Abstract
Background
Very early exercise has been reported to exacerbate motor dysfunction; however, its mechanism is largely unknown.
Objective
This study examined the effect of very early exercise on motor recovery and associated brain damage following intracerebral hemorrhage (ICH) in rats.
Methods
Collagenase solution was injected into the left striatum to induce ICH. Rats were randomly assigned to receive placebo surgery without exercise (SHAM) or ICH without (ICH) or with very early exercise within 24 hours of surgery (ICH+VET). We observed sensorimotor behaviors before surgery, and after surgery preexercise and postexercise. Postexercise brain tissue was collected 27 hours after surgery to investigate the hematoma area, brain edema, and Il1b, Tgfb1, and Igf1 mRNA levels in the striatum and sensorimotor cortex using real-time reverse transcription polymerase chain reaction. NeuN, PSD95, and GFAP protein expression was analyzed by Western blotting.
Results
We observed significantly increased skillful sensorimotor impairment in the horizontal ladder test and significantly higher Il1b mRNA levels in the striatum of the ICH+VET group compared with the ICH group. NeuN protein expression was significantly reduced in both brain regions of the ICH+VET group compared with the SHAM group.
Conclusion
Our results suggest that very early exercise may be associated with an exacerbation of motor dysfunction because of increased neuronal death and region-specific changes in inflammatory factors. These results indicate that implementing exercise within 24 hours after ICH should be performed with caution.
Introduction
Current recommendations for stroke exercise suggest that intervention should occur as early as possible, and many medical institutions start exercise immediately if the stroke patient’s medical condition is stable. Numerous previous studies have investigated the effects of early exercise following stroke. Early exercise studies in animal models of stroke have shown enhanced motor function recovery and reduced injury volume.1-4 Our lab has shown that compared with late or no exercise, early treadmill exercise after intracerebral hemorrhage (ICH) promotes functional sensorimotor recovery by preventing neuronal death and cortical atrophy while preserving dendritic structure.5,6 In addition, early post-ICH skills training increases postsynaptic density protein 95, amino-3-hydroxy-5-methylisoxazole-4-propionic acid, and microtubule-associated protein 2, all of which are involved in neuroplasticity.7-9 However, the AVERT study recently reported that very early rehabilitation following stroke promotes worse functional outcomes at 3 months compared with usual care.10,11 In addition, a clinical study examining different intervention initiation times showed that initiating an early mobilization intervention within 24 hours of stroke does not lead to favorable functional outcomes compared with initiating the intervention within 24 to 48 hours. 12 Subsequent analysis of AVERT has reported that brief, high-frequency, very early rehabilitation improves the likelihood of good functional outcomes when controlling for age and stroke severity; however, increasing total mobilization duration (min/d) during the early poststroke period is associated with reduced likelihood of good outcomes. 13 It has also been reported in animal studies that very early exercise following stroke adversely affects motor dysfunction and brain tissue. It has been shown that exercise 6 hours after stroke increases BAX/Bcl-2 and induces neuronal cell death in rats that received middle cerebral artery occlusion.14,15 Furthermore, early intervention is associated with increased expression of proinflammatory mediators (interleukin [IL]-1β) and cellular stress markers (Hsp70 and HIF-1α). 16
It has been reported that very early exercise activates microglia, which releases proinflammatory factors in rats with traumatic brain injury. 17 M1-type microglia that express proinflammatory factors are activated in the early stages of stroke, including during cerebral infarction and cerebral hemorrhage. Microglia expressing IL-1β are activated 6 hours after cerebral hemorrhage, and increased IL-1β mRNA expression is observed over the following 7 days. 18 Furthermore, astrocytes are reported to be involved in the expression of IL-1β after brain injury.19,20 M2-type microglia are gradually activated after ICH onset. 21 These M2-type microglia express the nerve growth factor IGF-1 and the anti-inflammatory factor TGF-β1.22,23 However, the effect of very early post-ICH exercise on the expression of microglial and astrocytic factors is not fully understood.
To reconcile these findings, this study investigated the effects of very early exercise after ICH on hematomas and edema as well as on neurons, synapses, and astrocytes. In addition, the Il1b, Tgfb1, and Igf1 mRNA expressions were examined.
Materials and Methods
Animals and Experimental Group
Adult male Wistar rats (250-300 g, n = 122) were pair-housed in clear Plexiglas enclosures at 23 ± 1 °C under a 12 h/12 h light/dark cycle. Food and water were freely available. Animal care and surgical procedures were performed according to the animal care guidelines of the Niigata University Committee on Health and Welfare. All efforts were made to minimize suffering and the number of animals used. All rats were divided into SHAM-operated (n = 44, SHAM) and ICH-operated rats prior to surgery. The ICH surgical rats were then divided into 2 groups: one without exercise intervention (n = 47, ICH) and the other with very early exercise intervention (n = 31, ICH+VET).
ICH Surgery
ICH was induced in the left striatum as previously described. 24 Rats were anesthetized with sodium pentobarbital (45 mg/kg, intraperitoneal administration) and placed in a stereotaxic frame. A small hole was drilled 3.0 mm lateral to the midline and 0.2 mm anterior to bregma. A 26-gauge needle (Hamilton, Reno, NV) was lowered 4.0 mm below the surface of the skull, and 1.6 µl saline containing 0.2 U of bacterial collagenase (type IV; Sigma-Aldrich, St Louis, MO) was infused over a period of 8 minutes using a microinjector (IMS-20; Narishige Scientific Laboratory, Tokyo, Japan). The needle was kept in place for an additional 8 minutes after completion of the infusion and, subsequently, was slowly withdrawn. The SHAM group was infused with 1.6 µl saline without collagenase.
Exercise Protocol
Pretraining on the treadmill was done for 3 days, from 4 to 2 days before the surgery, by increasing the running speed step by step from 6 to 11 m/min. The ICH+VET group was subjected to the running exercise for 60 minutes at 6 and 24 hours after the operation at speeds of 9 and 11 m/min, respectively. The ICH group was pretrained before surgery on a treadmill and kept in standard cages after surgery until sacrificed.
