Abstract
Introduction
Sudden mechanical perturbation of the spinal cord results in a complex array of neurological deficits, including permanent paralysis and loss of normal sensation. Pharmacological interventions to repair the damaged spinal cord and partially restore function have, unfortunately, failed to demonstrate efficacy in human clinical trials. While attributable to a number of factors (including the distinct possibility that there has been no measurable “effect”), one often-cited problem is that extensive heterogeneity in normal recovery from an acute spinal cord injury (SCI) masks potential treatment effects. 1
Biomarkers have been proposed to improve diagnosis and prognosis of acute SCI and, when implemented in a clinical trial, increase the likelihood of detecting a significant treatment effect. 2 To date, studies have focused on various cerebrospinal fluid and serum concentrations, including pro-inflammatory and neurofilament proteins,3,4 and the prediction of neurological outcomes derived from the International Standards for the Neurological Classification of Spinal Cord Injury (ISNCSCI). Somewhat surprisingly, potential biomarkers derived from conventional hematological tests, commonly performed by laboratories in acute care hospitals, have not been examined. The concentration of serum albumin represents one particularly notable oversight. Across a variety of neurological conditions, low serum albumin concentrations (ie, hypoalbuminemia) correlate with poor neurological and functional outcomes.5 -9
The goal of this study was to examine the utility of serum albumin concentrations early after SCI in predicting long-term neurological outcomes after an acute traumatic SCI. As a measure of neurological function, we examined the recovery of muscle strength according to the ISNCSCI. Serum albumin concentrations at 24 to 72 hours, 1, 2, and 4 weeks post injury were considered. Based on observations in other neurological conditions, our hypothesis was that low serum albumin would independently predict poor long-term outcomes.
Methods
Study Design and Data Source
This was a secondary analysis of a completed clinical trial. Specifically, we gathered data from a prospective phase III, placebo controlled, multicenter study testing the efficacy of GM-1 (monosialotetrahexosylganglioside) ganglioside therapy in acute, traumatic SCI.10,11 Full design, recruitment and enrollment details have been published previously. 11 The trial, which ran from 1992 to 1998, failed to demonstrate a significant treatment effect between GM-1-treated and placebo patients. Subsequent analyses of the data have been performed to characterize the trajectory and amount of spontaneous recovery from acute SCI.1,12,13
Cohort Definition: Inclusion Criteria
Of the 797 patients enrolled in the original trial, we included subjects with a baseline, 1,- 2,- and/or 4-week measurement of serum albumin concentrations AND a lower extremity motor score at 52 weeks. The workflow for the individuals included/excluded from our analysis is highlighted in Supplementary Figure 1.
Outcome (Dependent) Variables
Our analysis focused on prediction of 2 dependent variables. First, we considered the original trial’s definition of improvement (ie, “marked recovery at 26 weeks”). This endpoint was designed specifically for the Sygen trial in an attempt to dichotomize the efficacy of GM-1 administration. In brief, marked recovery was defined as at least a 2 grade equivalent improvement in the Modified Benzel Classification from the baseline American Spinal Injury Association Impairment Scale (AIS) score. For patients with AIS-A and B injuries at baseline, this meant a conversion to (at a minimum) AIS-C and -D, respectively (ie, sensory and motor function caudal to the level of injury). AIS-C and D patients were required to recover limited or unlimited walking function.
In addition to marked recovery at 26 weeks, we also examined motor scores at 1-year post injury. In brief, key muscles in the upper and lower extremities were examined according to the ISNCSCI. 14 Our analysis specifically focused on lower extremity motor scores, which represent neurological function caudal to both thoracic and cervical lesions.
Exposure (Predictor) Variables
The original Sygen trial involved the collection of routine blood chemistry, including serum albumin concentrations, at baseline (24-72 hours), 1, 2, and 4 weeks post injury. The blood chemistry data have not been previously published. Blood serum was analyzed by each of the participating centers according to institutional guidelines. To consider differences in serum albumin related to the center, baseline concentrations were examined across trial sites. This analysis was performed in AIS-A subjects (see Supplementary Figure 2). There were no significant differences in albumin concentrations between centers. Based on these observations, no further center effects were considered.
Primary Statistical Analyses
For the purposes of our primary analysis, placebo and GM-1 treated groups were pooled. The same pooling procedure has been done previously based on the null effect of GM-1 therapy. 15 Standard descriptive statistics were used to characterize subject characteristics and serum albumin concentrations. A 1-way analysis of variance was used to examine differences in serum albumin concentrations at baseline, 1, 2, and 4 weeks post injury based on 1-week AIS grade. Post hoc pairwise comparisons were Bonferroni corrected.
To examine the relationship between serum albumin and motor recovery, multivariable linear and logistical regressions were performed. Age, sex, treatment group (ie, placebo or GM-1), and injury completeness (AIS A/B and C/D at 1 week post injury) were included in the multivariable linear and adjusted logistical regression models as potential confounding factors. Effect sizes (unstandardized coefficient Β) and 95% confidence intervals (95% CI) were examined for linear regression for each serum albumin measurement (eg, baseline, 1, 2, or 4 weeks post injury). Coefficients greater or less than 0 with nonoverlapping 95% CIs indicate positive or negative association with motor scores at 52 weeks post injury, respectively. A similar logistical regression procedure was used to determine odds ratios (ORs) for marked recovery (yes/no) at 26 weeks. ORs greater or less than 1 indicate a positive or negative association between the variable and likelihood of achieving marked recovery, respectively. 95% CI that are nonoverlapping with 1 denote a significant OR. Reference categories for linear and logistical regression (ie, categorical variables) are shown in corresponding tables. Receiver operating characteristic (ROC) curves were examined to determine the sensitivity and specificity of albumin concentrations (baseline to 4 weeks) to predict marked recovery.
