Abstract
Background
Improved ability to predict patient recovery would guide post-stroke care by helping clinicians personalize treatment and maximize outcomes. Electroencephalography (EEG) provides a direct measure of the functional neuroelectric activity in the brain that forms the basis for neuroplasticity and recovery, and thus may increase prognostic ability.
Objective
To examine evidence for the prognostic utility of EEG in stroke recovery via systematic review/meta-analysis.
Methods
Peer-reviewed journal articles that examined the relationship between EEG and subsequent clinical outcome(s) in stroke were searched using electronic databases. Two independent researchers extracted data for synthesis. Linear meta-regressions were performed across subsets of papers with common outcome measures to quantify the association between EEG and outcome.
Results
75 papers were included. Association between EEG and clinical outcomes was seen not only early post-stroke, but more than 6 months post-stroke. The most studied prognostic potential of EEG was in predicting independence and stroke severity in the standard acute stroke care setting. The meta-analysis showed that EEG was associated with subsequent clinical outcomes measured by the Modified Rankin Scale, National Institutes of Health Stroke Scale, and Fugl-Meyer Upper Extremity Assessment (r = .72, .70, and .53 from 8, 13, and 12 papers, respectively). EEG improved prognostic abilities beyond prediction afforded by standard clinical assessments. However, the EEG variables examined were highly variable across studies and did not converge.
Conclusions
EEG shows potential to predict post-stroke recovery outcomes. However, evidence is largely explorative, primarily due to the lack of a definitive set of EEG measures to be used for prognosis.
Introduction
Stroke is a leading cause of long-term disability in the United States.1,2 Since stroke is heterogenous, functional ability and treatment response vary greatly among stroke survivors. 3 Currently, due to poor prognostic abilities, clinicians experience difficulty developing personalized treatment plans that maximize patient outcomes. Improved prognostic ability would direct treatment planning and provide individual patients with the maximally efficient course of treatment. Specifically, physicians may utilize patients’ recovery prognosis to determine the most appropriate discharge setting. Once patients are referred to rehabilitation, therapists may utilize patients’ recovery prognosis to set appropriate rehabilitation goals and administer individualized therapy. For example, for patients predicted to require a moderate level of assistance, therapists may focus on caregiver training, teach compensatory techniques, and introduce adaptive equipment. Alternatively, for patients with a prognosis of independence, therapists may focus on restoring function in daily activities, with goals targeted at improving strength and functional ability. Overall, improved prognostic ability can save both the patient and healthcare system time and resources while maximizing outcomes.
To aid with prognosis, many studies have investigated potential predictors of post-stroke outcome including initial clinical assessment, age, sex, time since stroke, and lesion volume. Meta-analysis shows that the initial motor score is the most significant predictor, while time since stroke, age, sex, and lesion volume do not predict recovery. 4 The prognostic utility of the initial clinical score for recovery, however, has recently been shown to be spurious.5,6 Specifically, the effect sizes reported for such findings are likely inflated, meaning the strength of the association between initial scores and recovery may be overly optimistic. 5
Outcome may be better predicted by neural function (i.e., the integrity of neural function within the residual neural circuits post-stroke). 7 In particular, electroencephalography (EEG) may be used to assess neural function and predict post-stroke recovery. While other instruments such as magnetic resonance imaging (MRI)8-12 and transcranial magnetic stimulation (TMS) 13 are also used to assess residual neural resources, EEG offers several compelling advantages. First, EEG provides a measure of direct, functional electrical activity of neuronal assembles in the brain that facilitate neuroplastic changes necessary for motor recovery 14 to occur, as opposed to structural MRI or indirect hemodynamic response in the brain measured with fMRI. 15 While EEG has poor spatial resolution compared to MRI, it has superior temporal resolution, capturing millisecond changes in neural activity relevant for function. 15 EEG also offers measurement of multiple aspects, including integrity of the afferent sensory tract16,17 and the corticospinal tract, 18 as well as local 19 and network 20 electrical activity in the cortex, whereas TMS is limited to measures of corticospinal 21 and corticobulbar 22 tract integrity. Furthermore, EEG has no contraindications, while approximately 20% of stroke survivors cannot undergo MRI or TMS due to contraindications such as metal implants in the body. 23 In addition, EEG costs less than MRI, can be performed at bedside unlike MRI, and continuous EEG monitoring is already used in clinical practice for some stroke patients in the acute hospital setting.
Overall, the objective of this study was to perform a systematic review and meta-analysis to determine the prognostic utility of EEG for post-stroke outcome. Qualitative synthesis of evidence exists in a recent review. 24 To further this knowledge, the present paper provides a quantitative synthesis of evidence with a meta-analysis. In addition, the previous review 24 examined 25 papers exclusive to acute/subacute stroke (<6 months post-stroke) in 4 outcome domains (i.e., mortality, function, epilepsy, and cognition). In contrast, the present paper synthesized 75 papers pertaining to both acute/subacute and chronic stroke (≥6 months post-stroke) in 9 outcome domains (i.e., independence, stroke severity, upper extremity, speech, whole body sensorimotor, balance/gait, cognition, mortality, and level of consciousness). It is important to consider prognosis in the chronic phase given accumulating evidence showing that neuroplasticity and subsequent recovery extends beyond 1-year post-stroke. 25 Through comprehensive qualitative and quantitative synthesis of the literature, we aim to provide an overview of the prognostic potential of EEG in predicting post-stroke outcomes.
Methods
Search Strategy
We followed the PRISMA guidelines for systematic reviews and meta-analyses 26 to examine the prognostic utility of EEG in stroke outcome. A literature search was conducted in PubMed (Medline), Scopus, and CINAHL databases. The search terms used were stroke and electroencephalography or EEG. We developed our search strategy based on consultation with a medical librarian and consideration of the literature. The search included papers published between 1965 and 2021 and was last searched on August 15, 2021.
Inclusion and Exclusion Criteria
Systematic Review
I. Publication a. Inclusion: i. Peer-reviewed journal paper. ii. Written in English.II. Study design a. Inclusion: i. Papers that acquired EEG for clinical and/or research purposes. ii. Papers that examined the relationship between baseline EEG and subsequent stroke related clinical outcome measures. iii. Papers that reported statistical analysis results for prognosis and/or provided data sufficient for independent statistical analysis for prognosis. b. Exclusion: i. Meta-analyses, reviews, clinical guidelines, case studies, commentaries, and trial protocols. ii. Papers that did not measure EEG. iii. Papers that did not include a clinical outcome measure. III. Participant characteristics a. Inclusion: i. Study participants had a stroke(s) of any type.
