Abstract
Background
Phantom limb pain (PLP) is a common and in some cases debilitating consequence of upper- or lower-limb amputation for which current treatments are inadequate.
Objective
This small clinical trial tested whether game-like interactions with immersive VR activities can reduce PLP in subjects with transtibial lower-limb amputation.
Methods
Seven participants attended 5–7 sessions in which they engaged in a visually immersive virtual reality experience that did not require leg movements (Cool! TM), followed by 10–12 sessions of targeted lower-limb VR treatment consisting of custom games requiring leg movement. In the latter condition, they controlled an avatar with 2 intact legs viewed in a head-mounted display (HTC Vive TM). A motion-tracking system mounted on the intact and residual limbs controlled the movements of both virtual extremities independently.
Results
All participants except one experienced a reduction of pain immediately after VR sessions, and their pre session pain levels also decreased over the course of the study. At a group level, PLP decreased by 28% after the treatment that did not include leg movements and 39.6% after the games requiring leg motions. Both treatments were successful in reducing PLP.
Conclusions
This VR intervention appears to be an efficacious treatment for PLP in subjects with lower-limb amputation.
This study has been registered at clinicaltrial.org with the title “Virtual Reality Treatment for Phantom Limb Pain,” registration number: NCT03338842
Introduction
Loss of limb is a common problem; almost 2 million people in the US have had an amputation. 1 Up to 90% of people with limb amputation experience the persistent sensation of the missing extremity, a phenomenon known as a “phantom limb” (PL). 2 Additionally, a significant proportion of individuals with a phantom limb—up to 85% in some studies—experience persistent and in some cases debilitating pain in the missing limb, a condition known as phantom limb pain (PLP). Although existing therapies provide pain relief in some cases, there is widespread agreement that current approaches fall short of bringing relief to most individuals with PLP. 2
Reflecting the many manifestations of PLP, a substantial number of theories regarding the cause of the disorder have been proposed, 3 falling into 2 major classes. The first emphasizes the role of the peripheral nervous system in the genesis of PLP and suggests, for example, that PLP may be the consequence of neuromas at the site of resection. Although this cause seems unlikely for PLP in all individuals, there is little doubt that peripheral factors contribute to PLP in some. 4 The second group of theories attributes PLP to changes in the central nervous system. Studies in both animals5,6 and humans7,8 have indicated that sensory cortex that had been responsive to input from the amputated or deafferented body part begins to respond to contiguous body parts. The “maladaptive plasticity” hypothesis suggests that cortical reorganization in primary sensory and motor cortex after amputation induces PLP. In particular, Flor and colleagues9,10 argued that PLP is associated with contiguous cortical regions invading the cortex representing the deafferented limb. They propose that this functional takeover occurs because of unmasking previously silent pathways or because of the creation of new cortical connections. In support of this claim, Lotze et al 10 reported that severity of PLP was correlated with the degree to which motor cortex for the face (indexed by lip movements) “invaded” motor cortex formerly devoted to the missing limb. Makin and colleagues,11-13 did not observe a relationship between PLP severity and shift of the face motor cortex,12,13 and other researchers have found that the representation of the missing limb is largely preserved.14,15 In the Makin studies, PLP was associated with reduced functional connectivity between brain areas13,16 or increased activity in somatosensory areas contralateral to the amputated limb. 12 Recent data from Raffin et al 14 suggest that the cortical remapping and functional connectivity accounts are not mutually exclusive, as it has been noted that intact cortical representation of the missing limb can coexist with the invasion of the neighboring areas. 14
Recognizing that no single account explains all of the complexities of the PLP syndrome, Ortiz-Catalan 3 proposed that PLP is caused by the “stochastic entanglement” of pain and somatosensory-motor circuitry. Following this hypothesis, amputation induces a chaotic state in the brain, which leads in a probabilistic manner to co-activation of connected brain networks. In normal circumstances, only a noxious stimulus induces the conjoint activation of pain, somatosensory, and motor networks. According to the stochastic entanglement hypothesis, the unstable state generated by the amputation leads to co-activation of the networks in the absence of noxious stimuli. Variability in PLP occurrence and characteristics across individuals is assumed to depend on the strength of the association between brain networks.
A variety of treatments targeting the sensorimotor cortices have been assessed in small studies. Mirror therapy may be the most extensively studied intervention. In mirror therapy for lower-limb PLP, participants attempt to move both legs in tandem, and they see the image of the intact limb reflected in a mirror at the location of the amputated limb, which tends to provide the sensation that the amputated limb is still present and under their control. This treatment is based on the suggestion that PLP results from the lack of correspondence between motor commands and proprioceptive and visual feedback from the amputated limb.15,17 Motor commands continue to be generated after the amputation, but the commands do not result in the anticipated sensory feedback in the absence of the effector. The mismatch between the predicted and estimated sensory states is interpreted as an error signal that (for reasons that are not explicated) induces PLP. Vision of the “amputated” limb executing the planned movement, as seen in the mirror, is assumed to reduce PLP by providing visual feedback for the movement, thereby eliminating the mismatch between action plans and sensory feedback.18-20 It has been shown in healthy individuals that in conditions where there is a mismatch between vision and somatosensory input, such as the rubber hand illusion, vision is predominant and action performance is generated based on visual rather than somatosensory feedback (e.g., Soechting and Flanders, 1989 21 ; Ambron et al, 2020 22 ). Therefore, manipulating vision of the limb may influence both action planning and the experience of body ownership.
Virtual reality (VR) has also been used to treat PLP. Some VR treatments are designed to mimic mirror therapy, so the apparent movement of the amputated limb follows the action of the intact limb.23-27 In other studies, the visually depicted “movement” of the amputated extremity is controlled by myoelectric recording from the distal amputated limb,28-31 surface EMG,32-34 or motion capture24,35,36 recorded from the residual limb.
Virtual Reality has advantages relative to mirror therapy. As interventions may take the form of diverse games rather than repetitive movements, subjects may be more likely to participate in therapy. Furthermore, VR provides participants with direct control of the motion and sense of agency of the phantom. 37 Perhaps most importantly, the movements of the 2 legs need not be mirrored in VR treatments, enabling a broader variety of interactions.
