Abstract
Background
Persons with multiple sclerosis (pwMS) experience walking impairments, characterized by decreased walking speeds. In healthy subjects, the self-selected walking speed is the energetically most optimal. In pwMS, the energetically most optimal walking speed remains underexposed. Therefore, this review aimed to determine the relationship between walking speed and energetic cost of walking (Cw) in pwMS, compared with healthy subjects, thereby assessing the walking speed with the lowest energetic cost. As it is unclear whether the Cw in pwMS differs between overground and treadmill walking, as reported in healthy subjects, a second review aim was to compare both conditions.
Method
PubMed and Web of Science were systematically searched. Studies assessing pwMS, reporting walking speed (converted to meters per second), and reporting oxygen consumption were included. Study quality was assessed with a modified National Heart, Lung and Blood Institute checklist. The relationship between Cw and walking speed was calculated with a second-order polynomial function and compared between groups and conditions.
Results
Twenty-nine studies were included (n = 1535 pwMS) of which 8 included healthy subjects (n = 179 healthy subjects). PwMS showed a similar energetically most optimal walking speed of 1.44 m/s with a Cw of 0.16, compared with 0.14 mL O2/kg/m in healthy subjects. The most optimal walking speed in treadmill was 1.48 m/s, compared with 1.28 m/s in overground walking with a similar Cw.
Conclusion
Overall, the Cw is elevated in pwMS but with a similar energetically most optimal walking speed, compared with healthy subjects. Treadmill walking showed a similar most optimal Cw but a higher speed, compared with overground walking.
Introduction
Persons with multiple sclerosis (pwMS) commonly experience gait impairments. Not surprisingly, walking is reported as one of the most important bodily functions in pwMS 1 and is essential for activities of daily life and participation in social roles. 2 The association between walking impairment on the one hand and perceived fatigue on the other stresses the importance of maintaining walking ability.3,4 On the longer term, gait impairment can lead to a decreased physical fitness and increased risk for comorbidities such as depression and cardiovascular impairments in addition to the severity of MS.5,6
A common parameter characterizing gait impairment in neurological disorders is a decreased self-selected (comfortable) walking speed. In pwMS, gait is affected by increased muscle tone, 7 impaired sensation and coordination, 8 and can further be negatively influenced by disease symptoms such as muscle weakness, 9 pain, and fatigue.4,10 Common altered spatiotemporal gait parameters are decreased step length and cadence and increased step width and double support phase, which all contribute to a decreased walking speed. 11
In healthy persons, the self-selected walking speed corresponds to the lowest metabolic energy demand in oxygen per kilogram bodyweight per distance travelled (mL O2/kg/m), referred to as the energetic cost of walking (Cw). The self-selected walking speed is the result of an optimal energy transfer between legs during the step-to-step transition 12 described by the inverted pendulum principle of gait.13,14 Walking at faster or slower speeds than self-selected increases the Cw. In healthy persons, the self-selected and thus energetically most optimal walking speed ranges from 1.27 to 1.67 m/s.15-17 Gait alterations due to disease symptoms can result in an inefficient energy transfer between legs, thereby increasing the Cw. For example, decreased step length due to muscular weakness of the knee extensors or flexors 18 increases the Cw. 12 Persons with MS, even with a low disability, have been shown to walk with an altered arm swing pattern that is possibly a strategy to compensate for other gait alterations. 19 In summary, gait alterations in pwMS could lead to an increased Cw as well as a decreased self-selected walking speed.
In most cases Cw is measured via indirect calorimetry where oxygen (O2) consumption and carbon dioxide (CO2) production are recorded during either overground or treadmill walking. Although overground walking is expected to resemble natural walking better, gait analyses in treadmill conditions benefit from the possibility of measuring biomechanical parameters of multiple consequent steps. However, the comparison between overground and treadmill walking conditions should be interpreted with caution due to different gait patterns between both conditions. Previous studies have shown that treadmill walking is characterized with increased cadence and decreased step length, compared with speed-matched overground walking. In addition, the preferred walking speed is slower during treadmill walking. 20 These differences are related to an increased Cw in treadmill walking.21-23 The increased Cw in treadmill walking is also supported by a study that reported an increased muscle activation compared with overground walking. 24 The energetically most optimal walking speed in both treadmill and overground conditions should be respected when conducting gait analyses. The difference in Cw between overground and treadmill walking is expected to be even greater in pwMS as walking is more challenging due to, for example, increased balance control. To our knowledge, the energetically most optimal walking speed in pwMS in both conditions has not been assessed yet. Investigating the relationship between Cw and walking speed can help understand the decreased preferred walking speed in pwMS, ultimately leading to tailored gait training for walking ability.
