Abstract
Background. An ischemic stroke is followed by the remapping of motor representation and extensive changes in cortical excitability involving both hemispheres. Although stimulation of the ipsilesional motor cortex, especially when paired with motor training, facilitates plasticity and functional restoration, the remapping of motor representation of the single and combined treatments is largely unexplored. Objective. We investigated if spatio-temporal features of motor-related cortical activity and the new motor representations are related to the rehabilitative treatment or if they can be specifically associated to functional recovery. Methods. We designed a novel rehabilitative treatment that combines neuro-plasticizing intervention with motor training. In detail, optogenetic stimulation of peri-infarct excitatory neurons expressing Channelrhodopsin 2 was associated with daily motor training on a robotic device. The effectiveness of the combined therapy was compared with spontaneous recovery and with the single treatments (ie optogenetic stimulation or motor training). Results. We found that the extension and localization of the new motor representations are specific to the treatment, where most treatments promote segregation of the motor representation to the peri-infarct region. Interestingly, only the combined therapy promotes both the recovery of forelimb functionality and the rescue of spatio-temporal features of motor-related activity. Functional recovery results from a new excitatory/inhibitory balance between hemispheres as revealed by the augmented motor response flanked by the increased expression of parvalbumin positive neurons in the peri-infarct area. Conclusions. Our findings highlight that functional recovery and restoration of motor-related neuronal activity are not necessarily coupled during post-stroke recovery. Indeed the reestablishment of cortical activation features of calcium transient is distinctive of the most effective therapeutic approach, the combined therapy.
Introduction
Ischemic injuries within the motor cortex result in functional deficits that may profoundly alter patients’ quality of life. Survivors are often chronically impaired with long-term disability. 1 To regain sensory and motor functions after stroke, spared neural circuits must reorganize.2-4 Multiple strategies have been developed to enhance neural rewiring which dramatically improved functional recovery 4 including pharmacological treatment, motor training, and brain stimulation. Among them, cortical neuromodulation techniques, such as transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tDCS), represent a promising non-invasive approach to improve cortical remapping. Nevertheless, these treatments can induce diffuse and non-specific activation in mixed neuronal populations,5-7 revealing the necessity for more targeted therapies. With the emergence of optogenetics, specific neuronal populations can be activated or inhibited achieving high temporal and spatial precision.8-11 Recently, optogenetics has been proficiently used to selectively modulate the excitatory/inhibitory balance of brain circuits affected by a stroke lesion.12,13 Repeated optogenetic neuronal stimulation of the ipsilesional hemisphere induced a significant improvement in neurovascular coupling response. 14 Furthermore, chronic optogenetic stimulation of the entire cortical mantle promoted behavioral recovery associated with the formation of new and stable thalamocortical synaptic boutons. 15 Nevertheless, information on the remapping of motor representation and motor-related cortical activation following optogenetic stimulation is largely unexplored. Further, no study investigated how stimulation-induced cortical remapping correlates with functional recovery.
When combined with motor training, cortical stimulation creates a pro-plasticizing milieu where spared neurons are more susceptible to experience-dependent modifications.4,16,17 Though several studies investigated the effect of combining neuronal modulation, such as TMS18-20 and tDCS,21-23 with robotic training, results are contradictory. Up to now, whether the combination of ipsilesional neuronal stimulation and physical training promotes motor recovery is still unknown. Further, though it is established that the combination of neuronal modulation and motor training plays a key role in post-stroke recovery, no investigation has yet addressed if possible physical improvement is supported by alterations in motor maps and in the distributed motor-related cortical activation.
Here, we designed a light-based stimulation protocol of peri-infarct excitatory neurons as a rehabilitative approach to achieve functional recovery, measured in a behavioral test different from the rehabilitation task.
Our hypothesis is that combination of optogenetic stimulation of peri-lesioned cortex and repetitive motor training of the affected forelimb boosts post-stroke recovery by promoting the establishment of a new excitatory/inhibitory balance between hemispheres. To dissect cortical remapping in motor-related neuronal activity in peri-infarct area, we took advantage of wide-field fluorescence imaging over the affected hemisphere. We found that longitudinal optogenetic stimulation restored forelimb functionality whereas motor-related functional activity did not recover. However, coupling optogenetic stimulation with longitudinal motor training of the impaired forelimb on a robotic platform halved the time required for a full recovery of forelimb function compared to optogenetic stimulation only. Furthermore, the rapid behavioral recovery was associated with the restoration of temporal features of calcium transient such as peak amplitude and slope. The analysis of motor-related activation maps in mice with combined therapy identified the peri-infarct area as the region of the cortex mostly involved in motor task. Finally, the combined treatment promoted the restoration of an interhemispheric balance between the two hemispheres, revealed by an increase of expression of Parvalbumin positive cells in the peri-infarct area, and plasticity marker, GAP43, both in per-infarct neurons and in contralesional hemisphere’s fibers.
Methods
Mice
All procedures involving mice were performed in accordance with regulations of the Italian Ministry of Health authorization n. 871/2018. Mice were housed in clear plastic cages under a 12 h light/dark cycle and were given ad libitum access to water and food. We used a transgenic mouse line, C57BL/6J-Tg(Thy1GCaMP6f)GP5.17Dkim/J, from Jackson Laboratories (Bar Harbor, Maine USA). This transgenic mouse line expresses the green fluorescent indicator in a subset of excitatory neurons, that is, pyramidal neurons. 24 Mice were identified by earmarks and numbered accordingly. Animals were randomly divided into 5 groups. Each group contained comparable numbers of male and female mice (weighing approximately 25g). Age of mice (ranging from 6 to 8 months old) was consistent between groups. Sample size (25 mice) was chosen according to a priori power analysis (Supplementary Materials, Mice subsection).
Experimental Design
Animals were distributed in 5 groups as follows: Sham n = 6; Stroke n = 4; Optostim n = 4; Robot = 7; Optostim+Robot (abbreviated in OR) n = 4 (Supplementary Figure 2).
•The Sham group consists of 6 healthy mice. During surgery instead of induced photothrombosis, we intraperitoneally injected saline and then we illuminated the primary motor cortex (+1.75 ML and +.50 AP). We then intracranially injected saline in the sensory cortex (+1.75 ML and −.75 AP). After 5 days of recovery from surgery, mice performed 5 days of motor assessment on the robotic platform in order to investigate cortical activation during a motor task in healthy conditions.
