Abstract
Phantom limb pain (PLP) is a frequent complication in amputees, which is often refractory to treatments. We aim to assess in a factorial trial the effects of transcranial direct current stimulation (tDCS) and mirror therapy (MT) in patients with traumatic lower limb amputation; and whether the motor cortex plasticity changes drive these results. In this large randomized, blinded, 2-site, sham-controlled, 2 × 2 factorial trial, 112 participants with traumatic lower limb amputation were randomized into treatment groups. The interventions were active or covered MT for 4 weeks (20 sessions, 15 minutes each) combined with 2 weeks of either active or sham tDCS (10 sessions, 20 minutes each) applied to the contralateral primary motor cortex. The primary outcome was PLP changes on the visual analogue scale at the end of interventions (4 weeks). Motor cortex excitability and cortical mapping were assessed by transcranial magnetic stimulation (TMS). We found no interaction between tDCS and MT groups (F = 1.90, P = .13). In the adjusted models, there was a main effect of active tDCS compared to sham tDCS (beta coefficient = −0.99, P = .04) on phantom pain. The overall effect size was 1.19 (95% confidence interval: 0.90, 1.47). No changes in depression and anxiety were found. TDCS intervention was associated with increased intracortical inhibition (coefficient = 0.96, P = .02) and facilitation (coefficient = 2.03, P = .03) as well as a posterolateral shift of the center of gravity in the affected hemisphere. MT induced no motor cortex plasticity changes assessed by TMS. These findings indicate that transcranial motor cortex stimulation might be an affordable and beneficial PLP treatment modality.
Keywords
Introduction
Phantom limb pain (PLP) is a common phenomenon following an amputation that leads to chronic disability. 1 In PLP, maladaptive plasticity associated with sensory deafferentation following an amputation is one of the contributors for excessive pain. 2 In this context, a potential neural target to modulate the dysfunctional sensorimotor circuits is the primary motor cortex (M1). 3 There are several approaches to modulate the motor cortex that have shown positive results. One of them is the direct stimulation of the M1 with a noninvasive brain stimulation technique: transcranial direct current stimulation (tDCS). 4 tDCS reduces neuropathic pain likely by changing motor cortex thalamic connectivity. 5 The activation of the thalamic pathways provides afferent inputs that counteract the effects of lack of sensory afferents, thus enhancing levels of thalamic and other pain-related inhibitory pathways. 6
In PLP subjects, Bolognini et al showed a short-lasting reduction of pain intensity after 1 session of anodal tDCS over M1 and a pain reduction for at least a week after 5 consecutive stimulation sessions. 7 A recent meta-analysis reported a statistically and clinically significant decrease of 1.5 points on the visual analogue scale (VAS) after M1 active tDCS in individuals with PLP. 8 Moreover, few studies have shown that tDCS over M1 increases pain threshold levels and enhances descending pain inhibitory control. 8
Another method that can modulate M1 plasticity and pain levels is sensorimotor representation techniques, such as mirror therapy (MT).9,10 The underlying mechanism of MT involves the bilateral activation of the sensory, motor, and visual cortex. 11 Previous studies show significant effects of MT and mirror visual feedback in motor excitability. 12 Some clinical trials and case reports on the effects of MT in PLP subjects have shown statistically significant analgesic effect. However, a meta-analysis with 9 randomized controlled trials comparing MT with a choice control showed that MT led to an inconclusive effect size for pain reduction. 13 Similarly, a systematic review showed an insufficient level of evidence to support use of MT as a first intention treatment in PLP. 14 Therefore, there is a need for well-powered testing the MT efficacy on PLP.
Given the potential shared neurophysiological mechanisms of tDCS and MT to modulate motor cortex excitability and given the role of M1 as a modulator of chronic pain, we hypothesized that the combination of these low-cost interventions would lead to stronger effects. Since tDCS is a neuromodulatory (only changes the likelihood of spontaneous neuronal firing), its optimal effect is achieved when combined with a behavioral intervention. 15
To address this gap in PLP treatment, we aimed to test whether tDCS combined with MT has a greater impact on PLP than any therapy alone in traumatic lower limb amputees. Second, we tested the effects of these interventions on motor cortex plasticity, by assessing cortical excitability and cortical mapping changes before and after treatment using transcranial magnetic stimulation (TMS). Last, we tested whether changes in motor cortex plasticity (paired-pulse TMS and mapping) were correlated with PLP changes. We hypothesize that combined tDCS and MT would induce larger PLP relief as compared with any therapy alone and no therapy. These effects would be driven by inducing greater cortical excitability modulation, specifically by increasing intracortical inhibition and facilitation and leading to changes over motor cortex mapping.
Methods
Study Design and Participants
We conducted a randomized, blinded, sham-controlled, 2 × 2 factorial clinical trial in Spaulding Rehabilitation Hospital, USA, and the University of Sao Paulo, Brazil. The study participants were placed into 1 of 4 treatment groups: (1) Active M1 tDCS + MT; (2) Sham M1 tDCS + MT; (3) Active M1 tDCS + covered MT; and (4) Sham M1 tDCS + covered MT. The study was double-blinded for tDCS intervention, but single-blinded for MT due to its behavioral nature. All participants signed an approved informed consent form before trial procedures. This clinical trial was registered in ClinicalTrials.gov (NCT02487966) before the inclusion of the first participant. Detailed protocol description is published elsewhere. 16
We defined phantom limb pain as the painful perception in a limb that no longer exists. 3
Eligibility Criteria
Unilateral traumatic lower limb amputation; oncological, congenital, and nontraumatic amputees were excluded because of underlying differences in the neuroplasticity profiles; 18 years or older; chronic PLP for at least 3 months (experienced regularly for at least once a week) after the residual limb has completely healed; at least 4 out of 10 (worst imaginable pain) on a numeric rating scale (NRS); stable on or no pain medications use for at least 2 weeks prior to enrollment. Exclusion criteria were the following: pregnancy; self-reported alcohol or illicit drug abuse within 6 months; contraindications to tDCS (ferromagnetic metal or implanted electronic medical devices in the head or neck); unstable medical conditions; a history of head injury with permanent neurological deficits (including cognitive decline that could affect the MT protocol); uncontrolled epilepsy or prior seizures within the last year; unexplained fainting spells or loss of consciousness during the last 2 years; a history of neurosurgery; MT within 3 months before enrollment. 16
Interventions
Transcranial Direct Current Stimulation
tDCS was carried out concurrently with the MT intervention using the Soterix Medical 1 × 1 tDCS device (Soterix Medical Inc). Two 35-cm2 surface sponge electrodes soaked in saline solution were used. The anodal electrode was placed over the M1 contralateral to the amputation side (over the C3 or C4, 10-20 system, due to the expected lack of spatial accuracy of the electrical current, and to avoid stimulation over the ipsilateral hemisphere) and the cathodal over the contralateral supraorbital area. 16 The stimulation was applied for 20 minutes at 2 mA in a daily session for 10 days. 7 The sham tDCS was applied with the same montage; however, the current was applied only for the first 30 seconds. The selection of the primary motor cortex contralateral to the amputated limb as the target area for the stimulation is based on prior literature showing this cortical area an effective target to modulate subcortical circuits—that is, thalamic connectivity—associated with pain control. 5 Additionally, prior literature has suggested that the analgesic effects from motor cortex NIBS are present regardless of the target area as well as the pain etiology. 17
Mirror Therapy
Participants were asked to perform a 12- to 15-minute sequence of exercise movement: (1) starting with light tactile stimulation; (2) active range of motion—flexion and extension; (3) functional task—pretend to write the alphabet with the lower limb; using the unaffected limb while watching its mirrored reflection superimposed over the affected limb simultaneously with the tDCS. 16 Before each exercise, the physical therapist explained and demonstrated the tasks, followed by a practice trial. During active MT, participants were asked to consciously relate the movement observed in the mirror to their phantom limb and instructed to keep their attention focused on the mirrored image. 16 In covered MT, the same tasks were performed with the covered mirror and participants were asked to imagine the movement as if it would be appearing in the mirror as a reflex of their phantom limb and instructed to keep their attention focused on the imagined mirrored image. After 10 combined sessions, the participants were instructed to perform 10 MT sessions at home over 2 weeks period. During this period, the study staff called subjects at least 3 times a week to assess and confirm protocol adherence. A detailed MT protocol is available in the Supplemental Table 1.
