Abstract
Introduction
Neurogenic bowel dysfunction is an aggravating, lifelong syndrome, which affects about 80% of patients with spinal cord injuries (SCIs). The estimated prevalence is 250 000 in the United States and up to 250 000 in Europe.1-5 Impaired spinal neural control of bowel and sphincter functions may cause serious clinical conditions, depending mainly on the level and severity of the spinal cord lesion. 6 The clinical presentation is typically dominated by stool retention, and, ultimately, severe constipation and fecal incontinence. 6
Moreover, neurogenic bowel dysfunction often leads to severe, potentially life-threatening complications, such as intestinal obstruction (ie, constipation up to the mechanical ileus), autonomic dysreflexia, urinary tract infections, sepsis, hemorrhoids, rectal bleeding, and prolapse.7,8 Additional factors related to SCI, such as loss of a rectal fullness sensation, difficulties in mobility, and impaired hand/arm function, may further complicate evacuation. 6 These aspects, together with the frequent dependence on a caregiver for bowel management, can severely limit working activities, social participation, and quality of life.6,8
Programs that aim to achieve the highest degree of independence in bowel management, by achieving continence and reliable bowel movements (ie, regular and time-efficient), represent a key aspect of rehabilitative treatments for patients with SCIs.9,10
Currently, several management strategies are available, and typically, several methods are applied either consecutively or in combination, including diet, oral medications, rectal stimulants, abdominal massage, and irrigation techniques. Eventually, different maneuvers might be applied, such as functional electrical stimulation of the skeletal muscles, and ultimately, surgical interventions, such as implanting a sacral anterior root stimulator, applying a Malone antegrade continence enema, and performing a permanent colostomy.6,11
However, despite all efforts in the rehabilitation phase and the different existing approaches, most patients with chronic SCIs report problems in bowel management.8,11 This issue might be a result of the largely empirical nature of current bowel management strategies because only a small number of studies is available, and those have limited methodological quality. 11
Patients with SCIs consider efficient bowel control, together with bladder function recovery, to be highly relevant medical needs. Thus, there is a need for high-quality research in this field. 12
Early predictions of bowel function outcomes would be instrumental for counseling patients and their families and for the prompt activation of structural interventions that might be necessary for the successful reintegration of patients into the community setting. 9 Moreover, it is important to identify patients who have a high probability of achieving efficient bowel control with standard care. 13 This identification would promote the optimization of future clinical trial designs for evaluating the efficacy of new bowel management interventions because it could facilitate the stratification of intervention groups for the likelihood of spontaneous recovery. 13
Although there are valid prediction models for locomotion, 14 upper-limb function, 15 and bladder function,16-18 no similar tool is available for bowel function. Thus, we aimed to derive and validate a model for predicting the achievement of independent bowel management, with reliable bowel movements and continence, at 1 year after traumatic SCI.
Methods
Study Design
The prediction model was derived and validated with data from patients included in the European Multicenter Study about Spinal Cord Injury (EMSCI; www.emsci.org) (ClinicalTrials.gov Identifier: NCT01571531). The EMSCI is a prospective longitudinal cohort study that started in July 2001. The study included patients with acute traumatic and ischemic SCIs, based on defined inclusion criteria, and the patients were managed according to common standard examinations (supplementary data, S1 Text).19-23 All patients were tested at fixed time intervals after the injury, as follows: between 0 and 15 days (very acute), between 16 and 40 days (acute I), between 70 and 98 days (acute II), between 150 and 186 days (acute III), and between 300 and 400 days (chronic). The examinations consisted of a standard set of clinical, neurological, neurophysiological, and functional assessments. The study conformed to the standards established by the Declaration of Helsinki and was approved by the local ethics committees of all participating centers. Before inclusion, patients were informed about the research protocol and signed written informed consent forms.
The present study conformed to the Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis Or Diagnosis (TRIPOD) statement (https://www.tripod-statement.org; S2 Text) and to the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) statement (https://www.strobe-statement.org; S3 Text).
Patient Populations
A previous study conducted by the EMSCI study group developed a prediction rule of ambulation after traumatic SCI. 14 According to that methodology, we performed a derivation and a temporal validation with 2 EMSCI cohorts.
