Abstract
Background
Motor performance is a complex process controlled in task-specific spatial frames of reference (FRs). Movements can be made within the framework of the body (egocentric FR) or external space (exocentric FR). People with stroke have impaired reaching, which may be related to deficits in movement production in different FRs.
Objective
To characterize rapid motor responses to changes in the number of degrees of freedom for movements made in different FRs and their relationship with sensorimotor and cognitive impairment in individuals with mild chronic stroke.
Methods
Healthy and poststroke individuals moved their hand along the contralateral forearm (egocentric task) and between targets in the peripersonal space (exocentric task) without vision while flexing the trunk. Trunk movement was blocked in randomized trials.
Results
For the egocentric task, controls produced the same endpoint trajectories in both conditions (free- and blocked-trunk) by preserving similar shoulder-elbow interjoint coordination (IJC). However, endpoint trajectories were dissimilar because of altered IJC in stroke. For the exocentric task, controls produced the same endpoint trajectories when the trunk was free or blocked by rapidly changing the IJC, whereas this was not the case in stroke. Deficits in exocentric movement after stroke were related to cognitive but not sensorimotor impairment.
Conclusions
Individuals with mild stroke have deficits rapidly responding to changing conditions for complex reaching tasks. This may be related to cognitive deficits and limitations in the regulation of tonic stretch reflex thresholds. Such deficits should be considered in rehabilitation programs encouraging the reintegration of the affected arm into activities of daily living.
Introduction
Reaching movements require perceptual-motor transformations depending on the spatial frame of reference (FR) in which they are performed.1
-6 A FR or system of coordinates consists of elements, the positions of which are defined by their distance from a specific point called the origin or referent point. The brain can use different FRs to accomplish motor actions. For example, reaching movements can be directed to a person’s body in an
Arm trajectories and interjoint coordination (IJC) differ for reaches made in egocentric and exocentric FRs. 7 Healthy individuals have no difficulty rapidly changing reaching patterns in different FRs, practically without learning (ie, 1-trial adaptation). 8 This behavior illustrates the principle of motor equivalence 9 : the ability to reach the same motor goal using different combinations of joint rotations, often referred to as kinematic redundancy.10,11 This type of rapid reaction, critical for everyday life, is related to sensory feedback and the cognitive ability to find motor-equivalent solutions through motor problem-solving.5,8,12 -15
Egocentric and exocentric reaches involve partially overlapping but different neural structures. The parietal cortex constructs multiple spatial FRs 5 and strongly interacts with the frontal cortex (premotor cortex, supplementary eye fields), which encodes object locations in different spatial FRs. 16 Egocentric coding is associated with the parietal and frontal cortices, whereas exocentric coding is associated with the temporal cortex.17,18 Damage to the parietal cortex and associated areas, such as that occurring after stroke, may lead to impairments in producing movements in both egocentric and exocentric FRs, 19 but rapid motor responsiveness in different FRs has not been investigated.
Upper limb (UL) motor impairment is a common and challenging sensorimotor deficit after stroke. 20 Compared with healthy individuals, movements of the affected UL after stroke are generally slower, more variable, more spatially and temporally segmented, and characterized by abnormal patterns of muscle activation and IJC, which may be related to deficits in the regulation of tonic stretch reflex thresholds.21,22 Stroke research has mainly focused on UL reaching in exocentric FRs. For example, in trunk-assisted reaching to beyond-reach exocentric targets, when trunk movement was suddenly arrested, healthy individuals preserved the hand trajectory and reaching accuracy by rapidly modifying shoulder and elbow movements. 23 However, those with stroke had difficulty modifying arm joint movements in response to trunk arrest. Only one study assessed differences in egocentric and exocentric movements in poststroke individuals during a finger-to-nose test. 7 Individuals with stroke had impaired IJC and used more trunk compensation for both egocentric and exocentric pointing phases compared with controls, 7 but the causes of movement deficits in different FRs were not elucidated.
Mechanisms underlying motor deficits after stroke may be better understood using perturbation methods compared with movement description alone. 24 Our objective was to identify deficits in producing pointing movements in egocentric and exocentric FRs in individuals with stroke. We hypothesized that individuals with stroke, unlike healthy controls, would have impairments in rapidly modifying shoulder-elbow IJC to reproduce similar reaching trajectories when trunk movements were present or blocked, in both egocentric and exocentric FRs. Because this behavior depends on sensory information and problem-solving, we also hypothesized that deficits in rapid response ability would be related to levels of UL sensorimotor and cognitive impairment. Preliminary results have appeared in abstract form. 25
Methods
Participants
Poststroke patients were included if they had a unilateral ischemic/hemorrhagic stroke in either hemisphere affecting cortical/subcortical areas at least 6 months earlier, had a score of >21/30 on the Montreal Cognitive Assessment (MoCa; α = .83, test-retest = 0.92), 26 and ~30° voluntary elbow movement in each direction (3-7 Chedoke-McMaster Arm Scale). 27 Those with severe biceps spasticity (>2/4 Modified Ashworth Scale), 28 unilateral neglect (line bisection, cancellation), apraxia (Boston Praxis Test), 29 or ataxia (finger-to-nose) 7 were excluded. Patients were excluded if they had arm pain, uncorrected vision, or other neurological/musculoskeletal problems affecting UL movement.
Eligible participants who agreed to participate signed consent forms approved by the Ethics Committee of the Centre for Interdisciplinary Research in Rehabilitation. They participated in 1 clinical and 1 experimental session.
Clinical Evaluation
For all individuals, handedness was determined using the Edinburgh Handedness Inventory. 30 UL impairment in stroke participants was assessed with the 66-point Fugl-Meyer Upper Limb Assessment (FMA-UL 31 ; interrater = 0.99, 32 intrarater = 0.95), 33 tactile (4 points), and proprioception (8-point) scales. 31 Cognition was assessed with the 30-point MoCa 26 Scale, and visual perceptual deficits were assessed with the Motor-Free Visual Perception Test 34 (α = .80; test-retest = 0.77-0.83) 34 on a 36-point scale, where <32 points denotes visual acuity loss. 35
Procedure
Participants reached without vision in egocentric (task 1) and exocentric (task 2) FRs in a randomized order according to a previously validated methodology. 8 Participants sat in an armless chair and wore a harness with an electromagnet attached to the back (Figure 1). When the trunk contacted the chairback, activation of the electromagnet prevented trunk movement. Participants with stroke used their more-affected arm and healthy participants used their nondominant arm to minimize the comparative advantage of comparisons with their dominant arm. 36 Participants practiced each task ~5 times with vision and feedback until performance stabilized prior to recording.
