Abstract
Background. Low-frequency repetitive transcranial magnetic stimulation (rTMS) over the unaffected motor cortex may improve motor function of the paretic hand after stroke. However, low-frequency rTMS might adversely affect bimanual movement by decreasing transcallosal function. Objective. The authors investigated whether combined administration of rTMS and transcranial direct current stimulation (tDCS) prevents deterioration of bimanual movement induced by low-frequency rTMS over the unaffected hemisphere. Methods. A total of 27 participants with chronic subcortical stroke were randomly assigned to receive either 1 Hz rTMS over the unaffected hemisphere, anodal tDCS over the affected hemisphere, or a combination of rTMS and tDCS. All patients performed a pinching motor-training task after stimulation. Bimanual movement and transcallosal inhibition (TCI) were evaluated after stimulation. Results. rTMS and rTMS-tDCS enhanced the motor training effect on the paretic hand. rTMS decreased bimanual coordination and reduced TCI from the unaffected to the affected hemisphere (TCIunaff-aff). rTMS-tDCS changed TCI balance of both hemispheres but did not affect bimanual coordination or TCIunaff-aff. The change in bimanual coordination was negatively correlated with TCIunaff-aff. Following stimulation, improvement in the pinch force in the paretic hand was negatively correlated with TCI balance. Conclusions. Inhibitory rTMS over the unaffected hemisphere transiently caused deterioration of bimanual movements for the current task in stroke patients. This short-term decline was prevented by combined administration of low-frequency rTMS over the unaffected hemisphere and anodal tDCS over the affected hemisphere. These responses to bihemispheric stimulation suggest possible caution and opportunities for the rehabilitation of hand function after stroke.
Keywords
Introduction
Rehabilitation interventions, including practice of the affected upper extremity and bimanual practice, 1 aim to increase gains after stroke. 2 Recent studies have aimed to augment plasticity and improve motor function using methods based on the interhemispheric competition model.3,4 The interhemispheric competition model proposes that motor deficits in stroke patients are a result of reduced output from the affected hemisphere and excess transcallosal inhibition (TCI) of the affected hemisphere from the unaffected hemisphere.
Repetitive transcranial magnetic stimulation (rTMS) is noninvasive brain stimulation that can alter the excitability of the human cortex for several minutes. High-frequency rTMS (more than 5 Hz) increases cortical excitability, whereas low-frequency rTMS (1 Hz or less) suppresses cortical excitability. 5 Application of rTMS takes advantage of this physiological peculiarity and has been proposed to promote functional recovery of the paretic hand in stroke patients.1,3,6 In particular, inhibitory rTMS over the unaffected hemisphere decreases TCI from the unaffected to the affected hemisphere and increases the excitability of the affected hemisphere; this would translate into improved motor function of the affected hand.7,8 However, inhibitory rTMS may deteriorate some aspects of motor function because of reduced TCI, which plays a role in bimanual movement.9,10 In fact, our preliminary study found that inhibitory rTMS therapy worsened bimanual movement. 11 Moreover, we found that activity of the affected motor cortex might compensate for deterioration of bimanual movement induced by inhibitory rTMS over the unaffected hemisphere.
Therefore, we hypothesize that excitatory stimulation over the affected motor cortex prevents deterioration of bimanual movement caused by inhibitory rTMS over the unaffected hemisphere. Recently, rTMS and transcranial direct current stimulation (tDCS) have been primarily used as noninvasive cortical stimulations.5,6 However, when a different rTMS apparatus, along with inhibitory rTMS, is used to perform excitatory stimulation, we cannot simultaneously stimulate both hemispheres because the stimulation coils overlap. 4 In contrast, tDCS can stimulate an area within a small space and stimulate both hemispheres simultaneously along with rTMS. Therefore, in this study, we added excitatory tDCS over the affected hemisphere along with inhibitory rTMS over the unaffected hemisphere. Additionally, we examine the TCI change after 3 stimulation protocols (low-frequency rTMS over the unaffected hemisphere, anodal tDCS over the affected hemisphere, and a rTMS-tDCS combination). The analyses address 4 questions: (1) Was combined administration of rTMS and tDCS more effective than unilateral stimulation in improving motor function in stroke patients? (2) Did any stimulation protocol change the bimanual movement? (3) Did any stimulation protocol change TCI? (4) Did TCI influence motor improvement and bimanual movement?
Methods
Participants
Our study population comprised 27 stroke patients. Inclusion criteria were as follows: (1) first-time stroke of more than 6 months’ duration, (2) only subcortical lesion confirmed by MRI or CT, and (3) motor deficits of the unilateral upper limb that had improved to the extent that patients could perform tapping tasks. Exclusion criteria included the following: (1) seizure, (2) intracranial metallic implant or pacemakers, and (3) intake of a drug that can modify brain excitability. Participants were randomly divided into 3 groups (Table 1). All participants gave written informed consent, and the protocol was approved by the local ethical committee of the Hokkaido University Graduate School of Medicine.