•The Stroke group consists of 4 mice. At the beginning of the protocol we evaluated forelimbs use via Schallert cylinder test. We induced a focal stroke in the primary motor cortex (+1.75 ML and +.50 AP). During the same surgery, we intracranially injected saline in the sensory cortex (+1.75 ML and −.75 AP). Two days after surgery we performed behavioral tests to identify alteration in forelimb use consequent to stroke. We performed behavioral experiments at the end of each week to longitudinally investigate spontaneous recovery. After 5 days of recovery from surgery, we stimulated Stroke mice, though not expressing Channelrhodopsin 2 (ChR2), with a blue laser for 20 days to evaluate possible artifacts due to repeated laser stimulation. After 25 days of spontaneous recovery, mice performed 5 days of motor assessment on the robotic platform in order to investigate motor-related cortical activation. 30 days after stroke mice were perfused.
•The Optostim group consists of 4 mice. At the beginning of the protocol we evaluated forelimbs use via Schallert cylinder test. We induced a focal stroke in the primary motor cortex. During the same surgery, we intracranially injected an adeno associated virus (AAV9-CaMKII-ChR2-mCherry) to induce the expression of ChR2 in the sensory areas. Two days after surgery we performed behavioral tests to identify alteration in forelimb use consequent to stroke. After 5 days of recovery from surgery, mice began the rehabilitation paradigm consisting in 20 days of optogenetic stimulation of the peri-infarct area. 25 days after photothrombosis, mice performed 5 days of motor assessment on the robotic platform in order to investigate motor-related cortical activation. At the end of each week of rehabilitation, we performed behavioral experiments to longitudinally investigate mice recovery. 30 days after stroke mice were perfused.
•The Robot group consists of 7 mice. At the beginning of the protocol we evaluated forelimbs use via Schallert cylinder test. We induced a focal stroke in the primary motor cortex. During the same surgery, we intracranially injected an adeno associated virus (AAV9-CaMKII-ChR2-mCherry) to induce the expression of ChR2 in the sensory areas. Two days after surgery, we performed behavioral tests to identify alteration in forelimb use consequent to stroke. After 5 days of recovery from surgery, mice began the rehabilitation paradigm consisting in 20 days of robotic training. At the end of each week of rehabilitation, we performed behavioral experiments to longitudinally investigate mice recovery. 30 days after stroke mice were perfused.
•The Optostim+Robot group consists of 4 mice. At the beginning of the protocol, we evaluated forelimbs use via Schallert cylinder test. We induced a focal stroke in the primary motor cortex. During the same surgery, we intracranially injected an adeno associated virus (AAV9-CaMKII-ChR2-mCherry) to induce the expression of ChR2 in the sensory areas. Two days after surgery, we performed behavioral tests to identify alteration in forelimb use consequent to stroke. At the end of each week of rehabilitation, we performed behavioral experiments to longitudinally investigate recovery. After 5 days of recovery from surgery, mice began the rehabilitation paradigm consisting in 20 days of robotic training followed by optogenetic stimulation of the peri-infarct area. 30 days after stroke mice were perfused.
Surgical Procedures
Mice were injected with a Rose Bengal solution (.2 mL, 10 mg/mL solution in Phosphate Buffer Saline (PBS)). Five minutes after intraperitoneal injection a white light from an LED lamp was focused with a 20X objective to illuminate the primary motor cortex (M1) for 15 min inducing unilateral stroke in the right hemisphere. During the same procedure, we delivered .5 μL of AAV9-CaMKIIa-hChR2(H134R)-mCherry (2.48*1013 GC/mL) 600μm deep inside the cortex at −.75 AP, +1.75 ML. For further information see Supplemental Material.
Robotic Platform
Animals were trained by means of the M- Platform,25,26 which is a robotic system that allows mice to perform a retraction movement of their left forelimb. Motor rehabilitation consists in a pulling task: first animal forelimb is passively extended by the linear actuator of the platform and then the animal has to pull back forelimb up to the resting position. Motor training is composed of 15 movements and after each movement the animal receives a liquid reward. For further information see Supplemental Material.
Optogenetic Stimulation
Daily optogenetic stimulation was performed on head-fixed awake mice by employing a 473 nm laser delivering 5 Hz, 10 ms light pulses. Laser power, ranging from .2 to .8 mW was daily adjusted according to the increment of the transfected area and the progressive lowering of stimulation threshold over the weeks. For further information see Supplemental Material.
Statistical Analysis
Results were considered statistically significant if their corresponding P value was less or equal to 0.05. All the statistical comparisons were performed simultaneously across all experimental groups.
The statistical programming language R was used for the statistical analysis of the Schallert cylinder data. We performed a repeated-measure ANCOVA analysis on raw data (without normalization with pre-stroke asymmetry index) using “Group,” “Week” and their interaction as qualitative variables, “pre-stroke asymmetry index” as a quantitative variable and the animal identifier as designator. Post-hoc analysis was then performed by computing linear contrasts for all pairwise comparisons among estimated marginal means for the variables “Week” and “Group.” Multiplicity adjustment was carried out with Tukey’s method.
OriginPro software (OriginLab Corporation) was used for all other statistical analyses. For calcium, forces and immunohistochemical analysis, a one-way ANOVA was used, with factor GROUP. For all ANOVAs that were statistically significant, multiple comparison among groups, or time points, were assessed using Tukey HSD test. For information regarding Schallert cylinder test, wide-field microscope, and image, forces, and immunohistochemical analysis see Supplemental Material.
