Abstract
Background. Patients who suffer from phantom limb pain can perceive tactile stimuli applied to the cheek on their affected side as if it were coming from their phantom limb, a phenomenon called “referred sensation.” Objectives. To investigate the analgesic effect produced by tactile feedback provided to the cheek during neurorehabilitation using visual feedback. Methods. Nine participants with phantom upper limb pain performed virtual reality neurorehabilitation exercises in which they repeatedly touched a target object with a virtual representation of their affected limb. We applied tactile feedback to their cheek when their virtual affected limb touched a virtual object (Cheek Condition). We also included 2 control conditions where tactile feedback was either applied to their intact hand (Intact Hand Condition) or not applied at all (No Stimulus Condition). We evaluated pain intensity on an 11-point rating scale and pain quality using the short-form McGill Pain Questionnaire before and after each rehabilitation condition. Results. The median pain-reduction rate in the Cheek Condition (33.3 ± 24.4%) was significantly higher than in the Intact Hand Condition (16.7 ± 12.3%) and the No Stimulus Condition (12.5 ± 13.5%; P < .05). Even patients who did not feel referred sensations reported significant pain reduction after the Cheek Condition. Conclusions. The analgesic effect of neurorehabilitative visual feedback during phantom limb movement is significantly improved by applying somatosensory feedback to the cheek on the affected side. Further studies are needed to extend these findings to objective pain measures and to elucidate the neural mechanisms that underlie the analgesic effect.
Keywords
Introduction
Following the amputation of an extremity, nearly all adult individuals will perceive phantom sensations originating from the body part that is now absent. 1 Fifty to eighty percent of patients are reported to experience painful phantom sensations following an amputation, 2 a phenomenon known as phantom limb pain (PLP). Although the mechanisms underlying PLP are still unclear, it has been revealed that the cortical representation of the affected upper limb within the primary somatosensory cortex (S1) shrinks and that the adjacent brain area (eg, the face or shoulder area) expands, effectively taking over areas once dedicated to the affected upper limb.2-4 Although several studies clearly show that the functional reorganization of the S1 somatotopic map is positively correlated with PLP,2,3,5,6 a recent study indicates that PLP is also associated with the preserved S1 cortical representation of a phantom hand. 7 Treatment options for PLP have generally been limited, and there is no clear consensus regarding an optimal treatment method.
Mirror visual feedback (MVF) therapy has been proposed as a means to alleviate PLP.8-10 It has been reported that MVF therapy can be an effective treatment option for PLP caused by either a brachial plexus avulsion injury or by a limb amputation. 11 The analgesic effect of MVF is thought to arise from the reestablishment of congruent sensorimotor integration via voluntary motor imagery of the phantom limb.12-14 However, the analgesic effect of MVF remains limited, 15 possibly because the visual feedback does not induce sufficient phantom limb motor imagery.16,17 Recent advancements in virtual reality (VR) and augmented reality technology could provide new means to induce greater phantom limb motor imagery.18-21
We have developed a multimodal virtual reality MVF (VR-MVF) system that enhances the reality of a patient’s experience by providing them an opportunity to interact, through a virtual affected upper limb, with virtual objects that generate simultaneous visual, auditory, and tactile feedback. 21 Because amputees could not feel stimulation on their affected hands, tactile stimuli were applied to the intact hand, which was thought to be effective means to provide feedback because of intermanual referral of tactile sensations. 22 However, in some participants, applying tactile stimuli to the intact hand did not improve the analgesic effect compared with when no tactile feedback was provided. Therefore, further research is required to determine the optimal body target to apply tactile feedback during VR-MVF rehabilitation.
Illusory perceptions are fairly common in PLP patients. For instance, Ramachandran et al reported that touching the cheek or shoulder of patients on their affected side can generate the illusory sensation that their phantom limb has been touched.4,23 This phenomenon is known as “referred sensation,” and it is reported that its underlying mechanism involves the functional reorganization of the brain.2,23,24 Some patients feel as if “their fingers are buried in their face,” whereas others feel referred sensations only when their cheek is touched. 23 In addition, referred sensations can also be induced artificially by demonstrating a slight conflict between visual and tactile information. 25 Visuotactile illusions adapting referred sensations have been reported to modulate the experience of pain,16,26 and VR technology can, therefore, be used to enhance the therapeutic benefit of MVF because it enables patients to easily experience these illusions.
Based on this illusory phenomenon, we hypothesized that stimulating the cheek of patients during VR-MVF therapy could induce vivid visuotactile illusions, which could then reduce pain more effectively than standard MVF therapy sessions that only provide visual feedback.
Methods
Participants
Nine patients suffering from PLP (P1-P9) participated in this study. Patient sociodemographic and clinical data are shown in Table 1. The cause of their PLP was either due to brachial plexus avulsion or due to arm amputation. The patients experienced both neuropathic pain, such as electrical shock pain and burning pain, and kinesthetic pain. No participant was in an acute stage. Participants with brachial plexus avulsion had very limited voluntary movement of their affected limbs, and even reported having difficulty imagining moving them.
