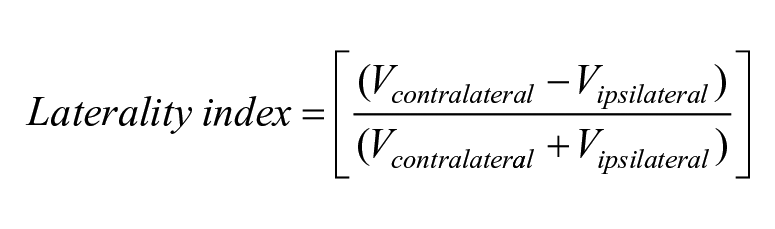Abstract
Background
Gross motor intervention designs for children with diplegic cerebral palsy (DCP) require an improved understanding of the children’s potential for neuroplasticity.
Objective
To identify relations between functional neuroplasticity and motor skill changes following gross motor interventions for children with DCP.
Methods
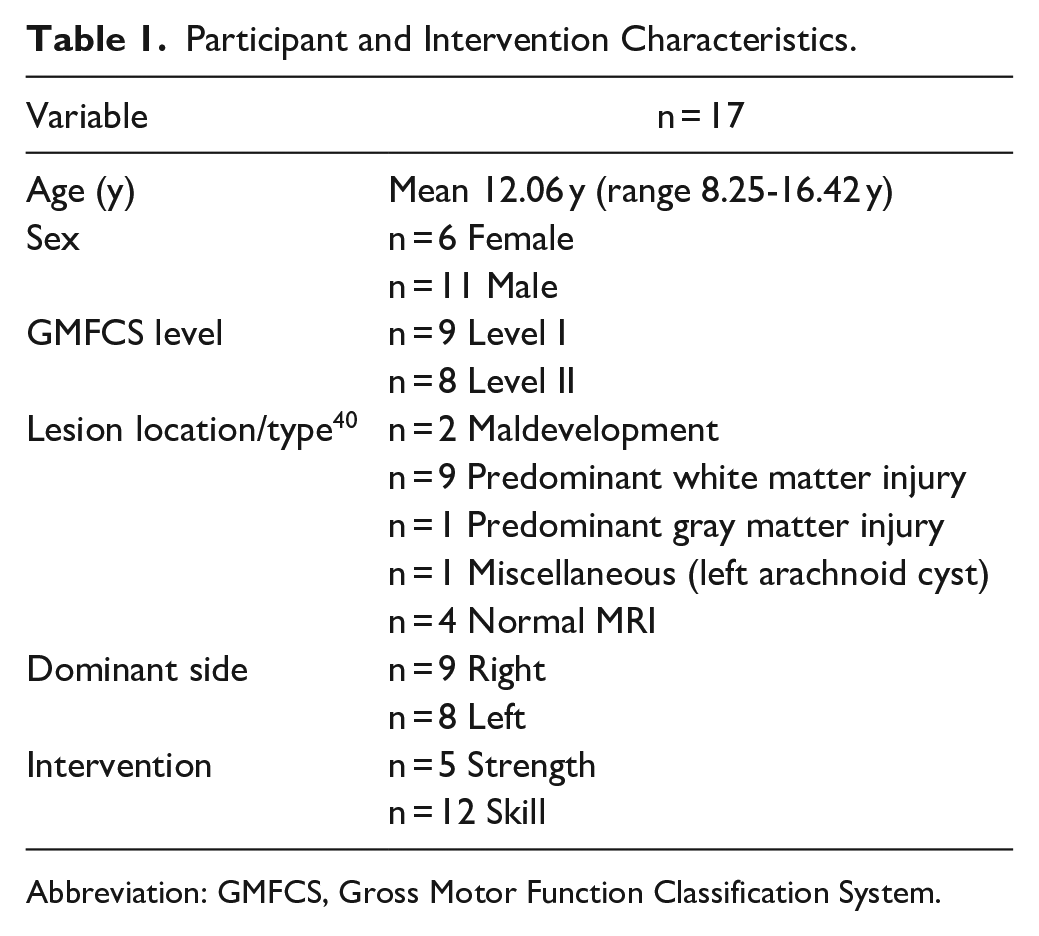
There were 17 participants with DCP (ages 8-16 years; 6 females; Gross Motor Function Classification System Level I [n = 9] and II [n = 8]). Each completed a 6-week gross motor intervention program that was directed toward achievement of individualized motor/physical activity goals. Outcomes were assessed pre/post and 4 to 6 months post-intervention (follow-up). An active ankle dorsiflexion task was completed during functional magnetic resonance imaging. The ratio of motor cortical activation volume in each hemisphere was calculated using a laterality index. The Challenge was the primary gross motor skill measure. Change over time and relations among outcomes were evaluated.
Results
Challenge scores improved post-intervention (4.57% points [SD 4.45], P = .004) and were maintained at follow-up (0.75% [SD 6.57], P = 1.000). The laterality index for dominant ankle dorsiflexion increased (P = .033), while non-dominant change was variable (P = .534). Contralateral activation (laterality index ≥+0.75) was most common for both ankles. Challenge improvements correlated with increased ipsilateral activity (negative laterality index) during non-dominant dorsiflexion (r = −.56, P = .045). Smaller activation volume during non-dominant dorsiflexion predicted continued gross motor gains at follow-up (R2 = .30, P = .040).
Conclusions
Motor cortical activation during non-dominant ankle dorsiflexion is a modest indicator of the potential for gross motor skill change. Further investigation of patterns of neuroplastic change will improve our understanding of effects.