Sensitivity Analyses
A sensitivity analysis was performed in the Sygen placebo group to further address the potential confound of GM-1 treatment effects (beyond including treatment group in the original linear and logistical regression models as a confounding variable). Additionally, a sensitivity analysis was performed to examine the impact of polytrauma on the relationship between long-term neurological outcomes and albumin concentrations. Polytrauma was defined as 3 or more of the following associated injuries at the admission/ entrance to study (ie, within 72 hours post injury): skin and soft tissue, head, ear/nose/throat, cardiac, pulmonary, gastrointestinal, genitourinary, or musculoskeletal (other than spine). Adjusted logistical and multivariable linear regression models were performed at 4 weeks to examine marked recovery and 52-week lower extremity motor scores.
Results
Cohort Summary
A total of 591 subjects were included in our analysis. Subject demographics and injury characteristics (including for those excluded from our analysis) are shown in Table 1.
Summary of Subject Characteristics, Albumin Concentrations, and Neurological/Functional Outcomes.
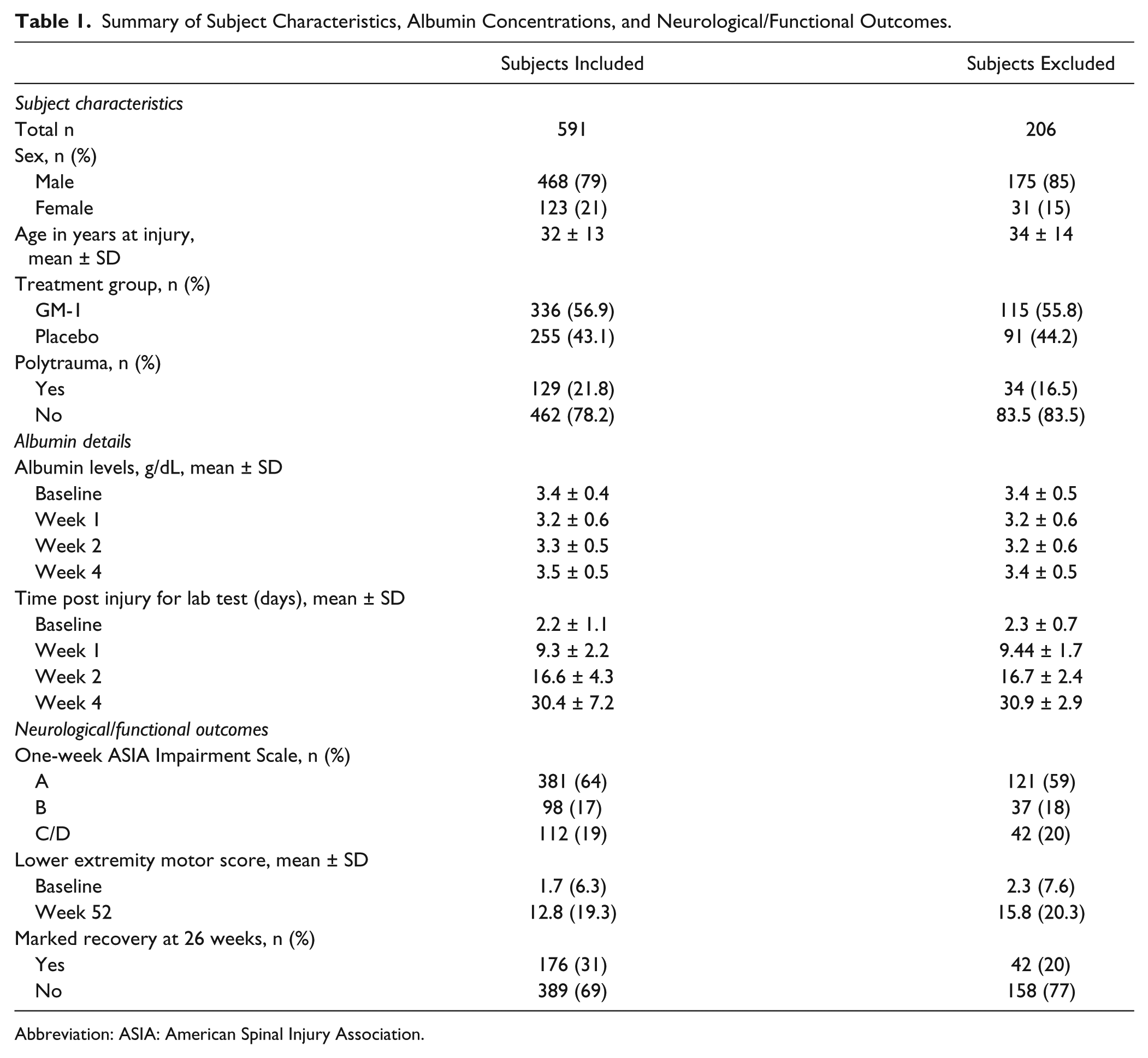
Abbreviation: ASIA: American Spinal Injury Association.
