Abstract
Background
Early-onset sleep disturbance is common following moderate to severe traumatic brain injury (TBI) and often emerges while patients are in posttraumatic amnesia (PTA). However, sleep disruptions during this subacute recovery phase are not well-defined, and research often utilizes indirect measures (actigraphy) that quantify sleep based on activity. This study aims to examine sleep macro-architecture and sleep quality directly with ambulatory polysomnography (PSG) and measure endogenous salivary melatonin levels for patients experiencing PTA following moderate to severe TBI.
Method
Participants were recruited from an inpatient TBI rehabilitation unit. Nighttime PSG was administered at the patient’s bedside. Two saliva specimens were collected for melatonin testing on a separate evening (24:00 and 06:00 hours) using melatonin hormone profile test kits.
Results
Of 27 patients in whom PSG was recorded, the minimum required monitoring time occurred in n =17 (adherence: 63%) at a median of 37.0 days (quartile 1 [Q1] to quartile 3 [Q3]: 21.5-50.5) postinjury. Median non–rapid eye movement (NREM) and REM sleep proportions were similar to normal estimates. Slow-wave sleep was reduced and absent in 35.3% of patients. Sleep periods appeared fragmented, and median sleep efficiency was reduced (63.4%; Q1-Q3: 55.1-69.2). Median melatonin levels at both timepoints were outside the normal range of values specified for this test (from Australian Clinical Labs).
Conclusion
This study reports that ambulatory PSG and salivary melatonin assessment are feasible for patients experiencing PTA and offers new insight into the extent of sleep disturbance. Further research is necessary to understand associations between PTA and sleep disturbance.
Introduction
In the initial stages following a moderate to severe traumatic brain injury (TBI), individuals experience a temporary state of altered consciousness commonly known as posttraumatic amnesia (PTA). 1 PTA is the recovery stage after waking from loss of consciousness (coma), where individuals display no continuous memory for day-to-day events. 2 Whereas the characteristic features of PTA are memory disturbance, disorientation, confusion, and uncharacteristic behaviors (agitation and restlessness),2,3 recent research suggests that sleep disturbance is also commonly experienced during PTA. 4
Sleep disturbance may occur at both the early 4 and chronic stages of TBI recovery. 5 In the overall TBI population, approximately 50% of individuals have sleep disturbance, and 25% to 29% are diagnosed with a sleep disorder at some point postinjury. 6 The elements of sleep that are often disrupted include sleep architecture (staging), 7 sleep quality, 8 and sleep-wake cycles. 9 Individuals therefore commonly experience various sleep problems and/or sleep disorders, 6 including poor sleep maintenance (50%), reduced sleep efficiency (49%), delayed sleep onset (36%), insomnia (29%), daytime sleepiness (27%), obstructive sleep apnea (25%), and hypersomnia (increased sleep need; 22%). 10 However, previous research has mainly evaluated sleep in the longer term (>6 months postinjury) 7 and once patients have emerged from PTA. Therefore, little is known about sleep disturbances in PTA following TBI.
A recent systematic review evaluated the limited research on subacute sleep disturbance post–moderate to severe TBI. 4 It identified a significant association between the stage of recovery and sleep-wake cycle disturbance (SWCD), whereby 79% of patients in PTA were classified with SWCD compared with 36% of patients who had emerged from PTA. 4 Patients in PTA also displayed significantly poorer sleep efficiency and fragmented rest-activity cycles (a measure of sleep-wake cycles). 4 Although this limited research suggests that sleep disturbances are exacerbated during PTA, sleep assessment is typically conducted in the later phases of recovery. The interaction between PTA and sleep disturbance is less clear. Beyond TBI severity, sleep disturbance may affect the emergence from PTA and recovery by interrupting the essential functions of sleep (eg, learning, memory consolidation, neurogenesis, and neuroplasticity). 11 The assessment and recovery of PTA is dependent on memory and new learning. 1 Therefore, earlier assessment, along with environmental modifications to promote sleep hygiene and medication review may help optimize sleep and emergence from PTA.
In terms of sleep assessment, polysomnography (PSG) is considered the gold standard method, 12 conventionally performed in a technician-attended laboratory setting, and involves objectively monitoring an individual’s physiological signals, which inform about sleep and architecture. However, portable PSG devices have also been developed that enable sleep testing in an unattended and naturalistic setting (eg, at patient bedside) and are generally less resource intensive than a laboratory setting. Research has reported that both portable and laboratory PSG yield similar results for sleep architecture. 13 Basic signals are recorded through sensors and include brain wave activity through electroencephalography (EEG; the only means to directly observing sleep architecture), 14 eye movement (electrooculography [EOG]), and muscle movement (electromyography [EMG]). Because of the challenges of PSG testing for patients who are typically confused and restless, 15 previous limited research has mainly opted for indirect measures of sleep during PTA, 4 such as actigraphy (a wristwatch with an inbuilt accelerometer quantifying sleep and wakefulness based on movement). However, actigraphy tends to be poorly correlated with PSG 16 and does not provide estimates of sleep architecture, 12 and reduced compliance with actigraphy has been reported in 28% of patients following TBI. 17 There are additional methods available for assessing sleep post-TBI; however, many of these methods (eg, clinical interviews, self-reported sleep diary, questionnaires) 18 are impractical for patients in PTA, who typically experience anterograde and retrograde memory disturbance. 3 Given these measurement limitations, the extent of sleep disturbance and changes to sleep architecture during PTA remains unknown.
