Abstract
Background. Poststroke individuals use their paretic arms less often than expected in daily life situations, even when motor recovery is scored highly in clinical tests. Real-world environments are often unpredictable and require the ability to multitask and make decisions about rapid and accurate arm movement adjustments. Objective. To identify whether and to what extent cognitive–motor deficits in well-recovered individuals with stroke affect the ability to rapidly adapt reaching movements in changing cognitive and environmental conditions. Methods. Thirteen individuals with mild stroke and 11 healthy controls performed an obstacle avoidance task in a virtual environment while standing. Subjects reached for a virtual juice bottle with their hemiparetic arm as quickly as possible under single- and dual-task conditions. In the single-task condition, a sliding glass door partially obstructed the reaching path of the paretic arm. A successful trial was counted when the subject touched the bottle without the hand colliding with the door. In the dual-task condition, subjects repeated the same task while performing an auditory–verbal working memory task. Results. Individuals with stroke had significantly lower success rates than controls in avoiding the moving door in single-task (stroke: 51.8 ± 21.2%, control: 70.6 ± 12.7%; P = .018) and dual-task conditions (stroke: 40.0 ± 27.6%, control: 65.3 ± 20.0%; P = .015). Endpoint speed was lower in stroke subjects for successful trials in both conditions. Obstacle avoidance deficits were exacerbated by increased cognitive demands in both groups. Individuals reporting greater confidence using their hemiparetic arm had higher success rates. Conclusion. Clinically well-recovered individuals with stroke may have persistent deficits performing a complex reaching task.
Introduction
Upper limb (UL) motor deficits following stroke affect a large proportion of acute stroke survivors with only a limited number regaining effective functional arm use. 1 There is a strong relationship between UL function and the ability to perform activities of daily living (ADL), with the caveat that even if motor recovery is scored highly on clinical tests, use of the paretic arm in daily life situations is diminished.2-4 The discrepancy between clinical measures of motor and real-world functional arm use after stroke may arise from deficits in the ability to adapt movement in unpredictable real-world environments. Sudden changes in the environment often require rapid and accurate motor adaptations to interact effectively with objects. In a previous study, well-recovered individuals with stroke were less successful than healthy age-matched controls in making online corrective endpoint trajectories to avoid an obstacle in the reaching path when seated. 5 This was due to delayed initiation of corrective movements in response to sudden obstacle appearance. Several stroke-related and sensorimotor impairments, including spasticity, sensory deficits, abnormal muscle recruitment patterns, decreased range of motion, and difficulty dealing with interjoint interaction torques affect UL movement production and adaptation.6-10 A decrease in overall motor abundance (i.e., ability to combine joint movements in different ways to achieve the same motor goal—termed motor equivalence) 11 is reflected in the altered adaptability of UL movement.5,12 Adaptability also depends on mental flexibility or the ability to change the movement plan according to task demands.13,14 Cognitive processes such as attention, information processing speed, and executive function (planning, task flexibility, reasoning, decision-making, and problem-solving), crucial for online movement correction, are often impaired after stroke.15-18
In addition to the ability to react to unexpected situations, real-world environments often require doing more than one task at a time (i.e., dual-tasking). 19 Competing attentional demands when dual-tasking may lead to performance decrements, especially when the attentional demand is high or attentional capacity is reduced. As poststroke individuals often have impaired cognition (including reduced attention), 19 dual-tasking may be diminished, such as that reported for gait and balance activities19-21 and pointing during standing. 12 For example, locomotor parameters were affected when a cognitive task (CT) was performed while walking. 22 Performing a CT in standing was associated with a decrease in anteroposterior and mediolateral sway and increased weight-bearing asymmetry. 22 To date, research in healthy subjects and neurological patients has focused on the effects of adding a CT during motor tasks such as walking or maintaining balance. Despite its potential importance for ADLs, there is relatively less information about dual-task effects on UL motor activities, which are presumably more cognitively driven and less automatic than gross motor activities such as walking.23,24 For example, Bank et al. 25 investigated the influence of on an auditory Stroop task on goal-directed UL movements. Healthy and stroke subjects controlled a virtual mouse to collect virtual pieces of cheese as fast as possible while avoiding a virtual cat (obstacle). Stroke subjects had greater difficulty avoiding the obstacle, were less successful, and moved slower than controls, suggesting differences between groups in capacity-sharing patterns. However, the study was limited by the heterogeneity of the stroke group, variability in dual-task performance and lack of kinematic outcomes.
While arm use may be limited by motor deficits in patients with greater stroke severity, well-recovered patients may have difficulties that remain undetected by conventional clinical tests. To optimally challenge cognitive–motor functions in well-recovered poststroke individuals, we used a reaching task requiring critical timing to avoid hitting a moving obstacle. To more accurately reflect everyday environments, we added a simple working memory task. Being able to detect complex arm motor function deficits and cognitive–motor interactions will allow clinicians and researchers to design rehabilitation programs that have the potential for greater carryover to everyday activities.
We sought to identify whether and to what extent motor and cognitive demands in well-recovered people with stroke affect the ability to avoid obstacles when reaching with the more-affected arm during standing. Since well-recovered patients with stroke may have subtle motor and cognitive deficits and motor problem-solving is involved in error correction, we hypothesized that (1) individuals with stroke would have greater difficulty in avoiding a moving obstacle during reaching while standing compared to healthy age-matched controls; (2) error correction ability would be more diminished in people with stroke than healthy individuals when cognitive demands are increased (i.e., dual-tasking); and (3) decreased success in avoiding obstacles would be related to lower clinical scores, patient-reported arm use, and self-efficacy. Preliminary results appeared in the abstract form. 26
Methods
Subjects
Demographic Characteristics and Clinical Scores.