Serum Albumin Concentrations
A 1-way ANOVA revealed significant differences in serum albumin concentrations by initial AIS grade (Supplementary Table 1). Post hoc comparisons indicated that serum albumin concentrations were lower among more severely injured individuals (Supplementary Table 2, summarized in Figure 1).
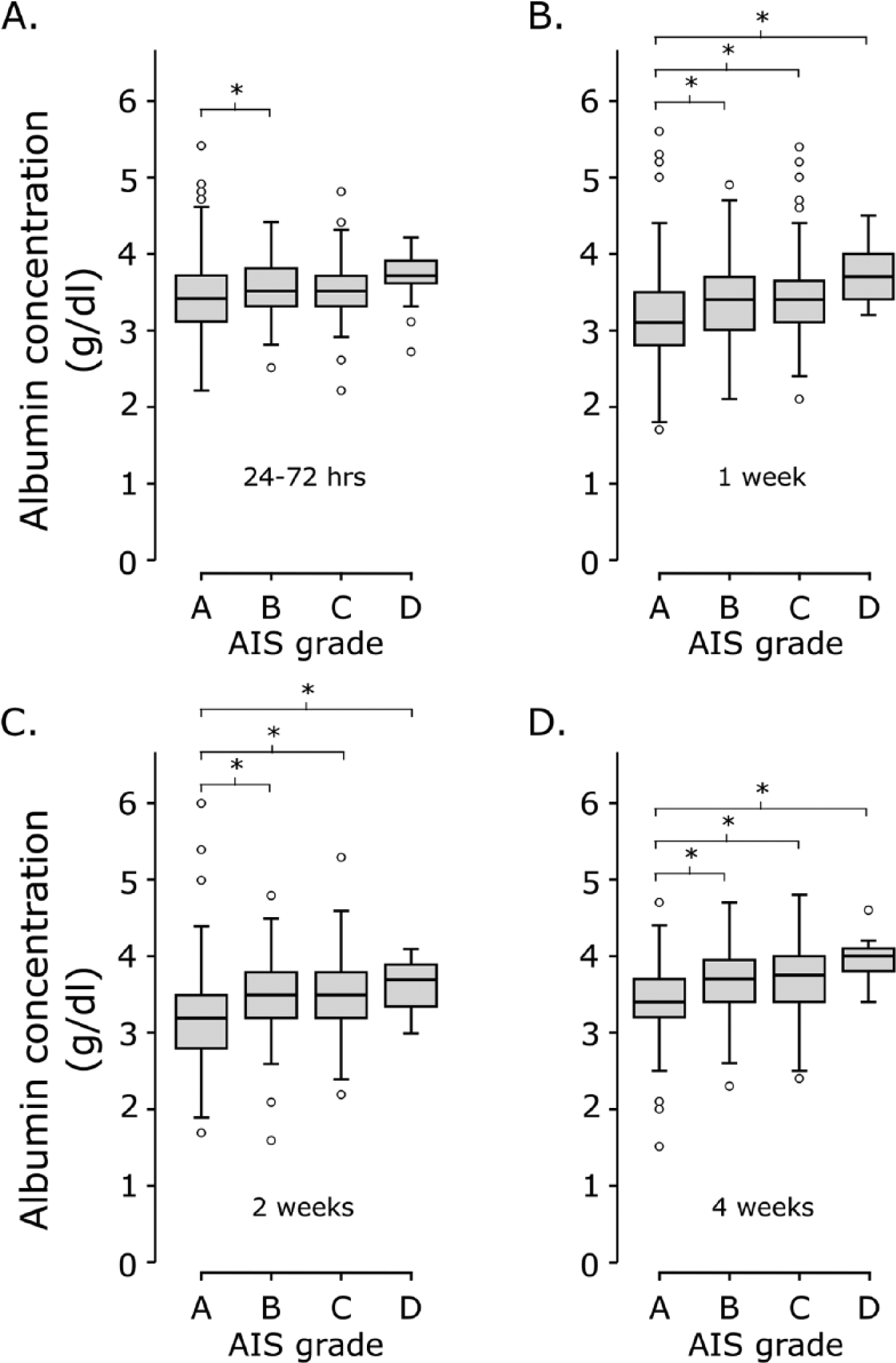
Relationship between albumin and injury severity. Serum albumin at 4 time points post injury (baseline, 1, 2, and 4 weeks, panels A, B, C, and D, respectively) by AIS grades. Based on a 1-way analysis of variance (results provided in Supplementary Tables 1 and 2), serum albumin concentrations were decreased in more severe (AIS-A) injuries at all 1-month time points after injury. AIS, American Spinal Injury Association Impairment Scale. *Denotes significant pairwise comparisons (
Predicting “Marked Recovery” at 26 Weeks
Unadjusted ORs were >1 and significant (lower 95% CI >1) at all time points for the association of serum albumin and marked recovery at 26 weeks (Table 2). These values indicate that higher serum albumin concentrations were associated with greater odds of marked recovery. Unadjusted models also indicated that incomplete injuries were more likely to experience marked recovery (OR < 1) compared with complete (reference group). As in the original trial, there was no significant differences in marked recovery between the treated (GM-1 administration) and placebo group (reference).