ClinicalTrials.gov Registry:
NCT02584491 and NCT02754128
Keywords
Introduction
Functional neuroplastic change following gross motor training may provide important indicators that link with movement skill improvement for children with spastic diplegic cerebral palsy (DCP), yet the potential patterns of neuroplasticity are poorly understood. DCP is within the bilateral spastic CP subtype, 1 and is characterized by a non-progressive brain injury occurring early in development that primarily affects sensorimotor control of the lower limbs, 2 with distal control being especially challenging. 3 For independently ambulatory children, interventions typically focus on gross motor coordination, balance, and/or muscle strength. 4 While gross motor gains are observed, intervention effectiveness varies among children and there is no consensus on the optimal approach. 5 Gaining an understanding of the functional neuroplastic changes that are related to functional skill gains will help inform future innovations to gross motor interventions.
Cross-sectional studies have begun to map structural, microstructural, and functional neural characteristics of children with DCP, highlighting atypical sensorimotor connectivity and function.6 -9 Patterns of injury are highly diverse, with bilateral lower limb corticospinal tract (CST) projections from the primary motor cortex (M1) often involved. Typical CST connectivity is from the M1 in 1 hemisphere to the contralateral body side, yet in children and adults with DCP, lower limb projections may stem from either M1.9,10 In fact, cortical activation of M1 in both hemispheres has been observed during unilateral lower limb movement using functional near infrared spectroscopy (fNIRS). 11
The majority of neuroplasticity research in CP focuses on the upper extremity. Location of M1 activity is often assessed using a laterality index, which is the ratio of activity between the contralateral and ipsilateral M1s. 12 Evidence suggests that an increased ratio of contralateral M1 activity is associated with better functional outcomes of the affected arm for children with unilateral CP, at both pre and post-intervention. 13 Notably, better ankle control has been associated with contralateral sensorimotor cortical metabolism, assessed using positron emission tomography (PET) in a small and diverse group of children with DCP. 14 Whether increased contralateral activation is associated with lower limb improvements following gross motor interventions has yet to be investigated.
The paucity of interventional plasticity research for children with DCP limits understanding of potential change and subsequent identification of optimal intervention approaches. In apparently the first study of locomotor-based training and neuroimaging in exclusively children with DCP, laterality of M1 activation was not investigated. Children with DCP were randomized to train on either a pediatric elliptical or motor-assisted cycle. While improvements were observed in device cadence, there were no improvements observed in gait or functional mobility 15 and no significant associations were found between changes in device cadence and resting state functional connectivity, volumetrics, or white matter microstructure using MRI. 16 Still lacking is an examination of neuroplasticity accompanying significant gross motor function improvements.
In the last decade, increased research of upper limb plasticity in children with unilateral CP has driven therapy advances. Initial, small sample studies of upper extremity plasticity in hemiplegic CP suggested uniform responses.17 -19 Subsequent studies highlighted the need to examine individual data to identify different response patterns.20,21 Evidence-based upper limb therapy options have emerged from concurrent investigations of hand function and neuroplasticity.22,23 These therapies employ high doses that may not be attainable in a gross motor, lower extremity context as fatigue is a limiting factor. 24 However, there is now potential for augmentation of upper limb therapies using technologies such as non-invasive brain stimulation, with cortical stimulation targets originally informed by neuroimaging studies. Disappointingly, there have not been equivalent advances for lower limb interventions for children with DCP.
Assessment of lower limb M1 cortical activation and laterality index calculation is challenging, as the target area is small and located in the midline of the cerebral hemispheres. Candidate modalities to map this M1 area are transcranial magnetic stimulation (TMS), PET, fNIRS, and functional MRI (fMRI). TMS can be used to evaluate cortical “hotspots” and CST connectivity. The strength of TMS electrical pulses required to stimulate cortical leg regions is high, causing discomfort that can be prohibitive for use in children. PET requires fasting and injection, making this an uncomfortable and invasive procedure that is not approved for pediatrics at many institutions. In contrast, functional MRI (fMRI) and fNIRS are appealing as non-invasive and painless methods to indirectly assess cortical activation during voluntary movement. While the mobility of fNIRS is ideal, the spatial resolution is limited and restricted to superficial cortex. Promisingly, fMRI can more precisely map cortical areas of activation during unilateral lower limb movements.25 -28
To assess lower limb-linked M1 activity and plasticity in children with CP, we developed an ankle dorsiflexion task-based fMRI paradigm designed to examine both the laterality index and activation volume (statistically significantly activated voxels). 29 Functional expansion, defined as an increase in the overall activation volume, is associated with early skill acquisition in healthy adults 30 and increased potential for lower limb skill learning in adult stroke. 25 As skills are mastered in healthy adults, the volume of activation becomes smaller. 31 Whether similar patterns might be seen in children with DCP is unknown.
The primary aims of this study were to detail changes and determine associations in M1 activity (measured by the laterality index and activation volumes) and advanced motor skills following gross motor interventions for children with DCP. We hypothesized that increased contralateral activity (as measured by the laterality index) would be associated with motor skill gains. The secondary aim was to determine whether M1 laterality or activation volume predicted motor gains at post-intervention (predictors: baseline M1 values) or follow-up (predictors: post-intervention M1 values).
Methods
In this prospective study, fMRIs and motor skills assessments were completed within a 10-day target window prior to the start of the gross motor intervention (baseline) and again after intervention completion (post-intervention). An additional assessment 4 to 6 months after intervention completion (follow-up) included only motor outcomes. This study was approved by the local institutional research ethics boards and registered on clinicaltrials.gov (NCT02584491 and NCT02754128). Informed consent/assent was obtained from participants and caregivers.
Participants
Children were recruited through 3 local pediatric rehabilitation centers, and were enrolled in 1 of 2 clinical intervention trials described below. Eligible children: (1) had a clinical diagnosis of spastic DCP; (2) were classified in Levels I and II of the Gross Motor Function Classification System (GMFCS) 32 ; (3) were aged 8 to 17 years; (4) were able to independently dorsiflex both ankles with a SCALE score 33 of ≥1; and (5) were able to cooperate with a MRI scan, determined in a screening visit. Exclusion criteria were: (1) standard MRI contraindications (eg, dental braces); (2) botulinum toxin injections within 4 months prior to study entry; (3) surgery within last 9 (muscle) or 12 months (bone); or (4) uncontrolled seizure disorder. A screening visit determined study eligibility, including practice of the MRI tasks in an MRI simulator to ensure compliance with scanning.
