Abstract
Introduction
Traumatic spinal cord injury (SCI) leads to physiological, psychological, and social impairments in thousands of individuals each year worldwide.1-3 Due to persistent efforts in basic and translational research, the understanding of the molecular and cellular mechanisms of SCI has improved in recent decades. This led to new insights into secondary injury mechanisms and their respective contribution to both neuronal damage and the inhibition of neuronal regeneration.1,4
As of date, no pharmacological intervention is available that enhances the degree of neurological recovery beyond that which is expected to occur spontaneously. 5 Nevertheless, improvements in diagnostics, prehospital and acute care management, early surgical intervention, and advanced rehabilitation methods resulted in higher life expectancy, and improved functional outcome in high-income countries.6-10
SCI is characterized by a distinct pathophysiological reaction: the primary mechanical injury caused by the initial trauma followed by the secondary injury maintained by a cascade of biological events.1,3,11 Secondary injury involves vascular changes, inflammation, neurotoxicity, and glial scarring that occurs minutes to weeks after the initial trauma.1,12-14 Following the acute injury phase, secondary injury mechanisms cause expansion of the lesion in both the caudal and cranial directions.3,13 Numerous therapeutic approaches under investigation aim to modulate these secondary mechanisms.7,10,13-16
Effort are ongoing to identify prognostic factors following SCI. 17 Some prognostic factors have already been established, including the severity of the initial lesion. 18 However, these factors neither provide insight into the molecular mechanisms of neurological regeneration nor provide an accurate prognosis of recovery after SCI. Moreover, injury severity alone does not adequately predict the degree of spontaneous neurological recovery. Prognostic biomarkers would be of paramount importance in clinical trials, and also inform clinical decision making. Therefore, a rising interest in quantitative biomarkers has evolved in recent years.3,19 A valid biomarker should reliably predict a clinical outcome. 20 In SCI, the clinical outcome mostly refers to neurological outcome/improvement over time. Numerous studies suggest, that biomarkers from either blood or cerebrospinal fluid (CSF) may be of diagnostic value in the assessment of severity of neuronal damage and even provide an insight into neurological recovery after SCI.21-24 Ideally, a biomarker should be present very early after the injury, not be altered by additional trauma or medication, and be useful in terms of quantification of injury severity and prediction of neurological outcome. The implementation of biomarkers in clinical practice may also provide an avenue to determine the injury severity of unresponsive patients, thereby supporting clinical decision making in acute SCI.2,25 Clinical research is in need of surrogate parameters in order to monitor the potential therapeutic capacity of novel treatment strategies. 26
The aim of this systematic review is to summarize available blood and CSF-derived biomarkers in SCI and evaluate their diagnostic and prognostic value. Furthermore, we aimed to investigate the time-dependency of detectable concentrations of included biomarkers.
Methods
We conducted a literature search in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The systematic review was registered at the international prospective register of systematic reviews (PROSPERO; NHS; https://www.crd.york.ac.uk; ID: CRD42019136029).
Search Strategy
Our search strategy was applied to the following electronic databases: PubMed/MEDLINE, CINAHL, Science Direct, The Cochrane Library, ISI Web of Science, and PEDro.
The databases were searched using the following key words and MeSH (medical subject headings) terms of the National Library of Medicine: “biomarker, blood serum, serum, cerebrospinal fluid, CSF, spinal cord injury, SCI, tetraplegia, quadriplegia, paraplegia.”
Reference lists of relevant articles were manually screened to identify any further articles not identified in the electronic database search.
The search strategy was limited to human studies and publications in English language. No other limits were applied to the search strategy.
Study Selection
Studies were included if they met the following inclusion criteria:
Study design: clinical trials, cohort studies, pilot studies
Patient population: patients with traumatic spinal cord injury
Studies involving at least 1 blood or CSF-derived biomarker
The titles and abstracts of articles retrieved from the electronic database search were screened to determine eligibility for inclusion. Two reviewers (IL and TH) applied the selection criteria independently. Discrepancies were resolved via consensus, and a third reviewer (GM) was consulted if consensus was not achieved.
Data Extraction
The following data were extracted from each included article: (1) characteristics of trial participants (acute or chronic SCI), (2) type of sample collection (blood serum or CSF), (3) the assessed biomarkers, and (4) the time points of samples collection. The synthesis of results includes candidate biomarkers investigated in more than 1 study.
Results
Study Selection
A flow-diagram of the search strategy and study selection is presented in Figure 1. Two researchers (IL and TH) screened potentially eligible articles. The literature search of studies published between 2002 and April 2019 captured 55 articles, of which 32 publications were excluded after reviewing the title and/or abstract. Seven duplicates were removed. The remaining 16 articles were obtained in full text. Eight of the 16 articles met the inclusion criteria and were selected for inclusion in this review.