Sociodemographic and Clinical Participant Information.
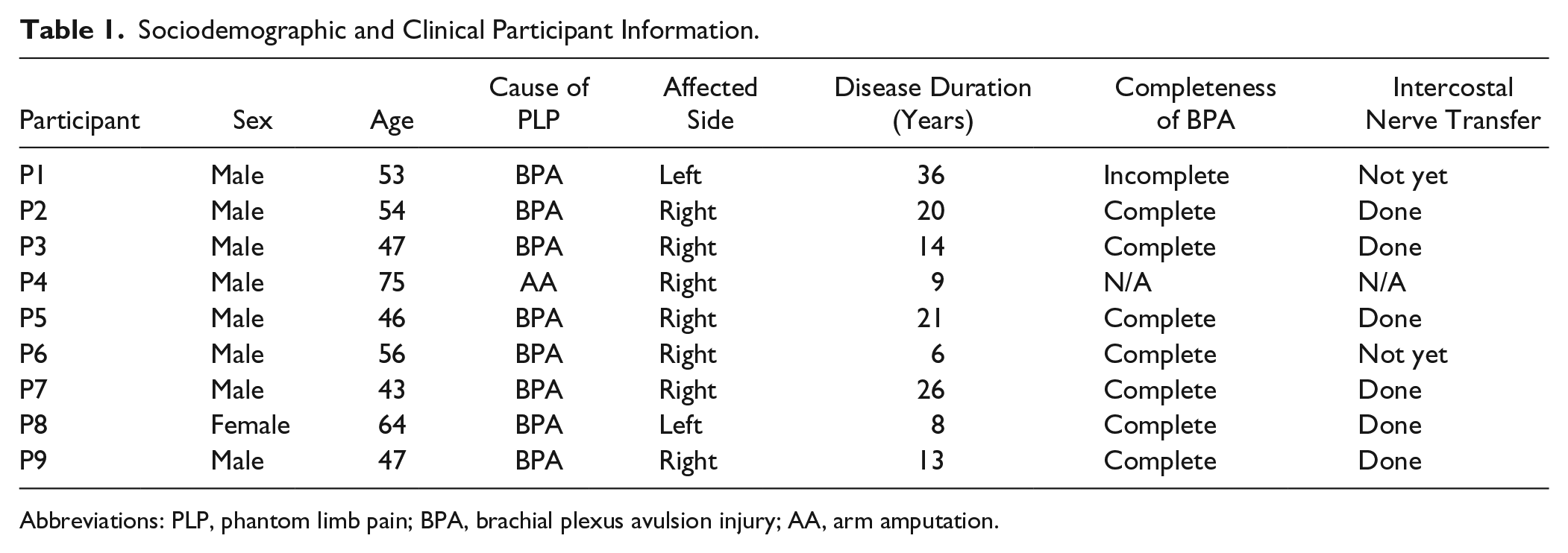
Abbreviations: PLP, phantom limb pain; BPA, brachial plexus avulsion injury; AA, arm amputation.
All participants were outpatients in the Anesthesiology and Pain Relief Center at The University of Tokyo Hospital. The study was approved by the Ethical Review Board of the Faculty of Medicine at The University of Tokyo and was conducted in accordance with the regulations of the ethical review board. We explained the nature and purpose of the study to all participants and obtained their written consent prior to their enrolment. This study is registered as a clinical trial in the University Medical Information Network (UMIN; trial ID: UMIN000017129).
Neurorehabilitation System
We used an adapted and improved version of the neurorehabilitation system originally developed by Wake et al.21,22 The Kinect sensor (Microsoft Corp, Redmond, Washington) was used to detect the movements of the intact limb, and an acceleration sensor embedded in an immersive head-mounted display (Oculus Rift; Oculus VR, Menlo Park, California) was used to detect the rotation angle of the head. The movements of the intact limb and hand were symmetrically converted to a mirror-reversed image that was displayed as the virtual affected upper limb in the VR environment. Participants could see both the virtual affected and the virtual intact upper limbs on the screen of the head-mounted display (Figure 1).
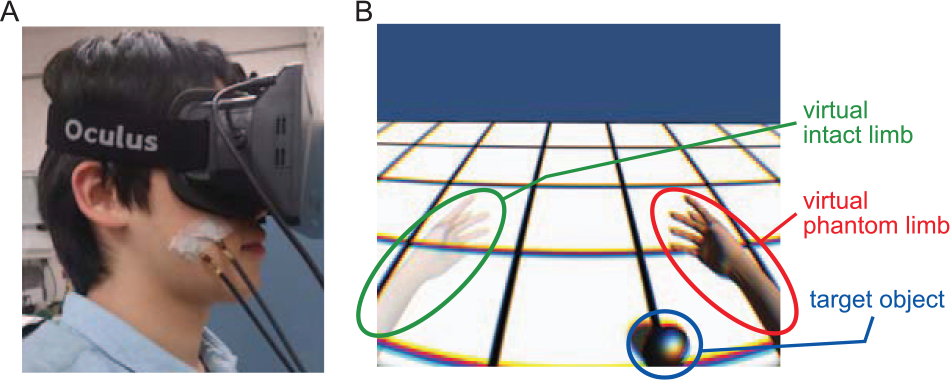
Experimental setup and the virtual reality (VR) environment. (A) A participant wearing a head-mounted display with vibrating motors attached to the cheek (or the intact hand) using skin-friendly surgical tape. (B) The VR environment. The left virtual hand corresponds to the intact limb and the right virtual hand to the affected hand. When the virtual affected hand, moving in a mirror-symmetrical manner to the intact virtual hand, touches the target object, it then disappears while auditory and tactile feedback is provided.