Meta-Analysis
I. Outcome measure a. Inclusion: i. Outcome measure common in at least 5 papers. b. Exclusion: i. Papers that utilized a modified outcome measure (e.g., dichotomization, proportion, and partial items). II. Statistics a. Exclusion: i. Papers did not provide relevant statistics or data needed to calculate relevant statistics, or authors did not provide data upon request.
Screening
Papers were screened by the primary and the senior author independently. Papers were initially screened based on the title and abstract. For papers that met the inclusion criteria based on the title and abstract, full-text papers were obtained and a subsequent screening was performed to determine if the inclusion criteria were met. The senior author completed 25% of the initial abstract screening and 19% of the full-text screening. In cases of discrepancy, resolution was found by a joint re-review of the paper.
Analysis for Systematic Review
Study characteristics were extracted from the selected papers, including patient characteristics, time since stroke, medical treatment, EEG protocol, EEG variable, outcomes, and findings. In addition, methodological quality of the papers was determined according to the modified Downs and Black Checklist.27-30 The modified version of the checklist27-30 was used due to the limited number of experimental intervention studies included in this review. Two independent raters determined quality of the papers included in the meta-analysis and one of the raters determined the quality of all remaining papers included in the systematic review.
Findings regarding the relationship between EEG and subsequent outcomes were extracted from each paper. The findings were classified as positive or negative based on the following criteria. (1) Findings with P<.05 were counted as positive and findings with P>.05 were considered as negative for regressions, correlations, odds ratios, t-tests, and ANOVAs. (2) In cases of multiple regression and/or ANOVA with other predictors (e.g., initial clinical score), the finding was considered as positive if EEG contributed to the statistical model. (3) If a P-value was not provided, correlation coefficients or predictive values ≥.6 were considered as positive and findings with <.6 were considered as negative. 31 Papers were then classified as “positive” if they presented only positive findings for EEG prognosis, “negative” if they presented only negative findings, and “mixed” if they presented both positive and negative findings.
The results of papers were qualitatively examined against study characteristics including sample size, time post-stroke at EEG and at outcome, EEG variable, number of EEG electrodes, outcome domains, and quality score to investigate the association between study characteristics and prognostic results.
Meta-Analysis Method
For each paper, we extracted a correlation coefficient between baseline EEG and a subsequent outcome measure. When an odds ratio was provided instead of a correlation, a transformation to the scale of a correlation coefficient Yule’s Q 32 was applied. Two papers33,34 that examined the same sample of subjects were treated as a single paper in the analysis. Nine papers33-41 reported EEG and/or outcome measure scores at 2 or more time points. Therefore, we included data at each timepoint. For 13 papers that did not provide the data needed to calculate relevant statistics, the authors were contacted via email. One author responded and provided additional data from which correlation was calculated and included in the meta-analysis.
The correlation coefficient of each paper was then transformed using Fisher transformation for normal distribution. 42 To estimate an average association between EEG and clinical outcome, a linear meta-regression was performed for each outcome, adjusting for sample size and study quality. Weighted sample size (=√[n/total n of included papers]) and weighted quality scores (=√[score/max possible score]) were used in the analysis. The overall adjusted effect was then calculated by multiplying the estimate of each variable included in the model by its average and adding these values to the regression intercept. In other words, the overall effect was calculated by solving the fitted regression equation for the population average. To account for multiple EEG and/or outcome measure times within a single paper, study ID was included in the analysis as a random effect for all models. Time of EEG, outcome time, time between EEG and outcome, and time post-stroke were adjusted for but did not significantly contribute to any of the regression models and were removed.