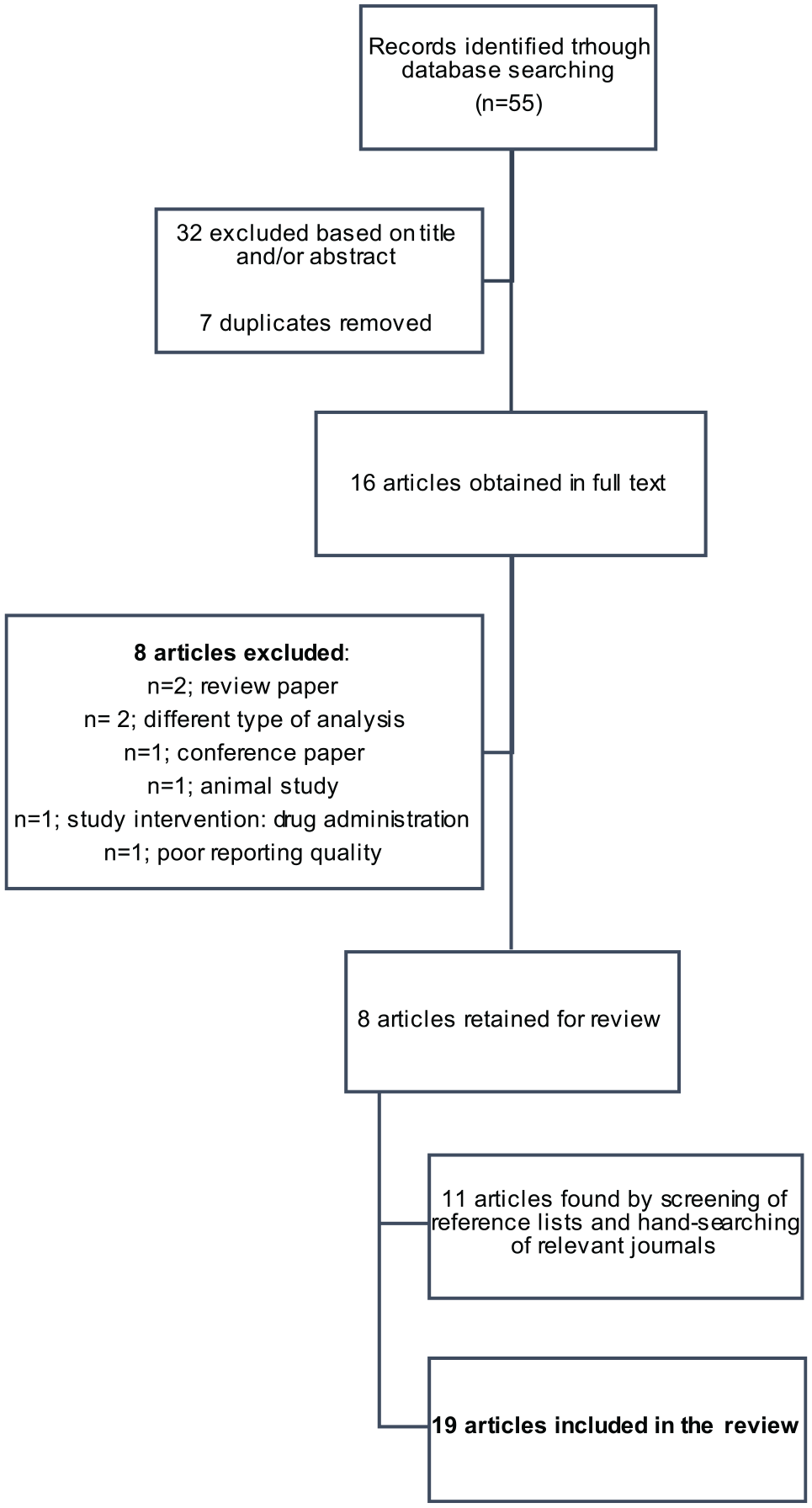
Flow diagram of study selection based on the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement.
Screening of reference lists and hand-searching of relevant journals obtained an additional eleven articles. Consequently, this review is based on data from 19 publications (Figure 1).
The independent reviewers (IL and TH) had discrepancies in selection criteria application in 2 articles. In both cases discrepancies were resolved via consensus discussion. Yang et al 27 collected human spinal cord specimen and the reviewers agreed to exclude this article due to limited comparability to blood/CSF samples. Hayakawa et al 23 collected blood plasma for analysis and the reviewers decided to include this publication because phosphorylated Neurofilament H (pNF-H) levels are expected to be comparable in serum and plasma.
Study Characteristics
Nineteen studies with 1596 patients were included in this systematic review. Thirteen studies collected serum samples for analysis, 1 study used plasma samples, 4 studies investigated CSF-derived biomarkers, and 1 study used both serum and CSF samples.
Results of Individual Studies
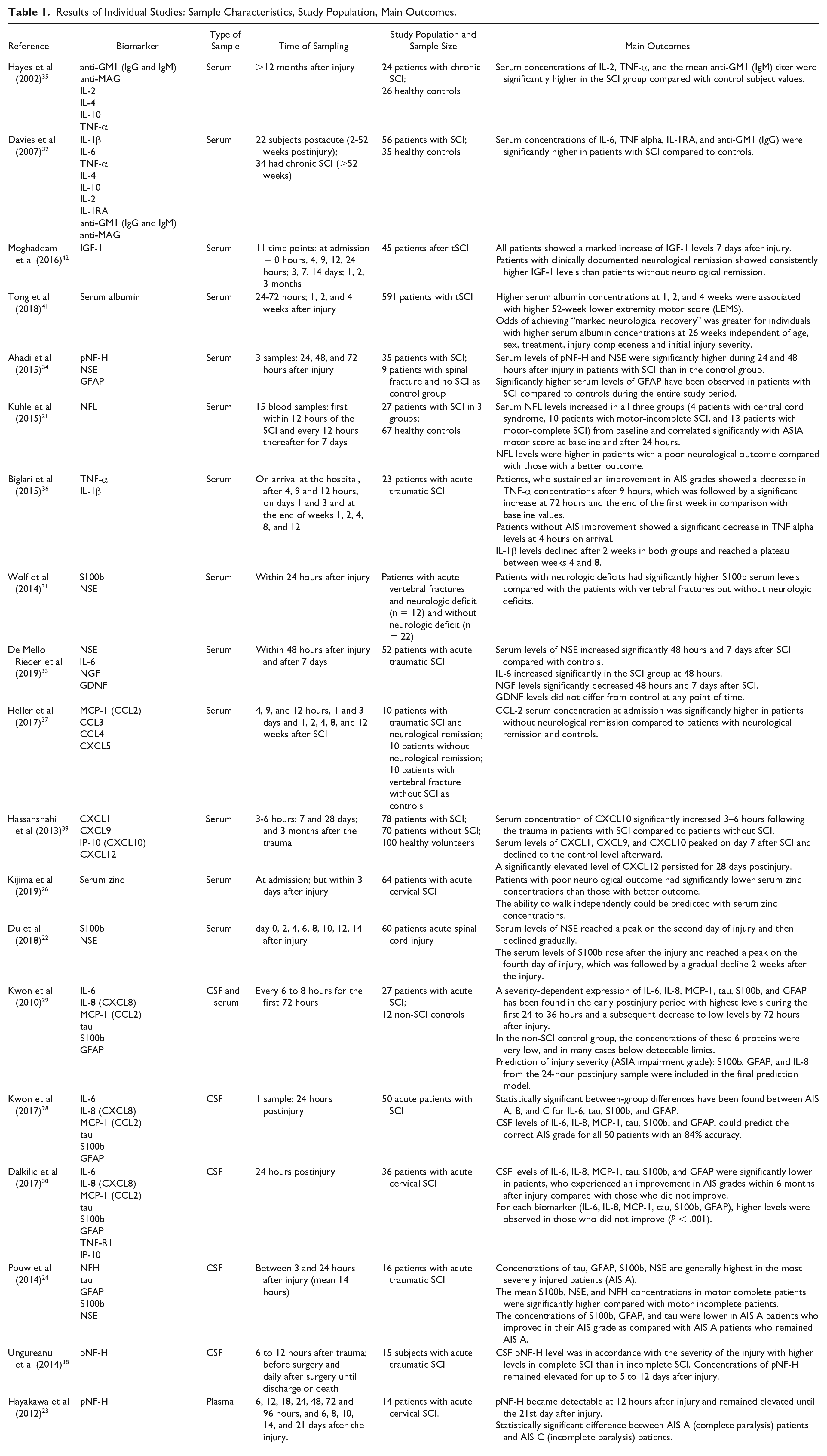
Table 1 illustrates the type of samples, time of sample collection, study population, and main outcomes of the included studies. A critical appraisal of the methodology of the included studies is provided in Supplementary Table 1.