Results
Peri-Infarct Optogenetic Stimulation Restores Forelimb Function but Not Cortical Activation Features
The main goal of this study is to find a neuronal substrate of functional recovery within the distributed motor-related cortical activity. To this aim, we compared behavioral and calcium imaging data from mice receiving three different treatments, optogenetic stimulation, motor training and a combination of them. First, the efficacy of repeated optogenetic stimulation of peri-infarct excitatory neurons was tested in stroke mice. A photothrombotic stroke was induced on the M1 of the right hemisphere in Thy1-GCaMP6f mice (Supplementary Figure 1A). We developed a rehabilitation protocol based on longitudinal optogenetic stimulation of peri-infarct excitatory neurons expressing Channelrhodopsin 2 (ChR2, Figure 1A and Supplementary Figures 1B–1D). Optogenetic stimulation was performed daily and consisted of three successive 30-sec laser stimulation trains, separated by 1-min rest intervals (Figure 1B). The optogenetic therapy lasted 4 weeks starting 5 days after stroke. We divided our sample into 3 groups (Figure 2A, Supplementary Figure 2 and Experimental design Materials and Methods section): Sham (healthy mice, no stroke), Stroke (stroke, spontaneous recovery), and Optostim (stroke, optogenetic rehabilitation). During the last week of our investigation, mice performed 5 days of motor assessment in order to evaluate motor-related cortical activity (Figure 1C). Experimental design: (A) Field of view graphical representation: ChR2-expressing neurons (red area) are stimulated by blue laser. Cortical activity is revealed in the right hemisphere during motor assessment. Grey cloud represents stroke core, black dot represents bregma. (B) Stimulation paradigm consisting of 3 stimulation train separated by 1-min rest intervals. (C) Graphical representation of M-Platform for motor assessment. Graph on the right shows an overlap of simultaneously recorded traces (Force, Fluorescence and Position) of an exemplary retraction movement. Optogenetic stimulation of peri-infarct area promotes the recovery of forelimb functionality but not the restoration of spatio-temporal cortical profiles: (A) Sham, Stroke and Optostim experimental timeline. (B) Pre- and post-lesion performance of Stroke (Grey) and Optostim (Blue) groups measured as Asymmetry Index in the Schallert cylinder test. *P 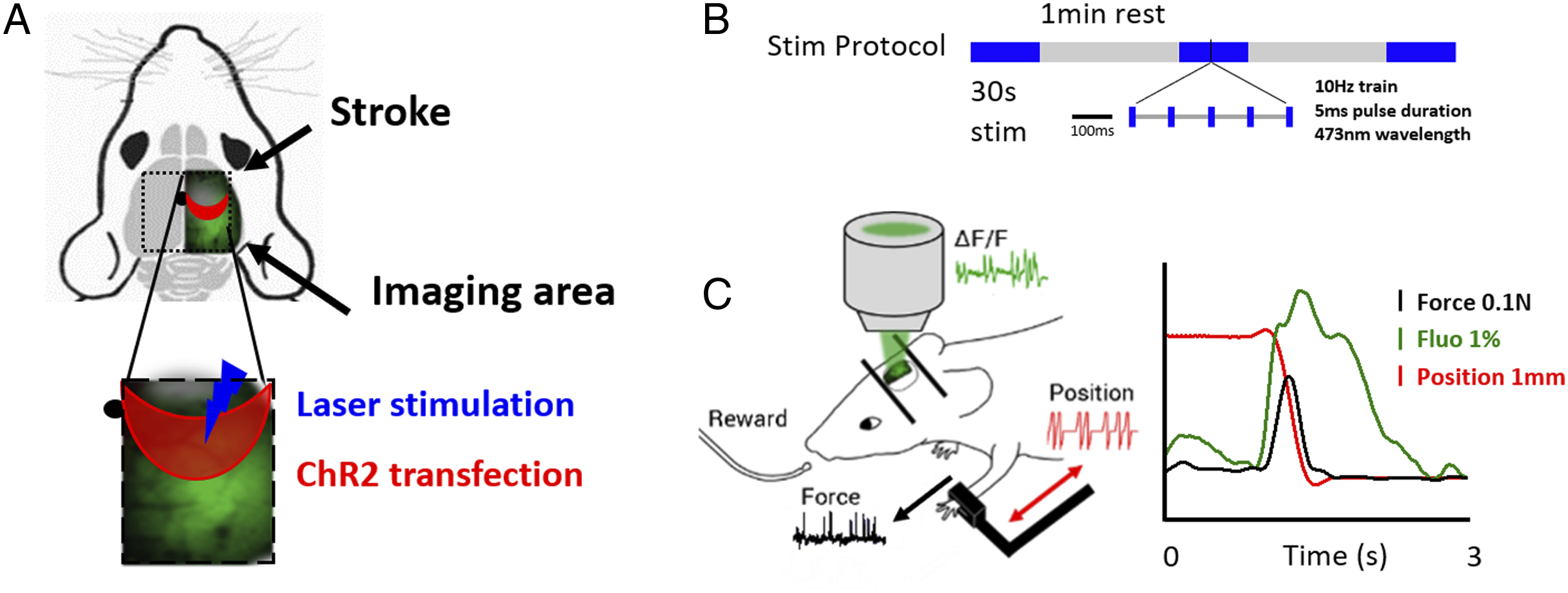
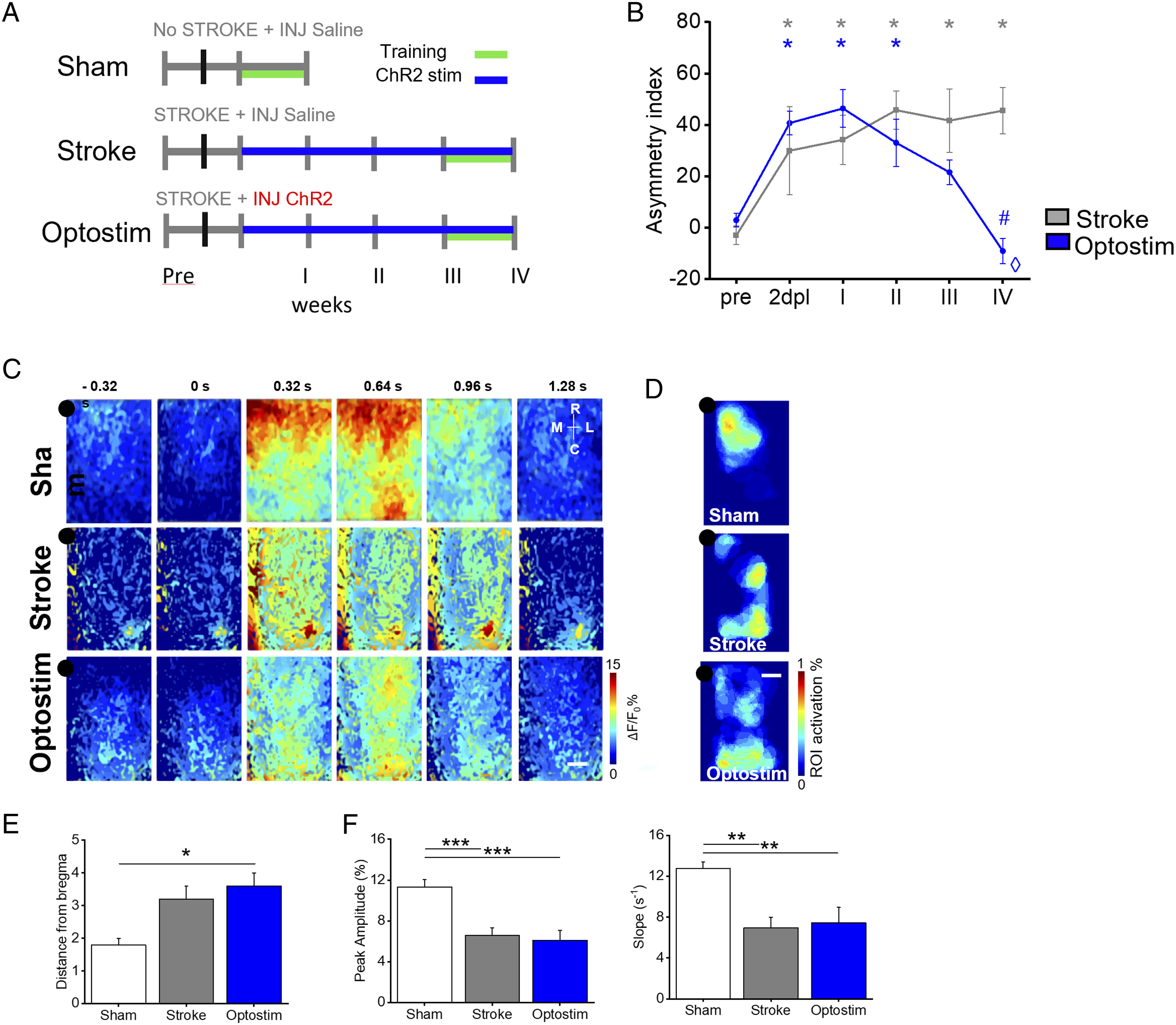
We longitudinally estimated functional recovery of forelimb functionality as a measure of forelimb-use asymmetry by performing Schallert cylinder test at the end of each week of treatment. In detail, the test quantifies the asymmetry index (A.i.) as the relative number of weight-bearing touches with one or both paws when the mouse descends and rears during the exploration of the cylinder walls. The longitudinal evaluation of the asymmetry index of spontaneously recovered mice (Stroke group), significantly different to pre-stroke conditions from the acute phase after stroke (2dpl), highlights the preferential use of the unaffected forelimb indicating the absence of spontaneous recovery up to 4 weeks after the insult (Figure 2B). Conversely, a full recovery of forelimb function was achieved after 4 weeks of daily optogenetic stimulation (Optostim group, Figure 2B). Indeed, the comparison of the asymmetry index at different time points within the Optostim group highlights that the use of forelimb became significantly different with respect to the acute phase after stroke (2dpl) after 4 weeks of treatment.
