Abstract
Keywords
Introduction
Intracerebral hemorrhage (ICH) accounts for 10% to 20% of all strokes, has a 30-day mortality of 50%, and often causes persistent disability. 1,2 Much is known about poststroke neuroplasticity mechanisms, such as synaptogenesis and neurogenesis, but most experimental studies and reviews focus on cerebral ischemia, 3,4 not ICH. Owing to differences in pathophysiology 5 as well as the location and extent of injury, one should not assume that findings in ischemia will apply to ICH. Thus, studies examining mechanisms of injury and repair following ICH are desperately needed to minimize injury from ICH as well as its behavioral consequences. Once an effective treatment is found for ICH, further study is needed to optimize treatment parameters such as dosage and timing.
Rodents, usually rats, are widely used to model ICH. The most common are the bacterial collagenase 6 and blood injection models. 7 Intraparenchymal infusion of collagenase, a bacterial enzyme that weakens the basal lamina of blood vessels, causes bleeding to occur over several hours. 8 The resulting hematoma engulfs and destroys tissue. The occurrence of spontaneous bleeding is a key advantage of this model. The other widely used model involves injecting autologous whole blood directly into the brain. The injection occurs over a few minutes with little or no spontaneous bleeding. Rapid cell death, because of mechanical trauma, occurs in both models. Furthermore, there is some ongoing cell death in perihematoma and distal regions in both models. 9,10 However, marked tissue loss continues for weeks in the collagenase model of striatal ICH, 8,11 which causes considerably more injury and substantially worse behavioral deficits (vs whole blood model) even when hematoma size is equivalent. 8 Post-ICH injury, which occurs in both models, has been targeted with numerous neuroprotective strategies, with varying success in animals 12 and consistent failures in humans.
Ongoing cell death is potentially of significant therapeutic importance. However, it is not known which model better mimics ICH in humans and which is considerably heterogeneous (eg, extent of injury, whether rebleeding occurs). Thus, it is important to use multiple models to better gauge experimental neuroprotectants, especially when one considers the range in human ICH and differences among models, such as in the extent of inflammation, 13 blood–brain barrier disruption, 8 and the pattern and amount of injury. 8 Likewise, neuroplasticity findings may differ among ICH models; thus, comparisons among models are needed to best understand post-ICH recovery, as has been done following cerebral ischemia. 14
Experimental findings in the collagenase model show that certain rehabilitation treatments, especially task-specific therapies such as skilled reach training, facilitate functional recovery after striatal ICH.
15-17
Interestingly, rehabilitation initiated after ICH can lessen cell death and the volume of tissue lost.
15,16,18
These neuroprotective effects, which one would expect to improve recovery, may be unique to the collagenase model because it seems to be the only ICH model where a
Thus, we presently evaluated whether rehabilitation (vs control treatment) improves recovery in rats subjected to whole blood infusion into the striatum, a common site of ICH in humans. Rehabilitation consisted of environmental enrichment coupled with skilled reach training in a modified staircase test. 16,17,19 Training began 1 week post-ICH and lasted for 2 weeks. Skilled reaching was measured with the single pellet task, a highly sensitive behavioral endpoint, 20 especially for this model. 21 Additional tests were not used to avoid the possibility that extensive testing would rehabilitate the animals. Dendritic arborization was measured at 1 and 7 weeks post-ICH to determine whether rehabilitation affects this form of neuroplasticity. Finally, lesion volume was assessed at 7 weeks to determine if tissue loss was affected.
Materials and Methods
Animals
Forty-eight male Sprague-Dawley rats (225-250 g, ~7-8 weeks old), bred at the University of Alberta, were used. Procedures complied with the guidelines of the Canadian Council on Animal Care and were approved by the Biosciences Animal Care and Use Committee at the University of Alberta.
Animals were randomly assigned to enhanced rehabilitation (ER) or control treatment and were housed in groups of 4, which remained constant throughout the study. Controls survived for 7 (n = 13) or 49 days (n = 14), whereas ER rats survived for 49 days post-ICH (n = 16). An additional 5 rats were excluded due to failure to learn (achieve >30% success) the single pellet reaching task (n = 4) or to display a reaching deficit following ICH (n = 1).