Tactile stimuli were provided by applying vibrating motors to the skin surface (4F442, T.P.C. DC Motor, 12 mm in diameter), the same type as those commonly embedded in mobile phones, that were controlled by an Arduino Uno circuit board (Arduino SRL, Torino, Italy) and that can be placed on any body part with surgical tape. When the virtual affected upper limb was stimulated in the VR environment, the motors were simultaneously activated for 150 ms. This stimulation duration is appropriate in terms of vibrotactile sensitivity. 27 Auditory stimuli were provided to participants through earphones, and events in the VR environment were accompanied by a stereo tone that lasted 0.5 to 1.5 seconds. Because participants were required to wear the head-mounted display, they could not see their surroundings. Therefore, to ensure their safety, we did not place any objects around or near the participants.
Protocol
Tasks
Participants were asked to “touch” virtual target objects appearing in the VR environment (Figure 1) with their virtual affected upper limb by moving their intact upper limb. When the virtual affected hand touched a target object, auditory (a collision sound) and tactile (vibration of the motor) feedback stimuli were presented. To promote congruent sensorimotor integration, the participants were asked to attempt to move both upper limbs symmetrically during the task.
Experimental Procedure
In a quiet room, participants were asked to sit in front of the Kinect sensor. Before beginning the rehabilitation procedure, each participant was interviewed to assess the following pain indicators: (a) level of pain on an 11-point rating scale (Numeric Rating Scale [NRS] 28 : 0 = no pain and 10 = severe pain); (b) quality of pain using an adapted Japanese version of the short-form McGill Pain Questionnaire 1. 29 We also assessed whether participants experienced “referred sensations” based on the method described in McCabe et al 30 : we provided tactile stimuli to the cheek, shoulder, and stump of each participant, in a random order, first with their eyes closed and then a second time with their eyes open. After each tactile stimulation, participants were asked to describe the sensation emanating from it.
The participants first had the opportunity to practice the different tasks before beginning the rehabilitation procedure. Because the tasks required a fair amount of concentration, we limited the duration of each task to 5 minutes.
The tasks were performed under 3 different conditions: (a) the “Cheek Condition,” (b) the “Intact Hand Condition,” and (c) the “No Stimulus Condition.” The latter 2 conditions served as control conditions. In the Cheek Condition, the vibrator was attached to the cheek on the participants’ affected side. When they touched a target object with their virtual affected hand in the VR environment, a synchronous tactile stimulus was applied to the cheek. In the Intact Hand Condition, the vibrator was attached to the participants’ intact hand. When they touched a target object with their virtual affected hand, a synchronous tactile stimulus was applied to the intact hand. This condition replicates the condition used by Sano et al, where the experimenters applied tactile stimuli to the patients’ intact hand to investigate the effectiveness of tactile feedback for reducing pain, and for improving the sense of agency. 22 When participants virtually touched a target object with their virtual affected hand in the No Stimulus Condition, no tactile feedback was delivered. Although previous studies were able to induce referred sensations of the phantom upper limb via tactile stimulation of the affected shoulder or the stump of the amputated limb,16,26,31,32 we did not include a similar condition. This was because some of our participants felt strong pain when their affected shoulder was touched, and because participants with complete brachial plexus avulsion could not perceive tactile stimulation of the affected limb. All participants were evaluated over 2 to 4 days, and 2 to 3 sessions were conducted on each day. The number of sessions performed varied between participants, and was based on several factors, including their medical condition, their degree of fatigue, their motivation level, and time constraints (Supplemental Table 1). The intersession interval was at least 5 minutes. Because these short intervals could result in a carryover effect between sessions, all conditions and sessions were distributed in a randomized order over multiple days. After each rehabilitation session, we asked the same interview questions that were asked before the session.
Analyses
As shown in Supplemental Table 1, the number of sessions performed by each subject also varied among the different experimental conditions. To exclude the potential influence of this difference, we removed excess sessions for each subject until each condition had an equal number of sessions performed. To avoid selection bias, we removed sessions based on the chronological order that they were performed.
The comparison of NRS scores across conditions was used to estimate the pain relief provided by each rehabilitation condition. Pain reduction rates were calculated as a percentage of the baseline measure: ([pre-rehabilitation NRS − post-rehabilitation NRS]/pre-rehabilitation NRS) × 100. We used the Wilcoxon signed-rank test to evaluate if pain measures in a given condition were significantly reduced. The Friedman test was used to examine whether the pain reduction rate was significantly different across the 3 conditions. If P < .05, we then applied a Bonferroni correction for pairwise comparisons. Effect sizes (r values) were calculated by dividing the test statistic by the square root of the number of observations 33 ; r > .5 generally indicates a large effect size. Furthermore, we used the Mann-Whitney U test to determine whether the presence of referred sensations altered the analgesic effect in each condition.
