Abstract
Background. Intracerebral hemorrhage (ICH) accounts for 10–20% of all strokes and is associated with high morbidity and mortality. Recent studies have identified serum biomarkers as a means to improve outcome prognostication in poor grade ICH patients. Poor prognosis of ICH patients and complex pathophysiology of the disease necessitate prognostic serum biomarkers to help guide treatment recommendations. Objective. The objective is to systematically review all biomarkers used to predict long-term functional outcome in patients with spontaneous intracerebral hemorrhage. Results. We identified 36 studies investigating the predictive utility of 50 discrete biomarkers. Data from 4865 ICH patients were reviewed. Inflammatory biomarkers (11/50) were most often studied, followed by oxidative (8/50), then neuron and astrocyte-specific (7/50). S100 calcium binding protein B, white blood cell count, and copeptin were the most often studied individual biomarkers. The prognostic utility of 23 biomarkers was analyzed using receiver operating characteristic curves. Area under the curve (AUC) values for all available biomarkers except neutrophil/lymphocyte ratio were acceptable. Twenty of the 23 biomarkers were characterized by at least one excellent AUC value. Vascular endothelial growth factor, glial fibrillary astrocyte protein, and S100 calcium binding protein B were characterized by outstanding AUC. Conclusions. We identified the inflammatory and neuron and astrocyte-specific biomarker categories as having the greatest number of significant individual biomarker predictors of long-term outcome. Further investigation utilizing cross-validation of prediction models in a second independent group and blinded assessment of outcomes for the predictive utility of biomarkers in patients with ICH is warranted.
Introduction
Intracerebral hemorrhage (ICH) accounts for 10%–30% of all strokes and is associated with high mortality and morbidity.1-4 Median 30-day mortality ranges from 15% to 50% 5 and mean functional dependency ranges from 12% to 39%. 6 Standard of care medical management includes close monitoring and treatment for blood pressure, seizures, increased intracranial pressure, and reversal of anticoagulant and antiplatelet medications. 7 Decompressive hemicraniectomy is used in severe cases of increased intracranial pressure, and minimally invasive surgical evacuation is under investigation as a promising surgical option.8-10 Treatment decisions and recovery depend on clinical characteristics including age, hematoma location, hematoma volume, and neurologic examination. 11
Recent studies have identified serum biomarkers as a means to improve outcome prognostication in poor grade ICH patients. 12 Some serum biomarkers have diagnostic and prognostic utility in other neurologic disease states as well, such as stroke and subarachnoid hemorrhage. 13 The application of these biomarkers can improve risk stratification and therefore guide implementation of tailored treatment modalities.
The poor prognosis of ICH patients and complex pathophysiology of the disease emphasize the need for prognostic serum biomarkers to help guide treatment recommendations. In this study, we systematically review the utility of serum biomarkers to predict long-term outcome after ICH.
Methods
We conducted a systematic review according to the Preferred Reporting Items with Systematic Reviews and MetaAnalyses (PRISMA) statement and Prospero (CRD42020176400).
Search Strategy and Selection Criteria
PubMed, Cochrane Library, and Google Scholar were screened using the terms “biomarkers predictive functional outcome intracranial (or intracerebral) hemorrhage” and “intracranial hemorrhage outcome biomarkers.” Queries were restricted to human studies investigating atraumatic spontaneous, supratentorial ICH of ≥30 patients in the English or Spanish language available as of December 31st, 2019. MeSH terms used in our queries were Biomarkers, intracerebral hemorrhage, and intracranial hemorrhage.
