Abstract
Background. Time outside therapy provides an opportunity to increase upper limb (UL) use during post-stroke hospitalization. Objective. To determine if a self-directed UL program outside therapy (Self-Empowered UL Repetitive Engagement, SURE) was feasible and to explore the potential effect of the SURE program on UL use and recovery. Methods. Twenty-three patients from an inpatient rehabilitation center who were ≤21 days post-stroke and had a Fugl Meyer UL (FMUL) score ≤50 and a positive motor evoked potential (MEP+) response were randomized (stratified by impairment) to either experimental group (SURE: individualized, UL self-exercise and use outside therapy for 6 hours/week for 4 weeks) or control group (education booklet). Feasibility was evaluated by program adherence, dropout rate, adverse events, and satisfaction. Potential effect was measured by paretic UL use via accelerometry weekly during the intervention, FMUL and Action Research Arm Test (ARAT) at baseline (Week 0), post-intervention (Week 4), and follow-up (Week 8 and Week 16). Results. Adherence to SURE was high: 87% program completion (mean 313±75 repetitions/day). There were no dropouts, no adverse events related to SURE, and patient satisfaction averaged 7.8/10. Experimental participants achieved an additional hour of UL use daily (range: .3–1.2 hours/day) compared to control. Significant improvements in FMUL and ARAT were observed in both groups from Week 0 to Week 4 and to Week 8 (P ≤ .002), which were maintained to Week 16. There were no differences between groups (P ≥ .119). Conclusions. SURE was a feasible self-directed program that increased UL use in MEP+ individuals with moderate-severe impairment early post-stroke. Further studies with larger sample sizes and potentially higher dose are required to determine efficacy.
Introduction
Upper limb (UL) recovery is often limited after stroke.1-3 Studies have shown that the highest rate of UL recovery occurs during the first 4 weeks post-stroke.4,5 It is during this early period that rehabilitation can interact with a heightened potential for spontaneous biological recovery to drive positive neuroplastic change and motor recovery after stroke.6,7 Although the optimal dosage during early rehabilitation is still unknown, early intervention studies8,9 and meta-analyses10-12 have shown that intensive task-specific practice is important for UL outcome after stroke.
In the clinical setting, UL practice is low. A recent systematic review demonstrated on average only 4–17 minutes of UL practice per therapy session per day during acute and subacute rehabilitation is performed. 13 Upper limb use outside therapy is dramatically lower than during therapy. 14 Thus, it is not surprising to find that paretic UL use throughout the day during inpatient rehabilitation is reported to be lower than non-paretic UL use.14-19 Stroke inpatients with moderate and severe UL impairment also demonstrate significantly less UL use compared to people with mild UL impairment. 14 This indicates it may be crucial to design targeted interventions to increase UL use based on UL impairment level, paying particular attention to those with moderate and severe UL impairment. It also suggests that stratification based on level of motor impairment is an important consideration in trial design. 20
Given time outside therapy during inpatient rehabilitation has been under-exploited, it is important to consider how to use this time to drive better recovery after stroke. Empowering individuals to perform self-exercise outside therapy could increase practice without a corresponding increase in resources. The Graded Repetitive Arm Supplementary Program (GRASP) demonstrated that it was possible to teach people with stroke to perform self-exercise outside therapy, increase UL practice, and improve UL outcome. 21 To maximize recovery, it is important to facilitate the use of paretic UL in daily functional activities in addition to self-exercises to overcome the reduced paretic UL use in people with stroke found in previous studies.14-19 A possible reason of the reduced paretic UL use might be due to the “learned nonuse phenomenon” 22 in which people with stroke have learned not to use their paretic UL to the full capacity. 23 Learned nonuse might be due to the reduced UL motor ability of people with stroke resulting in either nil or more clumsy and inefficient paretic UL movement that discourages UL use. 24 People with stroke then compensate with using non-paretic UL most of the time and eventually learn not to use the paretic UL, resulting in the paretic UL being “held in powerful inhibition.” 24 It is therefore important to educate and guide paretic UL use in functional activities during early rehabilitation to prevent learned nonuse further down the path of recovery.
For the people with moderate and severe UL impairment, the use of the paretic UL in daily functional activities is even more challenging as they do not have enough movement to work with 25 and often encounter problems performing reaching and grasping activities that are fundamental to daily functional activities. In addition, it has also been reported that people with moderate and severe UL impairment do not know what to do to increase use of their paretic UL in daily functional activities. 26 Though challenging, it is therefore crucial to teach people with reduced UL motor ability to actively engage their paretic UL in daily functional activities during early rehabilitation to increase UL use for the long term. The use of paretic UL in daily functional activities was limitedly addressed in GRASP. 21 To address this issue, we developed the Self-Empowered Upper Limb Repetitive Engagement (SURE) program, which includes self-exercise and education to facilitate use of the paretic UL in functional activities outside therapy for those with moderate and severe UL impairment. With the teaching of paretic UL use in functional activities in the SURE program, it was intended that the possibility for increased confidence in activities of daily living (ADL) performance, previously found to be associated with increased paretic UL use, 26 would result in an increase in paretic UL use. In working with the trainer to explore SURE program self-exercises/functional activities, people with stroke could learn problem-solving skills to enable paretic UL use in daily functional activities. Undertaking exercises during time outside therapy leads to efficient use of precious resources. Both problem-solving and utilization of resources skills are key skills in self-management. 27
As the SURE program is a new intervention, it is important to investigate the feasibility of the delivery of the program in the hospital setting and its potential effect on UL use and recovery. The first aim of this study was to investigate the feasibility of the delivery of the SURE program in the hospital setting. The second aim was to explore the potential effect of the SURE program on UL use and recovery. Following the recommendation of the Stroke Recovery and Rehabilitation Roundtable, 28 evaluation of motor evoked potential (MEP) status was used as a biomarker to stratify and select people with similar UL recovery potential into this study. Participants with positive MEP status (MEP+), a representation of corticospinal tract excitability, 29 were selected. The authors anticipated that the SURE program would be feasible for stroke survivors who are MEP+ and that it would have a positive potential effect on UL use and recovery.
Methods
Study Design
A randomized controlled Phase II pilot trial was undertaken.30,31 Participants were enrolled by the primary investigator. A non-team member generated a randomized allocation sequence in 3 blocks of 8, stratified by UL impairment using Fugl Meyer UL (FMUL) scores of 0–22, 23–40, and 41–50. 32 Group allocation was concealed by use of individual opaque envelopes. Envelopes were opened when a participant was assigned to their group by the trainer after baseline assessment.
