Abstract
Introduction
Repetitive peripheral sensory stimulation (RPSS) followed by 4-hour task-specific training (TST) improves upper limb motor function in subjects with stroke who experience moderate to severe motor upper limb impairments. Here, we compared effects of RPSS vs sham followed by a shorter duration of training in subjects with moderate to severe motor impairments in the chronic phase after stroke.
Methods
This single-center, randomized, placebo-controlled, parallel-group clinical trial compared effects of 18 sessions of either 1.5 h of active RPSS or sham followed by a supervised session that included 45 min of TST of the paretic upper limb. In both groups, subjects were instructed to perform functional tasks at home, without supervision. The primary outcome measure was the Wolf Motor Function Test (WMFT) after 6 weeks of treatment. Grasp and pinch strength were secondary outcomes.
Results
In intention-to-treat analysis, WMFT improved significantly in both active and sham groups at 3 and 6 weeks of treatment. Grasp strength improved significantly in the active, but not in the sham group, at 3 and 6 weeks. Pinch strength improved significantly in both groups at 3 weeks, and only in the active group at 6 weeks.
Conclusions
The between-group difference in changes in WMFT was not statistically significant. Despite the short duration of supervised treatment, WMFT improved significantly in subjects treated with RPSS or sham. These findings are relevant to settings that impose constraints in duration of direct contact between therapists and patients. In addition, RPSS led to significant gains in hand strength.
Trial Registry Name: Peripheral Nerve Stimulation and Motor Training in Stroke Clinical Trials.gov identifier: NCT0265878 https://clinicaltrials.gov/ct2/show/NCT02658578
Introduction
Increase in sensory input from the paretic hand in the form of repetitive peripheral sensory stimulation (RPSS) enhances motor performance in subjects with mild to moderate impairments. 1 Also, Carrico et al. 2 reported significantly bigger improvements in Wolf Motor Function Test (WMFT), Fugl–Meyer Assessment (FMA), and Action Research Arm Test after active RPSS compared to sham in subjects with moderate to severe motor impairments. Active RPSS or sham was administered for 2 h prior to 4 h intensive task-oriented motor (TOT) training over 10 days. Implementation of this protocol may be challenging in clinical practice, particularly in developing countries where most strokes occur. 3
Another intervention that may boost effects of training on amelioration of upper limb motor function is functional electrical stimulation (FES).4-9 Here, we compared effects of shorter durations of active RPSS or sham combined with FES and task-specific training (TST) in subjects with moderate to severe upper limb impairments in the chronic phase after stroke.
Methods
This single-center, randomized, placebo-controlled, parallel-group clinical trial compared effects of 18 sessions of active and sham RPSS followed by FES and TST of the paretic upper limb. Outcome assessors were blinded to group assignment and participants were naive to the experimental hypothesis. The protocol has been previously published. 10
Subjects
Adults with stroke (> 6 months) and upper limb FMA motor scores between 7 and 50 were included. For details, see Ref. [4].
Interventions
In the active group, 1.5 hours of RPSS of the median, ulnar, and radial nerves was performed. 10 In the sham group, the tibial, superficial peroneal, and sural nerves were stimulated. Average intensities of nerve stimulation (% of sensory threshold) were as follows: In the active group, 213±95% (median), 193±88% (ulnar), and 165±79% (radial). In the sham group, 92.6±52% (tibial), 174.8±48.6% (superficial peroneal), and 173.8±45.6% (sural). FES for 30 minutes, TST for 45 minutes, and instructions to perform functional tasks at home were administered to both groups. 10 Additional information is reported in Supplementary Materials.
Outcomes
The primary outcome was the WMFT (WMFT-TIME and functional assessment scale, WMFT-FAS) after 6 weeks of treatment.
The secondary outcomes 10 were the WMFT at 3 weeks as well as the following at 3 and 6 weeks: grasp and pinch strength; Motor Activity Log; active range of motion (ROM) of wrist extension and flexion; modified Ashworth scale; FMA; Barthel Index (BI); Stroke Impact Scale (SIS); Beck Depression Inventory Short Form (BDI).