Results
Systematic Review
Search results
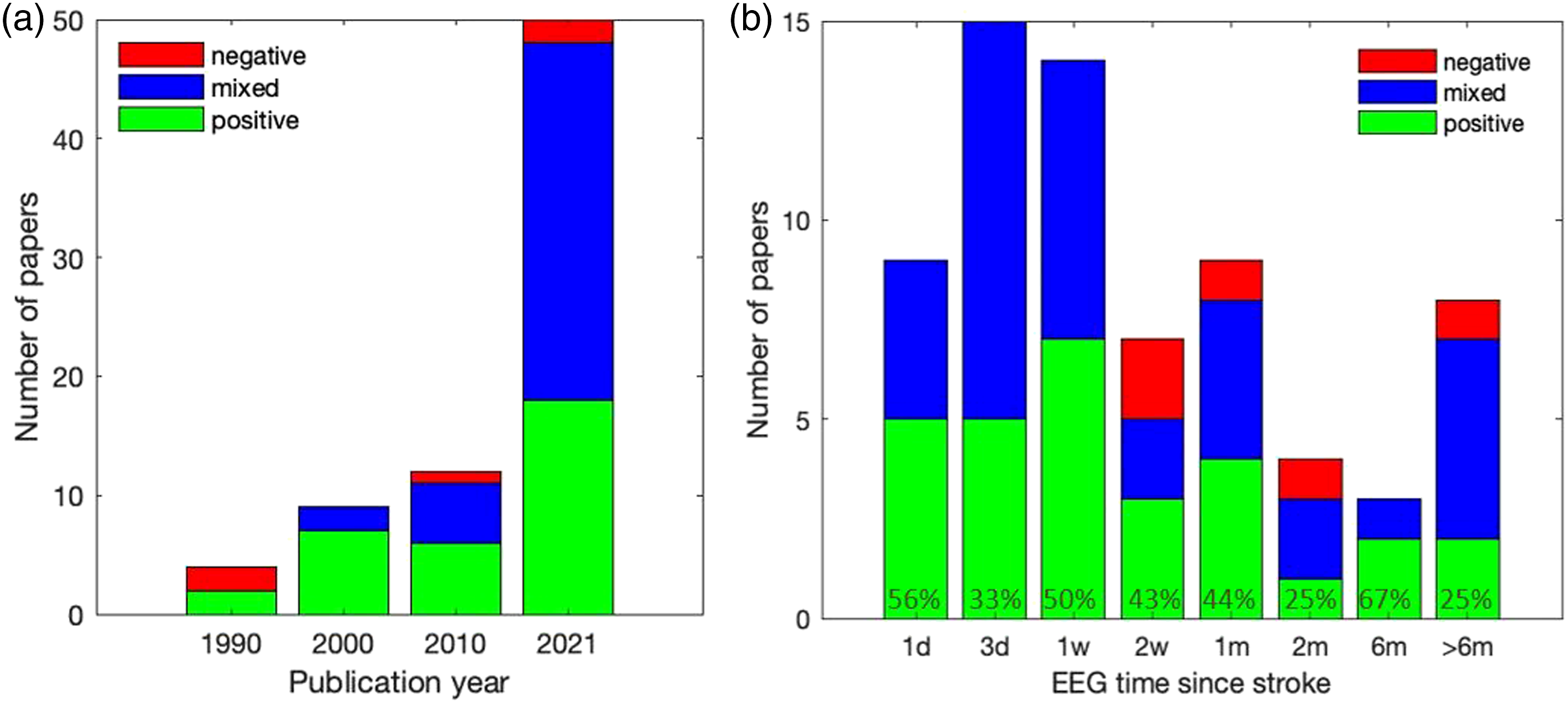
Results of the literature search and screening are summarized using the PRISMA 2009 flow diagram in Figure 1. A total of 75 papers met inclusion criteria and were synthesized for the systematic review. References for the included papers can be found in Supplement Material A. These 75 papers included a total of 4136 participants’ data, with the average age of participants in each paper ranging from 45 to 76 years. The majority of the papers were published in the last decade (Figure 2A). Of 75, 37 papers reported mixed results (i.e., both positive and negative), 33 only positive, and 5 only negative (Figure 2A). The detailed study information including characteristics of patients, EEG, outcome measures, and quality scores of each paper can be found in Supplement Materials B and A. PRISMA 2009 flow diagram. The number of papers published examining prognostic utility of EEG for post-stroke outcome over the years (A) and mean EEG times (B). The histograms shows the number of papers for each time period that reported (i) only positive, (ii) only negative, and (iii) mixed (i.e., both positive and negative) findings for EEG-based prognosis of post-stroke outcome. The upper limit of the bin is noted on the horizontal axis (e.g., bins=1981–1990, … , 2011–Aug 2021 in A, 0–1 day, >1–3 days, … in B). The last bar in B includes papers with EEG time ranging from 1 to 9 years post-stroke. Two papers with mixed findings and 4 papers with positive findings did not report the exact EEG time, thus are not included in B.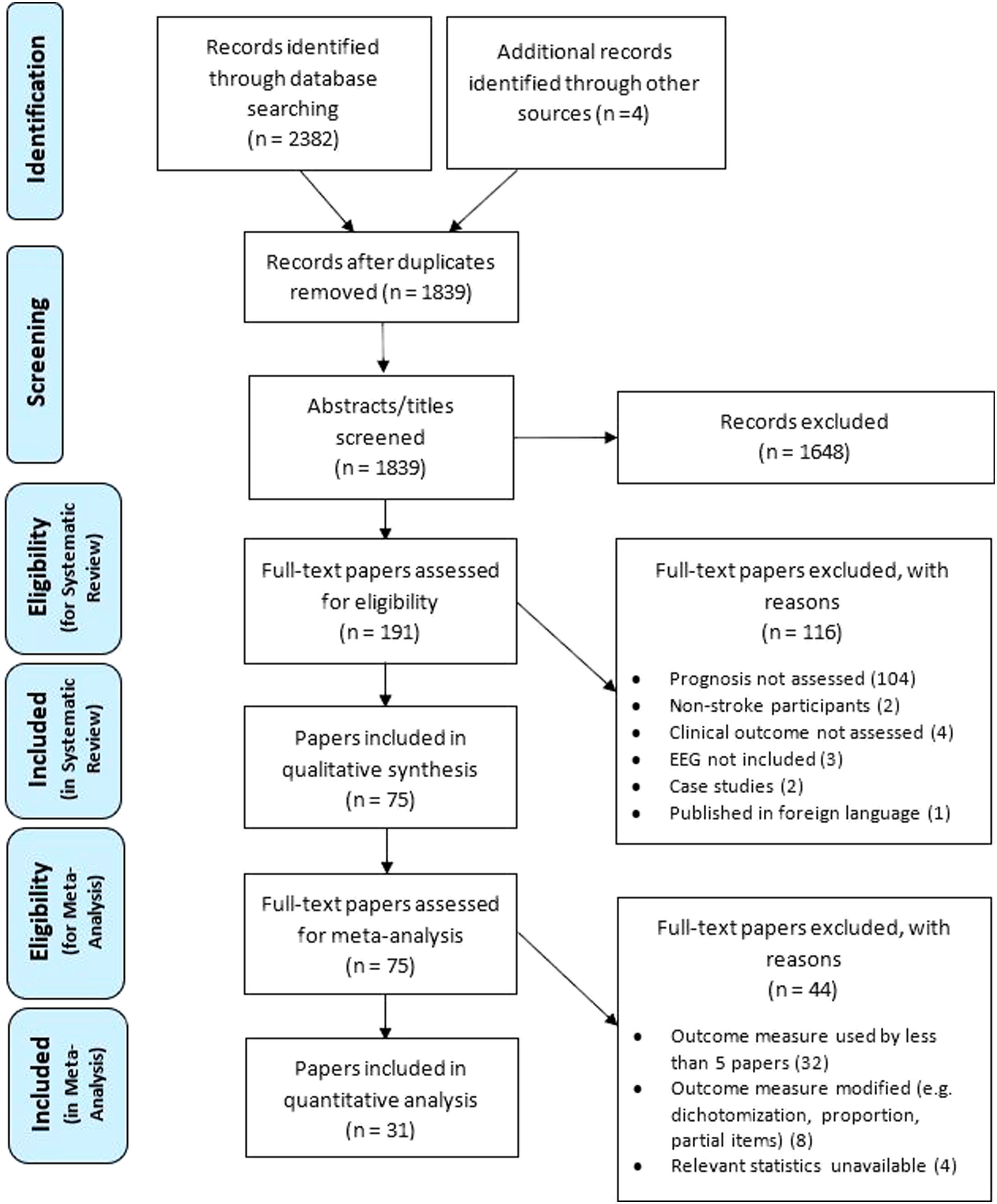
Time Since Stroke
Over 75% of the papers assessed EEG for prognosis within 1-month post-stroke (Figure 2B). Across all times post-stroke, 70 of the 75 papers found positive or mixed results for the predictive ability of EEG. Interestingly, positive findings were observed not only early post-stroke, but also at later times post-stroke (e.g., 6 months) (Figure 2B). This observation remained in chronic papers that assessed patients ranging from 1 to 8 years post-stroke. One negative prognostic result was found in 1 chronic paper at 9 years post-stroke in a small sample of 4 patients. 43
Type of Stroke
Of the 75 papers, 47 included only ischemic stroke and 28 included both ischemic and hemorrhagic stroke. However, of those that included both types of stroke, no papers compared prognosis between ischemic and hemorrhagic stroke. Thus, direct comparisons between stroke type could not be made. However, 2 papers compared ischemic stroke subtypes.44,45 Specifically, 1 study found that for posterior circulation syndrome, EEG within 3 days post-stroke was associated with 1-week stroke severity measured by the National Institutes of Health Stroke Scale (NIHSS) but not independence measured by the Modified Rankin Scale (MRS), while the opposite was seen for lacuna syndrome. 44 The other study found that EEG within a week post-stroke was predictive of 1-year MRS for both cortical and lacunar syndrome. 45
Treatment
Of 63 papers with mean EEG time ≤2 months post-stroke, 61 papers followed standard care, which encompassed inpatient hospitalization and/or inpatient rehabilitation therapy. The other 2 papers used a standardized treatment in which all patients received the same dose of a particular treatment (auditory discrimination training
46
and mechanical endovascular therapy
47
) (Figure 3). The other 12 papers with mean EEG time >2 months post-stroke used a standardized upper extremity treatment including standardized manual motor rehabilitation,48-52 visuomotor tracking training,
53
virtual reality training,
43
brain–computer interface training,39,54 semantic/phonological-based therapy,
55
and robot assisted therapy.56,57 Distribution of papers per study characteristics, including time since stroke, EEG protocol, EEG variable type, outcome domain, and outcome measure shows a lack of uniformity. * Power includes variables reflecting the amount of activity in given frequency bands.
