Abstract
Background. Rehabilitation aids recovery from stroke in animal models, including in intracerebral hemorrhage (ICH). Sometimes, rehabilitation lessens brain damage. Objective. We tested whether rehabilitation improves recovery and reduces perihematoma neuronal death. We also evaluated whether rehabilitation influences iron toxicity and inflammation, mediators of secondary degeneration after ICH. Methods. Rats were trained to retrieve food pellets in a staircase apparatus and later subjected to striatal ICH (via collagenase infusion). After 1 week, they were given either enriched rehabilitation (ER), including reach training with group housing and environmental enrichment, or control treatment (group housing). Rats in the first experiment were treated for 2 weeks, functionally assessed, and killed humanely at 1 month to determine brain levels of nonheme iron. A second experiment used a similar approach, except that animals were euthanized at 14 days to evaluate perihematoma neuronal death (FluoroJade), iron distribution (Perls), and astrocyte (GFAP) and microglia (Iba-1) activity. A third experiment measured levels of iron-binding proteins (ferritin and transferrin) at 14 days. Results. Striatal ICH caused functional impairments, which were significantly improved with ER. The ICH caused delayed perihematoma neuronal death, which ER significantly reduced. Hemispheric iron levels, the amount of iron-binding proteins, and perihematoma astrocytes and microglia numbers were significantly elevated after ICH (vs normal side) but were not affected by ER. Conclusions. Rehabilitation is an effective behavioral and neuroprotective strategy for ICH. Neither effect appears to stem from influencing iron toxicity or inflammation. Thus, additional work must identify underlying mechanisms to help further therapeutic gains.
Introduction
Intracerebral hemorrhage (ICH) is a devastating stroke that accounts for 10% to 20% of all cases. Mortality rates are high, and long-term neurological outcome is often poor.1,2 For instance, those with an ICH affecting the basal ganglia are often left with significant motor impairments that limit activities of daily living.3,4 Animal ICH models have been instrumental in identifying mechanisms underlying brain damage and for testing putative neuroprotectants,5-7 although none have yet been clinically proven to work. Unfortunately, few studies have examined recovery mechanisms after ICH.8-12 Many reviews13-16 also focus on ischemia, assuming perhaps that the mechanisms and timing of repair processes and the effectiveness of rehabilitation strategies are similar between stroke types—an unproven assumption.
Compared with ischemic stroke, it is rare for clinical studies to specifically target ICH patients either to test efficacy or mechanisms of action of a rehabilitation treatment. 17 However, several clinical studies have compared recovery after ischemic and hemorrhagic stroke with mixed results,1,3,18 which has also been recently evaluated in a rat model of ICH. 19 A greater number of animal studies have proven that rehabilitation promotes behavioral recovery after ICH,9,10,20,21 although such therapies are not always effective.22,23 As with ischemia,13,24 rehabilitation has been shown to promote growth of neuronal dendrites10,11,25 and astrocyte processes and density 9 following ICH. As in some situations with ischemic stroke, 26 rehabilitation can mitigate tissue damage when initiated either early or later after ICH.20,21,27 For example, DeBow and colleagues 20 were the first to show that forced use therapy started 1 week after ICH significantly lessened the final volume of brain damage in a rat ICH model. The mechanisms of this remarkable neuroprotective effect are not known. However, given that rehabilitation was given after 1 week, it makes sense to evaluate how rehabilitation affects pathophysiological mechanisms contributing to chronic cell death.
Unlike ischemia, a significant amount of damage after ICH results from mechanical trauma as blood dissects through tissue. This occurs rapidly, whereas secondary damage occurs over days, even weeks. For instance, injecting collagenase into the brain, which damages the basal lamina of blood vessels, causing a spontaneous bleed, leads to both rapid and chronic cell death.28,29 Indeed, in our lab, this model causes significant tissue loss occurring beyond a week post-ICH.8,30,31 Secondary damage results from many factors, including iron toxicity and excessive inflammation.32-34 Elevated “free” iron in the brain, originating from degrading erythrocytes, promotes free radical formation (eg, OH·) by Fenton reactions, thereby damaging proteins, lipids, and DNA.32,34-37 Along with these findings, chelator studies also strongly suggest that iron contributes to a considerable portion of secondary degeneration.32,38,39 Furthermore, injecting iron directly into the brain mimics many features of ICH (eg, edema and rapid cell death), including the occurrence of protracted cell death.38,40,41 Thus, iron toxicity is widely accepted to cause acute and chronic cell death.
Iron toxicity does not go unchecked. After ICH, there is a substantial and prolonged increase in iron-binding proteins such as ferritin (iron storage) and transferrin (iron transfer).32,42 Ferritin plays an important role in iron homeostasis through its 2 subunits: ferritin heavy chain (iron use within cells) and ferritin light chain (iron storage). 32 Transfer of iron within or outside the cell is a function of transferrin, and thus, it plays an important role in iron clearance. Furthermore, inflammatory cells such as microglia and macrophages are essential for neutralizing iron and clearing debris.32,33 Unfortunately, these defenses are overwhelmed after ICH. Thus, a more complete understanding of these and other processes are needed to develop effective neuroprotective strategies and perhaps also for optimizing and understanding rehabilitation therapies.
