Abstract
Background. Knowledge of how damage to brain regions and pathways affects central nervous system control of coordination of reach-to-grasp (RTG) following stroke may not be sufficiently used in existing treatment interventions or in research that assesses their effectiveness. Objective. To review current knowledge of motor control of coordination of RTG and discuss the extent to which this information is being used in research evaluating treatment interventions. Method. This review (1) summarizes the current knowledge of motor control of RTG coordination in healthy individuals, including speculative models and structures of the brain identified as being involved; (2) summarizes evidence of RTG coordination deficits in people with stroke; (3) evaluates current interventions directed at retraining coordination of RTG, including a review of the extent to which these interventions are based on putative neurobiological mechanisms and reports on their effectiveness; and (4) recommends directions for research on treatment interventions for coordination of RTG. Results. Functional task-specific therapy, electrical stimulation, and robot or computerized training were identified as treatments targeted at improving coordination of RTG. However, none of the studies reporting the effect of these interventions related results to individual brain regions affected, and neurobiological mechanisms underlying improved performance were only minimally discussed. Conclusions. Research on treatment interventions for coordination of RTG needs to combine measures of interruption to brain networks and how remaining intact neural tissue and networks respond to therapy with measures of spatiotemporal motor control and upper-limb function to gain a fuller understanding of treatment effects and their mechanisms.
The Problem
Upper-limb motor deficit following stroke is common, with approximately 88% of stroke survivors demonstrating symptoms, but only half of the people affected regain effective functional use of their arm. 1 More effective treatments are needed. One major functional goal for rehabilitation after stroke is to retrain coordination of reach-to-grasp (RTG). Interventions for coordination of RTG have been based primarily on type and severity of presenting symptoms such as muscle weakness, abnormal muscle tone, and poor neuromuscular control. However, there is a growing body of knowledge about how the central nervous system (CNS) controls coordination of RTG and how damage to brain regions and pathways affects performance. This information may be underexploited in existing treatment interventions, which may not be sufficiently based on an understanding of neurobiological mechanisms.
The Solution
Although treatment by therapists will ultimately be determined by the type and severity of presenting impairments, knowledge about the effects of particular lesions on interruption to local and remote brain regions known to have a role in coordination of RTG and the resultant consequences for movement organization underlying coordination of RTG can supplement this information to improve the specificity of our treatment strategies. We aim to provide a platform on which to guide next steps in research in this area that will ultimately lead to recommendations for clinical practice. To provide this platform, this appraisal has 4 aims: (1) to summarize the current knowledge about motor behavior and neurobiological mechanisms of RTG coordination in healthy individuals, including speculative models and structures of the brain identified as being involved; (2) to summarize evidence of RTG coordination deficits in people with stroke based on results of studies that have contrasted coordination in healthy individuals and those with stroke; (3) use results of a recently completed systematic review to identify and evaluate current interventions directed at retraining coordination of RTG, including an assessment of to what extent these interventions are based on knowledge of neurobiological mechanisms, and report on their effectiveness; and (4) to suggest directions for research that will lead to recommendations for rehabilitation of RTG coordination, in the future. We build on previous reviews that have addressed RTG in stroke survivors more generally 2 by concentrating on and expanding knowledge specifically concerned with coordination of RTG after stroke. The discussion will focus on the coordination of 2 components—transport and grasp—which constitute the main focus of recent research in this area and will be limited to unimanual coordination and to movements of the shoulder, elbow, wrist, hand, and digits in order to provide a detailed analysis of these components. Coordination of the arm with other body parts such as the trunk and the other arm or with grip lift force is also important, but these aspects are not the focus of this particular discussion.
Coordination of RTG in Healthy Adults
Kinematics of Reach to Grasp
During RTG, the hand follows a characteristic path as it moves toward an object, described as the “transport” component, defined as the change in position over time of the hand. Transport is commonly depicted by the velocity and acceleration profiles plotted against absolute time (usually in milliseconds or against normalized movement duration [each time point is expressed as a percentage of total movement duration]). The velocity profile of the hand is typically asymmetric (Figure 1B), with the peak velocity (PV) achieved within 50% of movement duration. The part of the movement occurring before PV is thought to be planned and activated prior to the beginning of the movement. 3 The time after PV, described as the “deceleration” phase, is thought to be controlled in a feedback manner, whereby visual or proprioceptive information made available to the CNS after the movement begins is used to correct the movement according to task goals and environmental demands. The acceleration profile increases from the start time to a peak acceleration and then decreases to a peak deceleration (PD) point, near to the time of object contact.