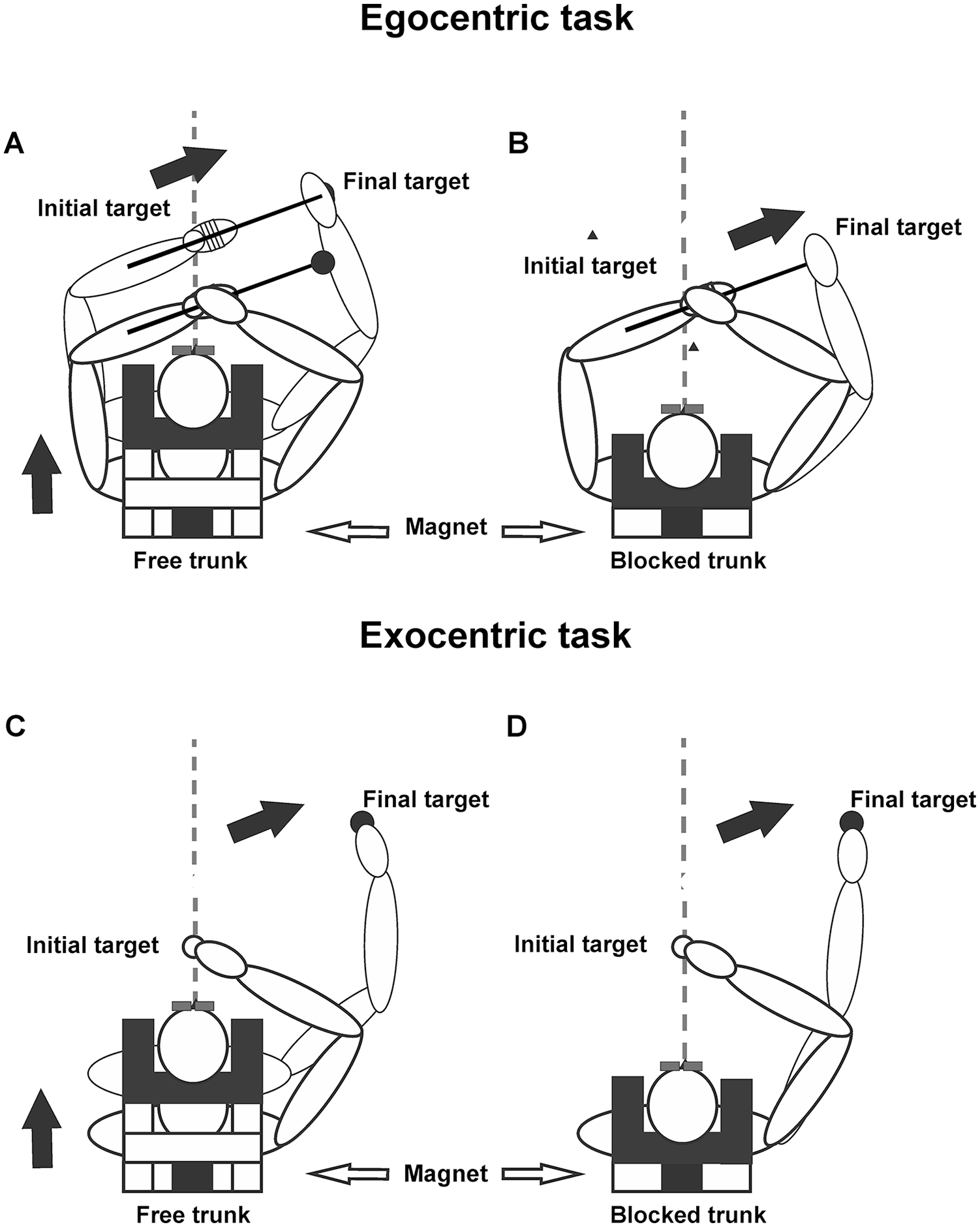
Experimental setup: The participant sat on an armless chair and wore a harness with an electromagnet plate that could be locked to an electromagnet attached to the chair back to prevent trunk flexion. Participants performed reaching along the contralateral arm in the egocentric condition (A, B), or between 2 targets in the exocentric (C, D) condition, while leaning the trunk forward with eyes closed. (A, C) When the electromagnet was not activated, the participant leaned the trunk forward while reaching: free-trunk condition. (B, D) When the electromagnet was activated, only arm movement could occur: blocked-trunk condition. Modified from Ghafouri et al. 8
For task 1, a 30-cm rod was attached to the contralateral arm from the midforearm in line with and extending beyond the forearm (Figures 1A and 1B). The arm was held in 90° elbow flexion in the horizontal plane in front of the body. A firm foam block under the contralateral axilla maintained the same relative arm position with respect to the trunk throughout the experiment. Participants held their ipsilateral index ~1 cm above the rod at the midforearm level and moved their hand ipsilaterally in the direction of, but not touching the rod/forearm in the horizontal plane with eyes closed, thus avoiding haptic and visual feedback. The movement was made within comfortable arm reach (ie, could be accomplished by arm movement alone). Participants were instructed to simultaneously lean their trunk ~20° sagittally with the lateral arm movement. We sought to determine how participants rapidly changed their arm IJC when moving the arm while leaning the trunk. This is similar to leaning forward to listen to someone while reaching for a cup placed within arm’s reach. The motor system naturally alters the UL IJC to accommodate for the additional (unnecessary) trunk DF. 37 A computer-generated tone signaled the simultaneous start of arm and trunk movements. A total of 50 trials were recorded. In 70% of randomly chosen trials, the electromagnet was unlocked (free-trunk trials, n = 35), whereas in the remaining 30% (15 trials), trunk movement was arrested by the electromagnet (blocked-trunk trials) simultaneously with arm movement onset. The free-trunk/blocked-trunk ratio was used to avoid anticipating the upcoming condition. 38 Locking or unlocking the electromagnet was soundless, so participants could not identify the trunk condition before trial onset. Participants were instructed to move at a comfortable speed and make similar arm and trunk movements in each trial whether or not trunk movement was blocked.
Task 2 consisted of arm movement in an exocentric FR (Figures 1C and 1D). Two fixed targets (1-cm3 cubes) attached to adjustable rods were placed 30 cm apart in the same direction and horizontal orientation as for task 1 but in external space. Participants made arm movements without vision from the initial to final targets while flexing the trunk, with the same number and type of trials (free, blocked) as in task 1.
