Abstract
Background. Drastic functional reorganization was observed in the ipsilateral primary motor cortex (M1) of a Paralympic long jumper with a unilateral below-knee amputation in our previous study. However, it remains unclear whether long-term para-sports are associated with ipsilateral M1 reorganization since only 1 athlete with amputation was investigated. Objective. This study aimed to investigate the relationship between the long-term para-sports and ipsilateral M1 reorganization after lower limb amputation. Methods. Lower limb rhythmic muscle contraction tasks with functional magnetic resonance imaging and T1-weighted structural imaging were performed in 30 lower limb amputees with different para-sports experiences in the chronic phase. Results. Brain activity in the ipsilateral primary motor and somatosensory areas (SM1) as well as the contralateral dorsolateral prefrontal cortex, SM1, and inferior temporal gyrus showed a positive correlation with the years of routine para-sports participation (sports years) during contraction of the amputated knee. Indeed, twelve of the 30 participants who exhibited significant ipsilateral M1 activation during amputated knee contraction had a relatively longer history of para-sports participation. No significant correlation was found in the structural analysis. Conclusions. Long-term para-sports could lead to extensive reorganization at the brain network level, not only bilateral M1 reorganization but also reorganization of the frontal lobe and visual pathways. These results suggest that the interaction of injury-induced and use-dependent cortical plasticity might bring about drastic reorganization in lower limb amputees.
Introduction
Structural and functional brain reorganization occurs following peripheral lesion such as amputation.1,2 For instance, motor-related areas such as the premotor cortex (PMC), precentral gyrus, and postcentral gyrus exhibited decreased cortical thickness after lower limb amputation (LLA).3,4 Additionally, gray matter (GM) volume in the vision motor-related and subcortical areas was also reduced following LLA.4-6 Moreover, functional connectivity between motor-related areas across hemispheres was reduced, accompanied by structural changes as represented by weakening of the corpus callosum fibers.4,7 These changes are interpreted as degenerative maladaptations of the brain after LLA and have been associated with phantom limb pain.8,9
Evidence of maladaptation after LLA is consistent across various studies; however, several studies have shown exceptional results regarding the reorganization of the ipsilateral primary motor cortex (M1) of the amputated limb. For example, increased corticospinal excitability ipsilateral to the amputated side has been demonstrated.10-12 Moreover, the drastic expansion of the M1 areas innervating lower-limb muscles and increased signal intensity in the ipsilateral M1 was observed with functional MRI (fMRI) during contraction of the residual limb in a long jumper with a unilateral LLA who holds the current long jump world record (8.62 m) but not in non-athletes with a unilateral LLA nor able-bodied long jumpers. 13 Undoubtedly, higher-level control of the athletic prosthesis is needed to achieve higher performance; therefore, it was inferred that ipsilateral M1 involvement would be required to optimize the motor function for precise prosthesis control.
In the corticospinal tract (CST), it is anatomically known that most parts of the fibers descend to the opposite side via the spinal cord, while up to 15% of intrinsic fibers could descend to the ipsilateral spinal cord directly in non-human primates.14,15 Although not fully understood in humans, studies using transcranial magnetic stimulation (TMS) have suggested the existence of an ipsilateral pathway, especially in the proximal muscles, even though functional significance has not been clearly defined. 16 Recently, several studies have reported that the ipsilateral motor pathway plays a crucial role in compensating for motor impairment in both upper and lower limbs after subcortical brain injury.17-21 This phenomenon can also be seen after spinal cord injury22,23; therefore, ipsilateral M1 activation might help compensate for the functional deficit after movement disorders.
Based on these studies, the human ipsilateral pathway descending from the ipsilateral M1 has intrinsic potential to contribute to the improvement of motor function. Although people with LLA do not suffer central nervous system injury, ipsilateral M1 activation could be considered a positive adaptation for the optimization of a residual limb as with other movement disorders. In the case of upper limb amputees, most researchers have clarified an enlargement and lateral displacement of the motor representation of residual limb, 10 while some studies found robust ipsilateral cortical activity when controlling their intact hands, regardless of the unknown mechanism of inducement. 24 Therefore, it is assumed that factors other than just amputation might be required to induce ipsilateral M1 involvement. Specifically, as para-sports require intense use and precise control of the prosthesis, this may very likely cause reorganization of the ipsilateral M1. It is well known that intense limb use induces structural and functional plasticity (ie, use-dependent/learning-induced plasticity), and such plasticity has been associated with motor performance.25-27 Thus, it is hypothesized that intense limb use after amputation would induce the involvement of the ipsilateral motor pathway descending from the ipsilateral M1 on the amputated side to optimize the motor function for precise prosthesis control. Nonetheless, whether long-term para-sports are associated with ipsilateral M1 reorganization remains unclear since only 1 amputee athlete and 4 amputee non-athletes were evaluated in our previous study. 13 Therefore, the purpose of the present study was to investigate the effects of long-term para-sports training on post-LLA brain activation. Additionally, to analyze whether GM volume was related to sports years, we also measured T1 images and performed voxel-based morphometry (VBM).
Methods
Participants
Population demographics and relevant clinical information from the participants.