To derive the prediction model, we extracted data from the EMSCI database for all patients with a traumatic SCI that occurred between July 2001 and December 2012. To validate the prediction model, we prospectively collected data for all patients included in the EMSCI who sustained a traumatic SCI between January 2013 and December 2014 (with 1-year follow-up data, ending at the latest in December 2015).
Predictive Variables
As potential predictors, we investigated the same variables analyzed previously to derive the prediction models of bladder function. 16 These variables included the following: patient age and gender, all variables derived from the International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI),24,25 and all variables from versions II and III of the Spinal Cord Independence Measure (SCIM).26,27 The ISNCSCI is a well-accepted neurological assessment strategy proposed by the American Spinal Injury Association (ASIA) to establish the level and severity of traumatic SCIs.24,25 This system rated the muscle strength of 5 key muscle groups in each limb according to a 6-level scale, where 0 indicated the absence of movement and 5 indicated complete movement against gravity and full resistance. Light-touch and pinprick sensation impairments were rated for each dermatome in the body on a 3-point scale, where 0 indicated no sensation, 1 indicated altered sensation, and 2 indicated full sensation. In addition, voluntary anal contraction and the sensation of deep anal pressure were examined. The ISNCSCI also recorded some cumulative scores, including the upper-extremity motor score (UEMS), lower extremity motor score (LEMS), and total light-touch and pinprick scores. Thus, the ISNCSCI provided assessments at the neurological level, motor level, and sensory level. The ASIA Impairment Scale (AIS) was used to grade 5 levels of severity, from A (complete lesion) to E (normal sensation and motor function) in all tested dermatomes and myotomes. ISNCSCI evaluations were performed by trained physicians, whose experience in the use of ISNCSCI was certified after a centralized EMSCI instruction course. 28 Motor and sensory scores and AIS grades were computed automatically by the validated ISNCSCI calculator developed by EMSCI (www.ais.emsci.org). 29
Functioning was assessed with the SCIM, a validated tool specifically developed to evaluate the capacity and level of independence in daily life activities of patients after a SCI.26,27 The SCIM tested patients in tasks relevant to spinal cord injuries in 3 activity domains: the self-care subscale (scores 0-20); respiration and sphincter management (scores 0-40), which included an item related to bowel management; and transfer (scores 0-40). The total SCIM score ranged between 0 and 100, with higher scores indicating more independence. After its introduction, 2 revised versions of the SCIM were developed: versions II and III, respectively.26,27 The 2 subsequent versions showed small differences from the first version, but they used the same subscales and total scores. The EMSCI applied SCIM version II in the first phase and later adopted version III. The version used in the initial evaluation was used in follow-up assessments. SCIM assessments were performed by health professionals (nurses, occupational therapists, physiotherapists) with specific instructions and experience in the use of this tool. All predictive variables were recorded within the first 40 days after the SCI. When available, variables were included (n = 1195) that were recorded at the acute I time point (16-40 days after injury). When the acute I assessment was missing, the very acute time point (within 15 days after injury) was considered (n = 55).
Outcome Measure
The primary outcome was independent bowel management with regular bowel movements and appropriate timing, with no or rare accidents (ie, fecal incontinence less than twice a month). This outcome was assessed with a bowel diary, which patients maintained for 1 year after the SCI (time point chronic in the EMSCI time schedule), and the outcome was measured with item 7 (sphincter management—bowel) of the SCIM.26,27
Patients were dichotomized, based on bowel function, at 1 year after SCI, as follows: (1) independent bowel management with regular bowel movements, appropriate timing, and no or rare accidents (less than twice a month; ie, the item 7 score was 10 points in SCIM version II and 8 or 10 points in SCIM version III), or (2) irregular timing, a very low frequency of bowel movements, or dependence on bowel management (ie, item 7 score <10 points, in SCIM version II, and <8 points, in SCIM version III).
Statistical Analysis
To derive the prediction model from the EMSCI data, we applied multivariable logistic regression analyses with the same statistical approach used previously to generate the prediction models of ambulation and bladder function after a traumatic SCI.14,16 Missing outcome data were anticipated, and a weighting factor approach was used to correct for missing data. Complete cases were weighted by the inverse probability of being a complete case.