Outcomes
Demographic variables were collected during baseline visits, including age, gender, ethnicity, study site, past medical history, current medication use, and others.
The primary outcome was the PLP intensity changes, indexed by the VAS from baseline to the last day of the home MT session (4 weeks after treatment, pain average over the past 24 hours). Additionally, we measured PLP intensity at 1- and 2-month follow-up visits as secondary outcomes. Participants rated the intensity of their PLP ranging from 0 (no pain) to 10 (worst pain imaginable).
Clinical information such as the severity and frequency of PLP, phantom limb sensation (PLS; non-painful sensations in the limb that no longer exists), and residual limb pain (RLP; pain felt in the part of a limb that remains after an amputation), the side of amputation, level of amputation, time since amputation, pain before amputation, previous treatment efficacy, and different PLS types were collected using the adapted version for the lower limb of the Groningen Questionnaire after Arm Amputation.
We collected the Beck’s Depression Inventory (BDI), Beck’s Anxiety Inventory (BAI), tDCS and single-pulse TMS side effects, and tDCS blinding questionnaire. tDCS and single-pulse TMS side effect questionnaires were collected after each procedure using a 4-point severity scale (none, mild, moderate, and severe) and a 5-point relatedness scale (none, remote, possible, probable, and definite).
TMS Assessment
All TMS assessments were performed at baseline and at 4 weeks after the intervention utilizing a Bistim2 device and a figure eight coil (Magstim Company LTDA). Motor-evoked potentials (MEPs) were stored using Powerlab 4/30 (ADinstruments) with a bandpass filter of 20 to 2000 kHz. 16 We investigated the cortical excitability using resting motor thresholds (rMT), MEPs (using 120% of the rMT or until reach an MEP of 1 mV), intracortical facilitation (ICF), and short interval intracortical inhibition (SICI) percentages following single and paired-pulse TMS protocols, respectively. We followed the latest TMS guidelines endorsed by the IFCN. 18 All cortical excitability measures were performed using the hand area cortical representation instead of the leg area because of the well-known reproducibility and reliability of these measurements in the hand area; large variability in the level of lower limb amputees; the leg representation in the brain falls over the interhemispheric fissure, which requires higher TMS intensities leading to major discomfort during the sessions. To assess the cortical reorganization, we performed a TMS cortical mapping assessment of the hand area (a 15-point grid [3 × 5 cm] was placed over the M1). The detailed TMS assessments protocol and the calculation of the parameter are described in the Supplemental Table 2, Supplemental Figure 1, and in our previous articles.16,19
Sample Size
We used the results of the study from Mehta et al as a reference to calculate the sample size for our primary outcome. 20 Using a bidirectional α of .05 and a power of 80%, we estimated the total need of 108 subjects (27 subjects per group). Additionally, estimating a conservative attrition rate of 20%, our target sample size was 132 participants. 16 The calculation was performed using GPower 3.1 software.
Randomization and Masking
The randomized list with a random block size of 4 and 8 was generated by an automatic web-based randomization program (http://randomization.com) and kept in sealed opaque envelopes. Participants were randomized in a 1:1:1:1 allocation ratio and stratified based on their baseline PLP level (less or equal of average 6 in VAS or more than 6 in VAS) and by study site. 16
Participants and study staff were blinded to group assignments throughout the trial. A person not involved in the trial prepared the sealed opaque envelopes with the randomization code. Randomization envelopes were only open immediately before the first session by the therapists not involved with the assessment of any outcomes.
Analysis
The primary outcome was pain changes (VAS scale) from baseline to the last day of the home MT session (at week 4). Baseline and PLP characteristics were reported with descriptive statistics for each group. We used 2-tailed tests in all analyses. The significance level of .05 and an intention-to-treat (ITT) analysis were used. To manage missing data, we used the last observation carried forward (LOCF) method. The normality assumption was tested using histograms and Shapiro-Wilk test. All analyses were performed using STATA version 16 (StataCorp, Inc).
We tested for interactions between tDCS and MT for the primary outcome. The treatment-interaction effect was assessed with a hierarchical linear regression model (dependent variable = PLP changes at 4 weeks; independent variable = tDCS * MT interaction). We used a hierarchical model test as it handles the issue of multiple comparisons efficiently compared to the traditional methods. 21 In the case of a nonsignificant interaction, the effects of tDCS and MT on outcomes were planned to be analyzed separately. For PLP, PLS, and RLP changes, the regression models were performed to account for covariate effects, and adjusted means were compared. Akaike’s information criteria were used to determine important confounders. The identified confounders were included in the final models as a way of adjustment. Durbin-Watson estimates, and Cook’s distance values were used for the analysis of regression diagnostics such as residual autocorrelation and influential cases.
Results for PLP, PLS, and RLP changes were adjusted for PLP-PLS index (PLS intensity subtracted from PLP intensity at baseline) and PLP_PLS category at baseline (high PLP and high PLS, high PLP and low PLS, high PLS and low PLP, and low PLP and low PLS categories; the categories were defined using the PLP and PLS sample distribution at baseline considering the mean as the cutoff). We considered these adjustments to assess specifically the treatment effects on PLP without influence on other phantom phenomena. We also tested for the effects of other covariates—based on our previous study. 22 The adjustment of P-values was only made for the co-primary outcomes. Effect size (Cohen d estimate) 23 on PLP changes for within-group and between groups (tDCS vs MT group) comparisons were calculated.
Additionally, we performed longitudinal models to test differences within and between groups across time (baseline, after intervention, 1-month follow-up, and 2-month follow-up) using repeated-measures ANOVA, including time * group interaction, with post hoc adjustments for multiple comparison (Bonferroni method).
Moreover, we performed a prespecified path analysis model for the PLP data changes to define if pain decrease within the tDCS group is explained by direct or indirect effects, that is, via improvement of secondary outcomes (depression, anxiety, PLS, and RLP). We used a sequence of linear regression models to build the paths; then we calculated the direct and indirect effects. 4
Finally, we performed a logistic regression model using responders as dependent variable; responders were defined as participants with PLP decrease of 2 or more points in the VAS scale at 4 weeks (the main outcome; 1 = PLP responders; 0 = PLP no responders). We included clinical and neurophysiological values in a multivariate model as possible predictors of response. A similar adjustment approach as for primary outcome analysis were followed. A receiver operating curve (ROC) and area under the curve (AUC) for the fitted logistic regression were performed to evaluate the discriminatory performance of the model. 24
Data Availability
The data that support the findings of this study will be available from the corresponding author, upon reasonable request.