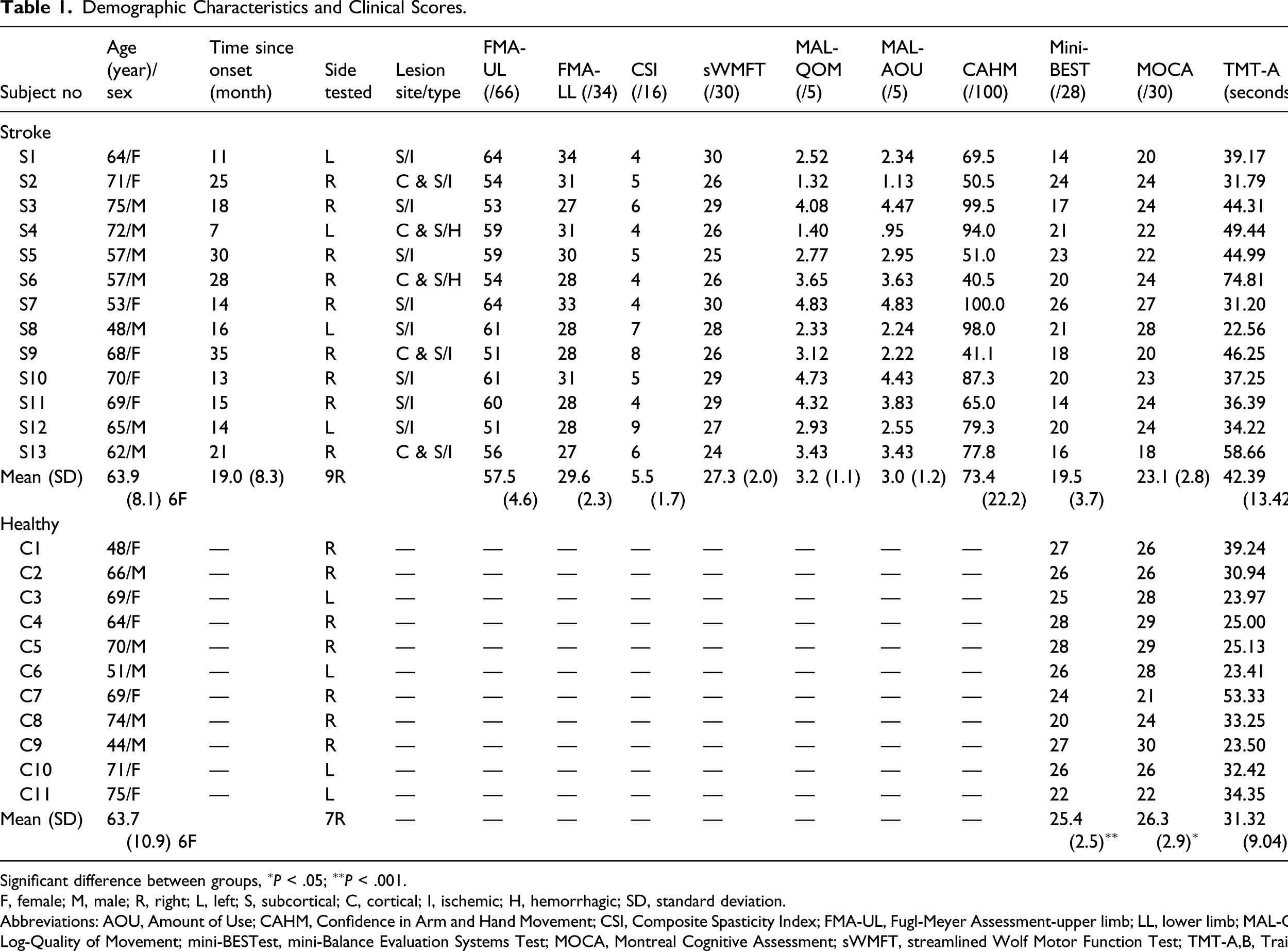
Significant difference between groups, ∗P < .05; ∗∗P < .001.
F, female; M, male; R, right; L, left; S, subcortical; C, cortical; I, ischemic; H, hemorrhagic; SD, standard deviation.
Abbreviations: AOU, Amount of Use; CAHM, Confidence in Arm and Hand Movement; CSI, Composite Spasticity Index; FMA-UL, Fugl-Meyer Assessment-upper limb; LL, lower limb; MAL-QOM, Motor Activity Log-Quality of Movement; mini-BESTest, mini-Balance Evaluation Systems Test; MOCA, Montreal Cognitive Assessment; sWMFT, streamlined Wolf Motor Function Test; TMT-A,B, Trail Making Test.
Clinical Evaluation
For the stroke group, an experienced physiotherapist evaluated impairments, activity limitations, and participation restrictions according to the International Classification of Functioning. 29 Sensorimotor impairment was quantified using the FMA-UL and lower limb assessments (FMA-LL; maximum score = 34), evaluating reflexes, voluntary movements, and coordination. 27 Elbow flexor spasticity was evaluated with the Composite Spasticity Index (CSI) 30 that assessed (a) resistance felt by the examiner to moderate-speed elbow flexor stretch, (b) biceps brachii tendon reflex excitability, and (c) wrist clonus in which scores of 0–9, 10–12, and 13–16 represent mild, moderate, and severe spasticity, respectively. Arm function was measured by the streamlined Wolf Motor Function Test (sWMFT) 31 that evaluated 6 UL functional tasks on a 30-point scale. Arm use in everyday activities (Motor Activity Log, MAL) 32 and self-efficacy (Confidence in Arm and Hand Movement scale, CAHM)33,34 were also quantified. MAL is a structured interview in which subjects rate how well (Quality of Movement, MAL-QOM) and how much (Amount of Use, MAL-AOU) they used the impaired arm in 28 UL activities during the preceding week on 5-point scales. The final score is the sum of all subscale scores (i.e., MAL-QOM or MAL-AOU) divided by the number of activities performed. The CAHM is a 20-item questionnaire in which individuals rate their confidence in performing a series of functional tasks involving the weaker arm or both arms in social, home, and community contexts. Items are scored from 0 (very uncertain) to 100 (very certain) and averaged.
In both groups, balance was evaluated with the mini-Balance Evaluation Systems Test (mini-BESTest). 35 The test includes 14 items addressing 4 dynamic balance domains (anticipatory postural adjustments, reactive postural control, sensory orientation, and dynamic gait) and is scored on ordinal scales (0–2) for a total score of 28-points per test direction. The Montreal Cognitive Assessment (MoCA) 36 measured overall cognition. Attention, information processing speed, and mental flexibility were assessed with Trail Making Tests (TMTs) A and B. 37
Experimental Setup
Subjects performed a motor task, a CT, and a dual-task, wherein the motor and CTs were performed simultaneously during a ∼2-hour experiment.
Motor Task
A reaching task was performed in a 3D virtual environment (VE) viewed through a head-mounted display (HMD; Kaiser Electro-Optics, ProView). Patients with stroke reached with their more-affected arm, whereas controls reached with their dominant or nondominant arm corresponding to the paired stroke subject. Arm and trunk movements were recorded and integrated into the VE in real time. Wearing regular footwear, subjects stood with their feet in their preferred mediolateral stance width. Foot position was marked on the floor and maintained throughout the session. In the initial position, the tested arm was alongside the body, the index fingertip rested on a lateral thigh support, the elbow was in 30° flexion, and the forearm and hand were in neutral between supination and pronation. The non-tested arm hung by the side.
The 3D-VE (CAREN2, Motek, BV, Amsterdam) consisted of a grocery store aisle with shelves located behind double sliding doors. Subjects controlled a forearm and hand avatar to reach a 5 cm diameter juice bottle on a shelf behind the door (Figures 1A, 1B, and 1D), located at a distance of 90% arm length in the midsagittal plane aligned with the sternum. Aside from other visual cues, 2 flanking objects (cans) enhanced the 3D perspective. The virtual scene was created to be ecologically valid by ensuring that the avatar was perceived as an extension of the subject’s own body with the bottle within perceived reaching distance.