Here, we evaluated 2 general objectives: (1) whether rehabilitation lessens perihematoma neuronal death, and (2) whether changes in iron levels, iron-binding proteins, or inflammation accounts for the neuroprotective effects of rehabilitation after ICH. We used the rat collagenase model of ICH because it causes spontaneous bleeding from multiple vessels, which has high face validity. As discussed, this model results in a well-defined profile of chronic cell death, behavioral impairment (walking and reaching deficits after striatal ICH), and responsiveness to rehabilitation. Enriched rehabilitation (ER), which consists of environmental enrichment and a skilled reaching task, 24 was used because it improves functional recovery and has been shown to decrease tissue loss after ICH, 27 as does a similar therapy. 20 A 1-week delay was used to mimic the delays encountered clinically (vs ultraearly interventions commonly used in animal research). We evaluated whether ER affected nonheme iron levels 32 days after ICH (11 days after the 2-week ER treatment). Similarly, the ongoing neuronal degeneration, iron-positive cells, lesion volume, astrocytes, and microglia were also evaluated 14 days post-ICH (ie, after 1 week of ER). Finally, protein expressions of ferritin H and L chain and transferrin were assessed 14 days post-ICH (ie, after 1 week of ER).
Materials and Methods
Animals
All procedures followed the Canadian Council of Animal Care guidelines and were approved by the Animal Care and Use Committee for Biosciences at the University of Alberta. A total of 84 male Sprague-Dawley rats weighing 200 to 225 g (~7 weeks old) were randomly assigned to control or ER treatment in 3 experiments. Rats survived for 32, 14, and 14 days post-ICH in experiment 1 (n = 16 each), 2 (n = 16 in control, n = 20 in ER), and 3 (n = 8 each), respectively. Rats were kept in a temperature- and humidity-controlled room under diurnal light cycles (on 7
Skilled Reaching (Experiment 1)
The staircase test, 43 which measures skilled reaching for sugar pellets (dextrose and fructose, 45 mg, Bio-Serv, Frenchtown, NJ), was used because it is highly sensitive to striatal ICH.20,44 Rats were trained over 4 weeks before ICH (two 15-minute sessions per day, 5 d/wk). They were tested on day 6 and on days 28 to 32 post-ICH. The rats that did not reach an average of ≥8 pellets per session over the final 3 days of baseline testing were excluded. Rats were food deprived to ~90% of their free feeding during these times, and their weight was slowly increased to accommodate natural growth. Note that behavioral training was conducted for experiments 2 and 3 in order to maintain procedural consistency with experiment 1. However, because there was no posttreatment assessment, which was a result of the timing of euthanasia, we did not formally evaluate the baseline data for the staircase or ladder other than to confirm adequate reaching ability and handedness.
Walking Ability (Experiment 1)
The horizontal ladder test, 45 which is sensitive to striatal ICH,20,44 measures a rat’s ability to traverse a 1-m long horizontal ladder with rungs randomly spaced 1 to 4 cm apart (3 mm diameter). Walking ability was assessed for 2 days (4 crosses/d) and videorecorded 2 days prior to ICH (all experiments) and on 6 (all experiments) and 32 days (experiment 1 only) post-ICH. Percentage success was calculated as (Successful steps/Total steps) × 100 (total of all ladder traverses in a day).
Intracerebral Hemorrhage
The collagenase model of ICH was used 28 as has been repeatedly done before.20,27,30,44 Rats were anesthetized with isoflurane (4% induction, 2% maintenance, 60% N2O, remainder O2) for aseptic stereotaxic surgery. Body temperature was maintained between 36.5°C and 37.5°C by a rectal temperature probe and a warm water blanket. A hole was drilled at 3.5 mm lateral and 1.0 mm anterior to the Bregma on the side contralateral to the more successful paw in the staircase test (number of pellets retrieved at the end of training). Overall, 45% of rats included in this study had ICH on the left side. A 0.7-µL solution of sterile saline containing 0.14 U of collagenase (Type IV-S, Sigma-Aldrich, Oakville, ON, Canada) was infused over 5 minutes through a 26-gauge needle (Hamilton syringe; Hamilton, Reno, NV) at a depth 6.5 mm below the skull. The needle was removed after 10 minutes to avoid back flow. A small metal screw was inserted in the hole, bupivacaine was applied, and the wound was closed.
Enriched Rehabilitation
The ER treatment, initially developed for ischemic stroke,
24
started 7 days after ICH and lasted for 14 (experiment 1) or 7 days (experiments 2 and 3). The latter time was because animals were killed humanely 14 days post-ICH, a time we expected delayed cell death to occur based on our experience with this model.8,30,31 The treatment consisted of reach training in a modified staircase apparatus and environmental enrichment housing for 5 d/wk consecutively.10,25,27 There were four 15-minute reach training sessions (2-hour intervals) during the day. Essentially, rats were allowed unlimited access to sugar pellets (45 mg size) located in 1 large well on the side of their impaired limb. The amount of sugar pellets obtained was recorded for each session, so that controls could receive the same amount in their home cages. Enrichment cages (width = 35 cm, length = 75 cm, height = 75 cm) contained ramps, tunnels, and various objects (eg, toys changed twice weekly) and were used from 5:30
Nonheme Iron Assay (Experiment 1)
Rats were killed humanely 32 days after ICH to measure the total amount of ipsilateral- and contralateral-to-ICH (2 mm anterior to and 4 mm posterior from the site of collagenase injection) nonheme iron using a Ferrozine-based colorimetric assay that used freshly prepared standard curves as previously described.31,46 Cerebellum and liver samples were also analyzed as controls.