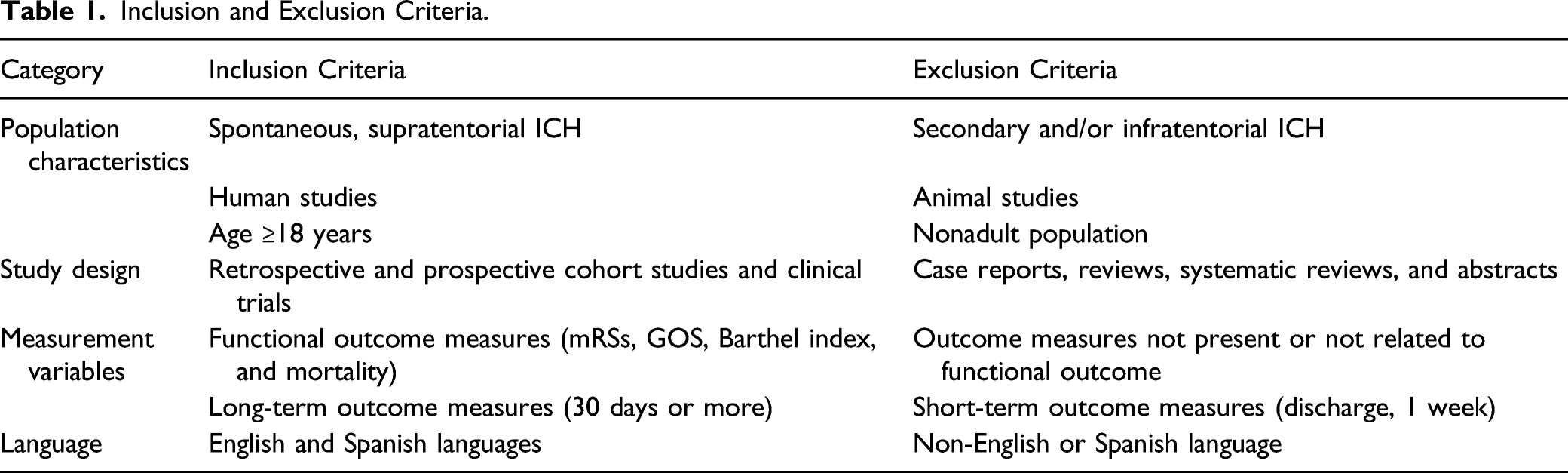
Inclusion and Exclusion Criteria
Inclusion and Exclusion Criteria.
Assessment of Study Quality
Two authors (NM and CR) independently assessed the 36 included studies using a modified version of the evidence methodological quality score adapted from previous systematic reviews on similar topics.14-18 Internal, statistical, and external validity criteria were scored 0 or 1, with maximum score of 14 (Supplementary Table 1). Studies where ≥80% of criteria were met were considered “high quality,” ≥ 60% but <80% were considered “moderate quality,” ≥ 50% but <60% were considered “low quality,” and <50% were considered “very low quality.” 19 Discrepancies between grading were resolved by a third author (MA).
Statistical Analysis
Descriptive statistics were used to analyze the data. Continuous data are reported as mean ± standard deviation. Categorical data are reported as proportions. We obtained the proportions of biomarkers investigated in each functional outcome group.
We pooled the AUC values of each biomarker category based on outcome used. AUC values were divided into 4 categories: Unreliable (AUC ≤.700), acceptable (.700 ≤ AUC <.800), excellent (.800 ≤ AUC <.900), and outstanding (AUC ≥.900). 20 Data were analyzed using Microsoft Excel, and Figures were constructed using Prism 8.3.
Results
Literature Search and Study Characteristics
The results of the electronic search and article selection process are shown in the PRISMA flow diagram (Supplementary Figure 1). In total, 502 articles were identified. Seventeen additional publications from associated clinical trials were manually added, comprising 519 total articles. After initial title and abstract screening, 79 articles were identified and subjected to a full text review. Forty-three studies failed to meet inclusion criteria due to population characteristics (18), study design (10), or outcomes measured (15). Thirty-six studies met the inclusion criteria and were included in our qualitative analysis. In aggregate, data from 4865 ICH patients were reviewed.