Results of Individual Studies: Sample Characteristics, Study Population, Main Outcomes.
Syntheses of Results
We classified the measurement properties in “diagnostic”, if the studies reported on injury severity or the mere presence of SCI (when the control group consisted of patients without SCI) and “prognostic”, if the included studies reported on neurological improvement by means of AIS grade conversion. Figure 2 provides a vast overview of the time ranges in which the samples have been collected in the included studies and the prognostic and diagnostic value of some biomarkers based on sampling time.
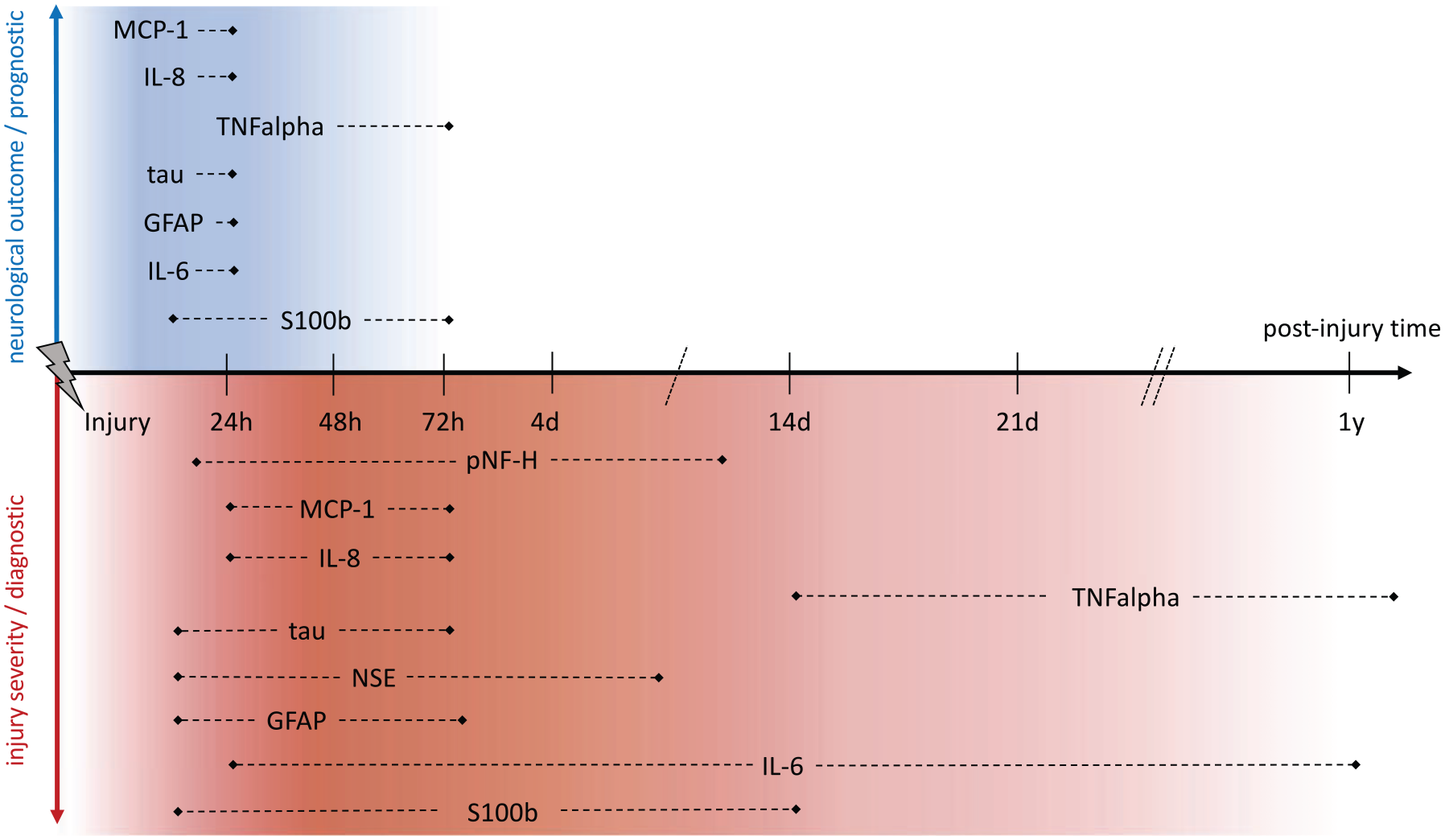
Prognostic and diagnostic value based on post-injury sampling time: The time ranges present the outer ranges of all the screened studies. “Prognostic” refers to a relation between the biomarker expression and neurological recovery. “Diagnostic” refers to an association between the biomarker levels and injury severity. S100b, S100 calcium binding protein B; IL-6, interleukin 6; GFAP, glial fibrillary acidic protein; NSE, neuron-specific enolase; TNFalpha, tumor necrosis factor–α; IL-8, interleukin 8; MCP-1, monocyte chemoattractant protein 1; pNF-H, phosphorylated neurofilament heavy.