Clinical Characteristics, Motor Functions, and TMS Parameters Before Stimulation
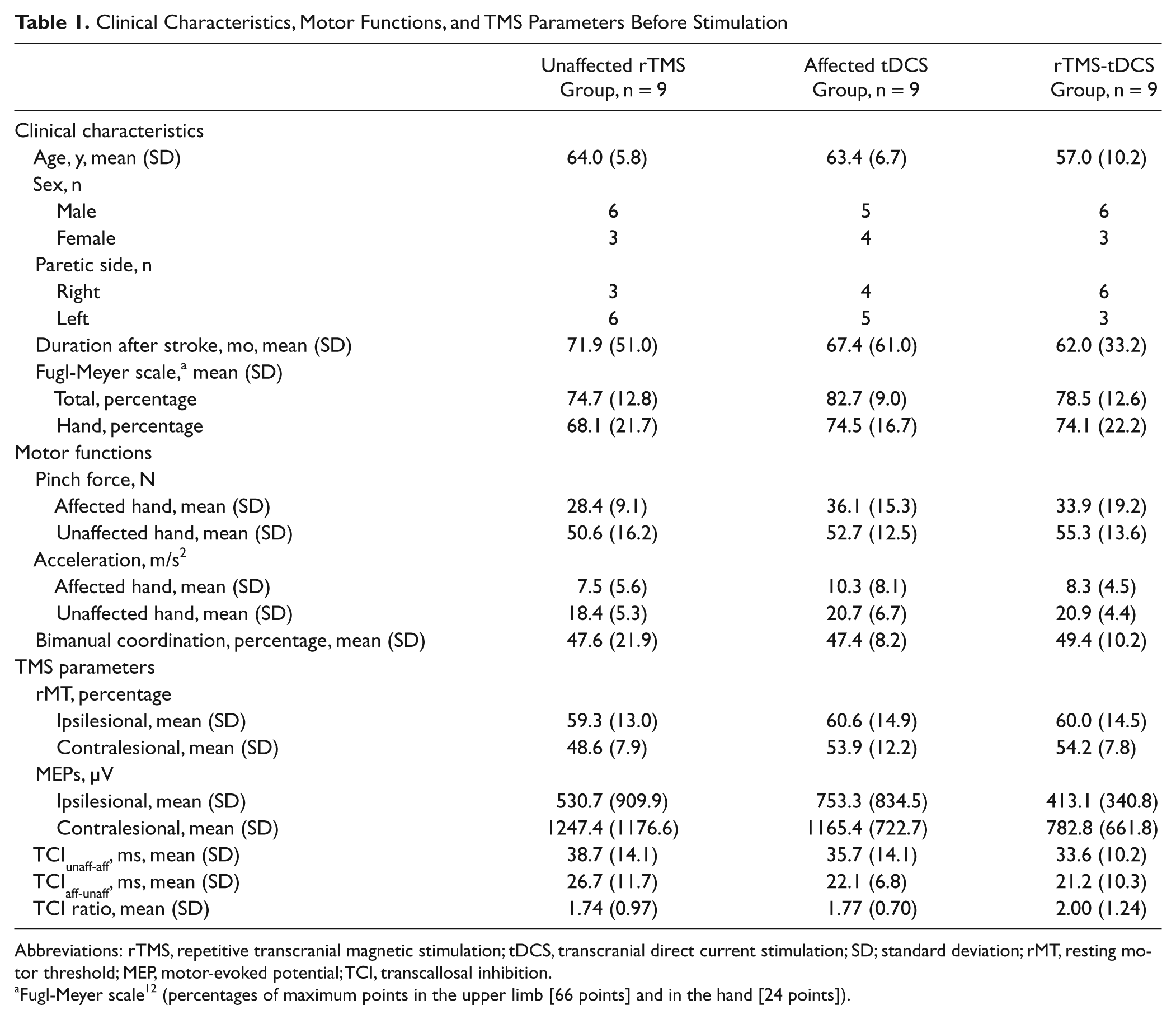
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; tDCS, transcranial direct current stimulation; SD; standard deviation; rMT, resting motor threshold; MEP, motor-evoked potential; TCI, transcallosal inhibition.
Fugl-Meyer scale 12 (percentages of maximum points in the upper limb [66 points] and in the hand [24 points]).
Time Course of Experiment
Measurements for assessing motor function (acceleration, pinch force, and bimanual coordination) and the parameters of TMS (resting motor threshold [rMT], amplitude of motor-evoked potentials [MEPs], and TCI) were performed prestimulation (Pre-Stim) and poststimulation (Post 1, immediately after stimulation; Post 2, 30 minutes after stimulation; and Post 3, 7 days after stimulation). Figure 1 shows the time course of the experiment.
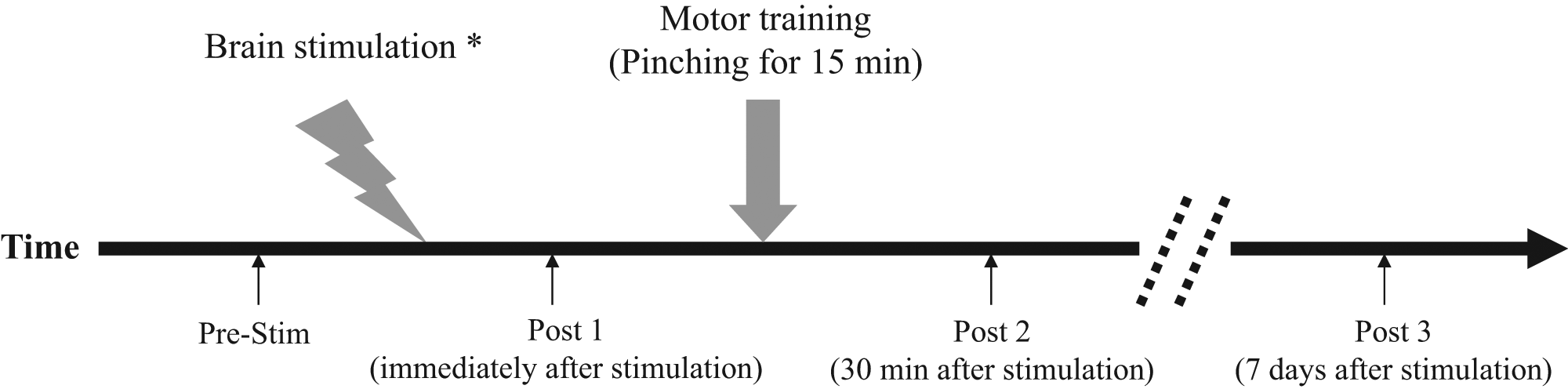
Time course of the experiment.