The onset of sleep disturbance likely derives from a complex interaction of factors, 11 such as the trauma compromising brain regions and systems involved in sleep and wake cycle regulation. 18 Accordingly, research in TBI samples has also evaluated endogenous melatonin hormone rhythms,19-23 which are considered a phase marker of the internal circadian clock. 24 The central circadian clock within the brain synchronizes various 24-hour physiological and behavioral rhythms, including melatonin production, temperature, sleep, and wakefulness 25 with the environmental day-night cycle, with melatonin being habitually secreted at night and reduced by morning. Limited TBI research20,21,23 has reported melatonin levels during the very early intensive care unit stage, where the phase of recovery and PTA status is uncertain. Studies have reported decreased melatonin secretion following TBI19,22 and more disrupted melatonin patterns with increasing injury severity. 21 This may be the result of the TBI disrupting brain regions (eg, pineal gland) involved in melatonin synthesis. 22
With the hypothesis that sleep architecture is disturbed after TBI, this pilot study aims to examine sleep macro-architecture (staging) and sleep quality in a naturalistic inpatient rehabilitation setting with overnight ambulatory PSG in patients in PTA following moderate to severe TBI. The secondary aim is to determine the nocturnal secretion of endogenous melatonin from saliva samples at 2 time points overnight.
Methods
Participants
This study was approved by the Epworth HealthCare Human Research and Ethics Committee (Study Number: 55212). Participants were prospectively recruited from consecutive admissions to the inpatient TBI Rehabilitation Unit at Epworth HealthCare where the criterion for admission is medical stability. The specialist TBI Unit contains a dedicated and secured low-stimulation PTA section.
Eligibility criteria for participation were as follows: (1) age ≥18 years, (2) sustained a moderate to severe TBI, and (3) in PTA. Participants were excluded if they were extremely agitated or behaviorally disturbed and would be unable to tolerate the study protocol. This was advised by patients’ treating multidisciplinary rehabilitation team (eg, rehabilitation physician, nursing and therapy staff). There were no criteria regarding time postinjury because this may vary depending on each patient’s acute care needs.
Measures
Westmead PTA Scale
The Westmead PTA Scale was developed in Australia as a standardized assessment scale for prospectively monitoring PTA 26 and is the most commonly used measure of PTA in Australasia. 1 It comprises 12 items that assess the patient’s orientation (date of birth, age, time, and place) and memory by recalling the assessor’s face, name, and picture cards of common objects. The Westmead PTA Scale is administered daily as part of routine inpatient clinical practice. When full scores are reached for 3 consecutive days, the patient is determined to be out of PTA on the first of these days and PTA duration is calculated from the time of injury. Longer PTA duration generally corresponds with increasing brain injury severity.
Polysomnography
PSG was objectively recorded at the patient’s bedside overnight using the Compumedics Somté (V1 system; Compumedics Ltd, Abbotsford, Australia), an ambulatory, portable, lightweight, multichannel PSG device. Portable devices are primarily used for the screening of obstructive sleep apnea 27 ; however, research has also used ambulatory PSG to evaluate sleep architecture and sleep quality. 28 With sleep architecture under investigation, the respiratory-specific channels of a standard PSG were not used. Current guidelines for recording parameters (ie, electrode placement) for in-laboratory PSG (type 1) recommend 3 channels of EEG, 29 with a minimum of 2 channels of EEG. 30 For unattended, ambulatory PSG (type 2) studies, a minimum of 1 EEG lead is required. 30 In this study, electrode placement included the following: 2 central EEG channels using mastoid reference leads, 2 EOG channels, and 2 masseter EMG channels. These parameters are adequate to measure the proportion of non–rapid eye movement (NREM) sleep stages (N1: the transition stage between wake and sleep; N2: low voltage activity with presence of sleep spindles and k-complexes; and N3: slow-wave restorative sleep), REM sleep (paradoxical sleep), total sleep time, sleep efficiency, number of awakenings, wake after sleep onset, and sleep latency. Sleep staging was scored in accordance with the American Academy of Sleep Medicine (AASM) Scoring Manual. 29 A recommended minimum PSG recording time is 6 hours.31,32 During PSG recording, sleep times were not imposed on participants who typically have disturbed sleep wake cycles. The Somté device was applied to correspond with the nighttime nursing shift schedule and the “lights out” period on the ward (21:00 to 06:00 hours).
Melatonin
Melatonin hormone profile test kits from Australian Clinical Labs (Australia) were utilized to collect 2 saliva specimens: at midnight (24:00 hours) and in the morning (06:00 hours). Kits contain 2 test tubes for saliva collection, which were externally analyzed by Australian Clinical Labs. Samples were frozen, thawed, and centrifuged prior to analysis via enzyme-linked immunosorbent assay. Melatonin production is inhibited by bright light, and therefore, the midnight sample is required to be taken in dim lighting. A Digitech Lux Meter (Australia) was used to determine the light intensity (lux). Melatonin levels were originally provided in picomoles per liter. To allow comparison with prior studies, the investigators converted this unit of measurement to picograms per milliliter. The conversion rate was (pmol/L/4.30 = pg/mL). 33
Injury Severity Score (ISS)
The ISS is an anatomical scoring system of trauma severity. 34 Body regions (head and neck, face, chest, abdomen, extremities, and external) are assigned an Abbreviated Injury Scale (AIS) score to classify injury severity. The AIS utilizes a 6-point scale ranging from minor to maximal (untreatable). The ISS is calculated by squaring the 3 highest AIS scores and summing these values. A senior physiotherapist specializing in trauma populations calculated an ISS for each patient using hospital medical records. Major trauma is defined as an ISS greater than 15. 35
Demographics and Injury Characteristics
Data on patient demographics (gender, age), injury characteristics (Glasgow Coma Scale, length of stay, injury mechanism), and medication use were collected from patients’ hospital medical records.
Procedure
Patients in PTA were admitted to Epworth’s inpatient rehabilitation unit following acute care within an external hospital. Consecutive admissions were screened according to the study criteria, and written informed consent was obtained from each participant’s designated person responsible (ie, family member or close other). Study measures (PSG and saliva collection for melatonin testing) were conducted separately over 2 nights while the patient remained in PTA. This occurred so that a full night of patients’ sleep could be recorded with PSG without interruption, as required by saliva sampling.