We report our experience from a small clinical trial. In a previous study, 38 we developed a VR system, which used immersive VR and inertial sensors mounted on participants’ intact and residual limbs to recreate and control the missing limb and intact limb independently. This system was tested in two individuals with lower-limb amputation, both of whom demonstrated pain reduction immediately after each session and across sessions. 38 The study reported here uses an updated version of our system to follow up these preliminary results in a larger sample of different individuals with lower-limb amputation and test the effect of the treatment over a larger period of time.
Seven participants underwent a 9-week-long treatment protocol consisting of 2 treatment phases. In the first phase (distractor treatment), participants were exposed to a VR environment (Cool!, https://firsthand.com/health-software/) that has been demonstrated to improve chronic pain 39 but that lacks the visual and auditory feedback of movement of an imputed “amputated” limb. After 5–7 sessions of Cool!, participants underwent 10–12 sessions of lower-limb treatment, in which they played a series of games using their virtual limb. The missing limb was virtually reconstructed by creating an avatar with two intact legs and visually superimposing it on the participant’s real body. Magnetic motion-tracking sensors placed above and below the knee on the participant’s intact and residual limbs tracked the real position of the limbs independently. These motions were used to control the avatar’s leg movements in the virtual world, providing participants with the sensation of control over the movements of both limbs. We predicted that both treatments would reduce PLP, but that the magnitude and duration of the benefit would be larger in the lower-limb VR than in the distractor treatment. Indeed, we expected the two treatments to differentially interact with PLP: the distractor treatment was expected to have a calming effect and move participants’ attention away from the pain; the limb treatment, in contrast, was expected to at least partially restore feedback of the missing limb, thereby reducing the mismatch between the motor command and visual feedback that may generate PLP.
Methods
Participants
Inclusion criteria were: participants had suffered a below knee (transtibial; stump length greater than 10 cm) amputation of 1 leg at least 4 months prior to enrollment and rated their average PLP as ≥4 over the preceding 4 weeks on a 10-point Likert scale. Exclusion criteria were: history of stroke, moderate to severe traumatic brain injury (operationally defined as loss of consciousness for more than 30 minutes), significant cognitive impairment (≤23 on the Montreal Cognitive Assessment 40 ), or symptoms of a current major psychiatric disorder. Patients continued their pharmacologic pain regimen without change during the study.
Participants were recruited at Moss Rehabilitation Research Institute, the Corporal Michael J. Crescenz Veterans Affairs Medical Center and Penn Medicine via referral from physical therapists and distribution of flyers. Fourteen potential subjects were identified; 4 were excluded because of cognitive impairment, 2 withdrew from the study after the first session (before the active VR treatment) because of time constraints. Eight participants were included in the study. One of those did not present with pain but rather with “uncomfortable” sensations that resolved completely after a single session; her data are not included here. The final sample was composed of seven participants. Two of these participants (SUBs 1 and 6) did not complete the final limb treatment sessions for personal reasons. One participant (SUB 4) withdrew after the 1-week follow-up session for unrelated medical reasons.
General Procedure
Our primary dependent variable was the average of the intensity ratings for the ‘current’ pain reported at the beginning and end of each session as measured by the Visual Analog Scale (VAS) of the short form of the McGill Pain Questionnaire. 41 Our secondary dependent variable was intensity of pain averaged across two ratings in each session as measured by the Brief Pain Inventory (BPI). 42 As scores on these two measures were very highly correlated (rho = .95, P<.001) and redundant, we report only the McGill VAS, but results for the BPI were qualitatively similar. Data were analyzed at the single case and group level.
Prior to beginning the treatment protocol, participants completed a battery of questionnaires measuring different aspects of PLP (i.e., intensity and quality), the impact of pain in daily life, and psychological well-being (i.e., depression and anxiety). This battery was presented again on 4 other occasions: immediately after the distractor phase treatment, immediately after the lower-limb phase treatment, and at 1, 4, and 8 weeks after completion of the entire treatment.
After this initial assessment, participants started the treatment, which included two phases: an initial distractor treatment phase and a subsequent lower-limb treatment phase. We used a multiple-baseline design: the total number of sessions in the distractor and limb treatment phases was constant across participants, but the number of sessions of distractor and limb treatment varied across participants (see Figure 2). Treatment sessions were delivered twice weekly.
Repeated Battery Data
In addition to the pain assessments performed at the beginning and end of each session, the protocol included a testing battery conducted prior to and after each of the two treatments. The battery comprised the pain interference subtest of the Brief Pain Inventory, the 12-Item Short Form Health Survey, 2 the Frenchay Activities Index, 3 the Hospital Anxiety and Depression Scale, 4 and the 13-item Pain Catastrophizing Scale. 5 The 12-Item Short Form Health Survey investigates how physical and emotional problems interfere with individuals’ work-related or personal activities. The Frenchay Activities Index provides information regarding the frequency with which individuals undertake activities of daily living such as meal preparation or hobbies. The Hospital Anxiety and Depression Scale 4 assesses the presence and degree of anxiety and depression, and the Pain Catastrophizing scale 5 measures attitudes toward pain.
