Abstract
Background
Vagus Nerve Stimulation (VNS) paired with rehabilitation improved upper extremity impairment and function in a recent pivotal, randomized, triple-blind, sham-controlled trial in people with chronic arm weakness after stroke.
Objective
We aimed to determine whether treatment effects varied across candidate subgroups, such as younger age or less injury.
Methods
Participants were randomized to receive rehabilitation paired with active VNS or rehabilitation paired with sham stimulation (Control). The primary outcome was the change in impairment measured by the Fugl–Meyer Assessment Upper Extremity (FMA-UE) score on the first day after completion of 6-weeks in-clinic therapy. We explored the effect of VNS treatment by sex, age (≥62 years), time from stroke (>2 years), severity (baseline FMA-UE score >34), paretic side of body, country of enrollment (USA vs UK) and presence of cortical involvement of the index infarction. We assessed whether there was any interaction with treatment.
Findings
The primary outcome increased by 5.0 points (SD 4.4) in the VNS group and by 2.4 points (SD 3.8) in the Control group (P = .001, between group difference 2.6, 95% CI 1.03-4.2). The between group difference was similar across all subgroups and there were no significant treatment interactions. There was no important difference in rates of adverse events across subgroups.
Conclusion
The response was similar across subgroups examined. The findings suggest that the effects of paired VNS observed in the VNS-REHAB trial are likely to be consistent in wide range of stroke survivors with moderate to severe upper extremity impairment.
Introduction
Stroke is a leading cause of adult disability. Upper limb impairment and inability to effectively use the arm and hand for functional daily tasks are common and persists in approximately half of people who have upper limb impairment at onset. 1 These limitations have a detrimental impact on quality of life and improving upper limb impairment and function are a priority for stroke survivors. 2
The use of vagus nerve stimulation (VNS) paired with rehabilitation to improve moderate to severe upper limb motor deficits associated with chronic ischemic stroke was recently approved the U.S. Food and Drug Administration. VNS augments task specific neuroplasticity by providing rapid cholinergic, noradrenergic, and serotonergic modulation. 3 VNS paired with rehabilitation leads to greater recovery of forelimb function in rodent models than either motor training or VNS alone.3,4 A combined analysis of data from 2 pilot feasibility trials of VNS paired with rehabilitation therapy5,6 found an improvement in impairment following VNS in people with long-term arm weakness after ischemic stroke. 7 In the recently published pivotal VNS-REHAB trial, there was a significant difference in change in Fugl–Meyer Assessment Upper Extremity (FMA-UE) score in favor of paired VNS following 6 weeks of in-clinic therapy. 8 There was also a higher clinically important response rate, defined as a greater than 6-point improvement in the FMA-UE score, and improvements in functional measures with paired VNS at 90 days after completion of in-clinic therapy. However, participants did not have a uniform response to VNS, so identifying those with a higher chance of responding could optimize prescription of this therapy. Pooled analysis of data from both pilot trials did not find any clear relationship between baseline variables and change in FMA-UE score with VNS, although lower baseline Fugl–Meyer score was associated with greater improvement across both treatment groups. 7 However, this analysis was based on a small sample size.
Here we perform a post-hoc subgroup analysis of data from the VNS-REHAB trial. We identified variables of interest based on known predictors of upper limb outcome. We examined whether the effect of paired VNS treatment differs by reported sex, age, time from stroke, severity of upper limb impairment, country of enrollment, paretic side, and whether there was cortical involvement of the index infarction.
Methods
The design and methods of the VNS-REHAB trial have been previously described. 9 The trial was a randomized, blinded, sham-controlled clinical trial conducted at 19 stroke rehabilitation centers in the USA and UK. A total of 108 participants were enrolled between Oct 2, 2017, and Sept 12, 2019. Participants with moderate-to-severe arm weakness, at least 9 months after ischemic stroke, were randomly assigned (1:1) to either rehabilitation paired with active VNS (0.8 mA, 100 μs, 30 Hz stimulation pulses, lasting 0.5 seconds) or rehabilitation paired with sham stimulation (0.0 mA). Participants received 6 weeks of in-clinic therapy (3 times per week; total of 18 sessions) followed by a home exercise program. All participants gave full informed consent. The trial was approved by the regulatory authorities and ethics committees/institutional review boards in the US and UK.
