Abstract
Background
Spinal cord injury (SCI) leads to various degrees of lifelong functional deficits. Most individuals with incomplete SCI experience a certain degree of functional recovery, especially within the first-year postinjury. However, this is difficult to predict, and surrogate biomarkers are urgently needed.
Objective
We aimed to (1) determine if routine blood chemistry parameters are related to neurological recovery after SCI, (2) evaluate if such parameters could predict functional recovery, and (3) establish cutoff values that could inform clinical decision-making.
Methods
We performed a post hoc analysis of routine blood chemistry parameters in patients with traumatic SCI (n = 676). Blood samples were collected between 24 and 72 hours as well as at 1, 2, 4, 8, and 52 weeks postinjury. Linear mixed models, regression analysis, and unbiased recursive partitioning (URP) of blood chemistry data were used to relate to and predict walking recovery 1 year postinjury.
Results
The temporal profile of platelet counts and serum levels of albumin, alkaline phosphatase, and creatinine differentiated patients who recovered walking from those who remained wheelchair bound. The 4 blood chemistry parameters from the sample collection 8 weeks postinjury predicted functional recovery observed 1 year after incomplete SCI. Finally, URP defined a cutoff for serum albumin at 3.7 g/dL, which in combination with baseline injury severity differentiates individuals who regain ambulation from those not able to walk. Specifically, about 80% of those with albumin >3.7 g/dL recovered walking.
Conclusions
Routine blood chemistry data from the postacute phase, together with baseline injury severity, predict functional outcome after incomplete SCI.
Keywords
Introduction
Traumatic spinal cord injury (SCI) is a devastating and life-changing event with no curative treatment currently available. 1 Individuals with incomplete SCI typically experience a certain degree of spontaneous recovery, especially within the first year after injury. Unfortunately, predicting who will experience spontaneous recovery, and to what extent, remains tumultuous at best because early biomarkers predictive for such recovery are sparse. In addition, standard clinical assessments are insufficient to fully depict individual and patient-relevant functional progress, which also limits comparability among clinical trials. 2 For example, sensory motor function is typically assessed using the American Spinal Injury Association (ASIA) International Standards for Neurological Classification of SCI (ISNCSCI) score, which is not able to detect a clinically meaningful change between 2 measurements. 3 Thus, the translational SCI landscape is in need of diagnostic measures and, most important, in need of surrogate markers predicting spontaneous recovery to assist in clinical decision-making in the course of treatment of individuals with SCI.4,5
Several studies have reported on blood and cerebrospinal fluid (CSF) proteins and their association with injury severity, and their prognostic value for neurological recovery after SCI.6,7 However, none of these proteins have been tested and validated as biomarkers in larger cohort studies. More important, such analytes require complex and specific methods for analysis, which are typically not available in a standard clinical setting, and in the case of CSF, incur substantial risk to the patient during sample collection. 8 Routine blood chemistry parameters might be a simple and feasible alternative to track spontaneous recovery after SCI, and indeed, previous literature reported about an association between serum albumin concentrations and the neurological outcome after SCI.9,10 However, literature on routine blood chemistry parameters and their potential to predict functional recovery in patients who sustained SCI is still tenuous. 7
Here, we aimed to (1) determine if routine blood chemistry parameters and their temporal evolution within the first year postinjury are related to functional recovery, (2) examine their predictive value in terms of neurological recovery in patients who sustained traumatic SCI, and (3) develop a cutoff value for such parameters that will assist clinical decision-making. For this, we evaluated the temporal pattern of readily available routine blood chemistry and established a model to predict functional outcome following SCI.
Methods
Study Design and Data Source
This was a post hoc analysis of data collected during the Sygen trial.11,12
Primary and Secondary Outcomes
The primary aim of this study was to determine whether routine blood chemistry parameters within the first year after injury are related to functional recovery after SCI. The secondary aim of the study was to evaluate if those parameters can predict functional recovery by means of walking ability (WA).
Predictor Variables and Covariates
The following blood chemistry data were included separately in linear mixed models calculations: white blood cell count, neutrophil bands, segmented neutrophils, lymphocytes, monocytes, eosinophils, basophils, platelet count (PLT), red blood cell count, mean corpuscular hemoglobin concentration, mean corpuscular hemoglobin, mean corpuscular volume, hematocrit, hemoglobin, albumin, alkaline phosphatase (ALP), amylase, creatinine, bilirubin, uric acid, urea nitrogen, calcium, creatine kinase, chloride, CO2 content, glucose, potassium, sodium, inorganic phosphorus, total cholesterol, serum glutamic-oxaloacetic transaminase, serum glutamic-pyruvic transaminase, and triglycerides. Blood samples were collected at baseline (24-72 hours postinjury) and after 1, 2, 4, 8, and 52 weeks. Blood samples were analyzed by each of the participating hospitals according to institutional guidelines. Importantly, Tong et al 9 examined baseline serum albumin concentrations across trial sites in the Sygen data set and found no significant differences between centers.
Neurological Assessment
The ASIA ISNCSCI scores 13 were used for assessing and classifying the neurological level and extent of SCI and were assessed at injury baseline and after 4, 8, 16, 26, and 52 weeks.
Functional Outcomes
WA was determined with the Modified Benzel Classification 14 after 4, 8, 16, 26, and 52 weeks. Patients with a Benzel score ≥5 points are considered self-ambulatory, whereby motor function allows walking with or without assistance. Patient-reported bladder and bowel (B&B) function were assessed at 8 and 52 weeks postinjury and was classified as normal, abnormal, or absent.
Statistical Analysis
All statistical analyses of blood chemistry outcomes, including their pattern of evolution and their association with neurological recovery, were completed in R (R Core Team, 2018, version 3.6.1; running under: macOS Catalina 10.15.4). Figures were compiled with Graph Pad Prism statistical software (Version 8, Graph Pad Software Inc, San Diego, CA), R, Biorender.com, and draw.io. Descriptive statistics for continuous variables and frequency counts for categorical variables were calculated. Pearson χ2 tests were applied to test the association between categorical variables. Independent t-tests were used to determine differences in continuous variables. We used Shapiro-Wilk tests to assess normality of distribution in blood chemistry outcomes. All measurements revealed nonnormal distributions.