Skilled Reaching Training/Testing
Rats were food deprived to 90% of their free feeding weight adjusted for natural gains over time. They were then trained in the single pellet task 20,21 for 4 weeks prior to ICH (Figure 1), which in previous work has been sufficient for most animals to acquire the task and reach asymptotic performance. We chose this test because of its sensitivity to ICH and treatment effects and because it is clearly distinct from the reaching apparatus used for rehabilitation (see below). Briefly, after determining paw preference, rats were trained to obtain 45-mg food pellets (Bio-serv, Frenchtown, NJ) that were located on a shelf just outside a vertical slot through which the rat reached (Figure 2A). Twenty trials were conducted per day over 5 days a week. Rats were subjected to an ICH 3 to 7 days following the return to ad libitum feeding. Afterward, reaching success was assessed on days 5 and 6 and on days 42 through 46 post-ICH in transiently food-deprived animals (˜10% reduction). For statistical analysis, we averaged the data from the last 5 days of training (“baseline”), days 5 and 6 post-ICH (post-ICH/pretreatment), and days 42 to 46 (final endpoint). Behavioral testing and analysis was done blinded to group identity.
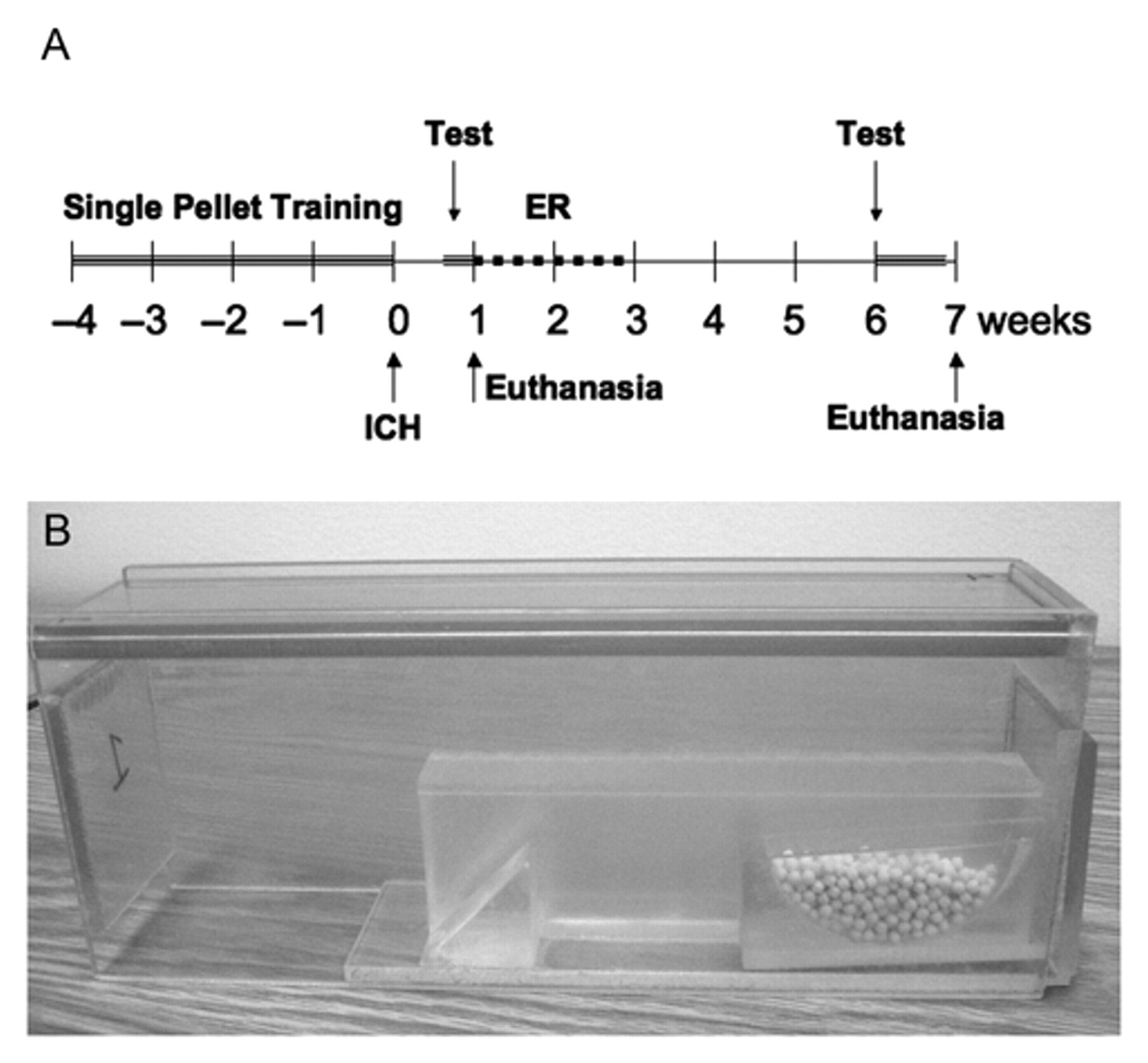
(A) Timeline of experimental procedures. (B) A modified Montoya reaching box used to provide task-specific reach training. This was coupled with daily environmental enrichment to form our enriched rehabilitation treatment
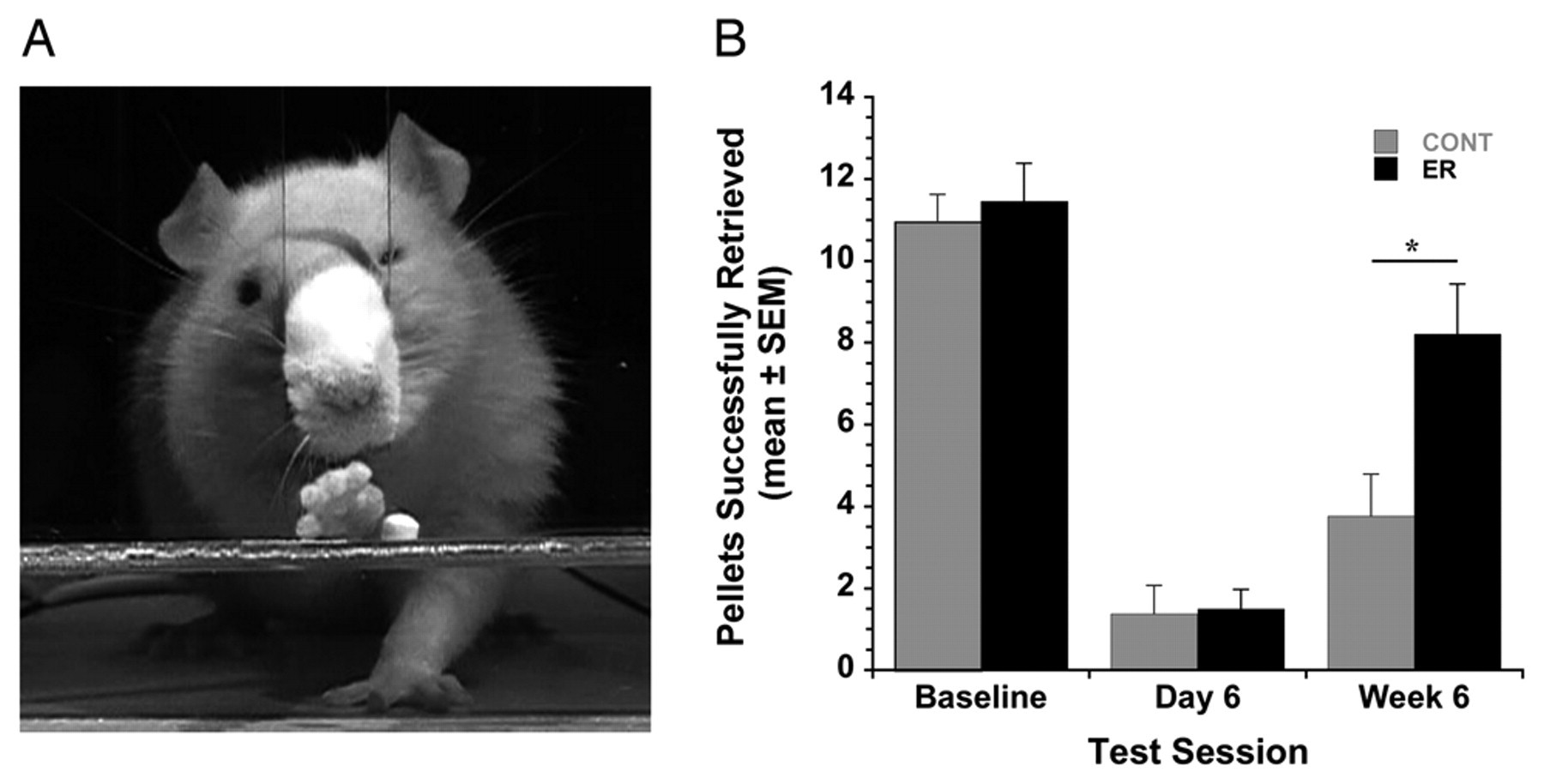
(A) The single pellet reaching task. (B) Reaching success was significantly improved by rehabilitation
Intracerebral Hemorrhage
Rats were anesthetized with isoflurane (4% induction; 1.75% to 2.25% maintenance in 60% N2O, 40% O2) for aseptic stereotaxic surgery. Briefly, an ICH was created by taking 100 µL of autologous blood from the tail vein and promptly infusing it, via a 26-G needle, into the striatum via a burr hole located at 3.5 mm lateral to Bregma (AP = 0) at a depth 6 mm below the skull surface. 7,21 Blood was manually infused over 10 minutes with a further 10-minute delay before removing the needle. The hole was sealed with a metal screw, a local anesthetic was applied (Marcaine, Sanofi, Markham, Ontario, Canada), and the wound stapled close. Body temperature was maintained at 37°C throughout surgery. The ICH was created on the side contralateral to the preferred paw in the reaching task.