Outcome Measures
The primary outcome for this analysis was the change in impairment measured by the FMA-UE score after completion of the 6-weeks in-clinic therapy. Secondary endpoints were change in FMA-UE score and Wolf Motor Function Test (WMFT) at day-90 after completion of in-clinic therapy, and clinically important response rate, defined as a greater than or equal to 6-point change in FMA-UE score at 90 days after in-clinic therapy 10 and a greater than 0.4-point change on the WMFT. 11 We also assessed occurrence of serious adverse events and adverse events related to device implantation and device use.
Subgroups of Interest
We identified the following variables of interest: reported age, sex (male vs female), country of enrollment (UK vs USA), severity of upper limb impairment, time since stroke, paretic side (right vs left) and whether there was cortical involvement of the index infarct. Severe upper limb impairment, time from stroke and age were dichotomized based on whether the baseline value was above or below the median (FMA-UE score of 34, time from stroke 2 years and age 62 years). Presence of cortical involvement was visually confirmed by a trained image analyst using data from the study specific MRI performed before VNS device implantation.
Statistical Analysis
All analyses used the intention-to-treat population. For the analysis of paretic side, only right-handed participants were included as the majority of participants were right hand dominant (48/53 [90.6%] in the VNS group and 50/55 [91%] in the control group). We calculated the mean difference and 95% CI for the FMA-UE and WMFT and the difference in response rates between active VNS and sham stimulation in the entire population and then by subgroup. We then assessed whether there was any interaction between subgroup factor and the mean difference in FMA-UE score at day 90 using a two-way ANOVA model including treatment and subgroup as factors. A P value of <.05 was used to determine statistical significance.