Experienced TBI ward nurses were trained to administer PSG by a senior sleep scientist. The Somté device was secured around the patient’s torso with a Velcro strap. Skin sites for electrode placement were prepped to increase tracings, by lightly abrading the skin with Nuprep Skin Prep Gel (Weaver and Company, Aurora, CO). Electrodes were secured using foam adhesive electrodes (for EOG and EMG leads) or Ten20 Conductive Electrode Paste (Weaver and Company; for EEG leads). Sleep scientists at Epworth HealthCare’s Sleep Unit (with more than 10 years of experience) analyzed PSG recordings. Overnight nursing staff would collect and refrigerate saliva specimens prior to analysis externally.
Statistical Analysis
A sample size of 12 patients of any specific group is the minimum figure suggested for pilot studies, 36 albeit in the context of a randomized controlled trial. Descriptive statistics were calculated for sample demographics, injury characteristics, PSG variables, and melatonin levels. Sleep data are often skewed, 37 and so medians, first (25th percentile), and third (75th percentiles) quartiles were calculated, as were nonparametric Spearman rank order correlations and Mann-Whitney tests. As is generally undertaken in sleep research, 38 we reported proportions such as the length of time spent in stage 1 NREM sleep divided by the total time spent asleep. The statistical analysis of proportions can potentially be problematic, in that this approach assumes that there is no correlation between the denominator (eg, total time asleep) and the ratio (eg, proportion of stage 1 sleep). Correlation between the ratio and the denominator may indicate that the size of the ratio or proportion does not remain constant across observed values of the denominator—in this case, total sleep time. 39 The above assumption was explored using Spearman correlations. The 95% CIs for the latter were obtained using the bias-corrected and accelerated bootstrap with 10 000 resamples. 40 Statistical analyses were conducted using IBM SPSS, version 26 (IBM Corporation, New York, 2019).
Results
Sample Characteristics
There were 36 patients recruited into this study, and of these, 4 patients declined PSG and 3 patients were unable to produce saliva for collection. There were patients who emerged from PTA and were excluded from analyses (n = 5 during PSG recording and n = 6 during melatonin testing). This was mainly a result of patients being determined to be out of PTA retrospectively as required by the Westmead PTA Scale (ie, data collection occurred during the 3 consecutive days when maximum scores were obtained). The analytic samples therefore consist of 17 patients with PSG and 26 patients in whom melatonin levels were determined (Figure 1).
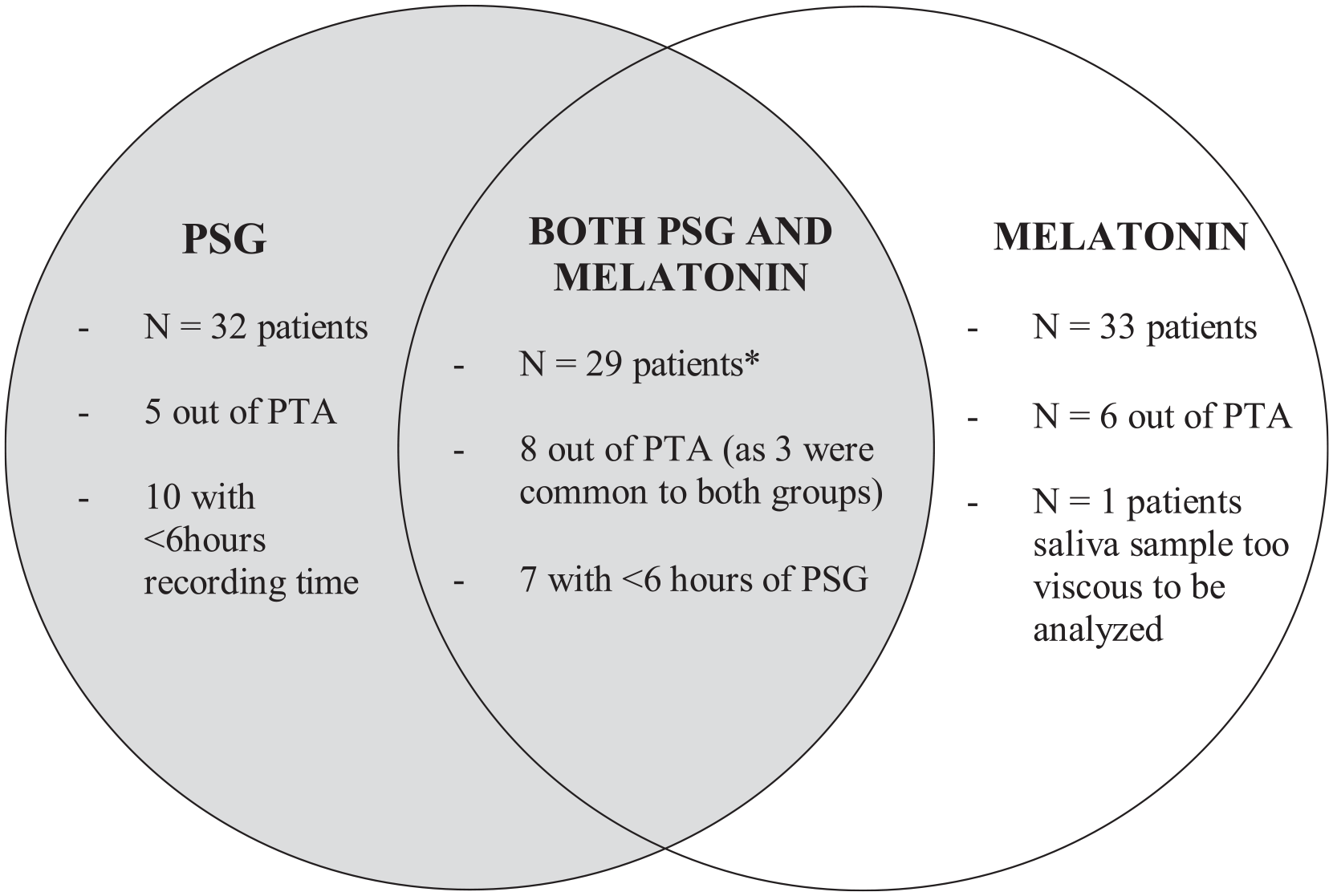
Number of patients grouped by measures completed.