After defining walking recovery as a clinically meaningful binary outcome, all parameters from routine blood chemistry were included in separate linear mixed-model analysis as part of a first, data-driven step of analysis. The blood chemistry data had missing data points. The percentage of missing blood chemistry parameters was 6.8% at baseline, 10% at week 1, 12.6% at week 2, 14.5% at week 4, 20.7% at week 8, and 41.8% at week 52. To account for this unbalanced design, we used linear mixed models, which are recommended for repeated-measures designs and allowed us to project a trajectory of blood values despite missing values. 15 Differences in blood chemistry outcomes were assessed across time and between patients who regained WA and patients who did not. Fixed effects of time and walking recovery along with the recovery-by-time interaction were included in random intercept models, which included random effects of subjects.
The significance of fixed effects was assessed by F-tests with a Kenward-Roger approximation of degrees of freedom. 16 Pairwise comparisons with respect to blood chemistry outcomes were performed between the groups with a Bonferroni P-value adjustment for multiple comparisons. A significance level of α <.05 was set and adjusted for multiplicity for all F-tests and Bonferroni corrected post hoc tests.
Blood chemistry parameters with an observed recovery-by-time interaction effect for walking recovery were plotted (Figure 2), and pairwise comparisons were performed between the group that had recovered WA compared with the group that had not. Blood chemistry parameters, which showed a significant recovery-by-time interaction effect in the linear mixed models and a significant difference in pairwise comparisons, at least in the blood sample at week 52, were considered to be associated with walking recovery.
To determine if the interaction effect was also true for another clinical end point, differences in these parameters were also assessed across time and between patients with normal B&B function compared with those with abnormal/absent function. Fixed effects of time and B&B function along with the function-by-time interaction were included in random intercept models, which included random effects of subjects.
Prediction of Functional Recovery
Our second step in the analysis was a hypothesis-driven approach based on the assumption that blood chemistry parameters that fulfilled the above-mentioned criteria (a significant recovery-by-time interaction effect and significant difference in pairwise comparisons) are associated with walking recovery. For this, we included those blood chemistry parameters in a LASSO (least absolute shrinkage and selection operator) regression analysis. LASSO regression is used to make prediction models in data sets with potentially intercorrelated independent variables, such as blood-derived biomarkers, in medical research. LASSO is a method to reduce intercorrelated coefficients to zero, which in turn minimizes overfitting and complexity in a model. The algorithm performs variable selection and regularization, thereby increasing generalization and interpretability by reducing the variance. 17 For cross-validation of the model fit, data were divided into a training and a testing set with a random 80-20 split. The LASSO regression was trained on the training set and subsequently used to test the predictive accuracy on the test set. The regression model was applied to the test data set of the original data and separately to the test data sets for the subgroups according to injury completeness. Blood chemistry outcomes from samples that were collected 2, 4, and 8 weeks postinjury served as independent variables.
To evaluate the performance of the proposed prediction model, we calculated precision, recall (sensitivity), specificity, positive predictive value (PPV), negative predictive value (NPV), accuracy (ACC), κ, F1 score, and the Matthews correlation coefficient (MCC). In addition, we assessed predictive ability of routine blood chemistry outcomes on walking recovery using area under the precision-recall curve (AUPRC). 18 AUPRC values range from 0% to 100%, where 0% is random classification and 100% is considered a perfect classification. The no-skill classifier, representing the threshold for random performance, is defined as 35% in our analysis. Recall is equivalent to sensitivity, and precision corresponds to PPV. Precision-recall curves have shown a higher informative value compared with receiver operating characteristics curves for evaluating binary classifiers in data sets with class imbalance because they consider the fraction of true positives among positive predictions. 19 In the Sygen data set, the number of patients who regained walking compared with those who remained wheelchair bound is strongly imbalanced (walking vs not walking = 109/59 in the group of patients with incomplete lesions and 11/365 in the group with complete lesions, respectively). AUPRC values were obtained by using the trained model to predict the test data.
Conditional Inference Trees
After an informed variable selection process based on expert opinion on the one hand to determine which parameters are considered clinically meaningful and for methodological reasons on the other hand, we finally adopted the data-driven machine learning approach of unbiased recursive partitioning by conditional inference (URP-CTREE) in our analysis. 20 The URP-CTREE algorithm is based on the iterative application of a variable selection and a population splitting procedure and allows for direct visual interpretation in a clinical setting.21,22 URP follows 2 steps, which are repeated after each split. In the first step, the algorithm assesses whether any of the predictors is associated with the outcome variable by multiple testing the Bonferroni-corrected P value. If at least 1 predictor is significantly associated with the selected outcome variable, a split in the data set is performed by selecting the predictor that showed the most significant association with the outcome (ie, the predictor with the smallest P value). In the second step, the algorithm calculates another possible split based on the new subset created from the previous split. If no statistically significant association between the included predictors and the outcome variable can be found, the algorithm stops producing splits.21,23 We chose URP because it is able to manage various independent and dependent variables and is not subject to parametric assumption.10,23 In this study, URP-CTREE was used to identify more homogeneous subgroups in the study cohort based on the clinical end point WA and several predictor variables (blood chemistry outcomes at postinjury weeks 4 and 8, baseline injury severity by ASIA Impairment Scale (AIS) grade, neurological level of injury, and B&B function at week 8). A simple illustration of the sequence of steps in the process of analysis is illustrated in Figure 1. The comprehensive workflow of statistical analysis is depicted in Supplementary Figure 1.