Rehabilitation Treatment
Treated rats received a combination of environmental enrichment (4 rats/cage in a large 3-level cage containing tunnels, ramps, and various “toys” cleaned and changed biweekly) and skilled reach training in the modified Montoya staircase test beginning 1 week after ICH.
16,17
Environmental enrichment lasted from 5:30
Histology
Six to 8 rats per group, randomly chosen, were used for lesion volume assessment whereas the others were used for evaluating dendritic arborization. In the first case, the rats were overdosed with pentobarbital (˜100 mg/kg) and transcardially perfused with phosphate-buffered saline followed by 10% neutral-buffered formalin. Brains were extracted, embedded in paraffin, and serial coronal sections (10 µm thick; 400 µm apart) were taken throughout and well beyond the lesion. Sections were stained with cresyl violet, and using Scion Image J (4.0; Scion Corporation, Frederick, MD) the tissue loss was expressed as a percentage of the unaffected hemisphere: 100 − (volume of healthy tissue on the side of the ICH/volume of tissue in the normal hemisphere × 100), as per established methods. 8,17,22
The remaining rats, also anesthetized with pentobarbital, were perfused with saline and the brains were immersed in Golgi–Cox solution for 2 weeks followed by 2 days in 30% sucrose. This tissue was then sectioned (200 µm) on a vibrating microtome (Leica VT 1200S; Leica Microsystems GmbH, Wetzlar, Germany) and developed to visualize the stain. 