We then assessed if behavioral recovery was associated with specific features of motor-related cortical functionality. To this aim, we performed wide-field calcium imaging of the affected hemisphere on GCaMP6f mice during the execution of a pulling task within a robotic device, the M-Platform.25,26 The M-Platform was previously integrated with a custom-made wide-field mesoscope to perform calcium imaging of motor-related activity and optogenetic stimulation.27-30 This robotic device allowed a detailed assessment of motor-related cortical activation during an active forelimb pulling task.
To evaluate possible spurious activation of ChR2 expressing neurons induced by the blue LED used for calcium imaging, we performed control experiments in Thy-GCaMP6f mice with or without ChR2 injection while performing 4 weeks of motor training. No significant differences in the peak amplitude and slope of calcium transients were observed between ChR2+ mice and ChR2- (Supplementary Figure 3A), demonstrating that ChR2 stimulation induced by the imaging LED was negligible or absent.
We then analyzed the spatial extension of motor representation in the ipsilesional hemisphere (Supplementary Figure 3B) by overlapping the movement-triggered activation maps obtained for each day of training. This comparison showed a segregated activation in healthy mice, whereas in Stroke and Optostim mice the maps covered most of the affected hemisphere, up to the caudal regions of the cortex (such as retrosplenial and visual areas Figures 2C and 2D), as quantified by analyzing distance from bregma of motor-related map’s centroid (Figure 2E). Then, we explored the possibility that temporal features of activation might be correlated with functional recovery in the Optostim group. To this aim, we investigated amplitude and slope of calcium transient as an estimate of the extent and the velocity of the rising phase of neuronal activation during movement execution. However, by analyzing the fluorescence transients averaged from the region of maximum calcium activation during active pulling, we observed no significant differences in amplitude and timing in Optostim mice compared to spontaneously recovering Stroke mice (Figure 2F). These results demonstrate that, even though recovery of forelimb function can be achieved by optogenetic stimulation alone, the spatiotemporal features of motor-related cortical activity in the ipsilesional hemisphere did not recover to pre-stroke conditions.
Combining optogenetic stimulation with motor training boosts the functional recovery and is associated with reshaping of motor-related activation maps and restoration of temporal features of calcium transients
In a previous study, we showed that a combined rehabilitation protocol of pharmacological inactivation and motor training was beneficial to achieve a proficient functional recovery.