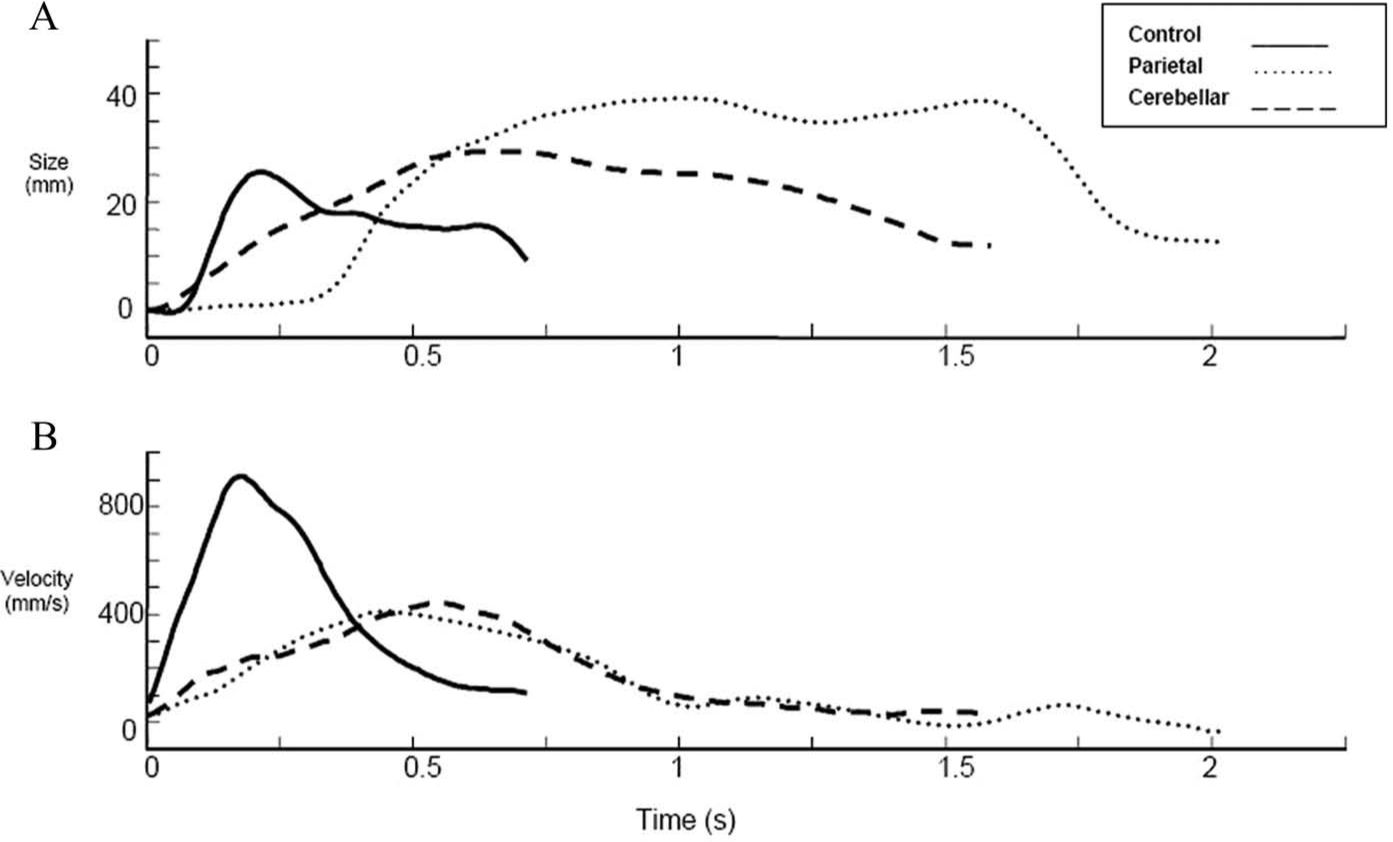
Sample grip aperture (A) and wrist velocity profiles (B) for fast reach-to-grasp movements. Movements were performed with the left hand using a precision grasp. The start position was midline with index finger and thumb together. The 10-cm high and 1.5-cm diameter cylindrical target was located 35 cm away in the right hemispace. Lines represent 1 trial each for a healthy control (black line), a parietal stroke participant (dotted line), and a cerebellar stroke participant (dashed line). For a patient with a parietal lobe lesion, the wrist velocity profile (Figure 1B) appears more asymmetrical than that of the control participant. The overall movement time is longer and the peak velocity is much lower and occurs relatively early in prehension. The prolonged deceleration phase suggests a more corrective mode of movement control, mediated with visual feedback as opposed to the movement being mainly preprogrammed. The profile also shows multiple wrist velocity peaks, suggesting corrective movement for spatial errors. A large amplitude for grasp aperture and maximum hand opening (MA) occurs relatively early in the movement. Poor control for aperture parameterization may result from impaired visual mapping and difficulties processing this information during the deceleration phase. In comparison to healthy wrist velocity profiles, we can see from Figure 1B that movement time is increased and PV is reduced for the cerebellar participant. The wrist velocity profile shows a prolonged deceleration phase and fluctuations (although less distinct than the parietal participant), supporting online corrections prior to grasp. MA for the cerebellar participant (Figure 1A) is large but smaller by comparison to the parietal participant.