Data Recording and Analysis
A 2-Certus bar Optotrak Motion Analysis System (Northern Digital, Waterloo, 120 Hz, 3-5 s per trial) recorded positions of 6 active LED markers placed on the ipsilateral index fingertip, ulnar styloid process, lateral humeral epicondyle, anterolateral acromions, and midsternum. Data were analyzed separately for each task (egocentric, exocentric) and group (healthy, stroke) and compared between conditions (free trunk, blocked trunk).
Mean (±SD) endpoint trajectories were computed in the egocentric FR that rotated with the trunk or the exocentric, motionless FR related to the room. For the egocentric FR, the reference frame origin was associated with the midpoint between the ipsilateral and contralateral acromial and sternal markers. For the exocentric FR, endpoint trajectories were determined from absolute
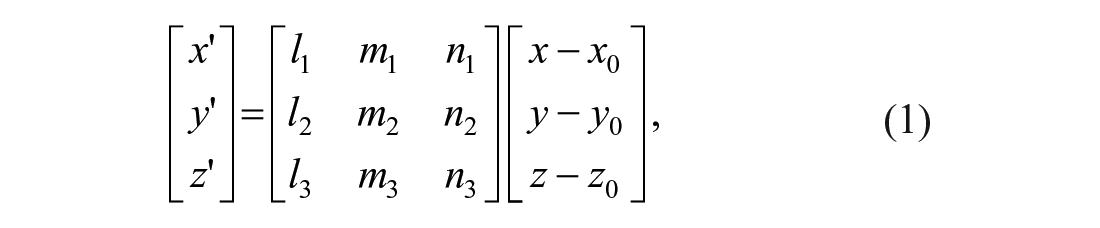
where (
Endpoint trajectories, shoulder and elbow joint ranges, and shoulder-elbow IJC for each task were analyzed. Endpoint trajectories were characterized by movement time, mean tangential velocity, trajectory straightness, and trajectory similarity. Raw
Similarity of averaged trajectories in free- and blocked-trunk conditions for each task and participant was evaluated by comparing slopes and times of divergence (divergence point [DP]) of the 2 trajectories. To determine the degree of trajectory overlap, the slope of the middle third of each trajectory was approximated with a straight line based on best-fit criteria, separately by condition, and the distance between lines was determined. DP times for averaged trajectories were found using
Arm movement was characterized by shoulder and elbow ranges and IJC between joint pairs: shoulder-flexion/elbow-extension and shoulder-horizontal abduction/elbow-extension. Joint ranges were computed as differences between movement onset and offset and measured in degrees. Elbow-flexion/extension angle was calculated from vectors formed between the ipsilateral ulnar styloid process and lateral epicondyle and the lateral epicondyle and the acromion where 0° represents full elbow extension. Shoulder-flexion/extension in the vertical plane was calculated using vectors formed between the ipsilateral acromion and lateral epicondyle markers and the vertical line through the acromial marker, where 0° was the position of the arm alongside the body. Shoulder-horizontal abduction/adduction was calculated from the horizontal vector between the 2 acromial markers and the vector between the ipsilateral acromial and lateral epicondyle markers, where 90° was the position of the arm held horizontally