30
The factors mainly associated with missing outcome data were the center, the year of inclusion, and age. Based on these parameters, we estimated the probability of missing data and defined a weighting factor (
All ISNCSCI scores that rated each side of the body were analyzed. However, for this study, instead of considering the 2 sides (right and left) separately, we grouped the 2 sides together, analyzing the best and the worst score of each side. In 80 patients, data were missing on the S4-S5 dermatome sensation, deep anal pressure, and voluntary anal contraction. Subsequently, the AIS grades for these patients were derived, based on the S1 score, by applying the method proposed by Zariffa et al. 31
A total of 182 covariates were considered for the model elaboration. These covariates are reported in the supplementary data (S4 Text). Based on the Akaike information criterion (AIC), and after applying a stepwise forward procedure, we identified potential predictors. 32 The AIC reflects the relative quality of a statistical model, given the independent variables used. It allows an evaluation of the extent of change in the quality of the model when parameters are added (or removed). Thus, the AIC is used to select a specific model. We created 182 logistic models by fitting only a single explanatory variable. Subsequently, we selected the model with the best AIC. Then, we added variables to create a multivariable model; each variable was retained, as long as it significantly improved the area under the receiver operating characteristics curve (aROC).
The receiver operating characteristics curve (ROC) is a method for representing the characteristics of a test. For all test values, the ROC
We described patient characteristics as the percentage or mean (SD), and we performed comparisons with parametric and nonparametric tests, as appropriate. Statistical analyses were performed with the R statistics package (R version 2.14.0, www.R-project.org/) and the Stata 14.2 statistics software package (StataCorp 2015; Stata Statistical Software: Release 14; College Station, TX: StataCorp LP). The statistics code is available on request.
Results
Patients
For the model derivation, 2366 patients with traumatic SCIs were enrolled between July 2001 and December 2012 from 18 EMSCI centers. The initial (very acute and acute I) ISNCSCI assessments were missing for 178 patients, and the late outcome, measured 1 year after injury, was missing for 938 patients. Consequently, the prediction analysis was performed on data for 1250 patients. Table 1 shows the clinical characteristics of patients included in the model derivation and those excluded from the analysis, because of the lack of a 1-year outcome. The 2 groups showed significant differences in age, the percentage of patients with paraplegia, and the percentage of patients with complete lesions (AIS A).