Interventions
Participants were enrolled in a lower extremity strength training or sports skill training intervention. 34 In brief, the lower extremity strength training intervention involved progressive functionally based muscle resistance exercises that could be single-joint, multi-joint, or bilateral while the sports skill intervention involved lower limb fundamental movement skill training such as running, jumping, and kicking. Each manualized 6-week intervention involved 16 one-to-one 45-minute sessions, with the motor task challenge progressively increased. To promote skill gains, the intervention dose and frequency were higher than standard care. Programs were individualized to focus on baseline goals for gross motor function/skills. Identical training methods were used for each leg, targeting symmetry of movement thus leading to increased use of the more affected side. All interventions were designed and delivered by interventionists trained at Holland Bloorview Kids Rehabilitation Institute. Further intervention details are reported elsewhere. 34
Motor Skill Assessments
Assessments were completed by study-trained physiotherapists blinded to scores from any previous assessments within this study. The primary motor outcome was the Challenge, a validated assessment of advanced gross motor skills designed specifically for children in GMFCS Level I and II that jointly considers accuracy and speed of performance.35,36 Test administration time is 45 to 60 minutes, completed on a 10-m “track.” Example items are running quickly with an exact stop at the end of the track, foot-to-foot dribbling a soccer ball down the track, and weaving between pylons while walking or running. Children complete 3 trials of each item. On the first trial, the child should be as accurate as possible. Two more trials are conducted to provide opportunity to improve accuracy, or if they did it perfectly, to try to speed up while staying accurate. Percentage scores reported are derived from the sum of “best trial” scores of 3 trials of its 20-items. Each item is scored on a 0 to 4 scale that first considers performance accuracy (scores of 0-2). If accuracy targets are reached (ie, score >2), performance time is rated (scores of 3 and 4). Participant (or caregiver-proxy) goals were identified using the Canadian Occupational Performance Measure (COPM) and its 1 to 10 rating scale to evaluate self-perceived performance (COPM-P). 37 All measures have been validated with children with CP, with excellent test–retest reliability (ICC >.75). Minimum detectable change is 4% to 6.5% points for the Challenge,35,36,38 with a minimally clinical important difference of 4.5% points, 36 while for the COPM, a 2 point difference is considered clinically important. 37 The dominant leg was determined using an established ball kicking task, where the leg used to kick the ball was identified as the dominant leg (and ankle). 39 Three trials were completed to assess consistency.
Image Acquisition
Imaging data were acquired on a 3T Siemens Trio MRI scanner with a 12-channel head coil. Acquired images included a high-resolution T1-MPRAGE (1 mm iso; TR/TE/TI/FA = 2300/2.96/900/9), a T2-weighted image (1.2 mm iso; TR/TE/FA = 9000/104/120), and an EPI task fMRI (19 oblique axial slices; 3 mm iso; TR/TE/FA = 1000/30/60). Axial slices were positioned to cover the motor cortex.
Visual stimuli for functional sequences were programmed using the software Presentation (Neurobehavioral Systems Inc., Berkeley CA, USA) and were displayed to participants using goggles or a mirror. The ankle dorsiflexion task sequence followed previously reported methods with an event-related design. 29 A visually presented “+” cued a single ankle dorsiflexion movement, while “O” indicated rest. In a pre-scan practice, children trialed the dorsiflexion movement in a mock scanner. Using a purpose-built wooden apparatus, 1 foot was securely fastened to a hinged pedal using Velcro straps. A foam wedge supported the calf, thigh, and knee in a flexed position. The apparatus limited dorsiflexion movement amplitude to 5°. An elastic band provided resistance to pedal movement. To normalize subjective task difficulty, resistance was individualized for each ankle so that a 5° dorsiflexion could be completed without knee, hip, or head movement. Resistance was constant between pre/post scans. 29 Children completed the unilateral dorsiflexion task on the dominant and non-dominant side.
Image Preprocessing
T1- and T2-weighted images were reviewed by a pediatric neuroradiologist, blinded to clinical presentation, to report type and location of brain injury. The MRI classification system was used to classify injury types. 40
Image preprocessing was completed using a combination of FSL (www.fmrib.ox.ac.uk/fsl), FreeSurfer (https://surfer.nmr.mgh.harvard.edu/), AFNI (https://afni.nimh.nih.gov/), and Matlab (Mathworks Inc., Natick MA, USA) software. Datasets were first aligned to dominant leg side, using results from the ball kicking task. 39 FSL’s Brain Extraction Tool was used to remove non-brain tissue from the T1-weighted image, which was then segmented using the Freesurfer recon-all tool to extract ventricle and lesion masks in Talairach space. Masks were then transformed back to native space, manually edited as required, and inverted. The masks were inputted in FSL’s FNIRT tool (ie, –inmask), which was used to non-linearly register structural and functional data to the Montreal Neurological Institute (MNI) 152 2 × 2 × 2 mm template.
fMRI data were corrected for slice timing and motion, registered to the MNI 152 2 × 2 × 2 mm template, spatially smoothed using a 6-mm full width at half maximum Gaussian kernel, and then baseline averaged. A maximum Euclidean displacement metric was calculated from the 6 rigid body transformation parameters for each volume.
Task fMRI Data Analysis
fMRI datasets were considered usable if: (i) mean maximum head movement (all planes) was ≤3 mm; (ii) >50% of volumes aligned with gamma convolved stimuli were motion-free; and (iii) correlations of head motion and stimuli were r < |.20|. Volumes with head motion >5 mm were censored from the dataset. For acceptable images, the convolved task stimulus file was inserted as an explanatory variable into FSL’s FEAT. Activation maps were thresholded at z > 2.3 with a corrected cluster significance threshold of P = .05.
