24 Any part of the pre-infarct M1 DFL area with intact vasculature at 3 mos post-infarct was considered to be spared M1 (Figure 1). The viability of this tissue was confirmed by the presence of ICMS-evoked movements in the zone with intact vasculature. Using this estimation technique, the areal extent of the M1 surface destroyed by the infarct as well as the cortical area spared by the infarct, was determined. PMV DFL area measurements were derived from the ICMS maps of PMV before and 3 months after the ischemic infarct and forced use in 3 monkeys, and after 18 months of forced use in a fourth monkey. Area differences were compared using a paired Student’s t-test. Comparisons with previous monkeys that experienced natural (spontaneous) recovery were conducted using a repeated measures ANOVA and Fishers LSD Post Hoc analysis. 25 To determine the differential effects of forced use vs lesion size, first, linear regression analysis was used to examine the relationship of lesion size to change in PMV DFL, and then a Student’s t-test was used to determine whether the change in PMV DFL was modified in forced use monkeys beyond what would be expected on the basis of lesion size.
After completion of these experiments, each monkey was given a lethal dose of sodium pentobarbital (100 mg per kg of body weight) and perfused transcardially for neurohistological examination.
Results
Behavioral Results of Infarct in M1
The mean baseline forelimb preference in the four monkeys was 99.4% with the left forelimb. Three of the four monkeys demonstrated a change in the preferred forelimb during the first week post-infarct in unjacketed Klüver board tests, resulting in a group mean preference for the previously dominant forelimb of 29.1% in post-infarct week 1. One of these monkeys returned to preferred use of the original forelimb at 3 mos post-infarct, while two maintained a preference for using the limb ipsilateral to the infarct.
In the first 2–3 days post-infarct there was little voluntary use of the affected limb for either locomotion or food procurement. There was an initial deficit in the ability to retrieve food pellets with the contralateral forelimb in 2 monkeys, (Figure 3, top). Percent successful pellet retrievals decreased in 2 monkeys post-infarct, then rapidly improved for one monkey (normal at week 2) and gradually improved for the other (normal at week 7). Behavioral performance measures of the contralateral hand for each monkey pre- and post-infarct in