Of the 36 studies, 31 were prospective cohort studies, 4 were retrospective cohort studies, and 1 study type was unidentifiable. No clinical trials were identified. The mean study size was 135 ± 92 ICH patients ranging from 40 to 423 patients. Functional outcome was measured by mRS (28 studies), GOS (3 studies), MBI (3 studies), and mortality (28 studies). Regarding the studies using mRS as a functional outcome, 25 characterized a “poor outcome” with a score greater than 2, while 3 of the 28 studies characterized a “poor outcome” with a score greater than 3. Functional outcome was evaluated at 30 (6 studies), 90 (16 studies), 180 (12 studies), and 365 days (3 studies) (Figure 1A). Number of biomarkers (A) with the number of studies evaluating their respective biomarkers at each time point, (B) classified by category, and (C) measured by functional outcome type. (D) Frequency distribution of study quality assessment scores.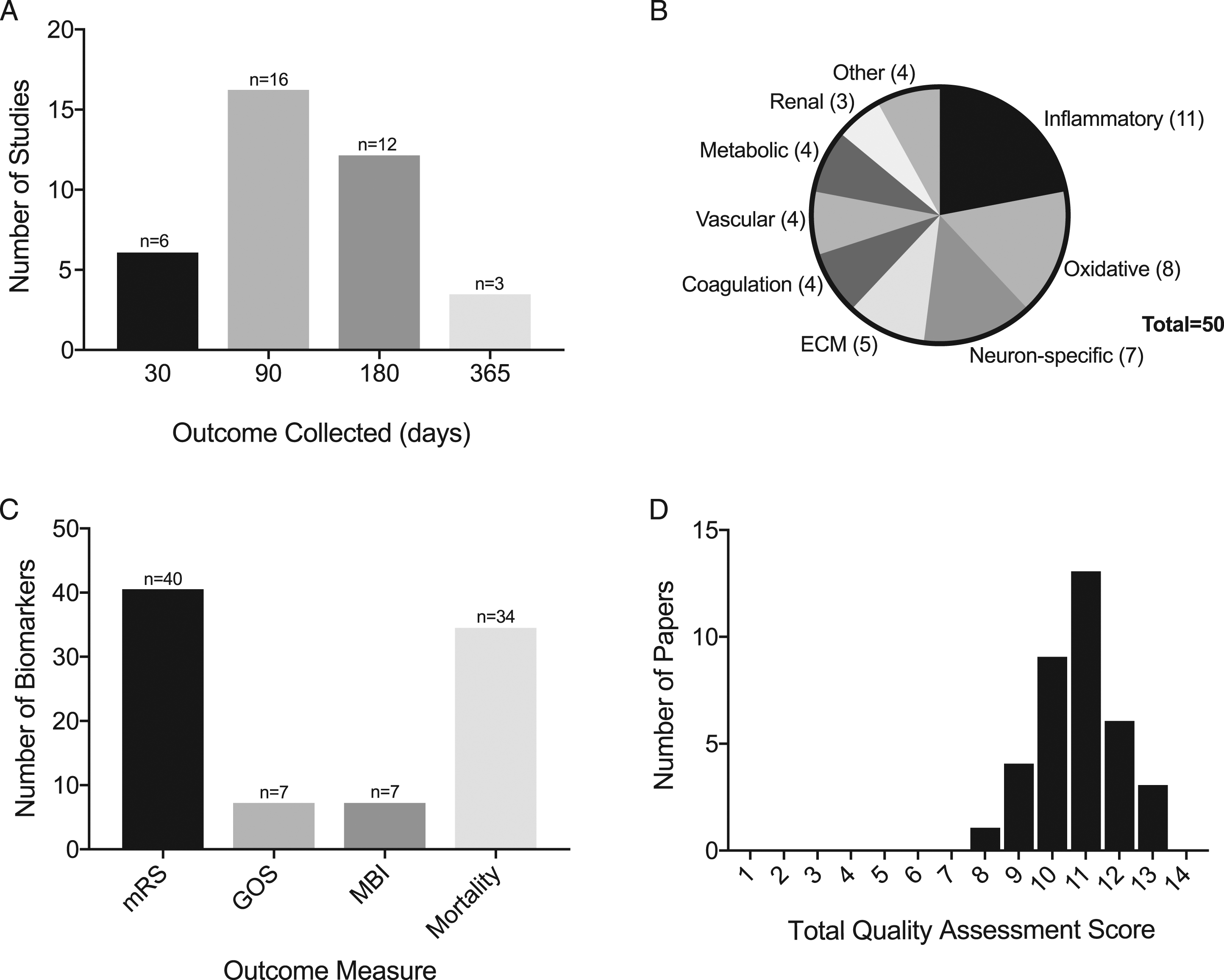