Participants
All people with stroke admitted to Tan Tock Seng Hospital Rehabilitation Centre, Singapore, from February 2018 to March 2019 were screened through medical records for inclusion. The inclusion criteria were diagnosis of stroke due to infarct/hemorrhage (first or recurrent) resulting in UL hemiparesis with a FMUL score ≤50; ≤21 days post-stroke; Montreal Cognitive Assessment ≥19; and MEP+ for paretic extensor carpi radialis. Motor evoked potential status was assessed using single-pulse transcranial magnetic stimulation over the motor area hotspot using a Magstim 200 device and a figure of 8 coil. The MEP assessment was completed by an independent trained research assistant. Motor evoked potentials were observed with surface electromyography recorded over the paretic extensor carpi radialis. Stimulation was delivered with the patient seated and their forearm at rest. If a resting MEP could not be achieved at maximum stimulator intensity, that is, 100%, participants were asked to extend their wrist during stimulation to determine if a MEP could be recorded during activation MEP. Participants were considered MEP+ if 4 MEPs of any amplitude with a consistent latency from a maximum of 8 trials were observed. 33 Participants were considered MEP negative (MEP−), and excluded from the trial, when no MEP was achieved at maximal intensity stimulation with activation.
Exclusion criteria were MEP− status; prior history of central nervous system disorders or structural brain lesions (e.g., Parkinsonism or brain tumor); medical history of psychiatric disturbance; bilateral stroke; cardiac disease limiting function by exertional dyspnea, angina, or severe fatigue; hemiplegic shoulder pain rated >5/10; peripheral nerve or orthopedic conditions that interfered with paretic UL movement; severe aphasia, neglect, agitation, or depression that in the opinion of the treating medical and therapy team could limit participation in therapy; and contraindications to transcranial magnetic stimulation, for example, pacemaker. This study was approved by National Healthcare Group Domain Specific Review Board Ethics, Singapore (2016/00600), and University of Queensland, Australia (2018000760), and conforms with the Declaration of the Helsinki. All participants provided written informed consent. There was a change in study protocol on June 14, 2018 based on new evidence 34 which led to the inclusion of individuals with hemorrhagic and recurrent stroke. Study reporting followed the CONSORT 2010 statement: extension to randomized pilot and feasibility trials 35 and the intervention was reported according to TIDieR guidelines. 36
Experimental Group
The experimental group received a SURE program booklet with instructions and illustrations of exercises and functional activities appropriate to their FMUL score (Level 1A for FMUL score =0–10 (severe); Level 1B for FMUL score =11–22 (severe); and Level 2 for FMUL score =23–50 (moderate)). 32 Each program booklet consisted of warm up, strengthening, simple physical tasks, and functional activities integrated into daily UL use such as wiping the face, wiping the table after eating, applying lotion to limbs, and drinking from a cup. The SURE program booklets included 20 exercises and 17 functional activities. Each participant was given five to eight exercises including at least one functional activity to practice. Exercises and functional activities were selected, graded, and progressed by the trainer based on individual UL motor ability and goals during each review. The trainer was a physiotherapist with 19 years of experience working with stroke patients. Participants were asked to practice the selected self-exercises each session (i.e., to perform one self-exercise for 20 repetitions, with 5 to 8 exercises each session, totally approximately 20 minutes/session), 3 sessions/day distributed across morning, afternoon, and evening, 6 days/week for 4 weeks. Participants were asked to use their paretic UL to perform the selected functional activities daily whenever needed across the 4-week intervention period. Participants were taught how to practice the exercises and functional activities during the first session and they were instructed to record their practice on a daily log (i.e., to tick on the box for each self-exercise after completing 20 repetitions for a particular time of day (morning/afternoon/evening)). Each participant’s program and daily logs were reviewed three times/week in Weeks 1 and 2, twice/week in Week 3, and once in Week 4 by the trainer. If participants were discharged from the hospital before the end of the intervention period, they continued the program at home with reviews occurring at the same intervals. Participants and the trainer were not blind to group allocation. The therapists delivering usual care were aware of the study but they were not aware of the group allocation. Participants’ caregivers, for example, spouses or close relatives, were invited to assist, monitor, encourage, or support the participants in SURE program performance. Nurses were minimally involved in assisting the participants in the SURE program performance.
Control Group
The control group received an education booklet with 10 modules on stroke: (1) general information; (2) recovery after stroke; (3) the rehabilitation journey; (4) knowing your medication; (5) health-frequently asked questions; (6) eating well for rehabilitation; (7) eating well to reduce recurrence; (8) activity scheduling; (9) fatigue management; and (10) fall prevention. The material was developed locally by the Tan Tock Seng Hospital Rehabilitation Centre Psychoeducation team in 2015 and was modified to form the Stroke Education Booklet for the purpose of this study. The same trainer went through and reviewed 2–3 modules per week to eventually cover all modules with participants at the same frequency as the reviews performed by the trainer in the experimental group.
Usual Care
Both groups continued with their usual care which typically consisted of two 45-minute therapy sessions daily, one of occupational therapy and one of physiotherapy. Content of usual care for the UL consisted of neuromuscular electrical stimulation, Armeo Power robotic device, hand bike, UL retraining, ADL training, and UL circuit group therapy.
Outcome Measures
Participants were assessed by a blinded assessor at baseline before randomization (Week 0), then at mid-intervention (Week 2), post-intervention (Week 4), and at two follow-up time points (Week 8 and Week 16). Assessors were occupational therapists trained by video for FMUL and group training for the Action Research Arm Test (ARAT) 37 assessments. Demographics including age, gender, diagnosis, time post-stroke, paretic side, National Institute of Health Stroke Scale at acute and rehabilitation admission, and Functional Independence Measures at rehabilitation admission and discharge were collected from medical records. Participants reported on their UL dominance, prior and after stroke living arrangement, and education level.
Feasibility was measured by adherence, dropout rate, adverse events related to SURE program, and participant satisfaction. Adherence to the SURE program was expressed as a percentage of prescribed exercise and functional activities sets completed as recorded in the daily log, based on individual prescriptions of 5–8 exercises/functional activities, 3 times/day for 6 days with each week totaling 90–144 sets (with 20 repetitions in each set). Dropout rate was defined as the percentage of participants that dropped out over the total number of participants commencing the SURE program. Adverse events were collected via reports by the trainer, the blinded assessors, and participants themselves (i.e., during the SURE program reviews, blinded assessments, etc.), as well as medical records during the whole study period. The adverse events, examined by two investigators, were then categorized by their relationship to the SURE program and the trial as unlikely, possibly, probably, or almost certainly related and by severity as non-life threatening or life-threatening. To determine if the program was associated with increased UL resistance to passive movement, pain, or fatigue, the Modified Ashworth Scale 38 was used to rate the resistance to passive movement of paretic biceps and finger flexors, a numeric rating scale was used to reflect paretic UL pain, 39 and the Stanford Fatigue Visual Numeric of fatigue level 40 was rated at Week 0, Week 2, and Week 4. To evaluate satisfaction, experimental participants, caregivers, and nurses were asked to complete a questionnaire using Likert scales (0 = not easy/not at all/not satisfied at all to 10 = very easy/greatly/very satisfied) at Week4 to rate the program’s ease of implementation, impact on UL recovery, and overall satisfaction. Nurses were also asked to rate the amount of assistance they needed to provide to enable participants to complete the SURE program in the ward using a Likert scale (0 = no need to help at all to 10=a lot of assistance).