Logistical Regression for Marked Motor Recovery and Serum Albumin Concentrations.
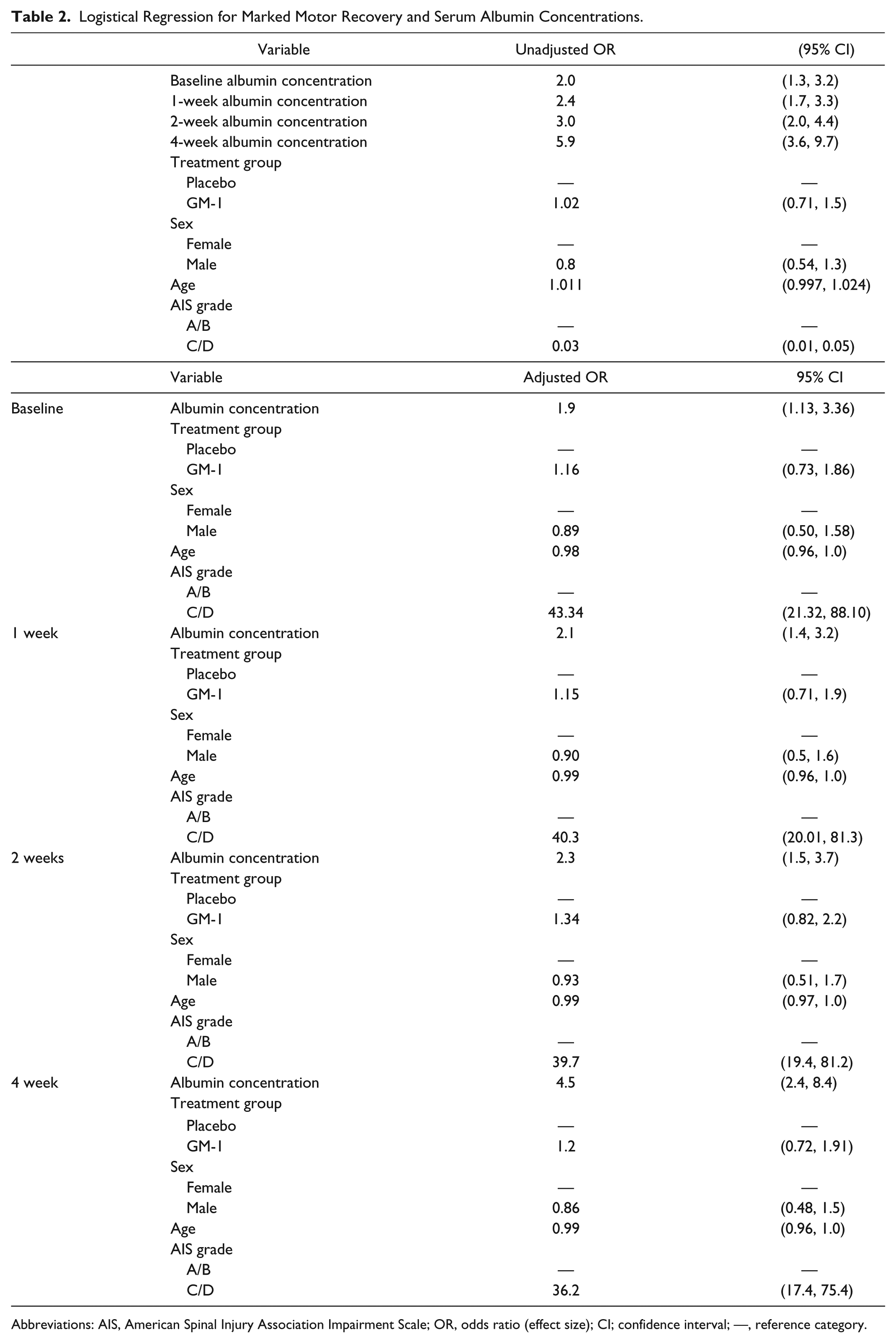
Abbreviations: AIS, American Spinal Injury Association Impairment Scale; OR, odds ratio (effect size); CI; confidence interval; —, reference category.
In adjusted logistical regression models (ie, age, sex, treatment, and initial AIS grades in one model), the association between serum albumin concentrations remained significant (Table 2). This indicates that subjects with higher serum albumin concentrations were more likely to achieve marked recovery at 26 weeks independent of age, sex, treatment, and injury completeness.
An ROC analysis indicated an increase in sensitivity and specificity for the prediction of marked recovery based on albumin concentrations from baseline to 4 weeks post injury (Figure 2A).