S100 Calcium Binding Protein B (S100b)
S100b is a calcium-binding protein localized in the cytoplasm and/or nucleus of cells and is involved in the regulation of cellular processes such as cell cycle progression and differentiation. S100b is predominantly expressed in glial cells and may function in astrocytosis and axonal sprouting. This protein was assessed in 6 studies.22,24,28-31 Elevated concentrations of S100b are thought to be the consequence of cellular death. Since different cell types express this calcium binding protein, systemic increase is of low specificity. 31 Kwon et al 28 reported significant between-group differences of S100b concentrations between AIS (American Spinal Cord Injury Association Impairment Scale) A, B, and C. The S100b concentrations within CSF correlated significantly with a conversion from AIS A to B or C. The CSF levels of S100b were significantly lower in those who improved 1 AIS grade over 6 months compared with those who did not.28,30 In another study, Kwon et al 29 found severity-dependent concentrations of S100b in the postinjury period in CSF as well as in serum with peak concentrations during the first 24 to 36 hours. Additionally, significantly higher mean CSF concentrations of S100b were found in patients with motor complete lesions compared with incomplete lesions. 24 Wolf et al 31 found significantly higher S100b serum levels in patients with SCI compared to patients with vertebral fractures without SCI. In the study of Du et al, 22 serum concentrations of S100b increased following SCI and peaked on the fourth day after the trauma, followed by a gradual decline. Collectively, concentrations of S100b were reported to be increased in complete SCI compared with incomplete lesions. Also, reports demonstrated a positive correlation of S100b concentrations with injury severity and a potential prognostic value with lower concentrations found in patients who improved neurologically.
Interleukin 6 (IL-6)
IL-6 is a pro-inflammatory cytokine actively secreted by different cell types involved in numerous inflammatory pathways. The protein is primarily produced at sites of inflammation and was assessed in 5 studies.28-30,32,33 Davies et al 32 found significantly higher serum concentrations of IL-6 in patients with SCI in the sub-acute injury-phase (2-52 weeks postinjury) compared with healthy controls. In contrast to these findings, another study found increased serum concentrations of IL-6 only within 48 hours but not after 7 days postinjury. 33
Statistically significant lower IL-6 CSF concentrations have been observed 24 hours postinjury in patients who showed neurological remission and a conversion from complete lesions to incomplete lesions correlated negatively with IL-6 concentrations.28,30 Kwon et al 29 observed severity-dependent concentrations of IL-6 within the first 72 hours after injury with higher concentrations in CSF and serum in more severely injured patients. IL-6 is dependent on injury severity with higher levels in AIS A patients. In general, lower concentrations were associated with neurological improvement. However, the detailed temporal pattern of serum IL-6 levels following SCI remains uncertain.
Glial Fibrillary Acidic Protein (GFAP)
GFAP is an astrocyte-specific intermediate filament protein, which is typically elevated in reactive astrocytes during plasticity, neurodegeneration, and after injury, where it is associated with glial scar formation. Passive release during cellular damage is thought to be the origin of elevated concentrations found in patients with SCI as reported by the included 5 studies.24,28-30,34 Significant between-group differences in CSF levels of GFAP were reported at 24 hours postinjury with higher concentrations in more severely injured patients. Also, lower CSF concentrations were observed in patients who improved by 1 AIS grade within 6 months of injury compared with those who did not. 28 These results coincide with the findings by Dalkilic et al, 30 where significantly lower CSF levels of GFAP were found 24 hours after the injury in patients with acute cervical SCI who experienced AIS grade improvement compared to patients without AIS improvement. Lower GFAP concentrations were found in AIS A patients who improved in their AIS grade as compared with patients who remained clinically complete. 24 Serological and CSF GFAP concentrations correlate positively with injury severity in patients with traumatic SCI within the initial 36 hours. 29 Significantly higher GFAP serum concentrations 24 hours after injury were observed in patients with SCI compared to patients with vertebral fractures without SCI. 34 Overall, the expression of GFAP in CSF and serum has been shown to be severity dependent and might serve as prognostic biomarker for AIS grade improvement in patients with acute SCI.
Neuron-Specific Enolase (NSE)
NSE is an intracellular protein found in mature neurons and cells of neuronal origin and is released in the event of cellular damage. Levels of NSE were measured in 5 studies.22,24,31,33,34 Ahadi et al 34 found significantly higher serum concentrations of NSE at 24 and 48 hours after injury in patients with SCI compared to patients with vertebral fractures and no SCI. NSE concentrations decreased after 24 hours postinjury. 34 Pouw et al 24 observed severity-dependent concentrations of NSE in CSF in patients with acute traumatic SCI within 24 hours after injury while highest values were measured in motor complete patients. Also, de Mello Rieder et al 33 observed significantly increased serum concentrations of NSE 48 hours and 7 days postinjury compared with healthy controls. Du et al 22 reported peak NSE serum concentrations on the second day following injury with a gradual decline after 2 weeks. In contrast to these results, Wolf et al 31 found no significant increase in serum concentrations of NSE 24 hours postinjury. Taken together, NSE might be of diagnostic value for SCI severity. None of the studies reported on the potential prognostic value of NSE in terms of neurological recovery.