To measure the amount of paretic UL use during the intervention period, all participants wore two tri-axial accelerometers (ActiGraph GT3X-BT, ActiGraph LLC, Pensacola, Florida), one on each wrist for at least 2 weekdays (maximum 3 depending on weekends) in Week 1. For Week 2, Week 3, and Week 4 of the intervention period, all participants wore one tri-axial accelerometer on the paretic wrist for 2–3 weekdays. Treating therapists recorded the start and stop time of therapy session when accelerometers were worn. Accelerometry data were sampled at 30 Hz and then converted to use counts (.01664 g/count) and integrated into 1-second epochs using ActiLife 6 software (ActiGraph; Pensacola, FL). Duration of non-paretic and paretic UL use and Week 1 use ratio of paretic/non-paretic UL use (based on previous UL stroke studies41,42) were derived from vector magnitude. Vector magnitude was defined as the combination of acceleration along the three axes, that is, √(x2 + y2 + z2). Every second with vector magnitude ≥2 was considered activity and used to calculate UL use.43,44 Accelerometry data of 24 hours (which included therapy time) were collected. Average number hours of UL use per day measured by accelerometry during that week were feedback to all participants verbally. No prior accelerometry data were collected before the intervention period. The accelerometry outcomes were calculated using custom scripts created in Matlab R2015a (Mathsworks, Nattick, IL) software. To measure the amount of paretic UL use after the intervention period, participants were assessed using the Rating of Everyday Arm-use in the Community and Home (REACH) scale at Week 8 and Week 16 follow-up. The REACH scale is a reliable self-report measure that captures how much people with stroke use their paretic UL outside the clinical setting. 45
The primary outcome for examining the potential effect of the SURE program on UL recovery was FMUL, and the secondary outcome was the ARAT. These outcomes were collected at all time points. The FMUL assessment is a clinical tool consisting of 33 items (scored 0–66) widely used to measure motor impairment in people with stroke. 46 A higher score denotes greater UL motor ability. 47 The FMUL has high intra-rater reliability and inter-rater agreement. 47 The ARAT comprises grasp, grip, pinch, and gross movement subscales. The measure is scored out of 0–57; a higher score denotes greater UL function. 37 The ARAT has high intra-rater reliability and inter-rater reliability in measuring UL function in people with stroke. 37 A minimal clinically important difference (MCID) of 10 points in FMUL 48 was used to evaluate whether participants achieved a meaningful improvement in their UL recovery from Week 0 to Week 16. An MCID of 12 points (dominant side affected) and 17 points (non-dominant side affected) 49 in the ARAT from Week 0 to Week 16 was used to determine if a meaningful improvement in UL activity level was achieved. Finally, to assess if participants from both groups achieved their expected proportional recovery, we compared the published rule (recovery = [66-FMUL (Week 0)]∗0.7) 29 to our data collected at baseline (Week 0) and Week 16.
Statistical Analysis
As a Phase II pilot randomized trial, n = 12 for each group was chosen for feasibility 50 and not powered to determine the effect of SURE program intervention. Data were analyzed as intention to treat. Normality of data was tested using Shapiro–Wilk tests. Comparison of baseline characteristics between the experimental and control groups was performed using unpaired t-tests for normally distributed data, Mann Whitney U test for non-normally distributed data, and Chi-square test for nominal data. All outcome data except for the Modified Ashworth Scale, UL pain, and fatigue were normally distributed.