in line with the 2 acromial markers, and adduction was horizontal movement of the shoulder toward the contralateral side. Trunk pitch angle was defined by the rotation of the plane determined by both shoulder markers and the sternal marker around the horizontal axis. For IJC between shoulder and elbow angles, 45° represented an equal contribution of shoulder-flexion or abduction and elbow-extension to the movement. Values <45° indicated greater elbow motion. DPs of each angle-angle IJC were computed as above for endpoint trajectories. Data analysis was done using custom MATLAB (v 2017, MathWorks, USA) software.
Statistical Analysis
Normality of distributions and homogeneity of variances were verified using Kolmogorov-Smirnov and Levene tests, respectively. 2 × 2 repeated measures ANOVAs or Wilcoxon signed-rank tests determined differences in movement variables, trajectory, and IJC slopes between conditions (free vs blocked) and groups (healthy, stroke). For between-group comparisons of trajectory and IJC DPs, independent
Results
Participants
A total of 14 poststroke patients were recruited from January to December 2018. Two patients were excluded because of noncompliance with inclusion criteria. In all, 12 poststroke patients and 13 older adults of similar age participated (Table 1). Stroke participants had mild hemiparesis and good UL tactile sensation/proprioception, except for 2 participants who had impaired elbow proprioception.
Demographic Data of Both Groups and Clinical Scores for Participants With Stroke (mean ± SD). a
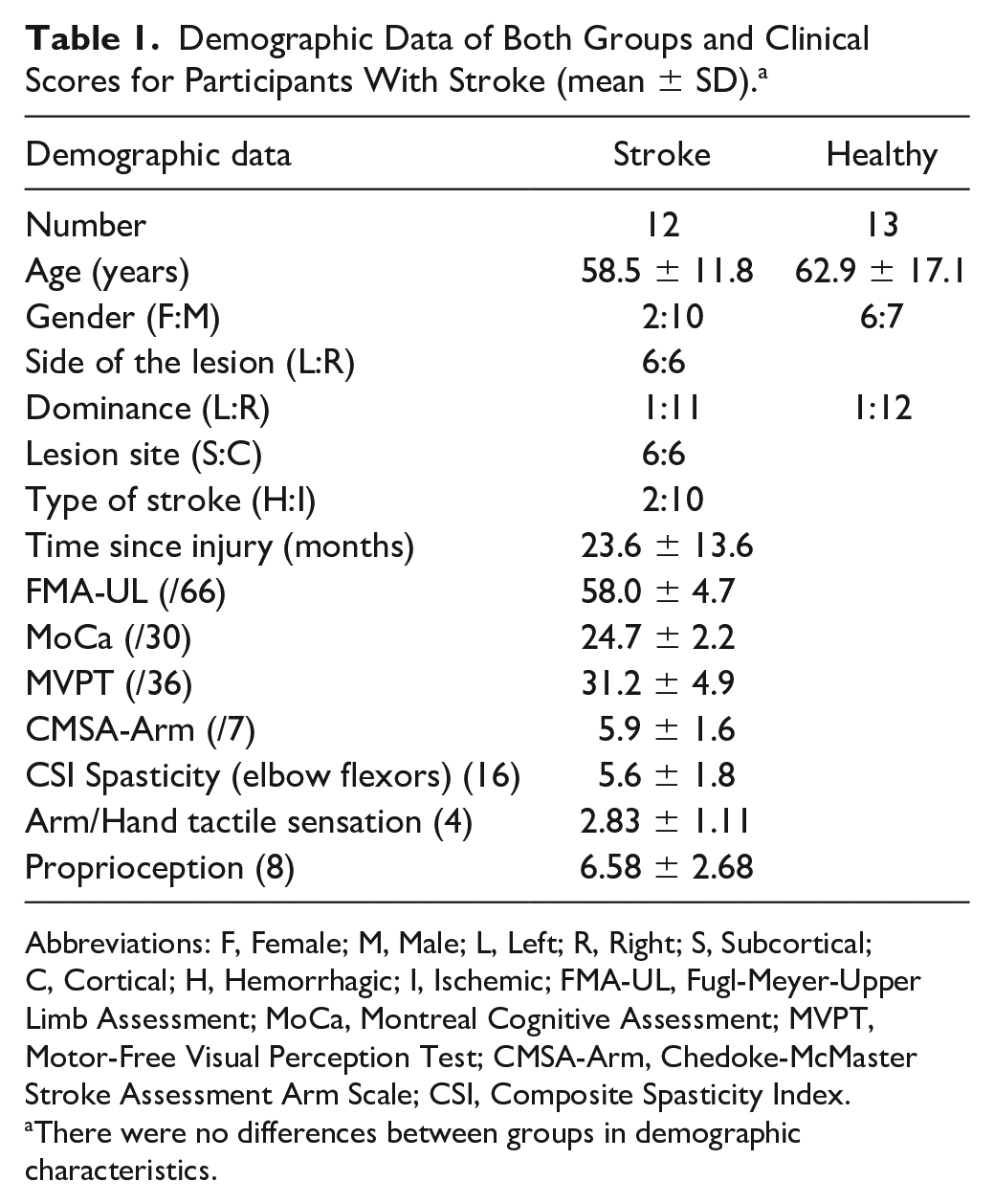
Abbreviations: F, Female; M, Male; L, Left; R, Right; S, Subcortical; C, Cortical; H, Hemorrhagic; I, Ischemic; FMA-UL, Fugl-Meyer-Upper Limb Assessment; MoCa, Montreal Cognitive Assessment; MVPT, Motor-Free Visual Perception Test; CMSA-Arm, Chedoke-McMaster Stroke Assessment Arm Scale; CSI, Composite Spasticity Index.
There were no differences between groups in demographic characteristics.
Healthy Participants
In healthy participants, trunk movement for both tasks in the free-trunk condition was effectively blocked by the electromagnet by ~90% (~20° to <5°; Table 2). For both tasks, endpoint trajectories for free- and blocked-trunk conditions were similar in each FR (Figures 2A and 3A) such that DPs occurred late in the reach. IJC patterns were similar for both conditions for the egocentric task (Figures 2B and 2C) but differed between trunk conditions for the exocentric task (Figures 3B and 3C).
Motor Performance Variables and Joint Ranges of Motion (Mean ± SD).
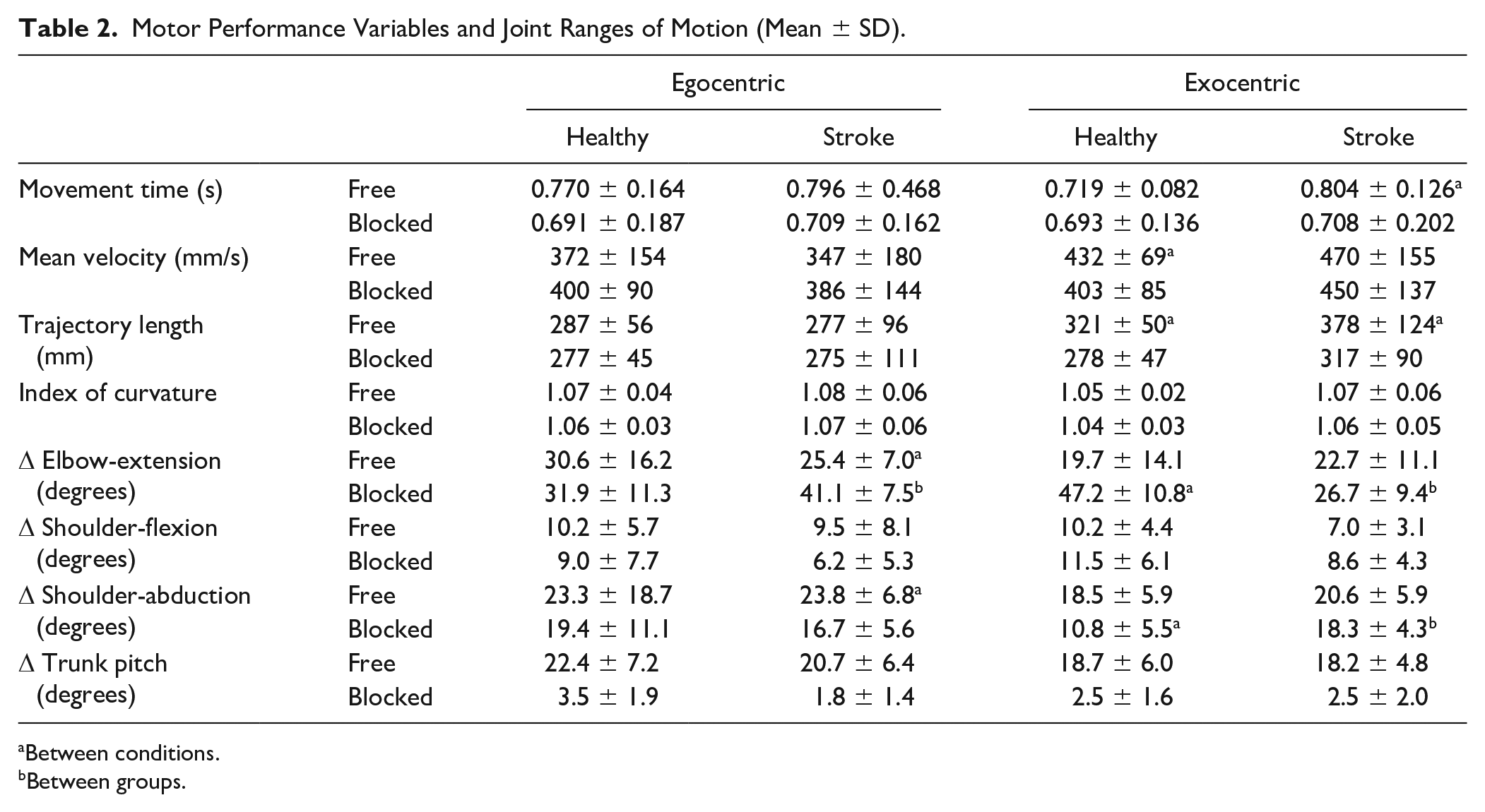
Between conditions.