Characteristics of Patients at the Time of Inclusion (Within 40 Days From Spinal Cord Injury).
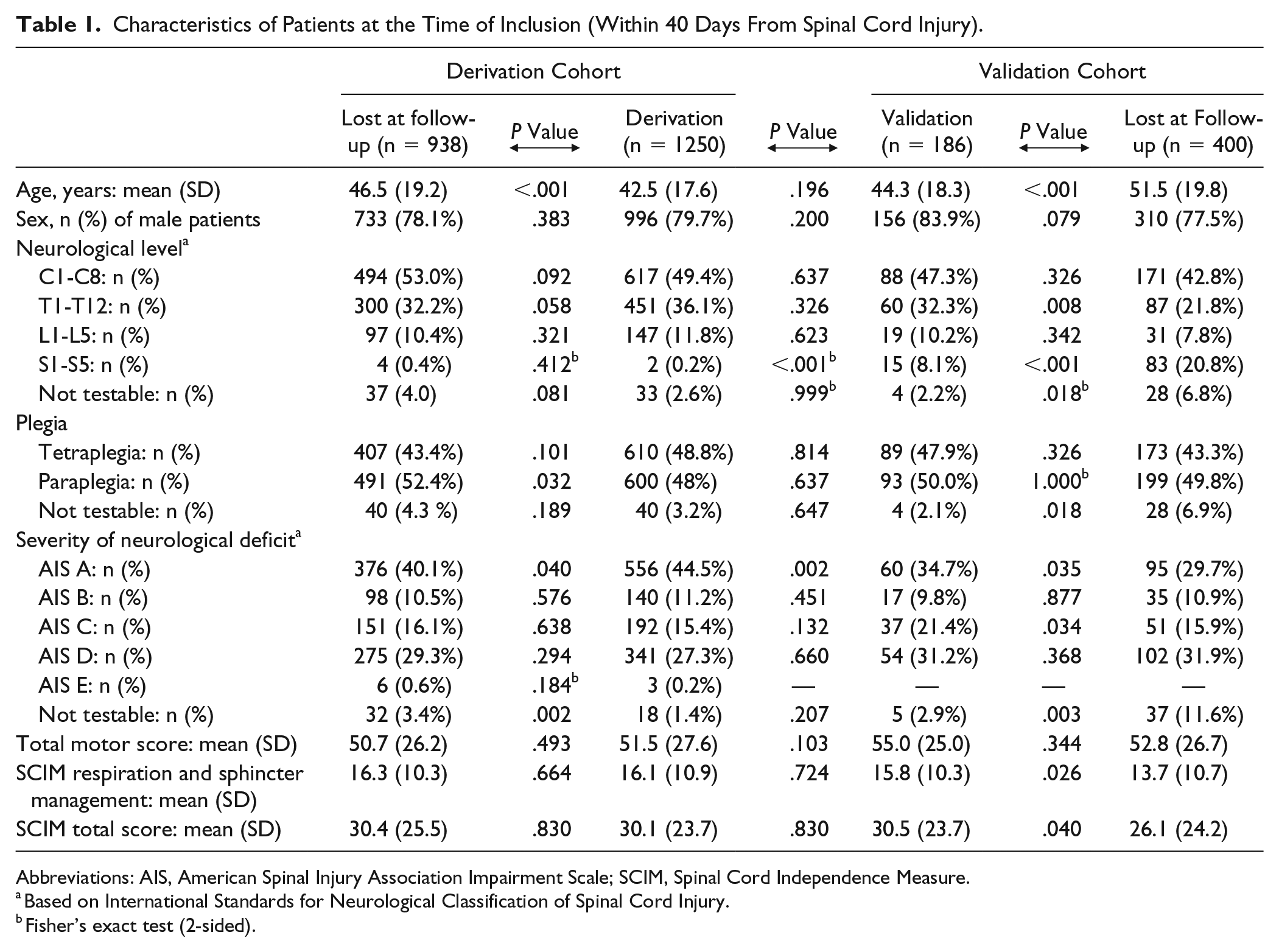
Abbreviations: AIS, American Spinal Injury Association Impairment Scale; SCIM, Spinal Cord Independence Measure.
Based on International Standards for Neurological Classification of Spinal Cord Injury.
Fisher’s exact test (2-sided).
In the derivation group, at the initial assessment (measured within 40 days from the injury), 167 (13.4%) patients exhibited independent, efficient bowel management. Of these, 153 (91.6%) patients showed unchanged bowel management at the 1-year follow-up.
At 1 year after the SCI, among all 1250 patients, 725 (58.0%: 143 of 254 females [56.3%] and 582 of 996 males [58.4%]) showed independent, efficient bowel management. Among these, 143 (19.7%) were female, and 263 (37.7%) had tetraplegia.
For model validation, a total of 586 patients with traumatic SCIs were enrolled between January 2013 and December 2014. The late outcome, measured at 1 year after injury, was available in 206 patients. However, complete initial ISNCSCI and SCIM assessments were missing for 17 and 6 patients, respectively (3 patients had incomplete data for both the ISNCSCI and the SCIM). Therefore, 186 patients with complete data sets were included in the validation analysis. The clinical characteristics at inclusion are shown in Table 1 for patients included in the model derivation and for patients lost at the 1-year follow-up. The derivation group included significantly more patients with complete lesions (AIS A) and significantly fewer patients with lesions at the sacral neurological level, compared with the validation group (Table 1). In the validation group, independent, efficient bowel management was observed in 33 (17.7%) patients at the initial assessment, and all these patients had maintained independent, efficient bowel management at the 1-year follow-up. At 1 year after the SCI, 122 (65.6%) patients showed independent, efficient bowel management. Among those, 19 (15.6%) were female, and 54 (44.2%) had tetraplegia.
Prediction Models
The first predictor identified was the ISNCSCI total motor score, a cumulative score defined as the sum of the UEMS and the LEMS. The aROC of the simplified model, based on this single predictor, was 0.837 (95% CI: 0.815-0.859; Figure 1 and the calibration plot in Supplementary Figure S5). The relationship between the ISNCSCI total motor score, measured at the time of inclusion, and the corresponding estimated probability of achieving independent, efficient bowel management at 1 year are shown in Figure 2 and Table S6.