All 50 biomarkers were tested via blood except D-dimer which was tested via cerebrospinal fluid. Some studies investigated multiple biomarkers using more than one functional outcome measure at more than one time point. In this systematic review, no direct comparisons were made between studies due to the significant variability between data. 21
Biomarkers Categories
In total, we identified 50 different biomarkers studied related to functional outcome (Figure 2). The breakdown by type of biomarker is shown in Figure 1B. The inflammatory group accounted for the largest number of identified biomarkers (11/50, 22.0%), followed by the oxidative group (8/50, 16.0%), and the neuron-specific group (7/50, 14.0%). The most studied biomarkers (WBC, S100B, and copeptin) were each assessed in 3 studies. Stratified by outcomes, Figure 1C shows that biomarkers were most commonly investigated using mRS as the functional outcome (40/50, 80.0%) followed by mortality (34/50, 68.0%). A summary of the studies included is in Table 2. Anatomical schematic illustrating biomarker classification. Summary of Biomarkers and Associated Outcomes.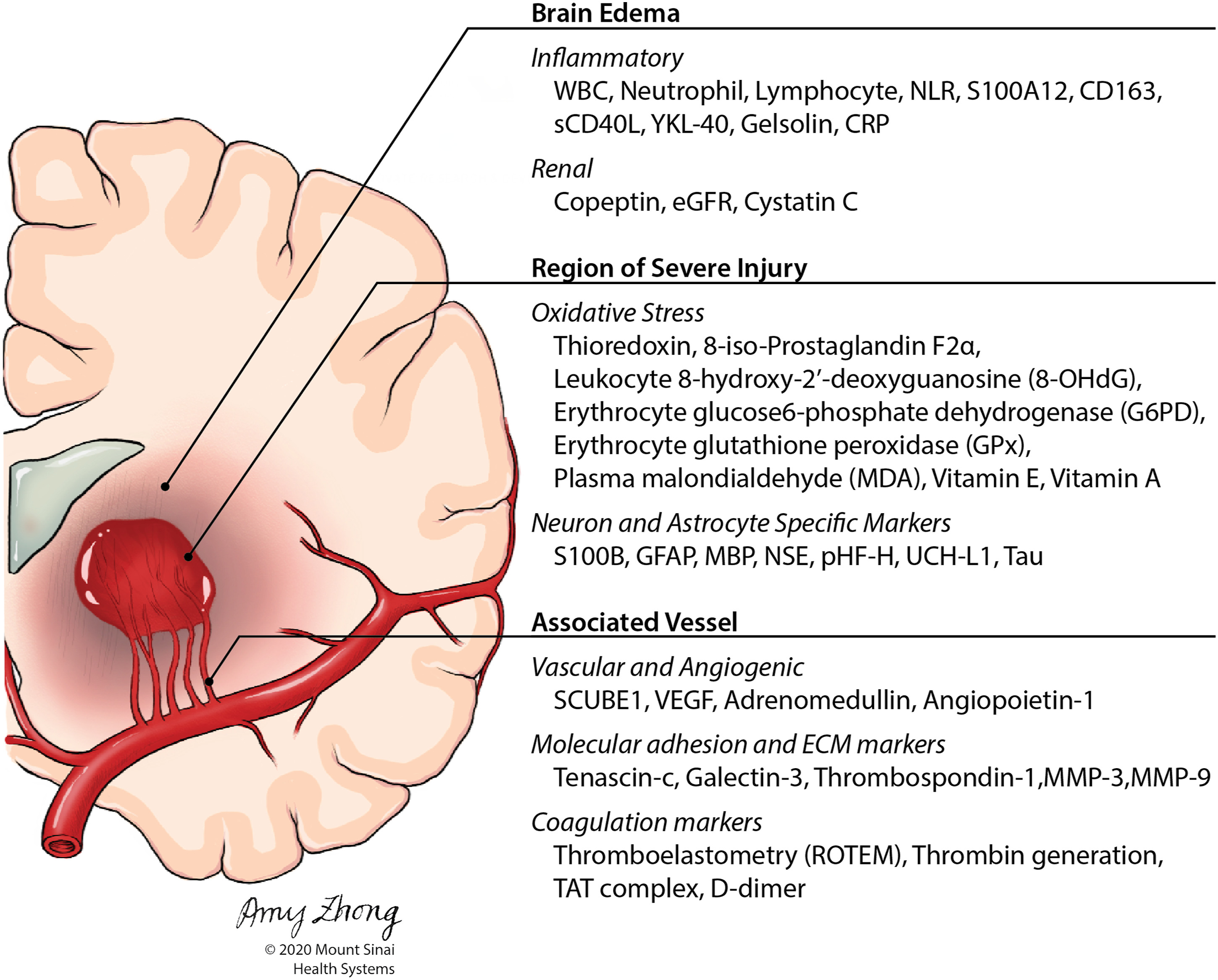

Study Quality Assessment Analysis
Summary of Quality Assessment Score Evaluation.