weekly epochs. Top: Percent Successful Retrieval Per Attempt. An attempt was scored each time the monkey reached onto the Klüver board and attempted to take a pellet from a well. The attempt was scored a success if the pellet was removed from the well. (A): Percent successful retrieval for all wells. (B): Percent Successful retrieval for well 5 only (the smallest well). Bottom: Normalized motor performance in weekly epochs. The motor performance index is the number of flexions per retrieval divided by the baseline (pre-infarct) flexions per retrieval for each animal from each well. (C): the normalized motor performance of the contralateral hand for well 4 and (D): for well 5 (the smallest wells). There was an initial (week 1) impairment in performance in all 3 monkeys on the smallest well (5).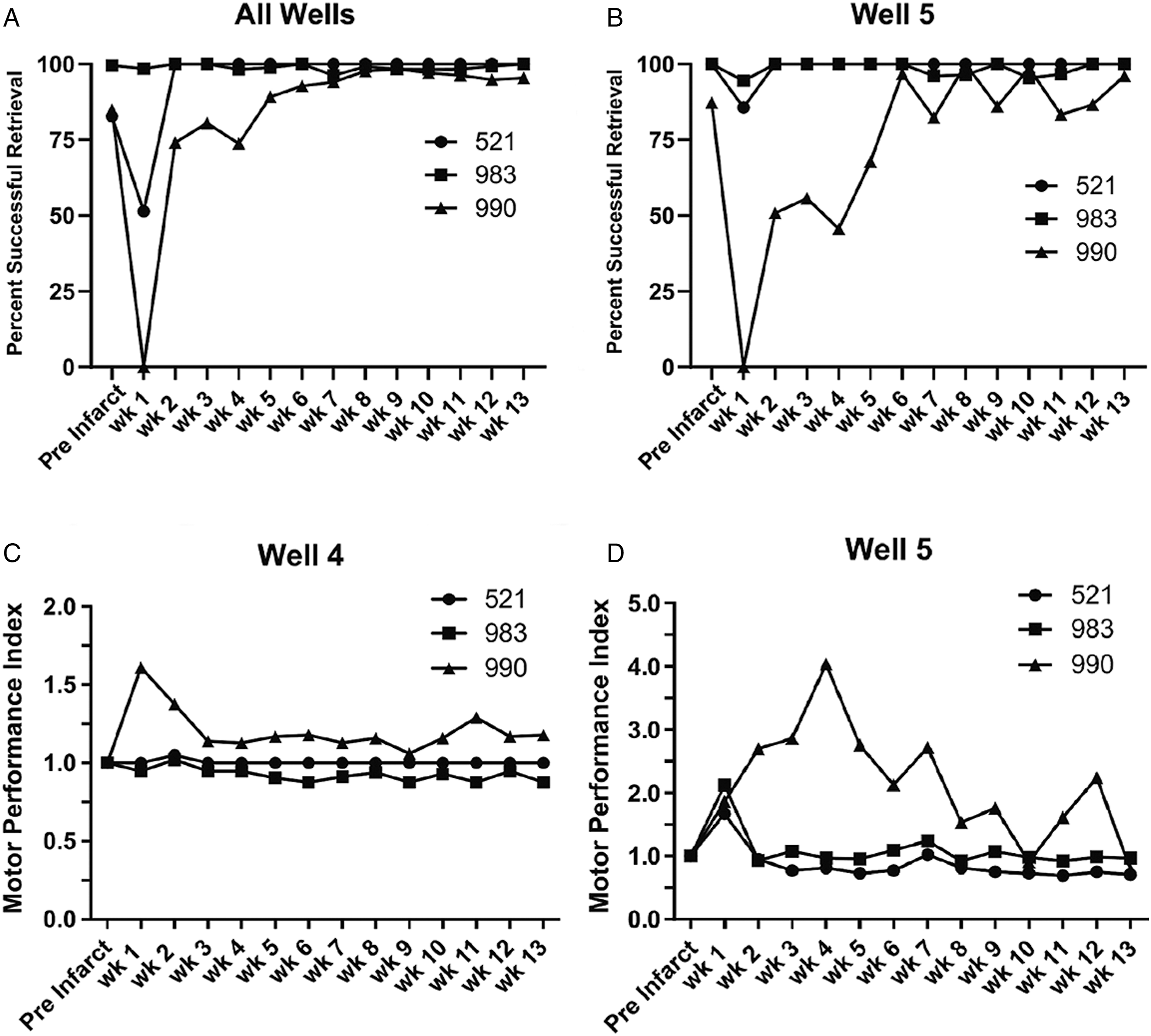
Evidence of a motor deficit was reflected in an initial increase in motor performance scores for the smallest well (well 5; Figure 3(D)). A rapid improvement in flexions per retrieval was observed in 2 monkeys; motor performance scores returned to normal in week 2. One monkey demonstrated an initial impairment on the largest wells (1-3). There was no significant post-infarct impairment in mean group performance on the largest wells (Figure S1).
Behavioral Comparison to Natural Recovery
Several behavioral comparisons suggest that even though the average lesion size was somewhat larger in the forced use monkeys, deficits were less severe than seen with natural recovery. 16 Naturally recovered monkeys could not retrieve pellets in week 1 and had a significant deficit in ability to retrieve pellets from the smallest well (well 5) until post infarct week 3. The mean motor performance index of naturally recovered monkeys on well 5 was 2.5X baseline in week 2, and although the group average gradually improved, it did not return to normal. Forced use monkeys had a mean motor performance index of 1.5X baseline at week 2 (2 monkeys having reached normal performance levels) and a group average that returned to normal at week 10 (Figure S2).
Naturally recovered monkeys displayed a deficit in mean motor performance on well 4 that returned to baseline levels at post infarct week 4; and a deficit on well 3 that returned to baseline in week 3. The mean well 4 performance of forced use monkeys returned to normal in week 2 (only one monkey exhibiting a deficit in weeks 1 and 2) and there was never a deficit in mean performance on well 3.