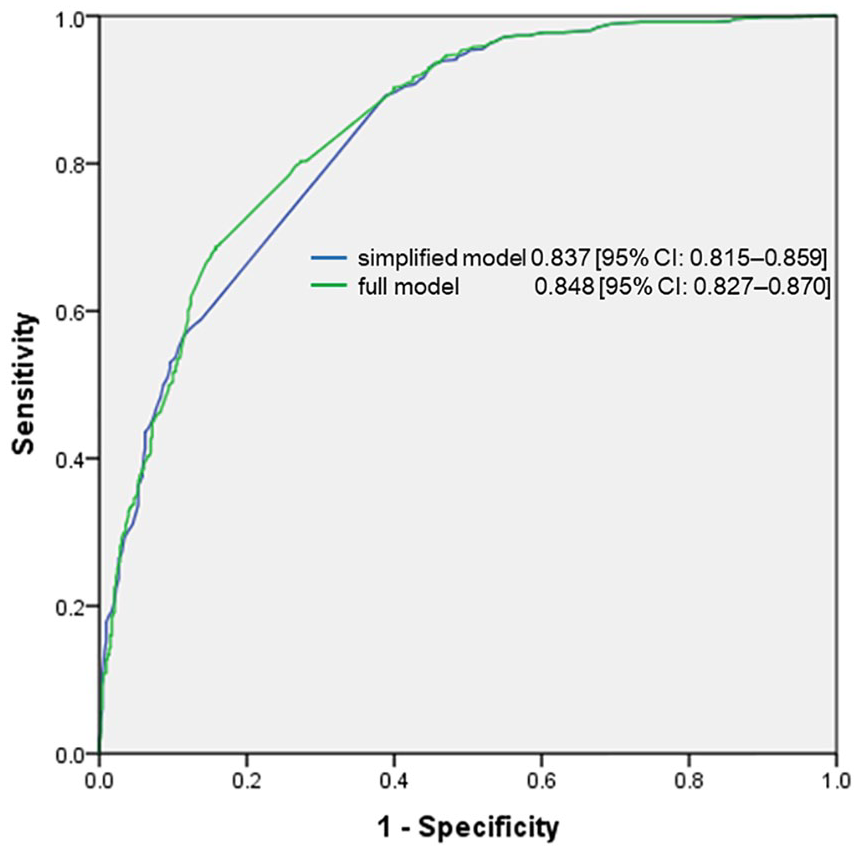
Receiver operating characteristics curve (ROC) and corresponding area under the ROC for 2 prediction models. The 2 models (simplified = based on the International Standards for Neurological Classification of Spinal Cord Injury [ISNCSCI] total motor score; full = based on the total ISNCSCI motor score plus Spinal Cord Independence Measure item 3a) predicted the achievement of independent, reliable bowel management at 1 year after traumatic spinal cord injury.
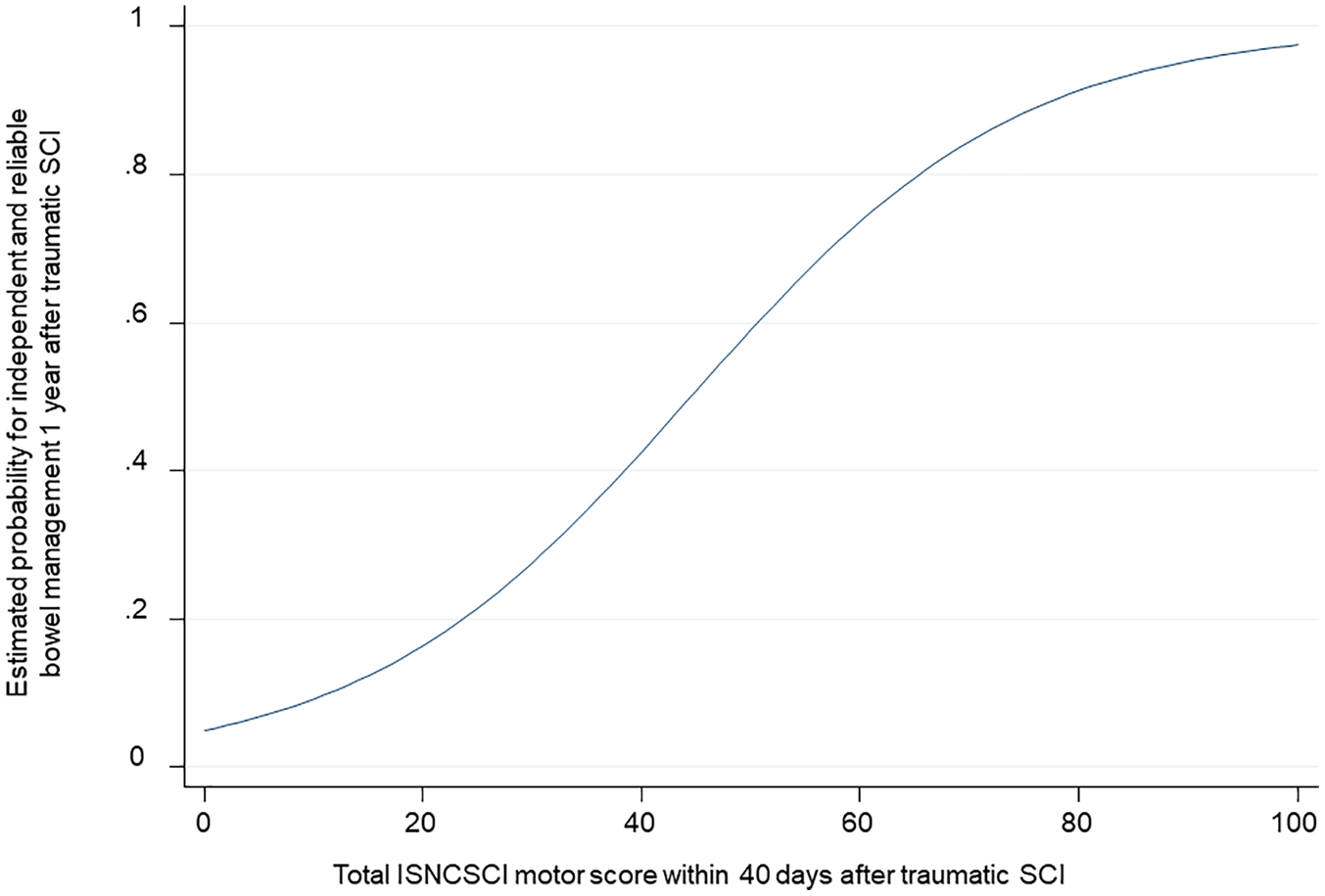
Relationship between the International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI) total motor score, evaluated at the time of inclusion, and the corresponding estimated probability of achieving independent, reliable bowel management at 1 year after traumatic spinal cord injury (SCI).
We then applied a sensitivity analysis to this model, after excluding 55 patients with only very acute measurements and 33 patients who displayed independent, efficient bowel management at inclusion. In this analysis, the aROC was 0.820 (95% CI: 0.768 to 0.883).
The second predictor identified was item 3a in SCIMs II and III—that is, independence in dressing the upper body. The addition of this second predictor to the first predictor conferred a small, but significant (
The complete function of the full model, based on the 2 predictors, and an example of its application is shown in the supplementary material (S8 Text).