31
We hypothesized if an even more effective recovery could be achieved by coupling optogenetic stimulation to longitudinal motor training of the affected forelimb on the M-platform. Thus, a rehabilitation protocol combining daily training on the M-Platform and optogenetic stimulation was tested (OR group, Figure 3A and Supplementary Figure 2). We choose to sequentially perform motor training and optogenetic stimulation of the peri-infarct cortex to avoid a potential bias on fluorescence signal during motor training (calcium imaging) due to the previous excitation of the cortex. The combined treatment was applied daily from the acute phase (5 days after stroke) up to 4 weeks after stroke. The OR group was then compared to healthy mice (Sham group), spontaneous recovery (Stroke group), and motor exercise alone (Robot group). Combining optogenetic stimulation with motor training boosts functional recovery and promotes the restoration of motor-related cortical functionality: (A) Experimental timeline for Robot and OR groups. (B) Pre- and post-lesion performance of Stroke (Grey), Robot (Green), and Optostim+Robot (Magenta) groups measured as Asymmetry Index in the Schallert cylinder test. *P ≤ .05 refers to repeated-measure ANCOVA analysis followed by post-hoc pairwise comparisons for Schallert asymmetry index values of Stroke, Robot and OR groups at different time points. Stroke group: p pre-2dpl = .0012; pre-Iw = .0001; pre-IIw <.0001; p pre-IIIw< .0001; p pre-IVw <.0001; Robot group: p pre-2dpl< .0001; pre-Iw<.0001; pre-IIw<.0001; p pre-IIIw<.0001; p pre-IVw<.0001; OR p pre-2dpl<.0001; pre-Iw<.0001; ⋄ P ≤ .05 refers to repeated-measure ANCOVA analysis followed by post-hoc pairwise comparisons for Schallert A.i. values of OR group 2dpl with respect to the other time points: 2dpl-IIw<.0001; 2dpl-IIIw<.0001; 2dpl-IVw<.0001; #P≤.05 refers to repeated-measure ANCOVA analysis followed by post-hoc pairwise comparisons for Schallert asymmetry index values of OR and Stroke groups. OR vs Stroke p wIII=.02; p wIV=.05 (for statistical pairwise comparison see Supplementary Materials Table 1). nStroke=4; nRobot=7; nOR=4. (C) Image sequence of cortical activation during pulling of the handle, from .32s before to 1.28s after force peak onset. Each row shows a representative sequence from a single animal of each group Sham (repeated from Figure 1), Robot and OR. Scale bar 1mm. Left panel shows the average thresholded ROI computed for each experimental group. Black dot represents bregma. Scale bar 1mm. (E) Graph shows the distance from bregma (average ± SEM) of maps centroid (Sham = 1.8 ± .2 mm; Stroke = 3.2 ± .4 mm; Robot = 2.9 ± .5 mm; OR = 2.2 ± .4 mm). (F) The graph shows the maximum of fluorescence peaks (average ± SEM) of calcium transient (Peak-Amplitude_Sham = 11.3 ± .7%; Peak-Amplitude_Stroke = 6.6 ± .7%; Peak-Amplitude_Robot = 6.3 ± .3%; Peak-Amplitude_OR = 10.9 ± .7%; *P < .05, **P < .005, ***<.0005 based on one-way ANOVA followed by Tukey’s correction: p Sham-Robot = .00005; p OR-Stroke= .003; p OR- Robot=.0005). (G) The graph shows the slope (average ± SEM) of the rising phase of fluorescence traces (Slope_Sham = 12.8 ± .6 s−1; Slope_Stroke = 6.9 ± 2.0 s−1, Slope_Robot = 6.5 ± .8 s−1; Slope_OR = 11.8 ± .7 s−1; *P < .05, **P < .005, ***<.0005 based on one-way ANOVA followed by Tukey’s correction: pSham-Robot = .0002; p OR-Stroke = .02; p OR-Robot = .004; for statistical pairwise comparison see Supplementary Materials Table 2); (H) Transgenic expression of GCaMP6f under Thy1 promoter (green) and representative PV immunostaining (red) of a Stroke and OR mouse. (I) Quantification of PV+ cells in all groups in the peri-infarct area (Stroke = 9.7 ± 2.2; Optostim = 13.0 ± 1.3; Robot = 14.6 ± .7; OR = 19.8 ± 1.0; *P<.05 based on one-way ANOVA followed by Tukey’s correction: p Stroke-OR = 0,0004; p Optostim-OR = 0.006; p Robot-OR = 0.02; for statistical pairwise comparison see Supplementary Materials Table 3). Scale bar 20µm.; nSham = 6; nStroke = 4; nRobot = 7; nOR = 4.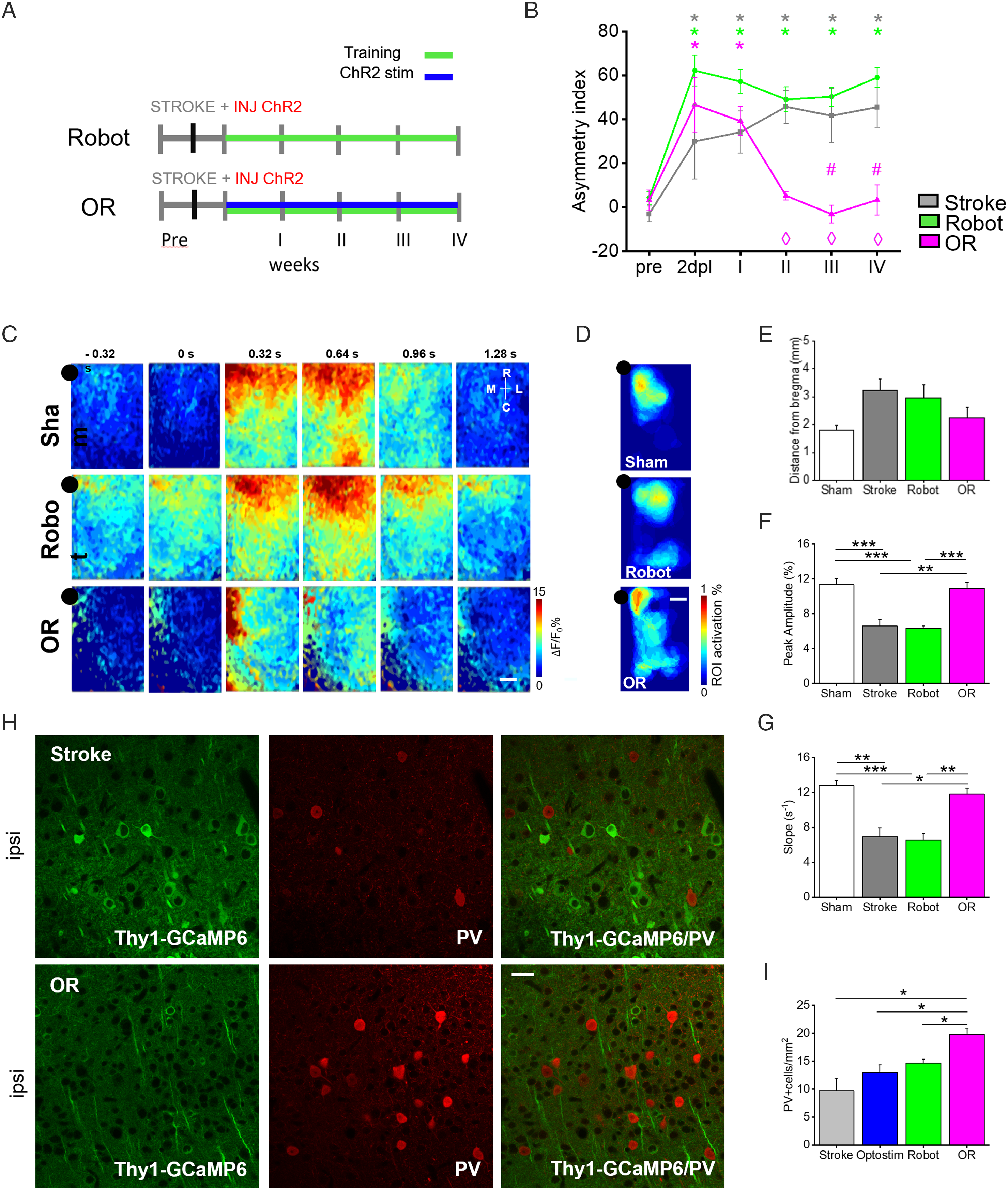
Training induced a progressive modulation of the force transients measured on the M-Platform during active pulling of the affected forelimb. A small reduction in amplitude and full-width half maximum, though not statistically significant, was seen both in OR and Robot mice (Supplementary Figure 3C). In addition, the progressive decrease of time-to-target visible in mice with combined therapy was in line with previous findings in robot-treated stroke mice. 26 We then evaluated alterations in spontaneous forelimbs use on Schallert cylinder test. In accordance with Spalletti and colleagues, 32 motor training alone was not able to restore pre-stroke performances (Figure 3B). Indeed by comparing the Asymmetry index (A.i). of Stroke and Robot mice no significant differences were revealed between groups. Conversely, the comparison of A.i. at different time points with respect to the acute phase after stroke (⋄) highlights that OR mice recovered forelimb functionality already on the second week of rehabilitation (Figure 3B), thus supporting the hypothesis that combined rehabilitation boosts recovery. Indeed, as emerges from the comparison with pre-stroke condition (*), the use of forelimb is shifted towards the non-paretic limb in the acute phase after stroke (2 days post-lesion and after I week of treatments). During the rehabilitation period (II-IV weeks of treatment) the asymmetry index was recovered in OR mice to pre-stroke levels, significantly different (#) from spontaneously recovered mice (III and IV weeks of treatment).