109
†Event related potentials are voltages generated in the brain in response to specific events or stimuli.
110
#Epileptiform activity is neural activity specific to epilepsy that occurs during a seizure.
111
‡Connectivity represents neuronal oscillations which underly the coordinated flow of information within networks of brain regions.
112
¶Results derived from an EEG analysis that utilized dipole modeling.
113
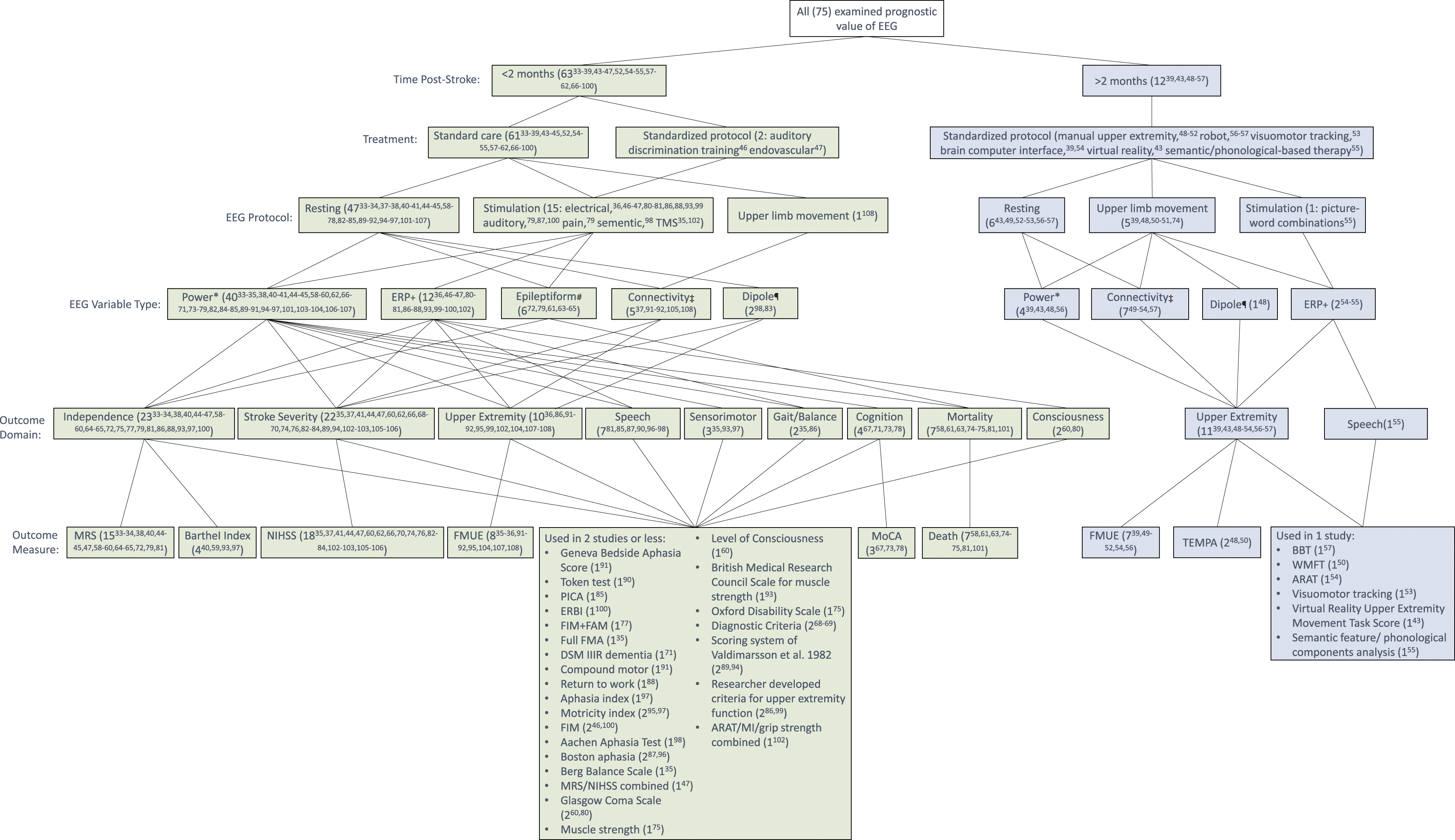
EEG Protocol
EEG was obtained for both clinical58-65 (n = 8 papers) and research33-41,43-57,66-108 (n = 67 papers) purposes. Of 63 papers with mean EEG time ≤2 months post-stroke, 47 papers obtained EEG during rest, 1 paper obtained EEG during upper limb movement, and 15 papers obtained EEG response to stimuli, the majority being electrical nerve stimulation in 9 papers (Figure 3). The other 12 papers with mean EEG time >2 months post-stroke obtained EEG during rest (n = 6 papers), upper limb movement (n = 5 papers), and response to stimuli (n = 1 paper).
EEG Variables and Outcome
EEG variables used varied considerably across papers. Thus, EEG variables were grouped into power, 109 event related potential, 110 epileptiform, 111 connectivity, 112 and dipole-based 113 EEG variable types (Figure 3). Outcome measures also varied considerably across papers, and were, therefore, grouped into outcome domains of independence, stroke severity, upper extremity, speech, (whole body) sensorimotor, balance/gait, cognition, mortality, and level of consciousness (Figure 3).