Behavioral Tests
Horizontal Ladder Test
The horizontal ladder test was performed using an apparatus with side walls constructed of transparent panels and metal rungs as previously described. 25 Rats (SHAM: n = 15; ICH: n = 14; ICH+VET: n = 13) walked across a ladder with varying distances between the rungs (1-3 cm). Rats were trained 3 times daily over 2 to 4 days before surgery. They were tested 3 times at 3 and 25 hours after surgery by an experimenter who was blind to the treatment group. The number of steps, correct placements (the midportion of the forelimb palm was placed on the rung), and errors (the forelimb fell between the rungs) of the contralateral forelimb at each crossing were counted. Foot placement scores were assigned according to the scale developed and validated by Metz and Whishaw. 25 The average values for these variables were calculated for the 3 daily trials.
Rotarod Test
Before ICH, rats were trained on the accelerating rotarod at a constant speed of 4 rpm until they remained on the rotating spindle for 60 s. Each rat was then given 5 trials on the accelerating rotarod, during which the spindle increased from 4 to 40 rpm over 5 minutes. 26 These assessments were performed at 3 and 25 hours after surgery by an experimenter blind to the treatment group.
Hematoma Volume and Edema Analysis
Hematoma Volume
Rats (ICH: n = 6; ICH+VET: n = 6) were deeply anesthetized using sodium pentobarbital, then transcardially perfused with 0.9% saline followed by 0.1 M phosphate buffer (pH 7.5) containing 4% paraformaldehyde. The brains were excised and 500-µm-thick frozen sections were prepared using a microtome. Immediately after sectioning, the slices were imaged with a microscope. ImageJ (US National Institute of Health, Bethesda, MD) was used to measure the hematoma area and the total brain area of each section. The ratio of the hematoma area to the total brain area was calculated for each section. The measured section values for each rat were averaged and used as the hematoma volume. This evaluation was performed by an experimenter blind to the treatment group.
Edema
Rats (SHAM: n = 7; ICH: n = 6; ICH+VET: n = 6) were humanely killed under deep anesthesia 27 hours following ICH, and their brains were excised and separated. Brain water content was determined using the wet/dry weight ratio method. The olfactory bulbs, brain stem, and cerebellum were removed, and the rest of the brain was immediately weighed to obtain the wet weight. The dry weight was obtained after drying the brain in an oven at 100 °C for 24 hours. The wet and dry weights were converted to percentage water content [(Tissue wet weight − Tissue dry weight)/Wet weight × 100].
Protein Expression Analysis
The entire striatum and sensory motor cortex of the injured side were excised at 6 hours (SHAM: n = 12; ICH: n = 12) and 27 hours (SHAM: n = 8; ICH: n = 8; ICH+VET: n = 7) postoperation and Western blotting was performed. Tissue samples were homogenized in lysis buffer containing phosphatase and protease. Samples were centrifuged at 12 000 rpm at 4 °C for 30 minutes. The protein concentration in tissue samples was determined by Biospec-nano (SHIMAZU, Kyoto, Japan). Protein samples (20 µg) were separated by 12% SDS-PAGE and transferred to polyvinylidenefluoride (PVDF) membranes (Millipore, MA). The membranes were blocked in 5% skim milk in phosphate buffered saline (PBS) containing 0.1% Tween 20 for 1 hour at room temperature, followed by overnight or 30-minute incubation at 4 °C with NeuN (1:2000, monoclonal rabbit, Cat No. 24307, Cell Signaling Technology, Beverly, MA), GFAP (1:1000, polyclonal rabbit, Cat No. ab116010, Abcam plc, Cambridge, UK), PSD-95 (1:2000, polyclonal rabbit, Cat No. ab18258, Abcam plc, Cambridge, UK), and β-actin (1:2000, polyclonal rabbit, Cat No. PM053-7, Medical & Biological Laboratories, Nagoya, Japan) primary antibodies diluted in 2.5% skim milk in PBS containing 0.1% Tween 20. The membranes were then washed and incubated with goat antirabbit immunoglobulin G–Horseradish peroxidase (HRP) conjugated (1:1000-2000, Cat No. 7074S, Cell Signaling Technology, Beverly, MA) for 1 hour at room temperature. After rinsing with buffer, the immunocomplexes were visualized using standard chemical luminescence methods (PS-3, Lumigen ECL Plus). The film signals were digitally scanned and quantified using densitometric image software (Lumicube; Liponics Inc, Tokyo, Japan). Expression was normalized to the respective β-actin levels.
mRNA Expression Analysis
The entire striatum and sensory motor cortex of the injured side were excised at 6 hours (SHAM: n = 9; ICH: n = 8) and 27 hours (SHAM: n = 8; ICH: n = 7; ICH+VET: n = 12) postoperation, and quantitative real-time reverse transcription polymerase chain reaction (RT-PCR) was performed using sets of gene-specific primers for Il1b, Tgfb1, Igf1, and Caspase-3. Total RNA was extracted using the TRIzol method. The RNA concentration was measured by Biospec-nano (SHIMAZU, Kyoto, Japan). Total RNA was used for 1-step real-time quantitative PCR with a 1-Step SYBR PrimeScript PLUS RT-PCR Kit (Takara Bio, Tokyo, Japan) and CFX Connect Real-Time PCR Detection System (Bio-Rad Laboratories, Inc, CA), according to the manufacturers’ instructions. The primer sequences were as follows: Actb, forward: 5′-GGAGATTACTGCCCTGGCTCCTA-3′, reverse: 5′-GACTCATCGTACTCCTGCTTGCTG-3′; Il1b, forward: 5′- CCCTGAACTCAACTGTGAAATAGCA-3′, reverse: 5′- CCCAAGTCAAGGGCTTGGAA-3′; Tgfb1, forward: 5′- CATTGCTGTCCCGTGCAGA-3′, reverse: 5′- AGGTAACGCCAGGAATTGTTGCTA-3′; Igf1, forward: 5′-GCACTCTGCTTGCTCACCTTTA-3′, reverse: 5′- TCCGAATGCTGGAGCCATA-3′; and Caspase-3, forward: 5′-GCAGCAGCCTCAAATTGTTGAC-3′, reverse: 5′- TGCTCCGGCTCAAACCATC-3′. Expression was normalized against the respective Actb mRNA level. The relative amounts of each product were calculated using the comparative Ct method.