Histology (Experiment 2)
At 14 days post-ICH, the rats were administered pentobarbital (100 mg/kg IP) and perfused transcardially with phosphate-buffered saline (PBS), followed by 4% paraformaldehyde. Brains were fixed overnight and then transferred to 30% sucrose in PBS. We took 40-µm coronal sections to determine lesion volume (cresyl violet), perihematoma cell death (Fluoro-Jade B), Fe3+ accumulation (Perls), and astrocyte (GFAP) and microglia activation (Iba1). The volume of tissue lost in serial sections (every 400 µm) extending through and beyond the lesion and including overt tissue damage and atrophy (eg, ventriculomegaly) was calculated as routinely done20,30,44:
The number of Fluoro-jade B positive (FJ+, degenerating) neurons 47 was determined in 3 coronal sections (200 µm apart) around the level of maximal injury. 40 Owing to the very large number of Perls-positive cells, we counted these in 1 coronal section at the level of maximum injury. Perls largely identifies ferric iron. 48 Although the number of these cells can be counted, the amount of iron per cell cannot be accurately determined with this method. Thus, counting Perls-positive cells does not replace quantitative methodology, such as that used in experiment 1.
For immunofluorescence, the slides were boiled in 0.1 mol/L citrate buffer (pH 6.3) for 15 minutes in a microwave (ie, improved antigen retrieval). After cooling the slides for 10 minutes, the tissue was blocked for 1 hour at room temperature in PBS-T (0.025% Triton X-100 in PBS) with 10% horse serum. The slides were incubated overnight in the following primary antibodies: mouse anti GFAP (1:400, Sigma, Canada) and rabbit anti-Iba1 (1:1000, Wako, Richmond, VA). The slides were subjected to three 5-minute washes using PBS and PBS-T, followed by 1 hour of incubation with 1:1000 dilution Alexa 488-conjugated donkey anti-mouse and goat anti-rabbit antibody (Abcam, USA). The slides were washed again and then cover slipped. One section per brain was counted at 20× for both Iba-1 and GFAP. 49 The microglia were counted based on their morphology as ramified, activated, and macrophage like (method adopted from Wasserman et al 49 ).
Western Blot (Experiment 3)
Rats were killed humanely as above and perfused with saline. Brains were quickly removed and frozen (−80°C). Subsequently, they were dissected into ipsilateral (containing remnants of the ICH) and contralateral striatum (−2 to +4 mm to injection site). These samples were homogenized quickly in 1× RIPA buffer containing protease inhibitors. Protein concentration was determined using the Bradford assay. We used 8% and 12% (w/v) acrylamide gels for transferrin and Ferritin H/L chain, respectively. 42 Total proteins amounting to 50 µg was loaded in each well during SDS PAGE electrophoresis. The proteins were electrotransferred onto nitrocellulose membranes and then blocked using Tris-buffered saline (TBS) with tween 20 in 5% nonfat milk (TBS-T) for 1 hour. The membranes were incubated overnight at 4°C with gentle rocking in the presence of mouse antiferritin H chain, antiferritin L chain, antitransferrin, or β antiactin at 1:1000 dilution in TBS-T. 42 After successive washes with TBS and TBS-T, the membranes were incubated with a 1:2000 dilution of the appropriate HRP-conjugated secondary antibodies in TBS-T for 1 hour at room temperature and further washed with TBS and TBS-T. Proteins were detected using Western Lightning chemiluminescence reagent plus (Invitrogen, Canada) and visualized using a Kodak Image Station 440CF (Kodak, NY). 50 Quantitative densitometry analyses were performed on 2 samples per region per animal (averaged) using Scion Image J (4.0; Scion Corporation, Frederick, MD).
Statistical Procedures
Surgical procedures were done blinded to group identity. It was not possible to be blinded to treatment administration. However, behavioral and histological assessments were done blinded.
The data are presented as mean ± standard deviation and were analyzed by repeated-measures and/or 1-way ANOVA using SPSS (SPSS v 17.0, SPSS Inc, Chicago, IL).
Results
Exclusions
In experiment 1, 10 rats were excluded (n = 5 in each group) because of a technical error. Two rats in experiment 2 were excluded in the ER group because they did not reach the baseline criteria in staircase training. One control rat was excluded from experiment 3 because of a technical error.
Experiment 1
An analysis of reaching success with the contralateral-to-ICH forelimb in the staircase test showed significant Day (P < .001) and Group (P = .007) main effects with a significant interaction (P = .001; Figure1A). Groups were not different prior to ICH (P = .753) or 6 days after ICH (P = .760), but the ICH did cause significant impairment (ie, day 6 scores lower than baseline, P < .001). As predicted, ER rats retrieved almost twice as many pellets at 32 days post-ICH (P = .003).