To evaluate SURE program feasibility, descriptive statistics of adherence, dropout rate, adverse events related to SURE, and satisfaction with the SURE program were calculated. To evaluate if the SURE program affected UL resistance to passive movement, pain, and fatigue, Friedman ANOVAs were performed across three time points (Week 0, Week 2, and Week 4) to examine within-group changes over time and Mann Whitney U tests to examine these outcomes between groups at each time point. To evaluate the potential effect of the SURE program on UL use, accelerometry data during the intervention period and REACH at two follow-up time points were compared between the experimental and control groups using linear mixed models with week x treatment group interaction and age as a covariate. Similarly, to evaluate the potential effect of the SURE program on recovery, linear mixed models with time points x treatment group interaction and age as covariate were used to compare the changes in FMUL and ARAT between the two groups. Statistical analysis was performed using SPSS version 25.0 for Mac (SPSS Inc., Chicago, Illinois, USA) with significance level set at P < .05.
Results
Participants
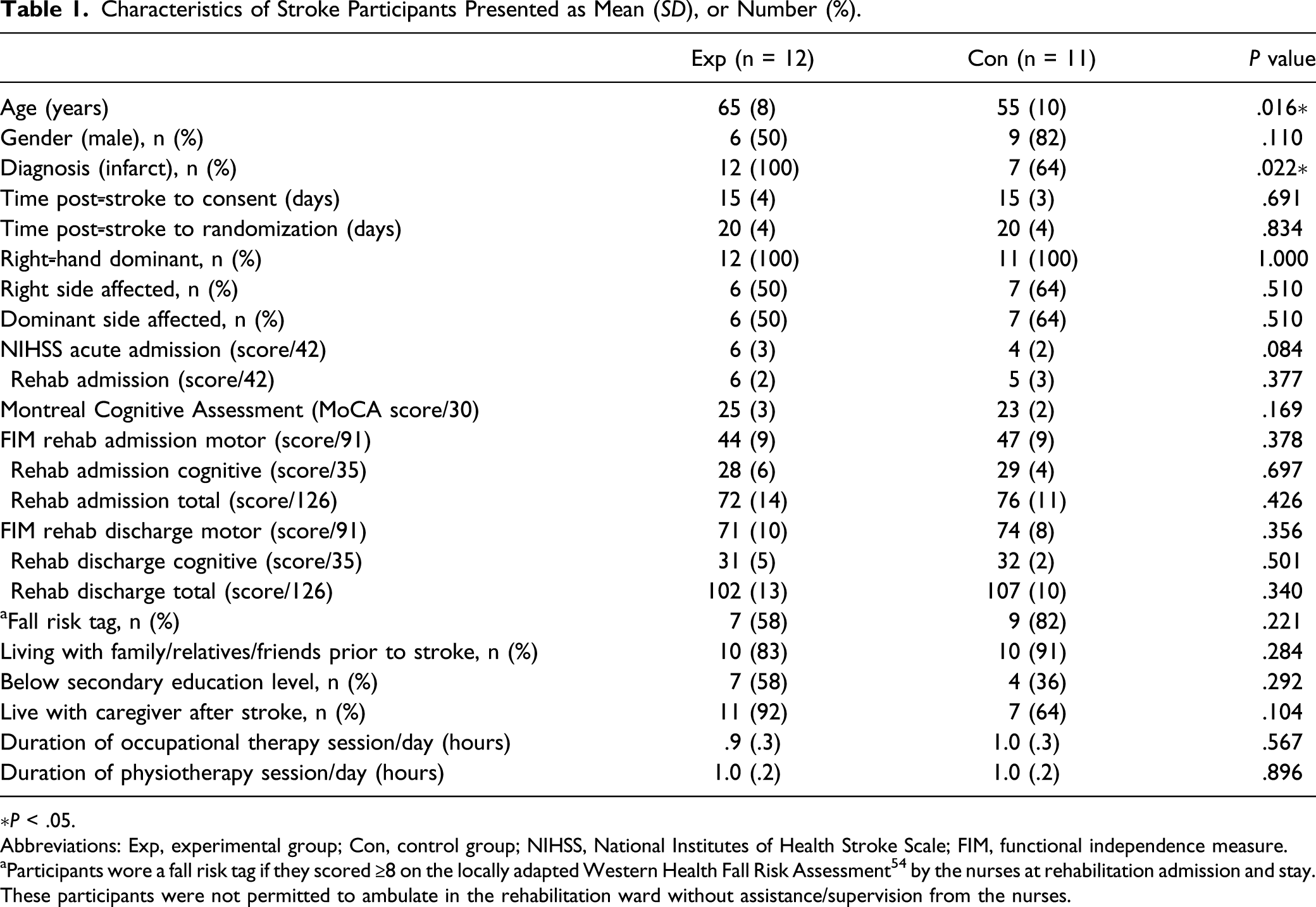
A total of 517 stroke patients were screened during the trial period (Figure 1). After 493 stroke patients were excluded, 24 eligible stroke patients agreed to participate and were randomized to the experimental or control group. In the experimental group, all twelve participants completed the intervention, and one participant did not complete Week 16 follow-up because of a second stroke occurrence prior to Week 16 follow-up. Data from this experimental group participant for Week 0 to Week 8 were retained for analysis as the second stroke occurred just prior to Week 16. One control participant had a second stroke at Week 2 and thus only had baseline data which was excluded from analysis. One experimental and one control participant developed redness and itchiness on their wrists when wearing accelerometers so stopped wearing accelerometers but continued to participate in the trial. One experimental and four control participants stopped wearing their accelerometers once they were discharged from the hospital as they refused or the discharge destination was uncertain. Participant characteristics were similar between groups except that experimental participants were significantly older (P = .016) and none had a hemorrhagic lesion (Table 1). Flow of participants through the trial. Characteristics of Stroke Participants Presented as Mean (SD), or Number (%). ∗P < .05. Abbreviations: Exp, experimental group; Con, control group; NIHSS, National Institutes of Health Stroke Scale; FIM, functional independence measure. aParticipants wore a fall risk tag if they scored ≥8 on the locally adapted Western Health Fall Risk Assessment
54
by the nurses at rehabilitation admission and stay. These participants were not permitted to ambulate in the rehabilitation ward without assistance/supervision from the nurses.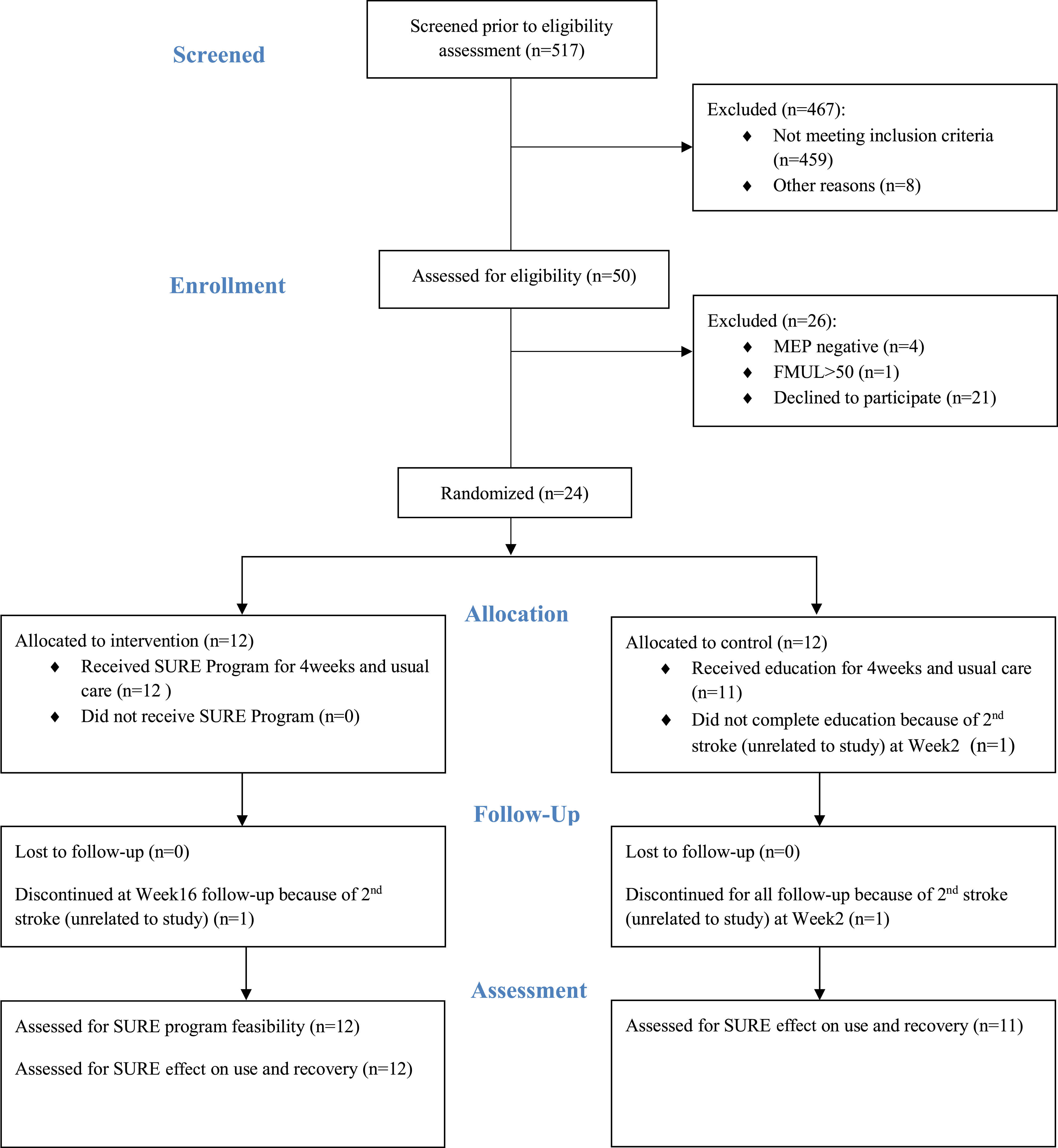
Feasibility and Safety of SURE Program
Adherence to the SURE program was high with an average of 87% (SD12, range: 66% to 99%) of prescribed exercises and functional activities completed over the 4-week program. Participants performed an average of 7330 repetitions across the 4-week SURE program (SD1798, range: 4060 to 10440), an average of 1876 repetitions/week (SD448, range: 1015 to 2610) and 313 repetitions/day (SD75, range: 169 to 435). In the control group, all modules of the education booklets were completed by all participants prior to discharge.