Results
About 108 participants were randomized to either the active VNS group (n = 53) or the sham stimulation group (n = 55). Baseline characteristics are shown in Table 1.
Baseline Demographic and Clinical Characteristics by Randomization Group in the Intention-to-Treat Population.
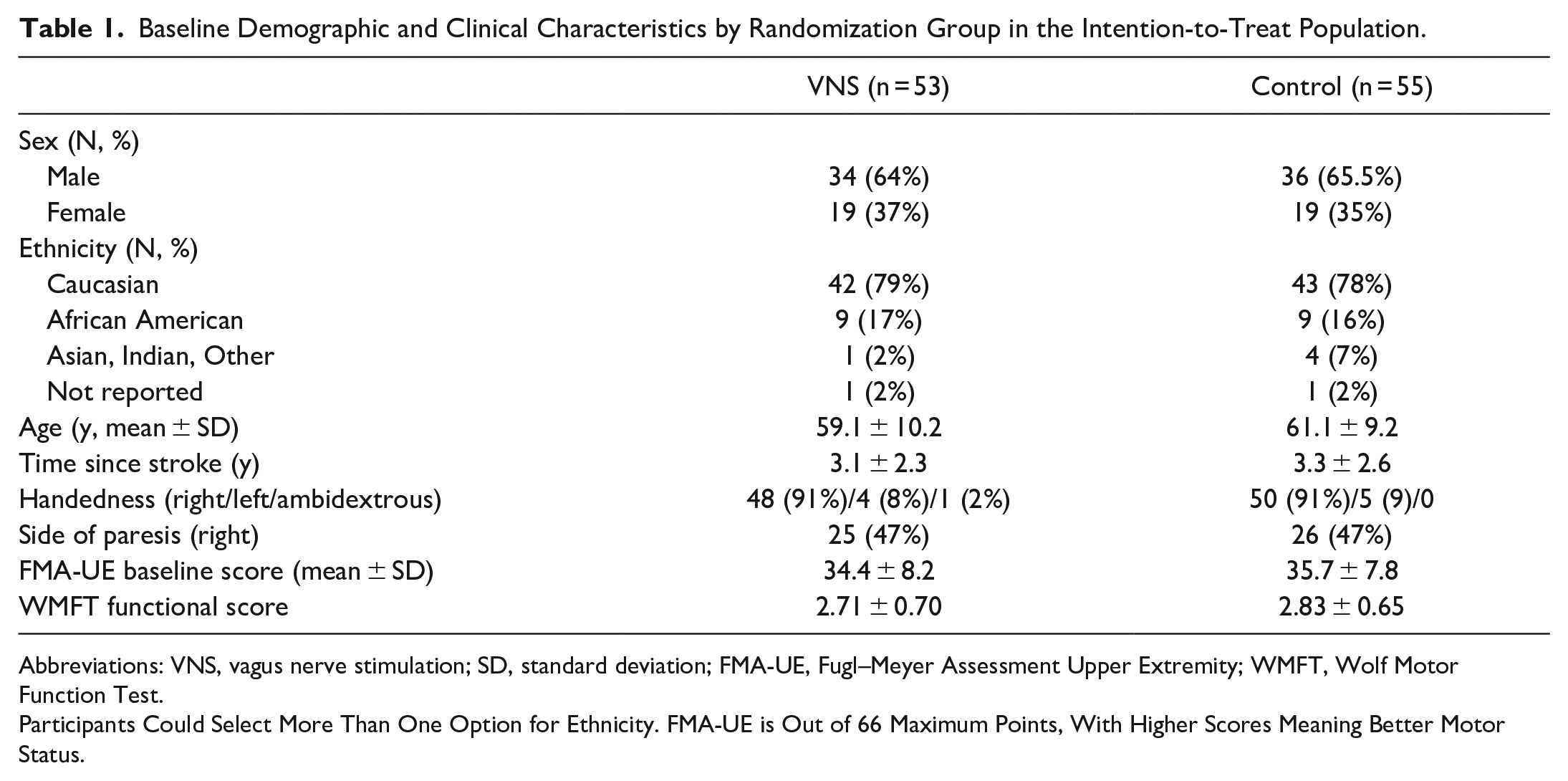
Abbreviations: VNS, vagus nerve stimulation; SD, standard deviation; FMA-UE, Fugl–Meyer Assessment Upper Extremity; WMFT, Wolf Motor Function Test.
Participants Could Select More Than One Option for Ethnicity. FMA-UE is Out of 66 Maximum Points, With Higher Scores Meaning Better Motor Status.
On the first day after completion of in-clinic therapy, the mean (±standard deviation) FMA-UE score increased by 5.0 points (4.4) in the VNS group and by 2.4 points (3.8) in the Control group (P = .001, between group difference 2.6, 95% CI 1.03-4.2). This difference was similar across subgroups of age, sex, country, stroke severity, time from stroke, paretic side, and presence of cortical involvement (Figure 1A). The between group difference in FMA-UE score at day-90 was 3.0 points (95% CI 0.8-5.1) and was similar across subgroups (Figure 1B). The between group difference in WMFT at day-90 was 0.3 points (95% CI 0.2-0.4) and was similar across subgroups (Figure 2B). The between group difference in response rates on the FMA-UE score and WMFT were 24% (95% CI 6%-41%) and 35% (95% CI 18%-52%), respectively, and were similar across subgroups (Figure 3). No significant treatment interactions were observed with change in FMA-UE or WMFT score and subgroups of interest at either day 1 or day-90 post completion of in-clinic therapy (all P > .05).