Analyses followed previously reported methods. 29 In summary, for calculation of primary outcomes of laterality index and volume, images were thresholded at 70% of the maximum z-score within the bilateral lower limb M1 region of interest (ROI). Clusters within the ROI were selected using a 2-step process based on cluster maxima/center of gravity values. First, values were required to be within MNI coordinate ranges of x = 20 to −20; y = −42 to −2; z = 36 to 84. Secondly, clusters were required to be within the precentral gyrus (motor cortex) of the Harvard Oxford Cortical Structural Atlas. 41 Selected data were binarized into 1 mask to calculate: (1) volume, defined as the total number of active voxels (V) in the M1 in both hemispheres; and (2) laterality index to quantify the number of active voxels in each hemisphere, with the midline voxel row excluded to avoid misclassification across midline. The laterality index was calculated as:
Laterality index scores vary from −1.0 to +1.0, with positive scores indicating greater contralateral activity. 12
Statistical Analyses
SPSS (version 24; IBM, Armonk, NY, USA) was used for statistical analysis. All analyses were intent-to-treat. Data were described (eg, means, SDs) and change scores summarized per outcome (Change 1: post minus pre; Change 2: follow-up minus post). Descriptive data of individual fMRI change values were examined for patterns by means of visual inspection of scatter and line plots.
Change in group (aggregate) fMRI outcomes over time were assessed using paired t-tests (normal distributions) or Wilcoxon Signed-Rank tests (non-normal). Repeated measures analyses of variance were used to assess motor skill outcomes over time, with post-hoc tests Bonferroni corrected for multiple comparisons. Mann–Whitney U tests compared scores between intervention groups to determine if there was a group effect.
The primary aim of exploring the relations between changes in fMRI (laterality index, volume) and changes in motor skill measures (Challenge, COPM-P) was examined using pairwise correlations. Correlations were evaluated using Pearson’s correlation (r), or Spearman’s rank order coefficient (rs) for non-normal distributions. For this analysis, correlations with both |r| > .40 and P < .05 (uncorrected) were considered significant. 42
The secondary aim of identifying fMRI predictors of Challenge change was examined using 2 linear regression analyses: (1) baseline fMRI measures (LI, volume) were included as predictors of Challenge change from baseline to post-intervention (Change 1); and (2) post-intervention fMRI measures were included as predictors of Challenge change from post-intervention to follow-up (Change 2). The forward regression procedure used probability of F < .05 (variable entry) and F > .10 (removal).
Results