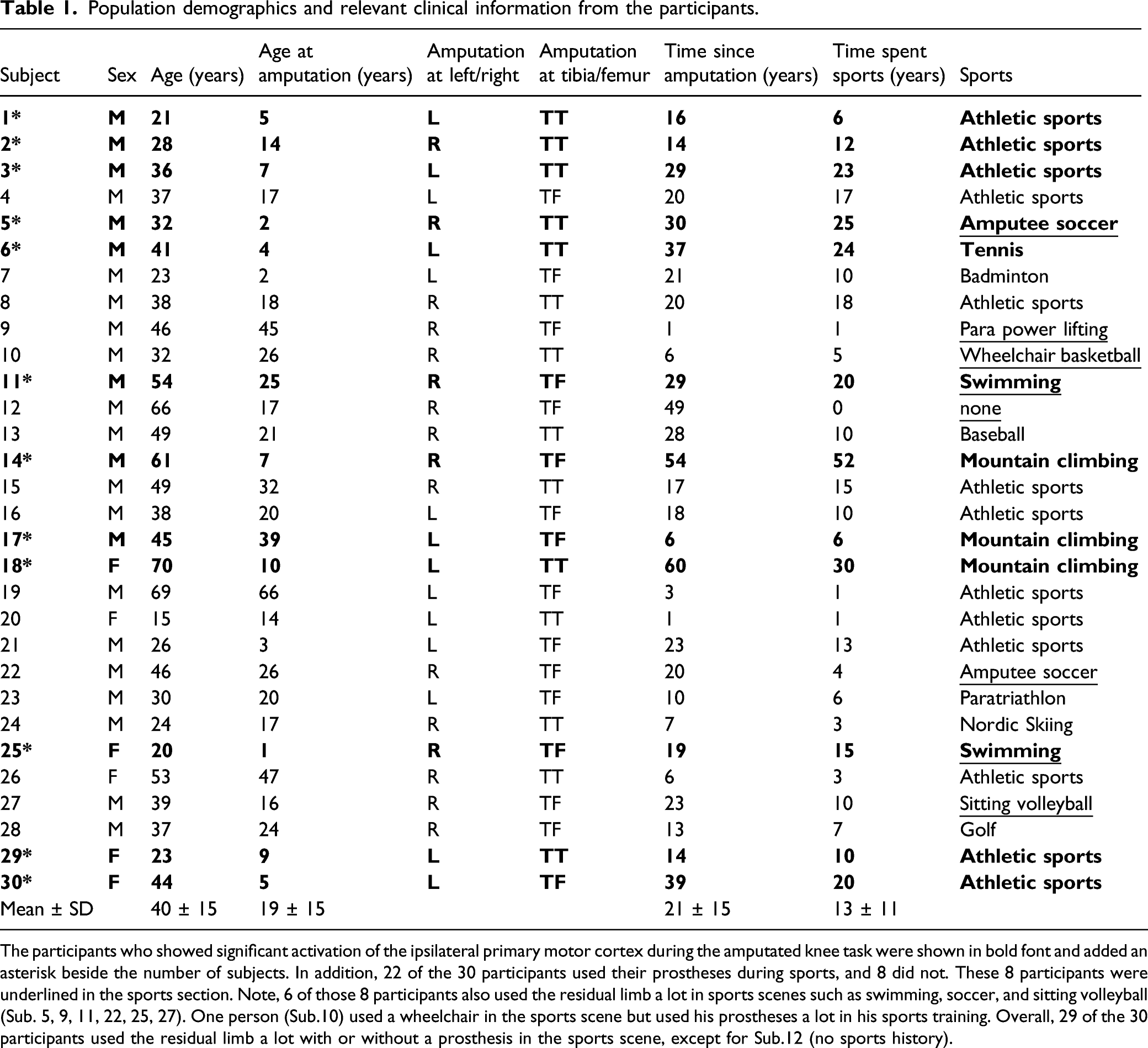
The participants who showed significant activation of the ipsilateral primary motor cortex during the amputated knee task were shown in bold font and added an asterisk beside the number of subjects. In addition, 22 of the 30 participants used their prostheses during sports, and 8 did not. These 8 participants were underlined in the sports section. Note, 6 of those 8 participants also used the residual limb a lot in sports scenes such as swimming, soccer, and sitting volleyball (Sub. 5, 9, 11, 22, 25, 27). One person (Sub.10) used a wheelchair in the sports scene but used his prostheses a lot in his sports training. Overall, 29 of the 30 participants used the residual limb a lot with or without a prosthesis in the sports scene, except for Sub.12 (no sports history).
Experimental Procedures
All participants performed rhythmic isometric muscle contraction tasks that comprised 6 motor tasks involving both legs (right and left) and 3 joints (ankle, knee, and hip) in an MRI scanner. The all participants were instructed to perform voluntary isometric contractions of the knee (rectus femoris) and hip joint (gluteus maximus) at constant rhythm of 1 Hz. The ankle joint on the non-amputated side performed the plantarflexion movement at 1 Hz, while an imagination task of cyclic plantarflexion from the first-person perspective without muscle contraction was assigned on the amputated side.
28
Before MRI measurement, the participants completed questionnaires, including 1) medical history: all previous diseases; 2) injury date: date of amputation and post-amputation duration, and 3) sports history: sport played by the participants at least once a week and number of years spent playing. For fMRI measurements, participants performed the contraction task using each lower limb joint. As a yellow fixation point was flashed on the screen at 1 Hz, instructions such as “L Knee” or “R Hip” were presented underneath. Subsequently, the participants contracted these muscles at a 20% maximum voluntary contraction (MVC) following the blinking yellow fixation point (Figure 1A). Experimental setup and group level brain activity. (A) The participants lay on their backs in the MRI gantry and were instructed to observe the screen and perform a contraction task on each lower limb joint during fMRI. (B) Brain activity in each task, as indicated by second-level group whole-brain analysis.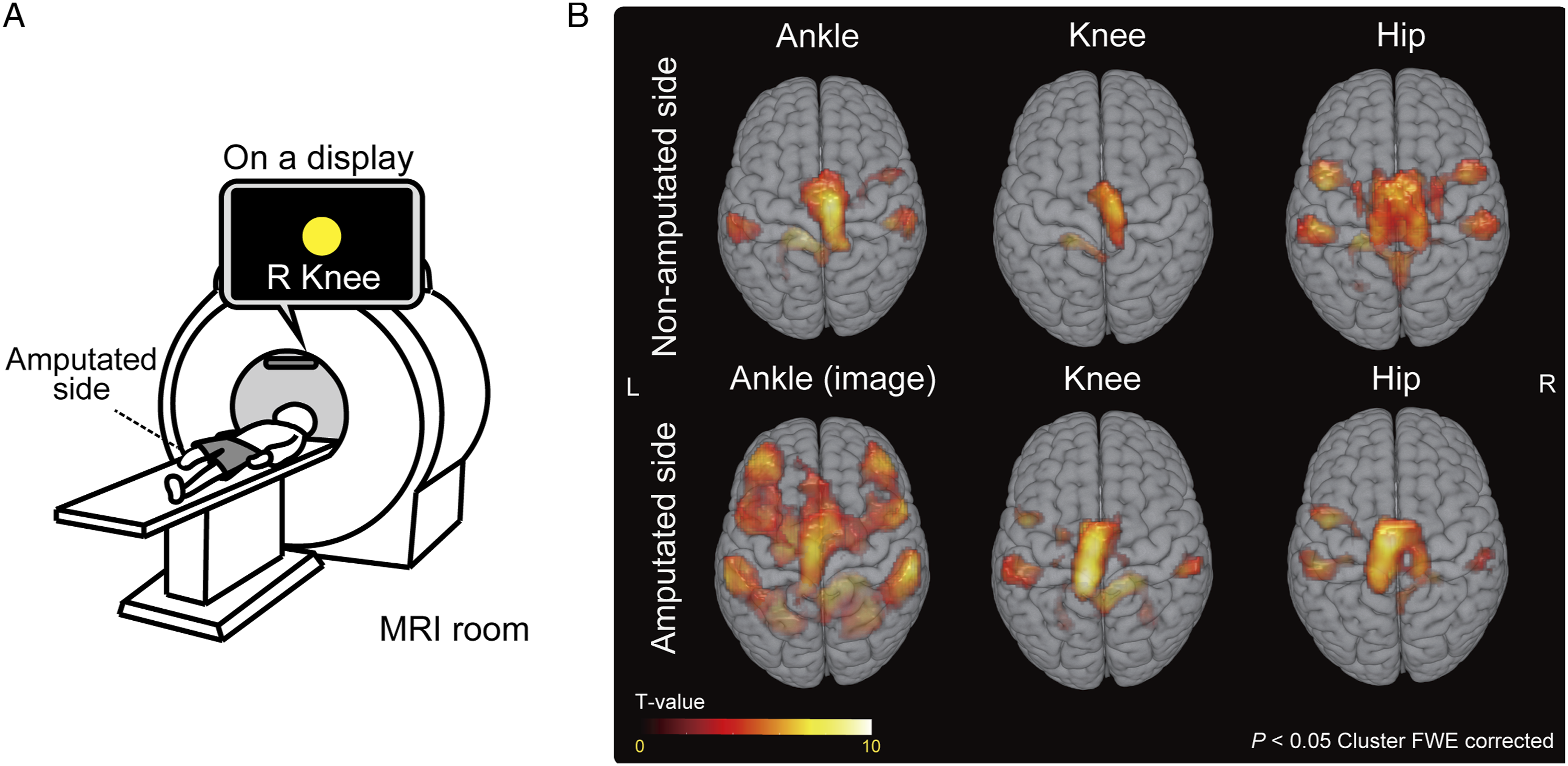
Before entering the MRI scanner, the participants underwent a practice session of contracting each muscle at 20% MVC without contracting other muscles during the task using surface electromyogram (EMG). Specifically, we conducted the following 2 measures to prevent unnecessary muscle contractions during the task. First, during the practice session, EMG was used to ensure that unnecessary muscle contractions for body stabilization did not occur. For correct learning of the task, the participants were asked to complete 20 consecutive sessions. Actual muscle activity is described in the supplementary material, and there was no unnecessary muscle contraction. Second, during the actual scan, 2 monitors, 1 on the side of the participant and the other on the parietal side of the head, showed whole body and checked constantly that the task was being performed correctly and without unnecessary muscle contractions. We monitored their bodies and confirmed that there was no visible movement due to the unnecessary muscle contraction during the task. As it is difficult to see muscle contractions of hip muscles directly under the supine position, we carefully checked and confirmed that there was no movement of the pelvis or femur accompanied with muscle contractions. Detailed methods and results are described in Supplementary file 1.