23 Five neurons in the contralateral striatum were drawn per animal, 14,19,24 Neurons were not drawn in the ipsilateral hemisphere because of poor staining of perihematoma tissue, which appears to be due to the ICH because staining problems were not evident elsewhere in the brain. The Sholl analysis of ring intersections estimated dendritic length, whereas branching order analysis gauged dendritic complexity. 23 All procedures were done by experimenters blind to group identity.
Statistics
Data are presented as mean ± SEM and were analyzed by analysis of variance (ANOVA) (SPSS version 15 [SPSS, Inc, an IBM Company, Chicago, Illinois]) with Tukey honestly significant difference (HSD) post hoc tests when appropriate. A
Results
As expected, baseline (mean of last 5 training days) skilled reaching success, which was ˜55%, was not different among groups (
Drawings of Golgi–Cox stained neurons (Figure 3A) underwent Sholl analysis (Figure 3B) to determine dendritic complexity. This analysis revealed a significant group effect (
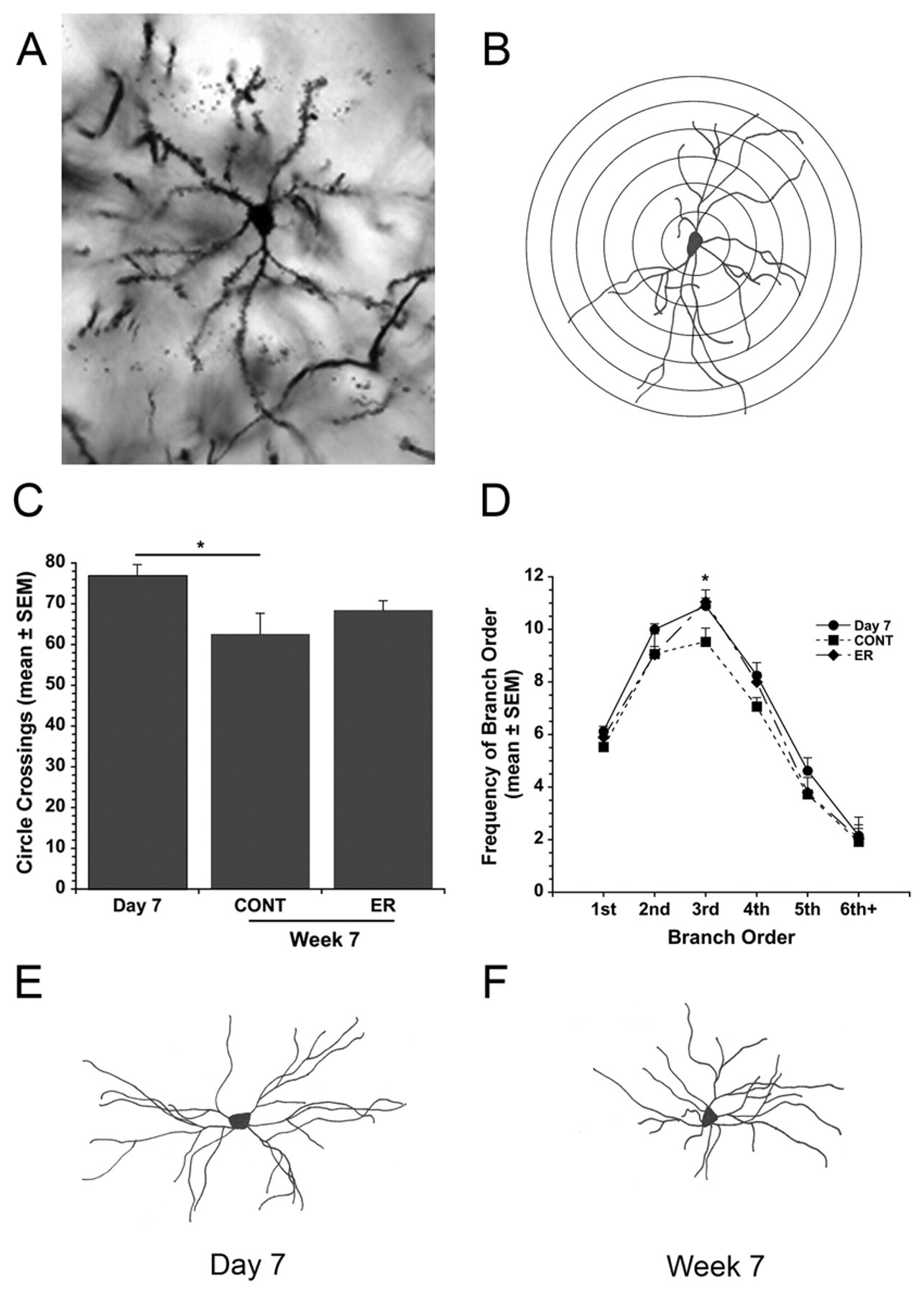
(A) Photomicrograph of a Golgi–Cox stained striatal neuron (20× objective). (B) Camera lucida drawing of a Golgi–Cox stained neuron with overlapping concentric circles used in the Sholl analysis. (C) Average number of circle crossings, reflecting dendritic length, at 7 days or 7 weeks post-ICH. (D) Branch order analysis showing similar complexity among groups. (E) Sample neuron at 7 days.(F) Sample neuron at 7 weeks
Tissue damage was largely restricted to the striatum, but the corpus callosum was also damaged as previously observed (Figure 4A). The percentage of tissue lost (Figure 4B) was not significantly different among groups (