Mean (SD) of Groups and Significance Level Across Baseline (Week 0), Mid-Intervention (Week 2), and Post-Intervention (Week 4) of Adverse Secondary Changes.
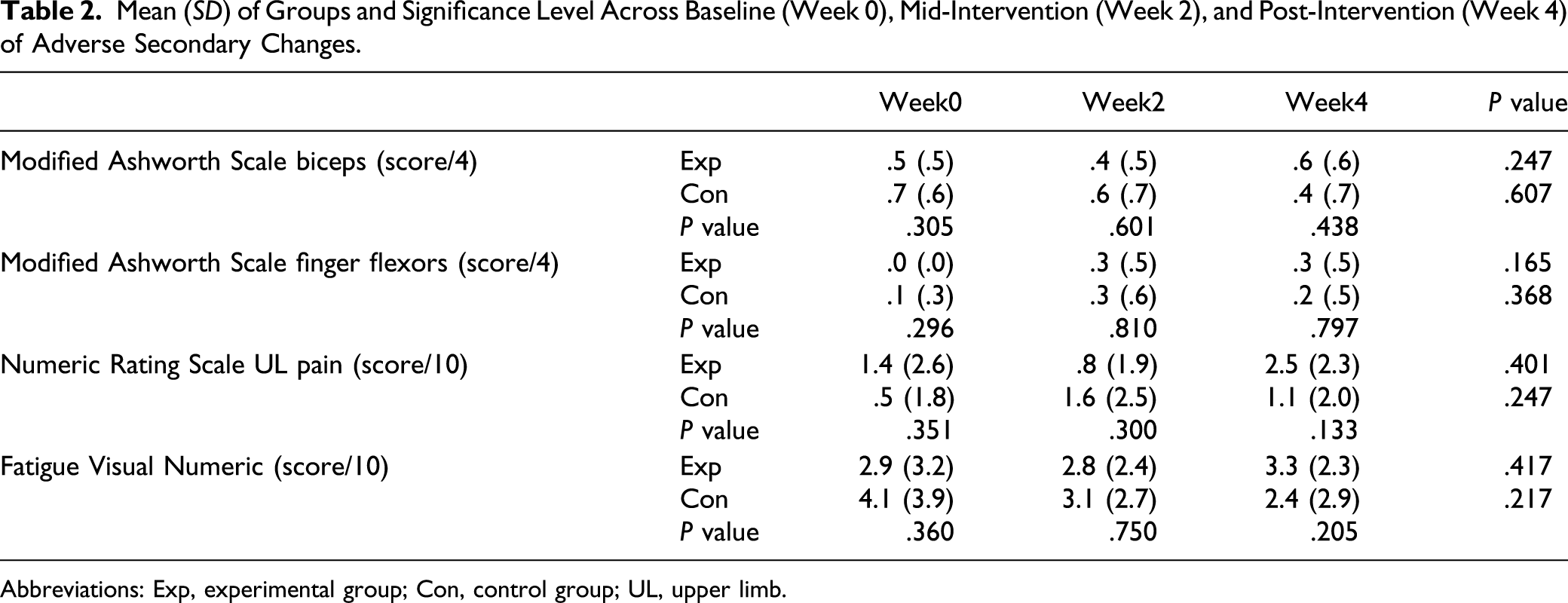
Abbreviations: Exp, experimental group; Con, control group; UL, upper limb.
All experimental participants completed the satisfaction survey. On average, participants rated the ease of carrying out the SURE program as 6.4/10 (SD2.6), the program helped with UL recovery as 7.5/10 (SD2.2), and overall satisfaction with the SURE program as 7.8/10 (SD1.8). Seven of the 12 participants had caregivers who assisted or supported them in the SURE program performance; six caregivers answered the questionnaire. On average, caregivers scored the ease of carrying out the SURE program as 7.5/10 (SD2.2), the program helped with UL recovery as 9.0/10 (SD1.3), and overall satisfaction with the SURE program as 9.2/10 (SD1.2). Ten nurses answered the questionnaire. Nurses scored the ease of carrying out the SURE program as 7.7/10 (SD1.6), the amount of assistance required by the nurses to enable participants to carry out the SURE program as 2.6/10 (SD2.4), the SURE program helping with UL recovery as 8.4/10 (SD1.5), and satisfaction with the SURE program as 9.1/10 (SD0.9).