Motor Function Evaluation
Bimanual tapping was conducted using a finger-tapping system (Hitachi Corporation, Tokyo, Japan) as a motor function evaluation. The system consists of 2 magnetic sensor coils, electronics, and a laptop PC. The finger-tapping system measures the distance between the 2 magnetic coils attached to the nail of each index finger and thumb using medical tape. Details of this system have been previously published. 13 The tapping task, using the index finger and thumb of both hands was paced by a metronome and performed as quickly as possible (frequency individualized between 0.5 and 1 Hz). The tapping frequency for each patient was consistent throughout the experiment. Participants performed the bimanual antiphase tapping for 1 minute in each section, because our preliminary study had revealed that rTMS changed the bimanual movement in antiphase but not in in-phase tapping. 11 Acceleration of both hands and bimanual coordination were evaluated by using the packaged analysis software of the finger-tapping system. The accelerations for 1 minute were averaged in each section. The time of the tapping trigger signal on the paretic and nonparetic sides were defined as TP and as TN, respectively. We defined bimanual coordination according to the following equation:
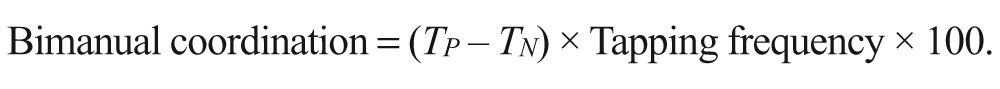
This equation indicated the accuracy of timing for the bimanual antiphase tapping; ideally, the value of bimanual coordination is 50%. We averaged the bimanual coordination values over 1 minute and calculated the absolute value of the difference between prestimulation and each poststimulation value in order to evaluate changes in bimanual movement. The tapping trigger signal was the defined time when the thumb and index fingers were most distant. We obtained bimanual coordination data from 22 patients. We excluded patients who were unable to conduct the antiphase tapping for 1 minute from the coordination study section (1 patient, rTMS group; 2 patients, tDCS group; 2 patients, rTMS-tDCS group). We used the data from the in-phase bimanual tapping task for acceleration evaluation in these patients.
The maximum pinch force of the affected hand was determined using a pinch gauge (Pinch Meter SPR-641; Sakai Medical, Tokyo, Japan). Participants were instructed to use only their thumb and index finger during the pinch force measurements. A total of 10 pinch forces were averaged in each session. Between Post 1 and Post 2, patients performed a pinching task using the paretic hand for 15 minutes as motor training, as described in a previous repot. 8 Patients were allowed to familiarize themselves with these motor evaluation methods on the day before the stimulation.
TMS Parameters
Single-pulse TMS was performed using a 70-mm figure-8 coil and Magstim 200 (Magstim Company, Dyfed, UK), and rTMS was applied using the same coil and a Magstim rapid stimulator (Magstim Company). The coil was placed tangentially over the motor cortex at an optimal site for the first dorsal interosseous (FDI) muscle. The optimal site was defined as the location where stimulation at a slightly suprathreshold intensity elicited the largest MEPs in the FDI. This position was marked on the scalp and used throughout the experiment. The rMT was determined separately for each stimulator and defined as the lowest stimulator output that could produce MEPs with a peak-to-peak amplitude greater than 50 µV in at least half of the 10 trials. The peak-to-peak amplitude of 10 averaged FDI responses obtained at 120% rMT was also determined using the Magstim 200.
In the TCI procedure, each hemisphere was stimulated 20 times (intensity, 150% rMT) during unilateral maximal tonic contraction of the ipsilateral FDI, as described previously. 7 Also, 20 electromyography (EMG) signals from the FDI were rectified and averaged for evaluation of TCI. TCI was quantified by the period of relative EMG suppression after the stimulus. TCI duration was measured from where EMG activity clearly decreased below background activity to where EMG activity again reached the background activity. 14 The mean amplitude of rectified and averaged EMGs for 100 ms prior to the stimulus was defined as the background activity. We evaluated the TCI duration from the unaffected to the affected hemisphere (TCIunaff-aff) and from the affected to the unaffected hemisphere (TCIaff-unaff). The TCI ratio was defined as TCIunaff-aff/TCIaff-unaff and used to evaluate the balance of both TCIs. In line with this definition, a low TCI ratio indicates that TCIunaff-aff is small relative to TCIaff-unaff.
Brain Stimulation
tDCS was delivered through 2 gel-sponge electrodes (surface area, 25 cm2; IOGEL, IOMED, Salt Lake City, Utah) by a battery-driven constant current stimulator (Phoresor II; IOMED). The anodal electrode was positioned on the affected hemisphere at an optimal site for the FDI, and the other electrode was placed over the contralateral supraorbital area, as described in previous reports.15,16 For the active conditions, patients received 1 mA tDCS for 20 minutes, whereas for sham stimulation, current was applied for 30 s only. Current intensity was gradually increased (at the beginning of the session) and decreased (at the end of the session) to diminish its perception.
We simultaneously applied the 1-Hz rTMS over the unaffected hemisphere and anodal tDCS over the affected hemisphere. In the rTMS-tDCS group, 1-Hz rTMS (90% rMT, 1200 stimuli) was applied over the unaffected hemisphere along with anodal tDCS over the affected hemisphere for 20 minutes. In the tDCS group, anodal tDCS was applied over the affected hemisphere, and sham rTMS was applied over the unaffected hemisphere at the same frequency and intensity used for the rTMS-tDCS protocol. Similarly, in the rTMS group, active rTMS was applied over the unaffected hemisphere, and sham tDCS was applied over the affected hemisphere. In the sham-rTMS method, we used electric stimuli and sound stimulation, as described in a previous report. 17
Statistics
Data analysis was performed by an investigator blinded to the stimulation condition. The data before stimulation (Table 1) were compared between rTMS, tDCS, and rTMS-tDCS groups by analysis of variance (ANOVA) or the χ2 test depending on variable type. The statistical difference on TMS parameters before stimulation between paretic and nonparetic sides was determined by paired t tests. The effects of stimulation and motor training were evaluated using an ANOVA for repeated measures with Time as a within-subjects factor and Condition (rTMS, tDCS, or rTMS-tDCS) as a between-subjects factor. Post hoc analysis was performed with Bonferroni’s correction. Any possible correlation between the changes in the various parameters was assessed using Pearsons correlation coefficient test for exploratory analysis. We performed statistical analyses with bimanual coordination data for 22 patients and other parameter data for 27 patients. All data except bimanual coordination were normalized by conversion to percentage change from the mean values of Pre-Stim.