Statistical Analysis
Statistical analyses were performed using Statcel4 (OMS, Saitama, Japan). The χ2 goodness-of-fit test was used to examine data normality. One-way ANOVA and Tukey post hoc tests were used to analyze between-group differences. Data that did meet assumptions of normality were analyzed by the nonparametric Kruskal-Wallis test, followed by the Steel-Dwass multiple comparison test. The criterion for significance was P < .05.
Results
ICH+VET Exacerbates Motor Dysfunction
The rotarod test evaluates the balance and coordinated motor function of the entire body. Postsurgery, the rotarod performance in the ICH and ICH+VET groups was significantly impaired compared with the SHAM group. After exercise, there was no significant difference in the rotarod performance between the ICH and ICH+VET groups (P < .05; Figure 1B). The horizontal ladder test was used to assess the coordinated motor function of the paralyzed forelimbs following ICH. Immediately after ICH, the ladder score decreased significantly compared with SHAM. In addition, the correct rate decreased significantly, and the error rate increased significantly. The ICH+VET group scores decreased significantly after exercise compared with scores in the ICH and SHAM groups (P < .05; Figure 1C). These results demonstrate that very early exercise following ICH exacerbates some motor dysfunction. Specifically, we observed deteriorations in skillful motor dysfunction, with no effect on whole body coordinated motor dysfunction.
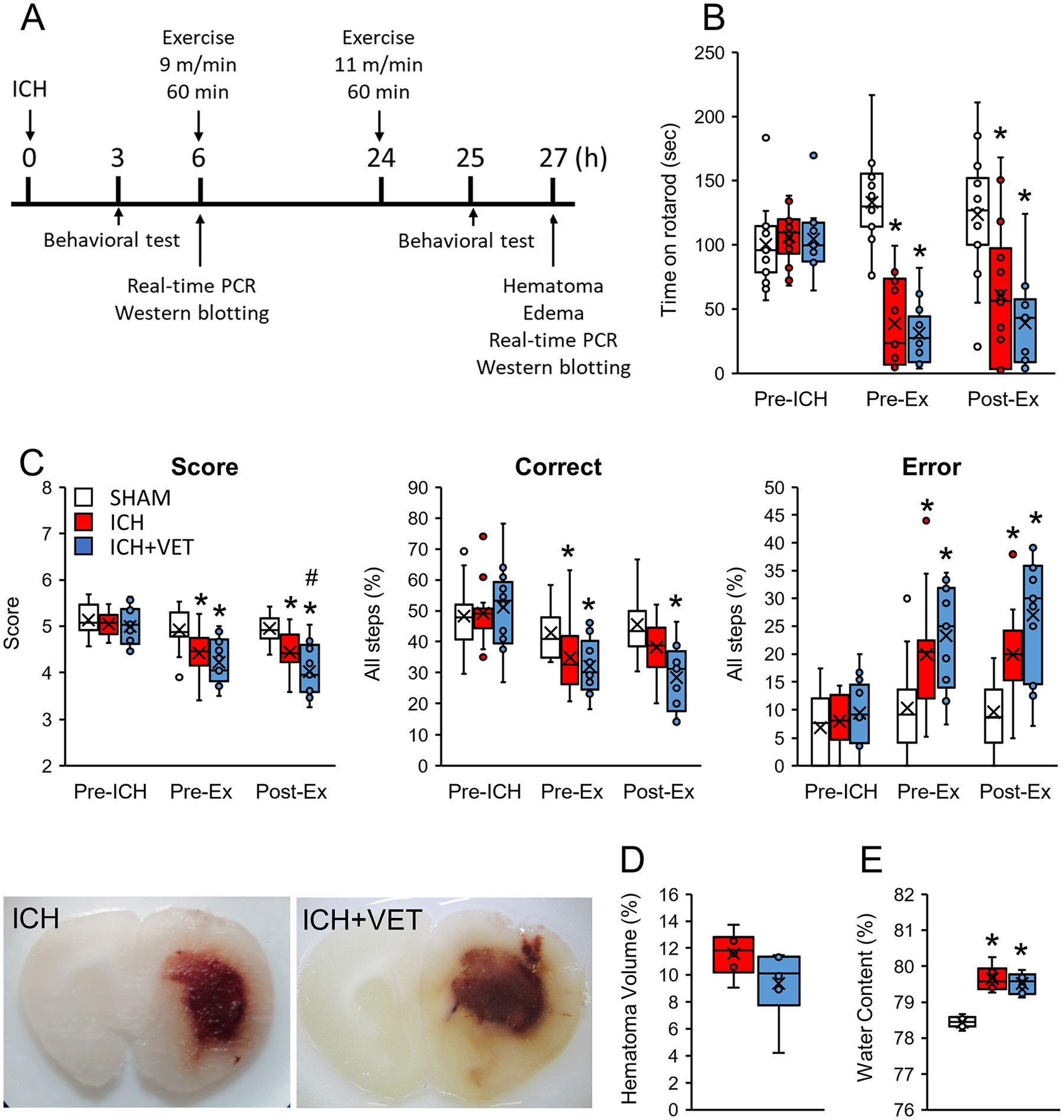
Experimental design and motor function assessment. (A) The experimental design. Only the ICH+VET group received the exercise intervention. (B) In the rotarod test, ICH significantly reduced performance with no further effects of ICH+VET (SHAM: n = 15; ICH: n = 14; ICH+VET: n = 13). (C) In the horizontal ladder test, ICH significantly reduced performance metrics, which were further reduced by ICH+VET (SHAM: n = 15; ICH: n = 14; ICH+VET: n = 13). (D) Hematoma and (E) cerebral edema were induced by ICH (n = 6) and not further altered by ICH+VET (n = 6).