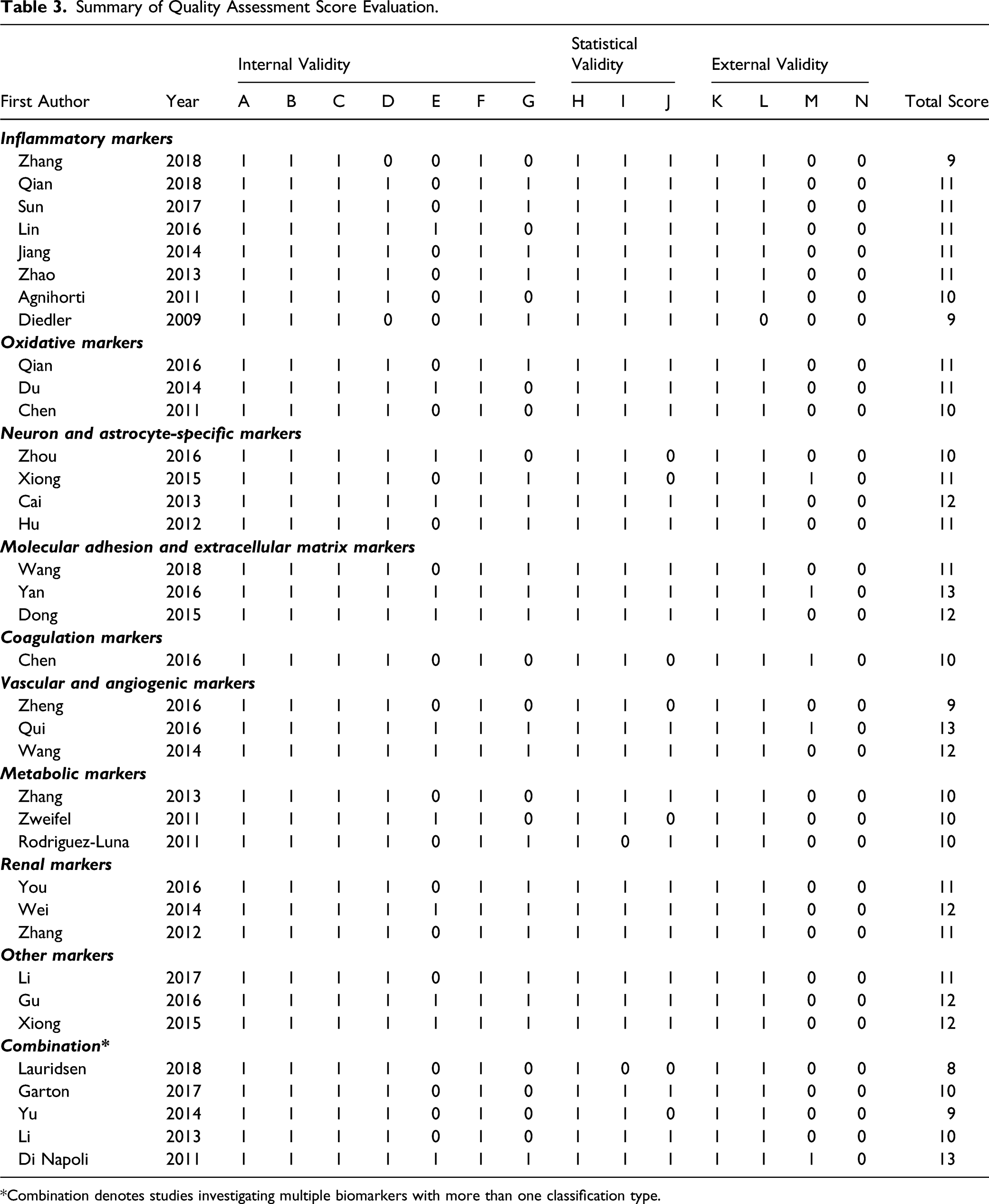
*Combination denotes studies investigating multiple biomarkers with more than one classification type.
Most studies properly validated predictor variables (94.4%), controlled for patient dropout in the statistical analyses (61.1%), and did not use blinded evaluators to assess functional outcome (63.9%). All other measures of internal validity were adequately met for all studies. Regarding statistical validity, most studies had an adequate sample size (94.4%) and tested the predictor variables for multicollinearity (80.6%). All other studies appropriately tested the relationship between predictor variables and outcome measures for statistical significance.