Results
Between July 5, 2015, and March 30, 2020, 387 patients were screened for eligibility. One hundred thirty-two eligible participants consented to participate, and 112 were randomly assigned to 1 of the 4 study groups (Figure 1). Twenty subjects were not randomized as they withdrew from participating in the trial; and 14 participants dropped out. The main reasons for dropouts were time commitment or lack of transportation to the research center. All randomized participants were included in the analysis of the primary endpoints. Time since amputation (months) was slightly imbalanced, especially in the active tDCS/covered MT group (56 ± 9.97). The rest of the characteristics were similar in all groups (Table 1).
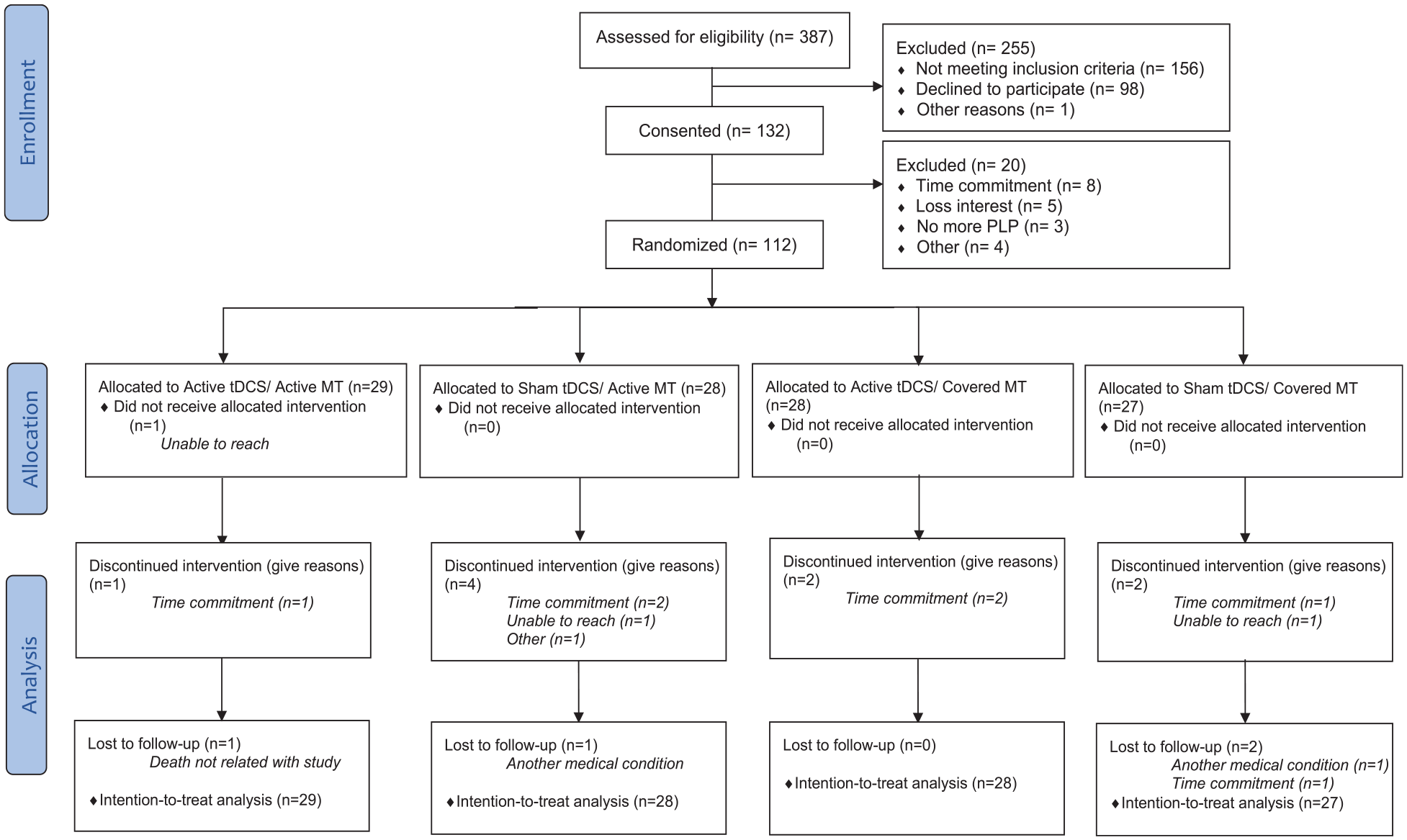
Study flowchart.
Baseline Characteristics a .
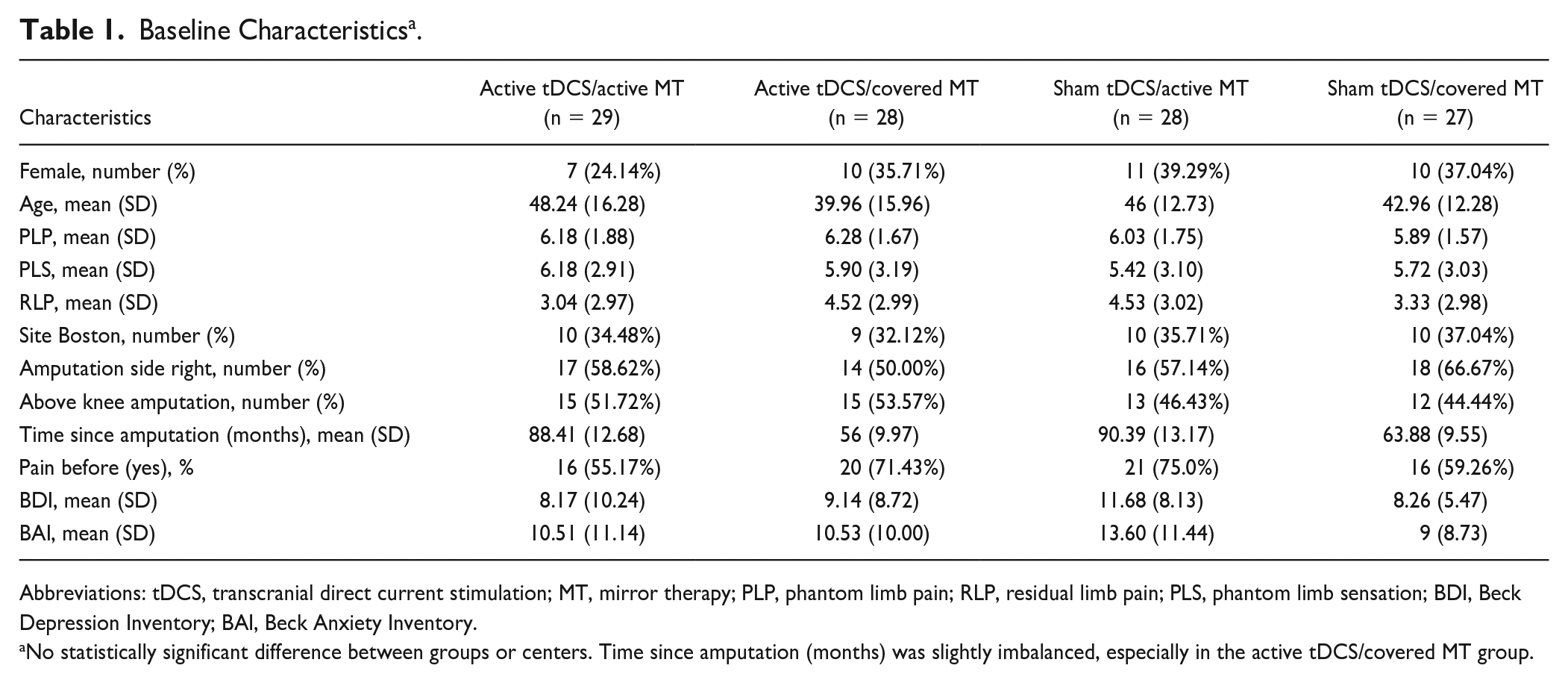
Abbreviations: tDCS, transcranial direct current stimulation; MT, mirror therapy; PLP, phantom limb pain; RLP, residual limb pain; PLS, phantom limb sensation; BDI, Beck Depression Inventory; BAI, Beck Anxiety Inventory.