Ethics
The protocol was approved by the institutional ethics committee (CAAE 70163217.5.0000.0068). All subjects provided informed consent. 10
Sample Size
Our goal was to include 40 subjects in the study. 10
Analysis
The data were analyzed with a generalized estimating equations 11 models with factors GROUP (active or sham) and TIME (baseline, 3, 6 weeks). All post hoc analyses were Bonferroni-corrected. Intention-to-treat (ITT) analysis was performed with and without the Last Observation Carried Forward (LOCF) for missing data. Per protocol (PP) analysis excluded subjects who participated in <80% treatment sessions. P-values ≤ .025 were considered statistically significant because 1 interim analysis was performed. Analyses of secondary outcomes were exploratory and not corrected for multiple comparisons. Additionally, we assessed attainment of Minimal Clinically Important Differences (MCID) 12 for WMFT-TIME (1.5 s) and WMFT-FAS (.2). Effect sizes (ES) were assessed with Cohen’s d.
Results
Participant Characteristics
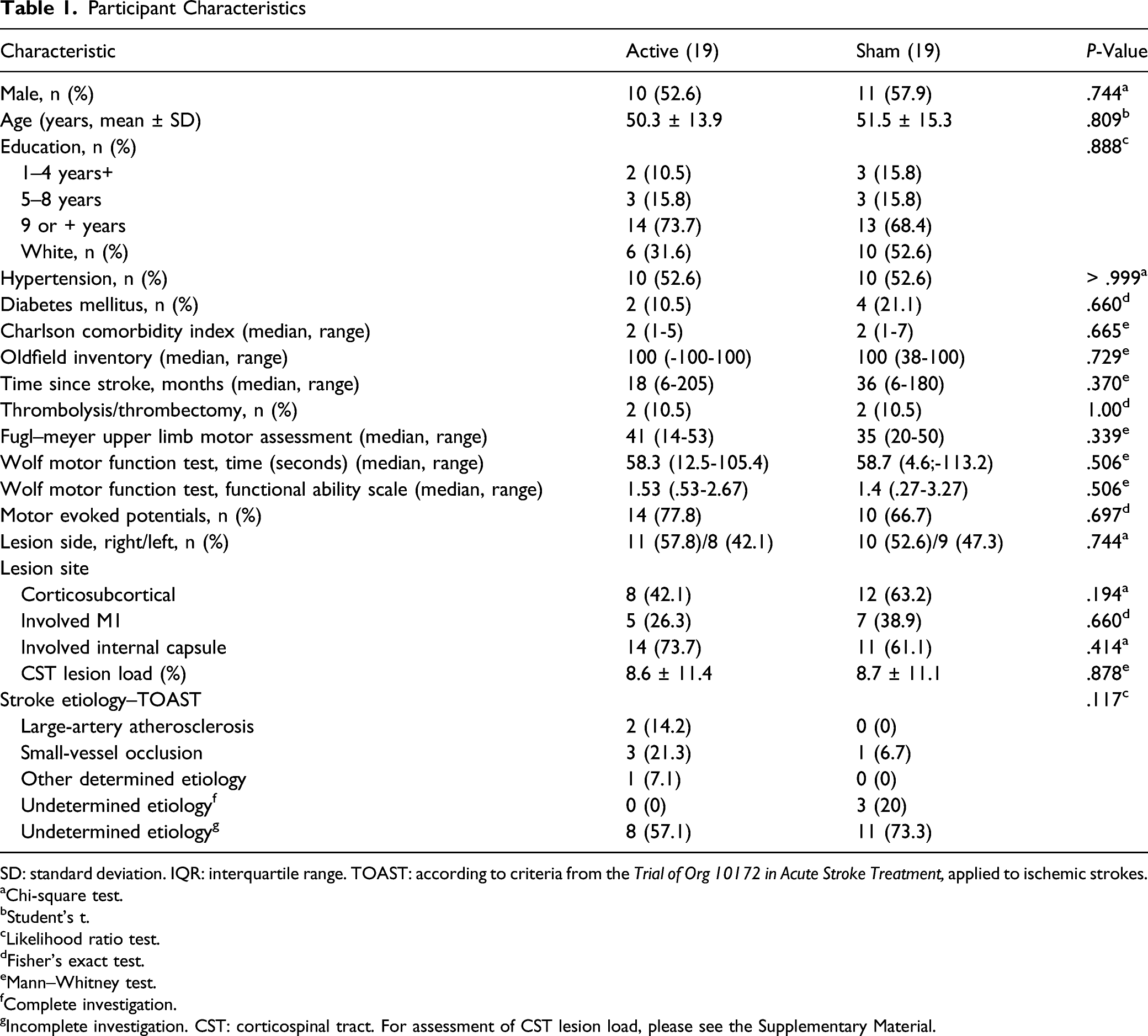
SD: standard deviation. IQR: interquartile range. TOAST: according to criteria from the Trial of Org 10172 in Acute Stroke Treatment, applied to ischemic strokes.