Table 1 displays the demographics and injury characteristics of the overall sample, divided into subgroups according to the measures completed. All patients were classified with severe (31.0%) or very severe (69.0%) TBI per the classification system for TBI severity 41 and had a median PTA duration of 47.0 days. Within patients’ medical histories, there were no recorded preinjury sleep disturbances and/or sleep disorders. No patients were taking sleeping medications at the time of the study.
Sample Demographics and Injury Characteristics by Study Measurement.
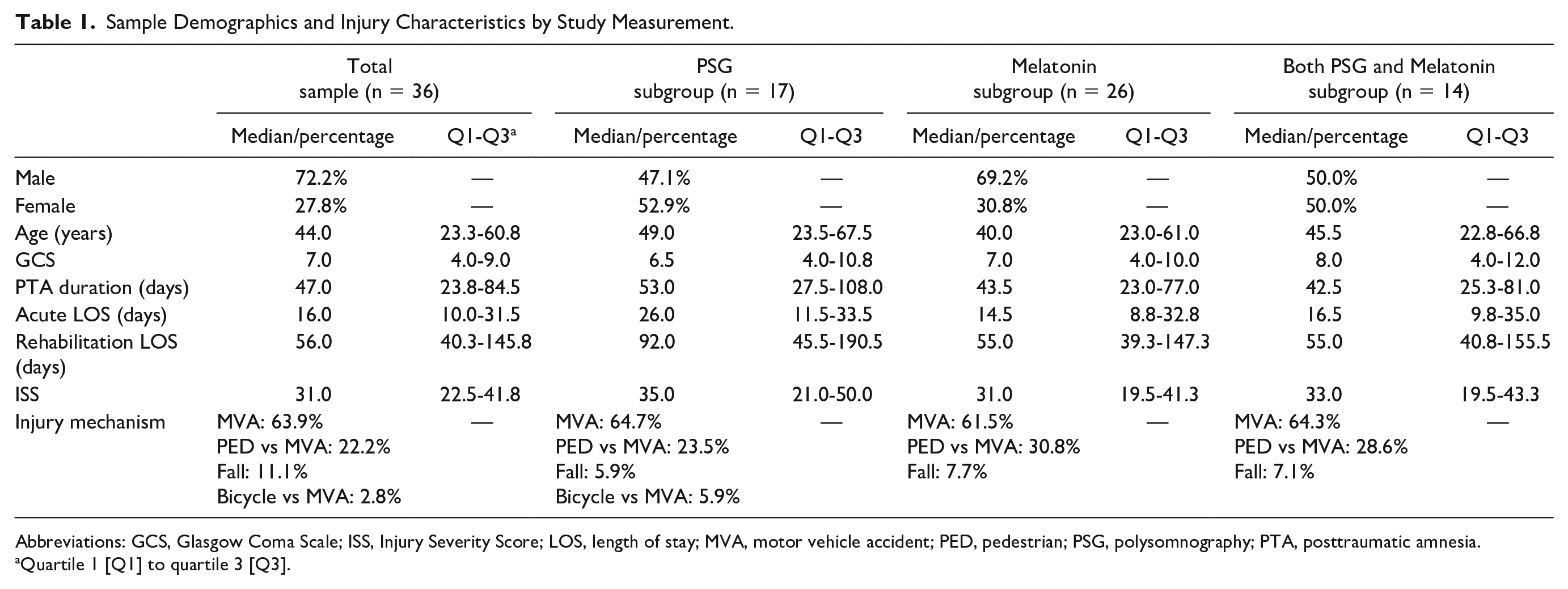
Abbreviations: GCS, Glasgow Coma Scale; ISS, Injury Severity Score; LOS, length of stay; MVA, motor vehicle accident; PED, pedestrian; PSG, polysomnography; PTA, posttraumatic amnesia.
Quartile 1 [Q1] to quartile 3 [Q3].
Polysomnography Group (n = 17)
PSG was conducted on 27 patients in PTA, of whom 17 patients had ≥6 hours of recording time (PSG adherence rate: 63%; Figure 1). Among the 10 patients with <6 hours of PSG, lead signals were either lost (n = 6) or the patient removed the device (n = 4). Within the analytic sample (n = 17), see Supplementary Material (available online) for the medications taken at the time of PSG.
Table 2 displays the results of PSG and general estimates of sleep stage and wake proportions in healthy young adults42,43 and Figure 2 displays patient hypnograms which represent PSG recordings. There were no statistically significant correlations between time (days) postinjury and PSG variables. Sleep duration was generally reduced (median: 5.3 hours; Q1-Q3: 4.9-6.1), difficult to initiate (with increased sleep onset latency) and appeared fragmented, with increased wake time during sleep periods (median: 120 minutes; Q1-Q3: 61.8-150.8). This equates to a median of 29.0% spent awake during sleep periods. The median sleep efficiency was therefore reduced (63.4%; Q1-Q3: 55.1-69.2), with 15 patients (88.0%) displaying a sleep efficiency below normal ranges (normal or good sleep quality across all age ranges ≥85%). 44 In terms of sleep macro-architecture, the median proportions of the broader sleep stages (NREM and REM) appear similar to normal estimates: 78.3% (Q1-Q3: 75.4-94.3) and 21.7% (Q1-Q3: 5.7-24.6), respectively. Within NREM, deep, slow-wave restorative sleep (N3) was generally reduced (6.4%; Q1-Q3: 0-19.1), and in 35.3% of the sample, it was entirely absent. This was offset by increased proportions of sleep time spent in the lighter sleep stages (N1 and N2). There were no statistically significant differences in sleep staging proportions (N1, N2, N3, REM, NREM) for patients taking opioids compared to patients not taking opioids.