Potential Effect on Upper Limb Use
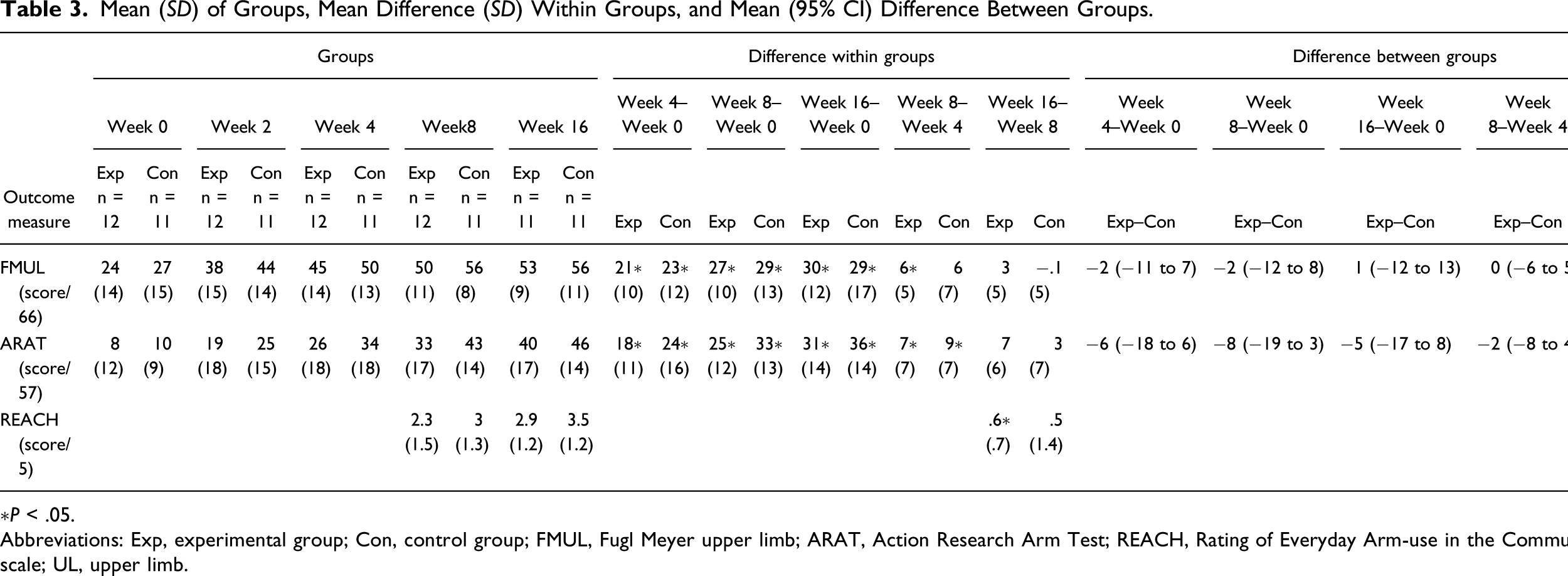
Experimental participants managed to use their UL approximately 1 hour per day more than controls in Week 1, Week 2, and Week 4 (range of mean difference: .9–1.2 hour, Figure 2A). During Week 1, experimental participants had significantly higher UL use than controls (experimental mean 4.5 (SD1.6) hours/day vs control mean 3.3 (SD0.7) hours/day; P = .036) and demonstrated no significant change in use across the weeks (P ≥ .088, Figure 2A and B). The control group had a lower level of UL use at Week 1, which increased over time (UL use higher in Week 4 than Week 1, P = .018, Figure 2C). The linear mixed model (week x group interaction, age as a covariate) revealed an increase in UL use in the control group between Week 1 and Week 3 and between Week 2 and Week 3 as compared to the experimental group (P ≤ .018). There was a significant improvement in the REACH scale from Week 8 to Week 16 in the experimental group (P = .009), but no difference in the control group (P = .237, Table 3). There was no difference in REACH scale change between groups from Week 8 to Week 16 (P = .863, Table 3). Graphs of 24 hours accelerometry UL use across Week 1 to Week 4 and comparison of UL use between experimental (exp) and control (con) groups: (A) comparison of UL use between experimental and control groups. ∗P < .05 between groups; (B) 24 hours accelerometry UL use across Week 1 to Week 4 for experimental group; (C) for control group. Each line represents an individual participant. Solid lines are participants with severe UL impairment, and dashed lines are participants with moderate UL impairment. Abbreviations: UL, upper limb. Mean (SD) of Groups, Mean Difference (SD) Within Groups, and Mean (95% CI) Difference Between Groups. ∗P < .05. Abbreviations: Exp, experimental group; Con, control group; FMUL, Fugl Meyer upper limb; ARAT, Action Research Arm Test; REACH, Rating of Everyday Arm-use in the Community and Home scale; UL, upper limb.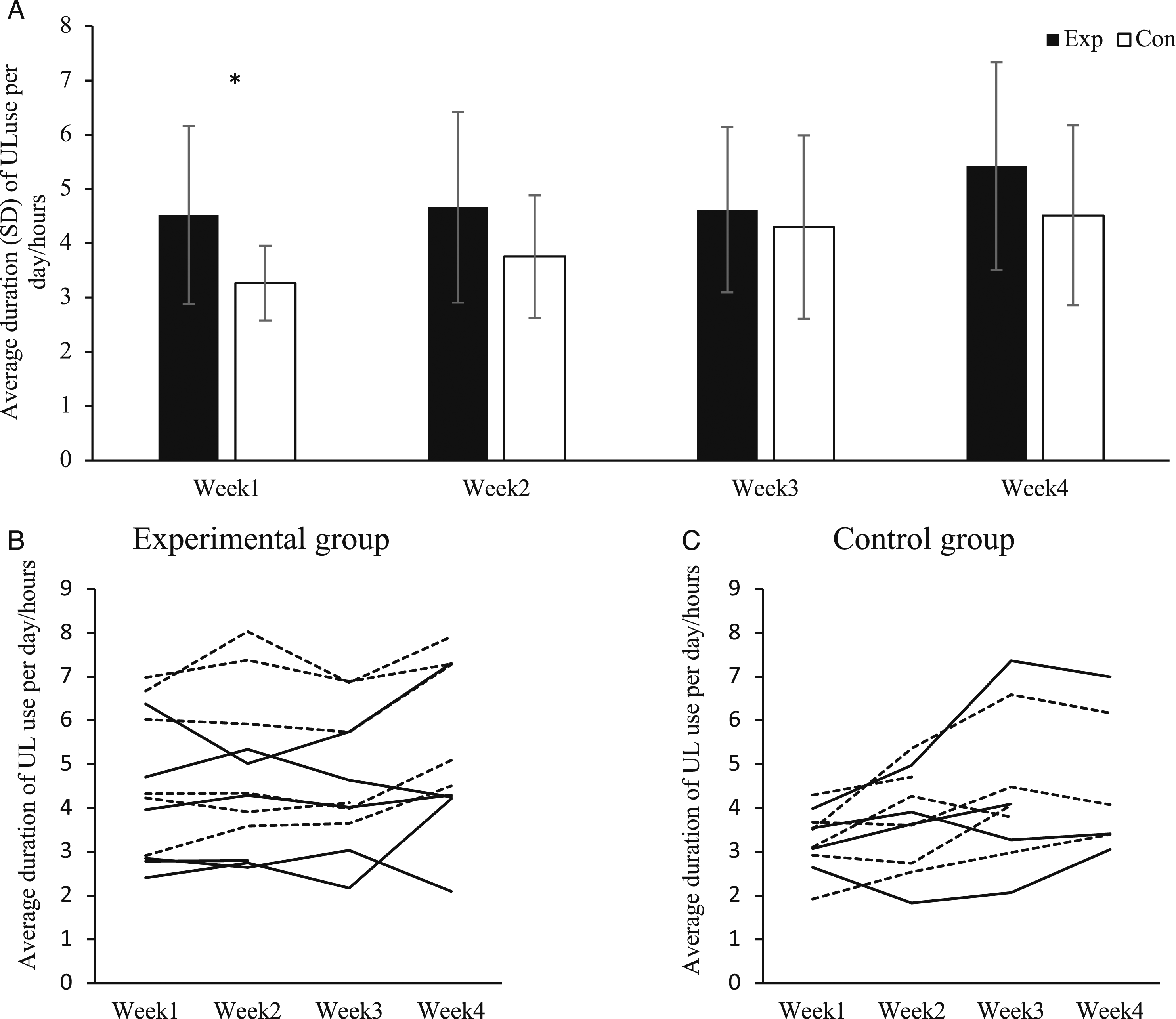
Potential Effect on Upper Limb Recovery
Participants from both groups demonstrated significant improvements in FMUL and ARAT from Week 0 to Week 4, Week 8, and Week 16 (P ≤ .002, Table 3, Figure 3). There were no significant improvements in FMUL and ARAT in either group from Week 8 to Week 16 (P ≥ .077, Table 3). There was no difference between groups in the improvement in FMUL and ARAT between all time points (P ≥ .119, Table 3). When