ICH+VET Did Not Affect Hematoma or Edema
We analyzed the effects of very early exercise on hematoma volume and cerebral edema. ICH-induced hematomas were not affected by very early exercise (P > .05; Figure 1D). Cerebral edema was also significantly increased in the ICH and ICH+VET groups compared with the SHAM group, but there was no significant difference between the ICH and ICH+VET groups (P > 0.05; Figure 1E). These results demonstrate that very early exercise may not adversely affect hematoma and cerebral edema.
ICH+VET Promotes Neuronal Cell Death and Synaptic Regression
We quantitatively examined the effect of very early exercise on neurons, synapses, and glial cells using the respective protein markers NeuN, PSD95, and GFAP. Six hours after ICH, the NeuN and GFAP protein expression levels were significantly decreased in the striatum (P < .05; Figures 2A and 2D). However, the protein expression of the synaptic marker PSD95 remained unchanged despite the decrease in the neuronal marker NeuN (P > .05; Figure 2B). Therefore, a PSD95/NeuN value was calculated as an index of the synapse-to-neuron expression. PSD95/NeuN was significantly higher in the ICH group than in the SHAM group (P < .05; Figure 2C). At 27 hours following ICH, persistently reduced NeuN protein expression was observed in the striatum of the ICH group (P < .05; Figure 2E). NeuN protein expression levels in the ICH+VET group were significantly lower than in the SHAM group (P < .01; Figure 2E). PSD95 protein expression did not significantly differ between the groups (P > .05; Figure 2F), but PSD95/NeuN was significantly higher in the ICH+VET group compared with the SHAM group (P < .05; Figure 2G). GFAP protein expression did not significantly differ between the groups (P > .05; Figure 2H).
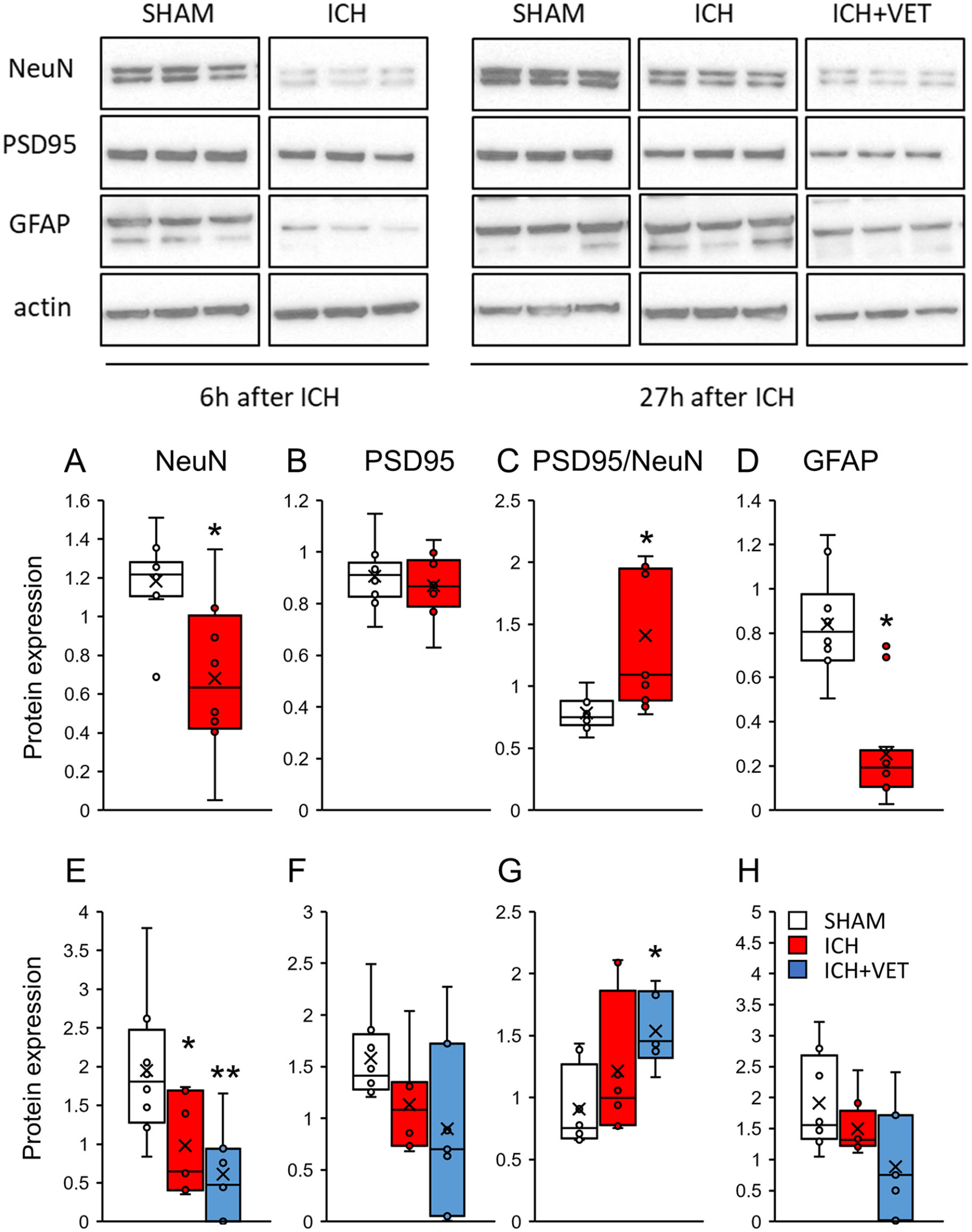
Protein expression in the striatum. Immediately after ICH (A) NeuN and (D) GFAP protein expressions were significantly reduced and (B) PSD95 protein expression was unchanged, but (C) PSD95/NeuN was significantly increased (SHAM: n= 12; ICH: n= 12). ICH+VET decreased (E) NeuN and increased (G) PSD95/NeuN protein expressions significantly more than ICH. There were no significant group differences in (F) PSD95 and (H) GFAP protein expressions (SHAM: n = 8; ICH: n = 8; ICH+VET: n = 7).