The grasp component consists of an opening phase, where the hand and aperture are adjusted to object shape and orientation, and a closing phase, where the hand and fingers close on the object. These phases are described as the change over time of the distance between the index finger and thumb (Figure 1A). The timing of hand opening and closing is adjusted according to the movement end goal. 4 Maximum grasp aperture occurs at around 50% to 70% of the movement duration. 5 Modulation of the palmar arches of the hand (oblique, distal transverse, and longitudinal arches) prepare the hand for grasping in 3 phases: transport shaping, from onset to peak wrist velocity; preshaping, from peak wrist velocity to time of grasp; and contact shaping, from grasp to PV of object transport. 6 Palmar arch modulation can vary for different types of grasps. 6
Models for Coordination of RTG
The coordination between transport and grasp required to ensure successful RTG is partly defined by an invariant temporal relationship between the 2 components, where the start time of the opening of the hand is correlated with the start time of hand movement toward the object, 7 and the time of maximum hand opening (MA) is correlated with the time of PD of the hand. 5 A relationship has also been demonstrated between MA and PV, where although PV occurs prior to MA, the 2 events are significantly correlated. 7
When either grasp or transport is disturbed, for example, by manipulations of object size or object location, 8 increasing the speed of transport, 9 presence of obstacles in the hand’s path, 10 or mechanical perturbations of the arm, 11 there is an adjustment made to the other component to compensate. This implies a comparative and correction process between the 2 components.
A hierarchical arrangement of motor control processes has been suggested where reach and grasp components are governed by independent motor synergies, which are in turn coordinated by a higher level control synergy. 12 Substantive data from both computer simulations and real individuals 13 support the idea that the reach is planned with respect to the expected duration to the target via a consistent hand closure time. There is a 2-way interaction between the neural processes controlling transport and grasp, so that the expected duration to the target of each of these trajectories is compared and adjusted, so that they are temporally matched. For example, when both object size and location are “perturbed” at movement onset, MA and PD are both delayed to allow them to be temporally matched again after adjustment to each component has been made. 8
A universal control law governing this interaction in different task conditions has yet to be discovered. Rand et al 14 found that a law that states that when hand distance to the object becomes less than a linear function of aperture size, hand velocity, and hand acceleration, grasp closure will be initiated, fitted experimental data well when both speed and distance were varied.
There is more support for the idea of distance from object being a controlling parameter than the time to close the hand. The time taken to close the hand remained constant over varying reaching amplitudes in 1 study 15 ; however, contrasting data show increasing time to contact with increasing object size, 16 whereas the distance travelled by the wrist after MA is relatively invariant under a variety of task conditions, such as alterations of object distance, presence/absence of objects, 17 and different involvement of body segments. 2
Alternative models for control of the hand have been proposed. Smeets and Brenner 18 suggest that the CNS determines independent trajectories for each digit based on the approach onto the object’s surface, supported by an experiment where response to an obstacle placed near the target object was guided more by placement of individual digits than grasp aperture. 19 Alternatively, some evidence suggests that grasp can be described by a small number of postural synergies and may be controlled based on a temporal weighting of these synergies. 20
Recently, a concept of threshold position control has been proposed, whereby a threshold posture is associated with a specific state of the neuromuscular system, and muscles are silent in this threshold state until there is a deviation from this state. 21 For example, the nervous system could specify a virtual distance between index finger and thumb for a grasp, the size of the object could cause an actual deviation from this, and grip force would be adjusted in response. Furthermore, others have considered RTG as a complex higher-order motor behavior consisting of 3 elementary components—transport, grasp, and orientation to the object (postural rotation of the hand), which unfold concurrently within a common time window, so that if the extent of hand rotation was increased, for example, the duration of the transport component would also increase. 22
Coordination of RTG requires the CNS to receive information about the movement, so that adjustments can be made. Feedback from vision and proprioception can be used in the latter stage of the movement to guide the hand as it moves toward the object. Evidence that this sensory information is used to aid coordination comes from studies of deafferented patients who, when deprived of vision, demonstrate a delayed grasp onset, longer grasp closure time, increased deceleration time, and reopening of grasp. 23 However, the minimum time needed for a motor response to this feedback to occur is estimated at about 100 ms. Before this, it is likely that the ongoing motor command is modified according to a comparison of target position with an instantaneous internal estimate of hand position. 24
Brain Areas and Pathways Involved in RTG Coordination
Corroborative data from neuroanatomical studies has also increased understanding of coordination of RTG. The earliest approach to understanding brain function was to correlate deficits in function with postmortem analysis of which brain areas were damaged, typically after stroke. Animal studies provided investigators with more control over the size and location of lesions but did not remove the problem that it is not possible to logically determine with a single case whether the area involved is a primary area subserving a particular function or a less influential part of a distributed network. A separate issue was how to determine whether the area in question subserves just 1 or multiple functions. Multiple case studies (to reveal lesion overlap) and cases of double dissociation (lesion A affects function X but not Y and lesion B affects function Y but not X) provided ways to resolve these issues. But further understanding came from the ability to produce reversible “virtual” lesions using transcranial magnetic stimulation (TMS) in healthy volunteers. A repetitive pulsed focal magnetic field was produced over an area of the cortex, resulting in temporary suppression of function of that brain area. The most direct evidence of the function of different brain areas has come from functional imaging techniques, such as functional magnetic resonance imaging (fMRI). These determine blood flow changes linked to increased metabolism of brain areas involved in carefully selected tasks.