No statistically significant difference between groups or centers. Time since amputation (months) was slightly imbalanced, especially in the active tDCS/covered MT group.
TDCS and MT Effects on PLP
The hierarchical regression models showed no statistically significant interaction between tDCS and MT (Supplemental Table 3), thus showing that both treatments’ effects were independent. As the interaction was not statistically significant, we then analyzed the main effects separately and we found a statistically significant main effect of tDCS on PLP (beta coefficient = −0.99, P = .04) when comparing active versus sham tDCS, controlling by PLP-PLS index and PLP-PLS category. For the main effect of MT, no statistically significant differences were observed when comparing PLP decrease between active and covered MT (beta coefficient = 0.71, P = .16). From the longitudinal models, we found no statistically significant interaction group * time for any group, although changes across time from baseline to the last follow-up were statistically significant for both groups (Figure 2A and B, Supplemental Table 4, and Supplemental Table 5).
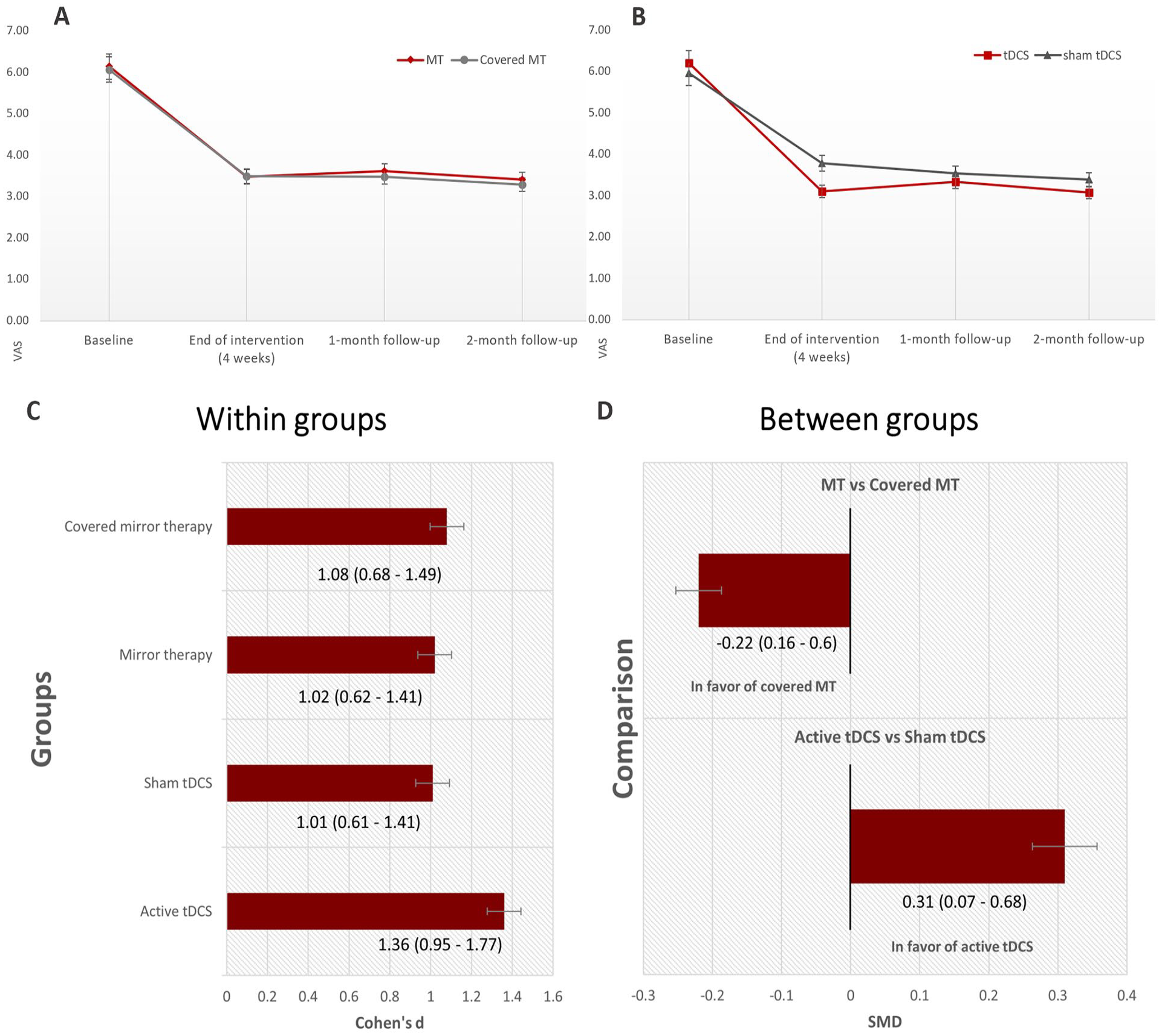
(A and B) PLP longitudinal changes (VAS) per group. We reported raw mean ± standard errors. The main time point was at the end of intervention. Sample sizes at follow-up were the following: 1-month follow-up = 91, 2-month follow-up = 86. (C and D) Effect sizes within (panel C) and between groups (panel D) of different therapies on PLP (ΔVAS). Measures calculated using Cohen’s d effect sizes formula and reported with 95% confidence interval. Note = bars represent standard errors.
In order to quantify the groups’ effects we computed within and between groups effect size (Cohen’s d) on PLP intensity (Figure 2C and D), we found a tDCS effect size (active vs sham) of 0.31 (95% confidence interval [CI]: 0.07, 0.68) showing higher pain decrease in the active tDCS group and a MT effect size of 0.22 (95% CI: 0.16, 0.60) representing smaller pain improvement in the active MT group (covered mirror induced larger pain decrease). Regarding the within-group effect sizes, all groups showed PLP improvement at 4 weeks, and the overall effect size was 1.19 (95% CI: 0.90, 1.47), representing a VAS reduction of 2.64 points. We found no influence of age, sex, or medications (as confounders or effect modifiers) in our models. The raw values (mean ± SD) per group are reported in Figure 3 and Supplemental Table 6.