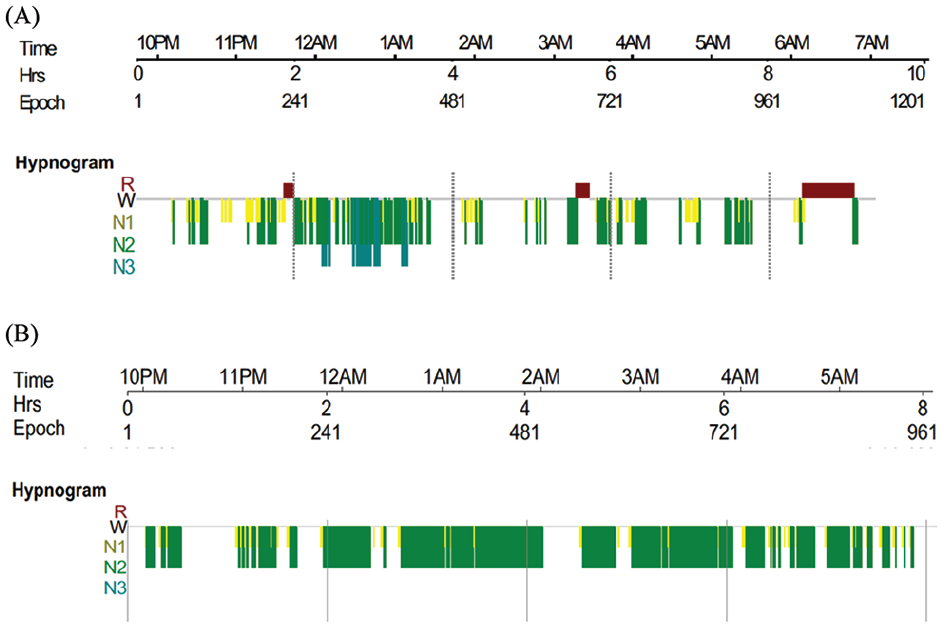
Examples of hypnograms to display PSG recordings. The colored bars represent each stage of sleep as indicated, and the gaps indicate wake time. (A) Patient displayed frequent awakenings throughput the sleep period, poor sleep efficiency and reduced N3 sleep. (B) Patient displayed an absence of N3 and R sleep.
PSG Results (n = 17).
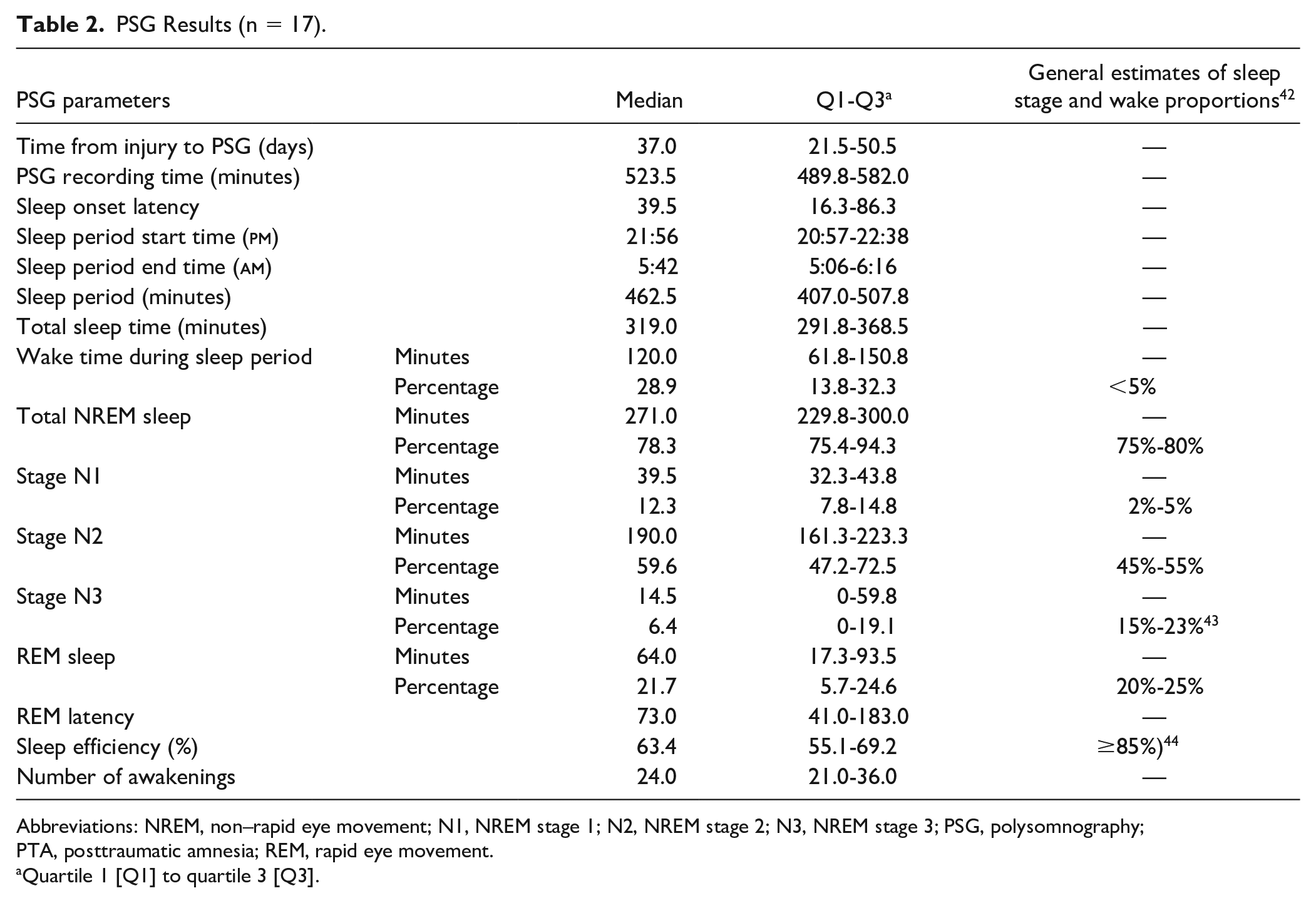
Abbreviations: NREM, non–rapid eye movement; N1, NREM stage 1; N2, NREM stage 2; N3, NREM stage 3; PSG, polysomnography; PTA, posttraumatic amnesia; REM, rapid eye movement.