The positive, mixed, and negative findings were spread across EEG variable types and outcome domains (Figure 4). More than half of papers assessed power (e.g., brain wave oscillation symmetry, delta to alpha power ratio, and peak frequency). As such, power had the most positive and most negative findings. The majority of papers (n = 71 papers) examined a single EEG variable type. Thus, direct comparison of prognostic potential across multiple EEG variable types is limited. Distribution of outcome domains and EEG variable types examined. The total sample size for the papers that reported (i) only positive, (ii) only negative, and (iii) mixed findings for EEG-based prognosis of post-stroke outcomes are presented in stacked bars. Papers that examined multiple outcome domains and/or EEG variable types are presented for each result. In addition to the 5 papers that showed negative findings in the previous figure,43,61,89,94,98 3 papers reported negative findings in only some of the multiple outcome domains35,97 or some of the multiple EEG variable types examined.
91
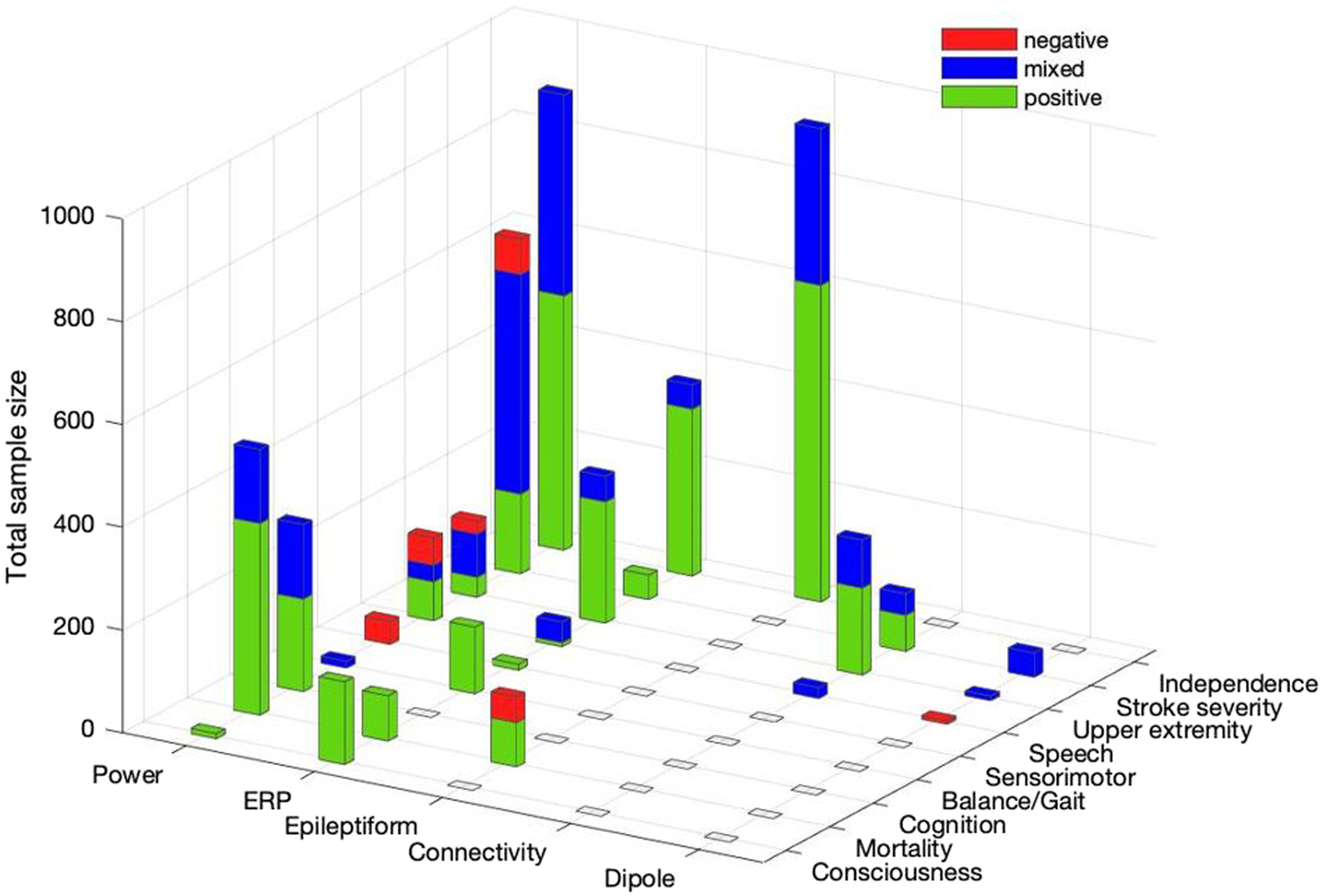
The most assessed outcome domain was independence (n = 23 papers with the total sample size of 2017, e.g., MRS), followed by stroke severity (n = 22 papers with the total sample size of 858, e.g., NIHSS) (Figure 4). Independence and stroke severity also had the largest number of papers with positive findings (n = 15 and 7 papers, respectively).
Quality Score, Sample Size, EEG to Outcome Time, Number of EEG Electrodes
The quality score ranged from 7 to 15, with a mean+SD = 11 ± 2 points, out of 16 points. To categorize the papers as “good,” “fair,” or “poor” quality, each paper’s quality score was divided by the maximum possible score of 16 to calculate a percentage. Thirty eight papers were found be of “good” quality (≥71%), 30 “fair” (54–70%), and 7 “poor” (≤53%).27,114 The poor quality was due to absence of variance estimates and/or actual probability values (e.g., reporting <.05 rather than exact P-values) and absence of description or adjustment for confounding variables, such as age and initial clinical score of patients. Poor quality was associated with earlier publication time, as 4 of the 7 poor quality papers were published between 1982 and 1994. Detailed quality score information including the number of points received on each item of the checklist and the total score for each paper can be found in Supplement Material C.