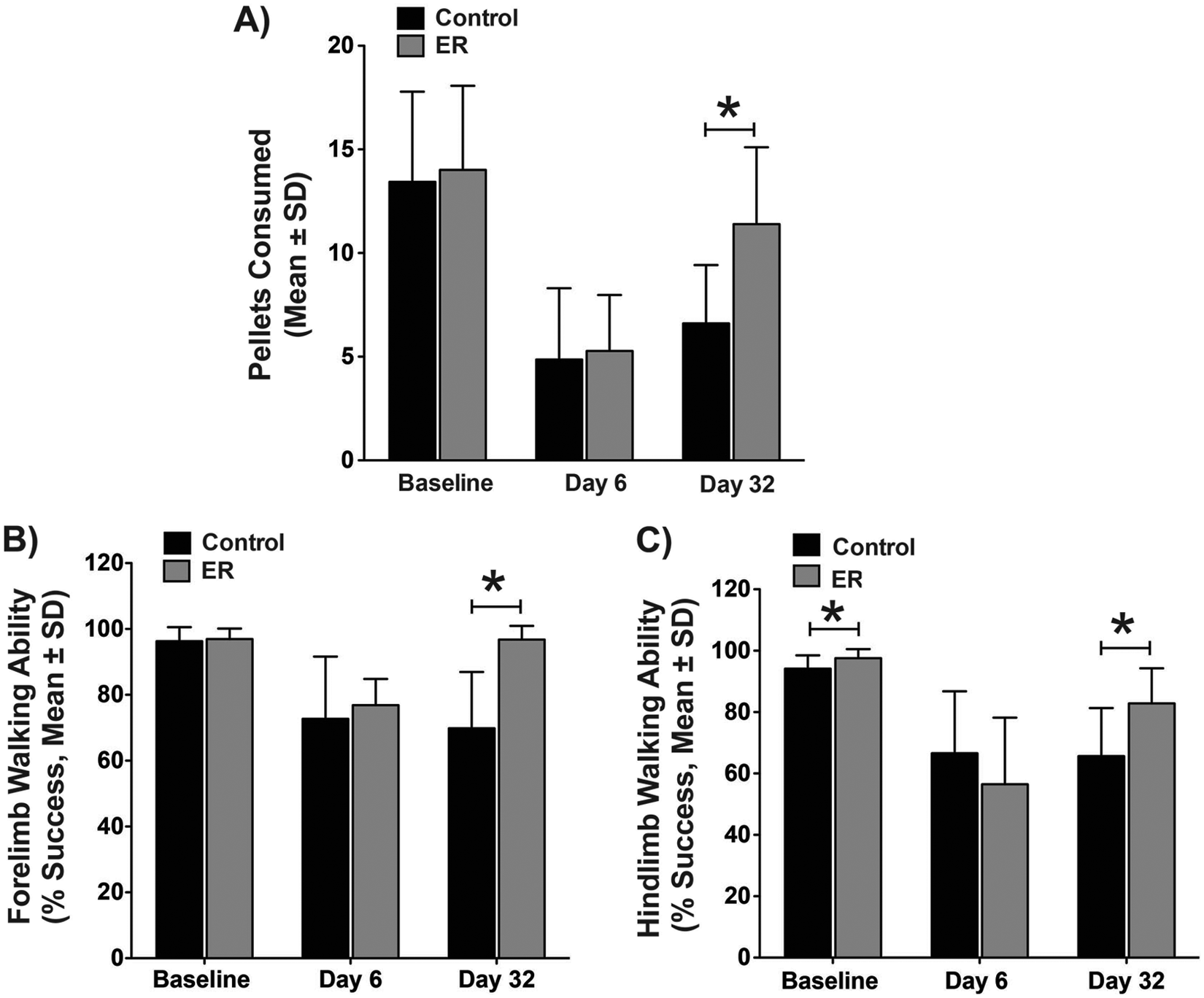
A striatal ICH caused significant skilled reaching (A) and walking impairments (B, C), which were significantly ameliorated by ER treatment. An asterisk denotes P < .05. There was a difference in baseline scores for the hind limb, but this did not meaningfully affect our findings as determined by an analysis on percentage baseline scores (not shown).
The contralateral-to-ICH forelimb (Figure 1B) stepping success was evaluated for the ladder task, revealing significant Day (P < .001) and Group (P = .004) main effects with a significant interaction (P < .001). Analysis of the contralateral-to-ICH hind limb stepping success (Figure 1C) revealed a significant Day main effect (P < .001) and interaction (P = .007) with a nonsignificant Group effect (P = .406). Owing to significant interactions, comparisons were made at each time for both limbs. At baseline, the groups had similar forelimb stepping success (P = .677), whereas the hind limb was statistically different (P = .045). There were no significant differences on day 6 after ICH for both forelimbs (P = .510) and hind limbs (P = .271). The ER treatment significantly improved forelimb (P < .001) and hind limb walking abilities (P = .008) at 32 days post-ICH. Owing to the statistically significant baseline hind limb difference, we also expressed postoperative data as percentage baseline, but this did not meaningfully change our results, which showed no difference between groups on day 6 (P = .190), whereas the ER group were significantly better on day 32 (P = .031, data not shown).
The ICH hemisphere had approximately twice as much nonheme iron (Figure 2) as the normal side (P < .001), but there was no effect of ER, including in the cerebellum (P = .450) or liver controls (not shown, average value ~107 µg of iron per gram of brain; P = .773).
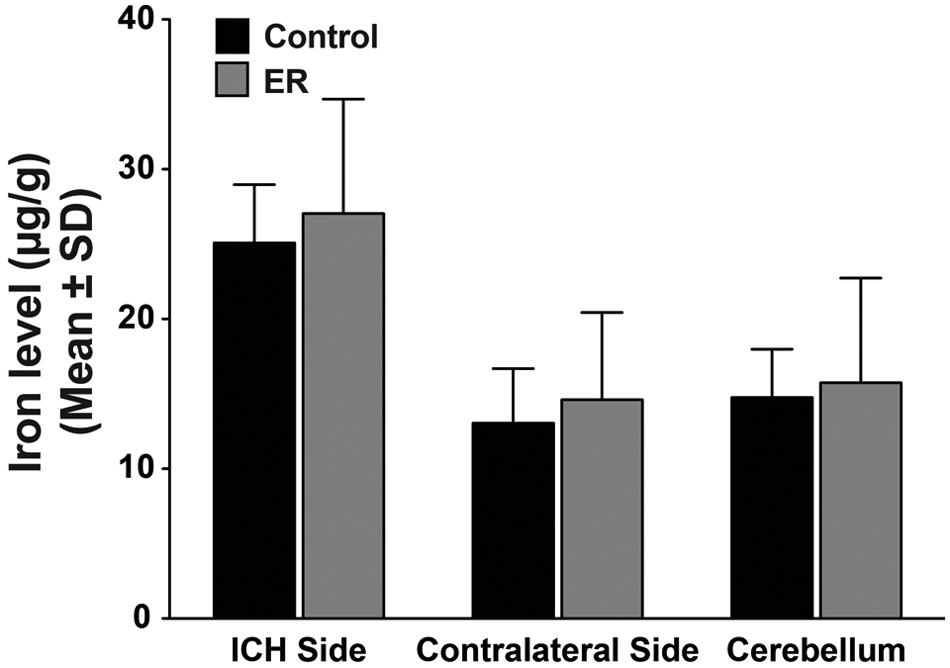
Nonheme iron levels were significantly higher in the injured hemisphere at 32 days after ICH (vs the contralateral side, P < .001). The ER treatment had no impact on any region sampled.