38
Experimental setup. (A) Standing subject viewed a 3D virtual environment of a grocery aisle with sliding glass doors through a head-mounted display. Target object was a juice bottle flanked by 2 cans. (B) Arm and sliding door positions for an unobstructed reach. (C) Arm position when reaching path was obstructed by a glass door sliding from the right side. (D) Example of unobstructed trajectory of the right arm. (E) Example of a trajectory of the right arm obstructed by the door sliding across the scene from the right side at the level of the arrow.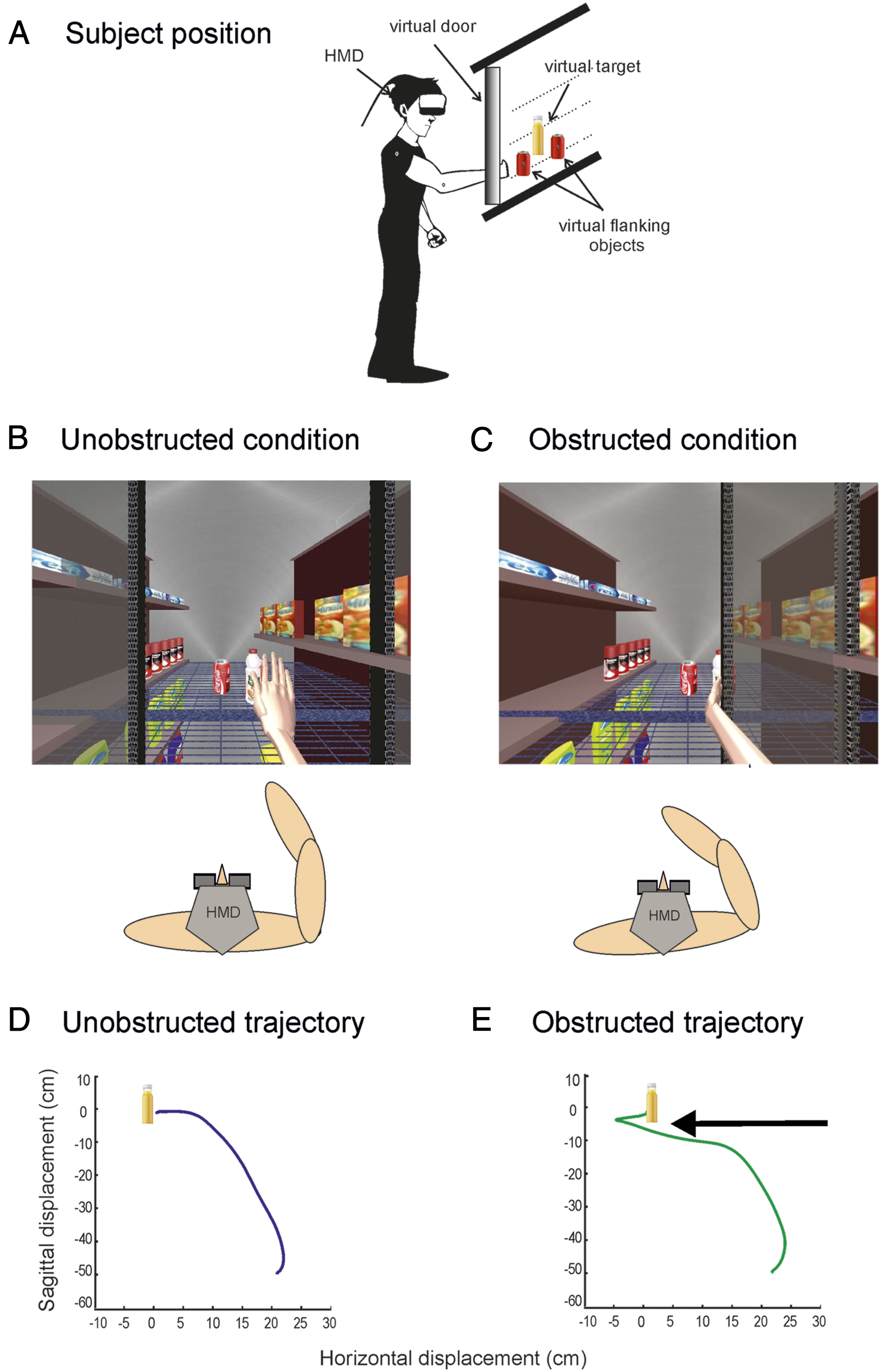
In 66% of random trials, the reaching path was obstructed by a transparent door that slid across the scene in front of the shelf from the left or right side (Figures 1C and 1E). When completely closed, the door obstructed 30% of the bottle. Door closure was timed so that corrective movements could only occur after the end of the initial arm movement to minimize online corrections. Door closure timing was based on findings that the shift in the motor command for reaching is completed by the time of endpoint peak tangential velocity. 39 Door closure was triggered at 10% of the subject’s mean arm peak tangential velocity so that the door completely closed by the time the arm reached peak tangential velocity.
Prior to recording, subjects completed 3 familiarization trial blocks. In the first block, at a computer-generated GO signal, subjects reached and grasped the bottle as fast and as accurately as possible without leaning/bending their trunk and returned the arm to the initial position (U trials). If the bottle was grasped within 5 seconds, a “ping” sound signaled success. The next 2 familiarization blocks consisted of practice of right/left door obstructed trials (RD/LD trials), until 10 consecutive successful reaches in each of the 3 familiarization blocks (n = 10–30 trials/block) were made. In RD/LD trials, a successful trial (S) was defined as one in which the subject could reach and grasp the bottle within 5 seconds without hitting the door with any part of their hand or arm. A failed (F) trial was signaled by the sound of cracking glass.
Cognitive Single Task
Prior to recording, subjects performed a 1-back CT testing auditory–verbal working memory. 40 Subjects sat in a quiet room and listened to a sequence of 4 random numbers between 1000 and 9999 presented consecutively and then repeated the third number in the sequence. The task was performed for 5 minutes and was audiotaped. Responses were recorded on paper for offline verification.
Experimental Procedure
Four experimental blocks were recorded, consisting of (1) unobstructed reaching (Block U and 15 trials), (2) randomly obstructed reaching (randomized single-task, Block R-ST, and 60 trials), (3) unobstructed reaching while performing the CT (unobstructed dual-task, Block U-DT, and 15 trials), and (4) randomly obstructed reaching while performing the CT (randomized dual-task, Block R-DT, and 60 trials). Blocks R-ST and R-DT were divided into two 30-trial blocks to avoid fatigue. Blocks included an equal number of the 3 trial types (U/RD/LD). A computer-generated random sequence was used for each block to minimize anticipation of the upcoming trial type. Blocks R-ST and R-DT were randomized across subjects. Subjects rested for 2–5 minutes between blocks to avoid fatigue.
Data Acquisition
Trunk and arm movements were recorded from 18 infrared-emitting markers for 5 seconds at 120 Hz with a 2-camera bar Optotrak Certus motion analysis system (Northern Digital; Waterloo, ON). Rigid bodies consisting of groups of non-coaxial markers were placed on the dorsal hand (6 markers) and mid-forearm (6 markers). Six individual markers were also placed on the mid-sternum, ipsilateral and contralateral acromia, lateral epicondyle, mid-dorsal wrist crease (endpoint), and third fingertip.
Data Analysis
Data were interpolated (third-order spline function) and filtered with a low-pass 10 Hz Butterworth filter. Endpoint tangential velocity was computed from wrist marker x, y, and z positional data. Movement initiation/end was defined as times at which tangential velocity rose/fell and remained above/below 10% of the mean peak tangential velocity for at least 50 ms calculated from Block U. 41
The endpoint trajectory during reaching was characterized by peak velocity, trajectory length, curvature, and smoothness. Endpoint peak velocity (mm/s) and time to peak velocity (TPV) values were computed from the wrist x, y, and z tangential velocity. Trajectory length was defined as the distance (mm) moved by the endpoint from movement initiation to end. Trajectory straightness was evaluated with the index of curvature (IC) defined as the ratio of total endpoint displacement to the shortest distance between initial and final positions. IC = 1 and IC = 1.57 denote a straight and semicircular path, respectively (π/2). 41 Trajectory smoothness was evaluated by the number of movement units (NMUs) defined as the number of peaks in the endpoint tangential velocity profile between movement initiation and end, where fewer NMUs indicate smoother reaching.