Primate studies have identified 2 parietofrontal neural circuits subserving control of coordination of transport and grasp: (1) a medial circuit, involving areas of the superior parietal lobule (medial intraparietal area/parietal reach region) and the dorsal premotor area 6, which carries information concerning object location (direction and distance), for hand transport and more proximal muscles 25 and (2) a lateral circuit, involving the inferior parietal lobule (in particular, the anterior intraparietal area) and the ventral premotor area 6, for grasping (object size and shape) and more distal musculature. 26 Studies inducing virtual lesions with repetitive TMS have confirmed these distinct roles of the dorsal premotor cortex, which has an important role in coupling between grasp and lift phases, 27 and the ventral premotor cortex, where disruption of neural processing via TMS just before object contact interferes with positioning of fingers on the object’s surface. 27 Although both circuits mainly subserve one or the other of the components, there is some overlap, so that both circuits are partially involved in both processes. The posterior parietal cortex (PPC) is involved in spatial monitoring, based on evidence that internal spatial monitoring is lost in monkeys with lesions in area 7 (in PPC) and that humans with lesions here show directional errors in reaching. 28 Because humans also show problems with grasp formation, it has been suggested that the PPC has a role in coordination as the hand nears the target. 29 The anterior intraparietal sulcus in the PPC is particularly involved in controlling grasping. 28 TMS to this area just after a perturbation of size of object to be grasped disrupts the online adjustment of maximum aperture in healthy people. 30 The parietal reach region neurons in the PPC are selectively active for reaches and encode target location. 31 TMS studies suggest that the PPC may be involved in determining current motor error to allow updating of muscle activation patterns, 24 so this area could be involved in the temporal comparison of transport and grasp.
The cerebellum provides an internal state estimate or sensory prediction that is used for online control of movements and to coordinate actions by the different effectors, including the eye, the hand, and the arm. 32 It may also integrate the independent motor processes for reach and grasp into 1 common motor program 33 and adjust the relative strength and timing of muscle activations based on internal predictions about the likely outcome of the motor command. 34 Thus, the cerebellum has a critical role in prediction of sensory consequences of motor commands. 35
Other areas discharging during RTG movements include the contralateral sensorimotor cortex and contralateral parietal cortex, bilateral premotor cortex (ventral and dorsal premotor areas), the supplementary motor area, 28 reticular formation, rostral mesencephalon, and superior colliculus. 36 The red nucleus, influencing movement via the rubrospinal tract, appears to have a role in the control of metacarpophalangeal extension during grasp. 36
Coordination of RTG in People With Stroke
The majority of existing studies of coordination of RTG after stroke have involved heterogeneous groups of participants with respect to brain areas and pathways affected, plus a small number of lesion-specific studies. Heterogeneous groups have demonstrated a longer movement duration,7,37-39 increased deceleration phase, 7 decreased movement smoothness (increased number of peaks in the velocity profile, or its derivatives, acceleration and jerk), 37 lower PV,7,39-41 increased variability of size and timing of PV, 41 and larger endpoint errors39,42 compared with healthy people. For grasp, there is increased variability7,39 and earlier occurrence of39,40 maximum aperture size, and the arches of the hand tend to be flatter than in healthy people, resulting in more arch modulation in the preshaping and contact shaping phases of grasp. 6 There is a cost to efficiency with such abnormalities, implied by optimization models for explaining movement control, such as minimum jerk, 43 torque change, 44 or variance of final arm position. 45
There are few studies that specifically address the temporal coupling between components of RTG. van Vliet and Sheridan 7 compared participants with stroke and healthy participants reaching for cups of different sizes. Although start of aperture and start of transport were significantly correlated in this heterogeneous group, this correlation was significantly weaker than in healthy participants when reaching for a larger cup. MA and PD were significantly correlated in stroke participants, but the correlation was weaker than in healthy participants when reaching fast for a small cup. In an alternative measure of hand-arm coordination during RTG, individuals with stroke exhibited poorer correlations between hand opening and elbow extension than healthy participants. 