Results
Participants did not report any adverse side effects during the course of the study. No difference was observed between the rTMS, tDCS, and rTMS-tDCS groups in terms of clinical characteristics, motor functions, or TMS parameters (Table 1). TCIunaff-aff was significantly longer than TCIaff-unaff before stimulation (mean 36.0 ± 10.8 vs 23.3 ± 9.7 ms; P < .001). The amplitude of contralesional MEPs was higher than that of ipsilesional MEPs (mean 1065.2 ± 874.1 vs 565.7 ± 724.9 µV; P = .002). Contralesional rMT was lower than ipsilesional rMT (mean 52.2% ± 9.5% vs 60.0% ± 13.6%; P < .001).
Pinch Force and Acceleration
A repeated-measures ANOVA for pinch force of the paretic hand showed a significant effect of Time (F6,72 = 27.250; P < .001) and interaction between Time and Condition (F6,72 = 2.510; P = .029). The post hoc test did not show a significant improvement in pinch force of the paretic hand immediately after rTMS, tDCS, or rTMS-tDCS. However, motor training induced an improvement in pinch force of the paretic hand following rTMS (Pre-Stim vs Post 2, P = .024) and rTMS-tDCS (Pre-Stim vs Post 2, P = .009) but not tDCS (Figure 2B). These improvements in pinch force of the paretic hand lasted for 7 days after rTMS (Pre-Stim vs Post 3, P = .010) and rTMS-tDCS (Pre-Stim vs Post 3, P < .001). The post hoc test showed no difference in pinch force between the rTMS and rTMS-tDCS groups across all sessions. A repeated-measures ANOVA for acceleration of the paretic hand showed a significant effect of Time (F6,72 = 19.303; P < .001) but not Condition, nor interaction between Time and Condition. Therefore, we did not find any difference between stimulation protocols for acceleration (Figure 2D). A repeated-measures ANOVA for pinch force (Figure 2A) and acceleration (Figure 2C) of the nonparetic hand did not show any significant interaction between Time and Condition; furthermore, no significant effect of Condition or Time was observed.
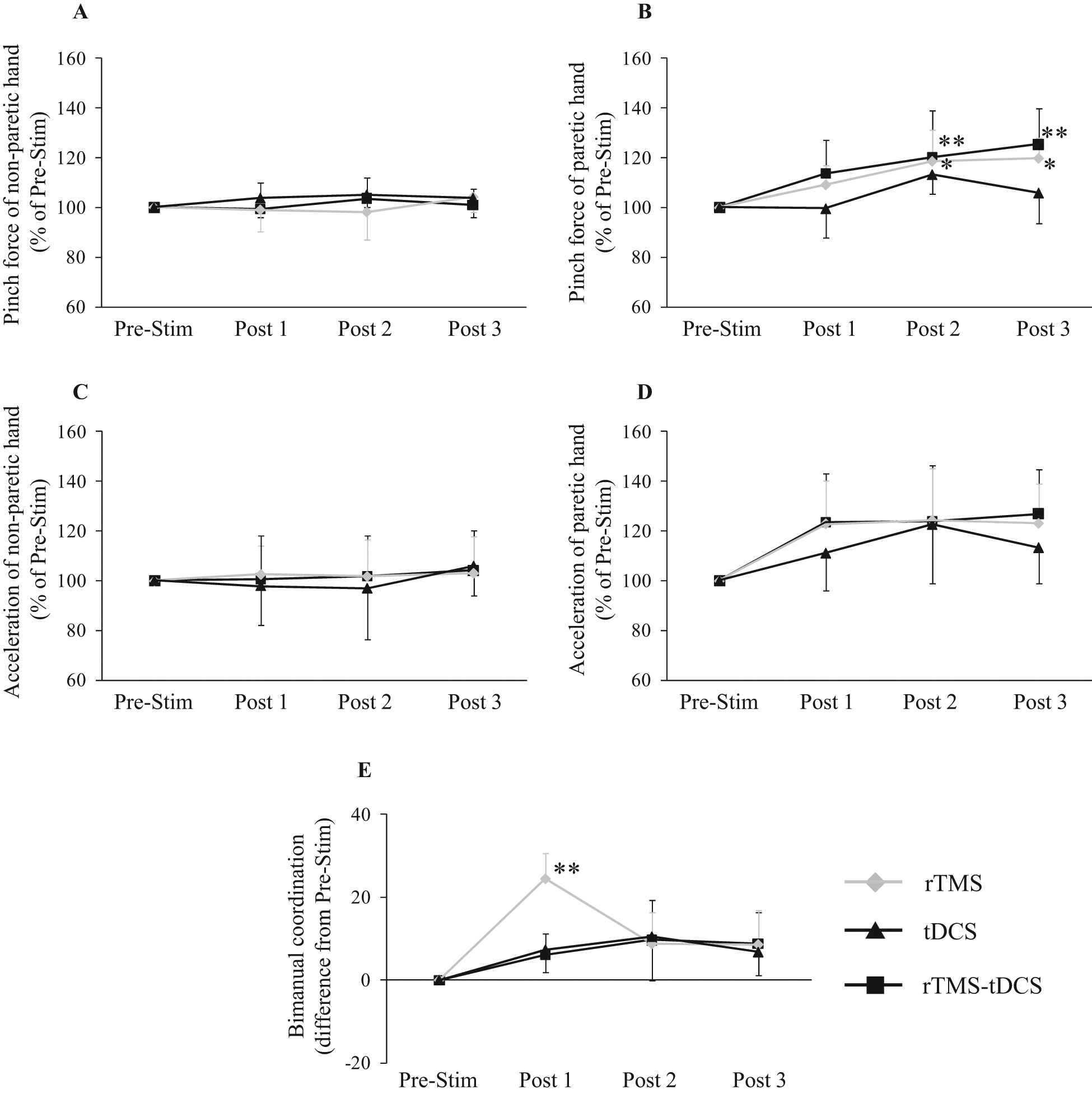
Changes in motor function after stimulation: A. Pinch force of nonparetic hand. B. Pinch force of paretic hand. C. Acceleration of nonparetic hand. D. Acceleration of paretic hand. E. Bimanual coordination. rTMS and rTMS-tDCS protocols significantly improved the pinch force of the paretic hand after stimulation and motor training (Post 2). These improvements in pinch force persisted for 7 days after stimulation and motor training (Post 3). Inhibitory rTMS significantly deteriorated the bimanual coordination (Post 1). However, the rTMS-tDCS protocol prevented the deterioration of the bimanual coordination induced by inhibitory rTMS.