aChi-square test.
bStudent’s t.
cLikelihood ratio test.
dFisher’s exact test.
eMann–Whitney test.
fComplete investigation.
gIncomplete investigation. CST: corticospinal tract. For assessment of CST lesion load, please see the Supplementary Material.
Results of Intention-to-Treat Analysis Without Last Observation Carried Forward. Medians and Ranges are Shown.
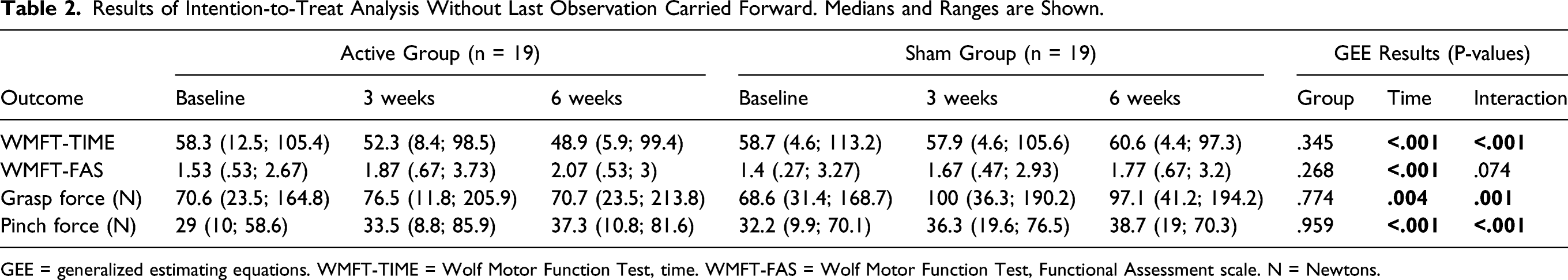
GEE = generalized estimating equations. WMFT-TIME = Wolf Motor Function Test, time. WMFT-FAS = Wolf Motor Function Test, Functional Assessment scale. N = Newtons.