Six hours after ICH, no protein expression changes were observed in the sensorimotor cortex (P > .05; Figures 3A-3D). In the sensorimotor area, NeuN and PSD95 protein expression levels were significantly lower in the ICH+VET group compared with the SHAM group (P < .05; Figures 3E and 3F). GFAP protein expression did not significantly differ between the groups (P > .05; Figure 3H). These results indicate that very early exercise after cerebral hemorrhage may promote neuronal cell death in the striatum and sensorimotor cortex.
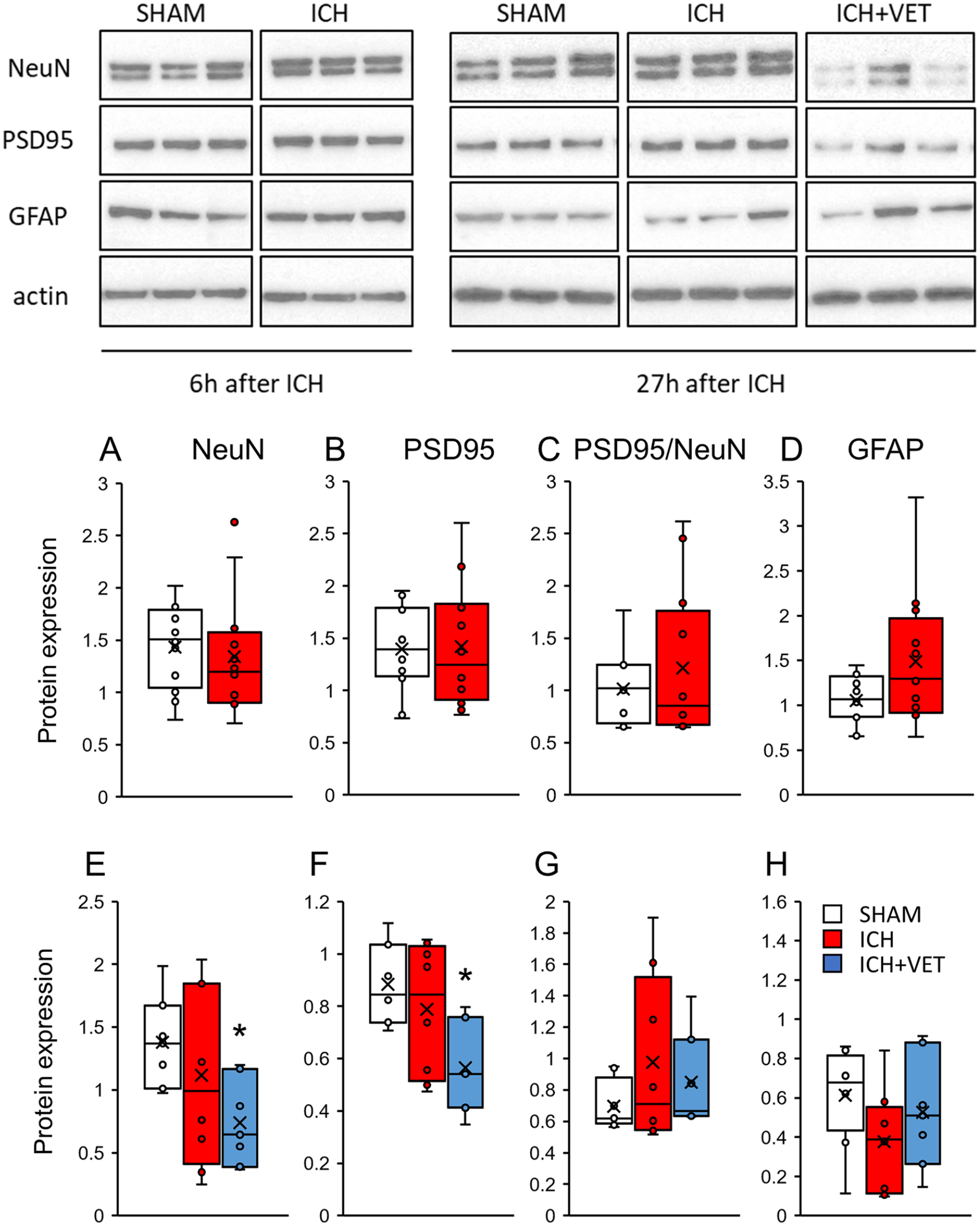
Protein expression in the sensorimotor cortex. Immediately after ICH (A) NeuN, (B) PSD95, (C) PSD95/NeuN, and (D) GFAP protein expressions were unchanged (SHAM: n = 12; ICH: n = 12). ICH+VET reduced (E) NeuN and (F) PSD95 protein expressions compared with SHAM, but there were no group differences in (G) PSD95/NeuN and (H) GFAP protein expression (SHAM: n = 8; ICH: n = 8; ICH+VET: n = 7).