Tau
Since neurons show high expression levels of tau, an intracellular protein involved in microtubule stabilization, increased extracellular concentrations may serve as indicator for neuronal damage. Concentrations of tau were evaluated in 4 studies.24,28-30 Kwon et al 28 found a significant negative correlation of CSF tau concentrations with AIS grade 24 hours postinjury (higher levels of tau were accompanied by a worse AIS grade). Additionally, CSF levels of tau were significantly lower in patients who experienced an AIS grade conversion.28,30 Additionally, Pouw et al 24 reported lower tau concentrations 24 hours postinjury in AIS A patients improving in their AIS grade with the highest increment of CSF tau concentrations in motor complete patients. This is in line with another study by Kwon et al 29 , who observed increased concentrations of tau in CSF and serum in the early post-injury period (within 30 hours) dependent on injury severity with higher concentrations measured in patients with complete SCI. In general, all included studies reported a positive correlation of CSF tau concentrations with injury severity.24,28-30 Collectively, tau might qualify as prognostic biomarker for neurological improvement after acute SCI.
Tumor Necrosis Factor–α (TNF-α)
The pro-inflammatory cytokine TNF-α is mainly secreted by macrophages and was assessed in 3 studies.32,35,36 Hayes et al 35 reported significantly higher serum concentrations of TNF-α more than 12 months after injury in patients with chronic SCI compared with healthy controls. In the study by Davies et al, 32 serum concentrations of TNF-α were significantly higher in patients with SCI in the subacute injury phase (2-52 weeks postinjury) as well as in patients with chronic SCI (>52 weeks) compared with healthy controls. Serum concentrations of TNF-α decreased more markedly within the initial 72 hours in patients who experienced AIS grade improvement. 36 In summary, TNF-α might serve as a prognostic biomarker for neurological recovery in acute SCI and seems to remain elevated in the chronic stages of SCI compared with the healthy population.
Interleukin 8 (IL-8) and Monocyte Chemoattractant Protein 1 (MCP-1)
IL-8, a chemokine with pro-angiogenic potency, is predominately produced and secreted by endothelial cells and monocytes/macrophages. Concentrations of IL-8 were assessed in 3 studies. MCP-1 is involved in immunoregulatory and inflammatory processes and was assessed in four studies.28-30,37 Kwon et al 28 and Dalkilic et al 30 investigated CSF levels of IL-8 and MCP-1 in patients with acute SCI 24 hours postinjury. Both studies found significantly lower CSF levels of IL-8 and MCP-1 in patients who experienced AIS improvement within 6 months postinjury compared with those who did not.28,30 Kwon et al 29 found severity-dependent concentrations of IL-8 and MCP-1 in serum and CSF samples within 30 hours postinjury. Heller et al 37 also found significantly higher MCP-1 serum concentrations in patients without neurological remission from an initial AIS grade A compared to patients with remission and controls. IL-8 and MCP-1 might serve as suitable biomarkers for injury severity with potential prognostic value for predicting neurological recovery.
Phosphorylated Neurofilament Heavy (pNF-H)
Levels of pNF-H were assessed in 3 studies in patients with acute SCI.23,34,38 Neurofilaments are intermediate filaments in neurons and important parts of the axoskeleton with increased extracellular concentrations serving as indirect maker of neuronal cell death. Ahadi et al 34 found significantly higher serum concentrations of pNF-H at 24 and 48 hours after SCI. Hayakawa et al 23 reported that plasma concentrations of pNF-H became detectable 12 hours after injury and remained elevated until 21 days postinjury in patients with acute cervical SCI. A statistically significant difference was observed in plasma pNF-H levels between AIS A and AIS C patients. 23 Ungureanu et al 38 reported 10 to 100 times higher CSF concentrations of pNF-H in complete SCI compared with incomplete SCI. In summary, pNF-H potentially qualifies as a biomarker for injury severity in the acute stage of SCI.
Interleukin 1 (IL-1) β
The cytokine IL-1β is an important mediator of the inflammatory response. IL-1β is predominately produced by monocytes and concentrations were evaluated in 2 studies.32,36 Davies et al 32 obtained no detectable serum levels of IL-1β from either patients with subacute (2-52 weeks postinjury) or chronic SCI (>52 weeks). Biglari et al 36 reported high fluctuations within the first 12 weeks after traumatic SCI in serum. Based on these conflicting early findings, serological IL-1beta concentration does not seem to qualify as a prognostic biomarker in acute or chronic stages of SCI.
Interleukin 4 (IL-4) and Interleukin 10 (IL-10)
IL-4 and IL-10 and were assessed in 2 studies.32,35 Both cytokines are secreted by leukocytes and are involved in adaptive immune response and shift toward Th2-cell response. The studies found elevated serum concentrations of IL-4 only in a small number of SCI subjects, while no detectable serum levels of IL-10 were obtained from either the healthy control subjects, patients with subacute (2-52 weeks postinjury), and chronic (>52 weeks) SCI. Thus, serum concentrations of IL-4 and IL-10 do not provide diagnostic or prognostic information in subacute and chronic SCI.
Interleukin 2 (IL-2)
IL-2 is predominantly expressed by activated T cells and is a regulator of T- and B-lymphocyte proliferation. IL-2 was evaluated in 2 studies.32,35 Hayes et al 35 reported significantly higher serum concentrations of IL-2 in patients with SCI compared with healthy controls. In contrast, Davies et al 32 found no significant difference between SCI subjects and controls in IL-2 serum concentrations. Both studies investigated patients with subacute and chronic SCI. Their results suggest that IL-2 is not a valid biomarker in subacute and chronic SCI.
Antibodies Against the GM1 Ganglioside Immunoglobulin IgG, IgM (Anti-GM1), and Antibodies Against Myelin-Associated Glycoprotein (anti-MAG)
GM1 is a glycoprotein associated with both the plasma membrane and intracellular organelles. It is involved in many functions in various cell types, particularly in neurons. Myelin-associated glycoprotein is a component of myelin in both central and peripheral nervous system. Anti-GM1 and anti-MAG were assessed in 2 studies on patients with subacute and chronic SCI.32,35 Hayes et al 35 observed a significant increase in serum anti-GM1 IgM-titer in the SCI group compared to controls, while no significant difference in anti-GM1 IgG-titer were observed. Conversely, Davies et al 32 found significantly higher serum concentrations of anti-GM1 IgG-titer in patients with SCI compared with controls. Both studies reported no significant difference between SCI subjects and controls in anti-MAG antibodies.