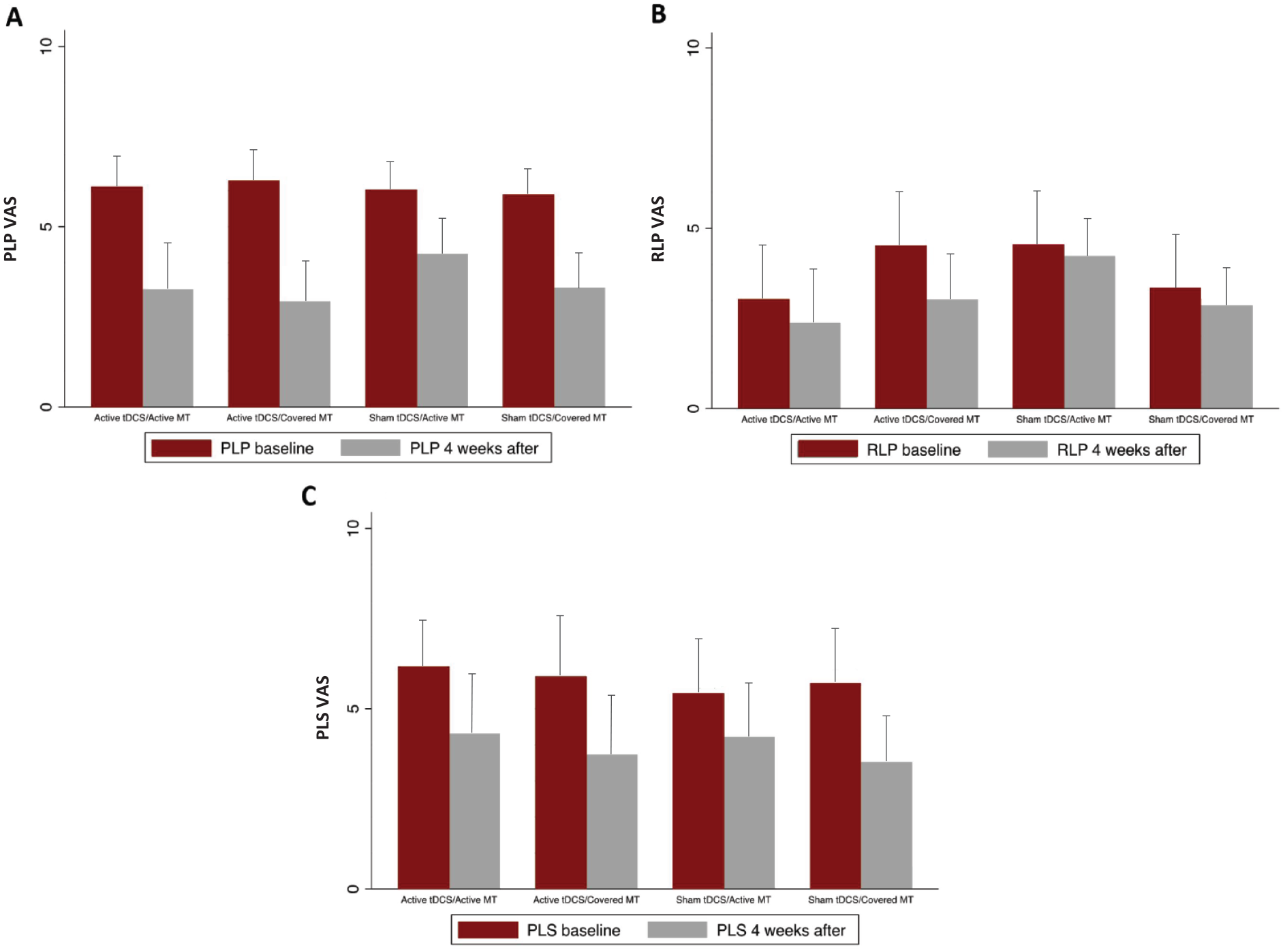
Baseline and after treatment values of PLP, RLP, and PLS intensities per group. A. PLP intensity. B. RLP intensity. C. PLS intensity.
Residual Limb Pain and Phantom Limb Sensation
No statistically significant between-group difference in RLP was found (P = .66). A statistically significant main effect of MT was observed in PLS (beta coefficient = −1.01, P = .03), controlling by the PLP-PLS index and PLP-PLS category; showing that the cover mirror has primary effects over PLS. Our longitudinal analysis showed no statistically significant group * time interaction on PLS. However, changes across time compared with baseline were statistically significant for both groups on PLS (tDCS: F = 23.43, P = .0001; MT: F = 23.85, P = .0001) and on RLP (tDCS: F = 11.36, P = .0001; MT: F = 11.39, P = .0001), including the active and sham groups. The overall within-group effect sizes on PLS was 0.61 (95% CI: 0.34, 0.89) and for RLP was 0.25 (95% CI: 0.03, 0.53) toward the reduction of pain (Supplemental Table 7 and Supplemental Table 8). We found no influence of age, sex, or medications (as confounders or effect modifiers) in our models. The raw values (mean ± SD) per group are reported in Figure 3 and Supplemental Table 6.
Path Analysis
We also conducted a mediation analysis to explore possible underlying mechanisms by which an indirect pathway—mood, anxiety, phantom limb sensation, or residual limb pain—could explain the effects of tDCS on PLP changes. We did not find that these variables (BAI, BDI, PLS, or RLP) mediated the effects of tDCS on PLP. In fact, tDCS intervention (direct effects) was the main pathway (coefficient = −0.57, P = .04) to explain PLP changes. The indirect pathways (via anxiety, depression, PLP, or RLP) were not statistically significant (Supplemental Figure 2). Besides, the comparisons among pathways’ effects (direct versus each indirect pathway) were not statistically significant (Supplemental Table 9).
Effects of the Interventions on Motor Cortex Plasticity
We included 99 participants in the multivariate models (8 subjects were not available for TMS assessment, and 5 were excluded from the analysis due to manufacturing defect of the coil used), and we showed that the tDCS intervention has an effect on intracortical excitability (Supplemental Table 10) specifically leading to (1) increases in the intracortical inhibition of the affected hemisphere (coefficient = 0.96, P = .02) and (2) increases of the intracortical facilitation of the affected hemisphere (coefficient = 2.04, P = .03), controlled by cortical excitability at baseline and opioid use (identified confounders). Furthermore, the tDCS intervention has a statistically significant effect on mapping changes of the hand area in the affected hemisphere (Figure 4), specifically, it leads to (1) less anterior COG of the affected hemisphere (coefficient = −0.63, P = .04) and (2) more lateral COG of the affected hemisphere (coefficient = 1.05, P = .04), controlled by the side of amputation and pregabalin use (identified confounders). We found no effects of tDCS on mapping volume. The MT intervention does not induce statistically significant changes in cortical excitability or mapping changes (Supplemental Table 11). No changes were found in the nonaffected hemisphere. TMS variables per group are reported in Figure 5.