Sample size ranged from 4 43 to 459 65 participants (median = 34 participants). EEG to outcome time ranged from 4 days 62 to 3 years 89 (median=2 months). The number of electrodes used ranged from 140,73 to 256,49,53,57 with 19 being the most used.33,34,41,46,60,62,63,72,77,78,82-84,90,106,107
The prognosis results of each paper are plotted against quality score, sample size, EEG to outcome time, and number of EEG electrodes in Figure 5. Papers with negative findings appear to have a combination of a (i) long time period between EEG and outcome measure (e.g., 2–3 years), (ii) low quality score, (iii) low sample size, and (iv) low number of EEG electrodes (Figure 5). Distribution of the quality score, sample size, EEG to outcome time, and number of EEG electrodes used (denoted by the marker diameter) across papers. Papers that reported (i) only positive, (ii) only negative, and (iii) mixed findings for EEG-based prognosis of post-stroke outcome are presented with the solid, segmented, and dotted lines, respectively. Papers that examined multiple outcome time points are presented for all time points (e.g., 1 study
36
reported negative findings for 2 months but positive findings for 6 and 12 month outcomes). Papers that did not report information on EEG or outcome times are not included in this figure.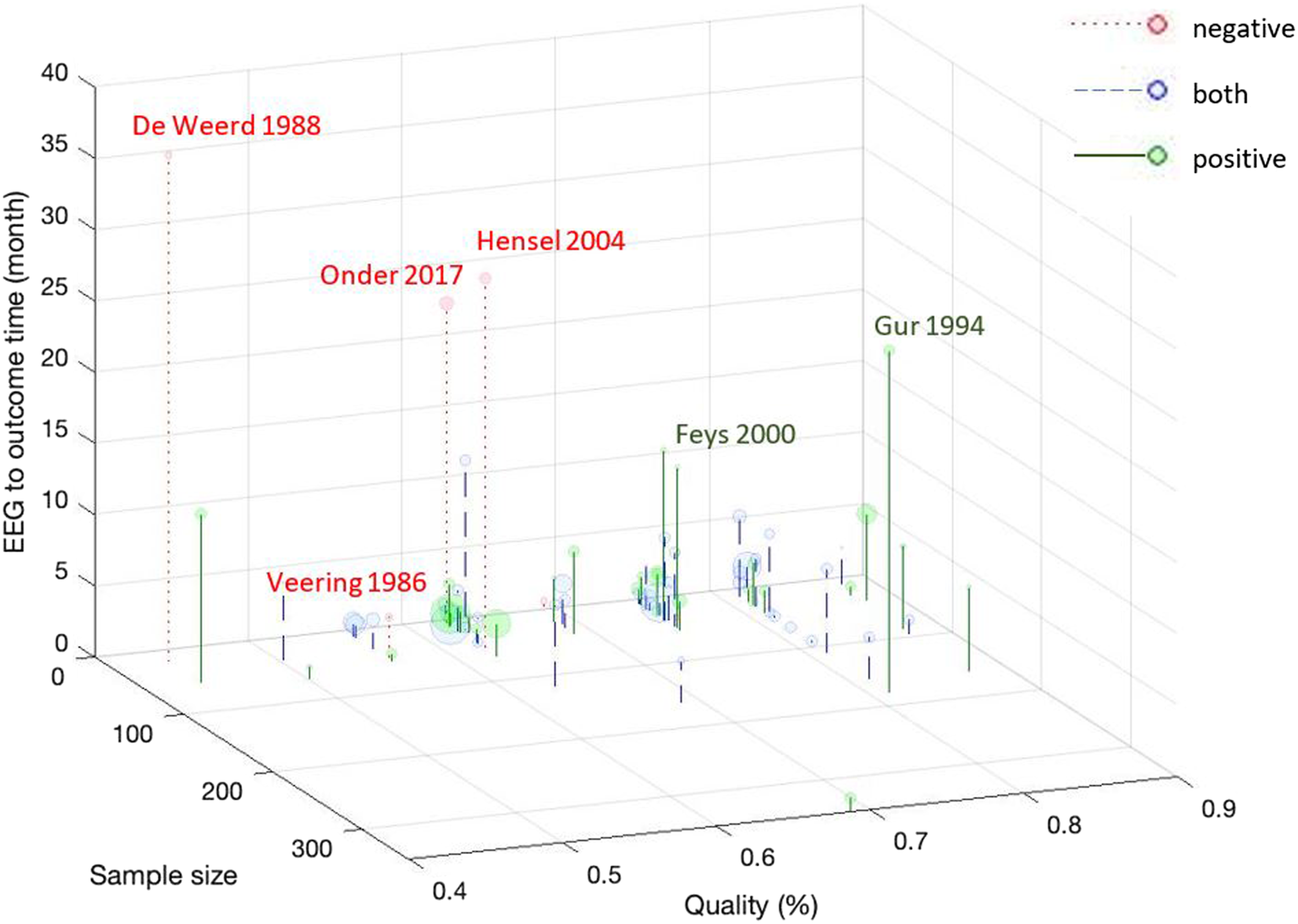
Prognosis Beyond Conventional Predictors
A total of 28 papers examined if EEG enhanced prognostic ability more than prognosis by the conventional predictor of baseline clinical score. Twenty-one papers found positive results. Specifically, the examined EEG variable(s) significantly explained variance in outcome after controlling for initial clinical score in 8 papers.48,59,64,73,83,90,92,107 In 7 papers, EEG further separated patients with good or poor prognosis after the consideration of the initial clinical score.38,45,54,65,99,102,104 In 6 papers, EEG correlated with outcome while initial clinical score did not.49,52,60,80,81,91 Mixed results were found in 6 papers, where EEG enhanced prognostic ability of conventional predictors only for some EEG variables,78,84 EEG time, 33 subgroup, 72 outcome domain, 74 and analysis method. 44 A negative result was found in 1 paper, in which only cerebral blood flow, not initial clinical score or EEG, predicted 3-year outcome. 89
Explorative Investigation
It was evident during the review that the majority of papers involved exploratory investigation. Specifically, 36 papers reported prognostic results for each of multiple EEG variables (e.g., simple correlations), including not only different EEG variable types (e.g., power and connectivity), but also multiple frequency bands (e.g,, delta, theta, alpha, beta, and gamma), multiple brain regions (e.g., ipsilesional and contralesional), multiple parameters (e.g., amplitude, latency, relative vs absolute power, power ratio, and dipole x, y, and z coordinates), and different tasks during EEG (e.g., eyes open vs close, movement preparation vs execution). In addition, 16 papers used an approach to statistically select a subset of multiple EEG variables for best prognostic results (e.g., stepwise regressions). Further, many papers examined prognostic results for multiple outcome times (n = 10 papers) and multiple outcome domains (n = 13 papers).