Arm movements were defined for the shoulder (flexion/extension and horizontal abduction/adduction), elbow (flexion/extension), wrist (flexion/extension and abduction/adduction), and trunk (pitch/roll/yaw). Shoulder flexion/extension angle was computed between vectors formed by the acromial and elbow markers with respect to the trunk position where the arm alongside the body was 0°. For shoulder horizontal abduction/adduction, 0° was defined as the horizontal projection of the upper arm vector on the horizontal plane. Elbow angle was defined by vectors formed between acromial, elbow, and wrist markers where a fully extended arm equaled 180°. Wrist movement was defined by vectors formed between the elbow, wrist, and fingertip markers. Three-dimensional reconstruction of trunk rotation (pitch/roll/yaw) was done using data from the sternal and acromial markers, where the initial position was 0°.
To determine where the trajectory of obstructed trials diverged from template unobstructed trials, a divergence point (DP) was calculated using endpoint tangential velocity/position phase profiles for each trial type (success/fail). Successful and failed phase profiles were averaged and superimposed on the mean template phase profile (U). The DP at which phase profile deviated from the template was identified by running a t-test (P < .05) as the first significant difference between the profiles that was maintained for at least 10 points. The DP was expressed as the absolute distance from the starting position. We also determined the endpoint speed at the time of divergence and expressed this as a percent of the maximal reach velocity of the U trial.
The motor task percent success rate was calculated for each condition as the number of successfully avoided ipsilaterally obstructed trials divided by the total number of trials in Block R-ST or R-DT, multiplied by 100. Similarly, the CT pxercent success rate was calculated as the number of correct responses to the total number of stimuli on the CT. The extent of change caused by the dual-task relative to single-task condition was calculated for primary outcomes and endpoint kinematics in U and successful trials as: dual-task cost = (DT − ST)/ST × 100, where a negative value indicates decreased task performance in the dual-task (DT) compared to the single-task (ST) condition.
Statistical Analysis
The primary outcome measures were success rate and DP in Block R-ST (Hypothesis 1), change in success rate and DP in Block R-DT compared to Block R-ST (Hypothesis 2), and correlation between success rates in Blocks R-ST/R-DT and clinical scores (Hypothesis 3). Secondary outcomes were changes in endpoint and joint kinematics in ST and DT conditions. Statistical analyses were performed with SPSS 23.0 (IBM Corp., Armonk, NY). Data normality was verified with Levene’s tests and sphericity with Mauchly’s test. Greenhouse–Geisser corrections were used when indicated. Initial significance levels of P < .05 were used for all tests, with Bonferroni corrections for multiple comparisons. For Hypotheses 1 and 2, the mean success rates and DP times between groups (control/stroke) were compared with Student t-tests.
DPs were compared between groups and trial types (success/fail and single-/dual-task) using 2-way repeated-measures ANOVAs. All other kinematics were compared between groups and trial types using repeated-measures ANOVAs with one between-subject factor (group: stroke/control) and one within-subject factor (condition: U/Success/Fail). Dual-task cost was analyzed with one between-subject factor (group: stroke/control) and one within-subject factor (condition: U/Success). Effect sizes are reported as Cohen’s d. 42 For Hypothesis 3, relationships between task success, arm use, and self-efficacy were determined with 2-tailed Pearson/Spearman tests. Student t-tests/Mann–Whitney U tests were used to compare baseline characteristics between groups and motor task success rates and CT error rates during the first 50% of trials and the second 50% of trials in both groups to confirm the absence of learning and fatigue effects.
Results
Compared to control subjects, stroke subjects had lower functional balance (mini-BEST, P < .001) and cognition scores (MOCA, P = .012; TMT-A, P = .03; TMT-B, U = 37.0, P = .047). All subjects could perform the obstacle avoidance task with and without dual-tasking with no evidence of learning or fatigue.
ST Condition
Primary Outcome Measures for all Subjects for the ST and DT Conditions. Bold Font Indicates Decreased Performance in DT Condition. Gray Font Indicates Improved Performance in DT Condition.
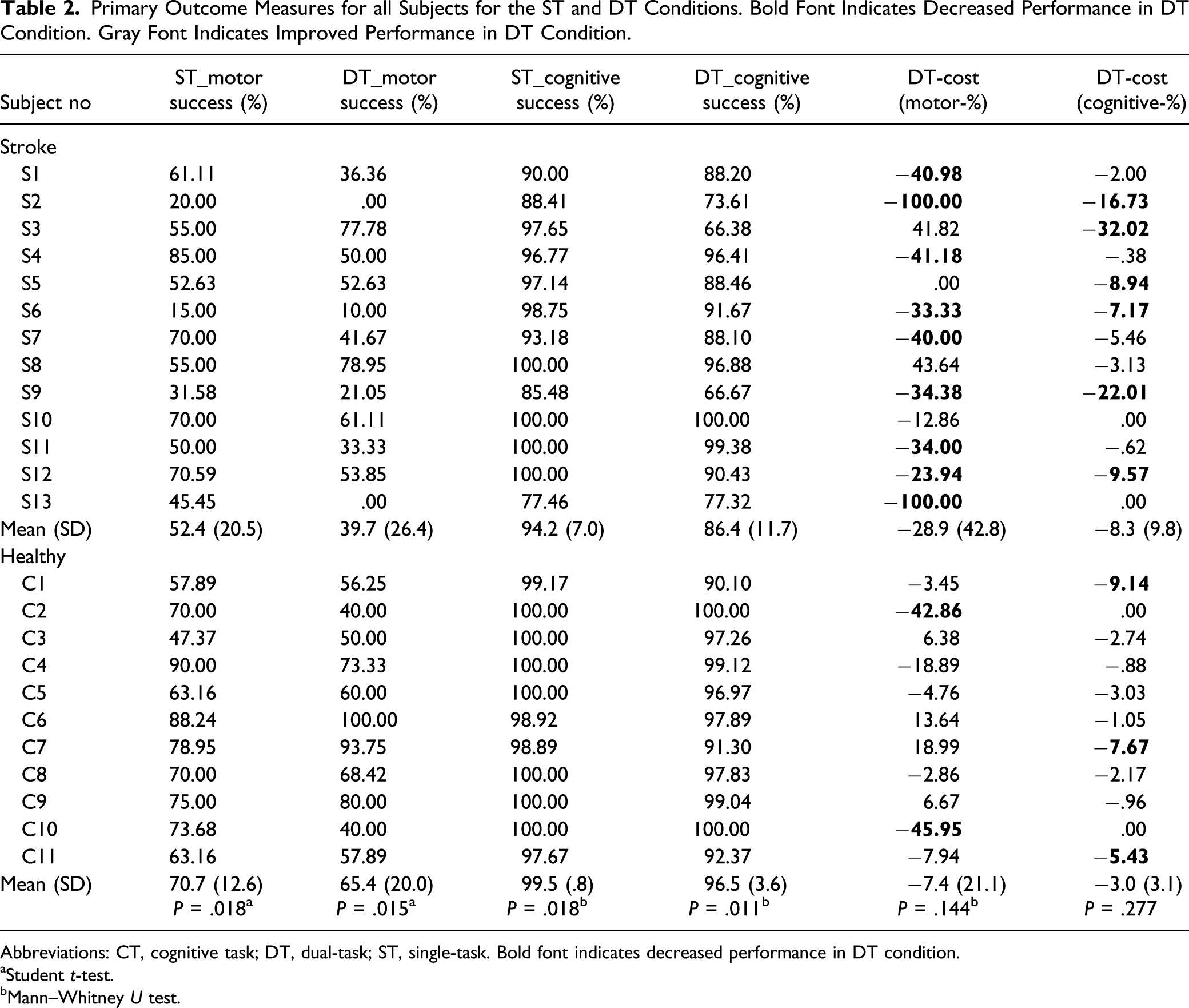
Abbreviations: CT, cognitive task; DT, dual-task; ST, single-task. Bold font indicates decreased performance in DT condition.