Subsequently, the participants entered the MRI machine while lying on their backs and watched the monitor while performing the contraction task as previously practiced over 3 fMRI scan sessions (Figure 1A). We intensively monitored their limbs during the task using 2 cameras to ensure proper task performance and that no other muscles were contracted, as mentioned above. Each session included an 8-s pre-trial dummy scan, 6 alternate task repetitions (6 movement types × 1 repetition), and rest periods. The 6 movements were randomly ordered to impede the prediction of the subsequent task. This order was consistent across all participants. Both task- and rest-periods lasted 20 s; therefore, each session lasted 268 s (Figure 1B), which was repeated thrice. During the tasks, T2* weighted (T2w) images were obtained using a 3.0T MRI (MAGNETOM Prisma, Siemens, Munich, Germany). Finally, high-resolution T1 weighted (T1w) structural images were acquired.
Scanning Procedure
All MRI data were acquired using a 3.0-T MRI scanner with a 64-channel head coil. We obtained functional T2w echo-planar images reflecting the BOLD signal using the following parameters: repetition time (TR) = 2000 ms, echo time (TE) = 25 ms, flip angle = 90°, field of view (FOV) = 192 mm, 39 contiguous axial slices acquired in interleaved order, thickness = 3.0 mm, in-plane resolution = 3.0 × 3.0 mm and bandwidth = 1776 Hz/pixel. These image acquisition parameters were chosen based on previous studies using fMRI during the same contraction task. 13 Siemens auto-align was run at the start of each session. For structural analysis, high-resolution T1w structural images were acquired using a 3D magnetization-prepared rapid acquisition with gradient echo pulse sequence using the following parameters: TR = 2000 ms, TE = 2.9 ms, flip angle = 9.0°, FOV = 256 mm, 176 contiguous axial slices, thickness = 1.0 mm, and in-plane resolution: 1.0 × 1.0 mm.
MRI Scans Flipping
Initially, we flipped all MRI scans of the participants with left-side amputations in the sagittal plane to normalize all amputated sides to the right.29,30 Therefore, the right and left hemisphere were considered as ipsilateral and contralateral to the amputated leg, respectively. The images were subsequently analyzed.
Task fMRI Analysis
For all sessions, the first 4 scans were excluded to allow for magnetization equilibrium. The remaining data were analyzed using Statistical Parametric Mapping 12 (SPM12, Wellcome Department of Imaging Neuroscience, London, UK; www.fil.ion.ucl.ac.uk/spm) software implemented in MATLAB (Mathworks, Sherborn, MA) using the standard preprocessing pipeline. Preprocessing was performed as follows: 1) Realignment: all images were realigned to the first volume; 2) Coregistration: the T1w structural and average echo-planar imaging (EPI) images from each fMRI session were aligned to superimpose the head-position information; 3) Normalization: all EPI scans were normalized to the Montreal Neurological Institute (MNI) space (TPM.nii in SPM12) by referencing normalization parameters of segmentation of the structural scan; and 4) Smoothing: EPI scans were smoothed using a Gaussian kernel of 8 mm.