Quartile 1 [Q1] to quartile 3 [Q3].
The proportion of N1 was negatively associated (Spearman ρ = −0.79; P < .001; 95% CI = −0.98 to −0.35) with total sleep duration, indicating that the longer the total sleep, the shorter the proportion of N1 sleep. On the other hand, the proportion of N3 was positively associated (ρ = 0.51; P = .037; 95% CI = 0.01 to 0.83) with total sleep duration. The proportion of N2 sleep was not significantly correlated (ρ = −0.24; P = .36; 95% CI = −0.74 to 0.33) with total sleep duration.
Melatonin Group (n = 26)
Reference ranges for salivary melatonin based on healthy population studies were provided by Australian Clinical Labs. These included the following: 10 to 40 pg/mL (24:00 hour sample) and <3 pg/mL (06:00 sample). During PTA, the median salivary melatonin levels at midnight and morning were (9.1 pg/mL; Q1-Q3: 1.2-18.5) and (17.3 pg/mL; Q1-Q3: 6.0-23.7), respectively. Among the sample, there was some variability in melatonin levels at each sampling point. However, melatonin levels at midnight appear generally reduced, with 14 patients (53.8%) displaying melatonin levels below the reference range (Figure 3). For morning melatonin levels, 21 patients (80.8%) were above the reference range (Figure 3). Median melatonin change between both time points was −1.6 pg/mL (Q1-Q3: −11.0 to 1.9), indicating that on average, melatonin levels did not decrease between midnight and morning. A total of 19 patients (73.1%) had either higher melatonin levels by morning or no change between sampling points (Figure 4). In patients for whom melatonin and PSG recordings were available (n = 14), there were no statistically significant correlations between melatonin levels at either time point and sleep staging proportions. Although β-adrenergic receptor blockers can suppress melatonin, 45 only 2 patients were on this medication, and their melatonin levels at both time points were not statistically significantly different from the rest of the sample.
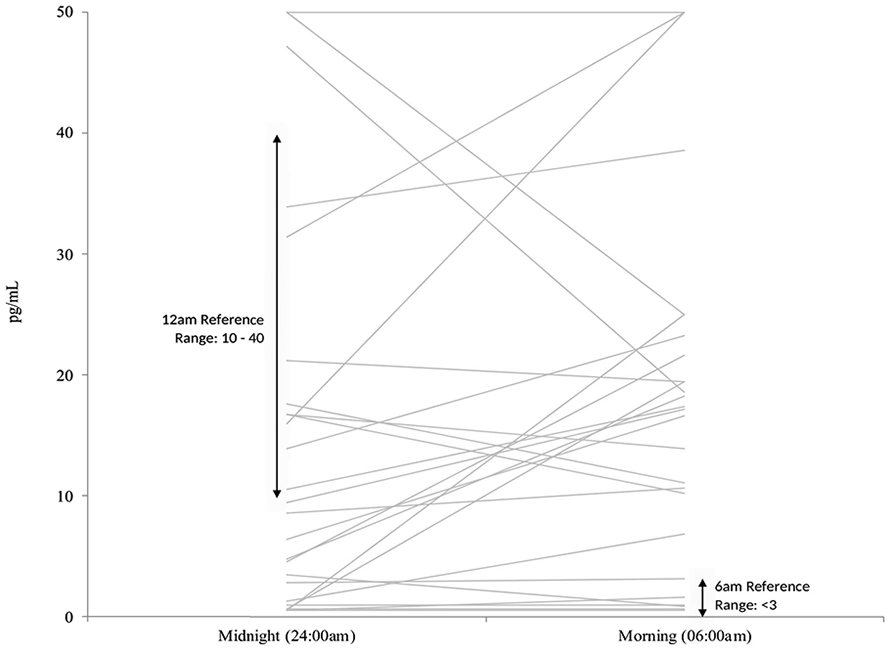
Melatonin level at midnight and morning for each patient (n = 26).