aStudent t-test.
bMann–Whitney U test.
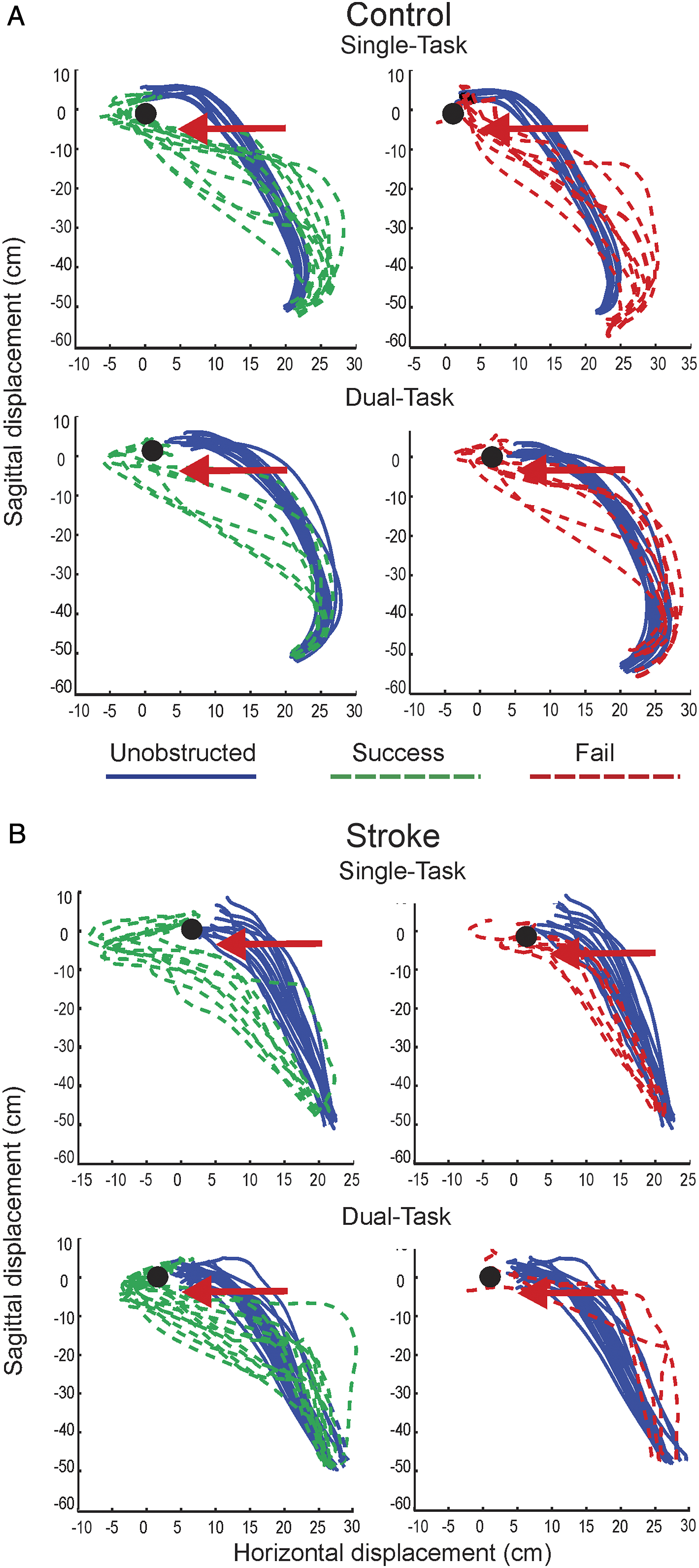
Examples of endpoint trajectories of the right arm in a control subject (C3) (A) and a stroke subject (S4) (B) showing unobstructed (blue/solid traces) and obstructed (green and red/dashed traces) reaching trials to a bottle (black dots) for single-task (top panels) and dual-task (bottom panels) reaches. The movement path and position of the obstruction (door) is shown by horizontal arrows in each panel. Successful trials are shown in the left panels and failed trials are shown in the right panels (top and bottom). Unobstructed reaches had consistent endpoint trajectory shapes in single- and dual-task conditions. Obstructed reaches had greater and earlier leftward deviation in both conditions in successful trials.