Virtual Reality Equipment
The VR treatments were presented with an HTC Vive TM head-mounted display, which provides a three-dimensional view of a virtual environment (see Figure 1). For the lower-limb treatment, custom software was used to generate an avatar with 2 complete legs that participants controlled. Information about the orientation of the upper and lower segments of each of the subject’s legs was provided by a 3D electromagnetic motion-tracking system (trakSTAR, Ascension Technologies Inc., Burlington, Vermont) with four 6DoF electromagnetic sensors; sensors were attached above and below the knees on both legs using Velcro straps. The sensors were applied during the distractor treatment, but data were not recorded. Participant using our system and VR games used in the treatments. From the upper-left panel, Quest for Fire (denoted on the experimenter console as “Chair”), Dog Food (“Doggie Feeder”) (upper row), Chess and Web Browser (lower row).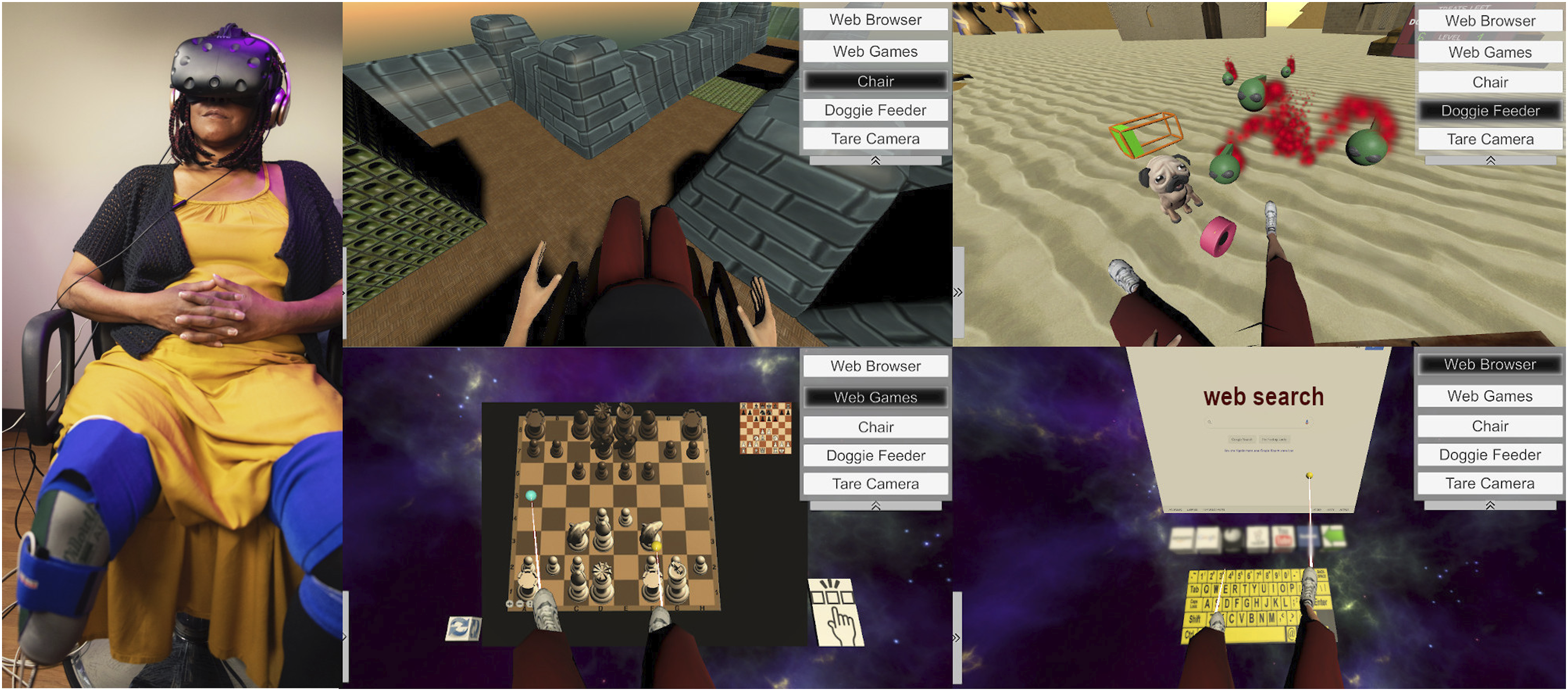
Distractor Treatment
The program Cool! (DeepStream VR Inc, 2014) delivers a VR environment in which participants navigate through a pleasant VR environment while listening to relaxing music, served as the distractor treatment. Participants do not see a rendering of their body and make no movements with their legs but can use their arms to make bubbles or feed seals. Each session in the Cool! environment lasted 55 minutes.
Limb Treatment
In this treatment, participants were actively using the motion of their residual leg and intact leg to control virtual limbs; our software calculated and displayed the configuration of each virtual leg from the orientation of the sensors on the thigh and the remaining portion of the corresponding leg below the knee. Both virtual legs were displayed intact, with the hip joints and knee joints controlled by the participant’s movements. Participants were presented with the following four games as shown in Figure 1: (1) Quest for Fire: participants use their virtual legs to maneuver a wheelchair around a virtual environment. The objective of the game is to access a fire separated from their avatar by a chasm. To reach the fire, the avatar must push blocks into chasms, thereby providing a traversable path. There are 18 levels of difficulty achieved by making the blocks more difficult to access and maneuver, and the environment more complex. (2) Dog Food: the participants’ task is to feed a puppy with candies and use their avatar legs to push away green insect-like invaders that are trying to eat the candy and approach the puppy on the floor near their legs. The degree of difficulty of the game is manipulated by increasing the speed and number of invaders. (3) Web Browser: participants use their virtual legs to navigate the internet by moving the cursor and typing on a virtual keyboard. This activity requires precise and controlled movements and provides access to the entire contents of the web, so that participants are able to locate material that interests them. (4) Chess, Checkers, Solitaire, Spades, 2048, and War Light: these games were available to all subjects and required them to move pieces or cards using their legs.
Auditory feedback regarding participants’ leg movements was provided during the games. For example, participants could hear the sounds associated with pushing blocks in the Quest for Fire game.
In each session, participants were required to play Quest for Fire or Dog Food for at least 30 minutes; these games required the continuous use of both legs. After that, they were free to continue with any game they preferred to play until the end of the 55-minute session.
Assessment of the VR System
During the third session of both the distractor and limb treatments, participants were asked to complete the Simulator Sickness Questionnaire 43 to test for possible side effects of VR (fatigue, nausea, headache, eye strain, difficulties in focusing, etc.) using a four-point scale (none, slightly, moderate, or severe). In addition, participants completed the System Usability Scale 44 to evaluate the degree of perceived complexity and usability of the VR system and the brief Slater-Usoh-Steed Presence Questionnaire 45 to rate their sense of being in the virtual environment.
Data Analysis
Data were analyzed at the single case and group levels. For each subject, we computed the average of the pain ratings collected at the beginning and end of each session to obtain a mean pain score that was used in both levels of analysis.
At a single-subject level, these scores were visually displayed and inspected for every participant; furthermore, to assess the effect of treatment for each individual, we used a variation of Cohen’s d statistic 46 that provides single-subject measures of effect sizes based on a comparison between the distractor and limb treatment, qualified by the distractor standard deviation (SD). As we were interested in obtaining an index for the limb treatment with respect to the distractor treatment, we computed the Cohen’s d based on the average pain ratings in the limb treatment minus the average pain ratings in the distractor treatment divided by the SD of the distractor treatment. We interpreted the magnitude of effect size following Beeson and Robey 47 : Cohen’s d of 4 represents a small effect size, 7 a medium, and 10.1 a large effect size.