Functional Reorganization in Ventral Premotor Cortex
Three mos forced use. Comparison of ICMS maps of movement representations in PMV before and after the infarct revealed substantial changes in the DFL representation in the PMV cortex ipsilateral to the experimentally induced infarct in each monkey (Figure 4). Concurrently, ICMS mapping and examination of M1 3 mos after the ischemic lesion revealed a decrease in the M1 DFL area in all monkeys (Figure 5(A)). Partial survival of cortical tissue in the M1 DFL area was seen in 2 monkeys (521, 990). The absolute size of the infarcted M1 area ranged from 11.2 to 19.6 mm2. This variation was, in part, due to the variation in size of the pre-infarct DFL in individual monkeys. The proportion of the original M1 DFL area that was infarcted ranged from 85.7% to 100% with a mean loss of 95.6 +/− 5.8%. The results of ICMS mapping of the PMV DFL before and after ischemic infarct in M1 in 4 monkeys. In 3 cases (521, 990 and 983) the second mapping procedure was conducted at 3 mos post-infarct with forced use. In the 4th case (950) the post-infarct map was conducted at 18 mos post-infarct with forced use 2 out of the first 3 months and all of the last 15 months. ICMS mapping revealed a net expansion in the PMV DFL representation in each monkey. Different colors represent the evoked movement(s) of particular body parts at near threshold levels at each penetration site. Scale bars = 1 mm. Distal Forelimb Area Change. The results of ICMS mapping of the M1 and PMV DFL in 3 monkeys before (Prelesion) and 3 mos after the ischemic infarct in M1 (Postlesion). (A): M1 