C-X-C Motif Chemokine Ligand 10 (IP-10)
Concentrations of IP-10 (=CXCL10) were evaluated in 2 studies. It acts as chemokine with various cellular origins.30,39 Hassanshahi et al 39 found significantly increased serum concentration of IP-10 at 3 to 6 hours following the trauma in patients with SCI compared to patients without SCI with a peak 7 days after SCI. IP-10 might qualify as a biomarker that relates to lesions to the spinal cord itself in early injury stages. Further research should analyze if injury severity is related to IP-10 levels.
Further Potential Biomarkers
All remaining biomarkers (NFH, NFL, serum albumin, IGF-1, TNF-R1, NGF, GDNF, IL-1RA, CCL3, CCL4, CXCL5, CXCL1, CXCL9, CXCL12, and serum zinc) were assessed in only 1 of the included studies.
The mean CSF concentrations of neurofilament heavy protein (NFH) was higher in patients with motor complete lesions compared with incomplete lesions. 24
Neurofilament light protein (NFL) serum concentrations were higher in patients with poor outcome (as defined by below median motor score). 21
Tong et al reported odds of achieving “marked neurological recovery” (i.e. at least a 2-grade improvement in Benzel score 40 ) were improved in individuals with higher serum albumin concentrations 26 weeks after injury independent of age, sex, treatment, injury completeness and initial injury severity. 41
Moghaddam et al 42 observed, that individuals with neurological remission showed consistently higher insulin-like growth factor 1 (IGF-1) serum concentrations within 3 months after injury compared to individuals without neurological remission.
Serum concentrations of C-C motif chemokine ligand 4 (CCL4) were significantly higher in patients with neurological remission. 37
Serum concentrations of C-X-C motif chemokine ligands 1, 9, and 12 (CXCL1, CXCL9, and CXCL12) were significantly increased 3 to 6 hours following SCI compared with controls. 39
Lower serum zinc concentrations at admission were related to poor neurological outcome and predicted the ability to walk independently. 26
The biomarkers tumor necrosis factor receptor superfamily member 1A (TNF-R1), nerve growth factor (NGF), glial cell–derived neurotrophic factor (GDNF), interleukin 1 receptor antagonist (IL-1RA), C-C motif chemokine ligand 3 (CCL3), C-X-C motif chemokine ligand 5 (CXCL5) did not yield significant results.30,32,33,37
Technical Methods
Technical procedures, types of samples tubes, clotting times, mode of centrifugation, storage of samples, type of the used assays, along with analytes and detection limits are illustrated in Table 2 for studies on blood-derived biomarkers and in Table 3 for CSF samples.
Technical Procedures for Studies on Blood-Derived Biomarkers.
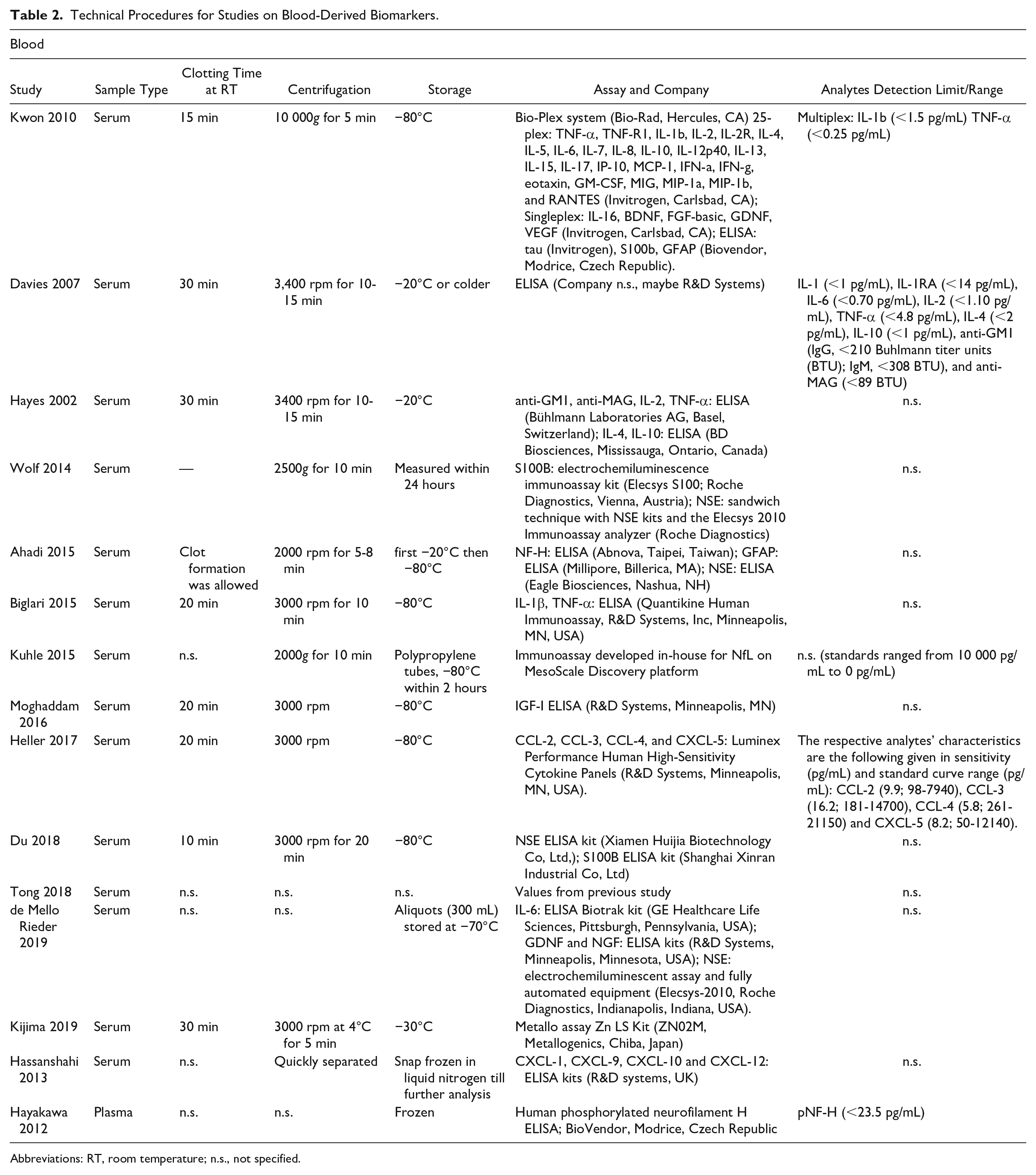
Abbreviations: RT, room temperature; n.s., not specified.