Between groups.
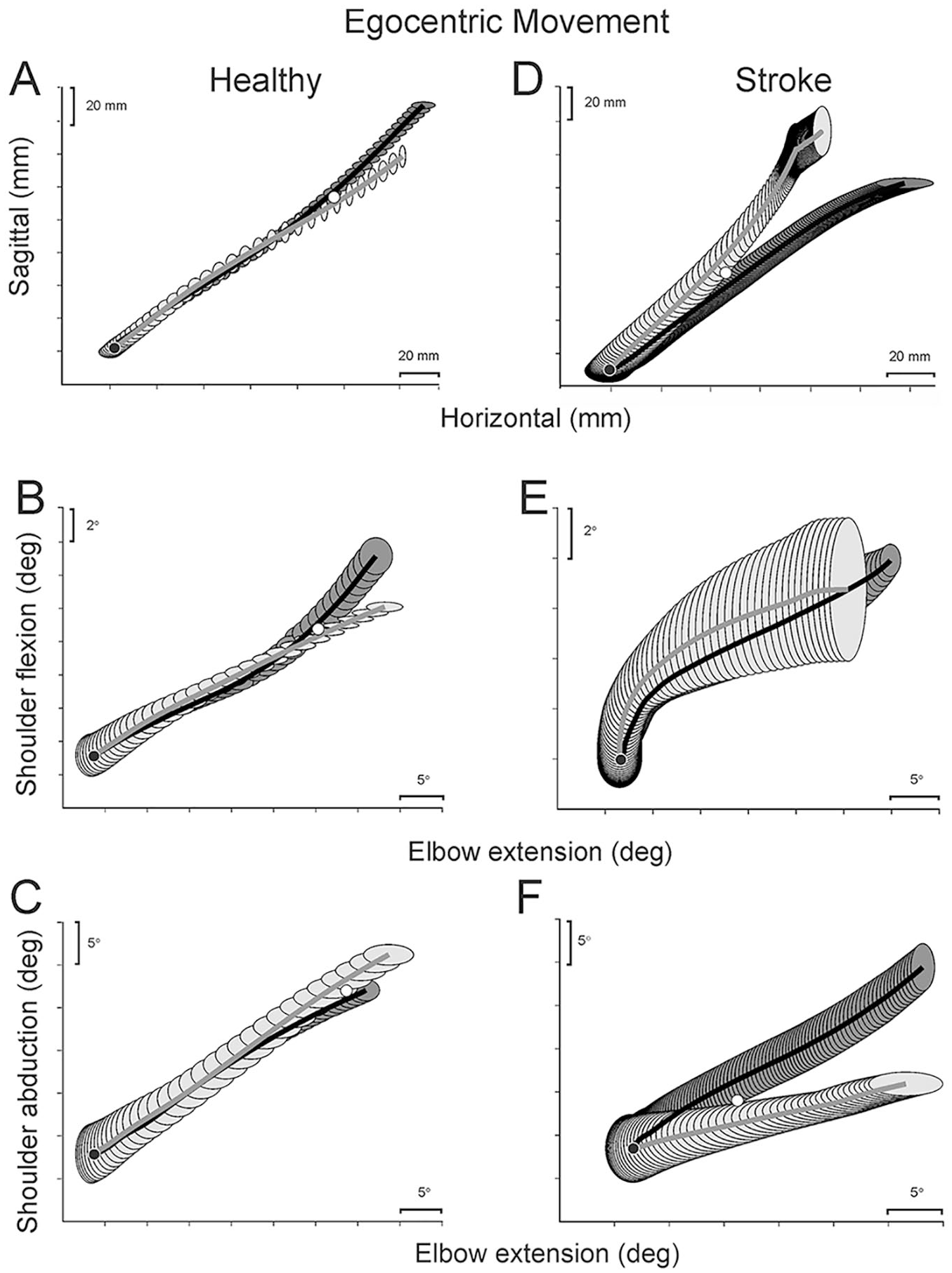
Mean trajectory (A, D) and shoulder-elbow interjoint coordination traces (B, C, E, F) of a representative healthy (A-C) and stroke (D-F) participant performing the egocentric task. Ellipses represent standard error values. Ellipse spacing represents time, where more tightly spaced ellipses indicate slower movements. Free-trunk traces are shown in black, and blocked-trunk traces are shown in gray. Black circles: initial positions; white circles: divergence points.
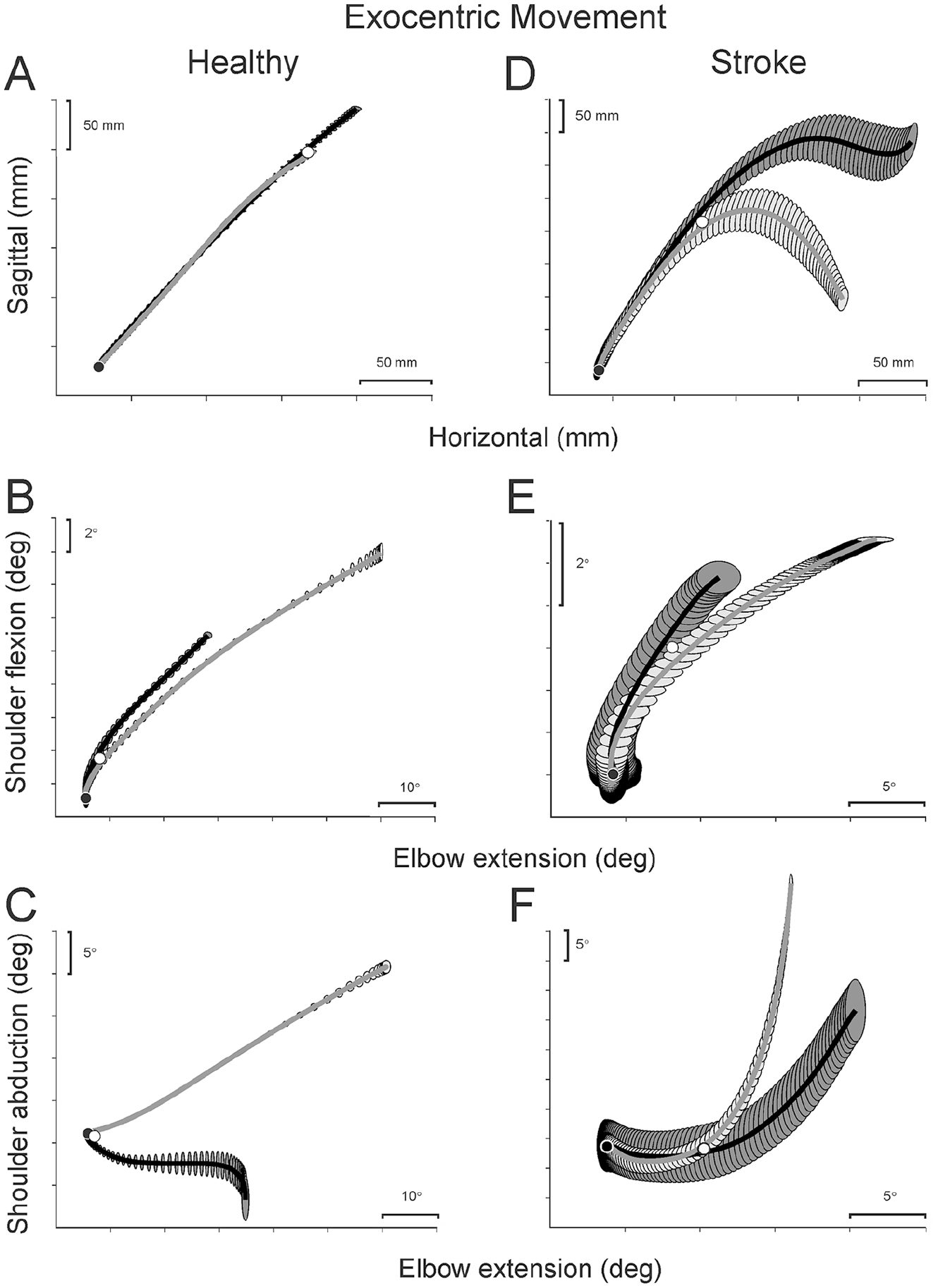
Mean trajectory (A, D) and shoulder-elbow interjoint coordination traces (B, C, E, F) of a representative healthy (A-C) and stroke (D-F) participant performing the exocentric task. Ellipses represent standard error values. Ellipse spacing represents time, where more tightly spaced ellipses indicate slower