Bimanual Coordination
A repeated-measures ANOVA for bimanual coordination showed a significant effect of Time (F6,57 = 17.208; P < .001) and an interaction between Time and Condition (F6,57 = 5.253; P < .001). The post hoc test revealed that bimanual coordination deteriorated immediately after rTMS (Pre-Stim vs Post 1: P < .001). This change did not last for 30 minutes after rTMS (Figure 2E).
Motor-Evoked Potentials and Resting Motor Threshold
A repeated-measures ANOVA for contralesional MEPs showed a significant effect of Time (F6,72 = 14.878; P < .001) and an interaction between Time and Condition (F6,72 = 2.786; P = .017). The post-hoc test revealed that contralesional MEPs decreased in amplitude immediately after rTMS (P = .003) and rTMS-tDCS (P < .001) but not tDCS (Figure 3A). Changes in contralesional MEPs did not last for 30 minutes after stimulation. A repeated-measures ANOVA for ipsilesional MEPs showed a significant effect of Time (F6,72 = 27.861; P < .001) and an interaction between Time and Condition (F6,72 = 2.295; P = .044). The post hoc test revealed an increased amplitude of ipsilesional MEPs (Figure 3B) immediately after rTMS (P = .002), tDCS (P = .011), and rTMS-tDCS (P = .001). Changes in ipsilesional MEPs lasted for 30 minutes after tDCS (Pre-Stim vs Post 2, P = .020) but not after rTMS or rTMS-tDCS. The change in ipsilesional MEPs did not last for 7 days after tDCS.
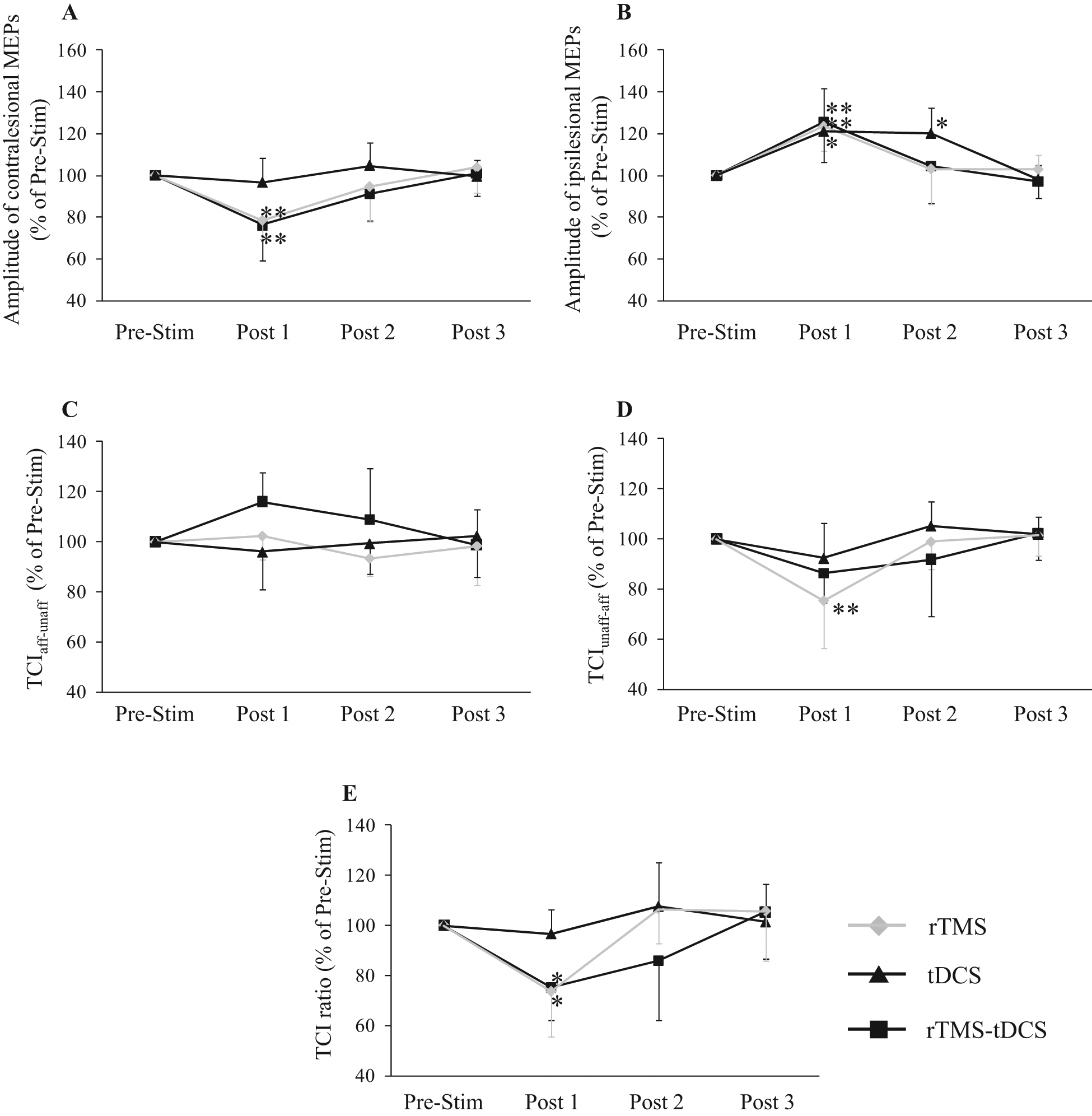
Changes in corticospinal excitability and transcallosal inhibition (TCI) after stimulation. A. Amplitude of contralesional motor-evoked potentials (MEPs). B. Amplitude of ipsilesional MEPs. C. TCI from affected to unaffected hemisphere (TCIaff-unaff). D. TCI from unaffected to affected hemisphere (TCIunaff-aff). E. TCI ratio. The amplitude of contralesional MEPs decreased immediately after rTMS and rTMS-tDCS (Post 1). Changes in contralesional MEPs did not last for 30 minutes (Post 2). The amplitude of ipsilesional MEPs increased immediately after rTMS, tDCS, and rTMS-tDCS (Post 1). These ipsilesional MEP changes lasted for 30 minutes after only t-DCS (Post 2). The change in ipsilesional MEPs did not last for 7 days after tDCS (Post 3). Inhibitory rTMS significantly reduced TCI ratio and TCIunaff-aff; however, rTMS-tDCS significantly reduced the TCI ratio but not TCIunaff-aff (Post 1). These TCI changes did not last for 30 minutes after stimulation (Post 2).