It is expected that pwMS walk at a slower energetically most optimal walking speed but with an elevated Cw due to disease symptoms. Also, treadmill walking is expected to be energetically more costly, compared to overground walking. By combining studies in which pwMS and healthy subjects have been challenged to walk at a range of self-selected and fixed different speeds, an overview will be provided of the relationship between walking speed and Cw in pwMS and healthy subjects. Therefore, the primary aim of this review is to determine the relationship between walking speed and energetic Cw in pwMS, thereby assessing the walking speed with the lowest energetic cost. The secondary aims were to assess the energetically most optimal walking speed in pwMS compared with healthy subjects and between overground and treadmill walking.
Method
A systematic literature review was performed of studies reporting the Cw and walking speed in pwMS. The systematic review was conducted based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 25 The protocol was registered and uploaded in PROSPERO CRD42020192661.
Search and Information Sources
A systematic literature search was performed in databases PubMed and Web of Science, until August 29, 2020. In order to identify all articles on the Cw and walking speed in pwMS, the following key terms were used reflecting the population, exposure, and outcome measures: multiple sclerosis AND walking AND energy metabolism. The full search string can be addressed in Appendix 1 (available online). Additional reference tracking of included articles was performed with similar key terms. Duplicates were removed before assessing eligibility criteria.
Eligibility Criteria and Study Selection
Studies were included when the following criteria were met: (1) persons with a diagnosis of MS, (2) walking speed and oxygen consumption or Cw were reported as outcome measure, (3) age ≥18, (4) ≥5 participants, and (5) articles published in English or Dutch. The following studies were excluded: (1) intervention studies without reported baseline outcome measures, (2) (systematic) reviews, meta-analyses, practice guidelines, and (3) cadaver or animal studies. All types of study designs were included. The articles were first screened on title and abstract based on the selection criteria. In case title and abstract did not provide sufficient information, the full text was consulted. In case of overlapping datasets, the study with the original, first published dataset was included. All studies were screened by 2 independent assessors (KT and BB) after which the included articles were compared to reach consensus.
Quality Assessment
After inspecting several instruments for quality assessments, the National Heart, Lung, and Blood Institute (NIH) quality assessment tool for observational cohort and cross-sectional studies was the best fit when considering the design of included studies (Supplementary File 2, available online). Notwithstanding, some adaptations were implemented to improve efficiency and specificity. Four criteria were excluded because studies retrieved for this systematic review did not comprise longitudinal studies. The criteria addressing exposure and outcome were given more importance by distinguishing (1) “whether a clear description was provided of the measurement of exposure/outcome” and (2) “whether this measurement was accurate and reliable.” Furthermore, to emphasize importance of accuracy of outcome measurement, one criteria was added (3) “was resting metabolism reported?” Finally, 4 criteria were added to evaluate the selection of controls (if applicable) following the Newcastle-Ottowa checklist for case-control studies. The final quality checklist consisted of 14 items across 3 domains: Study objective (1): Was the study question clearly reported in defining their aim and study population (2 items)? Risk of bias (2): Did the study control for risk of bias in their study design and analysis (5 items)? and Accuracy of outcome and exposure (3): Are exposure and outcome used accurate and reliable (7 items)? Each item was answered with “yes,” “no,” “cannot determine,” or “is not reported.” Items answered with “yes” were assigned 1 point. Items answered with “no,” “cannot determine,” and “not reported” were considered as a potential risk of bias or low quality and received no point. Next, the quality of each domain was determined as good, moderate, or poor based on the number of positive ratings, resulting in the following classification: Study objective: Good scored 2/2, moderate 1/2, and poor 0/2; Risk of bias: Good ≥4/5, moderate 3/5, and poor ≤2/5; Accuracy of outcome and exposure: Good ≥6/7, moderate 4-5/7, and poor ≤3/7. Two reviewers (KT and BB) independently rated the quality assessment, and in case no consensus was met, a methodologist as third assessor was consulted (AB). The quality assessment was used for describing the quality of the studies; however, no studies were excluded.