DFL Area. Large decreases in the DFL were seen in all 3 monkeys. Although the entire DFL was targeted for infarct, partial retention of spared DFL was seen in 2 monkeys. The decrease in absolute M1 DFL ranged from 11.15 mm2 in one monkey (521) to 18.49 mm2 in another (990). (B): PMV DFL. There was an increase in the total area of the DFL representation in PMV at 3 mos post-infarct in all 3 monkeys. This increase ranged from .9 mm2 (521) to 2.2 mm2 (990). (C): Average Change in PMV DFL area after 3 mos with forced use. Post-infarct maps of the PMV DFL area revealed a significant increase across 3 monkeys at 3 mos. Post infarct, with an average increase of ∼1.5 mm2. (paired t-test p = .0271; Error bars are +/− S.E).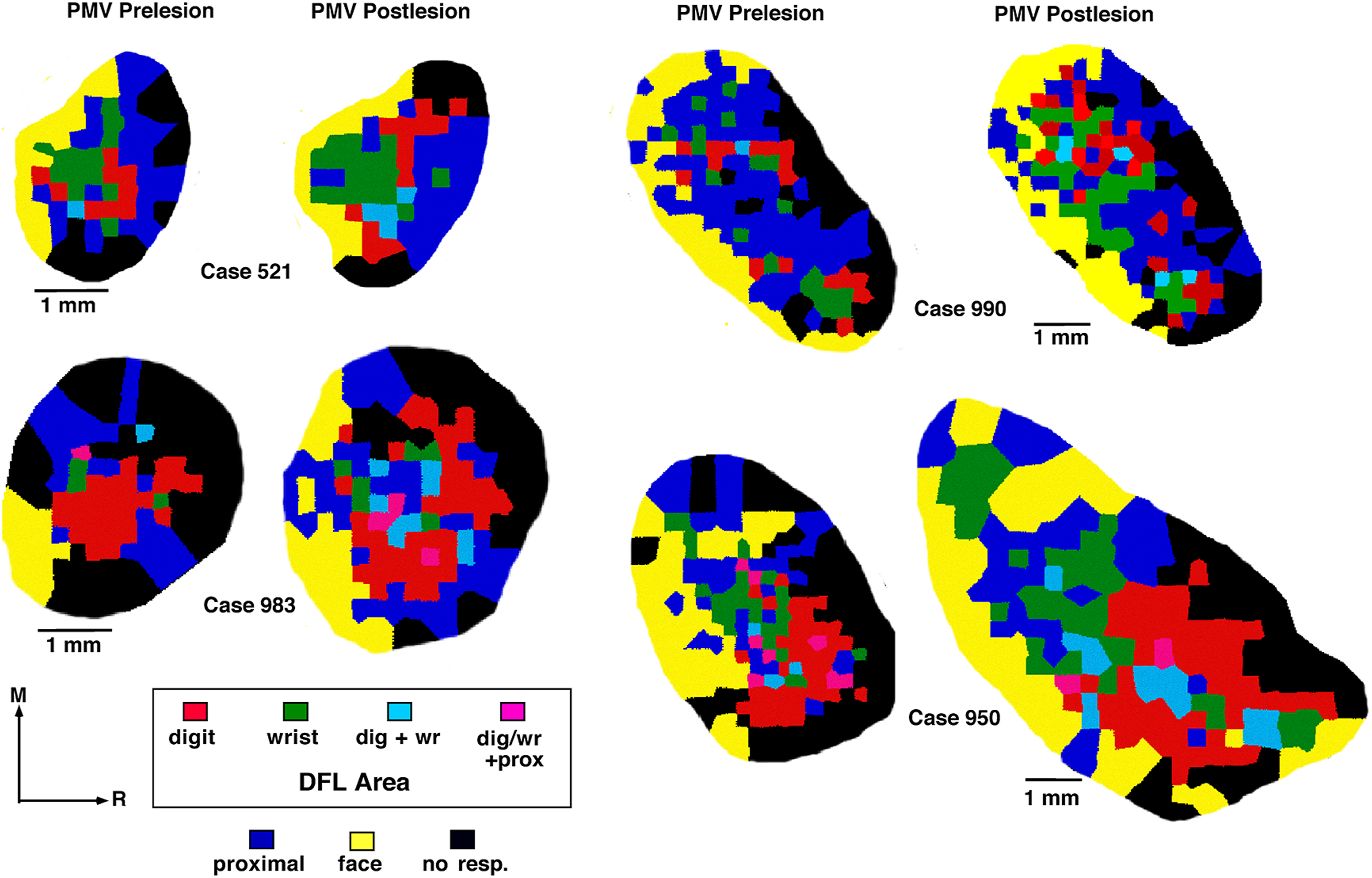
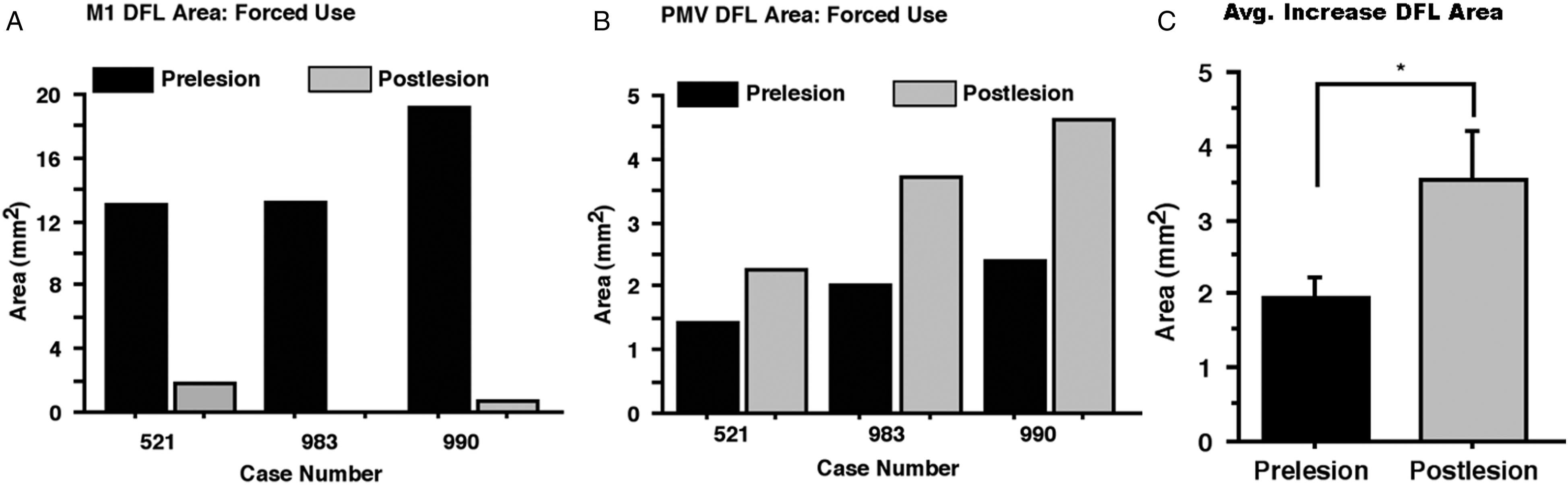
Measurements of the movement representations in PMV before and 3 mos after the infarct revealed an enlargement of the DFL representation in the PMV cortex ipsilateral to the experimentally induced infarct in each monkey (Figure 5(B)). This net areal expansion ranged from ∼.9 mm2 (i.e., from 1.4 to 2.3 mm2) to 2.2 mm2 (i.e., from 2.4 to 4.6 mm2). These increases corresponded to a 56.3, 83.7% and 92.9% increase relative to the initial PMV DFL area, with a mean increase of 77.4 +/− 10.9%. The absolute average increase in total PMV DFL area was significant in the 3 monkeys at 3 mos post-infarct (Figure 5(C), paired t-test: t = 4.118147; p = .0271). The monkey that had the smallest absolute and smallest relative infarct in the M1 DFL area had the smallest expansion in the PMV DFL representation (case 521).