Experiment 2
There were many FJ+ (recently dead) neurons at 14 days post-ICH. The ER treatment significantly reduced their numbers (P = .040; Figures 3A-3C). The total volume of tissue loss was not significantly different at this time (P = .394; Figures 3D and 3E).
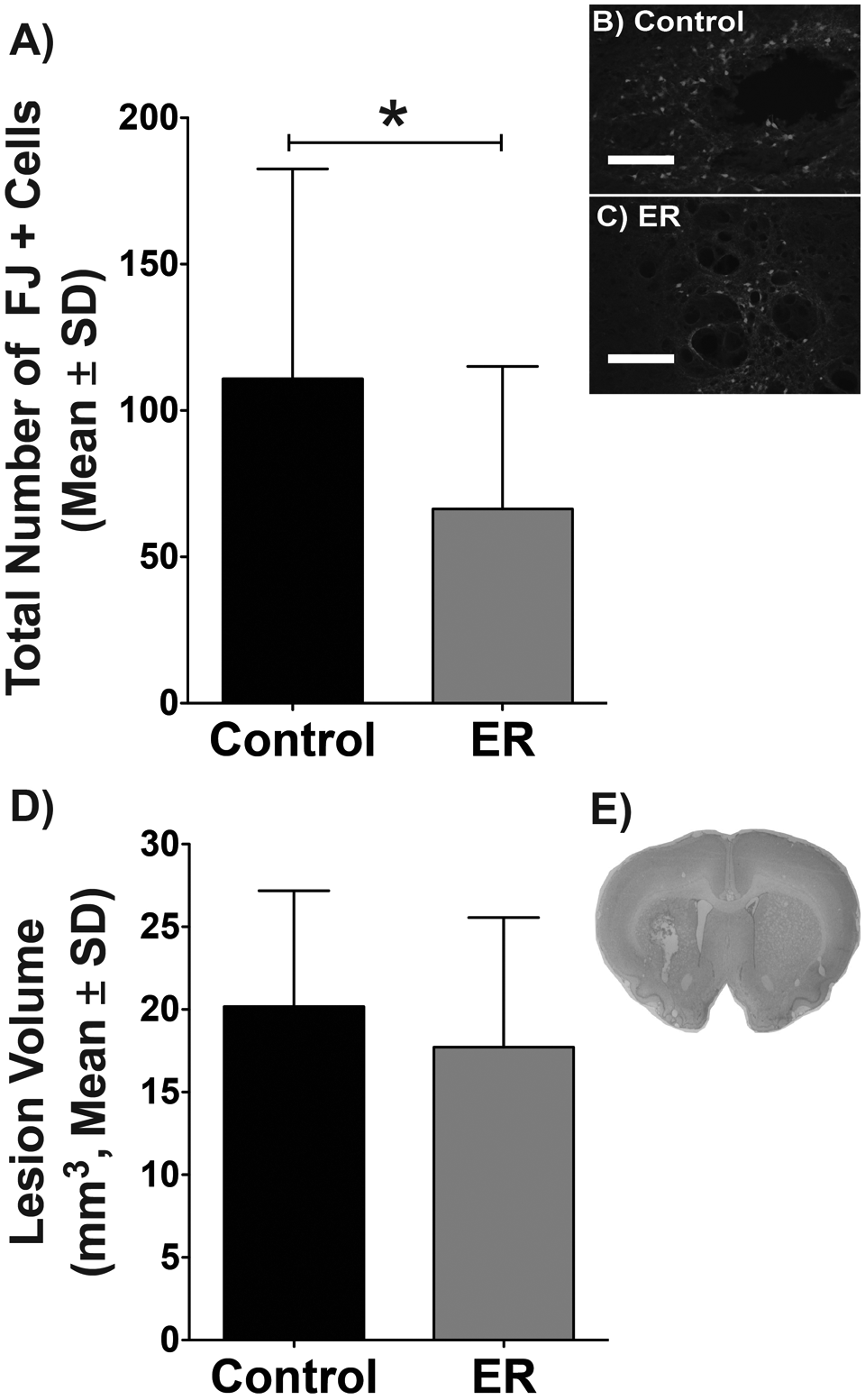
The ER group had significantly fewer degenerating neurons (Fluoro-jade positive, FJ+) in the perihematoma region than controls at 14 days post-ICH (A, *P = .040). Representative photomicrographs are shown in (B) and (C); scale bar = 500 µm. The volume of tissue loss was not different (D). A photomicrograph of the typical injury (E) illustrates the residual cavity and ventriculomegaly caused by the ICH 14 days earlier.
All rats exhibited numerous Perls-positive cells (presumptive macrophages and microglia) at 14 days post-ICH (Figure 4A), which was not affected by ER (P = .474; Figure 4B). Similarly, the number of GFAP-positive astrocytes were obviously increased in the perihematoma region (astrogliosis) but not affected by ER (P = .214, Figures 5A-5C). Finally, the numbers of ramified, activated, and macrophage-like microglia were substantial in the perihematoma/cavity region but not different between groups (P ≥ .351, Figures 5D-5H).