Meta-Analysis
Search Results
Results of the meta-analysis screening are summarized using the PRISMA 2009 flow diagram in Figure 1. Of the 75 papers included in the systematic review, 31 papers met the inclusion criteria and were synthesized for the meta-analysis. Quality scores ranged from 8 to 14, with mean + SD = 11 ± 2 out of 16 points. Eighteen papers were found be of “good” quality (≥71%), 12 “fair” (54–70%) quality, and 1 “poor” (≤53%) quality.27,114 The outcome measures examined were: (1) MRS 115 which measures the degree of disability/dependence in daily activities, (2) NIHSS 116 which measures stroke severity, and (3) Fugl-Meyer Upper Extremity Assessment (FMUE) 117 which measures upper extremity motor impairment.
Correlation Between EEG and MRS
Eight papers utilized MRS as the outcome measure. These papers presented 15 EEG and MRS correlations (Figure 6A) in a total of 202 participants. All papers assessed the EEG variable type of power. Linear meta-regression of the correlation between baseline EEG and subsequent MRS demonstrated a strong
31
adjusted effect of .72 (95% CI: .64–.78). Forest plots showing correlation coefficients between EEG and outcome with 95% confidence interval for MRS (A), NIHSS (B), and FMUE (C).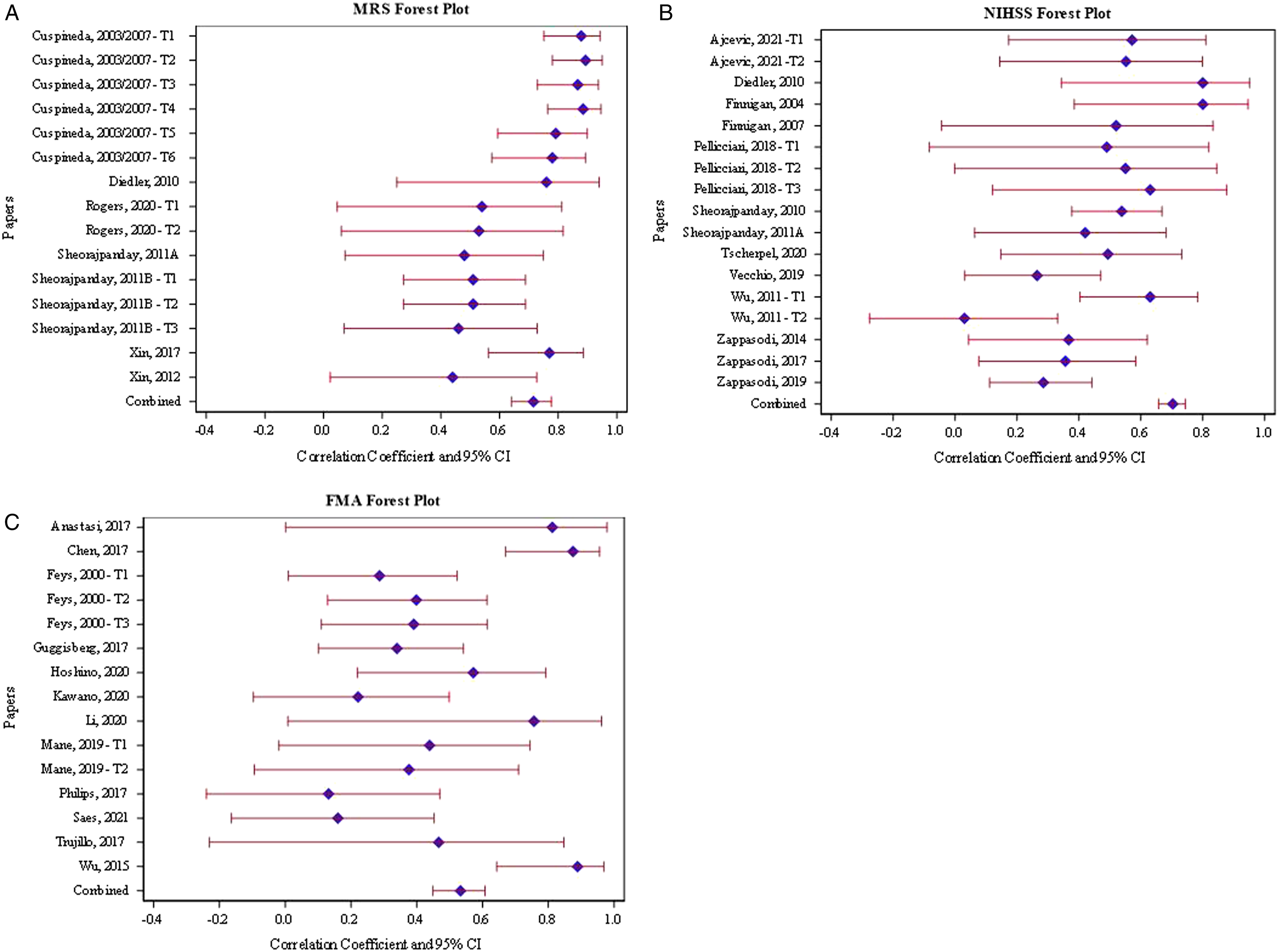
Correlation Between EEG and NIHSS
Thirteen papers utilized NIHSS as the outcome measure. These papers presented 17 EEG and NIHSS correlations (Figure 6B) in a total of 533 participants. They included multiple EEG variable types, including power (n = 12 correlations), connectivity (n = 4 correlations), and event related potential (n = 1 correlation). Linear meta-regression of the correlation between baseline EEG and subsequent NIHSS demonstrated a strong 31 adjusted effect of .70 (95% CI: .66–.74).
Correlation Between EEG and FMUE
Twelve papers utilized FMUE as the outcome measure. These papers presented 15 EEG and FMUE correlations (Figure 6C) in a total of 316 participants. They included multiple EEG variable types, including power (n = 6 correlations), connectivity (n = 6 correlations), and event related potential (n = 3 correlations). Linear meta-regression of the correlation between baseline EEG and subsequent FMUE demonstrated a moderate 31 adjusted effect of .53 (95% CI: .45–.61).
Discussion
Many papers have examined the prognostic utility of EEG in post-stroke outcome (75 papers for a total of 4136 participants). There has been a steep increase in the number of papers examining the prognostic utility of EEG in the last decade. This increase may be in part due to improvement in the computing resources to analyze EEG efficiently and in novel ways (e.g., connectivity and dipole/source analysis), along with the emergence of high-density EEG systems.