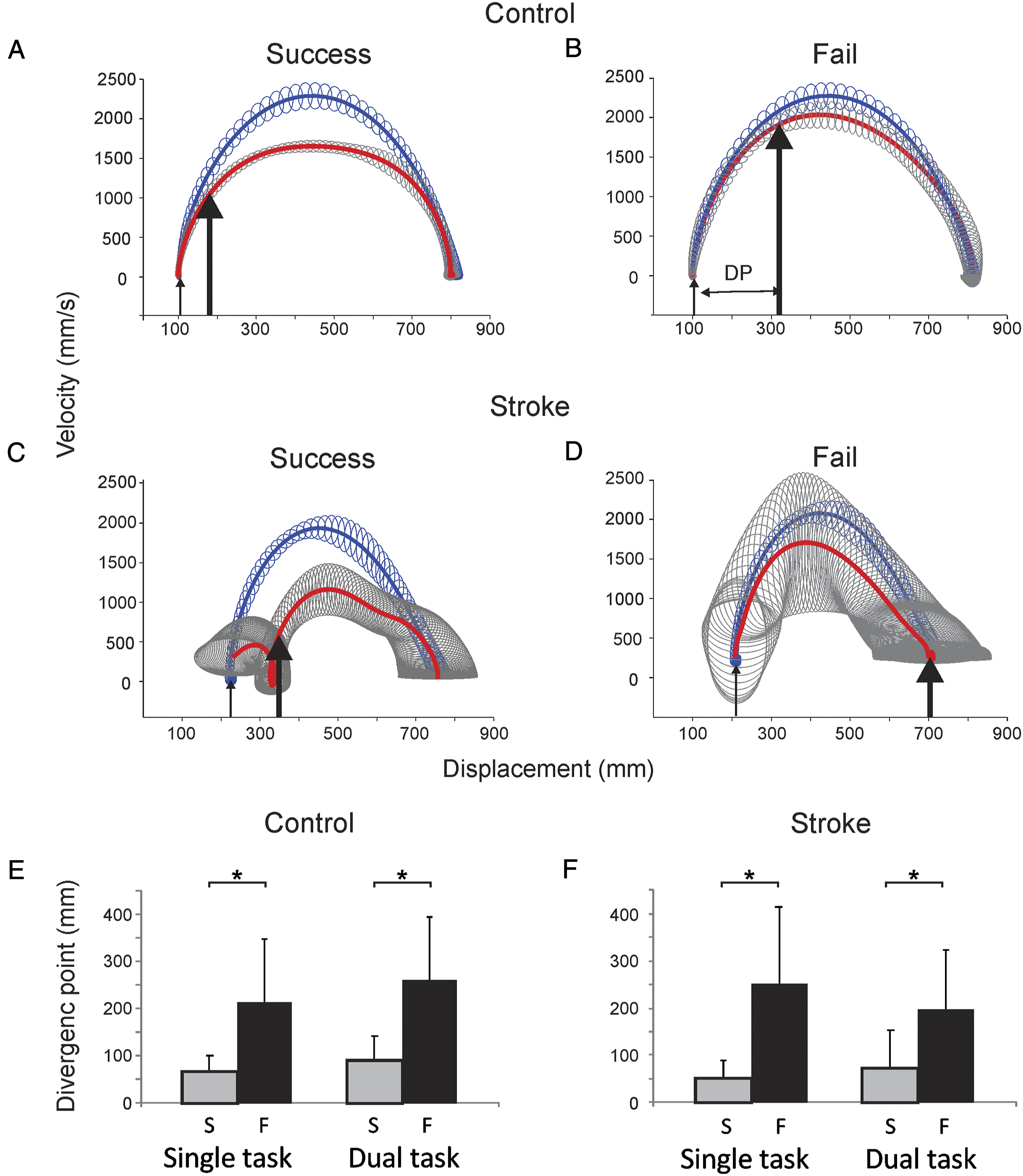
DP in representative control (A, B) and stroke subjects (C, D) for single-task reaches. Mean phase profiles are shown for unobstructed reaches (blue/upper traces) and successful (A, C) and failed (B, D) obstructed reaches (red/lower traces) with associated standard deviations (unobstructed-blue and obstructed-gray). Thin arrows indicate start position. Thick arrows indicate the point of the first significant difference between successful/failed reach and unobstructed reach phase profiles. The DP is expressed as the distance between the thin and thick arrows. (E, F) mean (±SD) of DPs of control and stroke subjects for all conditions. Endpoint trajectory paths diverged later in failed trials in stroke and control subjects in both conditions. Abbreviation: DP, divergence point.
Comparison of Mean (SD) Endpoint and Joint Kinematic Data Across Conditions and Between Groups.
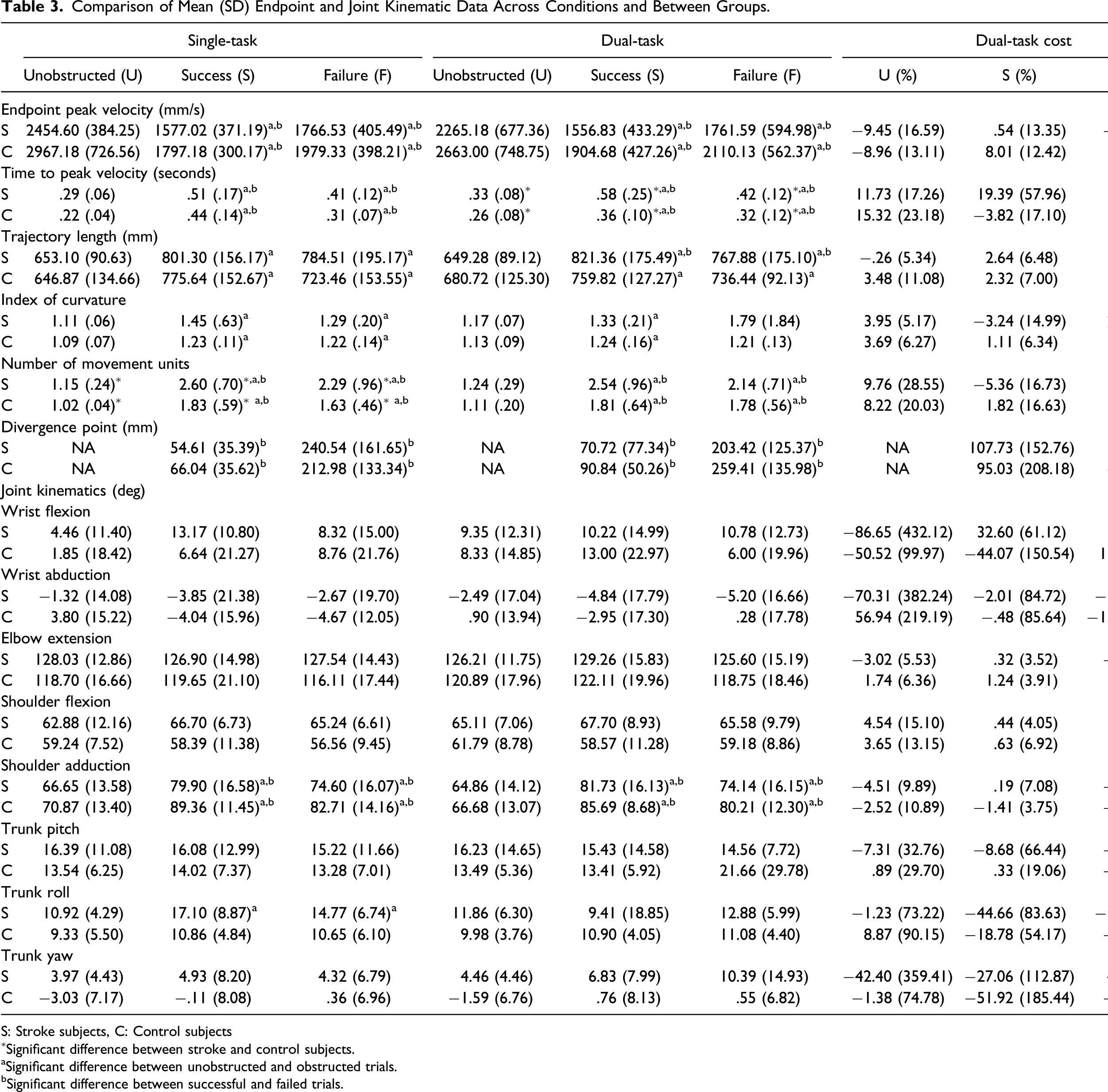
S: Stroke subjects, C: Control subjects
∗Significant difference between stroke and control subjects.
aSignificant difference between unobstructed and obstructed trials.
bSignificant difference between successful and failed trials.