Seventeen participants met study eligibility criteria. Participant and intervention characteristics are detailed in Table 1. Each child completed his/her intervention as per protocol. There were no significant differences between intervention groups for baseline or change scores (minimum P > .160). Interventions were individualized based on COPM goals, which were relevant to the fMRI ankle dorsiflexion task. Example goals: (1) improve balance on left leg to improve push-off, weight bearing, and gliding while on skateboard; (2) improve ability to stop and change direction at high speeds without falling; (3) improve soccer skills to steal ball from opponent with better foot skills and speed.
Participant and Intervention Characteristics.
Abbreviation: GMFCS, Gross Motor Function Classification System.
In total, collection of 66 functional images was attempted (non-dominant and dominant ankle for 17 participants at baseline and 16 participants at post-intervention). All participants were successfully imaged for at least 1 fMRI session. At each fMRI measure time point, data from 14 to 17 participants were available. One participant’s post-intervention MRI scan was not completed due to dental braces. All other missing data were due to image quality (excessive head movement) in 3 participants consisting of: non-dominant ankle task fMRI data at baseline (n = 2) and post-intervention (n = 2); and dominant ankle data at post-intervention (n = 2). These poor-quality data (n = 6) account for 9% of data collected.
Follow-up motor skill data consisted of 15 participants. One other participant had surgery and a second moved out of the country prior to the assessment. Table 2 reports scores for each measure.
Assessment and Change Scores.
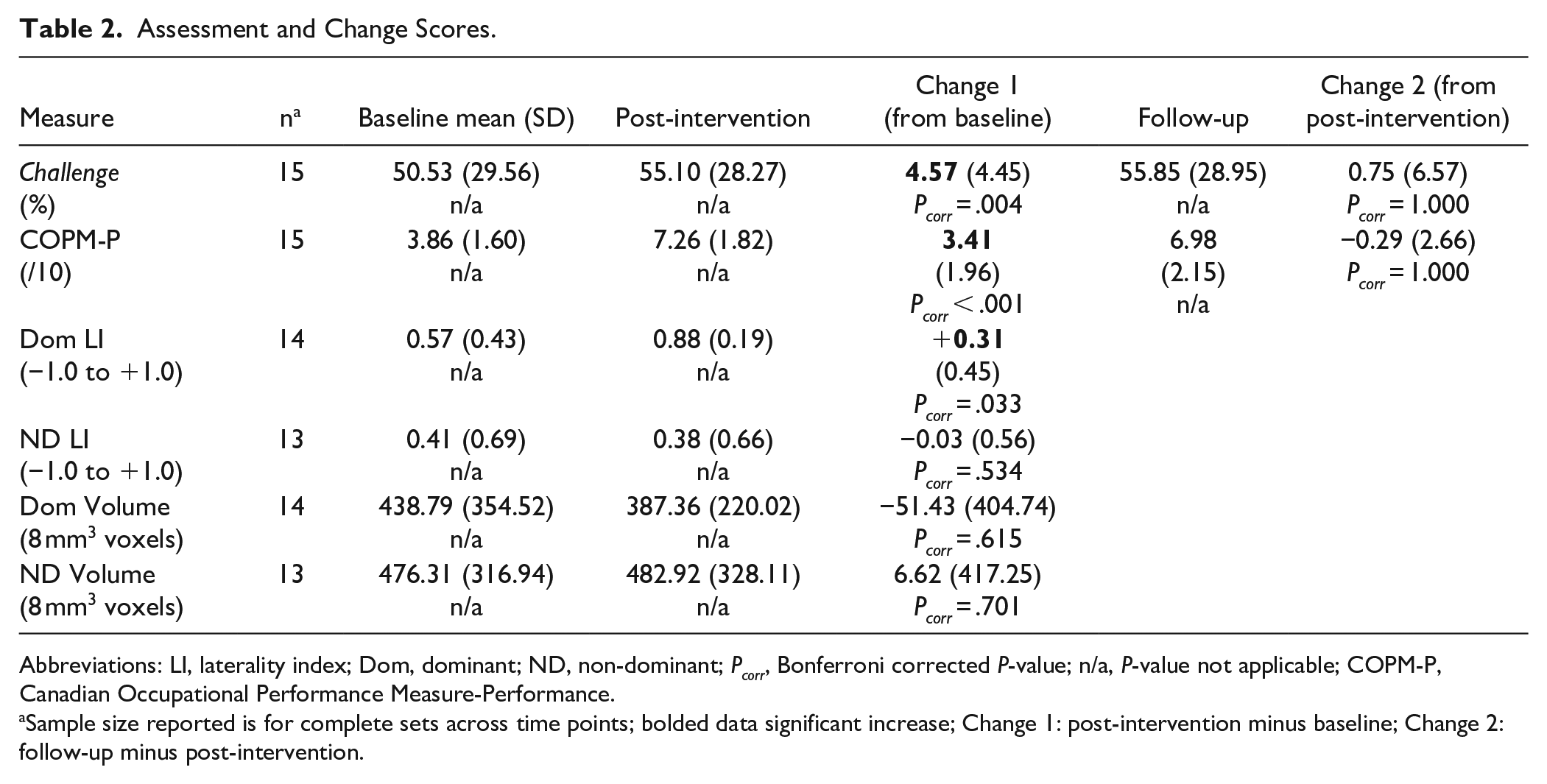
Abbreviations: LI, laterality index; Dom, dominant; ND, non-dominant; Pcorr, Bonferroni corrected P-value; n/a, P-value not applicable; COPM-P, Canadian Occupational Performance Measure-Performance.
Sample size reported is for complete sets across time points; bolded data significant increase; Change 1: post-intervention minus baseline; Change 2: follow-up minus post-intervention.
Neuroimaging Results
As seen in Figure 1, three patterns of individual (within child) task fMRI changes in dominant and non-dominant ankle laterality index were observed: (1) maintenance of contralateral activation at laterality index ≥+0.75 in 6/13 (non-dominant ankle) and 6/14 (dominant ankle) participants (see Figure 1, pattern 1 in green); (2) negative change, that is, increased ratio of ipsilateral activation, in 5/13 (non-dominant ankle) and 1/14 (dominant ankle) participants (see Figure 1, pattern 2 in red); and (3) positive change, that is, increased ratio of contralateral activation, in 2/13 (non-dominant ankle) and 7/14 (dominant ankle) participants (see Figure 1, pattern 3 in blue).
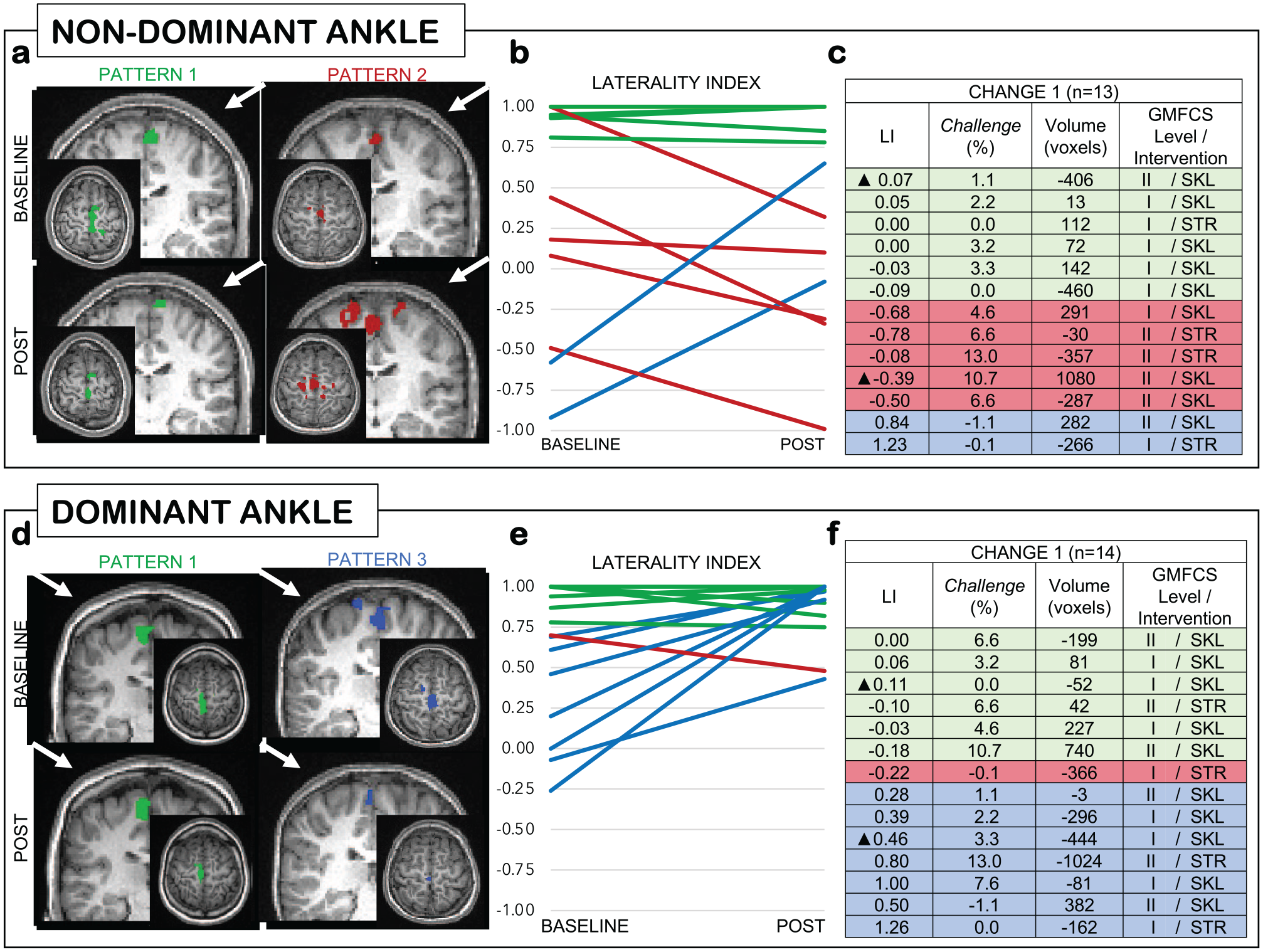
Individual patterns of task fMRI change. (A-F) Pattern 1 in green: maintenance of contralateral LI ≥+0.75; Pattern 2 in red: negative LI change (increased ipsilateral activation); Pattern 3 in blue: positive LI change (increased contralateral activation); A/d: white arrow indicates contralateral side; C/F: ▲ indicates MRI data presented.