The validation cohort confirmed that both models had very high predictive powers. The aROC of the model based only on the total motor score was 0.817 (95% CI: 0.754-0.881); the aROC of the model based on the 2 predictors—that is, the ISNCSCI total motor score and item 3a of the SCIM—was 0.836 (95% CI: 0.775-0.896). The addition of item 3a of the SCIM in the validation cohort did not significantly improve the model (
Discussion
Main Findings
The present study provided the first models for predicting the achievement of independent, reliable bowel management at 1 year after traumatic SCI. The aROCs of our 2 models (0.837 and 0.848) indicated high predictive accuracy—that is, the models displayed a high frequency of discriminating correctly between positive and negative cases in the prediction process. The first model relied on a single predictor—the total motor score of the ISNCSCI—which reflected the sum of the muscle strength of all key muscle groups evaluated. This predictor is commonly assessed as part of a standard neurological examination of patients with SCIs. Experienced examiners showed very good agreement on the total ISNCSCI motor score. 33 Collecting the values of this predictor was simple, rapid, noninvasive, and inexpensive, and its assessment required no specific instrument.
The second model included a second predictor—that is, item 3a of the SCIM (level of independence in dressing the upper body). The second predictor conferred a small improvement in the aROC for the derivation cohort, but no significant improvement for the validation cohort. Therefore, we recommend using the first model, which was based on the single predictor, the total motor score of ISNCSCI. Moreover, our study showed that at 1 year after a traumatic SCI, 58% of patients achieved independent, reliable bowel management.