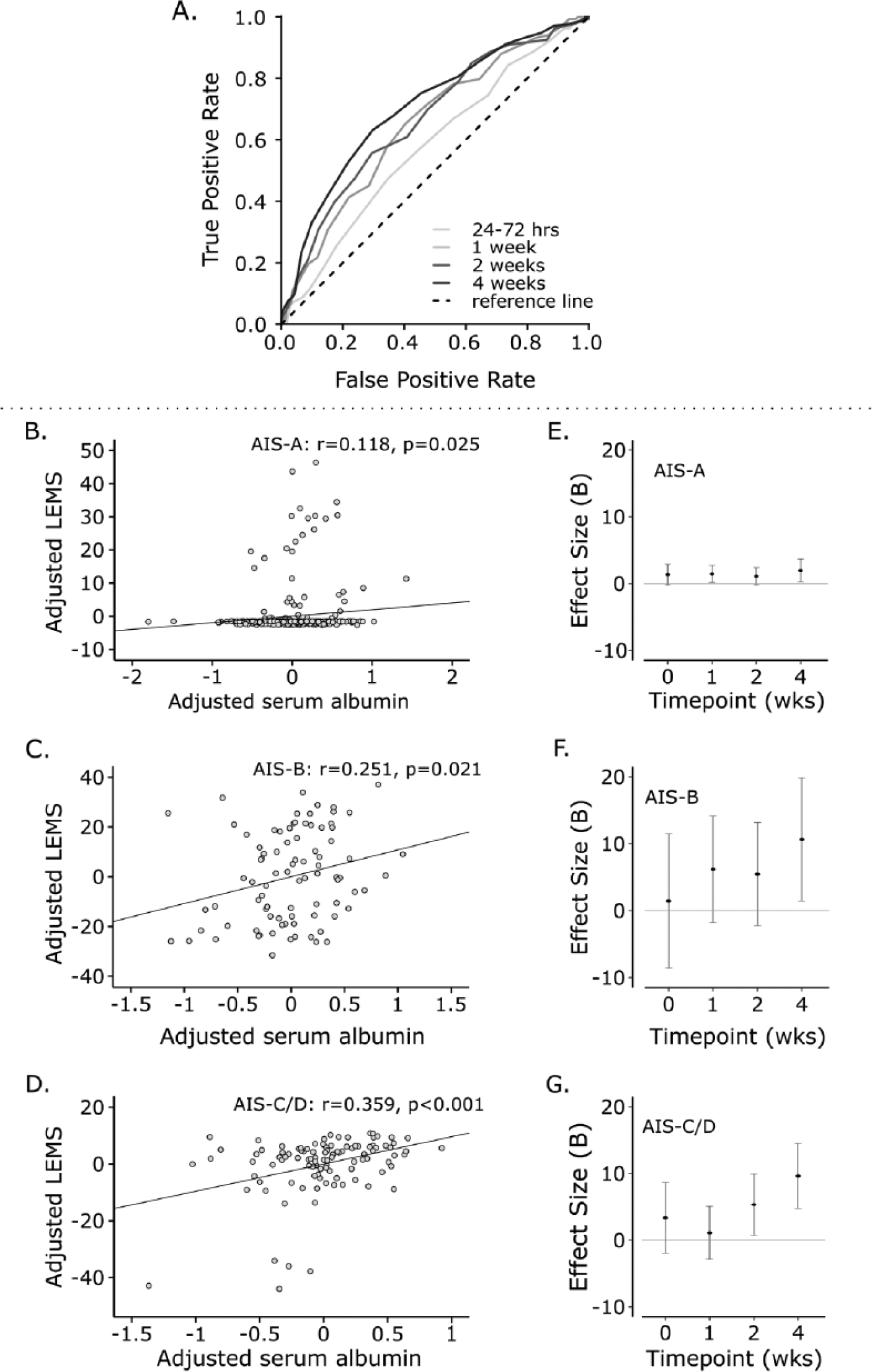
Relationship between albumin and 52-week motor scores. Receiver operating characteristic (ROC) curve analysis for serum albumin concentrations at baseline, 1, 2, and 4 weeks post injury and marked recovery (A). The area under the curve was significant for albumin concentrations at all time points (
Predicting Lower Extremity Motor Scores at 52 Weeks Post Injury
According to a multivariable linear regression analysis (adjusted for age, sex, treatment, and initial injury completeness), serum albumin concentration at baseline, 1, 2, and 4 weeks were significantly predictive of 52-week lower extremity motor score (Table 3). At all time points, higher serum albumin concentrations were associated with greater lower extremity motor scores (Β > 0 and
Linear Regression for 52-Week Motor Score and Serum Albumin Concentrations.