After preprocessing, statistical analyses were performed using 2 steps. First, we performed single-participant (first-level) analyses using a general linear model to depict the general brain activation features of each participant. Head motion parameters obtained during the preprocessing stage were included as nuisance regressors in each session to minimize the effects of head motion artifacts. We created the following 6 contrasts: (1) right (amputated) ankle contraction > rest, (2) left (non-amputated) ankle contraction > rest, (3) right (amputated) knee contraction > rest, (4) left (non-amputated) knee contraction > rest, (5) right (amputated) hip contraction > rest, and (6) left (non-amputated) hip contraction > rest. Subsequently, to determine the activated areas in each task of the group effect (second-level), we performed a one-sample t-test for each task based on a random-effects model. Statistical thresholds in both first and second-level analyses were set at a voxel and cluster level of P < .001 (uncorrected) and P < .05 (family-wise error [FWE] corrected).
To identify the activated brain regions correlated with injury and sports years, we used multiple regression models to determine the correlation of brain activity during each joint contraction with injury/sports years. When performing a multiple regression analysis, age, injury, and sports years were added as covariates. Statistical thresholds were set at a voxel and cluster level of P < .001 (uncorrected) and P < .05 (family-wise error [FWE] corrected), respectively. Furthermore, to verify whether the results differ depending on the amputated part, the TT and TF amputees were compared by drawing an approximate line based on the least-squares method in each group and calculating its slope. Finally, to assess whether correlations were amputated part-specific, we evaluated brain activity during the same task in the non-amputated side (ie control conditions and areas) using the inverted x-coordinate of MNI locations that were significantly correlated with sports years.
T1 Structural Image Analysis
The GM volume of each participant was calculated using the SPM12 standard preprocessing pipeline. T1w images were initially segmented for GM, white matter (WM) and cerebrospinal fluid, based on the International Consortium of Brain Mapping template for East Asian populations. Next, a normalization procedure was conducted, where GM images were registered to a standard MNI space to interpolate differences in each brain shape using high-dimensional DARTEL normalization. In the final preprocessing phase, a Gaussian kernel of 12 mm was used to smooth the normalized T1w structure images.
To identify whether the regional GM volume correlated with injury and sports years, we performed a correlation analysis of GM parameter estimates with injury or sports years using multiple regression models. Age, injury, and sports years were added as covariates. Further, we used total intracranial volume as a nuisance regressor to minimize the effect of each brain size. The statistical threshold was set at a voxel and cluster level of P < .001 (uncorrected) and P < .05 (FWE corrected). Furthermore, between-group differences were verified by drawing an approximate line based on the least-squares method in each group and calculating its slope.
Results
Task-Related Brain Activity
Brain Regions Activated During the Muscle Contraction Tasks in All Participants.
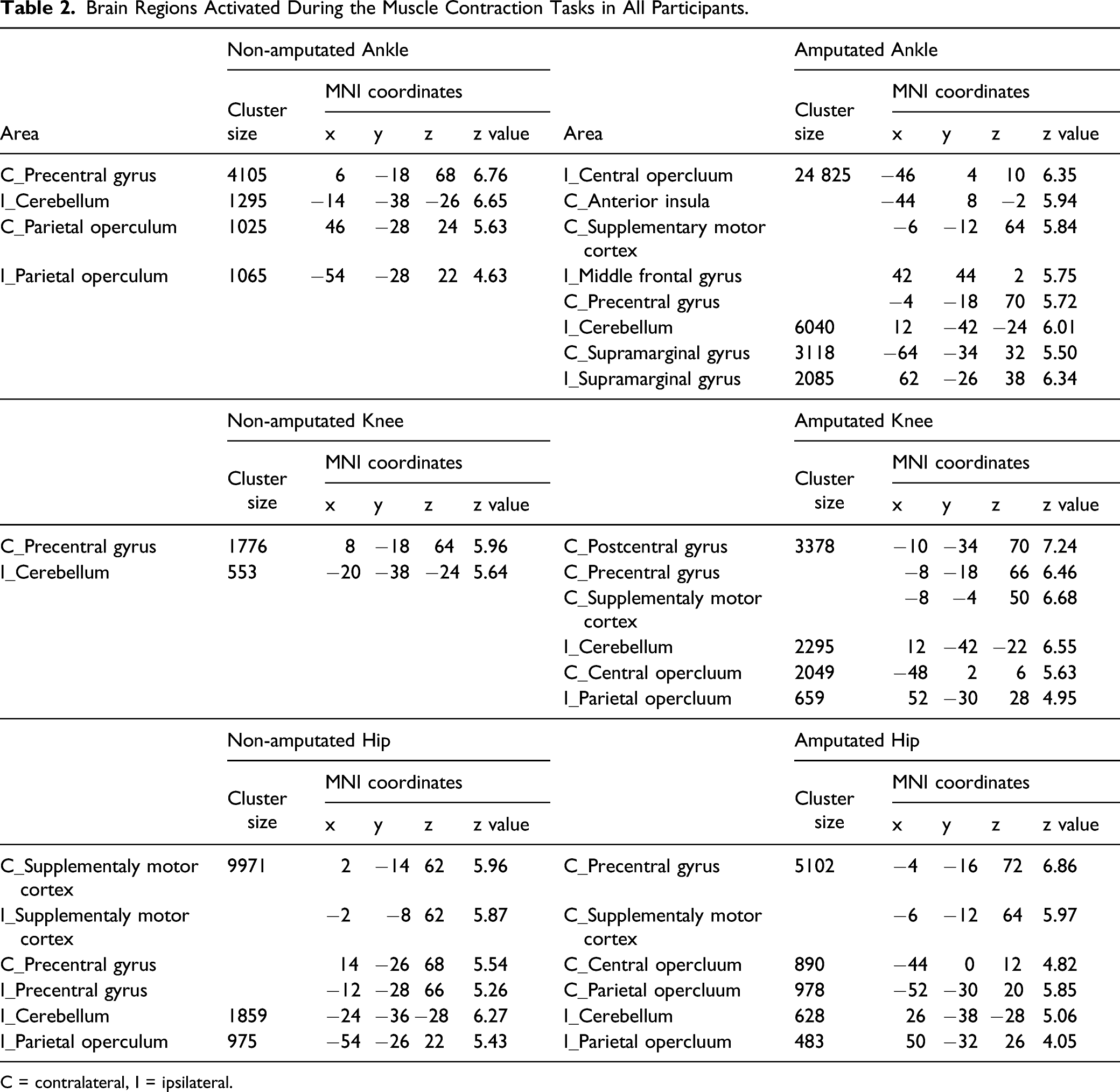
C = contralateral, I = ipsilateral.