The pain quality was evaluated with an adapted Japanese version of the short-form McGill Pain Questionnaire 1. We categorized pain descriptions into 2 main types as proposed by Melzack 34 : sensory pain (eg, throbbing pain, stabbing pain, and sharp pain) and affective pain (eg, tiring-exhausting pain, fearful pain). We calculated the pain reduction rate for each pain type and performed the same statistical analyses as those used for the NRS scores.
Results
All data below are expressed as the median score ± quartile deviation (QD).
Presence of Referred Sensations
Four of the 9 participants (P1, P5, P7, and P8) reported feeling referred sensations. One participant (P1) had a detailed somatotopic map of his phantom hand on his cheek. Another participant (P5) had a detailed somatotopic map on a relatively wide range of body parts: his phantom palm was felt on his cheek, and his phantom little finger and forearm were felt on his neck and occipital area, respectively. Although the other 2 participants also felt referred sensations when tactile stimuli were applied to their cheek, a detailed somatotopic map could not be established.
Phantom Limb Pain Reduction Rate
NRS scores were measured both before and after each intervention condition and the average pain reduction rates are shown in Tables 2 and 3. Pain was significantly reduced in the Cheek (P = .004) and Intact Hand (P = .016) conditions. The box-and-whisker diagrams illustrating the pain reduction rates, calculated using NRS scores, are presented in Figure 2. Statistical analyses showed that the pain reduction rates differed between conditions (Friedman test, χ2 = 14.8, P = .0006). Pairwise comparisons showed that the pain reduction rate in the Cheek Condition was significantly higher than in the Intact Hand (P = .018) and No Stimulus (P = .0006) conditions. The pain reduction rate obtained for the Intact Hand Condition did not differ from that obtained for the No Stimulus Condition (P = 1.00). The r values, which indicate the treatment effect sizes, were .68 (Cheek Condition vs No Stimulus Condition), .68 (Cheek Condition vs Intact Hand Condition), and .26 (Intact Hand Condition vs No Stimulus Condition).
NRS Scores Before and After Each Intervention a .
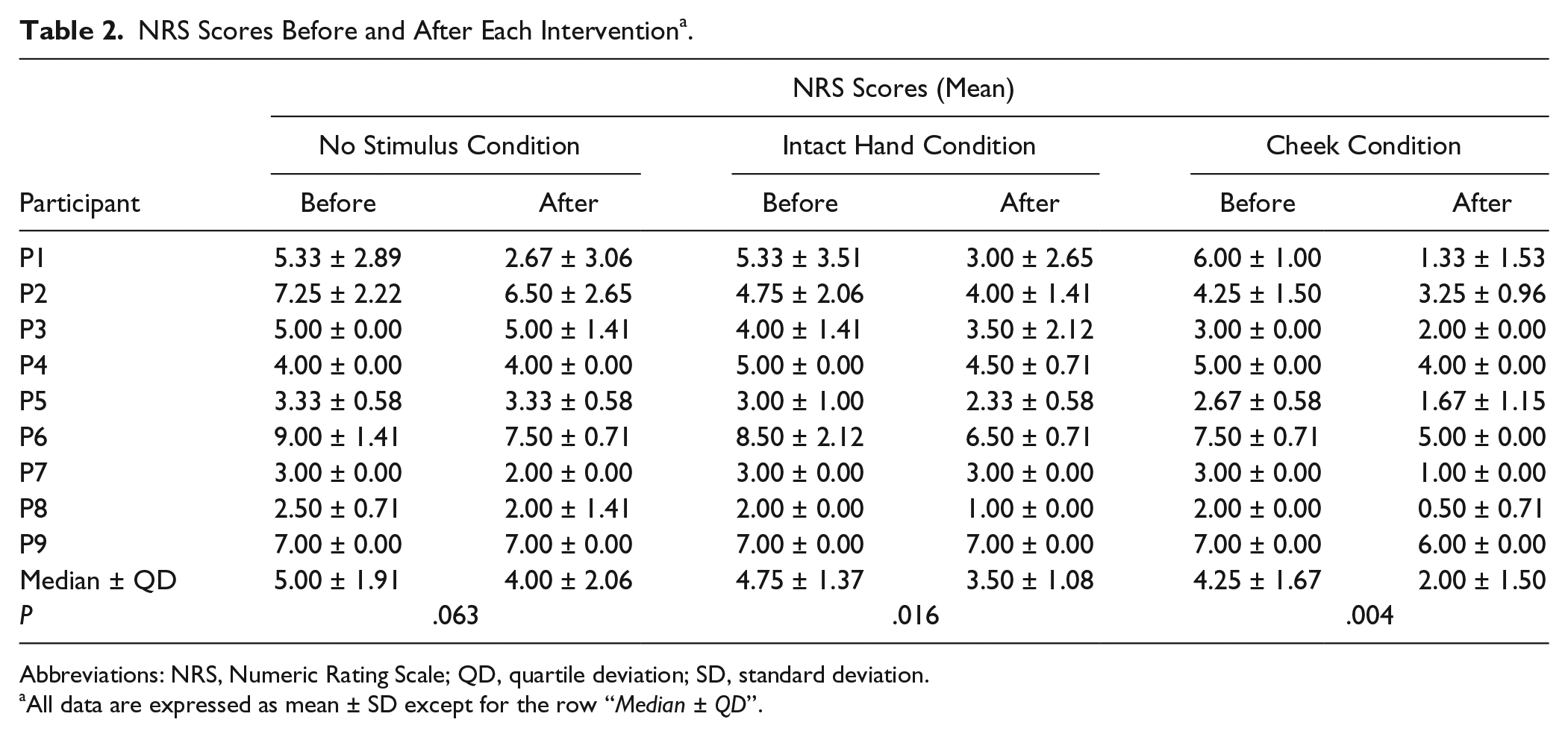
Abbreviations: NRS, Numeric Rating Scale; QD, quartile deviation; SD, standard deviation.