Regarding external validity, all studies specified relevant patient characteristics for patient selection and all but one study specified inclusion and exclusion criteria. Additional treatment effects on outcome measures were properly controlled for in only 5 studies (13.9%). No studies cross-validated their prediction model in a second independent group (Table 3).
Receiver Operating Characteristic Curve Analyses
Receiver Operating Characteristic Curve Analysis.
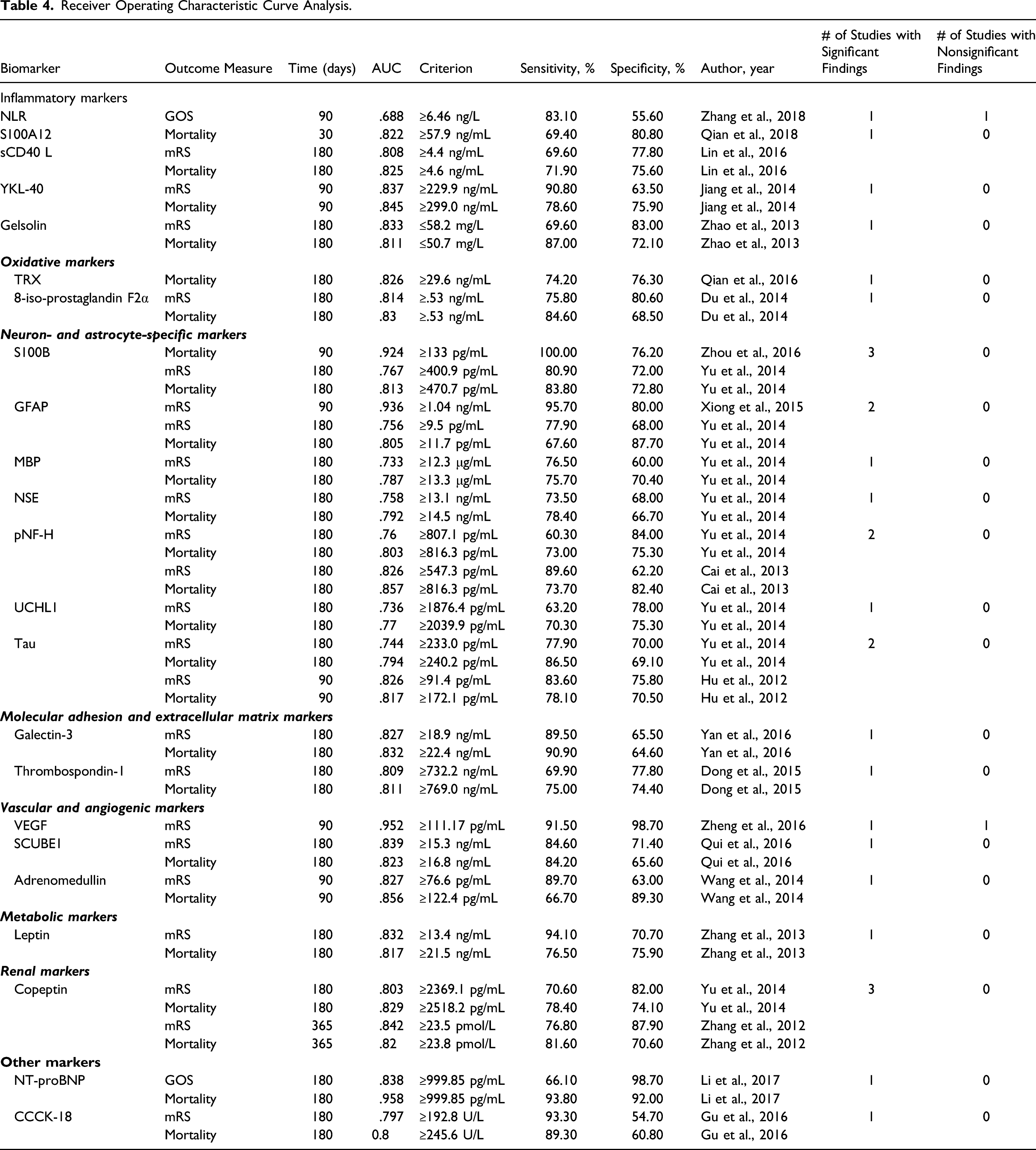
Discussion
Prognostic biomarkers for primary ICH have the potential to identify high-risk patients, guide treatment decisions, inform family members, and aid in clinical trial design. Currently, numerous biomarkers are under investigation. The most commonly investigated blood-based biomarkers for ICH prognosis are inflammatory, oxidative, and neuron and astrocyte-specific.23-25 This systematic review provides a comprehensive analysis of the available data on biomarkers in ICH. We found that inflammatory biomarkers were the most commonly studied and specifically white blood cell count (WBC), S100B, and copeptin were the most common individual biomarkers studied. All but one study was determined to be of at least moderate quality indicated by the QAS.