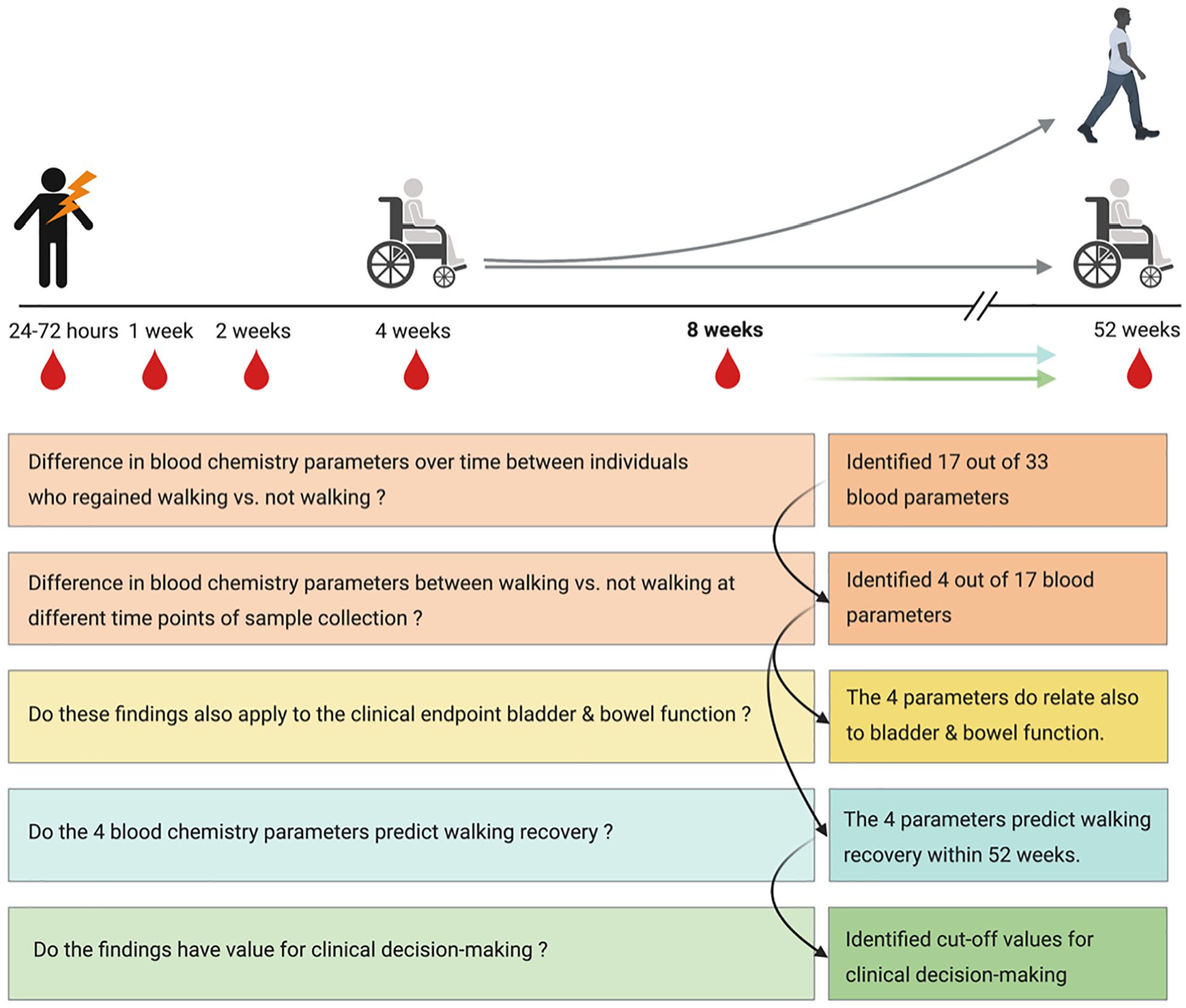
Graphical abstract: Sequence of steps in statistical analysis.
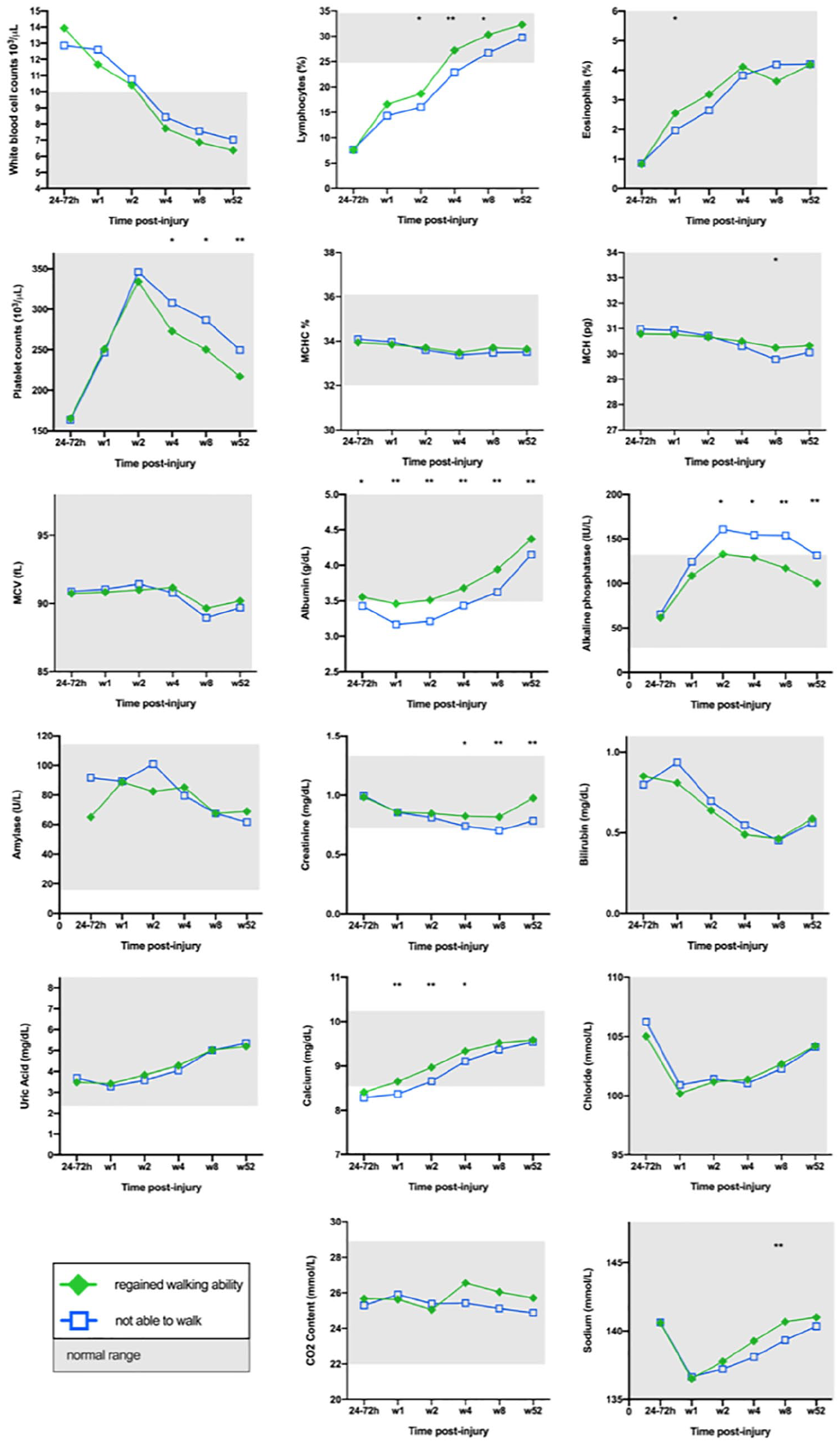
Blood chemistry parameters with an observed recovery-by-time interaction effect for walking recovery in the linear mixed models; mean values: independent t test with Bonferroni post-hoc correction: *p < 0.05, **p < 0.001.