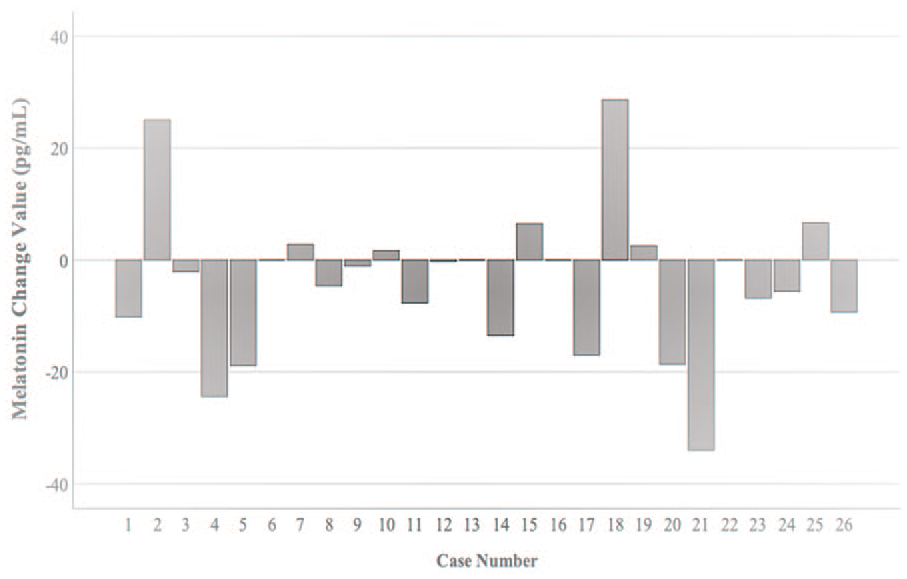
Change in melatonin value between midnight to morning for each patient. Change values greater than zero indicate that melatonin levels have decreased between the 2 sample times.
Discussion
To our knowledge, this pilot study is the first to report that the use of PSG, the gold standard in sleep assessment, was feasible for nearly two-thirds of patients experiencing PTA. The use of the ambulatory version of PSG overcame some difficulties that prevent this patient group attending an in-laboratory PSG. Overall, disturbances to sleep macro-architecture, sleep quality, and endogenous melatonin levels were common. One important finding was the reduction of deep, slow-wave restorative sleep. The median proportion of slow-wave sleep (6.4%) was well below normal estimates (15%-23%), and more than a third of the sample was unable to enter this sleep stage. Individuals displayed decreased sleep duration, and the quality of these sleep periods was fragmented. Collectively, sleep efficiency was decreased because of increased wake time during sleep periods. Results also appear to suggest disruptions to intrinsic mechanisms involved in sleep regulation.
Feasibility of PSG During PTA
This study demonstrated that gold-standard PSG was a feasible and tolerable method of assessing sleep during PTA for the majority of participants. This is an important finding, particularly because PSG involves more elaborate monitoring, 15 yet has a similar compliance rate to actigraphy (where 28% of patients post-TBI had reduced compliance). 17 Prior research studies, however have primarily opted for indirect proxy measures of sleep-wake cycles (such as actigraphy) because of their low invasiveness 46 and ability to monitor sleep over longer periods. Therefore, PSG has been generally applied during the later stages of TBI recovery, 7 where it is perceived as more likely to be tolerated. However, as has been discussed in a recent study, 47 this pilot study also suggests that a majority of patients in PTA may be able to tolerate some level of stimulation without an increase in agitation (eg, the application of PSG that is beyond the normal level of stimulation occurring during routine ward testing and daily interactions).
Although the use of PSG is feasible during PTA, tailoring its use may be required. PSG is commonly performed in a technician-attended laboratory setting, where individuals sleep overnight. However, in this study, ambulatory unattended PSG was applied in patients’ naturalistic hospital setting. This enables the patients to remain in a familiar environment with freedom to move, which is consistent with recommendations for PTA management. 3 It may also remove the “first-night effect” as seen with standard laboratory PSG, where sleep can be disturbed by a new and artificial environment. 48
Disturbances to Slow-Wave Sleep (N3) During PTA
Following TBI, alterations to sleep architecture can be difficult to identify because PSG findings can vary between individuals. 49 This is not unexpected because factors are known to influence sleep staging proportions (eg, age). 42 Although the results of this study should be viewed with caution given the sample size, overall, there still appears to be a reduction or absence of slow-wave sleep. This contrasts prior research, which demonstrated normal sleep staging proportions in acute TBI patients, where the majority were out of PTA. 28 Following a TBI, symptoms of insomnia (difficulty falling or staying asleep) are common 6 and are known to reduce slow-wave and REM sleep. 7 Although insomnia can also persist long-term, 50 chronic TBI studies, including a meta-analysis, have more commonly reported increased slow-wave sleep,7,8,22 which may be related to increased learning and neuroplasticity. 8 This study suggests that alterations to slow-wave sleep may be specific to PTA. It has been suggested that sleep fragmentation may prevent deep sleep. 46 In our study, individuals commonly displayed poor sleep quality, maintenance, and increased wake time during sleep periods. Slow-wave sleep is recognized as the most restorative sleep stage, 51 and research suggests its involvement in functions such as cognition (memory and attention), energy, hormone release, and metabolism. 52 It is, therefore, probable that disturbance to slow-wave sleep may affect the resolution of PTA, 46 given memory is a core assessment component of PTA measures. Research also suggests that patients with a quicker recovery of clearly delineated rest-activity cycles (a measure of sleep-wake cycles) are more likely to clear PTA. 46 Therefore, improving the consolidation of the sleep-wake cycle may be an important factor for PTA resolution and recovery. Decreases in slow-wave sleep are also common in sleep disorders such as obstructive sleep apnea. 52 Studies53,54 have reported both obstructive and central sleep apnea within an inpatient TBI rehabilitation setting. Future research may consider sleep apnea screening instruments during the PTA period because patients may not be able to tolerate additional respiratory monitoring during PSG. Chronic opioid use is also a risk factor for central sleep apnea, 55 and this medication can also reduce both slow-wave sleep and REM sleep. 56 In our study, the early use of opioids was not significantly associated with sleep staging proportions. Prior research during early TBI recovery 54 also found no relationship between central nervous system–active medications and sleep-related breathing disturbance.