Grasp strength improved significantly in the active but not in the sham group at 3 and 6 weeks. ES at 6 weeks was .35 in the active group. Pinch strength improved significantly in both groups at 3, and only in the active group at 6 weeks (ES, .61) (Figure 1). Changes in grasp strength at 3 and 6 weeks, normalized to baseline, in each subject in the active (left) and sham (right) groups. *P ≤ .025.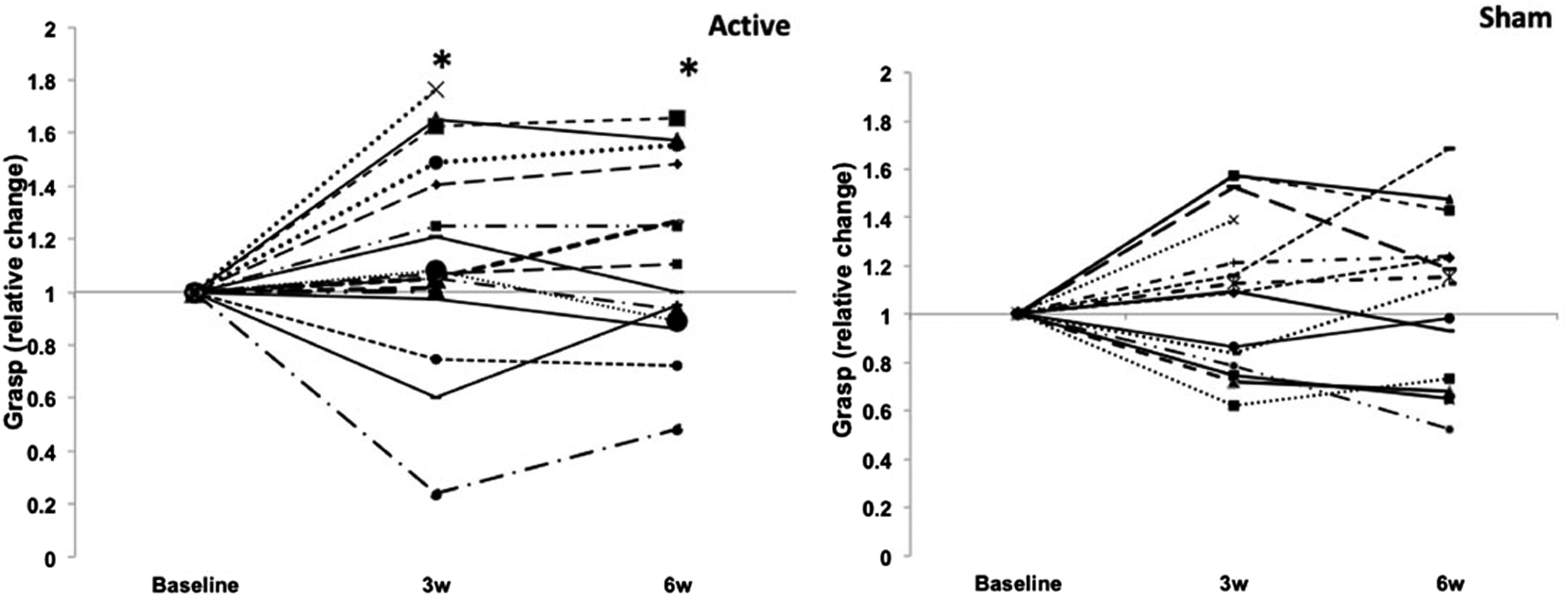
There were significant effects of time in both groups, with no interactions for WMFT-FAS (ES: .3 active, .31 sham), FMA, Minimum Acceptance Level, Read-Only Memory of wrist extension, Manufacturing Advisory Service, BI, some SIS domains, and BDI (Supplementary Material).
Intention-to-treat with LOCF and PP results were similar to results of ITT without LOCF, except for a lack of significant effects for grasp at 6 weeks in the active, and for pinch at 3 weeks in the sham group (Supplementary Material).
At 6 weeks, average changes in scores surpassed MCID for WMFT-TIME in both groups, and for WMFT-FAS only in the active group (Supplementary Material). MCID for WMFT-FAS was reached in 71.4% of subjects in the active, and in 42.9% in the sham group.
Post hoc analysis (Supplementary Material) was performed to assess the relation between the presence of Motor Evoked Potentials in the abductor pollicis brevis (MEPs), or the corticospinal tract lesion load at baseline (Table 1), and changes in the primary outcome at 6 weeks. According to ITT analyses, subjects with MEPs improved more than those without. Lesion load was inversely correlated with improvement, but the results were not statistically significant.
Discussion
The main result of this study was that WMFT improved significantly in both groups after 18 sessions of treatment (2.75 hsession). MCID was reached, on average, in both groups for WMFT-TIME and only in the active group for WMFT-FAS. Changes were greater in both groups compared to those described by Carrico et al 2 (for comparisons, see Supplementary Material). Two differences between patients´characteristics may have contributed to these findings: subjects randomized by Carrico et al 2 had more severe motor impairments and were, on average, 10 years older than those included in our study.