Inflammatory Biomarkers
Inflammatory changes in the perihematomal tissue contribute to secondary neuronal injury and poor functional outcomes following ICH. 26 Evidence suggests that leukocyte infiltration of adjacent parenchymal tissue elicits an immune response with a subsequent activation of microglia and cytokine release.27-30 Askenase et al 31 demonstrated that coordinated functional and metabolic reprogramming in CD14+ monocytes/macrophages occurs over time in the hematoma.
Inflammatory biomarkers were most commonly investigated (Figure 1B). We reviewed 4 studies investigating the predictive utility of 4 blood borne leukocytes: neutrophil, lymphocyte, monocyte, total WBC, and the neutrophil–lymphocyte ratio. The predictive utility of WBC count alone was investigated in 3 different studies but was not significantly associated with long-term functional outcomes in two of the studies.32,22 Other inflammatory biomarkers predicting long-term functional outcome in ICH include CRP,32,33 S100A12, 34 CD163, 35 sCD40 L, 36 YKL-40, 37 and Gelsolin (Table 2).37,38 Although inflammatory markers are non-neuron-specific, their substantiated predictive utility warrants further investigation.
Tumor necrosis factor-alpha (TNF-a) and interleukin-1 beta (IL-1b) are two well-known proinflammatory cytokines that were not included in this review. TNF-a and IL-1b are thought to exacerbate ICH-induced brain injury. While this has been shown in experimental and animal models, there is a paucity of human studies investigating these ubiquitous proinflammatory cytokines in the setting of primary ICH. 29
Neuron and Astrocyte-Specific Biomarkers
Astrocytes serve as the intermediary between the blood vessels and neurons. We identified 7 neuron-specific biomarkers which function as structural proteins, receptors, or enzymes, across 6 studies that increase following neuronal injury and blood–brain barrier disruption. Thus, increased levels are expected to be associated with poor long-term functional outcomes.25,39-43
Most biomarkers successfully predicted mortality at 90 or 180 days. Yu et al. demonstrated that the ability to predict outcome of several markers (NSE, MBP, GFAP, Tau, S100B, pNF-H, and UCH-L1) was comparable to the predicted capability of the NIHSS. However, none had an additive effect over the predictive capability of NIHSS. The AUCs of S100B and GFAP were outstanding (>.900) in one study each. These data suggest that neuron-specific biomarkers may accurately predict long-term functional outcomes.
Oxidative Biomarkers
Following ICH and subsequent hemolysis, oxidative stress secondary to hemoglobin toxicity and free radical generation causes brain tissue damage. 44 Of the 8 total oxidative markers investigated, increased levels of TRX, 8-iso-prostaglandin F2α, and 8-OHdG were significantly associated with poor long-term mRS and mortality. TRX and 8-iso-prostaglandin F2α were independent predictors of 180 mortality with excellent AUC values (Table 4).45,46 Although these two biomarkers had excellent AUC values, the other five oxidative biomarkers were not associated with long-term outcome. Additional studies investigating the predictive utility of oxidative molecules are needed.