ICH+VET Increases Proinflammatory Factors
At 6 hours following ICH, the Il1b mRNA expression was significantly increased (P < .05; Figure 4A), whereas the Igf1 and Caspase-3 mRNA expression was significantly decreased in the striatum (P < .05; Figures 4C and 4D). ICH+VET significantly increased the Il1b (P < .05; Figure 4E) and Tgfb1 mRNA expression in the striatum compared with SHAM (P < .05; Figure 4F). The Igf1 and Caspase-3 mRNA expression did not significantly differ between the groups (P > .05; Figures 4G and 4H). Similarly, in the sensorimotor area, the Il1b mRNA expression was significantly increased (P < .05; Figure 5A), whereas the Igf1 and Caspase-3 mRNA expression was significantly decreased (P < .05; Figures 5C and 5D) at 6 hours following ICH. The Tgfb1 mRNA expression did not significantly differ between the groups (P > .05; Figure 5B). At 27 hours following ICH, the expression of all quantified mRNAs was significantly increased in the sensorimotor area (P < .05; Figures 5A-5D). The Igf1 mRNA expression of ICH+VET in the sensorimotor cortex was significantly lower than that of ICH (P < .05; Figure 5G). The Caspase-3 mRNA expression was higher in the ICH+VET group than that in the SHAM group (P < .01; Figure 5H). These results indicate that ICH increases inflammation in the striatum. Notably, there was no increase in proinflammatory factors in the sensorimotor cortex, but ICH+VET suppressed the increases in nerve growth factors. Therefore, very early exercise after ICH may differentially affect the striatum and sensorimotor cortex.
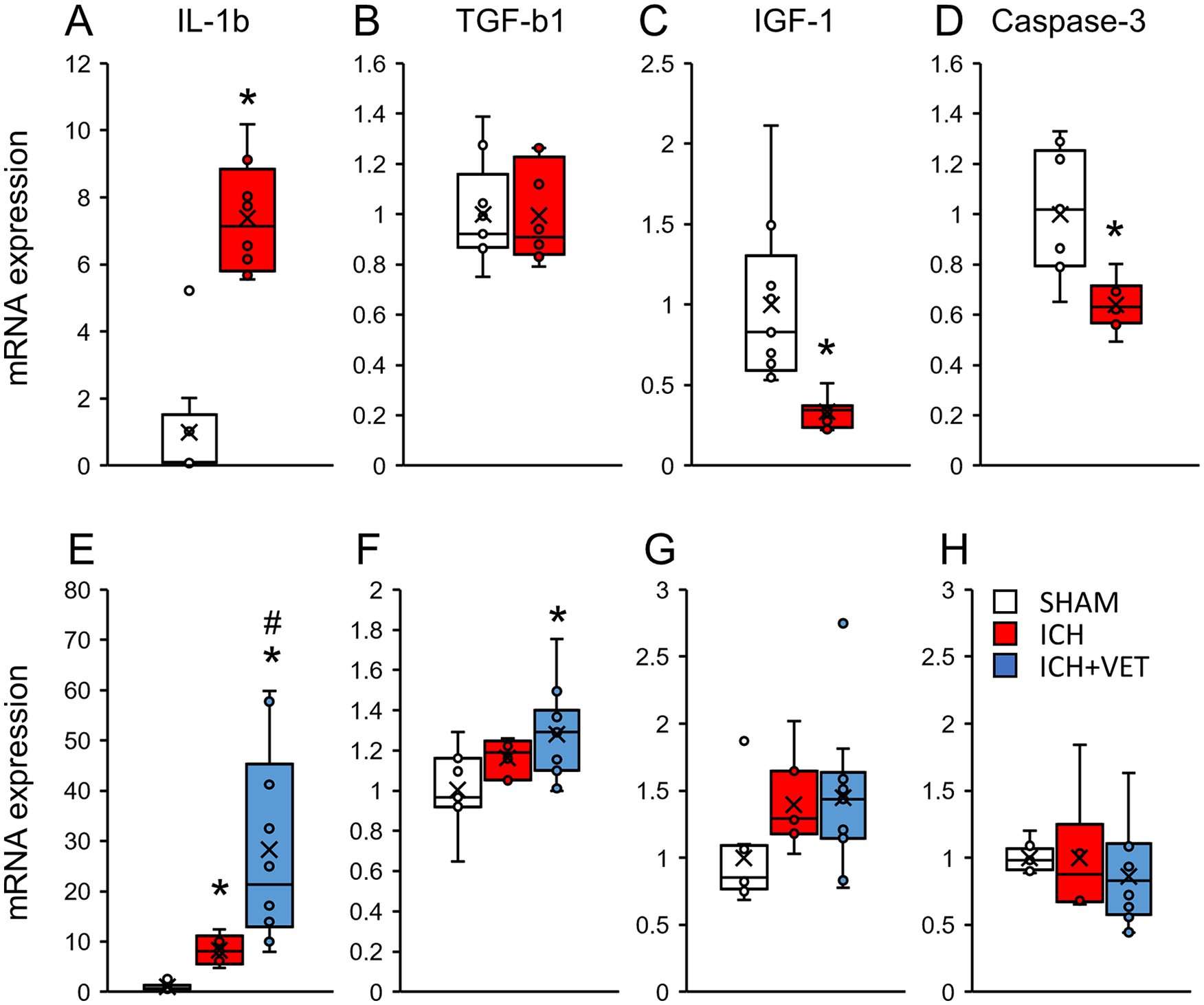
mRNA expression in the striatum. Immediately after ICH (A) the IL-1b mRNA expression levels were significantly increased, (B) TGF-b1 mRNA expression was unchanged, and (C) IGF1 and (D) Caspase-3 mRNA expression levels were significantly decreased (SHAM: n = 9; ICH: n = 8). ICH+VET increased (E) the IL-1b mRNA expression levels compared with ICH and (F) TGF-b1 mRNA expression compared with SHAM. No changes were observed in (G) IGF1 and (H) Caspase-3 expression levels (SHAM: n = 8; ICH: n = 7; ICH+VET: n = 12).