1 Michaelsen et al 46 found temporal coordination to be mostly intact in a heterogeneous group, with the time of MA and the temporal delay between time to PV and time to MA, expressed as percentage of movement duration, not significantly different from that of healthy participants. However, 4 out of the 19 stroke participants showed grasp timing abnormalities by keeping the grasp aperture constant throughout transport or suddenly opening the hand at the end of transport. Ability to adjust 1 RTG component in response to perturbations in the other is also affected by stroke. When transport speed was increased in 1 study, MA occurred later in the movement, but the latter adjustment was not as large as that in healthy individuals. 7 Stroke participants may therefore have trouble responding to changes in object size. There can also be an impaired ability to adjust timing of grasp for different sized objects. 47 The time of peak grasp aperture as a percentage of movement time of wrist transport represents a measure of the temporal coupling between hand transport and grasp formation. Several studies have noted an earlier peak grasp aperture as a percentage of movement duration.48,49 Peak finger extension velocity has also been measured as occurring earlier in the arm contralateral to the lesion, compared with the ipsilateral arm. 50
Thus, there is some preservation of the control strategy for coordination of RTG following stroke. Knowledge gained from studies of heterogeneous groups of patients is useful, but there is also a growing amount of information about the effect of lesions in specific brain regions involved in control of RTG, summarized in the following sections.
Lesions of the Parietal Cortex
Binkofsky et al 28 described the deficits of 6 people with stroke affecting the lateral bank of the anterior intraparietal sulcus in the PPC, who showed poor preshaping of the hand in the acceleration phase, increased and more variable aperture in the deceleration phase, and a later MA as percentage of movement duration compared with healthy participants. However, velocity profiles were almost normal, indicating that the performance of transport was preserved. Hand opening was also delayed by between 0.3 and 1.7 s in a case study following a large parietal lesion. 51
Movement during the transport phase tends to be preserved with a posterior parietal lesion, whereas the grasp showed deficits, including a larger-than-normal maximum aperture. 5 A lesion in the dorsal PPC, in a single case study, resulted in a lack of the usual modulations to changes in object size and location. 52 Patients with lesions in the superior parietal lobe and adjacent intraparietal sulcus commonly demonstrate optic ataxia, where misreaching (missing the target) and larger-than-normal maximum aperture 33 occurs to peripheral vision targets while looking at a central fixation point, but when fixating the target, no misreaching occurs. 34
Lesions of the Cerebellum
Cerebellar lesions appear to disrupt the relationship between transport and grasp aperture. 53 In 1 study, 6 patients with cerebellar lesions, in the hand ipsilateral to the lesion, showed greater variability in the velocity profile and more velocity peaks compared with healthy people 53 ; their grasp showed a larger and earlier MA expressed as a percentage of movement duration; and there was a shorter time from PV to MA. In another trial of pointing movements, 54 patients showed more variable endpoints and longer movement durations, with lower peak hand acceleration and deceleration and a longer deceleration compared with healthy individuals, and these deficits were accentuated in fast movements. The role of the cerebellum is likely to be greater in the planning rather than updating of aimed movements, 55 and unilateral cerebellar lesions are known to affect aimed movements of both arms. 56
In another study, 57 a cerebellar group showed within- and between-subject variability of time to MA, which occurred both before and after PD. MA for this group was also larger and showed multiple peaks, which, respectively, could suggest compensation and correction for inaccurate movements. Alternatively, decomposition or sequencing of movements may be the result of impaired parallel processing between the shoulder, elbow, and hand 58 or abnormal online processing of proprioceptive information during RTG. 59 Figures 1A and 1B demonstrate actual example wrist velocity and grasp aperture profiles for people with a cerebellar or parietal stroke and that for a healthy control from a study currently under way in our laboratory.
Lesions of the Posterior Limb of Internal Capsule (PLIC)
The RTG of 18 patients with internal capsule lesions was tested in 1 study, which found that in patients with PLIC lesions, MD was increased, time to PV was slightly increased, and there was a longer deceleration time and an even longer period closing in on the target and segmentation of movement, indicating abnormalities mainly in the latter part of reaching. 60 Patients with internal capsule lesions sparing the PLIC, however, were almost like controls in their performance. Grasp behavior was not measured, but it is likely that these changes in transport control will affect coordination of grasp—this has yet to be elucidated.