Notably, individual (first-level) analysis revealed that 12 of the 30 participants exhibited significant ipsilateral M1 activation during the amputated knee task. The median (25th and 75th percentile values) number of sports years for participants who showed and did not show significant ipsilateral M1 activation were 20.0 (11.5, 24.3) and 6.5 (3.0, 12.3) years, respectively. Figure 2 shows representative brain activity during the amputated and non-amputated knee task in 6 typical participants. These 6 individuals were relatively similar in age and injury years but differed in sports years. The 3 participants on the left with fewer sports years showed only contralateral M1 activation during the knee task. Contrastingly, participants with relatively longer sports years showed distinct bilateral M1 activation only during the amputated knee task. Representative brain activities of participants with relatively short and long sports years history. Representative brain activities during both amputated and non-amputated knee contraction in 6 typical participants. The 3 participants on the left with fewer sports years showed only contralateral M1 activation during the knee task. Contrastingly, participants with relatively longer sports years showed distinct ipsilateral M1 activation during only the amputated knee task.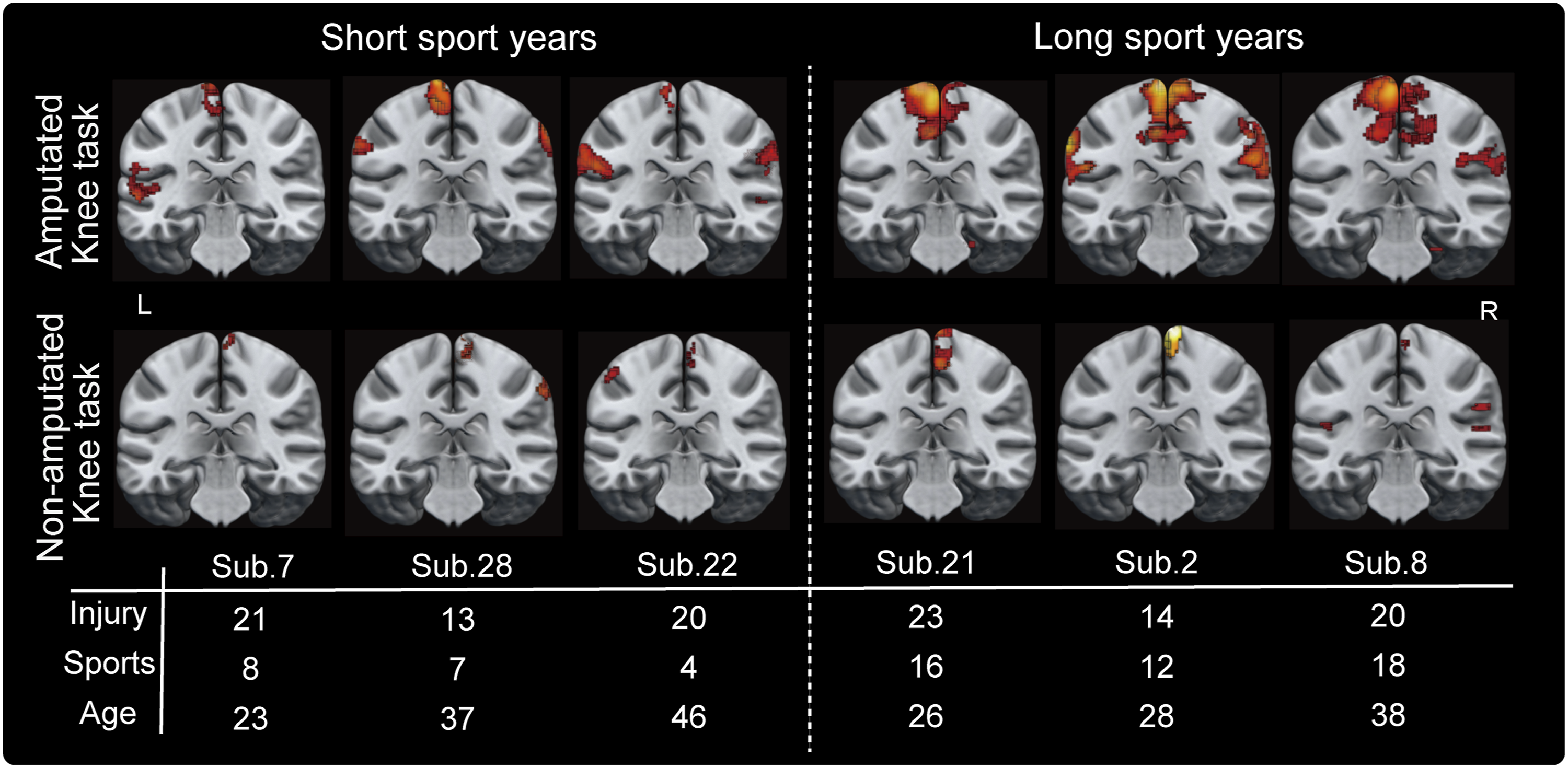
Association Between Brain Activity and Sports and Injury Years
Brain activity in the contralateral dorsolateral prefrontal cortex (DLPFC), primary motor and somatosensory areas (SM1), inferior temporal gyrus (ITG), and ipsilateral SM1 during the amputated knee task was positively correlated with sports years at a cluster level of P < .05 (FWE corrected) (Figure 3A). The correlated cluster of the M1 was defined as the SM1 because it exceeded the central sulcus and spanned the primary somatosensory