At a group level, we used permutation analyses to compare the session pain ratings between the first and last sessions of the distractor treatment, the session pain ratings from the first and last sessions of the limb treatment, and the first and last sessions of the entire (distractor + limb) treatment. An additional permutation test was carried out to compare the magnitudes of the effects of the two treatments (difference between the first and last session of the distractor treatment vs difference between the first and last session of the limb treatment). Permutation tests are often considered optimal for detecting differences in small samples. 48 One subject (SUB 5) was not included in the permutation analysis comparing the limb treatment and the distractor treatment as her pain ratings reached zero during the distractor treatment, so there was no opportunity to observe effects of the limb treatment.
For the test battery data conducted prior to and after the distractor and limb treatments, permutation analyses were used to test changes in scores resulting from the overall treatment protocol (difference between pre-distractor and post-limb treatments) as well as the distractor and limb treatments separately. Furthermore, a permutation analysis was performed to contrast the effects of the distractor and limb treatments, computed as the difference between pre- and post-treatment data for each subject. Note that for the battery data, we report data uncorrected for multiple comparisons throughout; results should be interpreted cautiously but are included as they may be of interest for future research.
Results
Single-Subject Analyses
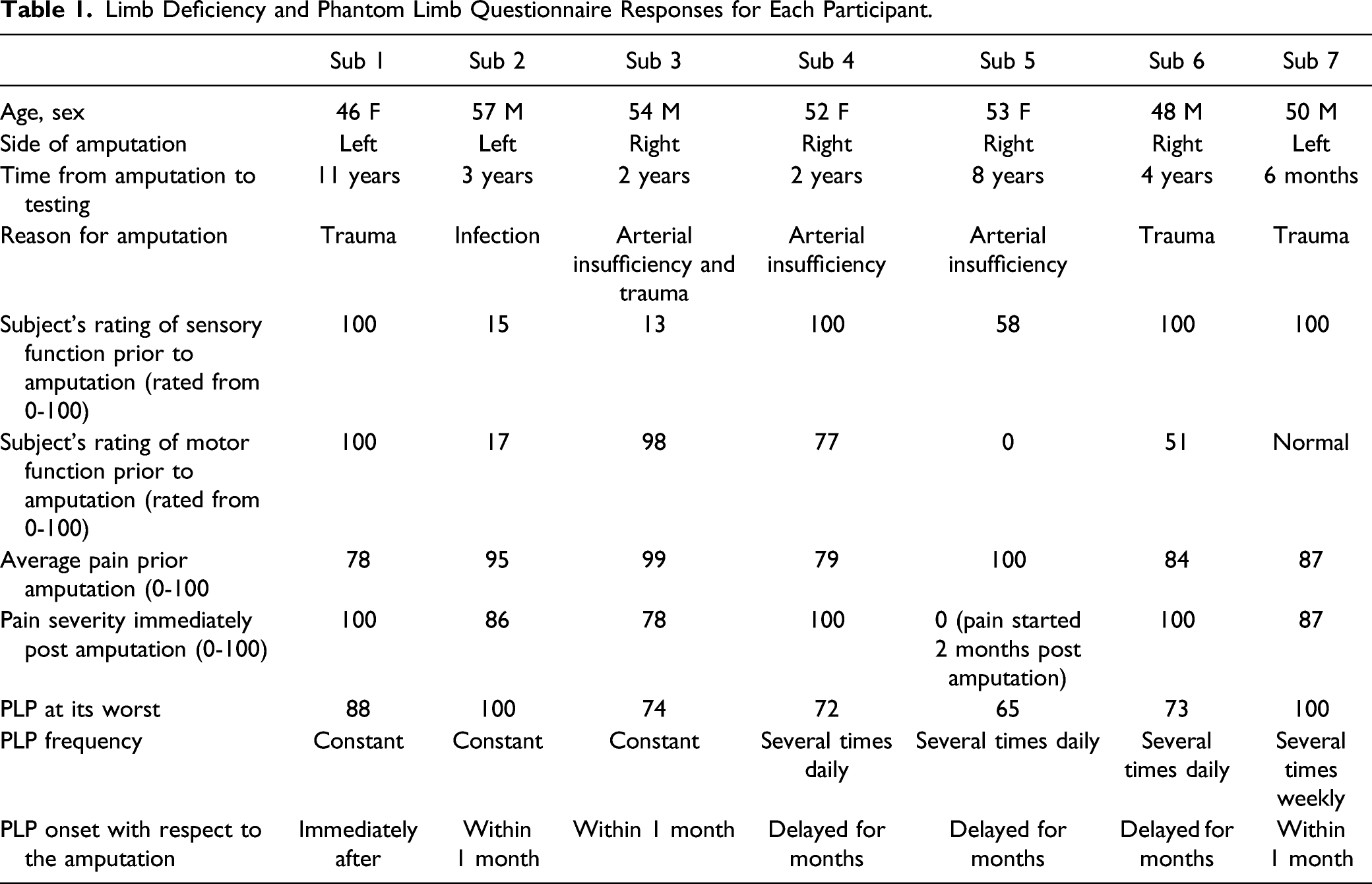
Limb Deficiency and Phantom Limb Questionnaire Responses for Each Participant.