All data are expressed as mean ± SD except for the row “Median ± QD”.
Pain Reduction Rates and Pairwise Comparisons.
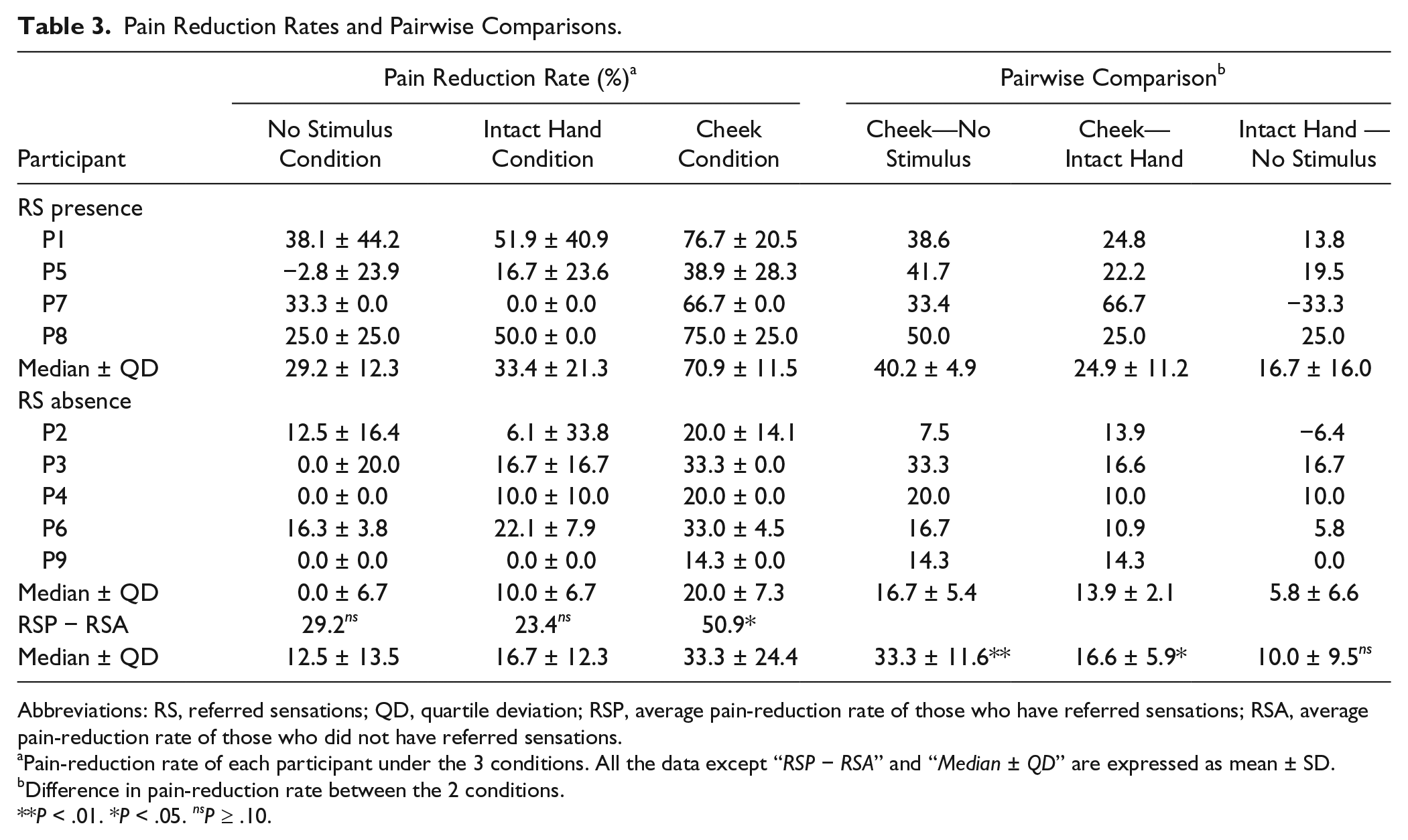
Abbreviations: RS, referred sensations; QD, quartile deviation; RSP, average pain-reduction rate of those who have referred sensations; RSA, average pain-reduction rate of those who did not have referred sensations.