A repeated-measures ANOVA for contralesional rMT showed no significant interaction between Time and Condition; furthermore, no significant effect of Condition or Time was observed (data not illustrated). A repeated-measures ANOVA for ipsilesional rMT showed a significant interaction between Time and Condition (F 6,72 = 4.056; P = .001) but no effect of Time or Condition. The post hoc test did not reveal any change in ipsilesional rMT (data not illustrated).
Transcallosal Inhibition
A repeated-measures ANOVA for TCIaff-unaff showed a significant interaction between Time and Condition (F6,72 = 2.479; P = .031) but no significant effect of Time or Condition. The post hoc test did not reveal any change in TCIaff-unaff after stimulation (Figure 3C). A repeated-measures ANOVA for TCIunaff-aff showed a significant effect of Time (F6,72 = 15.953; P < .001) and an interaction between Time and Condition (F6,72 = 2.271; P = .046). The post hoc test revealed that TCIunaff-aff decreased immediately after rTMS (Figure 3D, P = .001). The change in TCIunaff-aff did not last for 30 minutes after rTMS. A repeated-measures ANOVA for TCI ratio showed a significant effect of Time (F6,72 = 16.470; P < .001) and an interaction between Time and Condition (F6,72 = 4.130; P = .001). The post hoc test showed a significant reduction in TCI ratio immediately after rTMS (P = .019) and rTMS-tDCS (P = .038) but not tDCS (Figure 3E). The TCI ratio changes did not last for 30 minutes after stimulation.
Correlation Between Motor Function and TMS Parameter After Stimulation
In all groups, the change in pinch force of the paretic hand immediately after stimulation (Post 1) showed a negative correlation with TCI ratio (Figure 4A; r = −0.477; P = .012) but not other TMS parameters. The change in acceleration of the paretic hand showed no significant correlation with any TMS parameters. The change in bimanual coordination after stimulation (Post 1) was negatively correlated with TCIunaff-aff (Figure 4B; r = −0.486; P = .022) but not other TMS parameters.
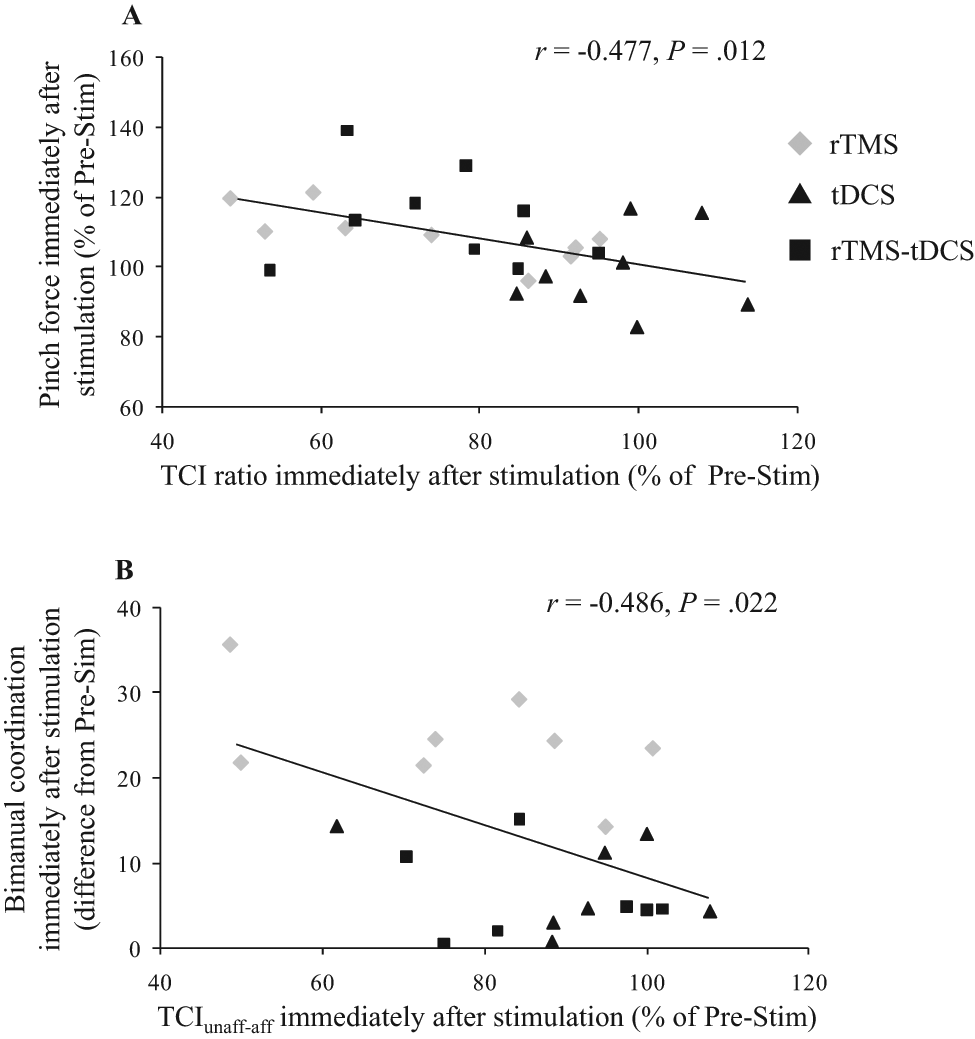
Correlation between transcallosal inhibition (TCI) and motor changes after all stimulation protocols. A. There was a significant negative correlation between the change in pinch force of the paretic hand after stimulation and TCI ratio. B. There was a significant negative correlation between the change in bimanual coordination after stimulation and TCI from the unaffected to the affected hemisphere (TCIunaff-aff). Abbreviations: rTMS, repetitive transcranial magnetic stimulation; tDCS, transcranial direct current stimulation; Pre-Stim, prestimulation.