movements. Free-trunk traces are shown in black and blocked-trunk traces are shown in gray. Black circles: initial positions; white circles: divergence points.
For the group, in the egocentric task, movement speed, trajectory length and straightness, shoulder and elbow ranges of motion, and IJC in the blocked- and free-trunk conditions were similar (Table 2; Figure 4A). However, for the exocentric task, movement was slower (
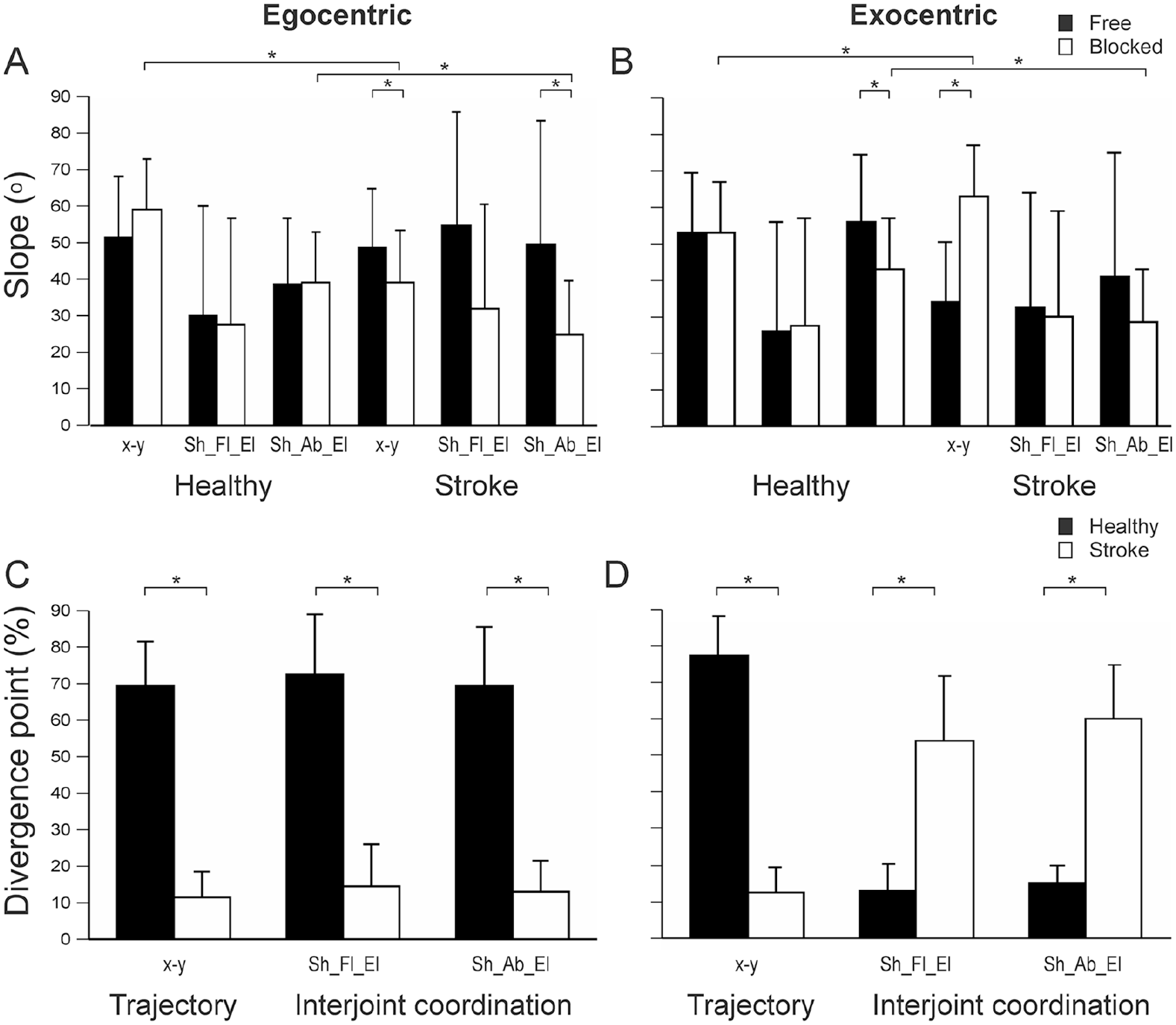
Slopes (A, B) and divergence points (C, D) of trajectories and shoulder-elbow interjoint coordination. (A, B): Mean (SD) values are shown for free (black bars) and blocked (white bars) conditions. (C, D): Divergence points for each pair of trajectories and interjoint coordination traces are shown for healthy (black bars) and stroke (white bars) groups. Statistically significant differences are shown with asterisks (*).