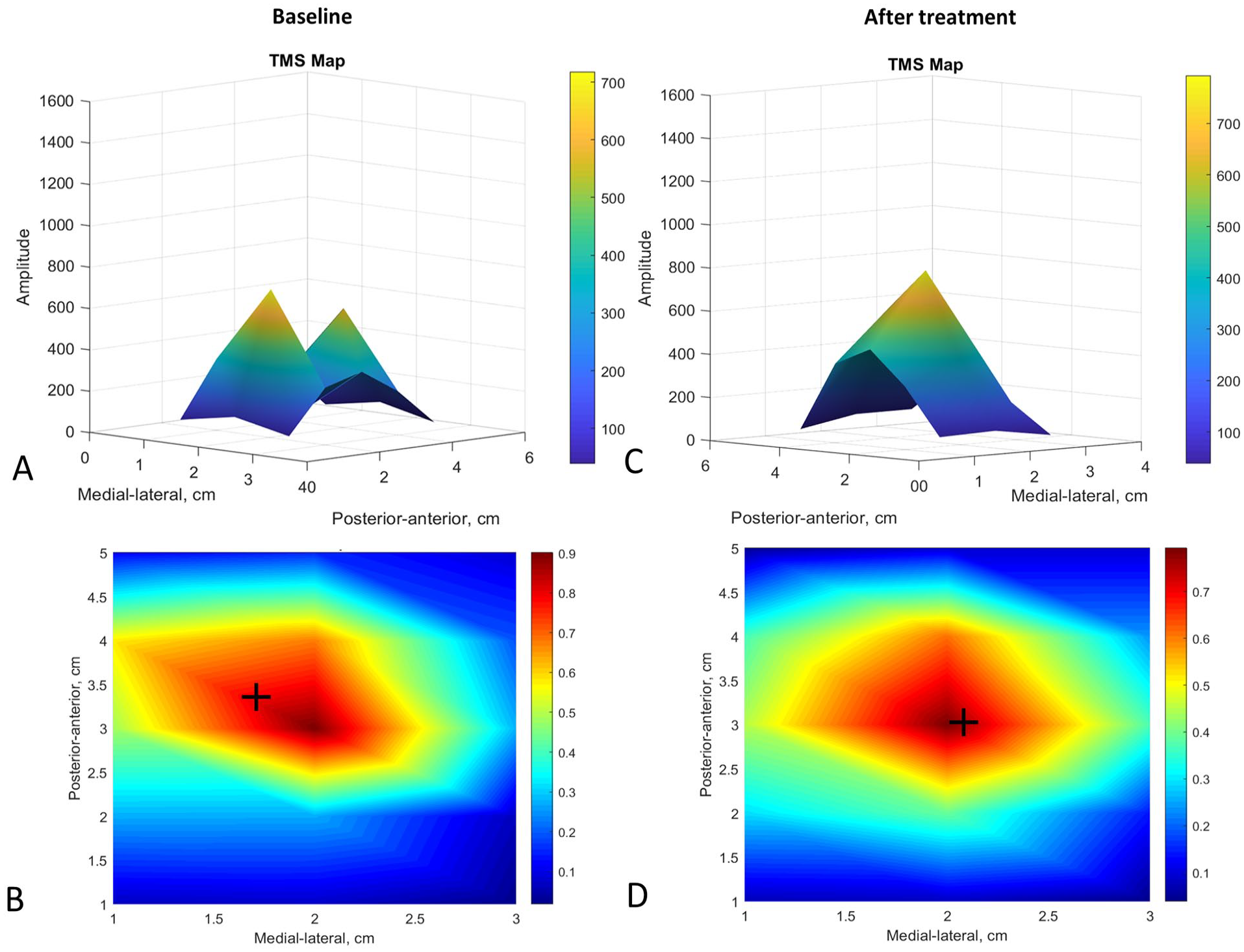
Cortical mapping changes after tDCS in the hand area of the affected hemisphere. At baseline (panels A and B) the cortical representation was disorganized and heterogeneous (panel A) with an anterior and medial center of gravity (COG) (panel B). After tDCS (panels C and D) the cortical representation reorganized, and the COG becomes less anterior (difference = −0.63 cm) and lateral (difference = 1.05 cm).
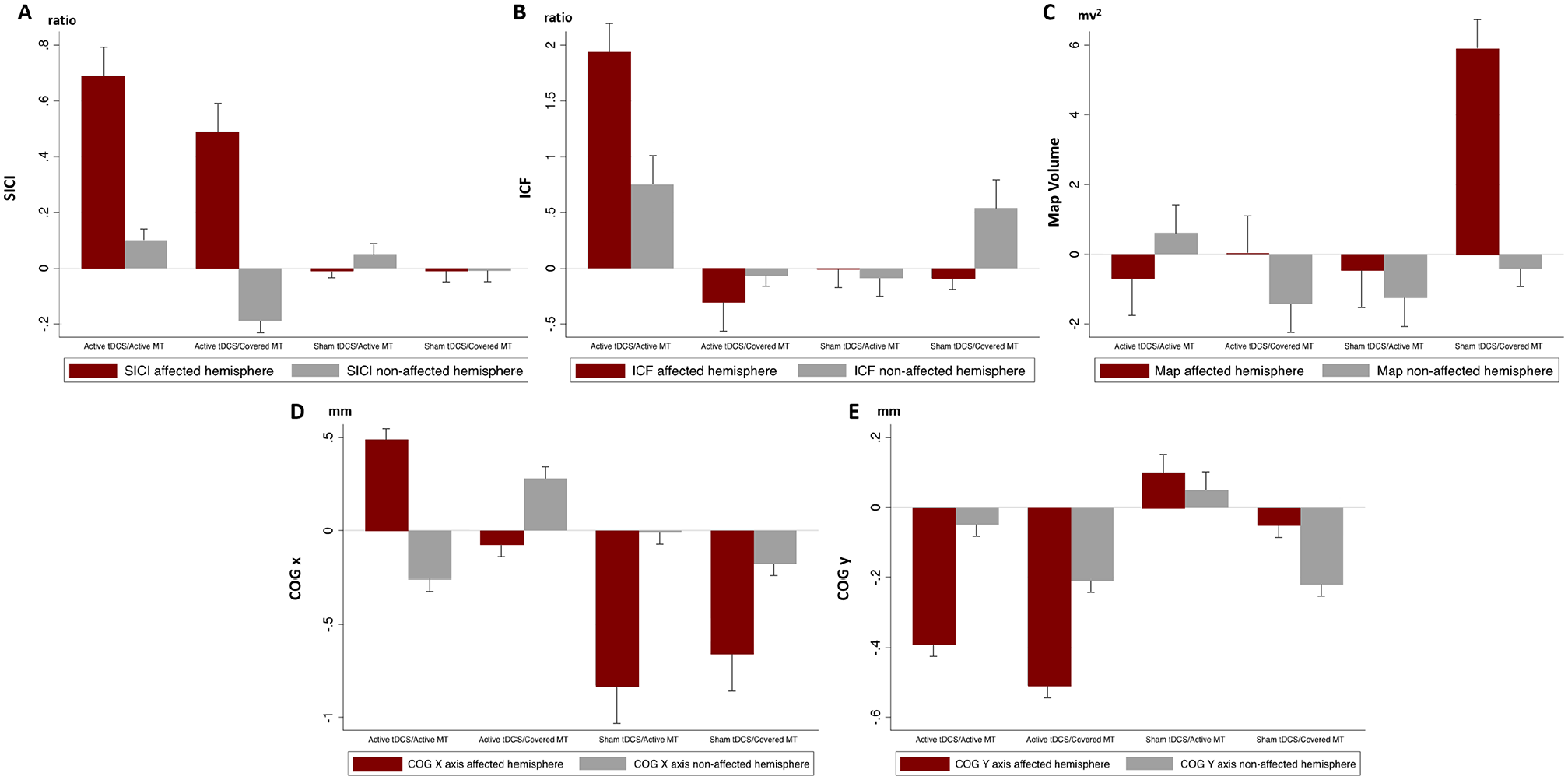
TMS variables changes from baseline per group. A. SICI ratio. B. ICF ratio. C. Map volume. D. Center of gravity x-axis value. E. Center of gravity y-axis value.
We assessed whether PLP changes were associated with changes in motor cortex plasticity. We found that PLP intensity changes are only positively correlated with SICI changes (measured as inhibition percentage) on the affected hemisphere (contralateral to the amputation) (coefficient = 1.00, P = .04) in the active tDCS group, that it was not present in the sham tDCS group (coefficient = 0.45, P = .56). This was not associated with other cortical excitability measures (MEP or ICF) or mapping changes (COG shift or volume change). Moreover, the PLS and RLP changes are not associated with cortical excitability or mapping changes.
Secondary Outcomes
No statistically significant differences within or between-group were found on the BDI or the BAI neither in the tDCS nor in the MT groups. We found no influence of age, sex, or medications (as confounders or effect modifiers) in our models.