We wondered if this fast behavioral improvement due to the combined treatment was mirrored into specific spatiotemporal features of motor-related cortical activity. Examples of temporal sequences of pulling-evoked cortical activation are shown in Figure 3C, together with the associated motor representations (ie the thresholded motor-related maps, see Supplementary Materials and Methods section) on the fourth weeks after stroke (Figure 3D). The motor representations of both Robot and OR groups partially resemble the Sham group, with a similarly short distance of the centroid of the motor representation to bregma (Figure 3E). Moreover, as shown in Figure 3D the region of maximum activation is confined to a segregated region of the cortex in the peri-infarct area. Conversely, the motor representation of the Stroke group (Figure 2D) is shifted more caudally and is divided into two regions diffusing from the sensory to the visual areas of the cortex. This means that daily motor training promotes the segregation of motor representation to the peri-infarct area. Interestingly, after combined therapy, the motor representation largely overlaps the caudal portion of the secondary motor cortex, according to the Paxinos and Franklin Mouse Brain Atlas. 33 We further investigated how the temporal profiles of calcium transients averaged over the motor representation area were altered after combined rehabilitation. Amplitude and slope in mice treated with motor training alone (Robot group) were comparable to spontaneous recovery (Stroke group). Conversely, the synergic effect of combined rehabilitation promoted the reestablishment of these salient features of calcium transient in peri-infarct area (Figures 3F and 3G). This positive trend towards pre-stroke conditions (Sham group), though not significantly different, started from the second week of rehabilitation in OR mice (Supplementary Figure 3D). The pronounced increment in peak amplitude and slope of calcium transient in combined rehabilitated mice became comparable to healthy condition after 4 weeks of combined treatment.
The modulation of cortical motor-related activity from excitatory neurons could be associated with a new excitatory/inhibitory balance 14 potentially compensating detrimental consequences of stroke. 34 We thus evaluated the density of parvalbumin-positive (PV+) cells throughout all cortical layers of the peri- and contra-lesional cortices (Figure 3H). In the peri-infarct region, a small but not significant increase of PV+ cell density was induced by the single treatments (Optostim and Robot, Figure 3I) with respect to spontaneous recovery mice (Stroke group). Interestingly, the PV+ cell density is significantly higher in peri-infarct cortex of mice with combined therapy (OR group) compared to non-treated (Stroke) animals (Figure 3I). Conversely, no significant differences were observed in the contralesional hemisphere in all groups (Supplementary Figure 4A). Together with the increased motor-related activation levels in pyramidal cells, these results suggest that the synergic effect of combined rehabilitation could promote the establishment of a new excitatory/inhibitory equilibrium in the peri-infarct and contralesional hemispheres.
Functional Recovery Is Associated With an Increased Expression of GAP43
To further identify molecular targets associated with functional recovery, we tested the presence of plasticity markers in the lesioned and contralesional cortex by immunohistochemical analysis. We examined the expression of the growth-associated protein 43 (GAP43), a plasticity marker involved in synaptic turnover and reorganization after stroke.3,32,35 While after optogenetic stimulation (Optostim group) or motor training (Robot group) GAP43 expression levels were comparable to spontaneously recovered mice (Stroke group), combined rehabilitative treatment promoted a massive expression of this neuronal plasticity marker in the peri-infarct region (Figure 4A). Though in the contralesional hemisphere no differences in GAP43+ cells were observed between groups (Supplementary Figure 4B), an increase in number and length of GAP43+ fibers was present in the homotopic areas on the contralesional hemisphere after all treatments (Optostim, Robot, and OR groups; Figure 4B). Nevertheless, only the combined treatment (OR group) showed significantly different density of GAP43+ neurites compared to spontaneously recovered mice (Stroke group). This result demonstrate that perilesional stimulation and motor training synergistically enhance the density of GAP43+ neurites in distal regions functionally related to the stroke core, which is possibly associated to axonal sprouting36-38 or dendritogenesis.39,40 Ex vivo evaluation highlights an increased expression