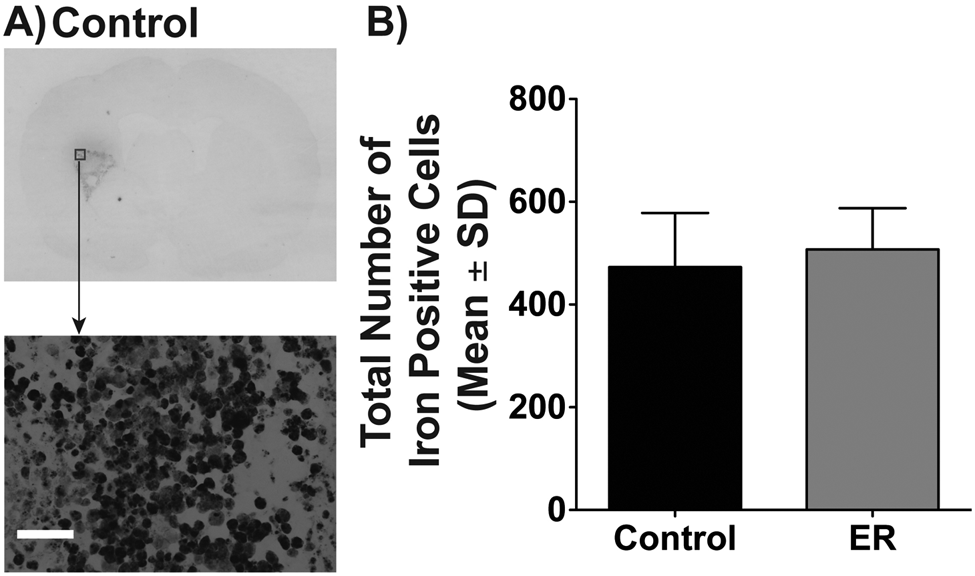
Representative photomicrographs of a control rat showing Perls staining, which largely identifies ferric iron loaded in macrophages and microglia (A); scale bar = 50 µm. Quantification showed no significant difference between groups (B).
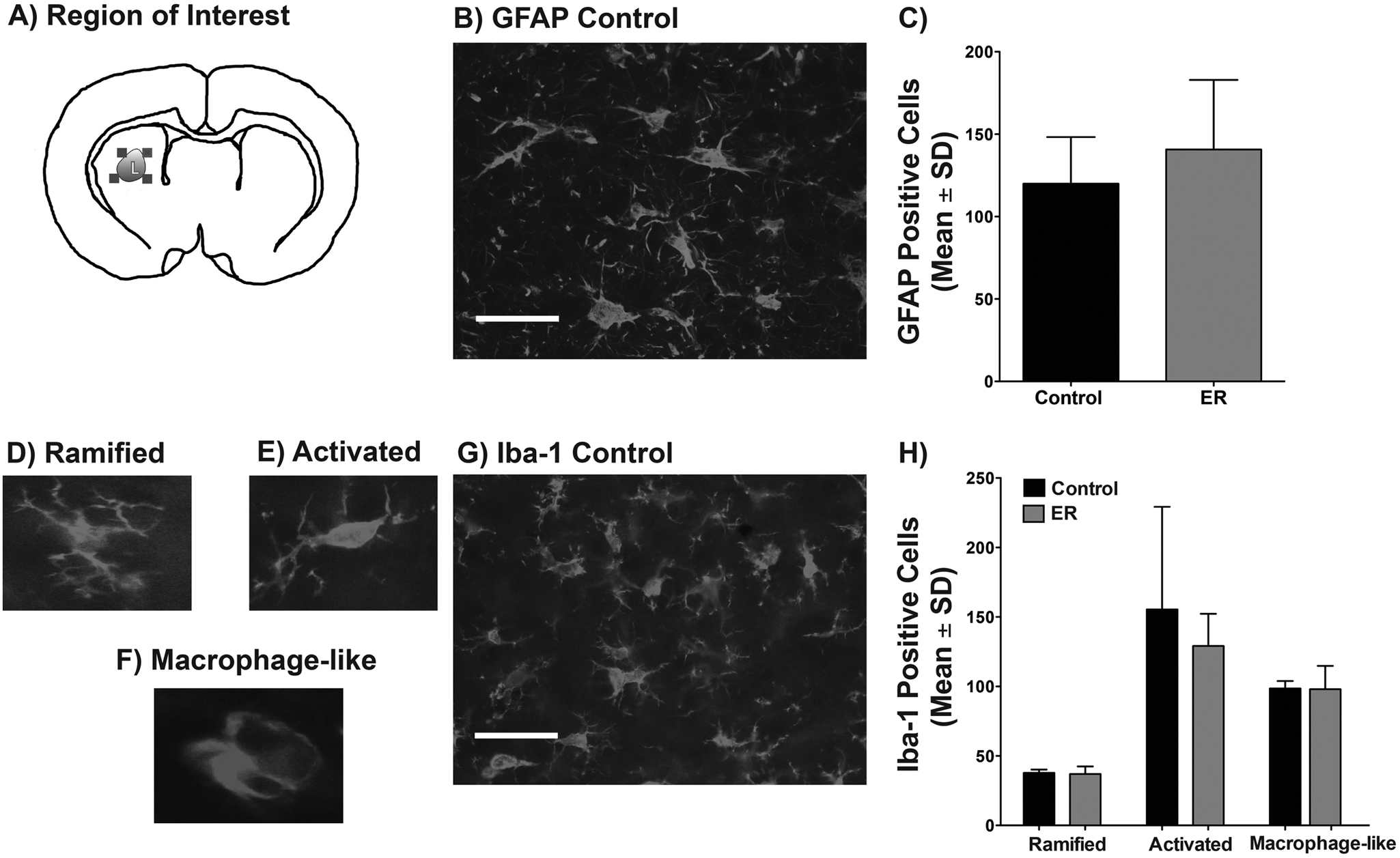
We counted GFAP and Iba-1 labeled cells in the perihematoma region (A). Labeled astrocytes from the control group are shown in (B); scale bar = 50 µm. Quantification showed no group difference (C). The different stages of macrophages are illustrated in (D), (E), and (F) along with a representative photomicrograph of a control animal in (G); scale bar = 50 µm. There were no group differences in the number of these cells (H). Histological controls (eg, omitting primary antibody) were negative.