During the course of a normal sleep period, individuals typically alternate between NREM and REM sleep stages in cyclic patterns. Slow-wave sleep is mostly present within the first third of the evening, and as the night progresses, it decreases and may disappear entirely. 57 However, in our study, there was a moderate, positive correlation between the percentage of slow-wave sleep and total sleep time, indicating that increased slow-wave sleep is more likely with longer periods of sleep. This trend may, therefore, be specific for patients in PTA, whereas N1 sleep (the transition stage between wake and sleep) displayed the opposite trend and is less likely with increased sleep periods. These results highlight that median staging proportions alone may provide an incomplete picture, and it is also important to consider their associations with its denominator (eg, total sleep time). The proportion of sleep stage may not be independent of total sleep time, and each appears to have diverse relationships with total sleep time. As well as there being a need for further research on sleep during PTA, there also needs to be more careful consideration into the analysis of correlations between proportion of stages of sleep and total sleep times, particularly when the latter is restricted, as it is in PTA.
Melatonin Production During PTA
In this study, melatonin levels were determined through saliva samples, which is generally less invasive than other biological fluids (eg, serum, urine). Overall, melatonin production at both time points was outside the reference ranges and was not significantly associated with sleep staging proportions. Nocturnal melatonin production normally peaks overnight (eg, between midnight and 5
Assessment of Sleep Disturbances During PTA and Future Directions
This pilot study suggests that disturbances to sleep architecture, sleep quality, and endogenous melatonin levels are common during PTA. These results are aligned with a recent systematic review, which found increased disturbances to sleep during PTA. 4 Overall, the research supports routine screening for sleep disturbance during PTA, whereby a formal assessment protocol needs to be established. This could include supplementing PSG with other methods, such as actigraphy, to capture daytime activity and sleep periods over longer periods. However, research is required validating such proxy sleep measures against PSG in individuals experiencing PTA. As discussed earlier, considerations also need to be given regarding the interaction between PTA symptoms, sleep disturbance, and various factors that may also influence sleep (eg, cognition, 59 agitation, environment, and medication). 18 This could lead to further research evaluating whether sleep disturbance during PTA should be considered a therapeutic target for promoting recovery and emergence from PTA. For example, a criterion for evaluating the recovery of PTA might be sleep improvement, with one study demonstrating that improvements in sleep efficiency as measured by actigraphy during PTA predicted the recovery of continuous memory (one component of PTA). 60
Study Limitations
This study has some limitations. It was designed as a pilot study to evaluate the logistics of utilizing ambulatory PSG and so included a small cohort of patients. However, there were challenges with performing the study methodology in patients who typically present as confused, disoriented, and/or agitated. In this study, there were interferences with PSG channel signals quality (22% of patients), which is not unexpected given the general agitated and restless nature associated with PTA and the unattended setting. Rates of data loss in unattended sleep studies generally range from 3% to 18%. 61 One limitation of PSG is that monitoring is typically only for 1 night, 15 and so daytime sleep and wake activity is not captured, which may contribute to nocturnal sleep disturbance. PSG is considered the gold standard and should be utilized when possible. However, for monitoring daytime activity and sleep over longer periods, future studies may need to consider supplementing PSG with other objective measures, such as actigraphy. Activity levels are reduced in a rehabilitation setting 62 and particularly during PTA, and actigraphy is known to underestimate sleep disruptions. 16 Given the pilot nature of this study, there was no maximum age limit. Sleep architecture can be influenced by age. 42 However, this study generally had a younger cohort, and controlling for age was difficult because of the small sample size. As discussed earlier, it is impractical to obtain direct input from patients given memory disturbance during PTA. Therefore, this study is unable to control for influencing factors on sleep, for example pain levels or preinjury sleeping habits. However, no patient had a documented history of preinjury sleep disturbance and/or sleep disorders. This study also did not control for other factors, such as the influence of nurse rounding or medication use. It would be difficult to isolate the effect of medications with the current sample size. To determine the overnight rhythm of melatonin, more frequent sampling is typically required (every 30-60 minutes). 63 However, this may be impractical for patients in PTA. Frequent sampling could either disrupt or completely deprive individuals of sleep. 63 This is particularly difficult in individuals who are already experiencing clinically significant changes to sleep. It is also generally used when small phase changes are relevant. 63 Melatonin was collected on a separate evening, so that sleep would not be disturbed during PSG recording. However, this is likely to have a negligible impact, and the environment remained the same for both measures.
Conclusions
Ambulatory PSG and melatonin assessment are challenging, yet feasible assessment tools for patients experiencing PTA. PSG also provides additional and valuable data, including sleep architecture. This study contributes to the emerging evidence that suggests considerable disturbances to both sleep architecture, specifically restorative sleep, and sleep quality in PTA.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683211011241 – Supplemental material for Assessing Sleep Architecture With Polysomnography During Posttraumatic Amnesia After Traumatic Brain Injury: A Pilot Study
Supplemental material, sj-docx-1-nnr-10.1177_15459683211011241 for Assessing Sleep Architecture With Polysomnography During Posttraumatic Amnesia After Traumatic Brain Injury: A Pilot Study by Bianca Fedele, Dean McKenzie, Gavin Williams, Robert Giles and John Olver in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors gratefully acknowledge the support of Epworth’s TBI Rehabilitation Nursing Team for their role in conducting the study methodology. This includes, but is not limited to, Jacky Ma, James Stribling, Michele Williams, Ed Cascayan, and Sally Lalande. We also wish to thank Compumedics Ltd (Abbotsford, Australia) for kindly providing the Somté PSG device for the duration of the study. Thank you to Dr Michelle Caldecott for your assistance with developing the study methodology and Natalie Swaby for assisting with the analysis of PSG recordings. Also, thank you to Dr Bridget Hill for calculating ISS scores.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RG reports personal fees from Compumedics Ltd (Abbotsford, Australia), for PSG analysis, outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Professor Jack Cade AM Intensivist Major Development Grant from the Epworth Research Institute (ERI).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