Technical Procedures for Studies on Cerebrospinal Fluid (CSF)–Derived Biomarkers.
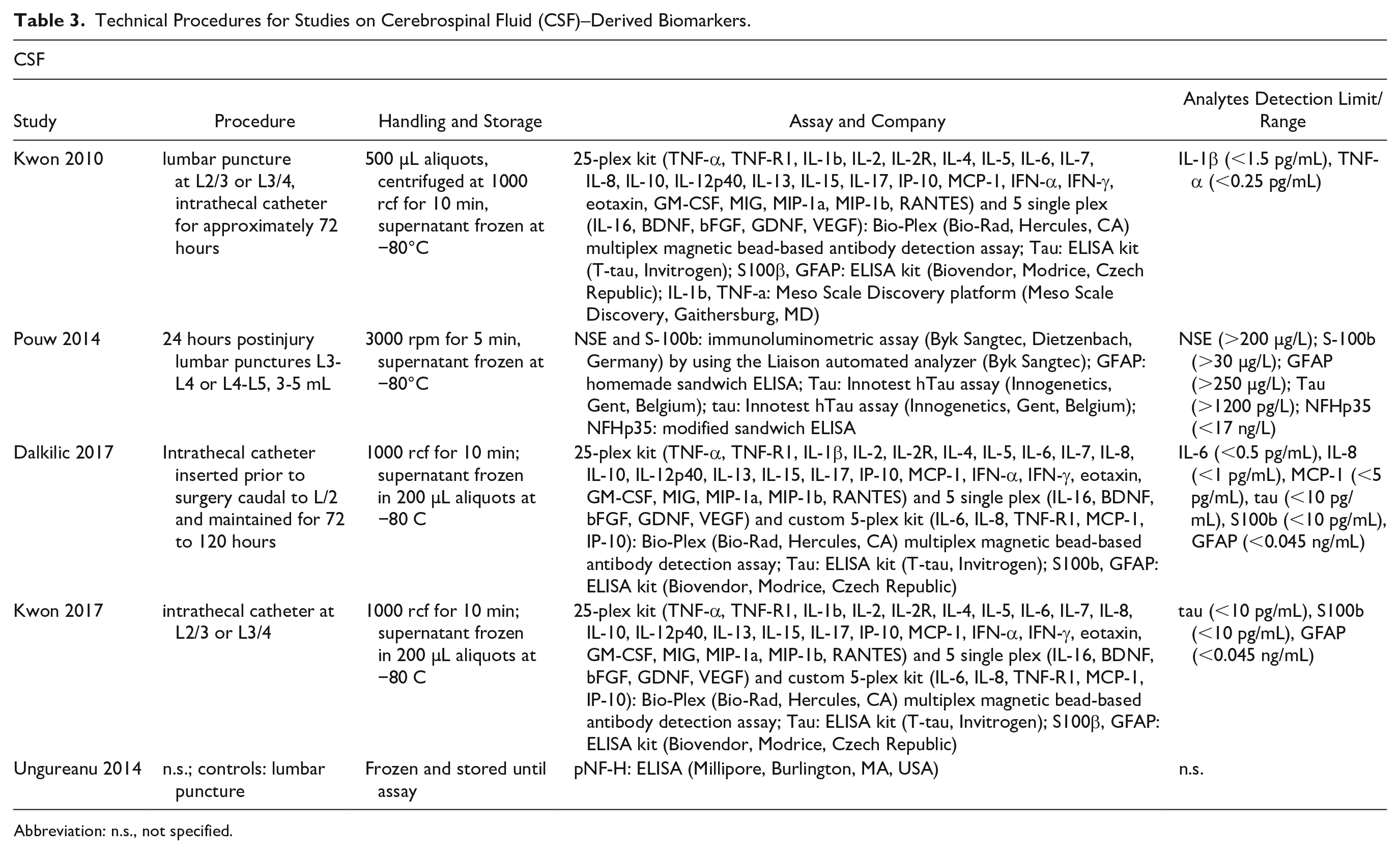
Abbreviation: n.s., not specified.
Nine studies processed the blood samples within 30 minutes after sample collection,22,26,29,31,32,35-37,42 whereas 6 studies did not specify the time between blood sampling and serum separation.21,23,33,34,39,41 Serum should be rapidly separated after sample collection, as blood cells secrete cytokines and thereby altering the sample. 43 For the studies by Ahadi et al, 34 Kuhle et al, 21 Tong et al, 41 and Hayakawa et al, 23 quick separation was not as essential, because the analytes measured in those studies are relatively stable and alterations due to longer contact of serum with the cells is considered unlikely. In contrast, IL-6 measured by de Mello Rieder et al 33 could have indeed changed if contact with whole blood lasted too long, 43 but this group did not specify the time frame.
Another important component in biomarker analysis is the storage temperature for long-term storage. Serum samples are stable for 2 years at −80°C. 44 Afterward, cytokines begin to degrade.43-45 At higher temperatures the degradation starts earlier. This issue could have affected the measurements in the studies by Davies et al 32 and Hayes et al. 35 All other included studies claimed samples storage at −80°C (in 1 case, –70°C), measured the samples on the same day 31 or had more stable analytes than cytokines. None of the articles stated storage duration.