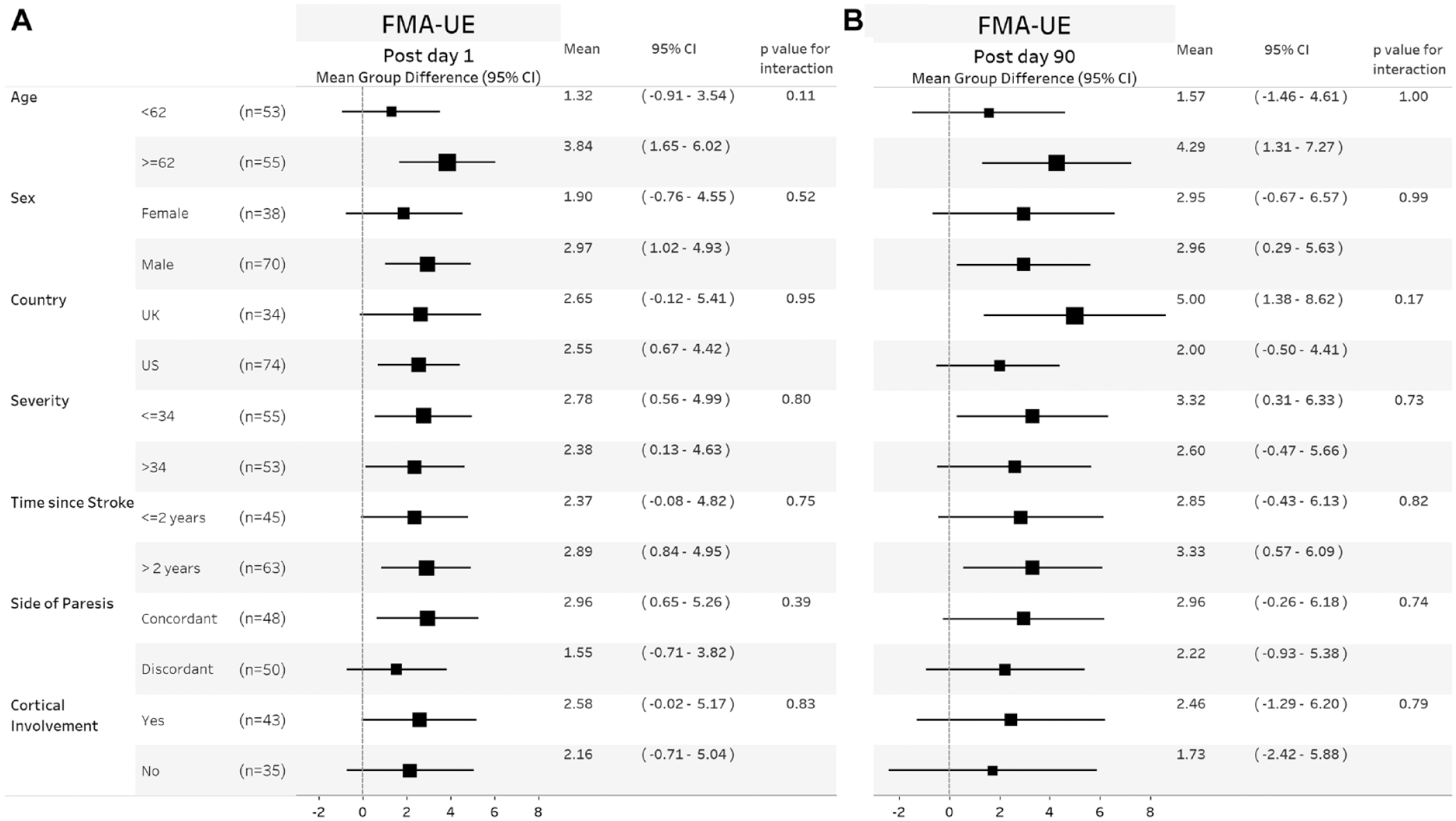
Forest plots showing mean group difference for FMA-UE change in score at day 1 (A) and day 90 (B) post completion of in-clinic therapy across different subgroups. Within each subgroup category, black square shows the mean group difference, and the size of the squares represents the degree of change. Horizontal lines denote 95% confidence. P values are given for the test of interaction between the group difference and subgroup of interest. Values to the right of the zero vertical line show a between group difference in favor of VNS.