Other Biomarkers
One of the most commonly studied biomarkers was copeptin, a surrogate marker for antidiuretic hormone (ADH). ADH levels are increased in syndrome of inappropriate ADH secretion (SIADH), a condition that causes hyponatremia and contributes to worse outcome patients experiencing a stroke. 47 In critically ill patients, copeptin levels have been shown to correlate with severity of disease.48,49 Three independent studies identified copeptin as an independent predictor of functional outcome and mortality at 90, 180, and 365 days with excellent AUC values (Tables 2 and 4). In ICH, copeptin is known to correlate with hematoma volume and is thought to be related to edema formation. 50
Molecular adhesion molecules have been implicated in ICH prognosis due to their roles in tissue remodeling and blood–brain barrier disruption. Degradation of the extracellular matrix by MMP-3 and MMP-9 results in cerebral edema, a secondary neuronal injury associated with poor outcomes. 51 In addition, tenascin-c (TNC) and thrombospondin-1 (TSP-1) have previously been implicated in aneurysmal subarachnoid hemorrhage. 52
Vascular and angiogenic biomarker levels change in response to inflammation, hypoxia, or platelet activation and have been studied in SAH, TBI, and ischemic stroke.51,53-55 However, their predictive utility in predicting long-term outcome in ICH is less clear. VEGF was identified as an outstanding predictor of 90-day mRS (AUC=.95). 53 However, these results are from a single study with short follow-up, and variables were not adjusted for in a multivariable model. Additionally, Li et al. 51 did not find a significant association between VEGF and long-term outcome.
There are scarce data investigating coagulation factors in the setting of ICH. Lauridsen et al. demonstrated that in patients without anticoagulant or antiplatelet therapy, local brain injury induced by ICH results in systemically activated coagulation. However, thrombin, ROTEM, and TAT were not significantly associated with long-term outcome in this cohort. 39 It is possible that coagulation factors are related to platelet activation in the recovery of ICH.
Leptin was an excellent predictor of mortality and poor outcomes at 6 months (AUC >.8) with increased serum levels predicting higher mortality and poor functional outcome. 56 Growth hormone levels have also been associated with high mortality and worse functional outcome, but no multivariate analysis was conducted in this particular study. 44 Similar to VEGF, higher LDL-C levels were observed in patients with low mortality as cholesterol decreases platelet aggregability, contributing to ICH hematoma volume and expansion risk. 57
Following ICH, hemoglobin released from erythrolysis binds iron in its ferrous (Fe2+) state and is then capable of generating reactive oxygen species and subsequent cellular damage. This results in an upregulation of iron regulatory proteins including ferritin and hepcidin, as well as others that have not yet been well-investigated in the setting of ICH such as transferrin and heme oxygenase. 35 Garton et al. 35 demonstrated that ferritin but not hepcidin was an independent predictor of 12-month functional outcome. This contrasted with Xiong et al. 41 who found that hepcidin was an independent predictor of long-term outcome. Additional investigation of iron regulatory proteins is necessary to ascertain their prognostic utility in ICH patients.
Study Limitations
This systematic review has several limitations. Multiple biomarkers are involved in more than one biological process, precluding uniform categorization. A more detailed analysis of the data is complicated by significant variability as the studies used different outcome measures at different time points.
Our study quality assessment score evaluation showed that the mean quality of all studies was considered moderate (Supplementary Table 2), and that many studies lacked internal and external validity. Only 13 (37.1%) of the 35 studies evaluated used blinded assessment of outcomes, and 21 studies (60%) adequately controlled for patient dropout. No studies cross-validated their prediction model in a second independent group and only 5 controlled for additional treatment effects on outcome measures. Also, 7 (20%) of the 36 studies did not conduct multivariate analyses to determine if the biomarker under investigation was an independent predictor of functional outcome.
Conclusions
Inflammatory as well as neuron and astrocyte-specific biomarkers contained the greatest number of investigated biomarkers, while S100B, WBC, and copeptin were among the most commonly investigated individual biomarkers in primary ICH. S100B, VEGF, and GFAP were the only biomarkers with outstanding AUC values. With the decreased cost and increased availability of proteomics and other omic analyses, multi-omic biomarker studies are likely to produce increasingly accurate multi-omic biomarker signatures in this disease process in the near future. The heterogeneity of the data analyzed in this review highlights the importance of further investigation for the predictive utility of biomarkers in risk stratification and management of primary ICH. Future studies should cross-validate prediction models in a second independent group, use blinded evaluators for outcome measurements, and adjust for treatment effects on outcome measures.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211041314 – Supplemental Material for Prognostic Utility of Serum Biomarkers in Intracerebral Hemorrhage: A Systematic Review
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683211041314 for Prognostic Utility of Serum Biomarkers in Intracerebral Hemorrhage: A Systematic Review by Zachary Troiani, Luis Ascanio, Christina P. Rossitto, Muhammad Ali, Shahram Majidi, J Mocco and Christopher P. Kellner in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary Material
Supplementary material for this article is available online on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