To summarize, there is some preservation of the temporal coordination of RTG after mild to moderate cortical stroke; however, a number of motor deficits have also been described. Research suggests that lesions involving the cerebellum may disrupt the control law governing the spatiotemporal coordination for prehension, although further studies are necessary to confirm this. The main types of lesions that will affect coordination of RTG will involve the parietal lobe, cerebellum, or PLIC. More studies are needed of coordination in groups defined by damaged brain regions and networks to increase depth of understanding of different effects, so as to inform future, more targeted therapies than we have at present.
Current Interventions for Retraining Coordination of RTG
A recent systematic review conducted by our group identified existing interventions targeted at coordination of arm and hand segments for RTG following stroke and the effectiveness of these treatments. 61 In all, 7 publications met the inclusion criteria of adult participants with stroke, an intervention or manipulation aimed at improving coordination of the upper limb during reach and grasp and a specific objective related to coordination of the upper limb. For the purpose of identifying interventions, the review was inclusive with regard to study design. To determine effectiveness of intervention, studies were required to have a measurement of upper-limb coordination. One randomized controlled trial, 2 case control studies, and 4 experimental studies without controls were included in the review. In all, 3 categories of potential intervention for improving hand and arm coordination after stroke were identified: functional task-specific therapy, electrical stimulation, and robot or computerized training. Functional task-specific training consisted of constraint-induced therapy with intensive training of the affected arm, including a range of object manipulations similar to daily life actions 49 : collaborative goal-setting real-world task practice, patient problem solving, confidence building and home practice coupled with repetitive task-specific training, 1 and hand opening and closing with arm movements during task-specific practice using actual objects. 7 Electrical stimulation involved stimulation to the median nerve to produce paresthesia in the absence of pain or muscle activation without task-specific practice. 48 Robot or computerized training consisted of opening and closing of the hand during assisted arm movements either with a robot 62 or a virtual reality exercise system with instrumented gloves.51,50 In view of the limited availability of good quality evidence and lack of empirical data, the authors of the review could not draw a definitive conclusion regarding the effectiveness of interventions aimed at improving hand and arm coordination after stroke. Improvements in hand and arm coordination during RTG were reported in 4 studies, whereas 1 study found no benefit. Two studies did not report specific effects of interventions for hand and arm coordination after stroke. Details and results of included studies are presented in Table 1, and further details are given in the published review. 61
Results for Hand and Arm Coordination During Reach to Grasp.

Abbreviations: JBI, Joanna Briggs Institute; Exp, experimental group; Ctrls, control group; CIMT, constraint-induced movement therapy; MT, movement time; SS, statistically significant; NS, not statistically significant.
“+” Refers to a positive difference in favor of the experimental group; 0, no difference; and “−,” difference in favor of the control group.
JBI evidence levels: 1, meta-analysis of experimental studies or 1 or more large experimental studies with narrow confidence intervals; 2, 1 or more smaller randomized controlled trials with wider confidence intervals or quasiexperimental studies; 3a, cohort; 3b, case-control; 3c, observational studies; 4, expert opinion, physiology bench research, or consensus.