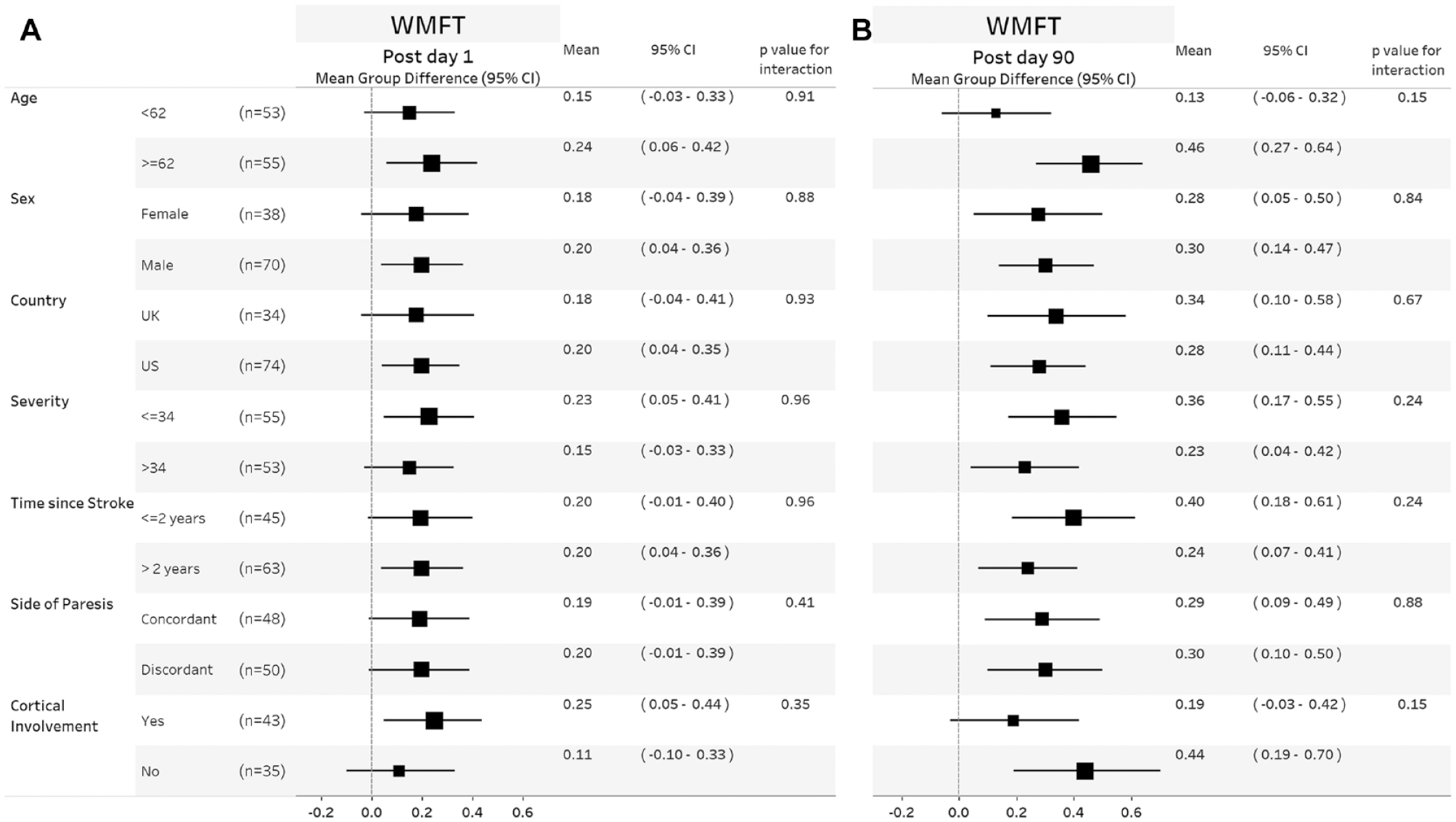
Forest plots showing mean group difference for WMFT change in score at day 1 (A) and day 90 (B) post completion of in-clinic therapy across different subgroups. Within each subgroup category, black square shows the mean group difference, and the size of the squares represents the degree of change. Horizontal lines denote 95% confidence. P values are given for the test of interaction between the group difference and subgroup of interest. Values to the right of the zero vertical line show a between group difference in favor of VNS. Values to the right of the vertical line show a between group difference in favor of VNS.