Detection of biomarkers was assessed by magnetic bead–based multiplex system in 2 studies.29,37 Kwon et al 28 used an additional normal sandwich ELISA. Wolf et al 31 made use of an electrochemiluminescence immunoassay and Kuhle et al 21 measured biomarkers with an in house developed immunoassay on Meso Scale Discovery platform. The majority of the included studies used ELISAs.
Discussion
This systematic review focused on serological and CSF-derived biomarkers. Over the past decade, research in the field of spinal cord tissue regeneration has focused on the immune response following SCI.3,14,46 Previously published data demonstrated some prognostic value of blood- and CSF-derived biomarkers for neurological outcome in patients with SCI.17,19 The level of some proteins (IL-6, IL-8, MCP-1, tau, S100b, and GFAP) is associated with neurological recovery and significantly lower serum and CSF levels were found in patients with AIS grade improvement compared to those without improvement.24,28,37 For the biomarkers pNF-H and NSE only diagnostic value was reported by the included studies. Concentrations of pNF-H are elevated in accordance with injury severity23,34,38 and levels of tau, GFAP, S100b, NSE are generally highest in the most severely injured patients.22,24 Protein levels were comparable between males and females, and not associated with age,24,39 which gives rise for a more objective assessment despite the heterogeneity in patients with SCI. However, certain issues with biomarkers in general, and particular in SCI research remain. We observed heterogeneities in study design, including timing of measurements, inclusion criteria, measurement methods with different reference values and ranges, and outcome measures among others. These differences complicate the comparability of available studies. Also, conflicting results preclude conclusions to be drawn for each biomarker.
In general, biomarkers have been investigated for numerous medical conditions. Preexisting conditions in SCI, especially inflammatory and malignant diseases, were previously shown to be associated with altered concentrations of various biomarkers.47-50 However, during the clinical routine in acute care of SCI, detailed medical history is not available in the majority of cases. In addition, concomitant injuries, especially traumatic brain injury (TBI), potentially interfere with systemic biomarker concentrations.51,52
Throughout the long-term clinical course of SCI recovery, patients are often affected by muscle dystrophy, infections, organ dysfunction, and immunological dysregulation, which further complicates biomarker research. 53 As such, these conditions need to be considered carefully in future mid- to long-term studies. Also, iatrogenic disturbance of biomarker concentrations represents important potential confounders. The ideal biomarker should be specific to lesions of the spinal cord, and independent of confounding factors.
A general issue in biomarker research remains the question as to whether alteration of biomarker concentrations is causative or the consequence of disease progression and poor prognosis, respectively. The inflammatory response was recently linked to both detrimental and beneficial properties in the secondary injury cascade. Timing of certain inflammatory pathways plays a key role in promoting either attenuation or promotion of SCI lesion progression.54-56 This phenomenon further complicates biomarker investigation and interpretation. Also, systemic concentrations of cytokines are highly sensitive to infections and immune dysfunction 57 making this group of biomarkers vulnerable to individual differences.
In the field of TBI research, several neural injury biomarkers (S100b, GFAP, pNF-H, NSE) have already been discovered and are used for diagnosis, intervention, and prognosis.58,59 Furthermore, radiologic markers were recently reported to show promising prognostic capacity.2,30 A combination of both, biological and imaging markers might increase sensitivity and serve as possible diagnostic tool in future SCI research. With improvement of measurement methods and the ability to assess numerous biomarkers simultaneously, further improvement in biomarker research is expected in upcoming years.
SCI research is in need of surrogate parameters to monitor potential therapeutics in clinical studies and to serve as potential pharmacological targets. Since SCI shows a great heterogenicity in clinical presentation, disease progression, and complications, large studies with comparable patient collectives are indispensable. Ideal surrogate parameters would allow to monitor important milestones of SCI such as glial scar formation, characteristics of local and systemic immunological response, and blood-brain barrier integrity. Further studies should not only examine the relationship of biomarker concentrations to injury severity but also examine temporal changes, and the relationship to spontaneous recovery. To evaluate if a marker is specific to lesions of the spinal cord itself and not just a marker of major trauma, future studies should also compare biomarker concentrations of healthy individuals with patients who sustained SCI.
Limitations
This review included studies reporting about biomarkers from either blood serum/plasma or CSF. Hematological parameters were not included for reasons of comparability, due to effects of storage duration and coagulation. It also should be acknowledged that the comparability between biomarker expression in CSF and serum or plasma is not entirely clear. Cytokine levels are reported to be higher in the CSF than in serum.2,29 Also, the results are not representative for the whole SCI population, since only patients with traumatic SCI were included. Animal trials were not included in the review, because it is considered unlikely that an animal model will be able to replicate the complexity of human injury. 19
Conclusion
The expression patterns of several biomarkers are highly dependent on postinjury time of sample collection. On the basis of the synthesis of the results, some biomarkers (S100b, IL-6, GFAP, NSE, tau, TNF-α, IL-8, MCP-1, pNF-H, and IP-10) showed promising capacity for indicating SCI severity and prediction of neurological recovery. An improved understanding of SCI-associated biomarkers may potentially help to further elucidate secondary injury mechanisms, discover novel therapeutic targets, and inform future SCI research. This understanding will hopefully guide future clinical decision making. We suggest definition of uniform criteria for study design in biomarker SCI research to improve comparability and promote translation to clinical routine.
Supplemental Material
NNR899920_Supplemental_Material – Supplemental material for Biomarkers in Traumatic Spinal Cord Injury—Technical and Clinical Considerations: A Systematic Review
Supplemental material, NNR899920_Supplemental_Material for Biomarkers in Traumatic Spinal Cord Injury—Technical and Clinical Considerations: A Systematic Review by Iris Leister, Thomas Haider, Georg Mattiassich, John L. K. Kramer, Lukas D. Linde, Adnan Pajalic, Lukas Grassner, Barbara Altendorfer, Herbert Resch, Stephanie Aschauer-Wallner and Ludwig Aigner in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by a research grant from the Paracelsus Medical University Research Fund (PMU-FFF; grant number: S-19/03/015-LEI) and the authors would like to express their sincere appreciation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