Most participants (12/14) had contralateral dominant ankle activation at baseline (range −0.26 to +1.00) and post-intervention (range +0.43 to +1.00). The non-dominant ankle laterality was more variable at baseline (range −0.92 to +1.00) and post-intervention (−0.99 to +1.00). Of the children with negative laterality index change for the non-dominant ankle, 4 of 5 were in GMFCS Level II.
Change Score Correlations
The primary aim of analyzing associations between changes in fMRI outcomes and motor skills revealed 1 significant pairwise relationship. Change 1 scores of non-dominant ankle LI and Challenge were negatively correlated (r = −.56, P = .045), indicating that a shift toward ipsilateral activation for the non-dominant ankle was associated with greater Challenge improvement. See Figure 2. Table S1 provides details of non-significant findings.
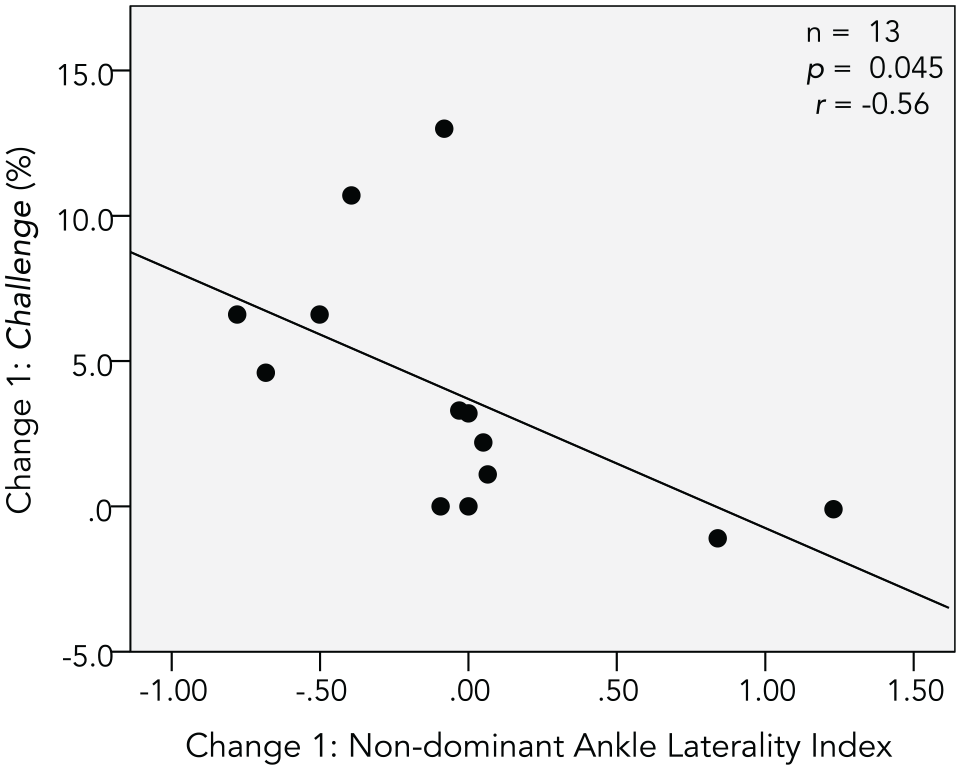
Significant Change score correlations.
FMRI Predictors of Motor Skill Change
For the secondary aim of identifying fMRI predictors of motor skill change, a smaller activation volume during non-dominant ankle dorsiflexion at post-intervention significantly predicted Challenge Change 2 scores (F(1,11) = 5.59, P = .040, adjusted R2 = .30, standardized B = −0.60). See Figure 3. Tables S2 and S3 provide details of non-significant findings.