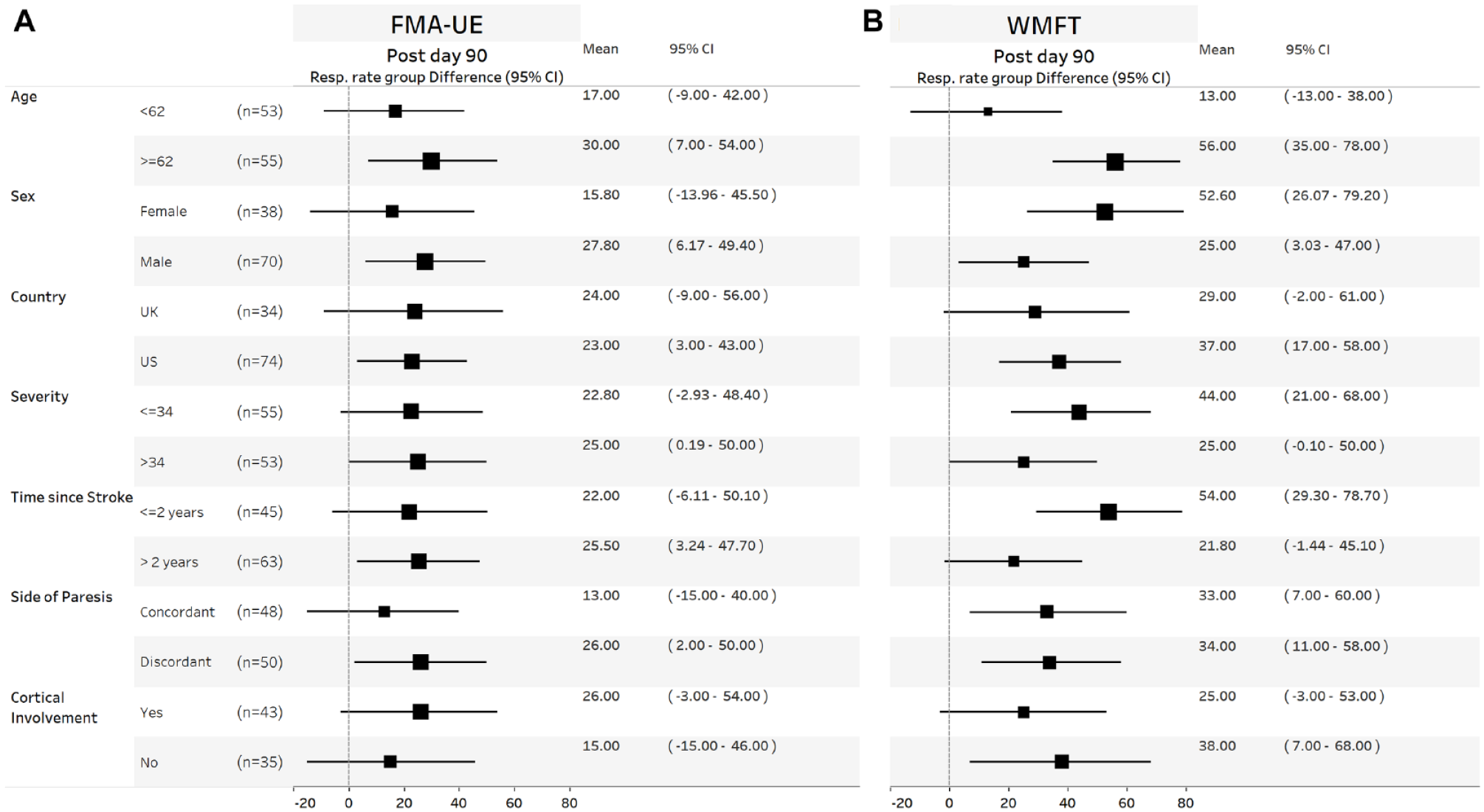
Forest plots showing mean group response rate difference for FMA-UE (A) and WMFT (B) at day 90 post completion of in-clinic therapy across different subgroups. Within each subgroup category, black square shows the mean group difference, and the size of the squares represents the size of the response difference. Horizontal lines denote 95% confidence. Values to the right of the zero vertical line show a between group absolute response rate difference in favor of VNS.
There were 15 reported SAEs in 12 participants. None were rated as related to study treatment or device implantation according to the investigators. There were 335 adverse events reported. The frequency of these events by subgroup is given in Table 2.
Table Shows the Number of Events Within Each Subgroup.