Experiment 3
The expressions of ferritin H and L chain and transferrin in the ICH hemisphere were significantly higher than in the contralateral hemisphere (P < .001), but the groups were not significantly different (P ≥ .174; Figure 6).
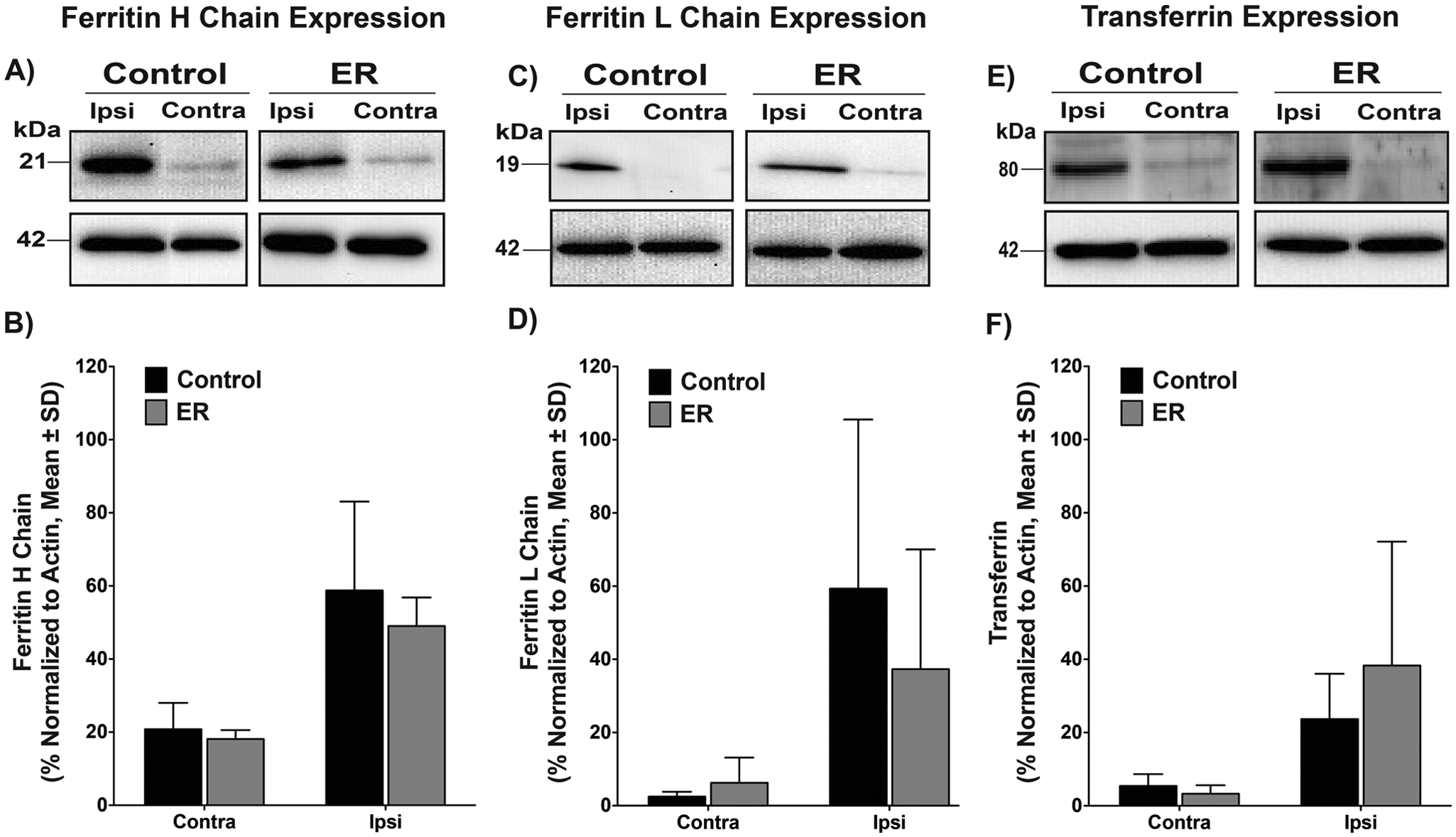
Representative Western blots showing the expression of iron handling proteins: ferritin H-chain (A), ferritin L-chain (C), and transferrin (E) at 14 days post-ICH. Data were normalized to actin (B, D, and F), showing significant differences between injured and contralateral hemispheres (P < .001) but no significant effect of ER treatment.
Discussion
ER training substantially improved recovery of walking and skilled reaching following a striatal ICH of moderate severity. Benefit occurred despite using only a 2-week regimen given after a 1-week delay. Furthermore, chronic neuronal death in the perihematoma striatum was found, and it was significantly attenuated with only 1 week of treatment. Thus, a modest rehabilitation therapy rescues many injured perihematoma neurons. Our study is the first to evaluate whether rehabilitation mitigates injury through affecting iron and inflammation. Although these are known effectors of perihematoma neuronal death, our results suggest that they are not involved in the neuroprotective effects of rehabilitation, but they may be potential targets to further augment behavioral therapies.