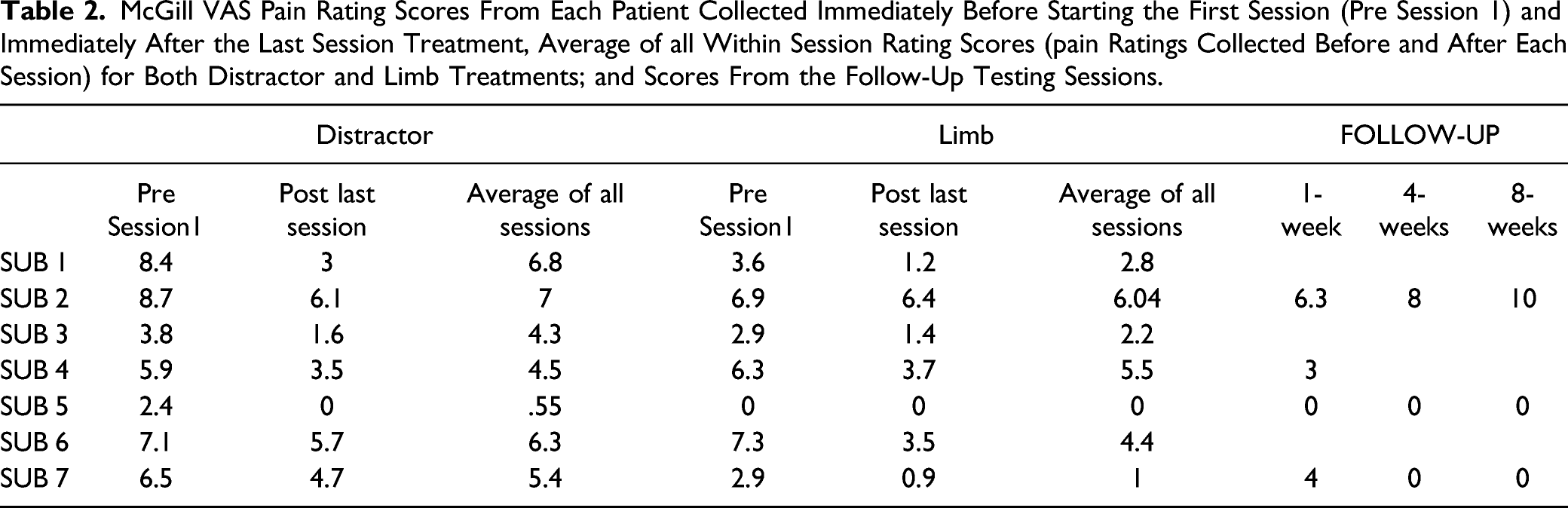
The McGill VAS pre- and post-session and average for each session of the distractor and limb treatments for every subject are presented in Figures 2 and 3. Table 2 reports the average pain scores in the distractor and limb sessions for each subject. Cohen’s d for the difference between the two treatments was 5.35 (small-moderate effect) in one subject (SUB 7) favoring the limb treatment; the remaining subjects showed non-significant effect sizes for the difference between distractor and limb therapies. Pain scores on the McGill VAS for each participant in each session. McGill VAS Pain Rating Scores From Each Patient Collected Immediately Before Starting the First Session (Pre Session 1) and Immediately After the Last Session Treatment, Average of all Within Session Rating Scores (pain Ratings Collected Before and After Each Session) for Both Distractor and Limb Treatments; and Scores From the Follow-Up Testing Sessions.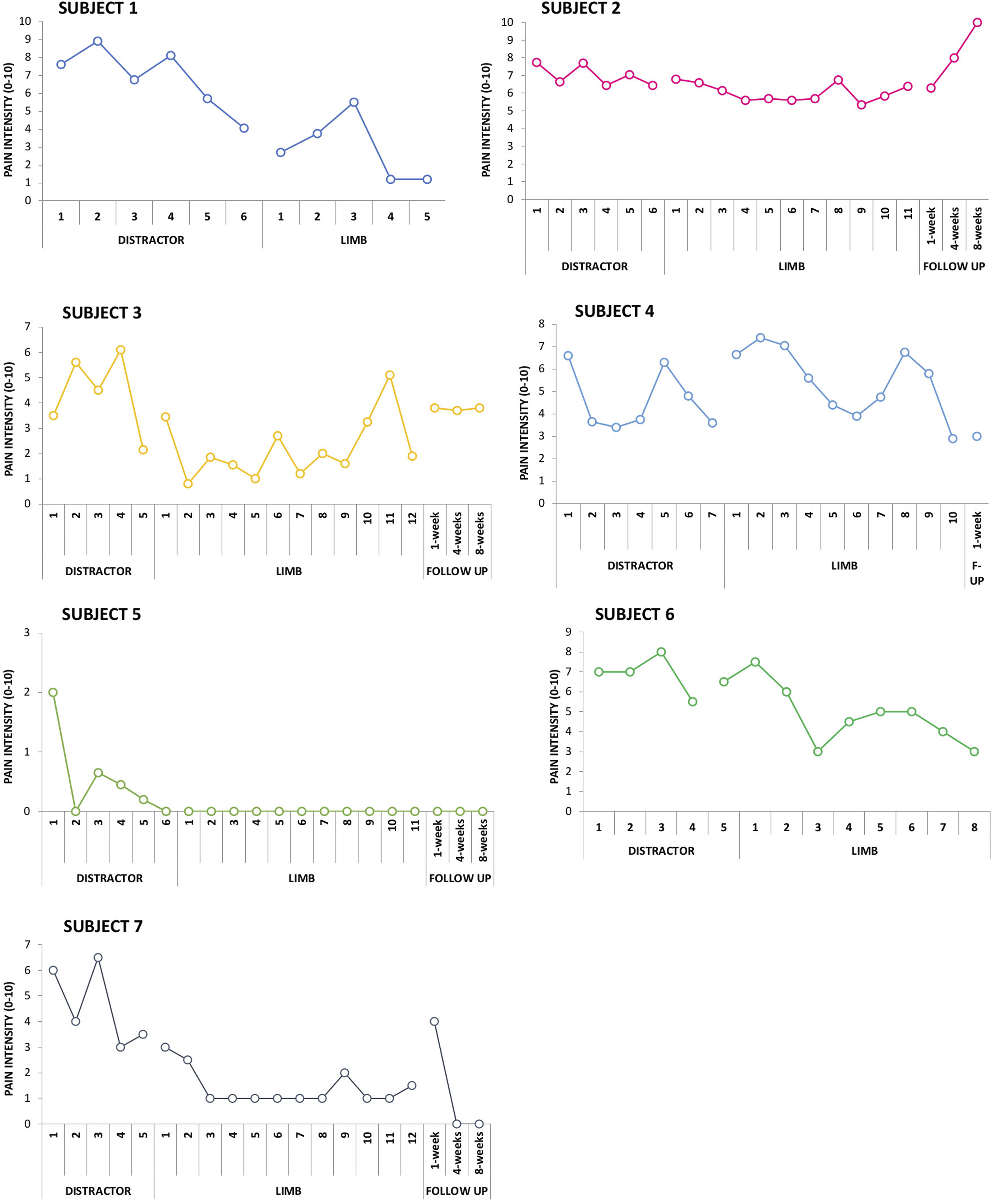
Group Analysis
Participants’ pain numerically decreased from the first (M = 53.9, SE = 8.6) to the last (M=38.7, SE=8.6) distractor session (n=7; absolute change −15.2; relative change −28%); and from the first (M=44.5, SE=7.6) to the last (M=26.9, SE=8.1) limb session (n=6; absolute change −17.6; relative change −39.6%). Permutation analyses showed a significant reduction of pain after both distractor (P=.046) and limb (P=.015) treatments, but only the comparison