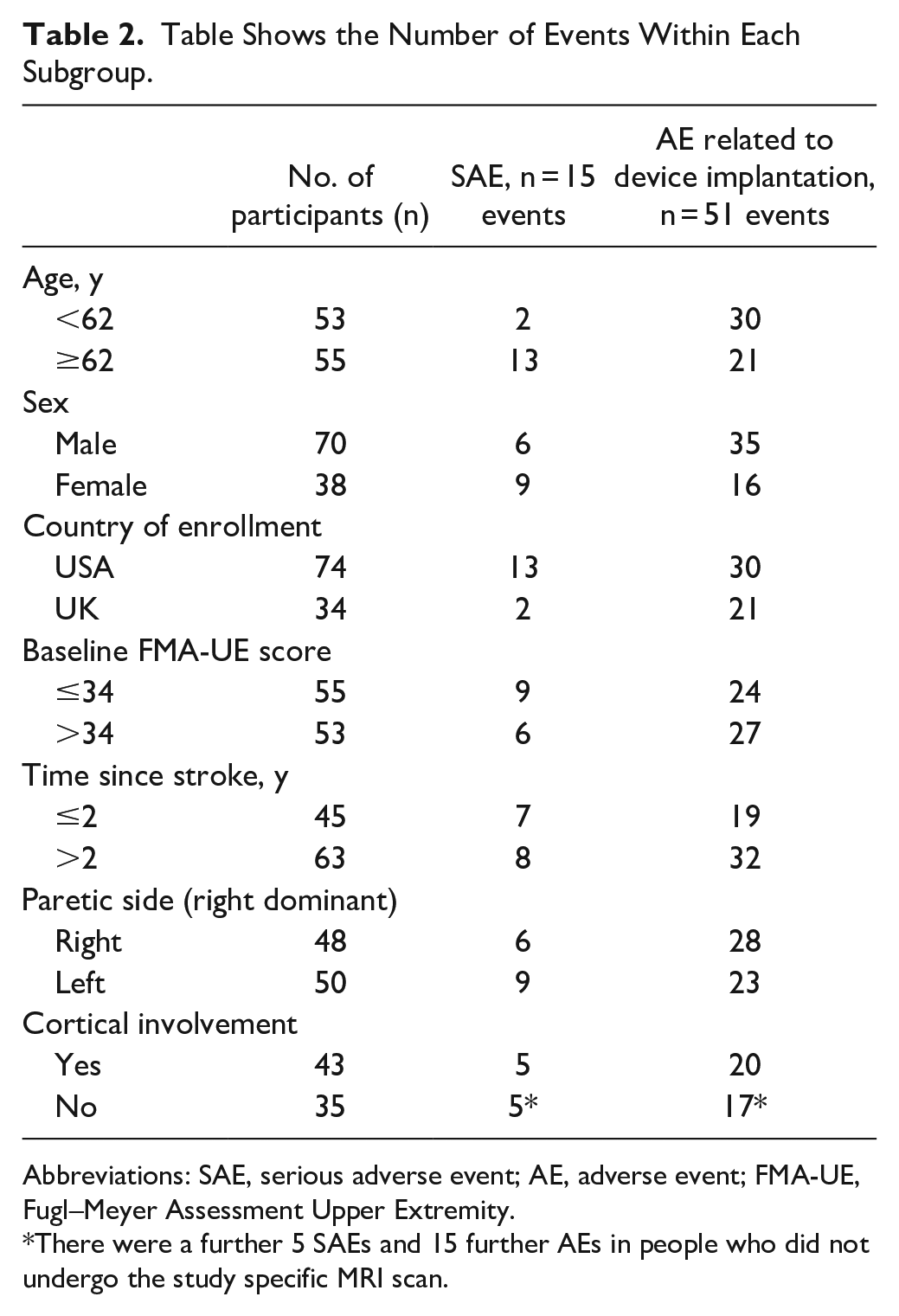
Abbreviations: SAE, serious adverse event; AE, adverse event; FMA-UE, Fugl–Meyer Assessment Upper Extremity.
There were a further 5 SAEs and 15 further AEs in people who did not undergo the study specific MRI scan.