There were no differences in trajectory slopes between conditions for either task (Figures 4A and 4B), with DPs occurring late in the movement (egocentric: 69.9%; exocentric: 77.4%; Figures 4C and 4D). For the exocentric task, shoulder-abduction/elbow-flexion IJC slopes were lower when the trunk was blocked (43.3° ± 15.4°) compared with the free condition (56.0° ± 23.9°,
Participants With Stroke
As in controls, the electromagnet effectively blocked trunk movement by ~90% (Table 2). In contrast to controls, movement trajectories made with and without trunk displacement did not coincide for either task (Figures 2D and 3D). IJC patterns of shoulder-elbow joint movement combinations differed from those of controls (Figures 2 and 3E and 3F).
For the group, movement speed, trajectory length, and straightness were similar between conditions for the egocentric task. However, for the exocentric task, whereas stroke participants preserved trajectory straightness in both conditions, movement time was longer by 0.96 s (
For the egocentric task, in the blocked-trunk condition, elbow extension was greater by 15.7° (
For both tasks, trajectory slopes differed between conditions. Trajectory slopes were 9.2° lower in the blocked- compared with the free-trunk condition for the egocentric task (
Trajectory DPs occurred early in the reach for both tasks (egocentric: 11.9%; exocentric: 12.3%). IJC patterns differed early in the reach for the egocentric task and in midreach for the exocentric task (IJC DP: 54.0%-60.0%). Data from 7 patients with MoCA scores <26 and from 2 patients with partial elbow proprioceptive impairment did not significantly differ from group means for any primary or secondary measure.
Comparisons Between Groups
For the egocentric task, the stroke group used ~9.2° more elbow extension in the blocked-trunk condition compared with controls (
For the exocentric task, stroke participants used less elbow extension (by ~20.5°;
Relationships With Clinical Scores
There were no differences in primary outcome measures between patients with left- and right-sided stroke. For the exocentric task, better cognition (MoCa) was related to later trajectory DPs (ρ = 0.825;
Discussion
We characterized the ability of healthy older adults and individuals with chronic stroke to reproduce the same (invariant) hand trajectories during reaches made in internal (egocentric) and external (exocentric) space. Arm movement in the same direction as the participant’s contralateral forearm was produced within an egocentric FR. Because the arm moved with the trunk, the same DFs were used whether the trunk was free or blocked. When the target was located in peripersonal space, reaching was produced in an exocentric FR, and the combination of DFs changed with different trunk conditions. The perturbation technique allowed us to identify deficits in the ability to produce dynamically stable movements involving multiple DFs by creating task-relevant IJC or synergies responsible for the principle of motor equivalence 9 in participants with mild chronic stroke.
Arm Movement in Different FRs in Healthy Participants
We confirmed previous findings that healthy participants could reproduce the same reaching trajectory when additional DFs are added to the task, such as when leaning the trunk or taking a step.37,40 Changes in motor behavior occurred by rapid condition-specific adjustments of shoulder-elbow IJC. Healthy participants produced similar endpoint trajectories by maintaining the same shoulder-elbow IJC regardless of the trunk condition in the egocentric FR or by rapidly adjusting the IJC according to trunk conditions in the exocentric FR. Results are consistent with previous studies of responses to perturbations of arm movements made in external FRs in healthy participants (eg, Ghafouri et al, 8 Shaikh et al, 23 Tomita et al, 40 Rossi et al 41 ). For example, healthy participants reproduced the same endpoint trajectory when the trunk was free or blocked when making planar within-reach 38 and beyond-reach movements.23,41 The rapid (~40 ms) changes in shoulder-elbow IJC could have resulted from vestibular and/or proprioceptive afferent influences on arm motoneurons (MNs) linked to trunk motion.41,42 Vestibulospinal signals resulting from forward head movement during trunk flexion and proprioceptive signals from hip flexors could trigger short-latency changes in ongoing whole body movement, as previously shown by galvanic stimulation (ie, vestibular) 43 and neck muscle vibration affecting proprioceptors.44,45 In contrast, poststroke individuals had difficulty producing similar hand trajectories, either because they did not maintain a consistent shoulder-elbow IJC (egocentric target) or appropriately adjust IJC (exocentric target) when trunk conditions changed (see below).
Motor Control Frameworks
Several approaches explaining how motor tasks are accomplished in a chosen FR have been proposed. The conventional, computational framework assumes that the brain directly specifies the desired motor outcome in a spatial FR, and transformations from one FR to another are based on mathematical computations (eg, Mussa-Ivaldi and Danziger 46 ). Recently, this approach has been questioned because of its inconsistencies with the principle of causality underlying physical and physiological laws and with nonlinear MN properties implying that their input/output functions are mathematically irreversible.15,47 This implies that preprogrammed motor output cannot be transformed into the requisite input synaptic signals to MNs to physiologically actualize the program.
Another way to consider how the control system shares task demands between different joints or body segments according to individual, task, and environmental constraints 48 is by the formation of coordinative structures or synergies (eg, Latash, 11 Kelso et al 49 ). Once a spatial FR is specified, muscle and kinematic synergies dynamically emerge from interactions between neuromuscular elements and environmental factors. 50 Experimental findings support the role of FRs in guiding specific actions, especially for UL 48 and whole body reaching. 40
In the physiologically based referent control framework, 47 posture and movement are controlled indirectly by specifying neurophysiological parameters influencing but remaining independent of the motor outcome. Parameters influence the origin or referent points of spatial FRs in which MNs and reflexes function and motor actions emerge. The system adjusts parameters based on sensory feedback until the emergent action meets the task demand. This is supported by findings that descending cortical MN influences can be decorrelated from EMG levels. 51 Thus, instead of directly specifying EMG output (ie, muscle activation levels), descending influences determine where, in body coordinates, a muscle begins to be activated. This is achieved by setting the threshold position, called the referent body configuration.