Results
Patient Cohort, Clinical Scores, and Functional Outcome Measures
We retrospectively analyzed data collected during the Sygen trial, in which the effect of the drug Sygen (GM-1 ganglioside) was examined.11,12 A total of 797 patients with SCI rostral to the T10 vertebral level (T12 spinal cord/neurological level) were included in the original trial. 12 In all, 24 patients had been lost to follow-up, and 47 had died during the study period. For the present analysis, we excluded patients with lesions below the T9 neurological level of injury (26 patients), because (1) T9 is the border of spleen innervation24,25; (2) the spleen is the main organ of degradation of blood components such as erythrocytes and platelets 26 ; (3) our main interest in the present study was blood chemistry parameters and their relation to neurological recovery; and (4) we, thus, aimed to avoid confounding effects caused by spleen innervation/denervation. Data from 24 patients were excluded as a consequence of incomplete or missing ASIA ISNCSCI scores at baseline and/or at the 1-year follow-up examination. Complete data sets were available for 676 patients (see Table 1 for subject characteristics). Routine blood chemistry data were available at baseline (24-72 hours postinjury) and at 1, 2, 4, 8, and 52 weeks postinjury. The AIS, including its subscores (motor and sensory scores), 13 was used as a clinical score to divide the cohort by injury completeness. WA and B&B function were chosen to serve as functional outcome parameters.
Summary of Subject Characteristics.
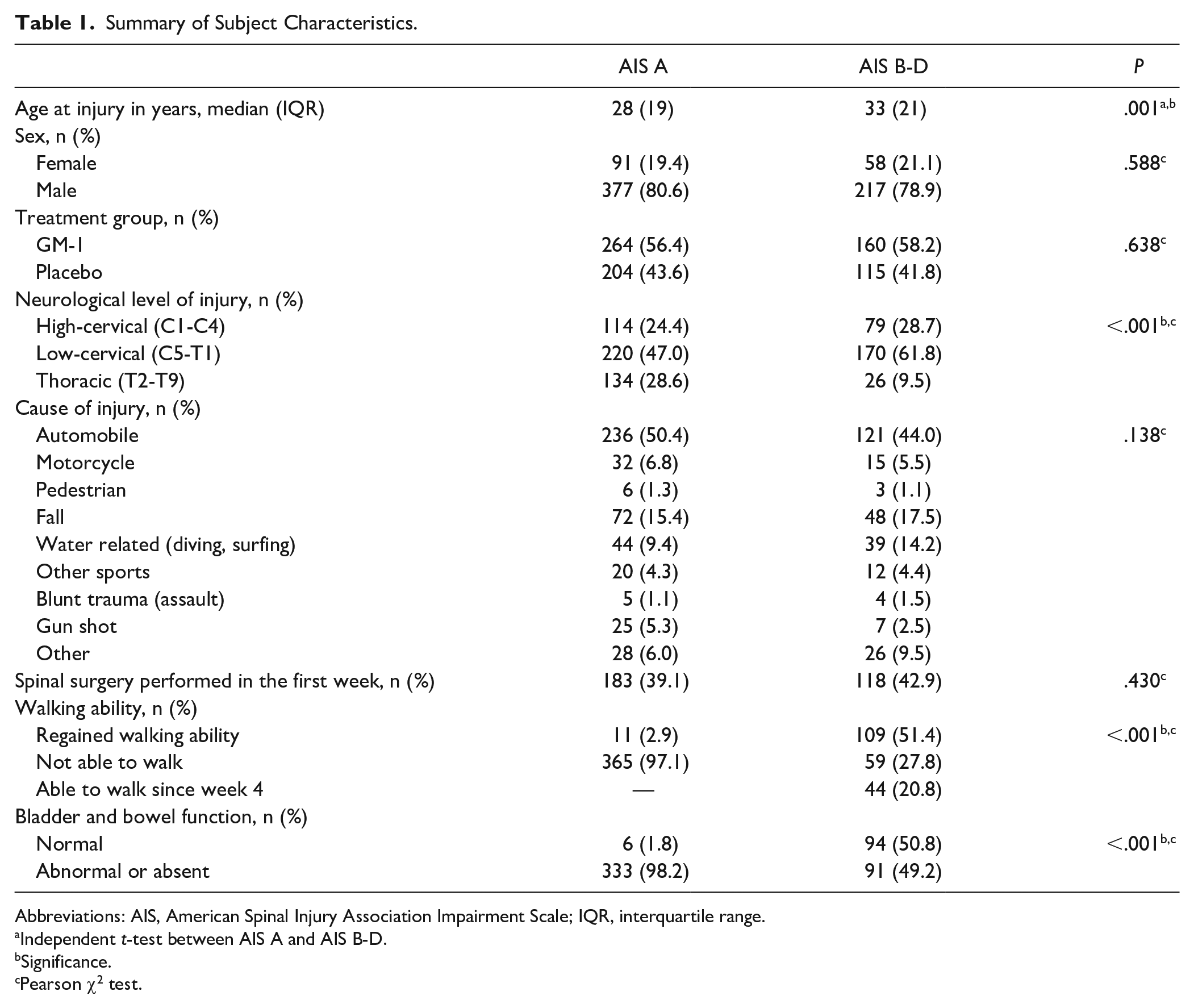
Abbreviations: AIS, American Spinal Injury Association Impairment Scale; IQR, interquartile range.
Independent t-test between AIS A and AIS B-D.
Significance.
Pearson χ2 test.
First, we reviewed the study cohort for the outcome parameter WA. Three groups of patients were identified: WA-W patients were able to walk already by 4 weeks postinjury (44 patients); WA-R patients were not able to walk 4 weeks postinjury but regained WA within 1 year (120 patients); and WA-N patients were not able to walk during the entire study period (424 patients). No significant differences comparing these groups were found between male and female patients, between patients who received GM-1 or placebo, and between patients who underwent no, early (within the first week postinjury), or late (beyond 1 week postinjury) spinal surgery (Supplementary Table 1). Therefore, those parameters did not influence the probability of regaining WA. A significantly higher percentage of patients were able to walk or regained WA in the group with incomplete (AIS B-D) lesions compared with patients with complete (AIS A) lesions (Table 1). Patients who were not able to walk 4 weeks postinjury (n = 544) defined the cohort in analyses, where WA served as end point. This group was divided according to walking recovery: individuals who regained WA (WA-R; 120 patients) and patients who were not able to walk (WA-N; 424 patients).