The Results in Context of the Literature
Liu et al 34 performed a cross-sectional study to identify potential predictors of severe neurogenic bowel dysfunction after a SCI. That study found that a higher spinal level, the completeness of nerve damage, and a longer duration with a SCI (more than 10 years) were associated with greater dysfunction severity. 34 In the present study, the level of injury and the AIS grade were considered among the potential predictors, but they did not emerge as main predictors in our analysis. However, it could be argued that the main predictor found in our study, the total motor score, reflected the degree of neurological impairment associated with the SCI, 24 and consequently, it was related to the spinal level and the completeness of nerve injury. Moreover, the study by Liu et al 34 showed major differences from the present study in terms of the design, sample, and outcome measures. They performed a monocentric study, which included 142 patients with chronic SCIs (injury durations were 1 to over 10 years). Their patients completed 2 questionnaires (the Neurogenic Bowel Dysfunction Score and the Beck Depression Inventory) through mail correspondence. In a recent study, the main predictor that emerged in the present study, the ISNCSCI total motor score, was also identified as a predictor of complete functional recovery at 1 year after SCI. 35
Belliveau et al 36 applied artificial neural network models to predict the self-reported ambulation ability and self-care activities at 1 year after discharge. However, their models for predicting nonambulation outcomes were only moderately accurate, and thus, they required further optimization. 36
Previous studies have shown that changes in sensory scores for the thoracic segments were modestly correlated with the changes in overall neurological and functional status because of the high variability and the difficulty in localizing these dermatomes accurately and repeatedly.37,38 Therefore, we opted to exclude the ISNCSCI thoracic sensory scores from the potential predictors in the present analysis.
In the present study, we did not evaluate the effect of injury duration on the bowel outcome. Many authors have agreed that at 1 year after SCI, bowel function stabilizes; thus, the 1-year bowel outcome might reflect the evolution of chronic bowel function.6,39-41 However, other authors have reported that function deteriorated with time postinjury. 42
Strengths and Limitations
The main strengths of this study were the large number of patients included and the amount of data analyzed to derive and validate the prediction models. To the best of our knowledge, our cohort was one of the largest ever analyzed regarding the evolution of bowel function after traumatic SCI.
Another strength of this study was the rigorous, methodological collection of prospective data from the EMSCI database. Of note, we recently applied the same methodology for deriving a prediction model of bladder function after traumatic SCI. 16 That model was subsequently validated in a large, independent cohort, based on data from the National Spinal Cord Injury Database, 18 on US patients with traumatic SCIs and, additionally, on patients with ischemic SCIs. 17
Finally, the outcome measure used in the present study (ie, the SCIM) was reliable and valid; it might be the best primary outcome measure for functional capacity in future phase 3 clinical trials. 43 It should be noted that the SCIM is an objective outcome measure; it does not consider any subjective evaluation of outcome or the level of patient satisfaction.
One potential limitation of the present study was that the EMSCI initially applied SCIM version II and, then, promptly adopted version III, after its introduction. Consequently, in our derivation cohort, some patients were evaluated with SCIM version II and others with SCIM version III. In contrast, all patients who were included at a later time for the prospective validation were evaluated with SCIM version III.26,27 The 2 versions of SCIM were slightly different in the scoring of the second predictor identified, item 3a of SCIM.26,27 This small difference in item 3a scoring between the 2 SCIM versions might partly explain why the addition of this second predictor conferred a small improvement of the aROC when applied to the derivation cohort and no improvement to the validation cohort. Another potential explanation might be the limited size of the validation cohort.
In our analysis, SCIM scores were treated as continuous variables, as suggested by Pasta. 44 This choice might be considered controversial, and it should be considered a methodological limitation of our analysis.
All our patients received rehabilitative treatment, including state-of-the-art management of neurogenic bladder and bowel dysfunction. Nevertheless, it should be noted that the treatments were not standardized among the different EMSCI centers; thus, confounding-by-center effects could not be excluded. However, despite the presumed heterogeneity in bowel management regimes applied in the various centers, our analysis identified a main predictor of recovery, which was confirmed in the prospective validation.