Discussion
We observed that the beneficial effects of paired VNS on measures of upper extremity impairment and function in people with chronic ischemic stroke were consistent across subgroups including measures of age, sex, impairment severity, time from stroke, stroke location, and paretic side. Our findings suggest the response to VNS therapy is consistent across the range of participants who met the trial eligibility criteria.
Paired VNS uses an implantable VNS device and is performed under general anesthesia. As with any device implant, there are associated risks. There is a reported rate of transient vocal cord palsy of approximately 3% following all types of VNS device implantations. 12 We saw no evidence of a difference in rate of reported (unrelated) serious adverse events and adverse events due to implantation by subgroup.
Our findings are consistent with a previous analysis of data from pilot studies of paired VNS therapy 7 and from studies exploring predictors of constraint-induced movement therapy where there is little evidence of a difference in response across subgroups. 13 In addition, pre-clinical studies of VNS paired with rehabilitation have also shown a consistent effect in different experimental models of aged versus young animals, cortical and subcortical infarction, and intracerebral hemorrhage.4,14,15 Although there are well described predictors of upper limb recovery after stroke, such as age, sex, lesion site, severity of motor impairment, and measures of evoked potentials identified in a large systematic review and meta-analysis, 16 studies of response to specific interventions were excluded. Recent studies have explored the predictive ability of biomarker-based algorithms to predict recovery of upper limb function when used within days of stroke. 17 These data incorporate measures of stroke severity and movement and motor evoked potentials but whether such tools can predict response to paired VNS therapy in chronic stroke requires further exploration. Time from onset of stroke and degree of impairment have been associated with response to robotic therapy in subacute stroke, 18 but not with more chronic impairment. Corticospinal tract excitability has been associated with response to robotic training after chronic stroke 19 and the degree of injury to the corticospinal tract is associated with response to therapy. 20 We did not gather data on corticospinal tract excitability in our study, so we cannot assess whether this factor would be associated with response to VNS.
This analysis has additional limitations. The small sample size constrains our ability to identify small but potentially important differences between subgroups and precludes us from further dividing the groups (ie, older females vs older males). In addition, the variables we have assessed are not exhaustive and there remain several variables of interest. We defined stroke severity according to the median of the baseline FMA-UE score within our study population. Other studies suggest that a score of greater than 31 corresponds with poor arm capacity on the Action Research Arm test score. 21 We therefore feel the median values is clinically informative for analyses, but we have not explored other cut-offs. We excluded people with significant sensory impairment from the study so cannot assess whether such people would benefit. In addition, people with a FMA-UE score of <20 were not included. Therefore, although we saw no evidence of a differential effect by stroke severity, we have not as yet acquired data on the most severely affected participants. This was also a post hoc analysis.
In summary, the response to VNS treatment was similar across subgroups of interest and there was no obvious important difference in rates of adverse events across subgroups. These findings suggest that the effect of VNS observed in the VNS-REHAB trial 8 are likely to be consistent in a wide range of stroke survivors with moderate to moderately severe upper extremity impairment. This should be clarified by further studies.
Footnotes
Acknowledgements
We would like to thank participants for their contribution to this research project. We thank all staff at VNS-REHAB study centers. We also wish to acknowledge the data safety monitoring board for their work during the trial (Chair Dr Joseph Edmonds, members Dr Mark George, Dr Scott Kasner, Dr Jaye Thompson). We also thank David Ng and Sean Spanyers at WuXi Clinical for guidance on the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SC serves as a consultant for Abbvie, Constant Therapeutics, MicroTransponder, Neurolutions, SanBio, Panaxium, NeuExcell, Elevian, Medtronic, Helius, Omniscient, and TRCare. JD has received reibursement for conference expenses to present trial data. NE, BT, DP, CP are employees of MicroTransponder Inc. SLW is a consultant to Enspire and serves on the Scientific Advisory Board of Saebo. GEF has received research grants, consulting honoraria, or both from Allergan, Ipsen, Merz, MicroTransponder, Ottobock/Hangar Orthopedics, Parker Hannifin, Revance Therapeutics, ReWalk, and Sword Health. The MGH Translational Research Center has a clinical research support agreement with Neuralink, Paradromics, and Synchron, for which LRH provides consultative input. The remaining authors declare no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was funded by MicroTransponder Inc.