When the PMV DFL representation was subdivided into component digit and wrist-forearm representations, large increases in digit and wrist areas were observed but with substantial variance, particularly in the wrist-forearm representation. The digit area increased in all 3 monkeys at 3 mos (71.9, 82.6, and 102%, respectively) with a mean increase of 85.5 +/− 8.8% (not significant, P > .05). The wrist-forearm area increased in all 3 monkeys (62.3, 264, and 100%) with a mean increase of 142.1 +/− 61.9% (not significant, P > .05).
Functional Reorganization With Forced Use Compared to Natural Recovery
The percent change in PMV digit, wrist and total DFL area after 3 mos of post-infarct forced use is compared to results from 5 monkeys that were naturally recovered for 3 mos (i.e., no forced use) in Figure 6, (A) & (B). The percent change in total DFL area after forced use (+77.4 +/− 10.9%) was significantly greater (>2X) than the changes seen in monkeys that recovered naturally (36.1 +/− 9.7%) (Figure 6(B); ANOVA (F1,6 = 7.35); **P < .05). One potential confounding factor in this comparison is that in the study of naturally recovered monkeys, the proportion of the M1 DFL that was infarcted ranged from 57.0% to 96.5%, compared with 85.7 to 100% in the present study. Since the percent change in PMV area is expected to be linearly related to the percent loss in M1 DFL area, the greater expansion may simply have been due to larger average lesions.
16
However, in the two naturally recovered monkeys with the largest M1 lesions (∼95%), the largest expansion in PMV DFL was 53.8%. This is approximately the lower limit of expansions in the forced use monkeys in the present study. Percent change in PMV DFL area 3 mos after M1 infarct in natural recovery and forced use monkeys. (A): Change in area of digit and wrist representations. (B): Change in total DFL area representation. There was significantly greater increase in PMV DFL area with forced use; ANOVA (F1,6 = 7.35; **P < .05). (C): Percent increase in PMV DFL area as a function of percent anatomical loss of M1 DFL area 3 mos post-infarct. Grey filled circles represent monkeys with forced use recovery and open circles