between pre- and post-limb treatment survived Bonferroni correction. No significant difference was observed in the comparisons between treatments (pain reduction following distractor treatment M=14.33, SE=7.9; following limb treatment M=17.58, SE=4.47; n=6; P=.37). (Figure 3) Pain scores on the McGill VAS for each participant pre and post each session.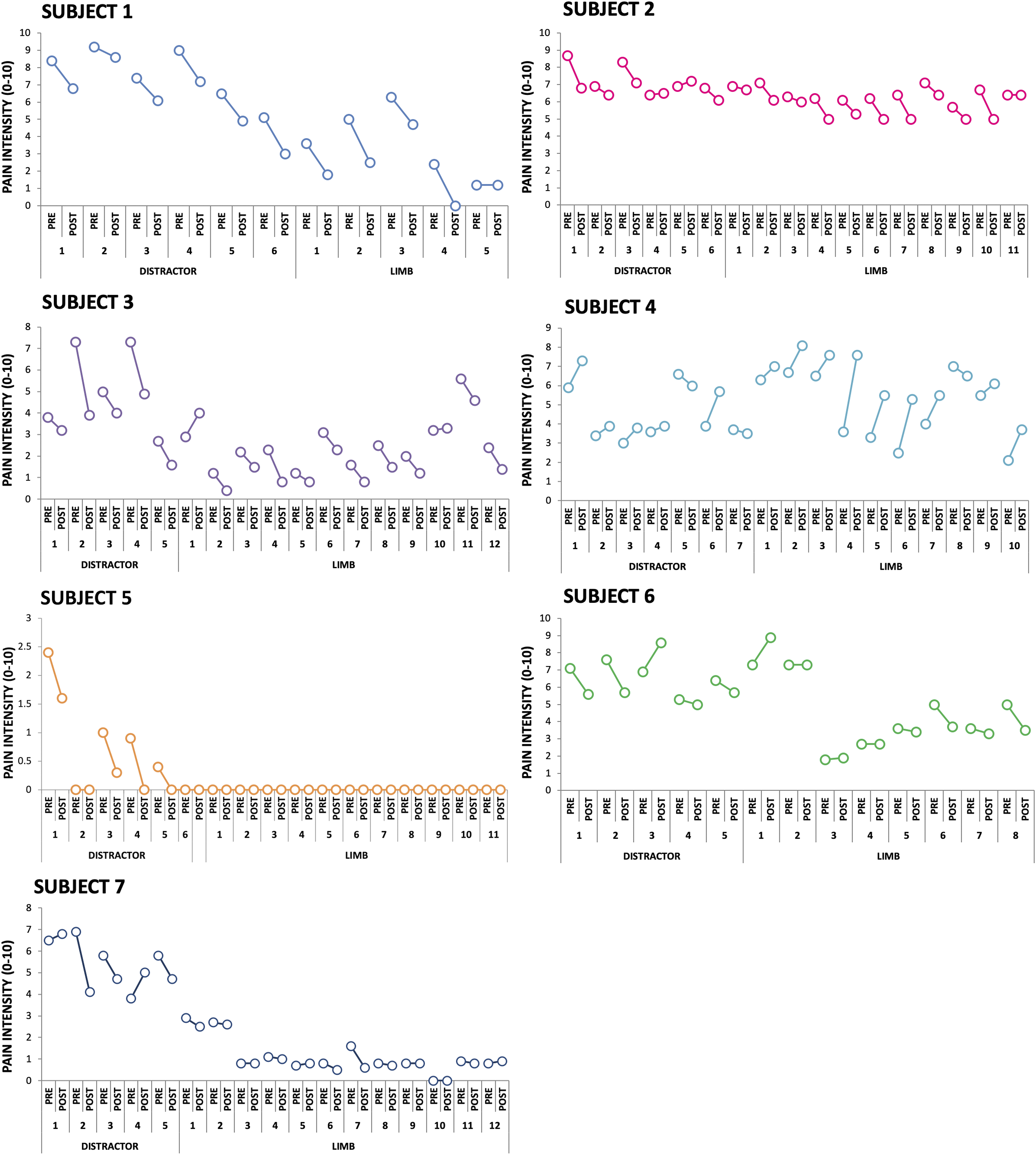
Virtual Reality Assessment
The games were well liked and created a sense of “presence”—that is, a feeling of being in the environment (see Figure 4). While there was not a significant difference across the activities in either presence or usability questionnaires, the ratings were generally satisfactory; no subject expressed dissatisfaction with respect to usability. Average rating scores for the presence questionnaire (left panel) and system usability scores (right panel).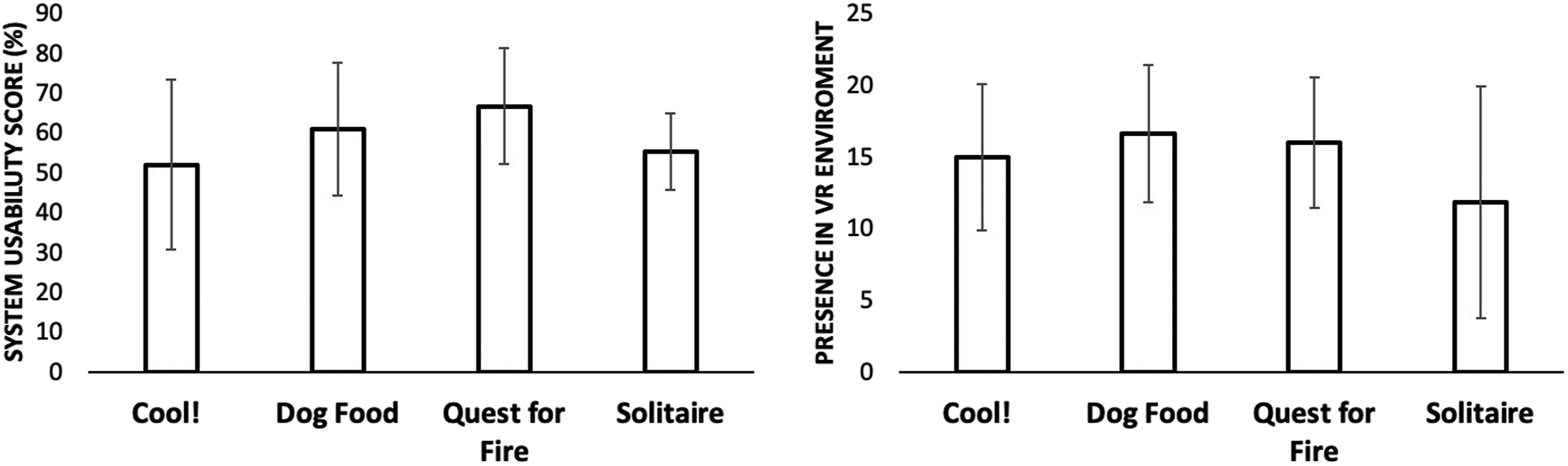
We encountered no significant adverse effects. Several subjects experienced mild cybersickness, an expected and non-significant adverse effect that did not cause any subject to withdraw from the study; for SUBs 1 and 4 this was noted in the first 2 sessions. We slightly modified our VR display after these subjects and noted only mild and transient cybersickness in one subsequent subject. SUB 6 reported mild, generalized headache with the VR therapy that resolved spontaneously; his headache was subsequently attributed by his primary care physician to sinusitis.