B&B function as secondary outcome parameter was assessed 8 and 52 weeks postinjury. For group comparisons, patients were allocated to 2 groups: B&B-N (normal function) and B&B-Ab (abnormal or absent function). In the study cohort, 100 (14.8%) patients had normal B&B function 1 year postinjury. No significant difference was found in terms of B&B function between male and female patients, between patients who received GM-1 or placebo, and between patients who underwent no, early, or late spinal surgery (Supplementary Table 7). A significantly higher percentage had normal B&B function in the group with incomplete lesions compared with patients with complete lesions (Table 1).
The Temporal Pattern of Blood Chemistry Parameters Relates to Functional Recovery After SCI
The primary aim of this study was to determine if parameters derived from routine blood chemistry are related to functional recovery after SCI. More specifically, we wanted to evaluate if the temporal evolution of routine blood chemistry parameters within the first year after injury is associated with walking recovery. For this purpose, we included routine blood chemistry outcome data in linear mixed model analyses accounting for different baseline values in blood chemistry parameters for each subject in the sample. Of the 33 available blood chemistry parameters, 17 showed a significant effect between walking recovery and their temporal pattern (Figure 2). Next, we investigated which blood chemistry parameters are actually different between the WA-R and the WA-N groups. The Bonferroni corrected pairwise comparisons at each time point of sample collection identified 4 out of the 17 blood chemistry parameters (PLT, albumin, ALP, and creatinine) that were significantly different between WA-R and WA-N (Figures 3A-3D; Table 2; Supplementary Tables 2-6) and are, therefore, considered to be associated with walking recovery. Besides the association with recovery of motor function, the 4 blood chemistry parameters were associated with B&B function, as discovered in linear mixed-model and pairwise comparison analyses (Figures 3E-3H; Supplementary Tables 8-12).
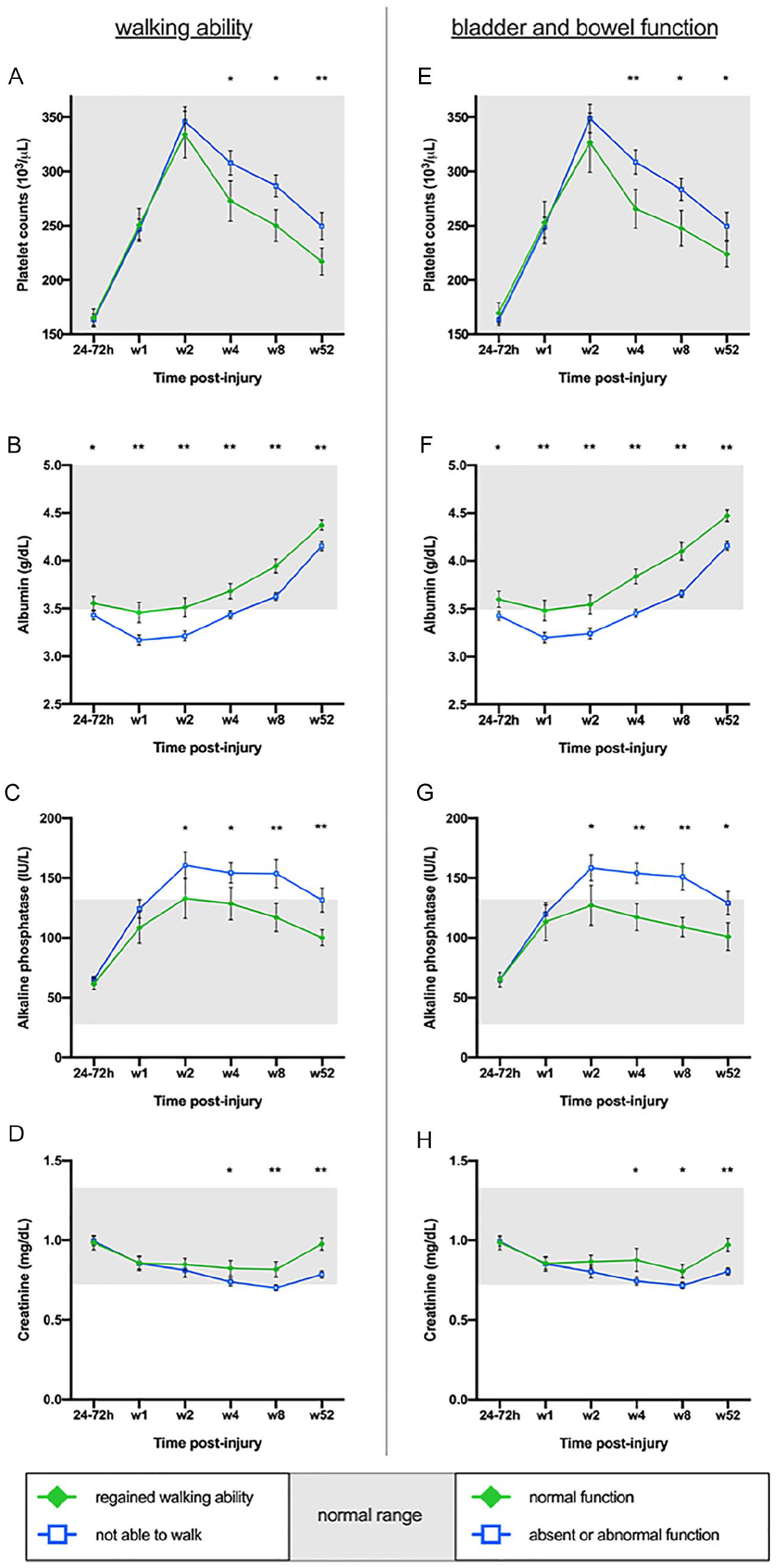
Association of blood chemistry parameters with functional recovery: Blood parameters (PLT, albumin, ALP, and creatinine) significantly correlate with functional recovery in the linear mixed model and in pairwise comparisons. Mean values; error bars show 95% CIs; independent t test with Bonferroni post hoc correction: *P < .05; **P < .001; (A) to (D): walking versus not walking; (E) to (H): normal bladder and bowel function versus absent or abnormal function.
Pairwise Comparisons Between Groups: Regained Walking Versus Not Walking.