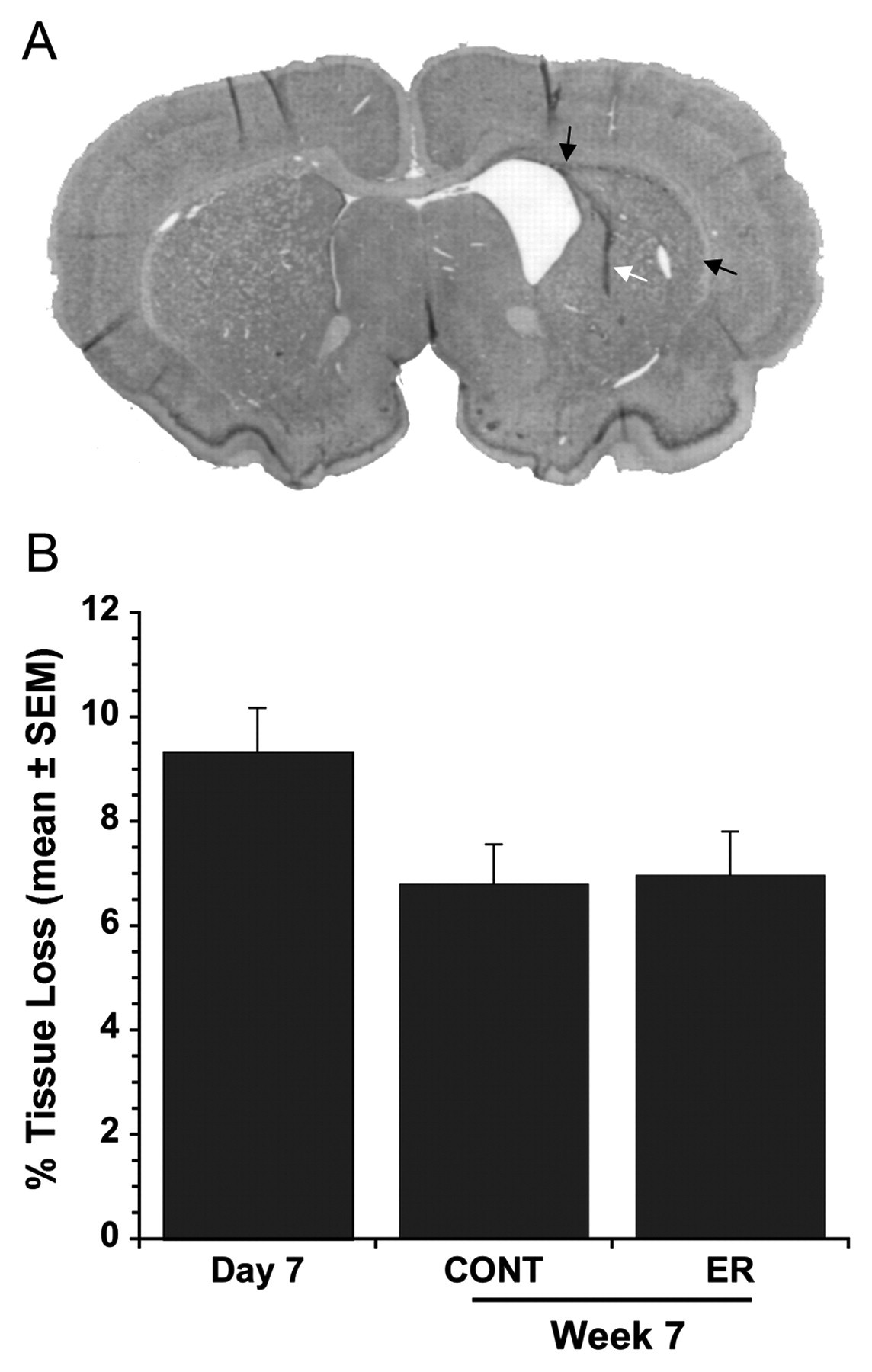
(A) Photomicrograph of typical injury at 7 weeks post-ICH (white arrow: injection tract; black arrows: hemosiderin along the corpus callosum). (B) The volume of tissue loss, expressed as a percentage of the contralateral side. There were no significant differences among groups
Discussion
The collagenase and whole blood ICH models are widely used to identify mechanisms of injury and to evaluate putative treatments. 5,12 However, differences between these models, such as in the progression of injury, and our current inability to identify the more clinically predictive model mean that preclinical assessments should be done in several models. 25 Presently, we show that ER, which was developed for focal cerebral ischemia 19 and recently shown to improve outcome after collagenase-induced ICH, 16,17 also substantially enhances recovery in the whole blood model. Cumulatively, these data and limited clinical findings 26-28 support the use of rehabilitation after hemorrhagic stroke, such as those with basal ganglia damage. Interestingly, our present data coupled with other recent findings also point to important differences between ICH models with respect to how rehabilitation improves recovery. Unfortunately, most clinical and experimental studies 3,4 examining mechanisms of recovery use ischemic stroke patients and related models and often use cortical not striatal injury. Thus, work specifically on mechanisms of recovery after ICH is warranted.