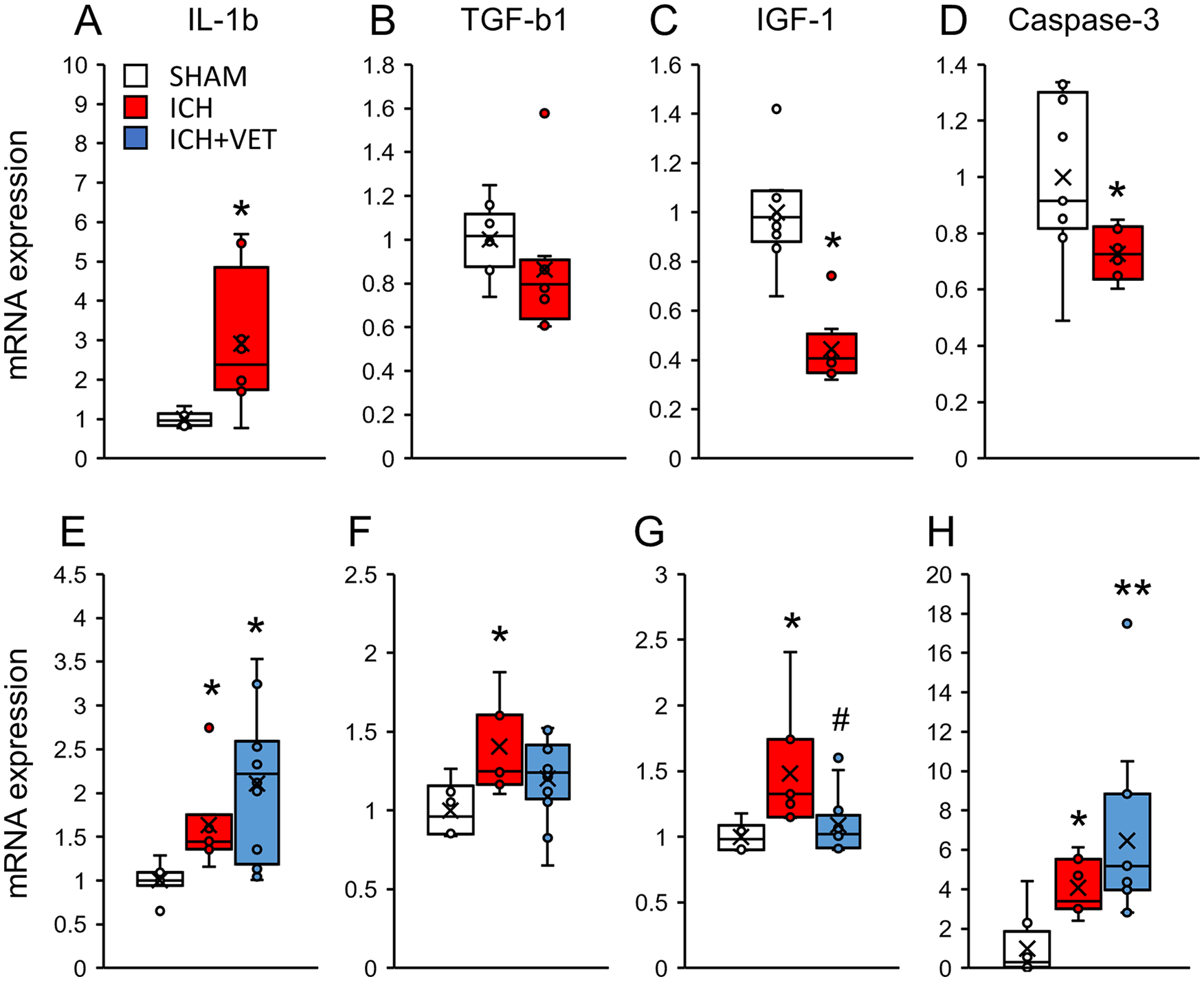
mRNA expression in the sensorimotor cortex. Immediately after ICH (A) the IL-1b mRNA expression levels were significantly increased, (B) TGF-b1 mRNA expression was unchanged, and (C) IGF1 and (D) Caspase-3 mRNA expression levels were significantly decreased (SHAM: n = 9; ICH: n = 8) ICH+VET did not increase (E) the IL-1b mRNA expression, but (H) Caspase-3 was increased compared with ICH. In addition, ICH+VET prevented the ICH-induced increase in (F) the TGF-b1 and (G) IGF1 mRNA expression levels (SHAM: n = 8; ICH: n = 7; ICH+VET: n = 12).
Discussion
This study examined the effects of very early exercise after ICH induced using the collagenase model. We found that very early exercise performed on a treadmill within 24 hours of ICH onset worsened motor dysfunction. In addition, we found a trend toward a decrease in neurons in the striatum and sensorimotor cortex. Specifically, increased proinflammatory factors were observed in the striatum, which may have played a role in promoting neuronal cell death. Although this increase in proinflammatory factors was not observed in the sensorimotor cortex, anti-inflammatory and nerve growth factor expression were suppressed. Furthermore, ICH+VET tended to increase apoptosis-promoting factors. These results indicate that very early exercise may promote region-specific effects after cerebral hemorrhage.
Very early exercise promotes an increased infarct volume following cerebral ischemia. 15 However, ICH+VET did not increase the hematoma volume or brain edema, indicating that these outcomes are not associated with the observed reduction in the number of neurons. Microglia activated by hematoma or cerebral edema, especially M1-type microglia expressing IL-1β, are likely to be involved. Previous studies in rats with cerebral ischemia and cortical damage have reported that very early exercise increased proinflammatory and proapoptotic factors.14-17 Similarly, the present study shows that ICH increased the proinflammatory factor Il1b mRNA expression at the site of injury. The Il1b mRNA expression after cerebral hemorrhage may be regulated by pyroptosis and M1-type microglia. Pyroptosis is a type of inflammation-induced regulated cell death that can be induced at the site of injury immediately after cerebral hemorrhage. 27 This programmed cell death can be caused by the action of a cell-side proteolytic enzyme called caspase-1, which induces the secretion of proinflammatory cytokines, such as IL-1β and IL-18.28-30 Administration of caspase-1 inhibitors suppresses pyroptosis and decreases the release of the proinflammatory factor IL-1β. 31 Given that we observed an increase in Il1b mRNA, it is possible that very early exercise following ICH may promote pyroptosis. Astrocytes, which express high levels of GFAP, have an important role in the release of IL-1β.32,33 In the present study, GFAP expression was unexpectedly decreased at 6 hours after ICH. A previous study reported that IL-1β repressed the transcription of GFAP, 34 with this effect, which is likely to be concentration and time dependent. 35 In the present study, the ICH-induced overexpression of Il1b mRNA might have led to IL-1β suppressing GFAP expression. Future studies will be needed to verify the relationship between IL-1β and GFAP following ICH.