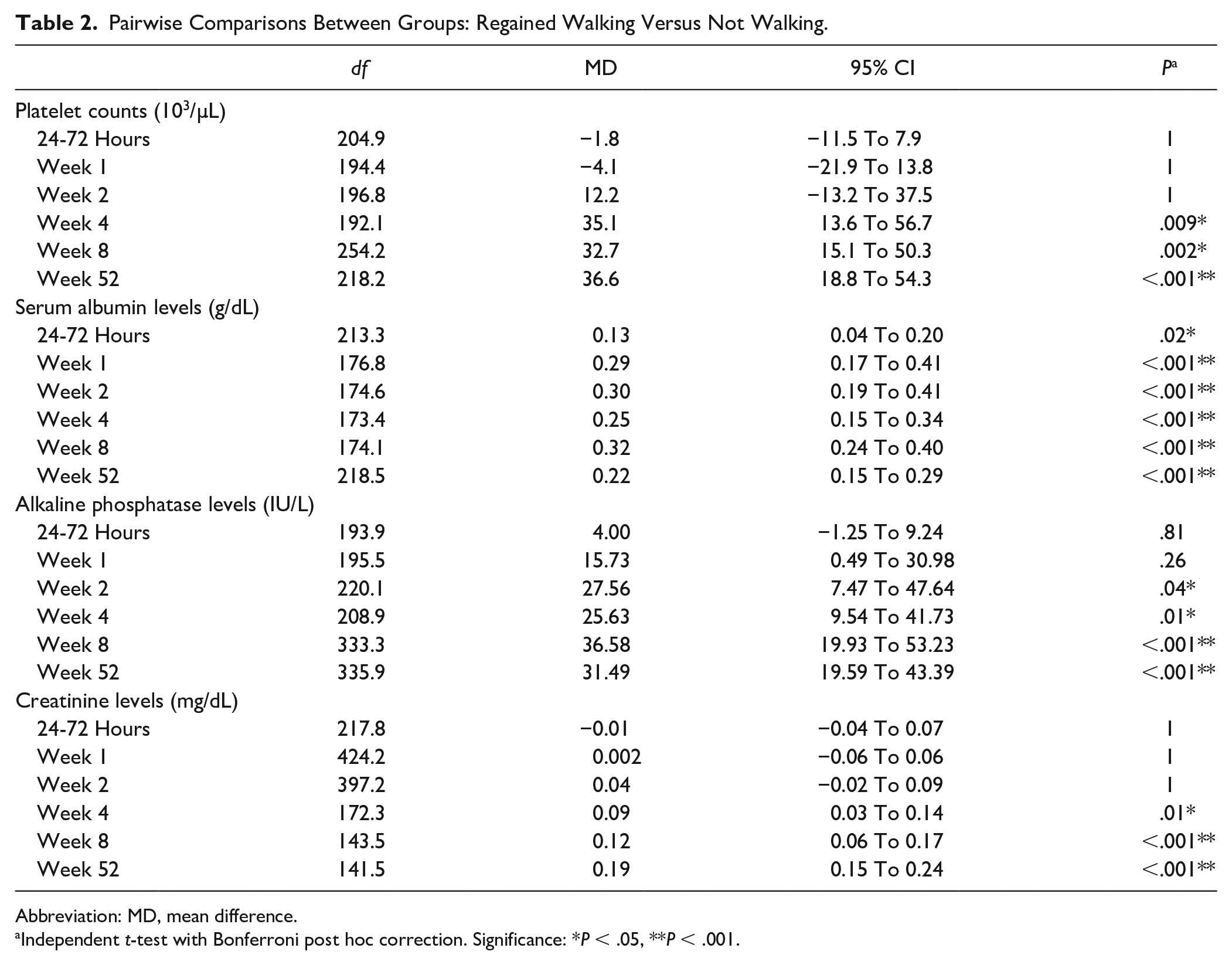
Abbreviation: MD, mean difference.
Independent t-test with Bonferroni post hoc correction. Significance: *P < .05, **P < .001.
Blood Chemistry Parameters Predict Functional Recovery
The secondary aim of the study was to develop a prediction model for walking recovery based on blood chemistry outcomes. Therefore, we investigated the 4 blood chemistry parameters identified (PLT, albumin, ALP, and creatinine) for their potential to predict walking recovery. Because these parameters did not differ between WA-N and WA-R during the first 2 weeks postinjury, we used data from the blood samples collected at 4 and 8 weeks postinjury in a LASSO regression model. We ran the LASSO regression model separately for the blood samples from weeks 2, 4, and 8. Participants with missing values in the respective blood chemistry parameters (PLT, albumin, ALP, or creatinine) were excluded from the regression analysis. The performance of the prediction model was assessed based on the AUPRC. AUPRC values were calculated by using the trained model to predict the test data (Figure 4A). The proposed prediction model revealed a high performance in the group of incomplete lesions (AIS B-D; n = 140), including blood chemistry outcomes (PLT, albumin, ALP, and creatinine) from samples collected 8 weeks postinjury (precision = 0.76; recall = 0.87; specificity = 0.69; PPV = 0.77; NPV = 0.82; ACC = 0.79 [P = .006]; κ = 0.57; F1 = 0.81; MCC = 0.57). The model produced an AUPRC of 0.74 (95% CI = 0.70-0.78; Figure 4A), indicative of good to excellent sensitivity and precision. Blood values were not predictive of walking recovery in patients with complete lesions (AIS A).