Pain-reduction rate of each participant under the 3 conditions. All the data except “RSP − RSA” and “Median ± QD” are expressed as mean ± SD.
Difference in pain-reduction rate between the 2 conditions.
P < .01. *P < .05. nsP ≥ .10.
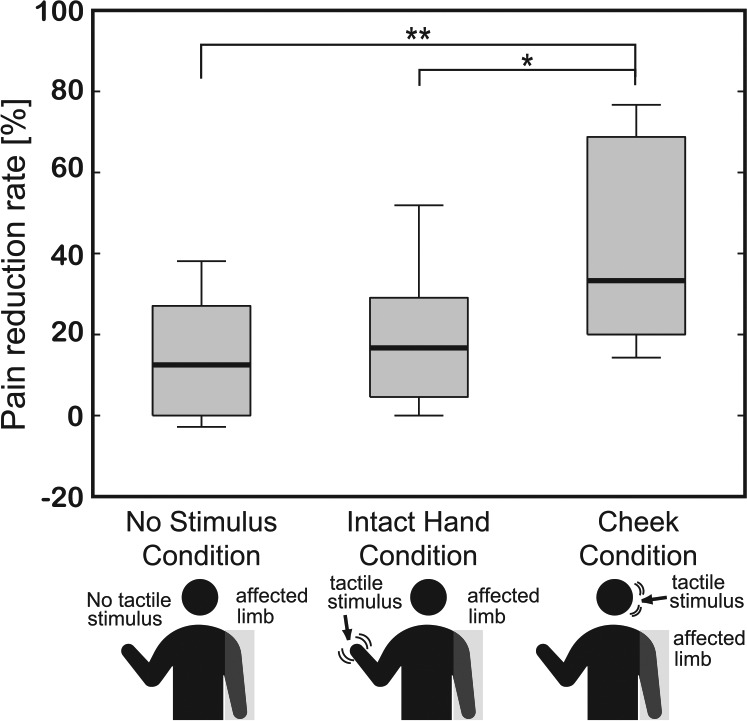
Pain reduction rates in the 3 experimental conditions. Reduction rates were calculated using NRS (Numeric Rating Scale) scores obtained both before and after the different experimental conditions. A significant difference was observed between the 3 groups (P < .01), with pairwise comparisons showing that the Cheek Condition produced a significantly higher analgesic effect than the other 2 conditions (vs No Stimulus Condition: P < .01; vs Intact Hand Condition: P < .05). **P < .01 and *P < .05.
Finally, the participants who always felt referred sensations experienced significantly greater pain reduction in the Cheek Condition than those who did not (P = .016; Mann-Whitney U test).
Quality of Phantom Limb Pain
Sensory and affective pain reduction rates were calculated using the scores obtained with the adapted Japanese version of the short-form McGill Pain Questionnaire 1, and are presented in Figure 3. The median pain reduction rates were 58.3 ± 18.2% (sensory pain) and 100.0 ± 27.1% (affective pain) for the Cheek Condition; 31.0 ± 13.9% (sensory pain) and 53.3 ± 38.5% (affective pain) for the Intact Hand Condition; and 10.0 ± 22.0% (sensory pain) and 29.2 ± 45.0% (affective pain) for the No Stimulus Condition. Statistical analyses showed that the sensory pain reduction differed significantly between conditions (Friedman test, χ2 = 10.5, P = .0054), but that the affective pain reduction did not (χ2 = 5.3, P = .072). Pairwise comparisons showed that the sensory pain reduction rate in the Cheek Condition was significantly higher than in the No Stimulus Condition (P = .0038). No other pairwise comparison produced a statistically significant result.