of plasticizing factor in OR mice: (A) Left: Transgenic expression of GCaMP6f under Thy1 promoter (green) and representative GAP43 immunostaining (red) of a coronal section (100µm thick) of a Stroke and OR mouse. Right: quantification of GAP43+ cells in the peri-infarct area in all experimental groups (Stroke = 25.0 ± 7.2; Optostim = 38.7 ± 7.3; Robot = 34.3 ± 2.4; OR = 70.7 ± 10.7; *P < .05 based on one-way ANOVA followed by Tukey’s correction: p Stroke-OR = 0.001; p Optostim-OR = 0.02; p Robot-OR = 0.004). (B) Left: transgenic expression of GCaMP6f under Thy1 promoter (green) and representative GAP43 immunostaining (red) of CL labeled fibers. Right: quantification of GAP43+ fibers in the CL hemisphere (Stroke = 2.4 ± 0.4; Optostim = 31.8 ± 5.4; Robot = 34.0 ± 4.7; OR = 54.4 ± 8.3; *P < .05 based on one-way ANOVA followed by Tukey’s correction: p Stroke-OR = 0,0007; p Optostim-Stroke = 0.04; p Robot-Stroke = 0.01; for statistical pairwise comparison see Supplementary Materials Table 3); Scale bar 20 µm; nStroke = 4; nOptostim = 4; nRobot = 7; nOR = 4.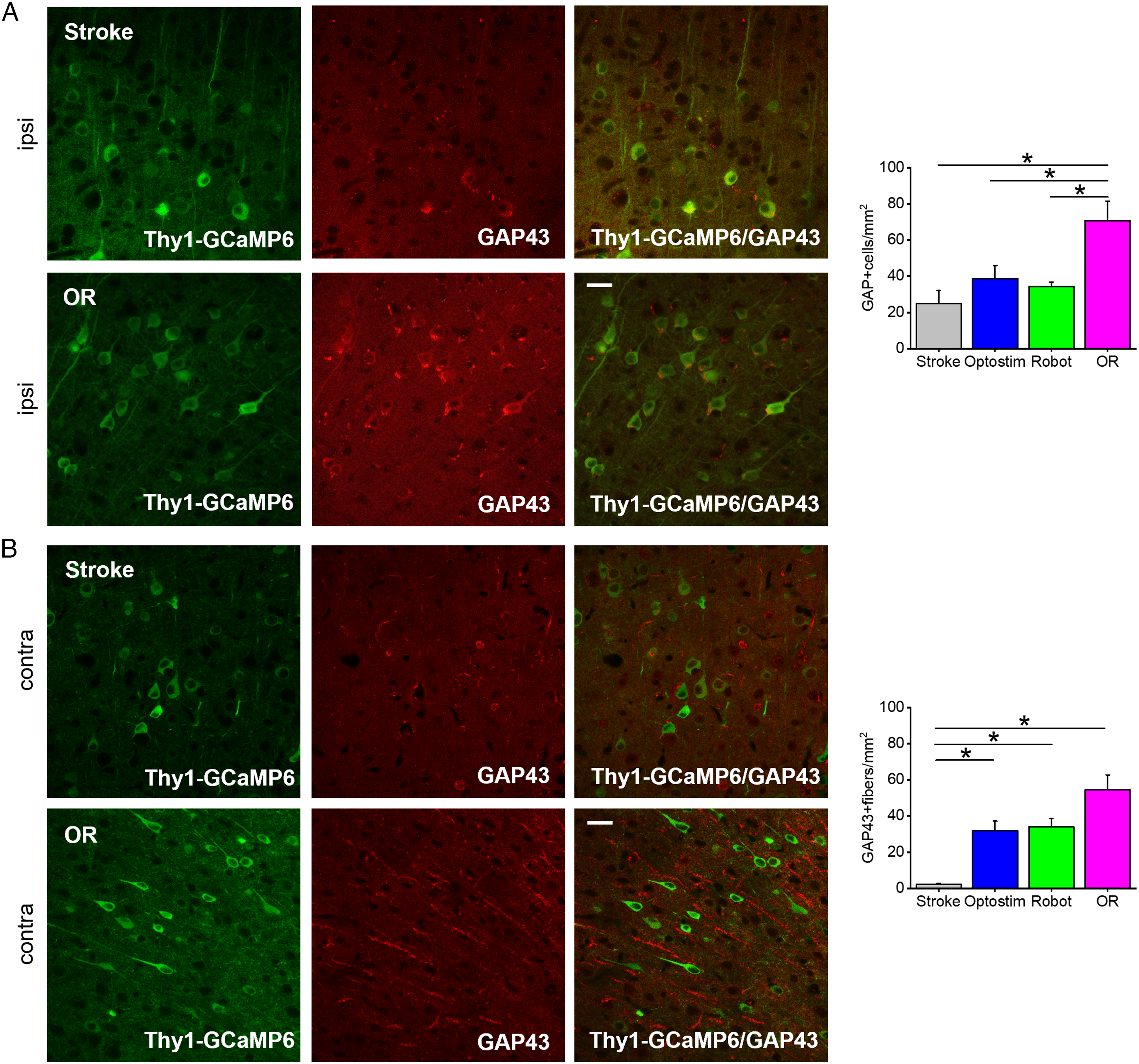
Taken together, the histological analysis shows that behavioral recovery and the associated cortical remapping induced by combined rehabilitation are supported by a plasticizing milieu promoted by the synergic effect of ipsilesional neuronal stimulation and repetitive motor training.
Discussion
The present study aimed at understanding the neuronal correlate of functional recovery. By taking advantage of recent advancements in optical tools, including in vivo fluorescence imaging and optogenetics, to characterize critical features of plasticity, repair and recovery after stroke,31,41-45 we investigated the motor-related distributed cortical activity in several rehabilitation paradigms. Among the treatments, both the optogenetic stimulation of perilesional excitatory neurons (Optostim group) and the combined therapy with optogenetic stimulation and motor training of the paretic forelimb (OR group) led to functional recovery. However, the novel combined rehabilitation paradigm validated here led to the behavioral recovery of forelimb functionality, significantly faster than optogenetic stimulation alone. Moreover the combinatory treatment induces the restoration of spatiotemporal features of cortical activity to pre-stroke levels. Based on these results, our hypothesis is that functional recovery is supported by the establishment of a new excitatory/inhibitory balance between hemispheres, revealed by the augmented cortical motor response flanked by increased expression of PV+ neurons in the peri-infarct cortex.
We first assessed the consequences of the optogenetic treatment consisting of 4 weeks of daily perilesional stimulation. In this work, we assess the motor functionality through Schallert cylinder test only in order to evaluate forelimb use preference after the insult and in different rehabilitative therapies; however, in future studies a combination of different motor tests will better evaluate different aspects of functional recovery such as fine motor control and forelimb dexterity. Previous works demonstrated the efficacy of post-stroke optogenetic stimulation on both the peri-infarct cortex14,15 and striatum 46 in promoting the recovery of forelimb sensory-motor abilities. In agreement with these findings, we revealed a remarkable improvement of forelimb functionality after 4 weeks of optogenetic stimulation of the peri-lesioned excitatory neurons. Our data also agree with the observation by Tennant and colleagues that even if optogenetic rehabilitative treatment enhanced the restoration of somatosensory cortical circuit function, the cortical area responsive to optogenetic stimulation and the peak of motor-related cortical activity after 4 weeks of treatment were not fully recovered to pre-stroke levels. Accordingly, our observations indicate that our optogenetic treatment per se does not recover pre-stroke spatio-temporal features of motor-related cortical activation. Indeed, a diffuse activation involving regions across the entire lesioned hemisphere is observed during active pulling of the paretic forelimb. In addition to these diffuse motor representation motor-related cortical activation profiles of the Optostim group have lower amplitude compared to pre-stroke conditions. Thus, our results support the hypothesis that optogenetic stimulation of excitatory spared neurons counteracts the increased excitability of the contralesional M1, thus balancing the excessive inhibitory drive onto the ipsilesional cortex. Taken together these results highlight that the restoration of pre-stroke features of cortical activity is not an essential requisite to achieve the recovery of forelimb functionality.