Repeated Battery Data Analysis
From the pre-distractor time point to the post-limb treatment time point, there was a 39% reduction in BPI pain interference scores (P=.06, n=5) and a 61% reduction in the HADS depression scores (P=.06, n=5).
Average Scores for Repeated Battery Assessments. Cells Highlighted in Gray Indicate the Significant (Dark Gray) or Trend (Light Gray) Differences Within Treatment Comparisons (i.e., Pre-Post).
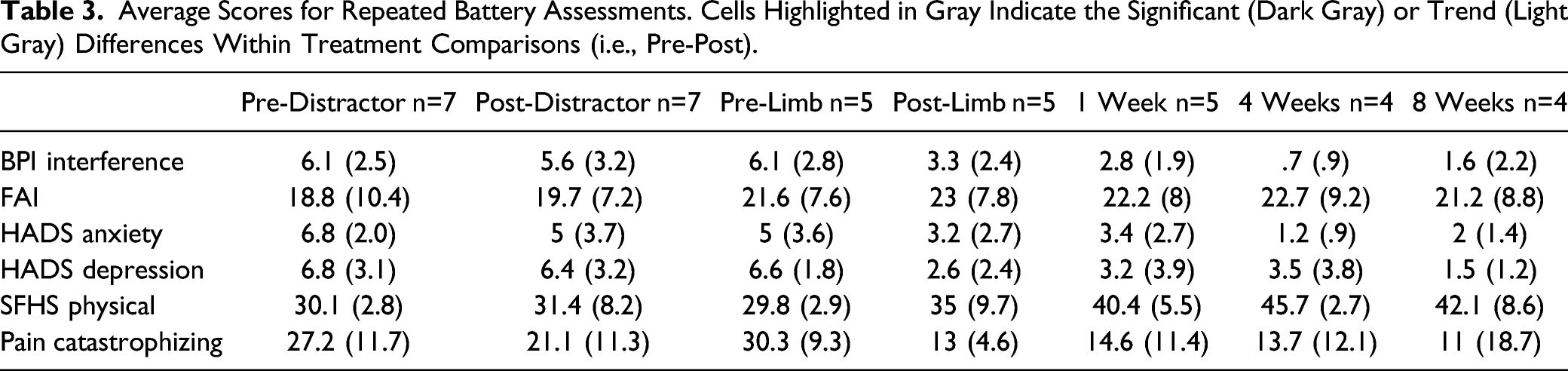
Finally, we ran permutation tests comparing the magnitude of treatment effects for pre-vs post-limb treatment as compared to pre-vs post-distractor treatment (n=5). For the BPI interference score, the decrease in scores was larger for the limb treatment (28%) than for the distractor treatment (15%; P=.03).
Discussion
A VR intervention comprising a combination of distractor and active limb treatments was successful in reducing lower-limb PLP in most participants. Group analyses showed a reduction of pain on our primary outcome measure after both treatments. Individual analyses demonstrated that the limb therapy was superior to distractor treatment in one subject, but the two treatments did not differ in other subjects. In line with previous clinical trial interventions with PLP,30,33 some improvement was observed in the majority of individuals who took part in the present study (85% of our sample); Subject 4 demonstrated a numerical pattern of increased pain during the intervention that may have been related to active systemic lupus erythematosus during that time. The activity required in both treatments may have thus triggered musculoskeletal discomfort in this specific individual. Interestingly, despite the within-session increase in pain, this individual showed an overall decrease in her pain severity across both treatments, suggesting that VR therapy was not detrimental.
Group-level analyses revealed a significant reduction of pain after both distractor and limb treatments. Although the two treatments showed similar positive effects on PLP, only the effect of the limb treatment survived Bonferroni correction, suggesting a more robust effect of this specific treatment.
This study explored the effect of VR in a group of lower-limb PLP amputees in which treatment was provided over the course of many sessions and followed up weeks after completion of treatment. The present study is distinguished from previous work with lower-limb amputees by the use of immersive VR rather than augmented reality 37 ; additionally, we report data from group analyses rather than single cases. 32 Furthermore, our VR treatment differed from some studies in that we did not employ repetitive and structured motor32,37 training; rather, participants were presented with a variety of virtual games that engaged the use of both limbs in a naturalistic fashion. However, it is unclear whether naturalistic activity or structured motor training is more beneficial in reducing PLP; future work should explore this question. Game-based interventions such as those evaluated in the present study have the advantage of being entertaining and engaging for patients. In a randomized controlled trial study, Rothgangel et al 37 assessed the effects of mirror therapy after 4 weeks of clinical training. PLP patients treated with mirror therapy showed a reduction in pain that was not statistically different from the control group, which was offered a sensorimotor treatment targeting the intact limb. In the same study, the authors also assessed the effect of classical or augmented reality-based mirror therapy after 10 weeks and 6 months of home-based training; there was not a significant effect of either treatment on PLP relative to the control group at either time point. In contrast, Lendaro et al 32 obtained results more comparable with the present study; they found a 68% progressive reduction in PLP across 12 weeks of VR treatment that remained evident at 1- and 3-months follow-up but not at 6 months. The treatment consisted of a set of motor exercises to be executed in VR in which measurements from surface electromyography controlled the motions of a visually displayed limb, which caused subjects to feel as though they were controlling their missing leg. We chose a magnetic motion-tracking system over electromyography to simplify system setup and more accurately display real limb movements in VR.