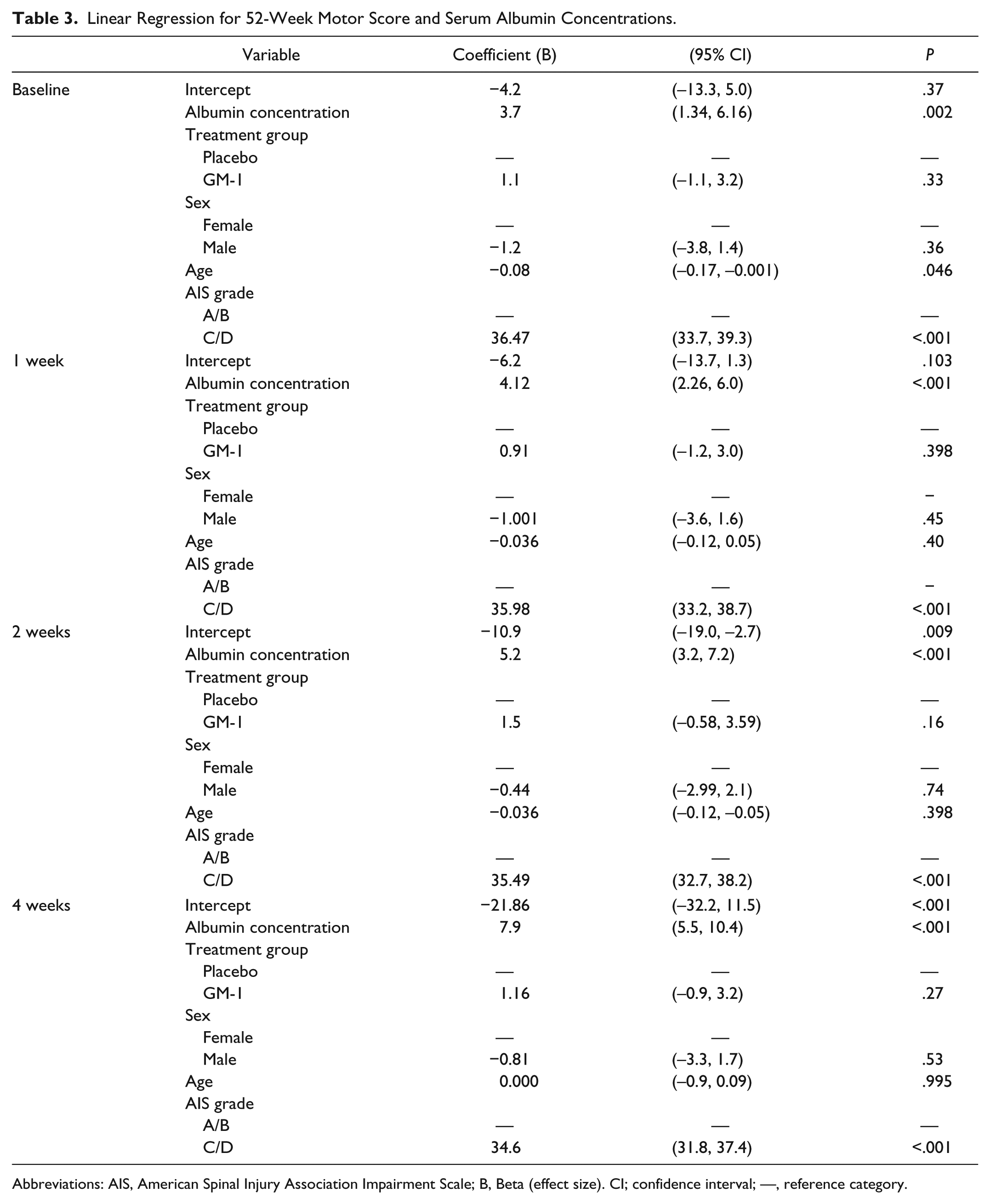
Abbreviations: AIS, American Spinal Injury Association Impairment Scale; B, Beta (effect size). CI; confidence interval; —, reference category.
Post Hoc Analysis of Individual Injury Severities and Serum Albumin Concentrations, and Bivariate Correlations Between Albumin Concentrations and Inflammation
A stepwise linear regression was performed to determine if albumin concentrations at 4 weeks improved the association between AIS grades and 52-week lower extremity motor scores. This analysis revealed a significant increase in the adjusted
Post hoc, we also performed separate linear and logistical regression analyses of individual AIS grades (A, B, C/D, see Figure 2). Serum albumin concentrations were significant in the prediction of lower extremity motor score by 4 weeks across all injury severities (Supplementary Tables 3-5). Marked recovery was significantly associated with albumin concentrations in AIS-C/D injuries at 2 and 4 weeks (Supplementary Tables 6-8). These results confirm the association between serum albumin concentrations and long-term neurological outcomes across different injury severities.
Correlations between albumin and inflammatory markers are illustrated in Figure 3. By 4 weeks after injury, significant correlations emerged between albumin concentrations and inflammation. These correlations suggest that lower albumin concentrations were associated with greater inflammation.