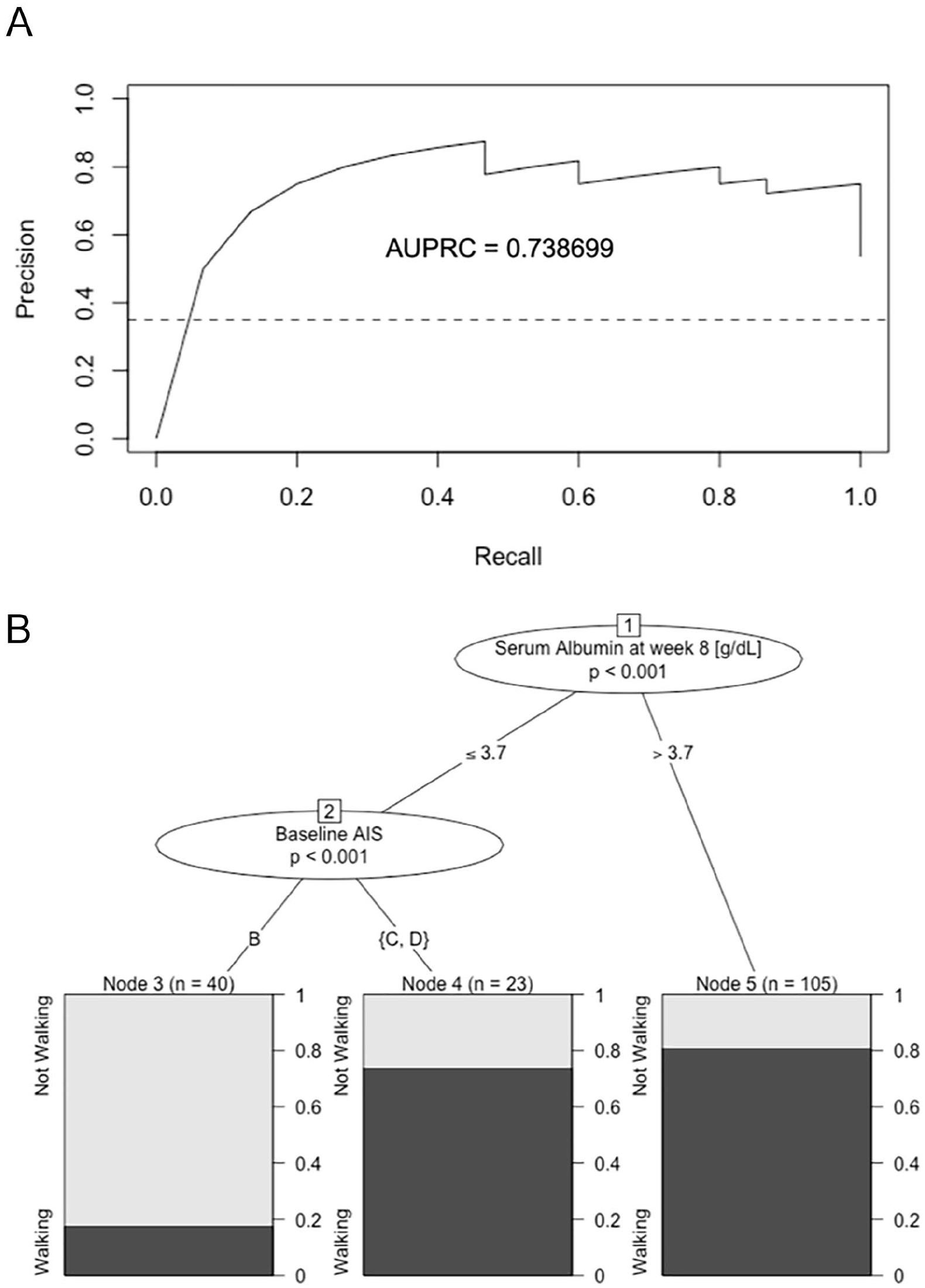
A. Predictive value of blood chemistry parameters on walking recovery: PR curve with AUPRC of the cross-validated LASSO regression model in the group of individuals with incomplete lesions (AIS B-D) using PLT, albumin, ALP, and creatinine concentrations at 8 weeks postinjury as independent variables and walking recovery as the dependent variable. The no-skill classifier (threshold for random performance) is shown as a dashed line. LASSO produced an AUPRC for predictability of walking recovery of 0.74 (95% CI = 0.70-0.78). B. Cutoff values in the prediction of functional outcome: URP-CTREE first node: serum albumin concentrations (in g/dL); second node: baseline AIS; 3 terminal nodes (3, 4, 5) for the end point distribution walking versus not walking (n, sample size; P, significance level).
URP Identifies Cutoffs in Blood Chemistry Parameters to Predict Functional Recovery
In the final step, we sought to establish a clinical application for the aforementioned findings. For this purpose, we used URP with conditional inference trees (URP-CTREE) with the aim of generating clinically relevant cutoff values. We included the blood chemistry parameters PLT, albumin, ALP, and creatinine from the samples at weeks 4 and 8 together with parameters that are already known to dictate prognosis such as injury severity, neurological level of injury, and B&B function. The URP-CTREE for the end point WA at week 52 based on a sample of 168 individuals with incomplete SCI (AIS B-D) produced 3 terminal nodes (3, 4, 5), for which the end point distribution (WA-R and WA-N) is shown in the diagrams in Figure 4B. The URP yielded 2 predictors for regaining the ability to walk within 1 year postinjury: (1) injury severity at baseline and (2) serum albumin concentration 8 weeks postinjury. The first node represents the cutoff value for serum albumin concentration of 3.7 g/dL (P < .001). In node 2, the subgroup with albumin levels ≤3.7 g/dL is further separated by baseline AIS (P < .001), resulting in 3 terminal nodes. From left to right, the terminal nodes represent subgroups from low percentage of individuals who recovered walking to high percentage of patients who regained walking within 1 year. In the albumin >3.7-g/dL cohort (n = 105), about 80% achieved walking recovery. In the cohort with albumin ≤3.7 g/dL and more severe injuries (AIS B; n = 40), less than 20% regained WA, whereas in the group of individuals with less severe lesions (AIS C-D; n = 23), approximately 75% achieved walking recovery (Figure 4B).
Discussion
Here, we demonstrate that the temporal profile of blood chemistry parameters, more specifically PLTs, serum levels of albumin, ALP, and creatinine are related to functional recovery after SCI. Predictive modeling demonstrated that the temporal profile of these blood chemistry parameters is able to predict walking recovery to some extent. According to our data, PLT and ALP levels are upregulated after SCI, followed by a downregulation 2 weeks postinjury, with a steeper decline in individuals who recovered walking. Platelets interact with different cells in the neurovascular system and are, therefore, thought to contribute substantially to the process of inflammation in central nervous system pathologies.27 -29 ALP correlates to muscle function after SCI 7 and has been shown to promote the neurotoxicity of extracellular tau, which is an indicator for neuronal damage. 30 Serum creatinine levels were significantly lower in individuals who did not recover walking, which is in accordance with previous literature reporting reduced creatinine levels in individuals with SCI. 31 However, the underlying mechanism of why these parameters are associated with walking recovery is a subject for further research.