Compared with ischemia, there are relatively few studies examining the effectiveness of rehabilitation treatments for ICH. Furthermore, the diversity among therapies (type, timing, and intensity) and models makes it difficult to identify those treatments that work best. Perhaps it is telling that in our lab, forced-use therapies with and without environmental enrichment work well,10,20,27 whereas simple exercises (running) or enrichment alone are less effective.22,51 Others have also reported that skilled training is more effective than unskilled training after ICH. 9 Furthermore, the present findings confirm the remarkable efficacy of this ER regimen after ICH,10,25,27 which is based on the seminal work of Biernaskie and Corbett 24 in ischemic stroke. Further work is needed to determine if the mechanisms of action are similar. In this regard, it is clear that ER and other rehabilitation therapies increase dendritic complexity after ischemia,13,24 and this is true for ICH too.10,11,25 However, delayed ER does not appear to be neuroprotective after ischemia, unlike for ICH. Certainly, this relates to differences between these stroke types with respect to the timing, extent, and underlying mechanisms of chronic cell death, which clearly occurs after collagenase-induced ICH8,30,31 (present data).
Our findings show that the number of degenerating perihematoma neurons, which we had not previously evaluated,20,27 was significantly reduced after 1 week of ER treatment (2 weeks post-ICH), but this did not lead to a significant reduction in lesion volume at that time. Although these findings appear contradictory, we suspect that it simply takes more time and perhaps more therapy (eg, the second week of our standard ER treatment) before there is a discernible reduction in the volume of tissue loss. Specifically, in controls there would be ongoing injury (lesion expansion) beyond 1 or 2 weeks, as we have repeatedly found after collagenase-induced ICH.8,30,31 At the same time, ER treatment would attenuate this progressive injury, thereby culminating in a long-term reduction in the volume of injury.20,27 Thus, it would take weeks to notice the cumulative effects of reducing perihematoma damage. Of course, other effects might also affect the volume of tissue loss (eg, ER promotes dendritic growth). Further work should evaluate whether rehabilitation also prevents cell death beyond the end of treatment and, if not, whether extending the duration of treatment would provide greater protection. An additional unresolved issue concerns the contribution of these salvaged neurons to functional recovery. Unfortunately, there is no obvious way to causally link neuroprotection from ER to improved behavioral recovery after ICH. The fact that ER also improves recovery in the whole blood model of ICH, 25 which does not result in as much chronic cell death, 30 suggests that behavioral benefit is not solely dependent on neuroprotection. Regardless, we have observed an approximately 30% reduction in tissue loss after collagenase-induced ICH from rehabilitation at long survival times,20,27 and it is difficult to argue that this would not be of behavioral significance. One must consider too that neuroprotective treatments rarely achieve this in any ICH model, let alone when the drug treatment is delayed for a week following ICH.
Nonheme iron levels were substantially higher in the injured hemisphere at 1 month following collagenase infusion. This is in line with our previous findings in this model, which used both a Ferrozine-based method and X ray fluorescence imaging, 31 and with studies using the whole blood model. 42 Our findings also parallel the whole blood model with regard to the accumulation of iron-positive cells (Perls stain), known to be mostly macrophages and microglia, and increases in ferritin and transferrin proteins. 42 In total, such findings point to a chronic degenerative/repair process. Rehabilitation had no discernible effect on any of these processes, thereby suggesting that ER is not mitigating iron load or the removal of degenerating erythrocytes and damaged brain tissue. However, several additional experiments are needed to confirm this. First, rehabilitation may influence oxidative stress by altering free radical defenses.52,53 Second, whereas ER did not influence the number or morphology of microglia, or reactive astrogliosis, it is possible that ER influenced the activity of these cells. Third, we did not examine multiple survival times following ICH; without this, it is possible that some effects were missed. Thus, ER may alter the rate of hematoma clearance or iron detoxification, but we expect that this is more likely following earlier interventions. We are currently examining these hypotheses. Along with this, ER likely has numerous other beneficial effects (eg, changes in growth factors) that aid in recovery and/or neuroprotection.
The collagenase and whole blood models have a number of similarities as well as important differences.5,7 Whereas it appears that key mechanisms of injury, such as iron toxicity and inflammation, are at play in each model, it is quite likely that their contribution varies by model and the fact that lesions are larger with collagenase than with a whole blood infusion of comparable hematoma size. 30 Neuroprotective treatments, therefore, may not be equally effective. Indeed, iron chelators, such as deferoxamine, appear to be more effective against whole blood1,38,39 than collagenase-induced ICH.54,55 The same argument regarding differential mechanistic and efficacy effects may be made for rehabilitation. Thus, until additional data are available to better validate these models, it makes sense to test treatments in both models as we have been doing. Additional studies are also needed to vary insult location and size along with other important variables, such as age, that are known to influence outcome.49,56
In summary, ER substantially improved functional recovery after striatal ICH. The fact that chronic cell death was observed and that ER attenuated this suggests that clinical rehabilitation therapies, at least when properly timed and at an appropriate dose, may be neuroprotective in humans suffering from ICH. Our study shows that rehabilitation at least does not markedly change 2 key mechanisms of injury: iron toxicity and inflammation. This suggests that cotreating against these injurious processes coupled with rehabilitation might be additively or synergistically advantageous. Finally, additional studies are needed to identify how rehabilitation improves recovery and reduces perihematoma injury after ICH. This information is needed to rationally devise more effective treatments that are especially needed in more challenging cases (eg, older individuals, greater intervention delays, and more severe stroke).
Footnotes
Acknowledgements
The authors thank Shannon Wowk, Yonglie Ma, Sampath Kumar Loganathan, and Gonzalo L. Vilas for technical assistance. The authors also thank Dr Joseph R. Casey for use of a Kodak Image Station.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Heart and Stroke Foundation of Canada supported this work. Alberta Innovates Health Solutions also provided a Senior Medical Scholar award.