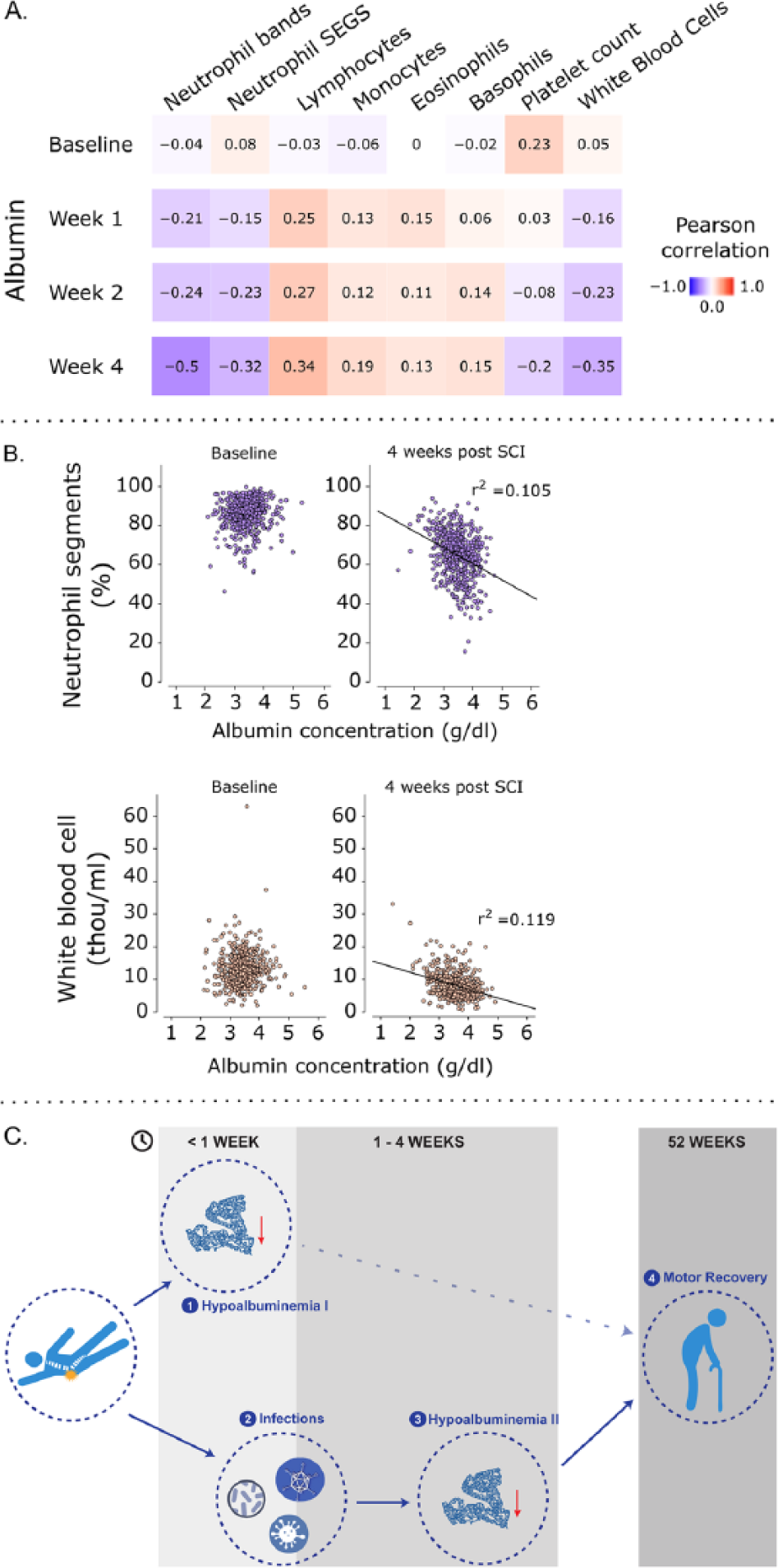
Relationship between serum albumin concentration and inflammation. Pearson correlations between serum albumin and inflammatory markers at 1-month time points after injury (A). Note that correlation coefficients increased from baseline to 4 weeks. Illustration of raw data showcasing the emerging relationship between serum albumin and inflammation (B). Based on these observations, the proposed link between early serum albumin concentrations, inflammation, and neurological outcomes at 52 weeks is highlighted in panel C. Hypoalbuminemia resulting directly from traumatic spinal cord injury (SCI; eg, blood loss) has little to no impact on long-term neurological outcomes (1). In contrast, infections arising in the initial days to weeks after injury (2) leads to a second phase or persistent hypoalbuminemia (3), contributing to poor neurological recovery (4). 21
Sensitivity Analysis of Placebo Group Only
The results from multivariable linear and adjusted logistical regression analyses of the placebo group confirmed the significant association between 4-week albumin concentrations and 52-week motor scores and marked recovery, respectively (Supplementary Tables 9 and 10). These results suggest the association between albumin concentrations and long-term neurological outcomes is unrelated to GM-1 administration.
Albumin concentrations at 4 weeks post injury remained significant (ie, linear and logistical regression) with the inclusion of polytrauma (3 or more associated injuries accompanying SCI) as a confounding factor (see Supplementary Tables 11 and 12). This indicates that albumin concentrations are associated with long-term neurological outcomes independent of polytrauma.
Discussion
In the present study, low serum albumin concentrations early after SCI were associated with poor long-term neurological outcomes. This was shown for both lower extremity motor scores and the recovery of 2 AIS/Benzel grades (ie, marked recovery), adjusting for initial differences in injury severity, age, and sex. Post hoc and sensitivity analyses confirmed the robustness of the association, ruling out several potential confounding factors (eg, treatment groups, injury severity, polytrauma). Based on these observations, the measurement of serum albumin concentrations represents a straightforward and routinely examined hematological biomarker to improve the accuracy of prognosis of long-term motor outcomes after traumatic SCI.