Another drawback of our study was the lack of external validation. This should be performed in the future, by applying our models to a sample of patients who were not included in EMSCI, to assess the generalizability of our findings. Moreover, the EMSCI data set did not provide a clear distinction between patients with upper versus lower motor neuron lesions. Future studies could evaluate the model performance in these 2 categories.
Finally, another limitation of our study was the substantial number of patients who missed the 1-year follow-up. However, the cohorts of patients with and without a 1-year follow-up showed only a few differences, in terms of baseline characteristics. Moreover, our analysis took this limitation into account with the weighting approach, which limited the possible impact of missing data.
Implications for Research
Currently, a large spectrum of conservative and surgical interventions is available for the management of bowel dysfunction after SCIs. 11 However, the lack of high-level evidence studies and the consequent empirical use of different strategies might be one of the principal causes of the frequent failure of bowel management programs. 11 Many authors have highlighted the urgent need for well-designed clinical trials to evaluate the efficacy of different interventions.11,19,39,40
The prediction model provided in our study might be used to identify patients with a high probability of achieving independent, efficient bowel management at 1 year after a SCI. Moreover, this information is essential for patient allocation in prospective studies. 13
Recently, a closed-loop optogenetic neuromodulation system has succeeded at targeting specific neurons to control urinary tract function. 45 These promising findings warrant animal and translational research to assess whether optogenetics could also play a role in the management of neurogenic bowel dysfunction in the future. 45
Implications for Practice
Our findings that 58% of patients with traumatic SCIs achieved independent, reliable bowel management at 1 year after injury were both statistically and clinically relevant. From a statistical point of view, the outcome distribution in our cohort (close to 1:1) made our sample an ideal data set for the derivation of a prediction model. From a clinical perspective, 1 of 2 patients achieved a positive bowel outcome at 1 year. Our prediction model can make it possible to inform all individuals involved in the rehabilitative process (patients, caregivers, rehabilitative team) at the beginning of the rehabilitative phase on the potential bowel outcome. Indeed, this prediction could provide a basis for early counseling for patients and family members. Clear counseling is a key point in psychological support for patients and caregivers, and it promotes compliance with the treatments recommended by the rehabilitative team. 9 Moreover, early definition of the rehabilitative goals is essential for optimizing resource allocation in the rehabilitative phase. For example, in the case of a very low probability of recovery, the team may promptly identify and instruct a caregiver who can take charge of bowel management after discharge. 9 Finally, a reliable prediction could promote the prompt prescription of appropriate durable medical equipment and environmental modifications to favor a successful discharge. 9 Moreover, several authors have highlighted that an early prediction of outcome would be necessary to identify patients with poor outcome with a conservative approach. These patients may require additional evaluations for surgical interventions.39,40 The optimization of bowel management could result in improving the level of care for patients with SCIs and reducing the heavy total costs to society for conservative bowel management.1,7,40,46
Conclusions
Our study provided the first model for predicting whether a patient with SCI is likely to achieve independent, reliable bowel management at 1 year after traumatic SCI. The use of our model could improve the design of future clinical trials and facilitate planning for the level of care that might be required in patients affected by neurogenic bowel dysfunction after traumatic SCI.
Supplemental Material
Supplemental_Material – Supplemental material for Bowel Outcome Prediction After Traumatic Spinal Cord Injury: Longitudinal Cohort Study
Supplemental material, Supplemental_Material for Bowel Outcome Prediction After Traumatic Spinal Cord Injury: Longitudinal Cohort Study by Chiara Pavese, Lucas M. Bachmann, Martin Schubert, Armin Curt, Ulrich Mehnert, Marc P. Schneider, Giorgio Scivoletto, Enrico Finazzi Agrò, Doris Maier, Rainer Abel, Norbert Weidner, Rüdiger Rupp, Alfons G. Kessels and Thomas M. Kessler in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Enrico Finazzi Agrò received a speaker honorarium from Astellas, Coloplast, Glaxo Smith Kline, and Wellspect; he is consultant for Allergan, Wellspect, and Laborie. Rainer Abel is member of an advisory panel of Coloplast.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Chiara Pavese is partially supported by an investigator fellowship from Centro Comunicazione e Ricerca, Collegio Ghislieri, Pavia, Italy. EMSCI is funded by International Foundation for Research in Paraplegia, Wings for Life, and Deutsche Stiftung Querschnittlähmung.
Supplementary material for this article is available on the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