represent naturally recovered monkeys. In general, the larger the infarct area and greater percentage of M1 DFL area lost, the larger the increase in the PMV DFL response area. A regression line was fit to the data (chg %PMV = 1.72 * %M1 loss - 96.52; F = 16.86; p = .0063; R2 = .738), indicating a significant linear relationship between % PMV gain and % M1 loss. (D): Actual minus predicted % PMV gain based on linear regression. PMV gain was somewhat less than predicted for natural recovery and somewhat greater than predicted for forced use. Note that in the long-term forced use monkey (black bar), the actual % PMV gain was over 120% greater than predicted by the model.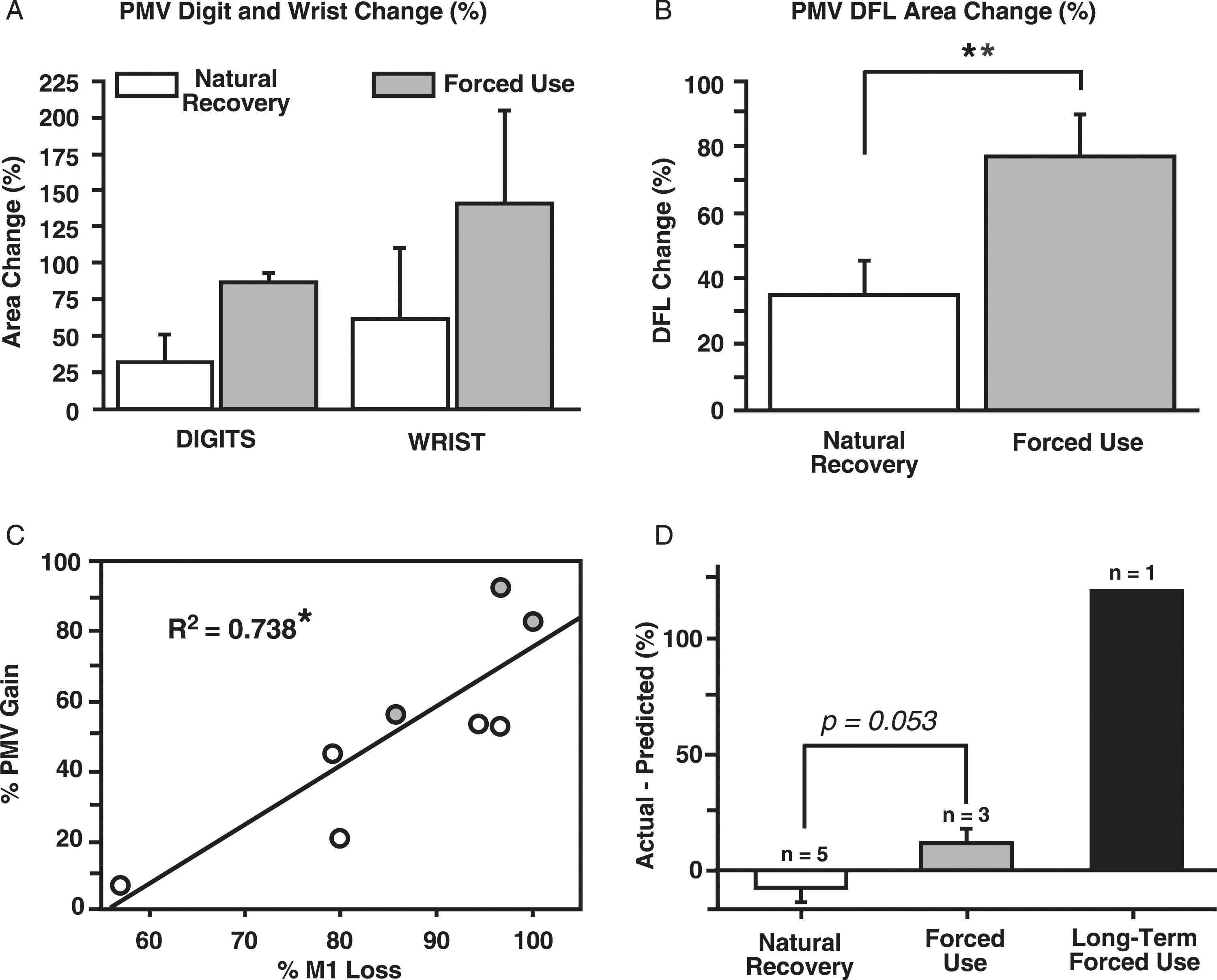
The relationship between M1 lesion size and PMV expansion in both forced use and natural recovery is further examined in Figures 6 C and D. In general, the larger the infarct area and greater percentage of M1 DFL area lost, the larger the increase in the PMV DFL area (Figure 6(C)). Linear regression analysis showed that the relationship was significant (F = 16.86; p = .0063; R 2 = .738). To explore the contribution of forced use to changes in PMV DFL, we plotted the residuals for the two groups based on the linear regression model (Figure 6(D)). The bar graphs show the deviation in actual % change in PMV DFL compared with the predicted change. The change in % PMV DFL was slightly less than predicted in the natural recovery group and slightly more than predicted in the forced use group. A t-test determined that these differences were not significant (t = 2.489; p = .0532), suggesting that M1 lesion size is the critical factor explaining the results.
Eighteen mos forced use. In the monkey subjected to long-term forced use (case 950), the PMV DFL area increased from 5.3 mm2 to 16.0 mm2 (Figure 4). This corresponds to a 201.9% increase in the PMV DFL representation. The expected increase based on the linear regression model was only 126.2%. The digit area increased 182% and the wrist-forearm area increased 312%. The area with DFL responses expanded into areas that previously were unresponsive or had evoked movements of the face in the baseline map.