In one previous study, low serum albumin concentrations were associated with increased risk of mechanical ventilation and mortality after acute SCI. 16 To our knowledge, our analysis represents the first time a conventional laboratory biomarker in the blood has been examined for the prediction of a neurological outcome after SCI. Our analysis benefited from a very large cohort of acute SCI patients (n = 591 of the original 797) sampled in the course of a clinical trial, with repeated blood chemistry assessments at baseline (average: 2 days), 1, 2, and 4 weeks. The value of serum albumin was demonstrated for 2 key measures of function, including the original trial endpoint designed to assess therapeutic efficacy (ie, marked recovery at 26 weeks). 10 In line with our observations, low serum albumin concentrations are associated with poor outcomes in traumatic brain injury,5,7 ischemic stroke, 6 amyotrophic lateral sclerosis, 9 and Guillain-Barré syndrome. 8
Application in Spinal Cord Injury Clinical Trials
Accurately predicting the extent of motor recovery after SCI has proven immensely challenging. 1 So far, the only strong indication for how much a person is likely to recover is sensory and motor sparing caudal to the injury site, measured according to the ISNCSCI. 14 There are several notable concerns with sensory and motor testing, including the subjective nature of characterizing manual muscle strength and grading sensation. Although progress has been made to better use data from the standard neurological examination for the purposes of prognosis and patient stratification, 17 extensive variability in recovery remains.
The concern in clinical trials is that variable spontaneous recovery will mask or exaggerate therapeutic benefits, either of which could result in the potential for an erroneous conclusion. This is especially problematic in small, early phase clinical trials. To improve the prediction of neurological recovery based currently on the international standards, objective predictive biomarkers are urgently needed.
Several factors make serum albumin concentration a feasible and convenient candidate biomarker that could improve the stratification of acute SCI patients. Albumin is a part of a standard blood chemistry workup, commonly evaluated in trauma centers and by physicians who are most likely to be involved in an acute clinical trial. In contrast, the measurement of specialized small molecules in serum and cerebrospinal fluid requires unfamiliar and more expensive assays.4,18,19 In addition, serum albumin is useful in the prediction of long-term motor outcomes based on measurements performed within the first month. This is important because many acute clinical trials aim to initiate therapies within a 1- to 4-week time window and will require stratification at early time points. Furthermore, serum albumin concentrations improved prediction beyond standard measures of injury severity (eg, AIS grades). This was confirmed in a post hoc linear and logistical regression analyses, including examination within individual AIS grades. The ISNCSCI is the best available tool for patient stratification in forthcoming clinical trials. Thus, the goal of a biomarker, in the short term, should be to complement these existing standards.
Mechanisms and Future Directions
Why serum albumin concentrations serve a prognostic role in neurological conditions remains largely unknown. Several factors may contribute to hypoalbuminemia after SCI, including acute trauma, infection, and surgical intervention (ie, “reduced hepatic synthesis, increased leakage into the interstitial space, and accelerated catabolism”). 20 In this way, low albumin concentrations may reflect a global picture of poor health, which is associated with limited neurological recovery. Hypoalbuminemia directly related to the initial trauma (ie, measured at 24-72 hours post injury) was only weakly related to long-term neurological outcomes (ie, 52-week motor score only).
One alternative emerging theory is that hypoalbuminemia reflects a state of pro-inflammation.
9
In terms of traumatic insult to the central nervous system, a number of candidate biological mechanisms link systemic inflammation and neurological deficits, including secondary damage and axonal degeneration.
22
As such, low albumin concentrations may represent a surrogate measure of systemic inflammation and
Limitations
The primary limitation of the current study is that we utilized retrospective data, from a nearly 20-year-old clinical trial. Of the original 797 subjects, our analysis focused on a cohort of 591. While a comparison of the Sygen data with more recent prospective collections of data have reported no differences in neurological recovery, 13 we cannot exclude that albumin concentrations were affected by the standards of acute care during the clinical trial period (eg, routine administration of methylprednisolone). Toward addressing this important limitation, next steps should examine serum albumin concentrations in an independent and recently injured cohort. Additionally, serum albumin concentration was only weakly associated with neurological outcome at baseline (24-72 hours according to our primary analysis) and only significant within individual AIS grades by 4 weeks post injury. As a result, application in very acute clinical trials (eg, neuroprotection studies) as a biomarker to stratify patient cohorts may then be rather limited.
In summary, we have demonstrated that serum albumin concentrations are a useful biomarker for the prognosis of long-term neurological outcome after an acute SCI. This has potentially important ramifications for study design and patient stratification in future SCI clinical trials.
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Author Contributions
BT was responsible for data entry, data cleaning, data analyses, interpretation of data, and drafting the manuscript. CRJ, JJC, LG, JMS, and SC were responsible for interpretation of data and revising the manuscript for intellectual content. FG was responsible for primary data collection, interpretation of data, and revising the manuscript for intellectual content. JLKK was responsible for study concept/design, study funding, interpretation of data, and drafting the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CRJ is supported by postdoctoral fellowships from the International Foundation for Research in Paraplegia, Craig H. Neilsen Foundation, and Swiss National Science Foundation. JJC is supported by The Branco Weiss Fellowship – Society in Science and the Michael Smith Foundation for Health Research (MSFHR). JLKK is a MSFHR and Rick Hansen Institute Scholar, and is also supported by Wings for Life (WfL), the International Foundation for Research in Paraplegia, and the Canadian Institutes of Health Research (ERA-NET-NEURON Program). JMS is an OSU Discovery Theme scholar and is supported by the Bundesministerium für Bildung und Forschung (ERA-NET-NEURON Program, #01EW1701A and #01EW1710), WfL, the Hunt and Curtis Endowment, and the National Institute on Disability, Independent Living, and Rehabilitation Research (NIDILRR grant number 90SI5020). NIDILRR is a Center within the Administration for Community Living, Department of Health and Human Services, USA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