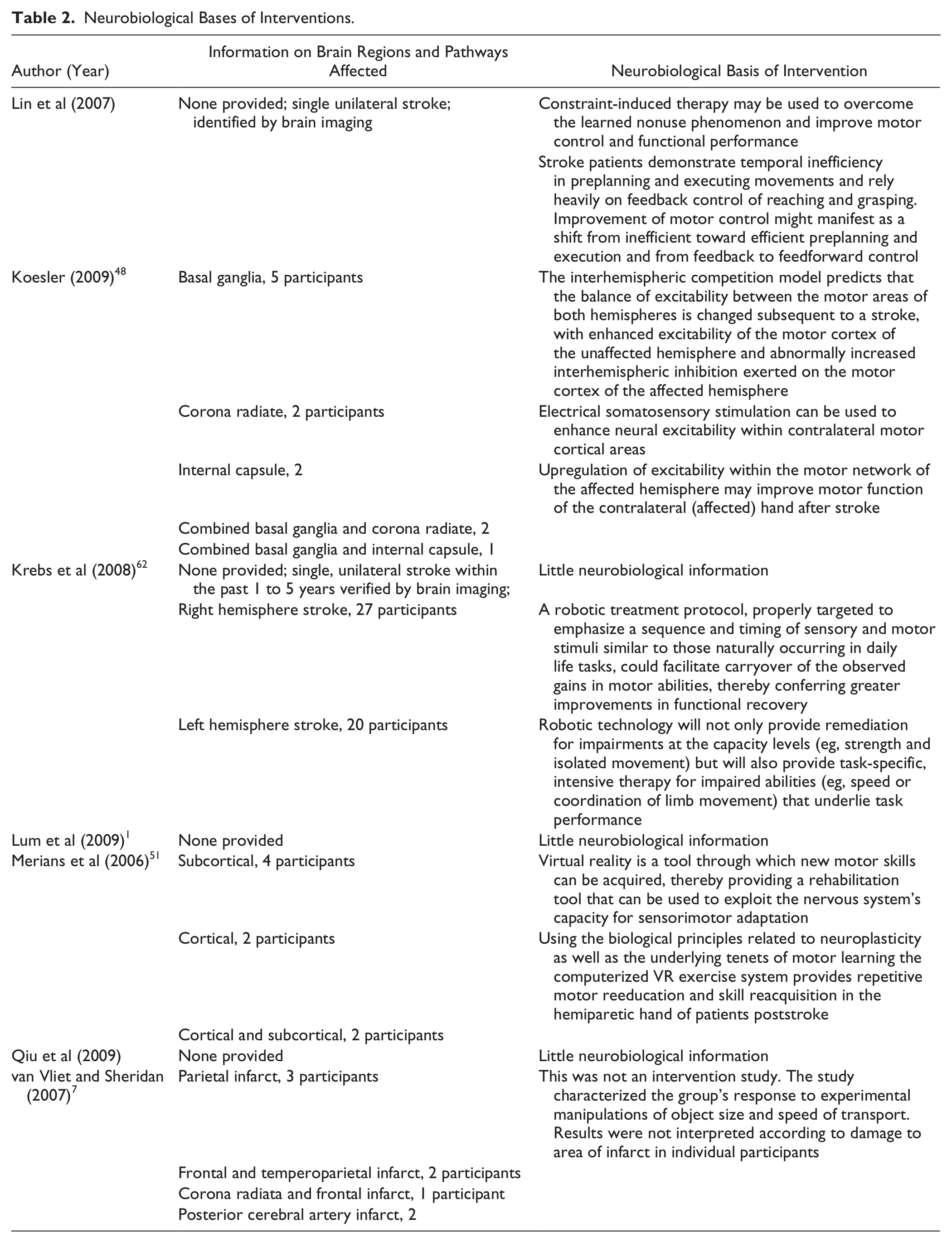
For the purpose of this point of view article, additional information was extracted from each of these included studies about the theoretical underpinning related to neurobiological information concerning brain regions and pathways and is presented in Table 2. Of the 7 studies, 3 provided details of the brain regions affected. None of the studies examined the effect of the intervention with respect to individual brain regions affected. One study demonstrated that people with an infarct in the area supplied by the middle cerebral artery, which with the anterior cerebral artery supplies blood to the parietal cortex, had a preserved ability to adjust grasp appropriately in response to changes in object size or movement speed. 7 This is in contrast to findings of studies of lesions of specific parietal areas—PPC 5 and anterior intraparietal sulcus, 63 both of which result in deficits in grasp. Therefore, it is probable that damage only to specific areas of the parietal cortex causes these deficits. Two studies48,51 discussed group results in the light of possible neuroplastic mechanisms behind improved performance. It was hypothesized by Koesler 48 that cortical excitability in affected cortical areas was increased by electrical somatosensory stimulation of the median nerve. Following stroke, there is hypoexcitability in the affected cortex with a compensatory increase in activation of the opposite hemisphere 64 ; therefore, any treatment that increases excitability of the affected cortex is likely to encourage more normal brain activation, which may result in improved coordination of RTG. The potential to improve will depend on corticospinal tract integrity, however. Those with a lack of corticospinal integrity may benefit more from bilateral arm therapies, which engage activation of the nonaffected hemisphere. 64 Merians et al 51 suggested that positive changes with virtual reality training were a result of brain reorganization mediated by intensive repetition of motor skills and also possibly by activation of the action/observation neural network. The studies highlighted the need for inclusion of kinematic measures that enable the assessment of spatial and temporal movement characteristics.
Neurobiological Bases of Interventions.
Suggested Future Directions for Research
It is common to interpret results in the context of impairment level. However, as yet, there appears to be little use of information about how different types of lesions have interrupted brain networks and how remaining intact neural tissue and networks respond to therapy in studies targeting coordination of RTG. It is recommended that future research should systematically develop a network-based model of recovery for coordination of RTG. Network-based models represent a paradigm shift from the standard lesion-deficit view and can be used to (1) characterize the deficit and recovery based on the functional-anatomical model of changes in the balance of activity across hemispheres and in functionally related remote locations, (2) predict ability to benefit from therapy using knowledge not only from clinical predictors but also functions served by viable brain tracts and networks, and (3) guide selection of individualized rehabilitation strategies based on knowledge of viable brain networks that may be accessed in therapy. 65 To discover the best interventions for particular individuals, research findings need to be also interpreted in the context of current information about damaged and preserved brain networks, motor control deficits, and biomechanical constraints such as contracture.