The majority of papers (70/75, 93%) showed all or some positive prognostic potential of EEG for post-stroke outcomes. Main observations are detailed as follows. First, prognostic potential was evident at both the acute and chronic phases post-stroke. While the majority of research has focused on prognosis within a few months post-stroke, there is evidence for chronic stroke patients with mean time post-stroke ranging from 11 months 50 to 8 years 56 that EEG is associated with improvement after a subsequent rehabilitation treatment. This evidence is aligned with general evidence of neuroplasticity in chronic stroke.118,119 This finding is encouraging for the clinical use of EEG for prognosis and may also have implications for participant selection in stroke recovery research studies which includes chronic stroke survivors exclusively in many cases. Stroke recovery research studies often result in findings that a treatment works best for a subset of the study sample. As a future direction, it is possible that EEG may be a useful tool to provide information to explain varying degrees of response to an intervention among individual patients.
Second, direct comparisons were not made between ischemic and hemorrhagic stroke types. While initial improvement is greater for hemorrhagic stroke compared to ischemic stroke, the time course of recovery does not differ between the 2 stroke types from 3 months post-stroke. 120 Despite the difference in etiology and initial severity level, response of the brain to the insult as captured by EEG may be relevant for recovery for both stroke types, although this needs to be empirically tested.
Third, among outcome domains, independence and stroke severity were most studied with the most positive findings (n = 15 and 7 papers, respectively). Meta-analysis also supports the strong relationships between EEG and MRS and between EEG and NIHSS. All papers that examined independence and stroke severity as an outcome were in acute/subacute stroke, with EEG performed on average 5 days post-stroke for both (ranging from a few hours to a month) and outcome measured on average 3- and 4-months post-stroke, respectively (ranging from a week to 1 and 3 years, respectively). Therefore, the translational potential of this evidence to standard acute/subacute stroke care is high, as the majority of the evidence is directly from that setting, involving EEG recording while patients rested.
While some ability of EEG to predict outcomes was seen for all outcomes studied, the results from other outcomes, such as upper extremity, speech, balance/gait, and cognition, were mixed. For all outcomes, besides sensorimotor, there was more evidence to support the predictive ability of EEG than evidence to refute it. In general, more research with methodological rigor is needed to determine the predictive ability of EEG for these outcomes.
Fourth, in over 95% of studies, EEG was able to increase prognostic ability compared to using the conventional predictor of initial clinical score alone. This is a critical point in the potential translation of EEG to routine clinical practice. The addition of EEG, while non-invasive, can be cumbersome and adds to cost of care. Evidence that prognostic ability is improved from what can be attained from standard of care is a critical factor in advocating for the addition of routine EEG in post-stroke patients. The practical extent of the consequences of better prognostic ability will need to be explored. It will be important for clinicians and hospital/clinical managers to ultimately realize a quality and/or cost benefit to the addition of prognostic EEG.
Lastly, prognostic potential was likely obscured due to methodological constraints. Variables such as EEG to outcome time, sample size, and number of EEG electrodes used may all contribute to differences in study results. The variety of EEG measures and lack of standardization also may mask results and hinders comparability of study outcomes. In addition, many studies had fair or poor quality evaluations due in large part to data not being fully reported; some quality issues were methodological in nature and may have influenced study results.
Evidence regarding prognostic utility of EEG is largely explorative. The majority of papers were exploratory in nature and did not have a priori hypothesized EEG variable(s) for prognosis. This explorative nature explains the large number of papers with mixed results due to the variety of EEG measures used. This may also explain the moderate relationship between EEG and FMUE found in the meta-analysis. The FMUE meta-regression model included a more even representation of each EEG measure (i.e., power, connectivity, and event related potential), whereas the MRS and NIHSS meta-regression models focused heavily on a single EEG measure (i.e., power). In general, there is emerging evidence that EEG has the potential to inform clinical decision-making and guide individualized treatment. However, consensus on the best EEG biomarker is needed for clinical translation to occur.
Limitations
Due to publication bias, the prognostic value found in this review may be elevated. However, such bias may have been mitigated since EEG prognosis is typically investigated as a secondary analysis in many papers. In addition, we were conservative in categorizing the results of each paper. Some papers concluded a positive prognostic result, while they were regarded as negative in this review based on the criteria described in the method section. Some papers hypothesized prognosis for one EEG variable and reported negative results for other EEG variable(s) as a negative control, which added to the number of papers with mixed results in this review. Some papers had an objective different from prognosis and happened to report correlations applicable to prognosis. Those results added to the negative results in this review, although these papers may not have chosen an EEG variable best for prognosis. The conservative approach used in this review was to identify a robust biomarker of outcome. The number of papers included in the meta-analysis was reduced because some papers applied outcome measures differently (e.g., dichotomization). This review did not include papers that were published in languages other than English.
Conclusion
Many studies examined the prognostic utility of EEG in post-stroke outcome in the recent decade. Prognostic evidence was seen at both the acute and chronic phases post-stroke, with mean time post-stroke ranging from immediately after the stroke 47 to 8 years. 56 The most studied prognostic potential of EEG is in predicting independence and stroke severity in the standard acute/subacute stroke care setting. This finding is also supported by the strong relationships between EEG and MRS and between EEG and NIHSS found in the meta-analysis. Furthermore, there is evidence that EEG improves prognostic ability beyond the conventional predictor of the initial clinical score. However, evidence regarding the prognostic utility of EEG is largely explorative, with many EEG measures used, primarily due to the lack of a definitive set of best EEG variables to use for prognosis. With continued advancement in computing capacity that enables source imaging, machine learning, and analysis efficiency, exploration of EEG biomarkers is expected to continue. In summary, EEG shows potential to improve post-stroke prognostic ability and inform clinical management, with a need to identify the best EEG measures for prognosis.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683221078294 – Supplemental Material for The Prognostic Utility of Electroencephalography in Stroke Recovery: A Systematic Review and Meta-Analysis
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683221078294 for The Prognostic Utility of Electroencephalography in Stroke Recovery: A Systematic Review and Meta-Analysis by Amanda A Vatinno, Annie Simpson, Viswanathan Ramakrishnan, Heather S. Bonilha, Leonardo Bonilha and Na Jin Seo in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-2-nnr-10.1177_15459683221078294 – Supplemental Material for The Prognostic Utility of Electroencephalography in Stroke Recovery: A Systematic Review and Meta-Analysis
Supplemental Material, sj-pdf-2-nnr-10.1177_15459683221078294 for The Prognostic Utility of Electroencephalography in Stroke Recovery: A Systematic Review and Meta-Analysis by Amanda A Vatinno, Annie Simpson, Viswanathan Ramakrishnan, Heather S. Bonilha, Leonardo Bonilha and Na Jin Seo in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: TThis study was possible through funding from NIH/NIGMS P20 GM109040 COBRE for Stroke Recovery, NIH/NCATS TL1 TR001451 and UL1 TR001450, and NIH/NICHD R01 HD094731.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