area (S1). All data points of clusters correlated with sports years are plotted in Figure 3B. The correlations between brain activity during the non-amputated knee task and sports years as control conditions using the above clusters (inverted x-coordinate of MNI locations) are shown in Figure 3B. Regarding the non-amputated knee task, there were no significant correlations between sports years and brain activity in the bilateral SM1, DLPFC, and ITG. As shown in Figure 3B, brain activity in each region of one TF amputee (Sub.14, the right-most plot in each graph) was relatively larger and might have affected the findings. Thus, a reanalysis was performed after excluding this participant, but the results were not affected. Correlation of brain activity during the amputated knee contraction with sports and injury years. (A) Brain regions that are correlated with sports and injury years. Regions significantly correlated with sports years are shown in red, and regions significantly correlated with injury years, in green. (B) All brain activity values in all major areas that were correlated with sports years are plotted as circles. Transtibial and transfemoral values were indicated by black and white circles, respectively. The approximate lines for the transtibial and transfemoral groups were shown as solid and dotted lines, respectively. The 4 graphs in the bottom row are plotted as a control condition for comparison. Cont = contralateral, Ipsi = ipsilateral, DLPFC = dorsolateral prefrontal cortex, ITG = inferior temporal gyrus, SM1 = primary motor and somatosensory areas, OFG = occipital fusiform gyrus.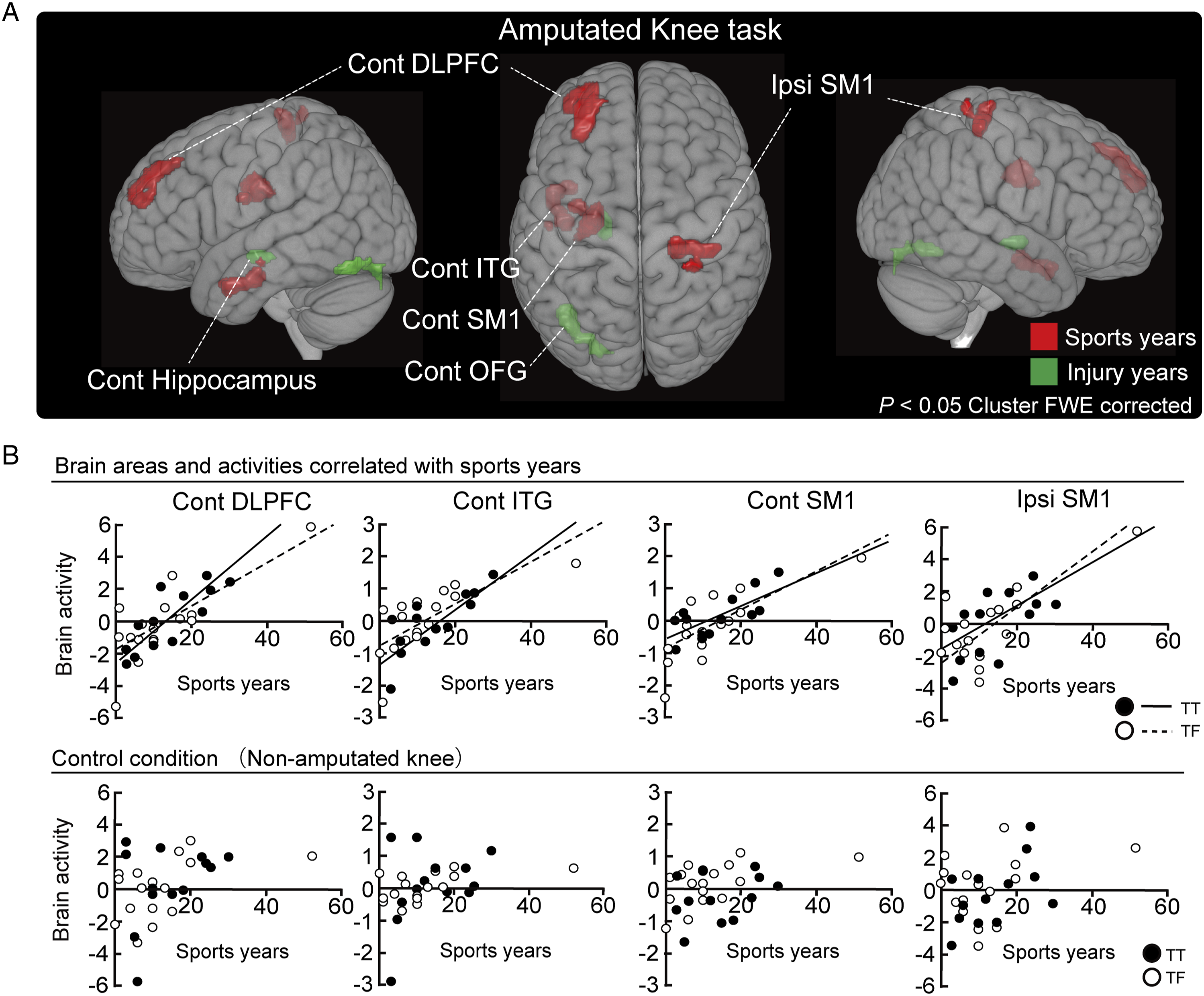
Activated brain regions during amputated knee contraction correlated with sports years and the result of VBM analysis.
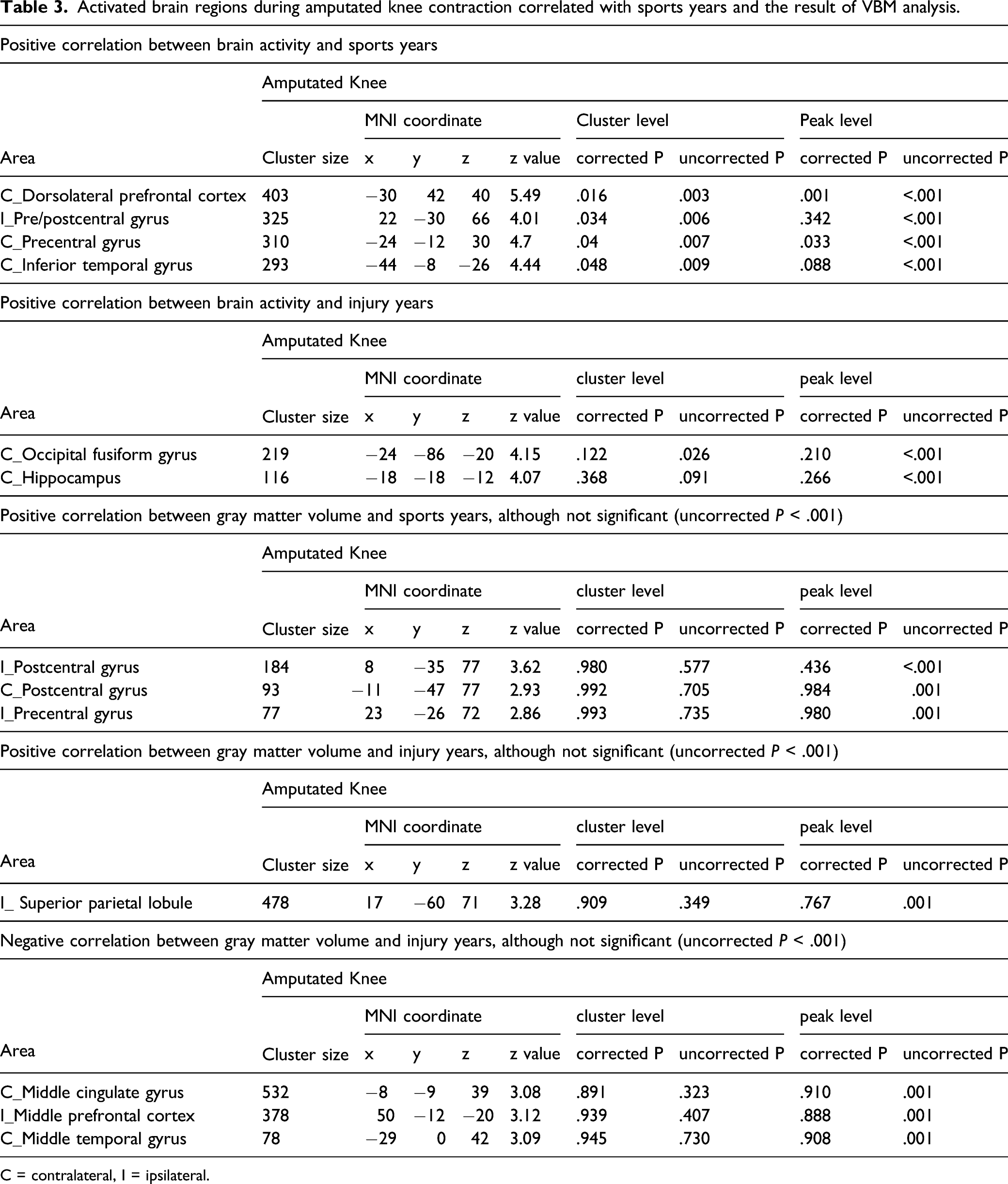
C = contralateral, I = ipsilateral.