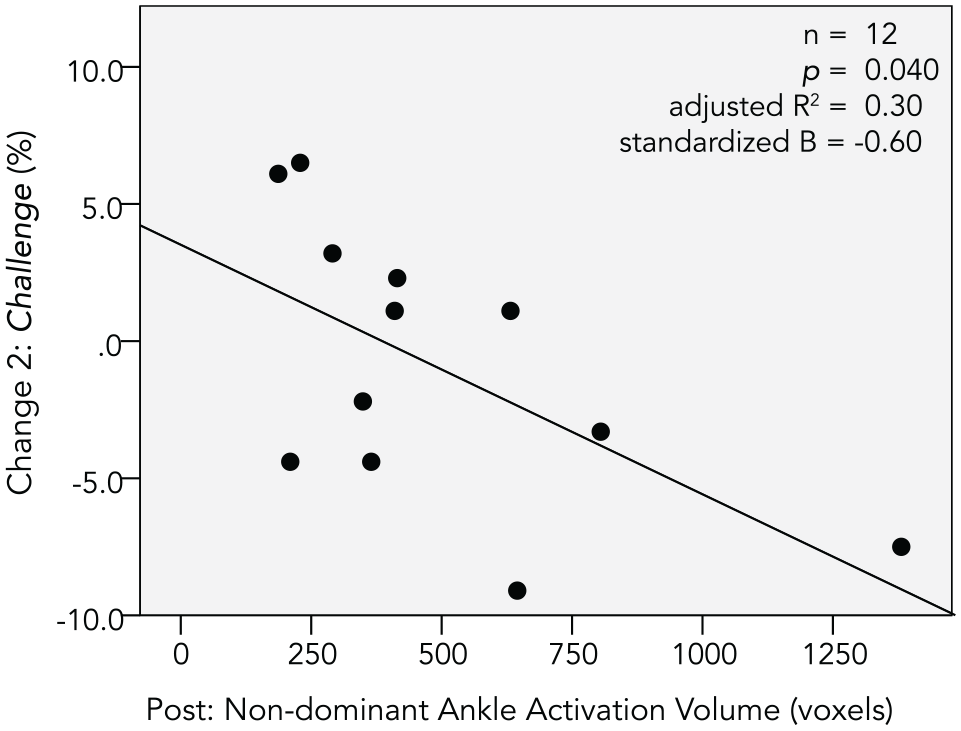
Significant fMRI predictor of motor skill change.
Discussion
To our knowledge, this is the first study with a focus on children with DCP to use task fMRI to examine changes in lower limb M1 functional activation following a gross motor intervention. Significant improvements in motor skills were observed, supporting subsequent analyses of associations of changes with fMRI outcomes.
Three individual patterns of laterality index changes were observed: increased ipsilateral activation (negative laterality index), increased contralateral (positive laterality index), and maintenance of high contralateral activation (laterality index ≥0.75). The most common responses to training were maintenance of or increases in contralateral activation, observed for 13 (of 14) dominant ankle and 8 (of 13) non-dominant ankle laterality index changes. As hypothesized, a significant increase in contralateral activity following intervention was observed for the dominant ankle. However, changes in non-dominant ankle laterality and changes in activation volumes were not significant. It is noteworthy that dominant ankle activations were more contralateral and less variable than the non-dominant side at baseline and post-intervention.
Increased ipsilateral activity for the non-dominant ankle was observed in 5 of 13 participants following intervention and was moderately associated with motor skill gains (Challenge). This was the only significant relation between fMRI outcomes and motor skill changes. One predictor of motor skill change was identified: a smaller activation volume during non-dominant ankle dorsiflexion at post-intervention was a predictor of higher Challenge change scores from post-intervention to follow-up. These significant relations observed between changes in non-dominant ankle motor cortical activity and gross motor skills suggest that the non-dominant side limits bilateral skill performance. Asymmetries in sensorimotor function, weight-bearing, and gait kinematics have been previously observed,43 -45 highlighting a need for non-dominant leg training. However, lower limb movements require bilateral coordination and interdependence, thus a unilateral training focus is not functional. Through targeted active involvement of both legs, the interventions in this study may have supported bilateral skill gains and neuroplastic change.
Results suggest that increased ipsilateral cortical activation during non-dominant ankle movements should not be discouraged for at least a subset of children with DCP. These results are concordant with recent findings that ipsilateral CST connectivity does not limit upper limb skill improvement in children with unilateral CP. 46 This atypical connectivity was previously thought to be maladaptive, but could also be a successful compensatory strategy after brain injury. For children with DCP, upregulation of cortical activity in both hemispheres may be beneficial as interest builds in using non-invasive brain stimulation in this population.47 -50 Further evidence is required to support this approach.
The predominant laterality index patterns of maintenance or increases in contralateral activation suggest that gross motor training can reinforce typical contralateral projections for both legs. 22 This was true for both interventions in this study, with no group differences observed between lower extremity strength training or sports skill training. Interestingly, laterality index change was also not associated with changes in activation volume, meaning that shifts in location of cortical activity cannot be explained by increased cortical recruitment. It is possible that patterns of change were driven by underlying CST connectivity that could be further investigated using diffusion tensor imaging. 51 A recent study in DCP did not find post-intervention changes in diffusion outcomes. However, this may be explained by the parallel lack of significant improvements in motor outcomes. 16 While the likelihood of white matter plasticity over the short-term is debated, white matter microstructure improvements have been demonstrated following intensive upper extremity training in children with CP. 52 A larger sample powered for subgroup analyses of each laterality index pattern would enhance understanding of the neuroplastic changes and relations with gross motor skills.
The significant relationship between increased ipsilateral activation for the non-dominant ankle and gross motor improvement was not expected. Of the 5 children with a shift toward ipsilateral cortical activity, 4 were in GMFCS level II. Higher GMFCS levels have been associated with greater structural damage in the brain, 53 decreased cortical volume, and poorer white matter microstructure (fractional anisotropy) within bilateral CST and corpus collosum. 54 Such damage may limit the potential to rewire unilateral lower limb control within 1 hemisphere, especially the non-dominant hemisphere. Interestingly, bilateral M1 CST connections in DCP may be present but not always engaged in functional activation. 9 Training may have unmasked existing bilateral CST connections, 30 thus supporting functional activation changes through previously unused ipsilateral pathways. For some children, it is also possible that the increased sensorimotor processing required for motor learning may necessitate dominant hemisphere contributions for improvements in non-dominant leg skills. Such bilateral CST connectivity is postulated to support fine motor learning of the affected hand in children with unilateral CP, 55 and has been observed using fNIRS during lower limb movements in children with DCP. 56
Regression analysis indicated that a smaller non-dominant ankle activation volume at post-intervention was a predictor of higher Challenge Change 2 scores (follow-up minus post-intervention). A small activation volume is associated with task mastery and a plateau in performance gains in healthy adults, 17 while in adult stroke, a large volume of activation has been hypothesized to indicate potential for continued lower limb skill learning. 25 Our results suggest that a large activation volume at post-intervention may be a flag that continued training is required to prevent loss of gains. Mean change was not significant from post-intervention to follow-up, however 6 participants showed a decline in motor skills of −2.2% to −9.1%, 3 of whom had the largest volumes at post-intervention. It is possible that a large volume of non-dominant ankle activation post-intervention indicates that continued training is required to consolidate skill learning to prevent losses. Without consolidation, children may not incorporate new skills into their daily repertoire. In this study, children with large activation volumes may not have received a sufficient therapy dose.