Discussion
Evaluation of bimanual coordination in antiphase movement can detect a deterioration of bimanual movement after inhibitory rTMS over the unaffected hemisphere. Moreover, this study presents the first evidence that combined administration of rTMS and tDCS can prevent the deterioration of bimanual movement induced by inhibitory rTMS in stroke patients.
Mechanism of Pinch Force Improvement After Stimulation
Inhibitory rTMS over the unaffected hemisphere increased the corticospinal excitability of the affected hemisphere; this effect is thought to be a result of the reduced TCIunaff-aff.7,8 rTMS-tDCS combination also increased the corticospinal excitability of the affected hemisphere. Enhancement of motor cortex excitability by brain stimulation can augment motor learning. 18 Therefore, artificially increasing cortical excitability could facilitate improvements in the pinch force in rTMS and rTMS-tDCS groups after motor training. However, we could not detect pinch force improvement after anodal tDCS over the affected hemisphere that increased the excitability of the affected hemisphere. In addition to enhancement of cortical excitability in the affected hemisphere, another mechanism may contribute to the improvement of the paretic hand after stimulation. In this study, the TCI ratio was reduced after rTMS and rTMS-tDCS but not tDCS. Moreover, the improvement of pinch force in the paretic hand after stimulation was negatively correlated with the TCI ratio. The ratio of TCIunaff-aff to TCIaff-unaff was high before stimulation because the TCIunaff-aff was longer than TCIaff-unaff in our stroke patients. Considering these findings, the reduction of TCI ratio indicated that TCIunaff-aff decreased relative to TCIaff-unaff. It is thought that TCI synapses on inhibitory interneurons. 19 Therefore, relative reduced TCIunaff-aff might induce disinhibition of the affected motor cortex. This disinhibition of the affected motor cortex may partly contribute to the functional improvement in the paretic hand by unmasking the latent networks. 20 However, regardless of the disinhibition hypothesis, we might be unable to detect the improvement of pinch force after tDCS because of the insufficient stimulation intensity of anodal tDCS. Previous studies have reported that high-intensity tDCS had a greater effect on function than low-intensity tDCS.21,22 Similarly, we might be unable to find that the rTMS-tDCS combination has an additional effect compared with rTMS alone.
Changes in TCI and Bimanual Movement After Stimulation
We found that the TCIunaff-aff decreased after inhibitory rTMS over the unaffected hemisphere; this result is consistent with a previous report. 7 However, rTMS-tDCS combination did not significantly decrease TCIunaff-aff. This result may be related to the activation of inhibitory interneurons by anodal tDCS. 23 TCI is likely mediated by transcallosal fibers originating from the motor cortex, which eventually synapse on inhibitory interneurons in the opposite cortex to produce their inhibitory effect. 19 The inhibitory interneurons of the affected hemisphere activated by anodal tDCS might lessen the reduction of TCIunaff-aff induced by inhibitory rTMS in the rTMS-tDCS protocol. However, anodal tDCS alone did not change the TCIunaff-aff in this study. A possible explanation is that the response of the inhibitory interneurons in the affected hemisphere to anodal tDCS is too weak to increase TCIunaff-aff because of the so-called ceiling effect in stroke patients, because TCIunaff-aff in stroke patients is already strong prior to stimulation. 24
The TCI ratio in the rTMS-tDCS groups decreased, despite the smaller reduction in TCIunaff-aff. Although there were no significant effects, rTMS-tDCS had a tendency to increase the TCIaff-unaff (Figure 3C). The changes in both TCIs may have contributed to the reduction in the ratio of TCIunaff-aff to TCIaff-unaff in the rTMS-tDCS groups. A possible explanation for the TCIaff-unaff change in rTMS-tDCS groups is that the affected motor cortex activated by anodal tDCS might more readily inhibit the unaffected motor cortex inactivated by inhibitory rTMS.
Inhibitory rTMS over the unaffected hemisphere deteriorated bimanual coordination and decreased TCIunaff-aff. It has been reported that TCI inhibits the generation of mirror movement14,25 and controls bimanual movement.9,10 Antiphase bimanual movement necessitates mirror movement suppression.26,27 Therefore, given that our TCI evaluation could detect changes in TCI function, inhibitory rTMS may worsen the antiphase bimanual movement by decreasing TCIunaff-aff (Figure 5A). In contrast, the rTMS-tDCS combination lessened the reduction of TCIunaff-aff, and as a result, rTMS-tDCS might prevent the deterioration of bimanual coordination induced by inhibitory rTMS (Figure 5B). This hypothesis is supported by the finding that bimanual coordination changes were negatively correlated with TCIunaff-aff changes. However, regardless of TCI changes, inhibitory rTMS over the unaffected hemisphere might deteriorate bimanual movement by inhibiting ipsilateral motor projection from the unaffected hemisphere. However, the contribution of ipsilateral projection to the hands is small compared with that to the proximal limbs.28,29 Moreover, it has been reported that ipsilateral projection might contribute to the instability of antiphase bimanual movement. 30 Therefore, it is unlikely that inhibitory rTMS deteriorates the antiphase bimanual movement of the hands by inhibiting ipsilateral projection.