On the other hand, although Robot trained mice did not achieve a functional recovery, their motor-related map is segregated to the sensorimotor regions, similarly to healthy mice (Sham group). Nevertheless, longitudinal motor training alone did not restore pre-stroke features of calcium transient. These results confirm that motor training alone promotes a task-specific motor improvement as previously shown by Spalletti and collaborators, 32 which is allegedly associated to the stabilization of motor representation. 47
Finally, the combined rehabilitative treatment triggered a synergic effect that connects behavioral improvement to recovery of pre-stroke motor-related cortical activation. Indeed, the combination of cortical stimulation and motor training induces a fast restoration of the forelimb function towards healthy conditions, as measured via asymmetry index analysis in the Schallert cylinder test. At the same time, flanking the stimulation with repeated exercise leads to both confinement of motor representations and restoration of temporal features of motor-related calcium transient in the peri-lesioned cortex, towards the pre-stroke condition. The strong involvement of spared neurons in the secondary motor cortex revealed in the motor representation of OR mice could play a leading role in regaining functional recovery. Our results highlight the synergic effect of the combined rehabilitation since counterbalancing the contralesional inhibition of peri-infarct cortex by optogenetic stimulation could enable the stabilization of spared circuitry achieved by longitudinal motor training.
We showed an increase in motor-related cortical activation mediated by excitatory cells in the peri-infarct area of OR mice compared to single treatments (Optostim and Robot groups) and spontaneous recovery (Stroke group). Together with the higher levels of parvalbumin expression, these results provide evidence that the combined treatment might promote the restoration of an excitatory/inhibitory balance in the peri-infarct cortex similar to pre-stroke conditions. It has been previously demonstrated that parvalbumin expressing neurons may contribute to long-range GABAergic projections.48,49 Moreover, Rock and collaborators 50 recently found a direct inhibitory pathway made up of PV neurons connecting corresponding cortical areas (ie motor, auditory, and visual cortices) in the two hemispheres through the corpus callosum. In our study, the increase of inhibitory cells in the peri-infarct cortex may counteract the iper-inhibition exerted by the healthy hemisphere through long-range cortico-cortical projections. Finally, previous studies demonstrated that modulation in parvalbumin expression is associated with achievement of skilled ability in motor task. 51 Our results are in line with what observed by Swanson and Maffei, who shows that PV expression is stabilized to higher expressions level once motor performance is saturated. In our study, the small increment of PV+ cells observed in the Robot group is strongly enhanced in OR mice. The combination of motor training with optogenetic stimulation further stimulates the inhibitory activity in the perilesional cortex.
The behavioral improvement is supported by the long-lasting expression of growth-associated factors in the perilesional area, as demonstrated by the increased number of cells expressing GAP43. It was previously demonstrated that this growth-associated factor promotes cortical remapping, 3 axonal sprouting,3,34,35 and dendritogenesis39,40 after stroke. This result extends previous findings by Cheng and collaborators showing that perilesional optogenetic stimulation after stroke promotes a global increase in GAP43 expression involving both hemispheres. 14 Also, our findings are in agreement with Spalletti and collaborators 32 showing that a combined rehabilitative treatment leading to functional recovery promotes an increase of plasticity markers expression. The recovery of the excitatory/inhibitory balance could be further supported by the axonal sprouting in the contralesional cortex, where a substantial number of GAP43+ fibers were detected, particularly in combined rehabilitated mice. The increment of GAP43+ cells in the peri-infarct area, in combination with GAP43+ axons in the contralesional region, suggests the rise of transcallosal axonal sprouting from the ipsilesional hemisphere.
One of the main results of the present study is that restoration of motor functionality can occur without recovery of spatiotemporal features of motor-related cortical activation that characterize pre-stroke mice, as in Optostim mice. On the other hand, segregation of motor representation is not necessarily entangled with functional recovery, as in Robot mice. These findings suggest that rehabilitation strategies for functional recovery might not essentially aim at the restoration of pre-stroke features of motor-related activity, as observed in a recent work by Cecchini and collaborators. 30 Nevertheless, the reestablishment of pre-stroke activation transients was a distinguished feature of the most efficient therapeutic approach, the combined therapy. In accordance with our previous findings 31 on a different combined rehabilitation paradigm, this study supports the hypothesis that the restoration of pre-stroke temporal features could be an important biomarker of the functional recovery.
To conclude, we developed a post-stroke rehabilitation therapy that exploits the synergic effect of peri-infarct optogenetic excitation and repetitive motor training to promote the stabilization of a new excitatory/inhibitory balance between hemispheres resulting in the recovery of forelimb functionality and spatio-temporal features of motor-related calcium transient. This study highlights how the outcome of rehabilitation therapies depends on a balance between stabilization of peri-infarct circuitry and fostering of cortical plasticity.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211056656 – Supplemental Material for Combining Optogenetic Stimulation and Motor Training Improves Functional Recovery and Perilesional Cortical Activity
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683211056656 for Combining Optogenetic Stimulation and Motor Training Improves Functional Recovery and Perilesional Cortical Activity by Emilia Conti, Alessandro Scaglione, Giuseppe de Vito, Francesco Calugi, Maria Pasquini, Tommaso Pizzorusso, Silvestro Micera, Anna Letizia Allegra Mascaro and Francesco Saverio Pavone in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgments
We thank K. Deisseroth for opsin plasmids. We thank Matteo Caleo, Cristina Spalletti and Claudia Alia for very useful discussion about the manuscript.
Author Contributions
E.C., A.L.A.M. and F.S.P. conceived the study. E.C., A.L.A.M. and F.C. performed experiments. E.C., A.S., G.d.V., M.P., processed data. F.S.P., T.P. and S.M. obtained funding support. E.C. and A.L.A.M. wrote the paper. All authors approved the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has received funding from the H2020 EXCELLENT SCIENCE - European Research Council (ERC) under grant agreement 692943 BrainBIT. In addition, it was supported by the European Union’s Horizon 2020 Research and Innovation Programme under Grant Agreements 785907 (HBP SGA2), and 654148 (Laserlab-Europe).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