scores were examined, by Week 8, 12 experimental participants (100%) and 10 control participants (91%) achieved an MCID on the FMUL test, and 10 experimental participants (83%) and 9 control participants (82%) achieved an MCID in the ARAT. Fifty-five percent of the experimental group and 82% of the control group achieved their expected proportional recovery. Graphs of UL recovery across Week 0 to Week 16: (A) Fugl Meyer UL score of the experimental group, (B) FMUL score of the control group, (C) ARAT score of the experiment group, and (D) ARAT score of the control group. Each line represents individual participant. Solid lines are participants with severe UL impairment, and dashed lines are participants with moderate UL impairment. Abbreviation: UL, upper limb; ARAT, Action Research Arm Test; FMUL, Fugl Meyer UL.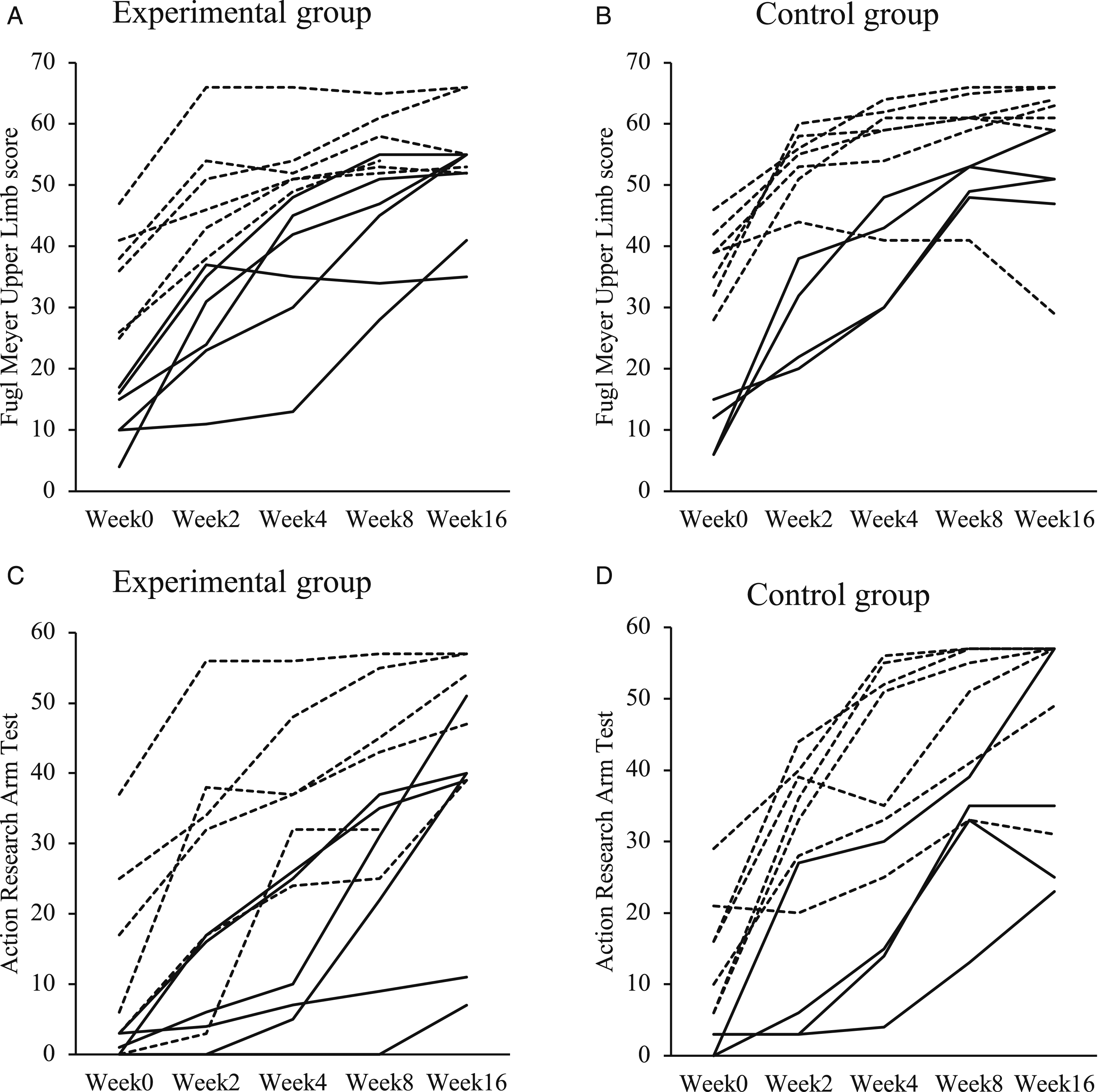
Discussion
Results of this study demonstrated that a self-directed UL program, incorporating UL exercise and promotion of UL use early during stroke inpatient rehabilitation, is feasible as shown by high adherence, no dropouts, and no adverse events related to the SURE program. Participants, caregivers, and nurses reported that participants experienced relative ease in carrying out the program and had a high level of satisfaction. Participants in the SURE program achieved >300 repetitions daily on top of usual care with no evidence of increased resistance to passive movement in paretic biceps and finger flexors, reports of UL pain, or fatigue when compared with controls. This demonstrates that people with stroke can perform a program involving exercise and functional use of the paretic UL safely outside therapy early post-stroke to achieve increased UL use.
Accelerometry results showed that experimental participants were able to achieve an additional hour of UL use per day by performing the SURE program when compared with the control group. Both groups demonstrated significant UL recovery until Week 8 (approximately 11 weeks post-stroke). This is consistent with other studies who propose that maximum recovery occurs during the first 2–3 months post-stroke, 5 suggesting a “sensitive” post-stroke period of spontaneous recovery and increased responsiveness to training. 51 However, the current study showed that increased use from the SURE program did not potentially effect UL recovery. It may be that use is still subthreshold compared to healthy older adults, 14 and a greater dose may be needed to effect recovery.