Safety Outcomes
No serious adverse event related to the intervention was reported during this trial. One death not related to the study was reported in the Brazil site. No difference between the groups was found in adverse effects (Fischer exact test, P = .99). The reported events were sleepiness (n = 14, 37.8%), neck pain (n = 11, 29.7%), tingling (n = 3, 8.1%), trouble to concentrate (n = 3, 8.1%), headache (n = 2, 5.4%), scalp pain (n = 2, 5.4%), skin redness (n = 1, 2.7%), and acute mood change (n = 1, 2.7%). The distribution of adverse event types was similar among experimental groups. The overall most common reported events were mild and transient scalp tingling and sleepiness.
Predictors of Response
We explored which predictor variables may be related to a reduction in PLP. We found that the presence of movement sensation (type of PLS) at baseline (odds ratio = 3.74; 95% CI: 1.35, 10.34) and the PLP-PLS index at baseline (odds ratio = 1.25; 95% CI: 1.03, 1.52) are the most important variables. This analysis was controlled by the intervention effects (group allocation) (Supplemental Table 12). The AUC of the model was 0.71 (Supplemental Figure 3).
Discussion
We found no interaction between tDCS and MT groups suggesting the independent effects of both interventions. Given the lack of interaction, the independent main effects analysis showed that active tDCS decreased PLP compared to sham tDCS when controlling for PLP-PLS index and PLP-PLS category. On the other hand, the main effect of MT was not statistically significant. Moreover, tDCS effects on PLP were mostly direct, thus not mediated by other covariates. Interestingly, the covered MT seemed to have a primary effect on PLS reduction.
TDCS was the only intervention to induce statistically significant plasticity changes in the cortical mapping, though these changes were not associated with PLP changes. Similarly, only tDCS stimulation was able to increase SICI response; and these changes were correlated with PLP decrease. This may explain the superior effects of tDCS. We also found that high PLP and low PLS presence and the existence of movement sensation were the best predictors for the treatment response. Therefore, motor cortex networks are plausible neural targets for PLP modulation, and a motor cortex-based treatment approach may be individualized based on PLP clinical phenotypes.
Additive Versus Synergistic Effects of TDCS
We found an independent and additive analgesic effect of M1 tDCS (within-group effect size of 1.36) to MT effect. This represents a reduction of 3 points in the VAS scale, which can be considered a clinically important difference (even though we found no effects on anxiety and depression)25-27 and higher than other available treatment modalities.13,14,28 Our findings are consistent with a recent meta-analysis 8 showing a statistically significant pain reduction of M1 tDCS in PLP patients. Therefore, our study considerably complements and expands the evidence on the analgesic effects of M1 tDCS in PLP.
On the other hand, a possible explanation for the lack of synergistic effect of tDCS and MT is that given both active and covered MT (combined with sham tDCS) had a similar effect on PLP, we were able to see only an additive effect of TDCS on both MT and covered MT. The next question would be whether these interventions are clinically meaningful or if they would be similar to a placebo effect. Although our study did not test such a hypothesis, if we compare the effect sizes within our sham tDCS group (groups of covered MT and MT, both combined with sham tDCS) to the effect sizes of placebo only interventions in published PLP clinical trials; we can see that our estimates were from 0.81 to 1.22, but the effect sizes of placebo interventions in other PLP trials were much lower (varying from 0.38 to 0.53).8,29,30 Therefore, given the larger effect sizes of the sham tDCS group combined with either MT or covered MT, it is plausible that both interventions had a real effect of pain decrease beyond just placebo effects, but not differences between them. However, the influence of significant placebo effect in our trial cannot be disregarded, further studies should measure predictors of placebo response (eg, treatment expectancy) to understand its effect on PLP.
In this context, the visual feedback provided by the real mirror may not be an essential component of the analgesic effect. The main pain effects may be due to the contralateral limb movement and the motor imagery that was also presented in the covered MT. Previous studies also showed that both active MT and covered MT were able to improve pain in amputees with PLP. 31 Similarly, the sensorimotor exercises without mirror feedback had similar effects on PLP compared to MT. 32 Moreover, the lack of standardization in the MT protocols as well as the control groups, reduce the precision of the effect estimate. 33 Given these results, one potential conclusion is that tDCS combined with sensorimotor therapy may lead to a higher statistically significant effect on pain reduction. Interestingly, the covered MT seemed to have a primary effect on PLS reduction, which can be secondary to the aforementioned effects from motor imagery without visual feedback. The motor imagery in the presence of visual feedbacks requires a large effort during the movement production, previous studies shows that amputees described an increase in movements and sensations during motor imagery with visual feedback. 34 Therefore, it is possible the visual feedback increased the difficulty of getting somatosensory engagement that could have been more present in the covered mirror group. Even though these sensations were previously associated with larger analgesic response to MT, their presence was highly variable between individuals. Further studies are needed to assess the effects of motor imagery on nonpainful phantom phenomena in amputees with and without PLP.
Effects of tDCS on Motor Cortex Plasticity
There were statistically significant changes in cortical mapping associated with tDCS intervention—less anterior and more lateral shift in the CoG of the affected hemisphere; however, the CoG shifts were not correlated with pain intensity or PLP decrease. These results support our previous findings 22 and previous fMRI and TMS studies in upper limb amputees. These studies show that the presence of shift in representation was present only in amputees with PLP when compared to amputees without PLP but was not associated with the pain intensity or the treatment response. 35 These results are aligned with previous literature supporting the idea that PLP may in fact be associated with a preservation of sensorimotor representation rather than a reorganization.36,37 Therefore, we hypothesize 2 independent circuits in PLP: one involved with cortical mapping representation and another one involved with PLP intensity. The cortical mapping representation seems to be correlated with the presence but not the intensity of PLP; this indicates that sensory deafferentation triggers PLP, but the intensity of PLP is likely modulated by circuits other than those inducing changes in cortical mapping. The directionality of the shift—less anterior and more lateral shift—should not be over interpreted since TMS mapping has low spatial resolution and changes in excitability could not accurately assess spatial location of structural brain changes. 38
The second important parameter we analyzed was intracortical excitability—inhibition and facilitation. We showed an increase in SICI response associated with the tDCS and PLP reduction. Our findings lend support to a previous study showing that excitatory rTMS stimulation of the primary motor cortex increases SICI response in patients with neuropathic pain—furthermore, these patients presented a decreased SICI response at baseline when compared with healthy subjects. 39 This study supports our hypothesis on the feedback loop between the M1 and the pain processing circuits. Therefore, increasing the activity of M1 by tDCS leads to increased inhibitory output and statistically significant pain relief. 40
Moreover, the tDCS intervention has a significant effect on increasing the ICF response; this seems to be a direct result of the motor cortex activation by tDCS. Previous literature also showed that higher ICF induced by anodal tDCS contributed to its analgesic effect. 41 It is also important to point out that different groups of cortical interneurons mediate the SICI and ICF responses, and even though there are interactions between them, an increase in SICI does not inhibit an increase in ICF. 42
In addition, the changes observed here were recorded over the hand representation of the motor area in lower limb amputees. Even though we expected changes in motor representation in adjacent areas to the amputation, as suggested by previous literature, 43 we cannot rule out site-specific excitability changes. 44 That said, these changes in hand areas representation strengthens our hypothesis that the shift in representation over the motor cortex goes beyond the amputated areas, since cortical mapping representation seems to be correlated with the presence of PLP. Moreover, the tDCS stimulation was also performed in the M1 (correspondent to the “hand” area) with the goal of reducing neuropathic pain by changing motor cortex thalamic connectivity. Prior studies have shown that cortical stimulation applied over the motor cortex—hand area—leads to analgesic effects, regardless of the etiology or location of the pain. 45
Path Analysis: The Effect of the Intervention Is the Main Explanatory Pathway
Using path analysis, we showed that the effects of tDCS on PLP relief were not mediated by the individual improvements on the depression and anxiety scales; or by PLS or RLP changes.