Our findings of reduced dendritic length over time (7 days vs 7 weeks) after whole blood–induced ICH are at odds with data in the collagenase model where a sustained increase occurs. 11 Similarly, ER significantly increased dendritic length in the contralateral striatum after collagenase-induced ICH, 17 whereas this effect was not presently observed in the whole blood model. We put forth several potentially complementary hypotheses to explain these findings. First, the contradictory findings may relate to insult severity. Specifically, greater injury sustained in the collagenase model may lead to greater recruitment of contralateral structures as part of the recovery process—a phenomenon observed after ischemia in rats and humans. 3,29 This also explains the failure to see a marked effect of ER after blood injection where ipsilateral structures are more important in recovery. Unfortunately, too few neurons stained with Golgi–Cox solution in the perihematoma region to test this hypothesis. We do not have a clear explanation for this effect, but it may stem from an interaction of iron released from degrading erythrocytes and the Golgi solution. Second, the time course of injury may be the critical factor as most cell death occurs rapidly in the whole blood model whereas a considerable amount of delayed injury occurs after collagenase infusion. 8,11 This protracted cell death may serve as an ongoing stimulus for brain plasticity in the contralateral hemisphere, leading to a sustained increase in dendritic length and complexity in the collagenase model not seen in the whole blood model. Finally, other model differences, such as in inflammation, 13 must be considered. These hypotheses can be evaluated in future studies (eg, varying insult severity), which should use additional techniques to overcome shortcomings of the Golgi–Cox method.
As discussed, ongoing cell death in the collagenase model is a potential target for traditional neuroprotective agents as well as rehabilitation therapies. Unlike the collagenase model, 15,16 however, we found that ER did not reduce lesion volume after whole blood injection. Thus, ER works through other means in this model. The most likely explanation is that, unlike in the collagenase model, there is little injury at a week or more after whole blood injection, 8 which we also found in this study. At present, it is not entirely clear whether a significant amount of delayed cell death (days/weeks) occurs after ICH in humans, an issue that could be addressed with repeated imaging. Should protracted cell death occur, then rehabilitation treatments, at least in some situations, may also rescue cells after ICH in patients.
Randomized clinical trials and meta-analyses confirm the benefits of rehabilitation after ischemic stroke, while emphasizing the importance of treatment intensity and early intervention. 30 Similar data are lacking for ICH, although it is assumed that rehabilitation is effective, as demonstrated in animal studies. Nonetheless, additional studies are needed to address unresolved issues such as the effects of treatment intensity and intervention delay on the efficacy and mechanisms of action of rehabilitation therapies after ICH. And it would be interesting to know how such factors interact with model differences (eg, location of injury, severity, and rate of cell death). Likewise, a better understanding of how hemorrhagic stroke differs from ischemic stroke is needed, specifically with respect to mechanisms of cell death and neurorepair. On the latter, further study comparing stem cell responses, changes in astrocytes, and growth factor levels among models and treatments would be particularly timely. Such data should lead to more effective treatments that are tailored and optimized for each insult. Certainly, this might include pharmacological cotreatment with traditional physical therapies.
In summary, ER significantly improved skilled reaching ability in the whole blood model, as we previously observed after collagenase-induced ICH. Thus, benefit, at least with enrichment and task-specific training, has been obtained across models and across a range in insult severity. In total, these data strongly support the use of such training regimens after hemorrhagic stroke affecting the basal ganglia, including those with severe residual deficits. However, putative mechanisms of action, such as dendritic growth and neuroprotection, are distinct from our work in the collagenase model. These findings highlight fundamental model differences and the need, at least for now, to use both in preclinical assessment of potential ICH treatments. Furthermore, while basic mechanisms of recovery may be similarly engaged after ischemic and hemorrhagic stroke, further work identifying more subtle differences, including the extent and timing of such processes, may allow us to better individualize neurorestorative and combination therapies to patient’s characteristics and needs.
Footnotes
Acknowledgements
The authors thank Yonglie Ma and Rosalie Paquette for technical assistance.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Research supported by a grant from the Heart and Stroke Foundation of Alberta, Northwest Territories, and Nunavut to Frederick Colbourne, who is a recipient of a senior medical scholar award from Alberta Innovates Health Solutions. Gergely Silasi was supported by a dissertation scholarship from the University of Alberta.