Discussion
Ventral premotor cortex has been previously identified as participating in various aspects of motor planning, execution, and learning in healthy individuals 26,27 and has been implicated in recovery after injury in both human 8,28 and animal studies. 29,30 Transcranial magnetic stimulation studies support the notion that premotor cortex contributes to functional motor recovery in human stroke patients. 8 Neuroimaging studies have shown that altered metabolic and hemodynamic changes occur in premotor cortex after forebrain injury. 9 Ventral premotor cortex activity associated with voluntary hand movements has been shown to increase during the initial recovery period after a lesion of M1 in macaque monkeys. 10 Human stroke survivors with intact premotor cortex have been shown to have better Functional Independence Measure (FIM) scores, especially in gait and mobility, than those that had damage to premotor cortex after middle cerebral artery occlusion. 28 In natural (spontaneous) recovery of upper extremity function after M1 injury in macaque monkeys, initial post-injury behavioral deficits are reinstated with pharmacological inhibition of PMV and dorsal premotor cortex. 30 These results suggest that reorganization in non-primary cortical motor areas such as PMV may provide a neural substrate for adaptive motor behavior that contributes to recovery of motor functions in the upper extremities after stroke.
Many of the neuroanatomical substrates that contribute to adaptive plasticity in the central nervous system are now well established. Increases in the number of synapses per neuron, enhanced dendritic arborizations, and strengthened synaptic responses have each been observed with exposure to complex environments or after skilled motor training. 31 -35 Similar changes have been seen in the intact cortex contralateral to cortical injury after skilled motor training. 33,36,37 Expansion of distal forelimb functional representations has been observed in M1 of both rats and monkeys following motor skill training. 22,24,38 Similar neuroanatomical and neurophysiological changes are expected to occur in anatomically and functionally connected pre-motor and sensory areas in both normal and cortically injured animals, and these changes may be enhanced with behavioral interventions such as skilled motor use or CIMT.
Multiple clinical trials in people after stroke have reported improvements in functional outcomes with CIMT or modified versions of CIMT. 39,40 One randomized controlled trial using an expanded version of CIMT resulted in a significant improvement in everyday use of the more-affected arm in adults with severe upper-extremity hemiparesis, that lasted long after stroke. 40 Functional changes after CIMT, including changes in cortical excitability, metabolic rate, and blood flow have been demonstrated in fMRI studies with human stroke patients. 41,42 Furthermore, structural changes such as increases in the amount of cortical gray matter in sensory and motor cortex have been shown to positively correlate with improvements in arm function following CIMT in human stroke patients. 43
Studies using forced use paradigms in rodent models of stroke have reported correlations between improved behavioral recovery and enhanced neurogenesis and white matter repair (oligodendrogenesis) with forced use. 44,45 Earlier rat studies suggested that enhanced structural plasticity and behavioral recovery promoted by post-stroke forced limb-use may be mediated through the cAMP/PKA/CREB signal transduction pathway. 46 Forced arm use was shown to result in better functional motor performance outcome measures than voluntary exercise alone and result in a quantitative change in the gene expression program linked to recovery through training after stroke. 47 Improved recovery has been demonstrated in a rat model of corticospinal tract (CST) injury with a concurrent increase in CST fiber growth and innervation density in the spinal cord in response to forced use. 48 Overall, these results suggest that areas interconnected with the damaged motor area may undergo substantial neuroanatomical changes with forced use therapy, that may contribute to enhanced recovery profiles.
It is feasible that non-primary motor areas such as PMV contribute significantly to functional recovery following injury in M1. Along these lines, it is important to note that increases in the PMV distal forelimb representation with large M1 lesions (>55%) with or without forced use ranged from 56–96.5% at 3 mos post infarct. In marked contrast, in naturally recovering animals after more limited M1 infarcts (29–49%), the DFL representation in the ipsilateral PMV was seen to decrease at 3 mos post-infarct. 49 These smaller M1 lesions have been shown to result in relatively mild and transient deficits. 50 These mild deficits may only require minor post-lesion learning or compensation strategies for which the remaining uninjured (intact) M1 DFL area would be sufficient for behavioral recovery. 50 Therefore, for the behavioral paradigm tested, small M1 lesions may not be a sufficient driving force to result in the expansion of the distal forelimb representation in premotor cortex.