Reaching peak velocities decreased in both groups when avoiding obstacles in successful trials (F2,21 = 80.21, P < .001, and d = 3.83; stroke: by ∼35.8%; controls: by ∼39.4%; Table 3) and in unsuccessful trials (F2,21 = 3.64, P = .07, and d = .81; stroke: by ∼28.0%; controls: by ∼33.3%). Both groups took longer to reach peak endpoint velocity in obstructed compared to unobstructed trials (F2,21 = 16.75, P < .001, d = 1.75) and in successful compared to failed trials (F1,22 = 31.32, P < .001, and d = 2.39). Endpoint trajectories were also longer (F2,21 = 9.75, P = .001, and d = 1.34) and more curved in obstructed compared to unobstructed trials (F1.18,21 = 7.26, P =.009, and d = 1.15). Endpoint trajectories in obstructed reaches were less smooth than those in unobstructed trials, and successful trials had more NMUs than unsuccessful trials in both groups (F2,21 = 37.74, P < .001, and d = 2.63). However, at the time of the endpoint divergence (DP) in successfully avoided trials, endpoint speed compared to the peak velocity of unobstructed trials was reduced more in the stroke group (controls: 51.8 ± 14.4%; stroke: 39.6 ± 12.9%, t = 2.034, and p = .05) and stroke subjects had more NMUs than controls in all conditions (F1,22 = 7.13, P = .01, and d = 1.14), which varied by condition (F2,44 = 3.70, P = .03, and d = .79).
Joint kinematics (Table 3) did not differ between groups or conditions except for shoulder adduction and trunk roll. Shoulder adduction was greater in obstacle avoidance trials compared to unobstructed trials in both groups (F1.54,33.86 = 28.71, P < .001, and d = 1.59) and greater in successful compared to failed trials (F1,22 = 12.91, P = .002, and d = 1.54). All subjects used increased trunk roll in obstructed compared to unobstructed trials (F2,22 = 12.35, P = .002, and d = 1.50). The amount of trunk roll was greater in stroke subjects compared to controls (group × condition – F2,21 = 3.39, P = .043, and d = .78).
DT Condition
Similar to ST, the stroke group was less successful avoiding the door than controls (DT success; P = .015, and d = 1.08; Table 2). Two subjects with stroke (S2 and S13) had no success in the DT condition and 2 (S3 and S8) were more successful in DT compared to ST (improved by >40%). All controls succeeded in at least 40% of DT trials. The stroke group made more errors on the CT condition than controls (DT–CT success: U = 28.0, P = .011, and d = 1.12), while the average DT cost did not differ between groups. Nine subjects with stroke had >20% reduction in performance in DT compared to 3 controls. In both groups, success was characterized by earlier trajectory divergence, DP (F1,17 = 24.50, P < .001, and d = 2.21).
Both groups maintained their unobstructed reaching peak velocity in the DT condition using similar kinematic patterns for reaching and obstacle avoidance as in the ST condition, that is, all subjects made slower movements to avoid obstacles (F2,18= 29.81, P < .001, and d = 2.44) and had longer times to peak arm velocity in obstructed compared to unobstructed trials (F2,18 = 20.38, P < .001, and d = 2.02). The TPV was longer in successful compared to failed trials (F1,19 = 5.88, P = .025, and d = 1.08). However, stroke subjects took longer than controls to reach peak velocity in successful trials (F1,19 = 8.47, P = .009, and d = 1.30) and had a significantly lower endpoint velocity at the time of trajectory divergence (DP, controls: 58.9 ± 17.6%; stroke: 35.9 ± 13.2%; t = 2.96, P = .01).
Endpoint trajectories were longer (F2,18 = 10.91, P = .001, and d = 1.48) and more curved in obstructed compared to unobstructed trials (F2,18 = 6.14, P = .008, and d = 1.11; Figure 2). Unobstructed trajectories were smoother than those in obstructed trials (F1,19 = 42.32, P < .001, and d = 2.78) and successful trials had more NMUs than unsuccessful trials (F1,19 = 7.6, P = .013, and d = 1.18). Joint kinematics did not differ between conditions or groups except for shoulder adduction that was greater in obstructed compared to unobstructed trials in both groups (F1,19 = 54.44, P < .001, and d = 3.22) and greater in successful compared to failed trials (F1,19 = 32.31, P < .001, and d = 2.48). Dual-task costs were similar for both groups (Table 2).
Relationship Between Motor Task Success and Clinical Measures
In the stroke group, task success in both conditions was related to CAHM scores (ST: r = .770 and P = .002; Figure 4A; DT: r = .669 and P = .012; Figure 4B) but not to arm motor impairment or other clinical measures. DT success was correlated with cognition (TMT-B time, r = −.624, and P = .023). Correlation between confidence in arm and hand movement scores and motor task success scores in (A) single-task and (B) dual-task conditions.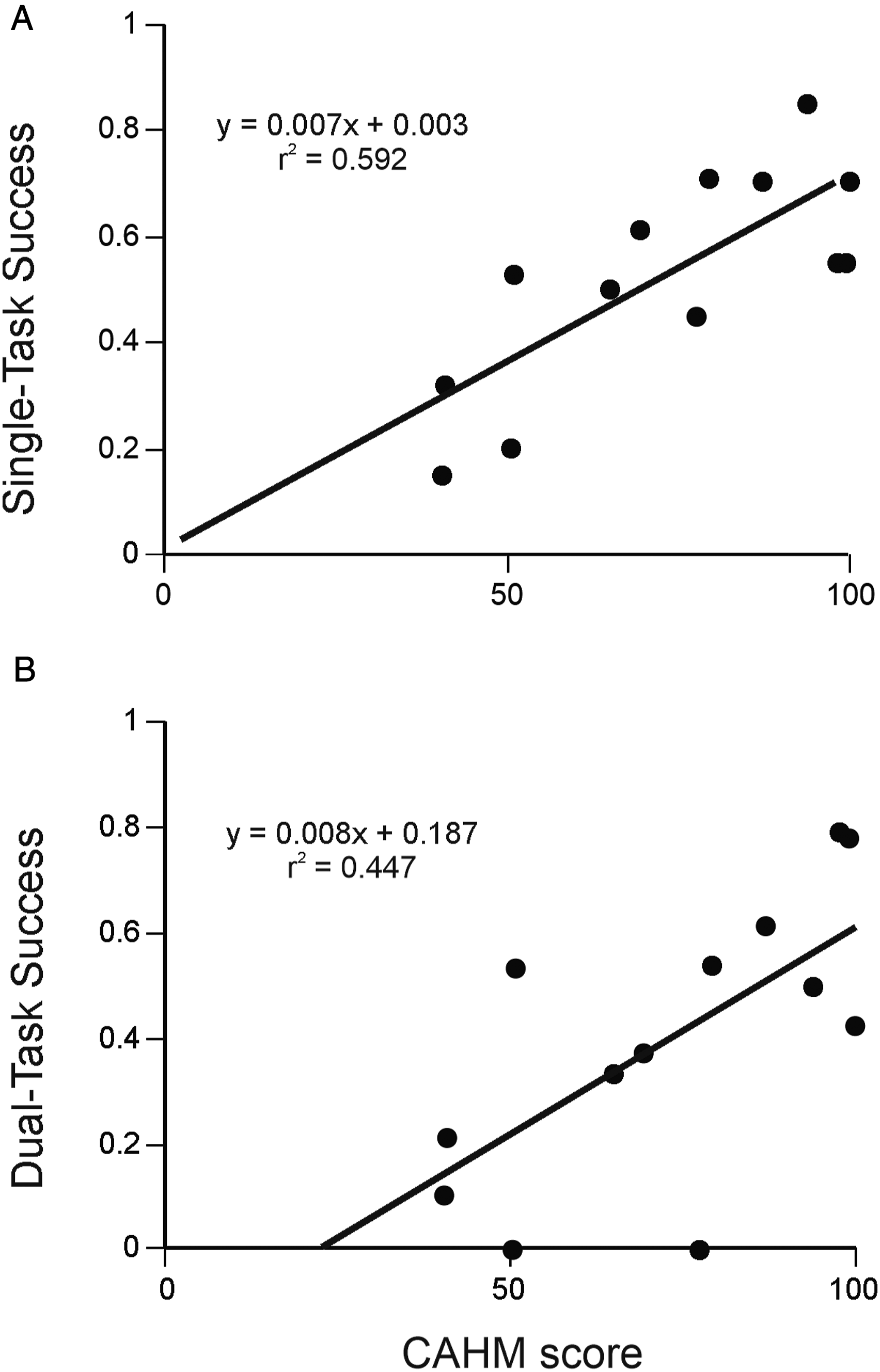
Discussion
Well-recovered patients with stroke had greater difficulty circumventing a moving obstacle while reaching from standing compared to healthy age-matched controls in both single- and dual-task conditions. For both conditions, decreased success may have been related to cognitive factors such as executive function delay in online modulation of the motor action.24,43 This is supported by findings that subjects with stroke could successfully avoid hitting the door only when they substantially decreased their endpoint speed. Patient-reported confidence in arm use moderately correlated with task success in both conditions.