Similar to M1-type microglia, M2-type microglia gradually increase after cerebral hemorrhage. 21 Our results suggest that M2-type microglia might have been activated by very early exercise because TGF-β1 tended to increase. However, the increase in TGF-β1 was limited and not equivalent to the increase in IL-1β, suggesting that M1-type microglia may have been preferentially activated compared with M2-type microglia.
In the present study, there was an increase in Il1b in the sensorimotor cortex immediately after ICH that was not observed in the ICH+VET group. Conversely, ICH+VET suppressed the Tgfb1 and Igf1 mRNA expression, an opposing effect compared to that observed in the striatum at the site of injury. Our previous work has demonstrated that the Tgfb1 and Igf1 mRNA expression was increased in the sensorimotor cortex 8 days following ICH and returned to normal levels 15 days after cerebral hemorrhage. 5 Continuous exercise starting the second day after ICH did not increase the expression of these mRNAs, 5 but exercise starting within 24 hours after ICH suppressed expression of these mRNAs. This may be a result of stress from exercise. Poststroke exercise raises corticosterone immediately after exercise ends. 36 Stress suppresses M2-type microglia activation,37,38 which may further suppress Tgfb1 and Igf1 mRNA expressions. The brain may be particularly susceptible to stress immediately after cerebral hemorrhage because of a significant loss of motor function and disruption of stress-tolerant neural circuits in the brain. Therefore, it is possible that the stress caused by exercise immediately after ICH suppressed the neuroprotective effect of M2-type microglia. In contrast to the striatum (injury site), ICH+VET tended to increase Caspase-3 expression in the ipsilateral sensorimotor cortex. Although the sensorimotor cortex was unaffected by the hematoma, apoptotic neuronal death was induced in the sensorimotor cortex after ICH and was accelerated by very early exercise. Damage to the corticospinal tract has been shown to induce apoptosis in the primary motor cortex, 39 and collagenase-induced ICH is known to damage the corticospinal tract, especially when the internal capsule is the site of injection. 40 Striatal hemorrhage also reduces the number of pyramidal cells in layer V of the sensorimotor cortex. 5 Therefore, very early exercise may facilitate pyramidal cell apoptosis in the sensorimotor cortex following damage to the corticospinal tract.
The present study analyzed the ratio of synaptic density to neuronal expression using PSD95/NeuN. We referred to a previous study that analyzed it using Synaptophysin/NeuN. 41 PSD95 protein expression at the site of injury was not altered by ICH or ICH+VET. However, PSD95/NeuN was significantly elevated immediately after ICH and tended to increase following ICH+VET. PSD95 is a major scaffolding protein and one of the most abundant proteins in postsynaptic thickening. It interacts with a variety of molecules and contributes to the maintenance of synaptic function and plasticity. 42 However, immediately following ICH, PSD95 interacts with the NMDA receptor subunit NR2B and nNOS. These interactions can lead to synaptic damage and cognitive impairment. 43 Furthermore, very early exercise increases PSD95-NR2B-nNOS complex activity and may contribute to the exacerbated motor dysfunction observed in this study.
Although previous studies have examined very early exercise in animal models of ischemic stroke, the present study is the first to show that very early exercise has a negative effect after ICH. Cerebral infarction and cerebral hemorrhage have different pathogeneses and recovery processes. 44 Indeed, in our previous study, we examined motor function recovery in cerebral infarction and cerebral hemorrhage rat models where brain injury occurred in the same region and was of the same size and found that the recovery process over time was different between infarction and hemorrhage models. In the very early poststroke stage, functional decline was more marked in cerebral hemorrhage than in cerebral infarction, but the subsequent recovery was faster in cerebral hemorrhage. 45 Because the progression of pathological and recovery processes following stroke-induced brain injury is different between cerebral hemorrhage and cerebral infarction, an early rehabilitative exercise intervention may have different effects in different stroke types.
There are some limitations to this study that could be addressed through further research. First, the mechanism leading to increased Il1b mRNA expression following ICH+VET needs to be clarified through histological and biochemical analysis of microglial activation and pyroptosis. Second, in this study, we interpret the decrease in expression of the neuronal marker NeuN as indicative of neuronal death. However, as previous studies have reported that NeuN level after cerebral ischemia does not correspond to the number of neurons, 46 it will be necessary to use other methods such as histological investigation to verify the presence of neuronal death in future studies. Third, we performed the behavioral assessment in the ICH+VET group within 24 hours of the exercise intervention, and our behavioral results may reflect an effect of fatigue. Therefore, it will be necessary in future studies to identify any effect of fatigue by performing exercise intervention and behavioral assessment in a concurrent SHAM+VET group.
Conclusion
This study shows that ICH+VET did not exacerbate hematoma volume or cerebral edema but increased motor dysfunction, proinflammatory factor Il1b mRNA expression, and neuronal cell death in the area of injury. In addition, it may have increased excitotoxicity by altering the PSD95/NeuN ratio. In the sensorimotor cortex, ICH+VET promoted neuronal death through apoptosis and suppressed increases in the mRNA expression of the anti-inflammatory factor Tgfb1 and the nerve growth factor Igf1. This is the first study to show that very early exercise within 24 hours of ICH can adversely affect brain tissue and exacerbate motor dysfunction. These results suggest that implementing exercise within 24 hours after ICH should be performed with caution. Future studies should identify the ideal exercise programs that will not adversely affect motor dysfunction.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI Grant Number JP 20K11269 and the Japanese Physical Therapy Association (19-A18).