The main question to be addressed in this study is whether 24 hr/day of forced use can improve motor performance and whether such functional gains are associated with greater reorganization in PMV. First, improvements in behavioral performance on the pellet-retrieval task was more rapid in the forced use monkeys compared to naturally recovered monkeys despite somewhat larger lesions. Second, the significant linear relationship between % PMV gain and % M1 loss demonstrated in previous studies was confirmed with the addition of the forced use monkeys with slightly larger lesions. Third, while PMV gain was somewhat less than predicted in naturally recovered monkeys, and somewhat more than predicted in forced use monkeys, this difference was not significant (p = .053). Thus, the magnitude of the change in PMV DFL was primarily driven by M1 lesion size, with only a small effect of forced use, expected with exceedingly long constraint periods. It should be noted that both groups experienced the same behavioral testing regimen, and the degree that 50 pellet-retrieval trials could introduce a training effect, if any, on map changes in PMV is unclear.
Although only one monkey underwent long-term forced use (∼18 months), the results revealed an expansion of PMV DFL area of over 200%. This amount of expansion in a spared motor area after an M1 lesion in primates is unprecedented and well beyond what the linear regression model predicted. This suggests that motor map reorganization can continue for at least years after the injury, and that forced use is sufficient to drive very large neurophysiological changes, at least with extended constraint.
Even if very long duration constraint can drive neurophysiological plasticity in spared cortical areas, the feasibility of such a protocol beyond implementation in laboratory animals is questionable, as potential dysfunction in the constrained limb must be considered. The use of the ipsilateral forelimb in pre-infarct unjacketed trials was minimal (0.6% average across monkeys), making it difficult to compare to post-infarct performance. It should still be noted that there were no obvious deficits in the ability to use the ipsilateral limb after prolonged jacketing. More direct evidence for functional decline in the constrained limb comes from a previous study in this laboratory, in healthy monkeys. 51 The effects of 5–18 weeks of unilateral forelimb restriction in four healthy squirrel monkeys, using a jacket and sleeve similar to that used in the present study were reported. After the restrictive device was removed, a small but significant atrophy of forearm muscles was observed. Motor skill on the pellet retrieval task was normal in two of the monkeys, and mild impairment was found in one monkey that resolved within several weeks. However, in one monkey with long-duration restriction (35 weeks), a more pronounced deficit was observed that partially resolved only after rehabilitation (restraint of the unaffected limb). 51 Thus, extended periods of forced use in human populations should be considered with caution. Although casting for up to 3 weeks in duration (e.g., in the treatment of orthopedic injuries) is typically deemed benign, longer periods may result in prolonged functional deficits after the restrictive device is removed.
Conclusions
The changes found in the DFL representation in PMV following ischemic infarct in the DFL representation of M1 in both this and previous studies indicate that neurophysiologic reorganization of cortical premotor areas occurs in response to cortical infarct in M1. 16,49 Further, the enhanced increases in the PMV DFL representation observed with forced use, when compared to the relatively smaller increases seen in naturally recovered animals, suggests that forced use therapy may be a catalyst for additional increases in premotor distal forelimb representation area after infarct in the M1 DFL area. Presumably, the substantially increased time devoted to using the impaired forelimb in daily cage activities might have a significant impact on the functional organization of spared cortical motor structures. However, the present results suggest that the magnitude of changes in PMV are primarily a function of M1 lesion size, and the impact of forced use on PMV is relatively modest, except with very long duration constraint periods that are unfeasible in clinical populations.
Supplemental Material
Supplemental Material—Reorganization of Ventral Premotor Cortex After Ischemic Brain Injury: Effects of Forced Use
Supplemental Material for Reorganization of Ventral Premotor Cortex After Ischemic Brain Injury: Effects of Forced Use by Shawn B. Frost, Daofen Chen, Scott Barbay, Kathleen M. Friel, Erik J. Plautz, and Randolph J. Nudo in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgments
The authors thank Diane Steffen Larson for technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH/NINDS [NS030853]; NIA [AG14635]; American Heart Association [0120624Z]; American Heart Association National [RI-G1093660] and the KUMC Training Program in Biomedical Research. Core facility support was provided by the Kansas Intellectual and Developmental Disabilities Center (NICHD [HD002528]). This work has previously been presented in abstract form. 52
Data Availability
Datasets of the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