Successful door avoidance was characterized by reduced peak velocity, longer time to reach peak velocity, more movement units, and increased shoulder adduction in both conditions. Stroke subjects made more online movement corrections to avoid hitting the door and used more trunk displacement to assist reaching compared to controls. For the ST condition, these results are similar to those of obstacle avoidance behavior during seated reaching in subjects with stroke, 5 while greater success in obstacle avoidance was achieved when reaching from standing (control: 55.9 ± 24.0%; stroke: 41.6 ± 26.4%, P = .15). The disparity is likely due to differences in target placement and reaching postures used in each study. When reaching from sitting, 5 the target was at shoulder height, whereas the target in this study was placed at mid-sternal level, which may have been easier to reach. The standing posture also allows for a larger number of degrees of freedom to participate in the reaching action, making more movement patterns available to accomplish the task. Based on the DP calculation, Baniña et al. 5 found that patients with stroke began movement corrections later in the reaching path compared to controls for seated reaches, which was not observed in our study. Given the greater number of degrees of freedom available to them, it is possible that when standing, patients with stroke could compensate for delayed movement correction with the arm by starting corrections earlier using proximal joints (i.e., trunk roll). The possibility of proximal recruitment contributing to task success could be investigated in future studies.
Although movement correction times were similar to controls, subjects with stroke had less smooth trajectories as evidenced by the greater number of NMUs. This suggests that the lower success rate could be a consequence of disruption in UL coordination or interruptions due to the triggering of spasticity/pathological movement synergies. 44 The reduced smoothness and greater movement disruption could also be due to a problem in the temporal organization of the movement. 45
Longer time to reach peak velocity and earlier hand path DPs differentiated between successful and failed trials in both groups. However, reaching speed was significantly slower during the initial part of the reach in patients with stroke for both conditions which may reflect slower cognitive processing speed and/or motor planning. 46 It is possible that in trials in which subjects initially moved their arm more slowly, they had more time to make movement corrections and were therefore more successful in avoiding hitting the door. The added cognitive load in dual-task condition may have exacerbated this difficulty. This result is similar to that of Bank et al. 25 who found that in a difficult obstacle-avoidance task, patients with stroke experienced greater cognitive–motor interference than controls. Contrary to our results, they found that within the stroke group, the amount of interference during dual-tasking was more related to motor than cognitive function. This discrepancy may be due to the difference in study samples. We included only well-recovered individuals, while Bank et al. 25 found that patients with more severe motor deficits had greater cognitive–motor interference.
Most patients in our study had reduced performance in one or both tasks suggesting insufficient attentional resources and a diminished capacity for dual-tasking, leading to a prioritization of one task over the other.18,46 The reduced attentional resources and its relation to task success rate in the stroke group were also verified by the moderate relationship between success rate and the time required to complete TMT-B. Slower reaction times have been reported in stroke during gait and balance dual-task activities.18,47 However, gait performance has occasionally been shown to improve while dual-tasking. For example, Kizony et al. 48 observed cognitive facilitation on gait speed in subjects with chronic stroke, in contrast to the cognitive interference usually observed in older subjects. Similarly, we found that 2 stroke subjects had more success in obstacle avoidance during the dual-task condition. This may be explained by a reduced conscious control of movement due to the cognitive distraction and greater automaticity, thus improving performance. 49 Overall, it is likely that diminished cognitive ability after stroke contributed to slower reaction times leading to delays in online modulation of arm movement but that this was mitigated by compensation from the recruitment of more proximal joints.
An interesting finding was that the level of confidence in arm ability rather than arm impairment was related to task success in both conditions. Confidence in one’s motor ability is thought to be a primary factor determining the amount of paretic limb use. 50 However, even patients with full clinical motor recovery (as indicated by FMA-UL) reportedly use their affected hand less often in daily activities. 50 This suggests that confidence in arm and hand movements should be targeted to improve performance in complex everyday tasks and spontaneity of arm use. It also suggests that a more in-depth cognitive evaluation may be required to assess the capacity to engage in complex motor tasks in individuals with stroke.
The generalizability of our results is limited for individuals with more severe or more acute stroke. The sample size was too small to determine the relative contribution of each factor to task success and may have inflated effect sizes. Results may have been influenced by the side and type of stroke as well as hand dominance. Furthermore, anticipatory postural adjustments and interjoint coordination that may provide a better understanding of movement differences between groups were not evaluated. Future studies may address these limitations.
Clinical Implications and Future Directions
Individuals with stroke considered to be clinically well-recovered still had deficits compared to controls when performing a complex obstacle-avoidance reaching task. This suggests that some cognitive and motor deficits that are overlooked by routine clinical evaluations may still pose difficulties in UL performance. Quantification of subtle motor, cognitive, and self-efficacy deficits may identify barriers to arm motor recovery. Knowledge of these deficits may be used to better tailor rehabilitation approaches for arm recovery.
Supplemental material
Supplemental material for this article is available online.
Footnotes
Acknowledgments
The authors wish to thank Rhona Guberek for her clinical evaluations and experimental assistance as well as Christian Beaudoin and Valeri Goussev for their technical support. Thanks are also extended to the study participants who volunteered their time for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Aditi A. Mullick was funded by Heart and Stroke Foundation of Canada. Melanie C. Baniña and Yosuke Tomita were funded by the Richard and Edith Strauss Doctoral Fellowship (McGill University). Mindy F. Levin held a Canada Research Chair in Motor Recovery and Rehabilitation.
Supplementary Material
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