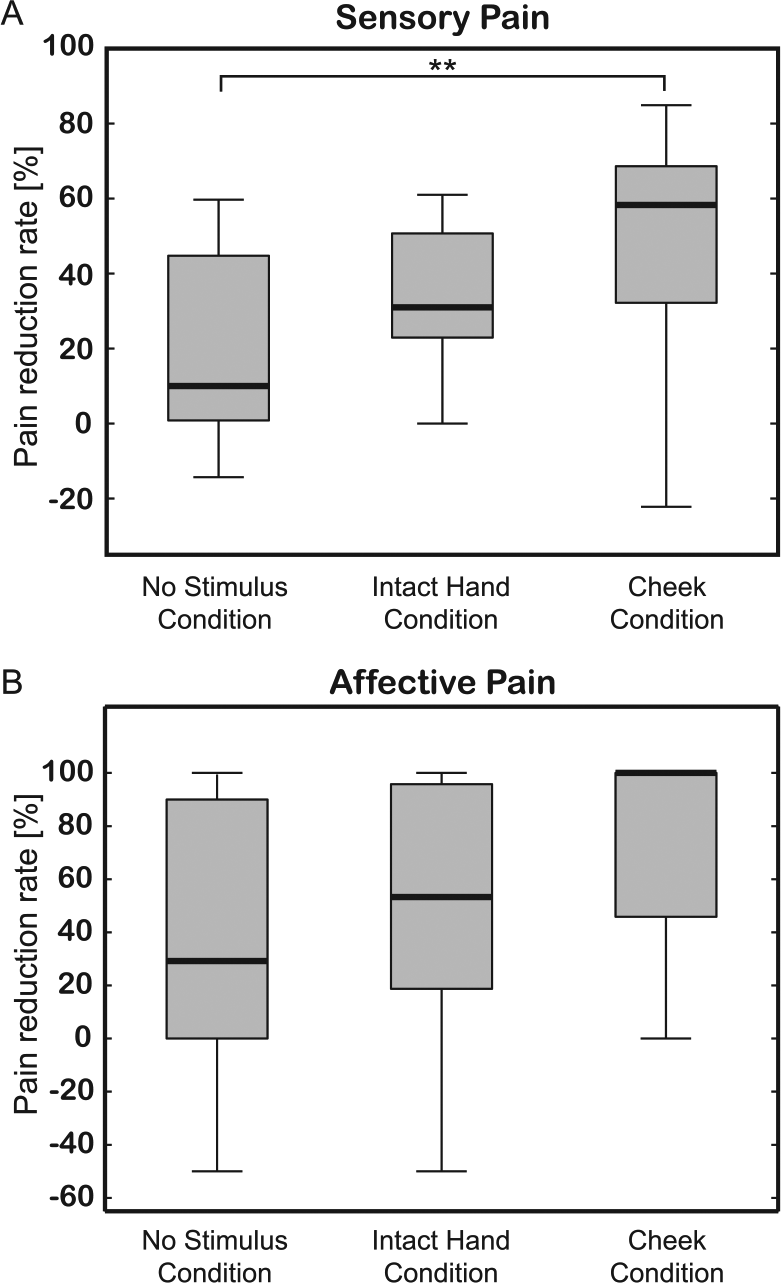
Pain reduction rates for sensory (A) and affective (B) pain in the 3 experimental conditions. Reduction rates were calculated using short-form McGill Pain Questionnaire scores obtained both before and after each experimental condition. A significant difference in sensory pain was observed between the 3 conditions (P < .01), with pairwise comparisons showing that the Cheek Condition produced a significantly higher analgesic effect than the No Stimulus Condition (P < .01). No significant differences were found for affective pain between the 3 conditions (P > .05). **P < .01.
Discussion
In the present study, we demonstrated that pairing the virtual touching of an object by a phantom upper limb with a vibratory feedback stimulus applied to the cheek on the affected side significantly decreased PLP compared with when the stimulus was either applied to the intact upper limb or not applied at all.
Several lines of evidence from other neurorehabilitation studies suggest that some pathological pain, including PLP, is induced by incongruent sensorimotor integration associated with an affected limb12,13 and is closely linked to the functional reorganization of the brain.3,24,35 As a result of this reorganization, when some patients with phantom upper limbs are touched on the cheek or shoulder on the affected side, they also perceive a phantom tactile sensation as if it were coming from that limb.4,23,32 This suggests that stimulating the cheek of patients on their affected side while they view a virtual affected upper limb touch an object might lead to an illusory spatial match between the visual and somatosensory representations of the virtual affected upper limb.
Such illusory cross-modal spatial matches could reinforce a sense of body ownership, 31 which is a key factor for the acquisition of voluntary motor imagery of a phantom limb.36,37 Some studies have revealed that voluntary motor imagery of a phantom limb plays an important role in alleviating PLP.12,14,20,38,39 We suggest that the illusory cross-modal spatial match about the virtual affected upper limb in the VR environment might enhance the sense of body ownership of the virtual affected upper limb, which in turn could improve the ability of patients to execute voluntary motor imagery with their phantom upper limb. We further propose that the acquisition of voluntary motor imagery of their phantom upper limb can help alleviate PLP.
Only 4 of 9 participants felt referred sensations every time their cheek on the affected side was touched. These same patients were shown to experience significantly greater pain alleviation in the Cheek Condition than those who did not feel referred sensations. However, participants who did not feel referred sensations still reported pain reduction in the Cheek Condition that was significantly greater than in the 2 control conditions. Thus, it can be speculated that applying tactile stimuli to the cheek on the affected side enhances the analgesic effect of visual feedback therapy even in the absence of referred sensations.
A simple simultaneous manipulation of visual and somatosensory information can induce referred sensations even in healthy people. For instance, a neuroimaging study demonstrated that S1 is involved in this kind of artificially induced referred sensations; the modulation of cortical representation in S1 was significantly correlated with the amount of felt referred sensations. 25 In this particular study, referred sensations were induced in the body part whose cortical representation in S1 was adjacent to the representation of the body part being touched. In the somatotopic map, the area corresponding to the affected upper limb is adjacent to the cheek area.2,3 Bases on these findings, we hypothesize that there is a possibility that the participants who did not report feeling referred sensations in our pre-experiment test, but reported significant PLP alleviation during VR-MVF, may still have felt artificially induced referred sensations in the phantom upper limb during the VR-MVF with somatosensory feedback to the cheek on the affected side.