Our findings, which showed that a self-directed UL program during early rehabilitation is feasible, are similar to that reported on the GRASP intervention. 21 However, in contrast to the GRASP findings which showed that increased UL practice from GRASP improved UL outcomes, 21 our study did not. The most likely explanation is that our exploratory trial was not powered to show a change in recovery having only 23 participants, whereas the GRASP study was powered with 103 participants. An appropriately powered future study is warranted to investigate the efficacy of the SURE program. Another possible explanation is that our participants in both groups started with much lower FMUL scores compared with GRASP participants (baseline FMUL: our study 24–27 (SD14–15), GRASP study 40 (SD13–14)). Coupled with intact corticospinal excitability (MEP +), our study participants collectively had a greater potential to improve with time; hence, the increase in UL use in SURE might not be able to demonstrate a differential effect on recovery between our experimental and control participants.
Our study importantly compared the potential effect of increased UL use in MEP+ stroke survivors stratified based on UL impairment level. By using corticospinal tract excitability as an UL recovery biomarker, we were able to streamline stroke participants with similar UL recovery potential to allow for a more accurate evaluation of the potential effect of the SURE program on UL recovery. When stroke participants are recruited based only on clinical measures, it has the potential to mix individuals with poor UL recovery potential with those with good UL recovery potential and therefore reduce or washout the effect of the intervention. The non-significant effect of SURE on UL recovery may possibly be due to the proposal that people with MEP+ followed proportional recovery regardless of UL therapy dosage.29,34 Thus, an increased dosage in SURE program may not effect UL recovery in some people. Another possible contrasting hypothesis is that an addition of 1 hour daily UL use, with a total of 24 hours for the 4-week intervention may have been insufficient to push participants beyond proportional recovery expectations. Thus, the question of whether intensive UL therapy during early post-stroke would increase proportional impairment resolution above 70% still requires further investigation. 29 Further feasibility and investigative studies with higher UL dosage are required to answer this question.
Interestingly, even when there were no significant improvements in FMUL and ARAT in both groups after Week 8, the experimental group continued to show an increase in UL use. Even after the SURE program ended, the experimental group demonstrated a significant increase on the REACH scale which was not observed in the control group. Although the difference was not significant when comparing the two groups, this highlights that the SURE program might be able to enhance the ability of people with stroke to use their paretic UL during time outside therapy and beyond rehabilitation to drive their own recovery. This is important as stroke rehabilitation services are often limited to the first 3–6 months after stroke, sometimes less depending on geographical location. Therefore, it is vital for people with stroke to be empowered to drive their own recovery beyond rehabilitation to achieve better outcomes. 52 The SURE program provides the education to empower the people with stroke and together with regular reviews by the trainer guides, monitors, and encourages paretic UL use. A possible reason for the increased UL use after Week 8 in the experimental group might be due to the increased confidence in ADL performance from their participation in the SURE program as increased self-efficacy in ADL performance has been found to be associated with increased UL use after stroke. 26 Further investigation is warranted to confirm the empowerment effect of the SURE program and identify the component responsible for enhancing the ability of people with stroke to use their paretic UL outside therapy and beyond rehabilitation.
Study Limitations
It is acknowledged that our study is not without limitations. First, our small sample size means our study was not powered to detect a change in UL recovery by the SURE program. Hence, there is a limited generalizability of our study results. Nevertheless, the results of our study can be used to estimate the sample size required for a larger randomized SURE program trial. The results of our study can also be used to guide the design of a dose-response trial to determine the optimal dosage for the SURE program to have an efficacy on UL recovery in MEP+ stroke survivors. Second, a potential source of bias existed as participants were not blinded to the intent of the study. In general, the controls in our study showed interest in using their paretic UL by asking how to increase their paretic UL even though they were in the control group; thus, they might have increased UL use on their own during the intervention period even without the SURE program. This is commonly observed in stroke rehabilitation trials 53 and warrants consideration in future trials to ensure sufficient contrast between the experimental and control groups is maintained. The possibility of intervention leak in this study was low for several reasons. First, 70% of our participants wore a fall risk tag due to their high fall risk and were only allowed to ambulate with nurses’ supervision or assistance. Hence, most participants were in bed or seated next to their bed most of the time outside therapy. In addition, recruitment was spread across for more than 1 year resulting in only a few participants present at one time in a 90 bedded rehabilitation center. Another possible source of increased UL use in the controls could have been the wearing of accelerometers and the weekly feedback on amount of UL use given to all participants. The attention given by the trainer to both groups might also have resulted in potential increased UL use in the controls. However, these potential sources of increased UL use in the control group were also applicable to the experimental group, thus resulting in similar bias of increased UL use to both groups.
Conclusions
This study demonstrated that SURE is a feasible self-directed UL program of exercise and functional activities performed outside therapy that can potentially increase UL use by approximately 1 hour per day when compared with the control group during early rehabilitation post-stroke in people who are MEP+. Further appropriately powered studies are required to investigate the efficacy of the SURE program on people with stroke. The effect of SURE in empowering and enhancing the ability of people with stroke to use their paretic UL to drive their own recovery outside therapy and beyond rehabilitation is of clinical interest and should warrant further investigation.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211032967 – Supplemental Material for A Self-Empowered Upper Limb Repetitive Engagement Program to Improve Upper Limb Recovery Early Post-Stroke: Phase II Pilot Randomized Controlled Trial
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683211032967 for A Self-Empowered Upper Limb Repetitive Engagement Program to Improve Upper Limb Recovery Early Post-Stroke: Phase II Pilot Randomized Controlled Trial by Lay Fong Chin, Kathryn S. Hayward, Audrey Lik Ming Chai and Sandra G. Brauer in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgments
We thank the patients, nursing, and therapy staff of Tan Tock Seng Hospital Rehabilitation Centre. We thank Keng He Kong (Senior Consultant, Tan Tock Seng Hospital Rehabilitation Centre) for his advice on the research design and Chloe Teng Sok Ching (Senior Physiotherapist, Tan Tock Seng Hospital Rehabilitation Centre) and Yan Ming Soh (Principal Occupational Therapist, Tan Tock Seng Hospital Rehabilitation Centre) for their assistance in data collection. We thank Asad Khan (Associate Professor in Biostatistics, School of Health and Rehabilitation Sciences, The University of Queensland) for his advice on statistics and Dion Scott (Principal Scientific Officer, School of Health and Rehabilitation Sciences, The University of Queensland) for assisting in the development of the accelerometry processing scripts.
Authors’ Note
Clinical trial registration: ClinicalTrials.gov_NCT03425890.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Rehabilitation Research Institute of Singapore Rehabilitation Research Grant (grant number RRG1/16015). LFC is supported by National Medical Research Council Singapore (MH 95:03/8-1031). KSH was supported by a National Health and Medical Research Council Early Career Fellowship (1088449). The Florey Institute of Neuroscience and Mental Health acknowledges the strong support from the Victorian Government and in particular the funding from the Operational Infrastructure Support Grant.
Trial Registration
ClinicalTrials.gov_NCT03425890.
Supplementary Material
Supplementary material for this article is available online on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