Two previous studies tested the effectiveness of multiple sessions of VR treatment in individuals with upper-limb amputation.30,50 These studies demonstrated a reduction of PLP intensity across treatment sessions30,50 that, in the report of Ortiz–Catalan, 30 persisted for several months. In conjunction with these studies and the report of Lendaro et al, 32 our data suggest that multiple VR sessions provide a benefit that increases over sessions and, in some subjects, persists for weeks to months.
Several factors are likely to have led to an underestimation of the potential benefit of the active limb VR treatment in our study. First, all patients except one had been suffering from PLP for many years and had tried multiple alternative treatments (medication, mirror therapy) with little or no success. Only 1 participant (SUB 7) started the treatment within 6 months of amputation. This individual showed a strong reduction of pain intensity (from 6–7/10 at the beginning of the treatment to 1/10 at the end of the treatment) and frequency (from several times a week to once a month at the end of the treatment). Although they must be interpreted with caution, these data suggest that the efficacy of VR treatment should be explored early in the course of the disorder. We are unaware of studies that have addressed the effect of VR therapy in an acute or sub-acute setting.
A second factor that may have led to an underestimation of the effect of limb VR therapy is that all subjects received an alternative therapy prior to limb VR therapy. Indeed, we observed a positive effect for both distractor and limb treatments. PLP was reduced by the distractor treatment, and the limb treatment produced additional pain reduction. As the limb treatment always occurred after the distractor treatment, it is difficult to partition out the effect of the limb treatment from the overall VR protocol and the placebo effect. However, both individual and group data suggest that the beneficial effect of the limb treatment was somewhat larger than that of the distractor treatment. Additional studies with larger samples will be required to further assess this possibility.
Although both the distractor and limb treatments produced positive results, we speculate that they differ with respect to mechanism. It is likely that the distractor therapy reduced pain at least in part by diverting attention from the pain39,51; as there was a reduction between sessions, however, this account does not adequately explain the benefit and suggests that the relaxing VR environment may have a persistent down-regulating effect on pain perception.
The pain reduction with the limb treatment is likely to rely upon an additional mechanism, namely the provision of visual feedback that is able, in part, to substitute for the missing proprioceptive and sensory afferent information. Indeed, our system aimed at reducing the mismatch between the intact motor commands and visual feedback from the amputated limb. The evidence of a significant reduction of PLP after the limb treatment supports the hypothesis that PLP is related, in part, to the incongruence between predicted and expected sensory feedback and that the restoration of the impaired representation of the limb can reduce PLP. The results of Lendaro et al 32 are consistent with this speculation.
In addition, the battery data revealed a numerically beneficial effect of the limb treatment in the reduction of depression (HADS) and in pain interference with daily life (BPI). A decrease in PLP interference with daily activities had previously been observed in a clinical study with upper-limb PLP 30 ; we provide evidence of this effect in subjects with lower-limb amputation. Although the small sample size mandates a cautious interpretation, the effects are encouraging. These data suggest that being able to see and have control of the amputated limb may improve attitudes towards pain, help in the daily management of pain and improve emotional sequelae of pain.
There were no significant or unanticipated adverse effects of the VR treatment. Two subjects experienced moderate cybersickness when playing the Quest for Fire game. This game might have increased motion sickness because participants rotated 360° in the VR environment using only the motion of their legs; the VR background moved accordingly, while their actual body remain stationary. Indeed, previous work had suggested that the lack of correspondence between absence of self-motion, provided with vestibular information, and visual motion generate cybersickness, proposing the control and reduction of scene movement as a possible way to reduce this symptom. 52 Although descriptive, we note that the limb treatment was engaging and entertaining. Several participants were not keen to stop the limb treatment, as they wanted to continue to play; several asked if they could purchase our limb treatment.
We note that the games used in the limb treatment sessions received adequate scores with respect to “presence” and system usability; in fact, scores were slightly higher for our games such as Quest for Fire and Dog Food as compared to the commercially available Cool! Efforts to enhance these games are underway.
Our study suffers from a number of limitations, including the small sample size and the fact that only subjects with below-the-knee amputations were included. Feedback from some potential participants suggested that the number of visits to the lab was an impediment to participation. We are currently exploring home-based treatments in individuals with PLP with above- and below-knee amputations.
Another limitation of the present study is that the distractor treatment, which we hypothesized at the outset would have smaller effects than the limb treatment, was always presented first. This choice was predicated on the anticipated small sample size, which was insufficient to assess order effects as would be required had we used a crossover design with balancing of order across subjects. Additionally, the design we used quasi-randomly assigns subjects to baselines (or initial treatments) of staggered lengths such that changes after the switch point between treatment A and B can be attributed to the effect of treatment rather than the passage of time or non-specific practice effects. 53 This design is optimal to use with randomization tests and permutation analyses, which assess whether the obtained data could have occurred in Phase A or B by chance. Moreover, we did not include a washout period. We are therefore unable to assess whether the apparent benefits of the limb treatment are instead attributable to additive effects of both treatments. Future studies should investigate the long-term effects of both treatments independently.
Finally, as both the distractor and active VR treatments reduced PLP, one might speculate that the pain reduction was attributable to a placebo effect. We elected to compare active VR therapy to a treatment demonstrated to reduce pain (Cool!) to ensure a stringent test of VR therapy. The fact that active VR treatment reduced pain to a greater degree than distractor therapy by some measures argues against—but does not disprove—an account based on a general placebo effect. Future studies, including those currently planned, will address the role of placebo effects in VR therapy for PLP.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIH Clinical Center (R21NS099645).