The motor skill and fMRI response variability in the current study may reflect different dose needs across participants. Ultimately, the goal of interventions should be to achieve targeted gains while at the same time minimizing the dose that is required for a child to realize these gains. Recommendations for upper limb training call for high practice dose and frequency, 57 but the schedule may dilute individual responses 58 and add unnecessary burden for children who are best responders. In the context of gross motor interventions, achieving a similar minimum dose as recommended for upper limb training (ie, >60 hours) 57 may require reduced session frequency and a shift to a longer intervention program duration, as fatigue is an identified factor during sub-maximal gross motor activities for children with CP. 24 It is clearly necessary to establish realistic recommendations for gross motor intervention dose and frequency, in conjunction with investigations of predictors of motor skill gains to individually tailor training dose. Continued neuroimaging research will inform the value of imaging outcomes as predictors to consider for children with DCP within a multivariate International Classification of Functioning, Disability and Health (ICF) 59 -encompassing model.
It is a challenge to have sufficiently powered neuroimaging studies in CP. Contributing factors include poor data quality, missing data, and brain lesions that can distort neuroimage pre-processing and analyses. Acknowledging these challenges, our group previously systematically tested and refined methods for neuroimaging data collection and analysis in children with CP. 29 Our data loss of <10% provides an estimate to inform recruitment targets for future neuroimaging studies. Recruiting as homogeneous a population as possible in CP may also benefit future research. Research trials often group children with DCP and unilateral CP together, however the different clinical and brain injury profiles of these 2 groups 60 increases heterogeneity for neuroimaging analyses. 61 Recent cross-sectional imaging studies in DCP have included participants in GMFCS Levels I to III 11 or I to IV, 14 and have included children and adults. In comparison, our study had narrow inclusion criteria yet still had diverse injury types, including 4 participants with normal MRIs, estimated to found in less than 20% of children with a clinical diagnosis of CP. 40 Indications of relations of brain lesions with motor function and gait suggest that analyses of structural images may also prove useful. 53 Ideally plasticity profiles would be created for each injury type, which would require multi-center studies to achieve required sample sizes. 61
Limitations
The sample size and missing data are acknowledged limitations in this longitudinal study. To enhance reliability of task fMRI data collection and analyses, task parameters were kept constant for each scan.29,62 Following these same strategies, stable cortical activation patterns during ankle dorsiflexion have been reported in adult stroke. 28 Misclassification of leg dominance is possible, yet similarity in dominant side laterality index values and patterns of change supports the assigned classifications. Future work could investigate whether dominant and non-dominant assignment correlates with motor impairment using a measure such as the SCALE. 33 Our study did not include imaging at the long-term follow-up, which may have provided insight into cortical activation patterns that support maintenance or continued motor skill gains. Finally, we did not systematically collect data on botulinum toxin use or the specific timing for the most recent injection (if one was received), and therefore were not able to account for this as a potential confounding variable in the analyses. However, we required participants to be 4 months post-injection and would expect any effect at this time to be minimal.
Future Directions
We found 3 different patterns of outcomes in this longitudinal study; this was not expected and hence the study was underpowered to support subgroup analyses. Thus, investigations of the patterns of change and of injury type are identified as high priority for next steps in moving this research forward. While this first exploration of functional plasticity was hypothesis-driven with a focus on M1, changes in other areas of the motor network may also relate to gross motor skill development and be the focus of future studies. Further, complex relations of change have been highlighted in CP, 42 and diverse individual factors that may not have been captured in our measures could be included in future work.
Conclusions
This is the first study to specifically focus on task-based functional neuroplasticity in association with gross motor skill improvements following gross motor training for independently ambulatory children with DCP. Results indicated that increased ipsilateral activity during non-dominant ankle motor cortical activation is associated with motor skill change, and that a smaller volume of activation during non-dominant ankle dorsiflexion at post-intervention predicted continued movement gains 4 to 6 months post-intervention. These differing patterns of neuroplastic change would benefit from further investigation in a larger population.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683221143503 – Supplemental material for Functional Neuroplasticity and Motor Skill Change Following Gross Motor Interventions for Children With Diplegic Cerebral Palsy
Supplemental material, sj-pdf-1-nnr-10.1177_15459683221143503 for Functional Neuroplasticity and Motor Skill Change Following Gross Motor Interventions for Children With Diplegic Cerebral Palsy by Alicia J. Hilderley, F. Virginia Wright, Margot J. Taylor, Joyce L. Chen and Darcy Fehlings in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank the participants and families for their time, energy, and support of this research. We also thank clinicians and research team members involved in this study, including Gloria Lee and Marlee Vandewouw.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a Pedal-with-Pete grant from the Amercian Academy for Cerebral Palsy and Developmental Medicine (AACPDM), the Holland Bloorview Kids Rehabilitation Hospital Foundation Chair in Paediatric Rehabilitation and was conducted as part of the Childhood Cerebral Palsy Neuroscience Discovery Network (CP-NET) of the Ontario Brain Institute (OBI). Funding from OBI partially paid for MRI fees. Funders were not involved in study design, data analysis, manuscript preparation, or publication decisions. The authors have stated that they had no conflicting interests.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.