In addition, there is a possibility that our pre-experiment test overlooked some participants who felt referred sensations every time their cheek was touched. We examined the presence of referred sensations using a comparatively simple method proposed by McCabe et al 30 ; however, in hindsight, the more in-depth method proposed by Knecht et al 24 may have more accurately detected the presence of referred sensations.
There are 2 main hypotheses that can explain the mechanism that underlies the perception and control of motion and posture. The first is based on the notion of an efference copy—a copy of efferent motor commands. Some researchers have suggested that the prediction of sensory signals based on an efference copy is compared with actual sensory feedback and that the discrepancy between the 2 sets of information is utilized to generate the following motor command.13,40 It is believed that deafferented patients cannot properly control the posture and motion of their phantom limbs due to the absence of sensory feedback generated from the movement. Therefore, it can be speculated that presenting multisensory information that is congruent with phantom limb motor commands can reestablish sensorimotor integration and consequently help acquire phantom upper limb voluntary motor imagery.
The alternative hypothesis is based on the notion of a referent control. This hypothesis posits that actual body configuration results from the summation of the referent body configuration, which is determined before producing a motion, and the deviation from the referent configuration delivered by sensory feedback. 41 It is believed that the referent configuration remains present for some time after deafferentation, even though adequate sensory feedback is absent. Consequently, patients cannot move their phantom limbs voluntarily. However, this hypothesis does suggest that patients could control virtual representations of their phantom limbs voluntarily during VR-MVF, which provides relevant afferent information, such as congruent visual, tactile, and auditory feedback cues, and delivers the proper deviation from the referent configuration.
Given the existence of multiple hypotheses that can explain the effects of VR-MVF on PLP, further studies are needed to elucidate the specific mechanisms that underlie the analgesic effect of VR-MVF with tactile feedback to the cheeks and to develop a better understanding of the relationship between the referred sensations and the analgesic effect of our VR-MVF.
The main contribution of the present study is the demonstration of the significant analgesic effect provided by the application of tactile stimuli to the cheek on the affected side during VR-MVF, which provides multimodal sensory feedback associated with the movement of a virtual affected upper limb.
One remarkable advantage of stimulating the cheek to induce referred sensations is that this method can be applied to all PLP patients, including those who suffer from complete brachial plexus avulsion. Although the present study is the first to use referred sensations during VR-MVF therapy, previous studies have used them for other rehabilitation and prosthesis training.16,31,42 In these studies, for instance, tactile stimuli were often applied to the stump of the amputated limb to induce referred sensations of the phantom limb. However, patients with a complete deafferentation of the limb (eg, brachial plexus avulsion or cervical spinal cord injury) cannot benefit from this approach because they cannot perceive any tactile sensation on the affected limb. In the present study, we have shown that stimulating the cheek of patients with a complete deafferentation is a viable alternative, which constitutes a significant scientific advance for pain management in clinical settings.
It should be noted, however, that other factors could have influenced the present results. For example, auditory feedback during VR-MVF might have also contributed to the observed analgesic effect. Indeed, previous studies have shown that providing auditory stimuli during MVF enhances the therapeutic effect.17,43 However, this additional effect cannot account for differences observed between our experimental conditions, because they were equally present in all conditions. Additionally, there is a possibility that the redirection of attention during VR-MVF provided a distraction from the pain that contributed to the perceived pain reduction.44,45 We cannot exclude the possibility that the effect of the distraction was the largest in the Cheek Condition, followed in order by Intact Hand Condition and No Stimulus Condition, leading to the present results. This should be resolved in further studies.
Another potential limitation of the present study is that our pain measures, the NRS scores and the short-form McGill Pain Questionnaire responses, were subjective in nature. Further studies are needed to collect objective pain measurements, such as those provided by functional brain imaging 46 and electroencephalography, to corroborate our findings. These methods would also have the added benefit of helping to determine the underlying neural mechanisms responsible for PLP and for the analgesic effect of VR-MVF therapy. Furthermore, the present study investigated only the immediate analgesic effects of applying synchronous tactile feedback stimuli to the cheek during VR-MVF therapy and was limited to a small sample size. Future studies should attempt to replicate our findings with larger sample sizes and investigate the long-term analgesic effects of VR-MVF therapy combined with cheek stimulation.
In conclusion, the present study provides a novel and clinically relevant method to alleviate PLP by applying somatosensory feedback to the cheek on the affected side during VR-MVF therapy.
Footnotes
Acknowledgements
We are grateful to Mr Naoki Wake for developing a prototype of our VR-MVF system.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education, Culture, Sports, Science and Technology Grant-in-Aid for Scientific Research on Innovative Areas for “Constructive Developmental Science” (24119002, 24119006).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