The availability of in vivo neuroimaging technologies makes this possible. Techniques such as high-resolution structural MRI can be used to reveal changes in brain gray matter density, diffusion-weighted imaging and tractography can reveal integrity and structural connectivity of brain white matter, and functional MRI, TMS, and positron emission tomography (PET) can reveal brain connectivity while performing tasks. With these advances, it should now be possible to identify specific groups of patients according to their underlying neurobiology and how this affects recovery of neural networks and function. TMS and fMRI measures have been used to predict upper-limb function in chronic stroke, where the presence of motor-evoked potentials (MEPs) in response to TMS predicted higher Fugl-Meyer upper-limb scores compared with cases where MEPs could not be elicited as a result of damage to the motor cortex. 64 For those without MEPs, a greater amount of asymmetry in fractional anisotropy of the internal capsule predicted poorer function. Furthermore, 3 specific groups of stroke patients have been identified according to which descending tracts are detectable with DTI, with best clinical scores for a group with traceable fibers in both anterior pons (pyramidal tract) and posterior pons (alternate motor fibers), followed by a group with fibers traceable only in the pyramidal tract, and lowest scores for a group with no traceable fibres in either the pyramidal tract or alternate motor fiber tracts. 66
This knowledge can then be combined with measures of motor deficit and/or effectiveness of learning-based interventions to inform the direction of individualized targeted therapy. For example, it is likely that patients with both motor cortex damage and asymmetry in PLIC over a critical point (fractional anisotropy value > 0.25) have limited functional potential. 64 In the context of interventions for RTG, 2 training approaches (unilateral and bilateral motor arm training) had different impacts on task-related brain activation. 67 This approach will also help with treatment planning. For example, in a study of robot-assisted therapy, structural MRI was obtained at baseline to outline the infarct, and then the degree to which stroke injured each of 4 descending white matter tracts was investigated (from the primary motor cortex, supplementary motor area, dorsal premotor cortex, and ventral premotor cortex). Improvements were strongly and negatively correlated with injuries to tracts descending from the primary motor cortex and dorsal premotor cortex in chronic stroke survivors. 68 Improvements did not show a significant correlation with motor outcome measures, thus highlighting the importance of not relying only on impairment levels to interpret study results. This kind of approach has not yet been applied in studies of arm-hand coordination after stroke, either in terms of predicting coordination recovery or the likely effect of targeted interventions for coordination. Because coordination involves specialized brain regions and pathways, it cannot be assumed that findings for more generalized upper-limb tasks also apply to recovery of coordination, and so specific investigations of coordination need to be conducted, incorporating both neurobiological measures and motor performance measures that reveal coordination ability, such as measures of spatiotemporal control of transport and grasp using motion capture systems. 49
Individuals present with a wide variation of brain lesions and functional deficits, and this heterogeneity makes it challenging to detect significant effects of interventions in studies where individuals are grouped for statistical analysis. Many of the studies that are available describing coordination deficits after stroke recruit participants with substantial heterogeneity. This, combined with the need for individualized treatment based on damaged and preserved neural networks, suggests that individual results should be presented in reporting study results, and statistical analyses that take account of this individuality should be used. As an example, in the study of robot-assisted therapy above, 68 correlational analyses were used to examine relationships between tract-specific injury and behavioral gains. Analyses may also be directed at subgroups of patients with interruption to specific cortical or subcortical regions of a particular system, 69 or individual patient data may be compared with expected variation in a group of healthy controls to determine standardized differences in brain imaging indices. 70 Also, individual case studies shed light on how people with particular lesions respond to specific treatments. 51 Finally, noninvasive mapping of interruption to specific pathways, such as corticofugal fibers, may be undertaken in individual stroke patients relative to trajectories established in healthy controls to infer disconnections and how these may relate to motor impairment and recovery. 71
Conclusion
There is a growing understanding of models and structures of the brain identified as being involved in CNS control of coordination of RTG and how these change in response to injury as a result of stroke. The basis for reported current interventions directed at retraining coordination of RTG are not well explained in terms of neurobiological mechanisms and require further investigations to assess their effectiveness. Research on treatment interventions for coordination of RTG needs to combine measures of interrupted brain networks and how remaining intact neural tissue and networks respond to therapy, with measures of spatiotemporal motor control and motor impairment to gain a fuller understanding of treatment effects and their mechanisms.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
During the writing of this manuscript, Paulette van Vliet and Leeanne Carey were financially supported by ARC (Australian Research Council) Future Fellowships, and Trudy Pelton was supported by The Stroke Association, UK.