To generate motion in an FR, the system specifies a referent arm-trunk configuration, R, at which all arm muscles are silent—that is, reach their activation thresholds (Figure 5). At the initial configuration, the endpoint position corresponds to position Ri. To produce movement, the system shifts the referent body configuration to Rf, producing a referent trajectory (solid line). This is done in advance of the actual arm trajectory (dashed line) that emerges as a deflection from the referent trajectory as a result of proprioceptive reflexes and the environmental interaction of the arm (ie, gravity, inertia). The system tends to minimize the difference between the actual and referent body configurations regardless of the number of DFs involved.37,38
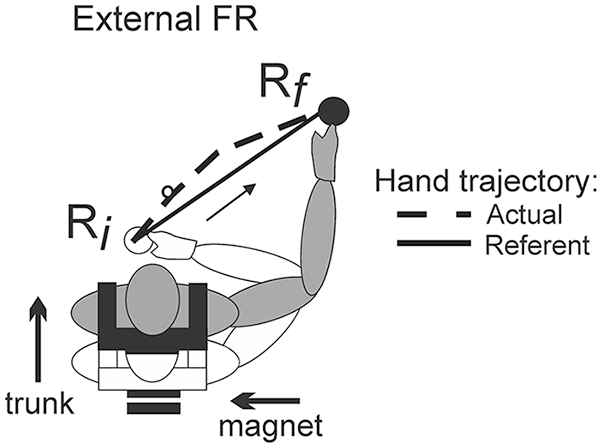
Schematic diagram of the referent arm-trunk configuration for a movement made in the external frame of reference (FR). The hand traces the referent hand trajectory (solid line) between the initial position Ri (open body configuration) and the final position Rf (gray body configuration). The actual hand trajectory (dashed line) deviates from the referent trajectory because of the interaction of the limb with the environment (ie, gravity, inertial forces). Muscle forces emerge as a result of the deflection of the actual from the referent hand trajectory.
By gradually changing the referent arm-trunk configuration from Ri to the final value, Rf, the system generates a hand trajectory along the stick in task 1. Relative to the trunk, this trajectory remains the same whether or not the trunk moves. In other words, with repeated trials, the system may reproduce shifts from Ri to Rf, resulting in the same hand trajectory and IJC regardless of the trunk condition, as observed in our study. Similarly, the system may produce a referent hand trajectory from an initial to a final target in an external FR. In this case, trunk motion influences the hand position, but the system maintains the actual hand trajectory by changing the referent arm-trunk configuration and minimizing the difference between the actual and referent trajectories. Kinematically, this would be associated with a change in the IJC, as observed in task 2.
Arm Movement in Different FRs in Stroke
Individuals with mild stroke could not produce similar endpoint trajectories with and without trunk displacement for both tasks, evidenced by earlier trajectory divergences compared with controls (Figure 4). This was accompanied by deficits in the ability to reproduce (task 1) or quickly alter (task 2) elbow and shoulder joint excursions when the trunk was unexpectedly blocked. Deficits in rapid motor responses in stroke have been demonstrated previously for reaching in exocentric FRs,23,38,52,53 but this is the first study to illustrate deficits in egocentric FRs. There are several important differences between the current and previous studies invalidating direct comparisons. Specifically, in Shaikh et al, 23 participants with chronic stroke made more diagonal trunk-assisted reaching, whereas in the current study, reaches were made within arm’s reach and were more horizontal. In Shaikh et al, participants reached along a horizontal table, whereas in the present study, although participants were instructed to reach in the horizontal plane, movements were done in 3D space. Participants participating in the 2 studies differed, such that Shaikh et al 23 included participants with mild to severe motor impairment—thus, the need to evaluate sitting balance—whereas in the current study, all participants had only mild impairment and had no apparent sitting-balance deficits.
Deficits were present in the hemiparetic arm, regardless of arm dominance, side of stroke, and mild cognitive deficits, in individuals who had only mild clinical impairment (mean FMA of 58/66). Because all participants had stroke in the middle cerebral artery area resulting in UL hemiparesis, it is likely that damage to the tempero-fronto-parietal cortex resulted in deficits in the ability to plan the specification of spatial FRs according to the minimization principle described above (Figure 5). This is supported by previous findings that, unlike healthy participants, individuals with chronic stroke have deficits in regulating the threshold of the stretch reflex of elbow flexors and extensors throughout the entire elbow range. 21 The stretch reflex threshold can be considered as the origin point of the spatial FR for that joint. 2 Deficits in threshold regulation at either the planning or execution level may lead to limitations in the production of task-appropriate reciprocal muscle activation and could have contributed to deficits in rapid responses to task conditions in the present study.21,54
Several participants with stroke had low MoCa scores. Behavioral markers of motor skill, such as the endpoint trajectory DP between free-trunk and blocked-trunk conditions, were correlated with cognitive scores but not with UL impairment levels (ie, FMA, sensation). This suggests that cognitive deficits may have led to difficulty in the formation of spatial FRs. Cognitive skills are important for rapid planning and execution of corrective movements. 55 Although information processing speed and executive function are the processes most impaired after stroke,56,57 other domains such as attention, memory, and visuospatial and constructive skills may also be affected.56,58 Poststroke individuals with greater cognitive impairments had lower gait velocity and poorer performance during dual-tasking memory tasks 59 and poorer UL motor recovery. 60 Furthermore, deficits in motor problem-solving were reported for a complex exocentric reaching task in well-recovered individuals with stroke, who used altered UL movement strategies to modify reaching trajectories to avoid a suddenly appearing obstacle. 52 Individuals also initiated corrections later in the reaching path compared with controls, resulting in a reduced margin of error. Similarly, in a task involving reaching to a target placed beyond arm reach in standing while flexing the hips, well-recovered individuals with stroke had difficulty modifying arm and trunk movements to a perturbation to maintain smooth and precise endpoint trajectories. 40 However, the association between cognitive deficits and motor problem-solving should be further investigated in a larger study with more detailed neuropsychological testing.
Limitations
A deeper understanding may have resulted if we had included the analysis of muscle activity and more detailed lesion information. Our results apply only to patients with mild stroke without apraxia or other perceptual deficits and cannot be generalized to patients with more severe stroke. Trunk stability was not assessed, but because all patients could perform the forward leaning task without falling, it is unlikely that they had significant trunk impairment. In addition, we did not evaluate reaching with the less-affected arm in which mild deficits have been previously reported, nor the effect of arm dominance, 61 and comparisons were made only with the nondominant arm of healthy participants, which may have introduced some interpretation bias.
Conclusion and Clinical Implications
Altered ability to adequately adjust shoulder-elbow IJC for both egocentric and exocentric arm movements suggests that individuals with mild stroke had deficits in higher-order motor skills that are not routinely evaluated clinically. These deficits may result in a loss of dexterity that may affect their ability to use their more-affected arm. Indeed, previous studies have shown that even though patients may score highly on clinical tests, they do not fully integrate their more-affected UL in daily activities. 62 In addition, cognitive deficits may affect the ability of individuals with stroke to produce movement in different FRs. Deficits in motor planning related to complex tasks should be considered in UL rehabilitation programs to improve UL recovery and arm use.
Footnotes
Acknowledgements
The authors would like to thank Kaci Bouattou, who contributed to some of the data collection, and Rejean Prevost as well as the volunteers who took part in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