In addition, in contrast with our findings of significant decreases in WMFT in the active as well as in the sham group, Carrico et al (n = 36) reported significant decreases only in the active group. 2 Likewise, in a larger study that delivered 10 sessions of 4 hour intensive TOT following active RPSS (n = 33) or sham (n = 22), WMFT-TIME only improved significantly in the active group. 13 In both studies, no instructions to perform tasks at home were given. Conversely, another study from the same group 14 (n = 19) found significant improvements in WMFT-TIME after active RPSS or sham followed by 4-hour modified constraint induced-therapy (CIMT) that included instructions to perform tasks at home and wear a mitt on the unaffected arm for 90% of waking hours. Together with the results of the present study, these findings suggest that the performance of motor tasks at home may have led to gains in WMFT-TIME in both groups. Moreover FES, delivered to both groups in the present study but not in previous work, may have enhanced effects of TST. More work is required to assess whether FES can augment effects of RPSS or training in subjects with moderate to severe upper limb impairments after stroke.
Modest improvement at 6 weeks in pinch and grasp strength in the active but not in the sham group according to ITT analysis were consistent with reports of increase in force after single sessions of peripheral stimulation1,15 or 2 weeks of CIMT. 16
The main weakness of this study is the >20% attrition rate in both groups. Compliance with rehabilitation sessions represents a challenge to clinical trials and routine patient care. Furthermore, despite the results of debriefing, subjects may have suspected that they were in an active or sham group. We opted for lower limb stimulation as previously described 10 in order to mimic active treatment, without the same effect. Further studies are necessary to compare patients’ perceptions about different types of controls for RPSS.
Our hypothesis-generating findings should encourage larger trials to assess the efficacy of RPSS on strength in the chronic phase after stroke. Despite attrition and the shorter duration of supervised training compared to traditional CIMT, 17 this study showed clinical benefit in WMFT scores with an intervention of shorter duration than prior reports in the literature in subjects with comparable motor impairments. It remains to be confirmed if the presence of MEPs in hand muscles may help select subjects more likely to respond to training.
Improvements in WMFT scores after 3 weeks of treatment indicate that a closer evaluation of effect time-course across a treatment regimen may shed new light about the possibility of decreasing the number of sessions as well as the duration of interventions, while still deriving clinical benefit. If confirmed by larger trials, these results will have implications to settings of limited duration of direct contact between therapists and patients—or instance, due to costs of therapy or concerns about SARS-CoV-2 transmission.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211046259 – Supplemental Material for Repetitive Peripheral Sensory Stimulation as an Add-On Intervention for Upper Limb Rehabilitation in Stroke: A Randomized Trial
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683211046259 for Repetitive Peripheral Sensory Stimulation as an Add-On Intervention for Upper Limb Rehabilitation in Stroke: A Randomized Trial by Adriana B. Conforto, André G. Machado, Nathalia H. V. Ribeiro, Ela B. Plow, Sook-Lei Liew, Claudia da Costa Leite, Artemis Zavaliangos-Petropulu, Isabella Menezes, Sarah M. dos Anjos, Rafael Luccas, Paul Hunter Peckham and Leonardo G. Cohen in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgments
We thank Danielle Pires, Paloma de Freitas and Eduardo A. Mello for participation in data collection.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Machado reports other from Enspire DBS, outside the submitted work. In addition, Dr. Machado has a patent 8,190,263 issued to Cleveland Clinic. Dr. Liew reports grants from the National Institutes of Health during the conduct of the study. Dr. Conforto, Dr. Ribeiro, Dr. Menezes, Dr. Dos Anjos and Dr. Luccas received funding from the National Institutes of Health (grant R01NS076348-01) during the conduct of the study. The other authors declare that there are no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Institute of Neurological Disorders and Stroke, National Institutes of Health (grant R01NS076348-01). The sponsor had no roles in study design, collection, analysis and interpretation of data, in the writing of this report and in the decision to submit the article for publication.
Supplementary Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