Finally, URP-CTREE for the end point WA at week 52 defined a cutoff value for the level of serum albumin, which might facilitate monitoring of disease progression in patients after incomplete SCI. 22 In individuals with incomplete lesions and an albumin concentration >3.7 g/dL in the sample at week 8, about 80% regained WA independent of injury severity. In the group of individuals with albumin levels <3.7 g/dL and more severe injuries (AIS B), less than 20% regained walking, whereas in individuals with less severe lesions (AIS C-D) around 75% recovered walking (Figure 4B). In the Sygen study cohort, serum albumin was consistently higher in the group who regained WA compared with individuals who remained wheelchair bound. Previous literature stated that low serum albumin concentrations along with hypoproteinemia and malnutrition are predictors of adverse clinical outcomes and mortality in patients with SCI.9,32,33 Although this has been discussed extensively in the literature, the relation between albumin, malnutrition, and morbidity risk is not entirely clear. 34 Additionally, albumin supplementation has shown no effect on morbidity and outcome in critically ill patients with hypoalbuminemia. 35 Wong et al 33 postulated that undernutrition is associated with, but not proven to be a cause of, poorer clinical outcomes after SCI. 33 One theory suggests that albumin is an indicator for the general state of inflammation.9,36,37
Our findings add to the emerging body of research in this field by proposing serum albumin as a potential surrogate marker for monitoring disease progression, thereby enabling caregivers to continuously evaluate and improve the rehabilitation process. Our results are further supported by the work of Vo et al, 10 who reported that in individuals whose minimum albumin levels during the first year fell below 3.1 g/dL, less than 20% achieved marked recovery as defined by improvement by 2 or more AIS grades. The underlying reason why albumin concentration qualifies as a prognostic biomarker in various neurological conditions is to be further explored.
It has to be acknowledged that these findings are not applicable to the whole SCI population because we excluded patients with lesions below the T9 neurological level to eliminate a potential bias caused by spleen innervation and because thoracolumbar and lumbar injuries have a known different recovery pattern.24 -26,38
Also, our findings merely apply to incomplete SCI because we only included data sets of individuals with incomplete lesions in the prediction models because the number of patients who regained independent ambulation compared with those who remained wheelchair bound is strongly imbalanced (walking vs not walking = 11/365) in the group with complete lesions, which is in accordance with previous literature. 39
In this study cohort, half of the patients received GM-1 ganglioside. No significant differences were found in terms of WA as well as B&B function between patients who received GM-1 and the placebo group (Supplementary Tables 1 and 7). Because individuals given GM-1 and placebo were randomly distributed in the study groups, it is assumed that group allocation does not interfere with generalizability of our findings.
Certain progress has been made in SCI biomarkers, in particular in CSF-derived analytes and imaging methods.40,41 However, CSF sample collection is invasive and often accompanied by complications. 8 Spinal cord imaging techniques allow for high-resolution imaging of the lesion site and predict long-term neurological outcome after SCI.40,42 However, availability and feasibility of these techniques remain an issue in clinical routine. In contrast, routine blood chemistry represents a worldwide accessible, standardized, cost-effective, and validated method of measurement in clinical practice. The possibility of using blood parameters as biomarkers in SCI is an emerging field, and our findings can be translated to clinical practice immediately. A recent study by Brown et al 7 also concluded that some parameters from routine blood chemistry could serve as prognostic factors for motor and sensory scores after SCI.
We decided to use the ability to walk as the clinical end point in our calculations because the measurement of neurological improvement over time by AIS conversion is problematic as a result of ceiling and floor effects that involve AIS A, C, and D categories 43 and would, therefore, reduce the informative value of our analysis to the cohort of individuals with AIS B lesions.
Because the window of opportunity for neurological recovery persists for several months, and SCI leads to various degrees of lifelong deficits in most cases, we hypothesized that the relationship between the temporal evolution of blood chemistry parameters and neurological recovery will provide improved insight into the progression of recovery as opposed to 1 single time point of measurement. Our prediction model based on LASSO regression revealed the highest performance, including blood chemistry parameters from samples collected 8 weeks postinjury. However, it has to be acknowledged that in order to properly plan rehabilitation, a prognostication earlier than 8 weeks postinjury would be desirable. Generally, the informative value of biomarkers within the first weeks after injury might be reduced because of polypharmacy 44 causing potential alterations in blood chemistry. Thus, this study adds to research for the development of potential biomarkers in SCI by means of blood chemistry data. Also, our findings introduce the opportunity of a prognostic tool in clinical practice that can be evaluated straightforwardly by clinicians and future clinical trials because blood chemistry is well established in clinical routine.
Limitations
The main limitation of this study is that it is based on a retrospective analysis on data from a clinical trial that was initiated in 1992 when standards in clinical practice differed substantially from current treatment standards. Administration of high-dose methylprednisolone in the early postinjury period was standard of care in the 1990s after the NASCIS II trial.1,45 Therefore, blood chemistry data within the first postinjury weeks might have been altered by methylprednisolone administration.
Also, WA is defined as being able to walk with assistance or unassisted in our analyses. Future research should also assess walking distance, the ability to walk on different ground surfaces, and the use of walking aids.
Conclusions
The temporal evolution of PLTs, serum levels of albumin, ALP, and creatinine derived from routine blood chemistry is associated with walking recovery in individuals after traumatic SCI. Also, blood chemistry parameters investigated in this study provide predictive value for neurological recovery after traumatic incomplete SCI. For clinical application, baseline injury severity (by AIS grade) and a cutoff value of 3.7 g/dL in serum albumin concentrations at postinjury week 8 have been shown to facilitate the prognosis of whether an individual with incomplete SCI will regain walking ability.
Supplemental Material
sj-pdf-1-nnr-10.1177_1545968321992328 – Supplemental material for Routine Blood Chemistry Predicts Functional Recovery After Traumatic Spinal Cord Injury: A Post Hoc Analysis
Supplemental material, sj-pdf-1-nnr-10.1177_1545968321992328 for Routine Blood Chemistry Predicts Functional Recovery After Traumatic Spinal Cord Injury: A Post Hoc Analysis by Iris Leister, Lukas D. Linde, Anh Khoa Vo, Thomas Haider, Georg Mattiassich, Lukas Grassner, Wolfgang Schaden, Herbert Resch, Catherine R. Jutzeler, Fred H. Geisler, John L. K. Kramer and Ludwig Aigner in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank Fred Geisler for making the Sygen database available to our research group. The project was supported by a research grant from the Paracelsus Medical University Research Fund (PMU-FFF; Grant Number: S-19/03/015-LEI, Leister), Wings for Life Research Foundation (#2017_044, Jutzeler and Kramer), and the Swiss National Science Foundation (Ambizione Grant PZ00P3_186101, Jutzeler). The authors would like to express their sincere appreciation.
Authors’ Note
John L. K. Kramer and Ludwig Aigner have shared senior authorship.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by a research grant from the Paracelsus Medical University Research Fund (S-19/03/015-LEI, Leister), Wings for Life Research Foundation (#2017_044, Jutzeler and Kramer), and the Swiss National Science Foundation (Ambizione Grant PZ00P3_186101, Jutzeler).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