GM Volume in Association With Sports and Injury Years
Table 3 summarizes the statistical results of VBM analysis. No significant results were obtained from the VBM analysis.
Muscle Activity During the Task
The results of the EMGs in the practice sessions showed that EMG of only the primary target muscle increased during the task, and contraction of other muscles was not observed in any participant (Supplementary file 1).
Discussion
This study suggested that long-term, routine para-sports participation after LLA can induce functional reorganization in the ipsilateral M1. Twelve of the 30 participants who had a relatively longer history of para-sports participation (median: 20.0 years) exhibited significant ipsilateral M1 activation during the amputated knee task. Additionally, the number of sports years was positively correlated with brain activity in the contralateral DLPFC, SM1, and ITG of the amputated limb during knee muscle contraction on the amputated side. No significant correlation was found in the structural analysis. Moreover, there were no differences between the amputated part, that is, TT and TF, of all results, likely because the thigh muscles in both TT and TF amputation are key in precisely controlling the prosthesis. These results indicated an interaction between amputation-induced and use-dependent brain reorganization in individuals with LLA who continue to participate in long-term para-sports.
Brain Activity During Contraction Tasks in LLA and Contribution of Prosthetic Sports Participation
The contralateral M1 was mainly activated during the ankle and knee task of the intact leg, consistent with the results of a similar evaluation in able-bodied individuals.13,31–33 Contralateral activity in the M1 is associated with the lateral CST. 32 The lateral CST originates from the contralateral M1 and is crucially involved in controlling the movements of the distal extremities.33–35 Therefore, contralateral M1 activation is assumed to contribute to unilateral limb contraction via the lateral CST. Compared with the non-amputated knee task, the amputated knee task showed higher activation in wider regions, including the contralateral M1, bilateral cerebellum, and bilateral operculum. Notably, the averaged image showed no significant brain activity in the ipsilateral M1 (Figure 1B). However, in the individual level (1st level) analysis, 12 of the 30 participants who showed significant activation in the ipsilateral M1 during the amputated knee task had a relatively longer history of para-sports participation (median sports years: 20.0 years). Contrastingly, the median sports years for the remaining 17 participants (no ipsilateral activation) was 6.5 years. The typical brain activities in Figure 2 suggested that the number of sports years was associated with activation of the ipsilateral SM1. Participants who had a relatively longer sports participation history and who are represented on the right side of each representative brain activation in Figure 2 showed significant ipsilateral SM1 activation. Conversely, participants who had a relatively shorter sports participation history and who are represented on the left side in Figure 2 showed only contralateral SM1 activation. All participants, on the left and right sides, who showed different activation patterns, were equal in age and injury years but differed in sports years. In the analysis of the correlation between sports years and brain activity, there was a significant correlation of sports years with brain activity in not only the hemisphere contralateral to the amputated limb but also in the ipsilateral SM1 (Figure 3A). This result was not observed in the control area (inverted x-coordinate of MNI locations) on the non-amputated side (Figure 3B), indicating that this functional reorganization was specific to the amputated parts. Although below the statistical threshold, the ipsilateral SM1 tended to adapt structurally and with increasing sports years (Table 3). Accordingly, it was inferred that long-term para-sports participation could induce functional and structural reorganization of the ipsilateral SM1 to improve the motor function of the amputated limb.
Bilateral M1 activity was observed for both the left and right hip tasks, which is a well-known attribute of the proximal muscles receiving stronger projections of the ipsilateral M1 than the distal muscles via the corticoreticular pathway.17,36 Moreover, activated areas during the amputated ankle imagery task differed from those during other tasks since no muscle contraction was involved. Consistent with previous findings, the DLPFC, SMA, and cerebellum were activated during motor imagery.28,37,38
Possible Mechanisms of Involvement of the Ipsilateral M1 for Amputated Leg Contraction
The neural mechanism underlying the involvement of the ipsilateral M1 in amputated leg contraction can be presumed in 2 steps. First, a decrease in GABA levels in the ipsilateral and contralateral M1 after mitigating interhemispheric inhibition caused by post-LLA-induced compensational plastic changes, as inferred from WM structural analysis by Li et al. 8 Reduced microstructure in the corpus callosum (inhibitory interhemispheric pathways) in LLA was reported, which could induce plastic changes in both hemispheres by decreasing GABA levels. GABA is considered an important inhibitory neurotransmitter39,40 with decreased GABA inhibition unmasking pre-existing synaptic connections and easily causing synaptic plasticity 40 ; therefore, this key transmitter for short-term expansion of cortical representation could induce long-term reorganization similar to that observed in the present study through intense amputated limb use. Hordacre and Bradnam 12 reported that in the M1 contralateral to the amputated side, there were decreased GABA receptors and mitigated interhemispheric inhibition during the first walk after LLA. Furthermore, there was an earlier reduction in GABA receptors, which mediated inhibition, in the M1 ipsilateral to the amputated side than in the contralateral M1. Therefore, LLA-induced interhemispheric inhibition could be mitigated by reduced GABA levels in the bilateral hemisphere, which would induce bilateral M1 reorganization.