As shown previously, the primary somatosensory and motor cortex play a key role in PLP development and maintenance and could be the main neurophysiological pathway to explain our direct effects. 3 Another potential pathway involves the limbic system since it is a central relay that works filtering and transmitting information of nociceptive stimulus and pain perception. 46 However, we found no changes in depression and anxiety, despite of the strong analgesic effects. This is aligned with previous studies that assessed the psychological factors associated with limb amputees, they found no difference of anxiety and depression between phantom pain and no phantom pain groups, 47 or did not show any correlation between anxiety/depression and pain levels. 48 This lack of correlation was also present in other clinical trials showing significant analgesic effect but no significant group difference in depression and anxiety.49,50 Therefore, this absence of mediatory effects of depression and anxiety suggests a lesser involvement of mood-related limbic pathways in PLP compared to somatosensory and motor pathways.
PLS as the Best Predictor of Response for tDCS
We found that PLP-PLS index and movement sensation were the best predictors for tDCS effects on PLP, meaning that participants with less PLS intensity compared with PLP intensity, or presence of movement sensation experienced greater analgesic effects. In our previous study, we found that movement sensation is a protective factor for the PLP intensity. 22 Previous results showed that the movement sensation in the phantom limb is associated with higher activation in the contralateral M1 and somatosensory cortices than controls. 43 However, the other types of PLS engage a more extensive region, including parietal and occipital networks. 43 Therefore, we hypothesize the presence of movement sensation and predominance of PLP (higher PLP-PLS index) could represent a clinical sub-phenotype of patients with PLP. In this context, participants who have PLS can likely trigger somatosensory compensation mechanisms for the lack of sensory afferents. Therefore, tDCS of the motor cortex would have a less significant role, as shown in our results. On the other hand, covered MT had a significant effect on PLS; therefore, behavioral therapies focusing on somatosensory plasticity may have an effect mainly in participants with an active PLS system. Clinically sub-phenotyping patients with PLP provides the possibility of personalized pain treatment using noninvasive brain stimulation or other behavioral therapies.
The current study has several notable strengths, such as the large sample size and the inclusion of clinical and cortical plasticity outcomes. However, this study is not without its limitations. Despite this being a comprehensive study, our sample included only traumatic lower limb amputations; therefore, our results are not totally generalizable to all amputee population. Nevertheless, the lower limb is the most frequent amputation location, and this homogeneous sample allows us to better understand the cortical plasticity changes after the intervention. Finally, we performed a phenotyping post hoc analysis; thus, these findings should be interpreted cautiously as hypothesis-generating results.
Conclusion
In summary, this is the largest mechanistic clinical trial to date investigating amputees with lower limb amputation and PLP. Our results provide additional data supporting the effects on motor cortex stimulation to reduce PLP, the correlation between motor plasticity and PLP changes and finally, how behavioral therapies such as somatosensory training may modify PLP.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683211017509 – Supplemental material for Effects of Combined and Alone Transcranial Motor Cortex Stimulation and Mirror Therapy in Phantom Limb Pain: A Randomized Factorial Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683211017509 for Effects of Combined and Alone Transcranial Motor Cortex Stimulation and Mirror Therapy in Phantom Limb Pain: A Randomized Factorial Trial by Muhammed Enes Gunduz, Kevin Pacheco-Barrios, Camila Bonin Pinto, Dante Duarte, Faddi Ghassan Saleh Vélez, Anna Carolyna Lepesteur Gianlorenco, Paulo Eduardo Portes Teixeira, Stefano Giannoni-Luza, David Crandell, Linamara Rizzo Battistella, Marcel Simis and Felipe Fregni in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683211017509 – Supplemental material for Effects of Combined and Alone Transcranial Motor Cortex Stimulation and Mirror Therapy in Phantom Limb Pain: A Randomized Factorial Trial
Supplemental material, sj-docx-2-nnr-10.1177_15459683211017509 for Effects of Combined and Alone Transcranial Motor Cortex Stimulation and Mirror Therapy in Phantom Limb Pain: A Randomized Factorial Trial by Muhammed Enes Gunduz, Kevin Pacheco-Barrios, Camila Bonin Pinto, Dante Duarte, Faddi Ghassan Saleh Vélez, Anna Carolyna Lepesteur Gianlorenco, Paulo Eduardo Portes Teixeira, Stefano Giannoni-Luza, David Crandell, Linamara Rizzo Battistella, Marcel Simis and Felipe Fregni in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-3-nnr-10.1177_15459683211017509 – Supplemental material for Effects of Combined and Alone Transcranial Motor Cortex Stimulation and Mirror Therapy in Phantom Limb Pain: A Randomized Factorial Trial
Supplemental material, sj-docx-3-nnr-10.1177_15459683211017509 for Effects of Combined and Alone Transcranial Motor Cortex Stimulation and Mirror Therapy in Phantom Limb Pain: A Randomized Factorial Trial by Muhammed Enes Gunduz, Kevin Pacheco-Barrios, Camila Bonin Pinto, Dante Duarte, Faddi Ghassan Saleh Vélez, Anna Carolyna Lepesteur Gianlorenco, Paulo Eduardo Portes Teixeira, Stefano Giannoni-Luza, David Crandell, Linamara Rizzo Battistella, Marcel Simis and Felipe Fregni in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-4-nnr-10.1177_15459683211017509 – Supplemental material for Effects of Combined and Alone Transcranial Motor Cortex Stimulation and Mirror Therapy in Phantom Limb Pain: A Randomized Factorial Trial
Supplemental material, sj-docx-4-nnr-10.1177_15459683211017509 for Effects of Combined and Alone Transcranial Motor Cortex Stimulation and Mirror Therapy in Phantom Limb Pain: A Randomized Factorial Trial by Muhammed Enes Gunduz, Kevin Pacheco-Barrios, Camila Bonin Pinto, Dante Duarte, Faddi Ghassan Saleh Vélez, Anna Carolyna Lepesteur Gianlorenco, Paulo Eduardo Portes Teixeira, Stefano Giannoni-Luza, David Crandell, Linamara Rizzo Battistella, Marcel Simis and Felipe Fregni in Neurorehabilitation and Neural Repair
Footnotes
Author Contributions
MEG, KP-B, and CBP are the first equally contributing authors. DD and FGSV are the second equally contributing authors. FF had the initial idea and designed the study. FF, CBP, and FGSV set up the MT intervention and design of the TMS assessments. MEG, CBP, DD, FGSV, ACLG, PEPT, KP-B, and SG were involved in participants’ recruitment and data collection. KP-B, MEG, and FF performed the data analysis. FF, MEG, CBP, KP-B, DD, FGSV, ACLG, PEPT, and SG were involved in the manuscript writing. DC was the medical collaborator and assisted also in the recruitment. MS and LRB coordinated all data collection in the Brazil site. All authors completed a critical review and revision of the article and approved the submitted version. FF is corresponding author and had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by an NIH RO1 grant (1R01HD082302-01A1).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