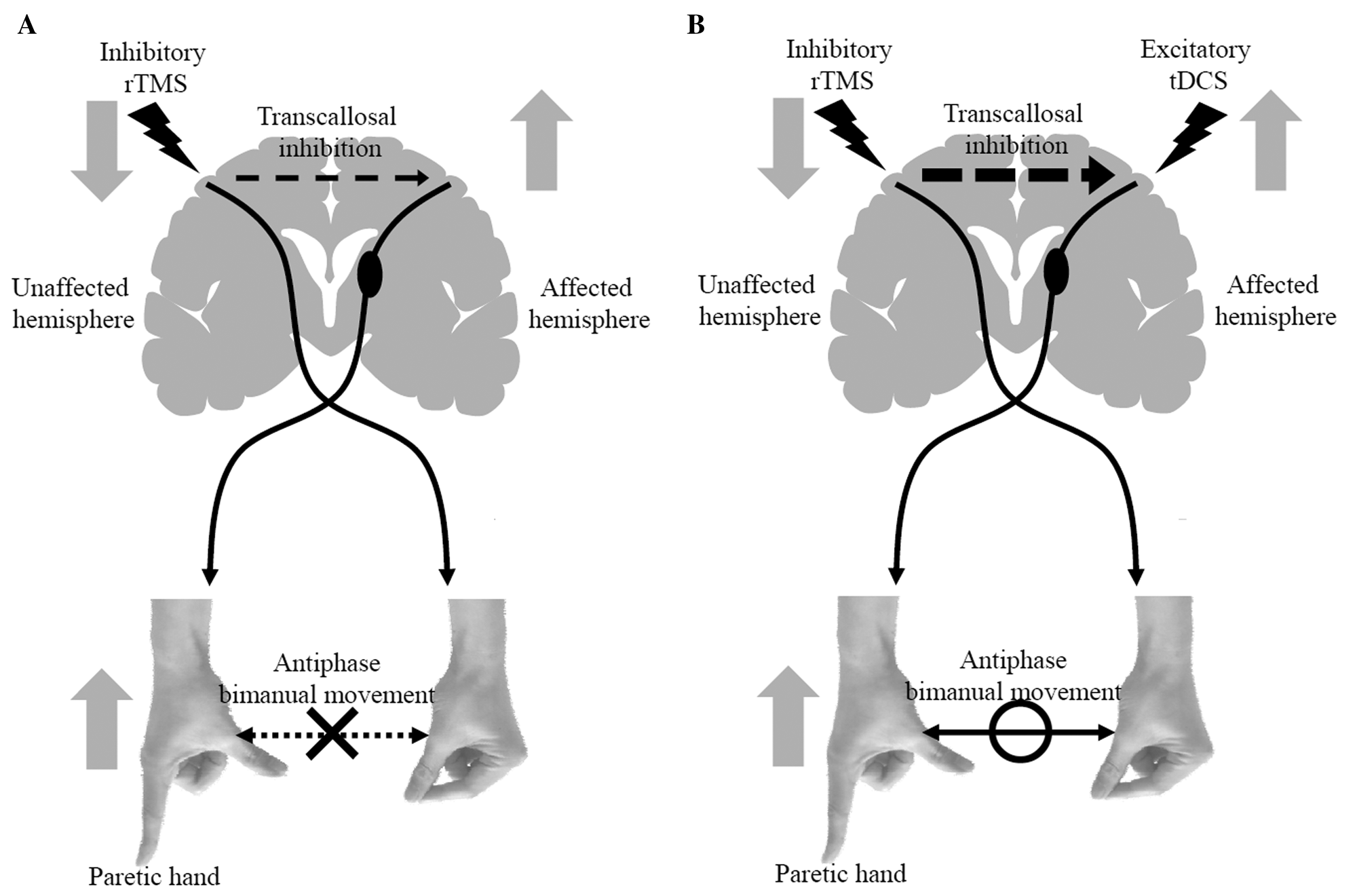
Mechanism of motor function change after brain stimulation in stroke patients. A. Inhibitory rTMS over the unaffected hemisphere: inhibitory rTMS decreased excitability of the unaffected motor cortex and increased excitability of the affected motor cortex by reducing transcallosal inhibition (TCI) from the unaffected to the affected hemisphere. Facilitation of the affected motor cortex improved pinch force of the paretic hand in stroke patients. However, antiphase bimanual movement deteriorated because of reduction of TCI, which controls bimanual movement. B. rTMS-tDCS combination: anodal tDCS along with inhibitory rTMS also decreased excitability of the unaffected motor cortex, increased excitability of the affected motor cortex, and improved pinch force of the paretic hand in stroke patients. rTMS-tDCS combination lessened the reduction of TCI induced by inhibitory rTMS and prevented deterioration of antiphase bimanual movement.
In addition to the lower reduction of TCI, the affected motor cortex directly enhanced by anodal tDCS might have also contributed to the absence of bimanual coordination deterioration in the rTMS-tDCS groups. Our preliminary study using electroencephalogram coherence had indicated that activity in the affected motor cortex might reduce the deterioration of bimanual coordination by rTMS. 11 The enhanced cortical excitability of the affected hemisphere by anodal tDCS might prevent the deterioration of bimanual coordination induced by inhibitory rTMS.
Limitations and Future Studies
In this study, we did not perform functional motor tests that are more relevant to stroke patients.31,32 Moreover, our bimanual coordination evaluation is still an experimental paradigm for stroke patients, and further work is needed to validate the effects of brain stimulation on bimanual movement. Another methodological problem we had in our study was the use only of the single-pulse TMS technique to evaluate TCI. In addition to our method, TCI can also be measured by the paired-pulse technique.33,34 The single-pulse method has been reported to detect changes in TCI after rTMS and motor training in stroke patients.7,35 However, the single- and paired-pulse methods do not represent the same phenomenon, and they should be considered as complementary measures of TCI. 34 Therefore, further investigation is required to clarify the changes seen in TCI after brain stimulation using our method and the paired-pulse method.
Bilateral tDCS methods have been reported to be useful in bihemispheric stimulation, excluding rTMS, in stroke patients.36,37 These bilateral tDCS methods are easy to follow; however, it is possible that inhibitory tDCS over the unaffected motor cortex inhibits the neighboring premotor cortex, which contributes to bimanual movement.38,39 As a result, the inhibitory tDCS over the unaffected hemisphere may deteriorate bimanual coordination regardless of TCI changes. Future studies should perform bilateral tDCS using a small electrode capable of local stimulation 40 to evaluate the effect of bimanual movement.
In conclusion, inhibitory rTMS led to deterioration of antiphase bimanual movement in chronic stroke patients. A combination of inhibitory rTMS over the unaffected hemisphere and excitatory tDCS over the affected hemisphere could prevent the deterioration of bimanual movement induced by inhibitory rTMS in stroke patients. These findings are pertinent to the design and optimization of brain stimulation strategies for stroke patients.
Footnotes
Acknowledgements
The authors would like to thank Mami Onodera for technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or publication of this article: this work was supported by a research grant from the Akiyama Life Science Foundation (NT).