The second step is that intense post-LLA use of the amputated limb promotes bilateral reorganization, which we termed the “sports-induced reorganization” step. Strength training could uniquely modulate GABA-mediated inhibition between hemispheres and ipsilateral corticospinal excitability in healthy subjects. 41 Even in an LLA case, Hordacre et al11,12 revealed enhanced ipsilateral M1 excitability of amputated limbs using TMS, accompanying the progression of acute-phase post-LLA prosthetic rehabilitation. Moreover, there were longitudinal changes in interhemispheric inhibition over time accompanied by changes in GABA levels during the acute rehabilitation phase. Our correlation analysis of injury years and brain activity did not show significant results in ipsilateral brain regions (Figure 3A), suggesting that para-sports training to be a necessary component for facilitation of the ipsilateral motor-related pathways. In summary, this evidence of adaptation after amputation suggests that, initially, altered microstructure in the corpus callosum, and decreased GABA levels occur in both hemispheres (ie, first step [injury-induced reorganization]) after amputation. Subsequently, continuing para-sports overcomes this reorganization as a natural response in the ipsilateral M1 (ie, second step [sports-induced reorganization]). It is important to note that GABA was not measured in our study; therefore, careful interpretation is needed. Future studies should clarify how GABA is altered during the second step of this adaptation.
Overall, this ipsilateral M1 functional reorganization was not seen in the able-bodied athletes in a similar experiment 13 and was considered to be a unique result that revealed new properties of human brain plasticity.
Reorganization in the Frontal Lobe and Visual Pathways Induced by Long-Term Sports Activities
Correlation analysis of sports years showed significant correlations with the ipsilateral SM1 as well as the contralateral SM1, DLPFC, and ITG. Reorganization in the contralateral SM1 appeared to be a natural consequence because it was previously reported that cortical expansion accompanies intense limb use and motor learning. 27 In contrast, although there is no direct anatomical connection between the DLPFC and M1,42,43 DLPFC activation decreases ipsilateral M1 corticospinal excitability.42,44 In fact, the DLPFC is known to be involved in motor control, for example, in force control 45 and motor memory maintenance, 46 which might have contributed to the task of regulating force to 20% MVC in this study. Conversely, both M1s inhibit the other M1 via transcallosal inhibition. 47 Therefore, we assumed that activation of the DLPFC specifically caused the suppression of the inhibitory function of the M1 on the contralateral side to the amputated side (ie, the same side as the DLPFC) and resulted in the disinhibition of the ipsilateral SM1.
The ITG is known as a core region of the ventral stream related to visual information processing. 48 Moreover, cortical plasticity of the ventral stream was also shown to accompany motor learning. 49 In fact, several studies have demonstrated that ITG hypertrophy and increased brain connectivity occur in able-bodied athletes.50,51 Therefore, it is possible that visuomotor learning was enhanced by the increase in sports years which in turn induced the reorganization of the ITG in the current study; however, this assumption is speculative, and further studies are needed.
Limitations of the Study
First, we used sports years as an index to quantify the approximate activity level over a long period of time. However, we did not assess physical activities in detail; therefore, careful interpretation of our results is required. Since activity level is an important point in the discussion of use-dependent plasticity, a future longitudinal study would be necessary to specifically identify the characteristics of individual physical activities, including the sports events, intensity, frequency, and other activity levels in daily life.
Second, we did not record the EMG during the MRI scan, although it was recorded and confirmed during the practice session, and all participants could contract the agonist muscle at 20% MVC without other muscle activity (Supplementary 1). Therefore, we cannot exclude the possibility of muscle activity differing between the MRI scan and the practice session. Without EMG monitoring, it is difficult to exclude the possibility that isometric contraction (which does not accompany joint movement) of proximal muscles (eg gluteus maximus or paraspinal muscles) influenced the ipsilateral M1 activity. However, we believe that activity level, at least in homologous muscles, was comparable as the participants performed the contraction similarly in both sessions. This was further confirmed by our intensive camera monitoring of their legs during the task.
Finally, we did not recruit age-matched able-bodied participants as controls. Therefore, this study alone cannot infer the possibility of long-term sports practice inducing ipsilateral M1 activation, irrespective of limb amputation. However, we previously measured able-bodied athletes and observed no ipsilateral M1 activation during the same contraction tasks. 13 This result could support our conclusion that both amputation and long-term para-sports are needed for further reorganization in the ipsilateral M1.
Conclusion
Our fMRI study revealed post-LLA functional brain reorganization in the ipsilateral M1. Specifically, 12 of the 30 participants who had significant activation in the ipsilateral M1 during the amputated knee task had a relatively longer history of para-sports participation. Moreover, participants with LLA who continued long-term para-sports also exhibited reorganization in the contralateral DLPFC and ITG. This indicates that long-term para-sports could lead to extensive reorganization at the brain network level, not only of the M1 but also of the frontal lobe and visual pathways. Our findings support the development of neurorehabilitation, including brain-targeted rehabilitation training or non-invasive stimulation after LLA.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211056660 – Supplemental Material for Para-Sports can Promote Functional Reorganization in the Ipsilateral Primary Motor Cortex of Lower Limbs Amputee
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683211056660 for Para-Sports can Promote Functional Reorganization in the Ipsilateral Primary Motor Cortex of Lower Limbs Amputee by Tomoya Nakanishi, Nobuaki Mizuguchi, Kento Nakagawa and Kimitaka Nakazawa in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgments
We would like to express our gratitude to the participants for taking part in this experiment. Furthermore, we would like to thank Advanced Bioimaging Support for their advice on MRI data acquisition and analysis techniques.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid for the Japan Society for the Promotion of Science (JSPS) Fellows to T.N. [grant number 19J21542], and a Grant-in-Aid for Scientific Research (A) from JSPS to K. Nakazawa. [grant number 18H04082].
Data Availability
The dataset generated and analyzed in this study will be available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