Data Extraction
After study selection, the following data were extracted: population characteristics of pwMS (and if applicable for healthy subjects): number of subjects, gender, age, height, and weight; MS characteristics: time since diagnosis, type of MS (benign, primary progressive [PP], secondary progressive [SP], or relapsing remitting [RR]) and disability reported as Expanded Disability Status Scale (EDSS) score or Patient-Determined Disease Steps (PDDS) score; Walking condition: treadmill or overground. In case of intervention studies, only baseline measurements were included which could be considered as either observational cross-sectional study designs or, in case of comparison to healthy subjects, as case-control studies.
Data Synthesis
The main outcome measures were reported as walking speed, converted to meters per second (m/s), and Cw expressed as consumed milliliters oxygen per kilogram bodyweight per distance travelled in meters (mL O2/kg/m). Cw was calculated manually in case of O2 consumption; walking speed (or derivatives such as step length and step time) and body weight were reported. Studies reporting the net Cw were converted to gross Cw by summing the resting and walking O2 consumption. Narrative summaries were separately described in a table for studies including pwMS (n = 29), pwMS and healthy subjects (n = 8), overground (n = 19) walking, and treadmill walking (n = 10). Data of walking speed versus Cw were also visualized in figures that included the line of best fit using a second-order polynomial function through the data with IBM SPSS Statistics for Windows, Version 25 (IBM Corp). The energetically most optimal walking speed was calculated from this function and described for pwMS, healthy subjects, overground walking, and treadmill walking.
Results
The PRISMA flow-diagram of the included studies is presented in Figure 1. Initially, a total of 205 articles were found of which 59 duplicates were removed. After screening title and abstract based on eligibility criteria, 88 studies were excluded. After full text screening, 25 studies were excluded according different criteria. Additionally, 4 studies were excluded due to use of apparent overlapping datasets,26-29 leaving a remaining 29 studies for analysis describing the Cw at different speeds in pwMS and pwMS and healthy subjects. Eight studies included healthy subjects, 19 studies were included for overground walking, and 10 studies were included for treadmill walking.

PRISMA flow diagram for study selection depicting the number of records identified, included and excluded, and the reasons for exclusions.
All subject characteristics are presented in Table 1. In total, 1535 pwMS (71% female) with mean age 46 ± 10 years, height 1.69 ± 0.09 m, and weight 75 ± 17.2 kg and 190 healthy subjects (55% female; 42 ± 8 years; 1.70 ± 0.06 m; 70.6 ± 12.9 kg) were included. For pwMS, the single patient disability levels ranged in EDSS from 1 to 6.5 (n = 13) and PDDS from 1 to 4 (n = 11), reflecting no to moderate disability. Mean time since diagnosis was 10.2 ± 7.6 years.
Characteristics of Included Studies a .

Abbreviation: n.s., not specified. Subject characteristics: number (female); age; height; weight, Disease characteristics: Expanded Disease Disability Scale (EDSS)/Patient Determined Disease Step (PDDS); disease duration; Primary Progressive (PP), Secondary Progressive (SP), Relapsing Remitting (RR); Walking condition: surface, (passive) walking aid allowed such as canes and walkers, metabolic system; Walking speed: meters per second (m/s); Cost of walking: gross milliliter oxygen per kilogram bodyweight per meter (mL O2/kg/m); Oxygen consumption during walking (VO2): milliliter per kilogram bodyweight per minute (mL/kg/min).
All characteristics are presented as mean or median values and in case available ± standard deviations.
Indicates conversion from net to gross Cw by summing the resting O2 consumption.
Indicates calculation Cw from data available in article.
Quality Assessment
As presented in Table 2, almost 60% of the studies (17/29) were considered to have good quality on the domain “study objective,” and none of the studies were of poor quality concerning this item. Risk of bias was poor for 28% (8/29) of the studies, and for the domain “accuracy of outcome and exposure,” 24% (7/29) were rated as poor quality. No studies were assessed as good quality on all domains; the same applies to poor quality. The following items were assessed poorest where all studies but one scored “no”: “Were the outcome assessors blinded to the exposure status of participants?” and “Was a sample size justification, power description, or variance and effect estimates provided?”
Study-Specific Assessment of the Modified NIH Quality Assessment Tool (14 Item Across 3 Domains) a .

Cells with a check mark indicate a “yes” reported. Blanks are items reported as no/not reported. The overall domain score in light grey indicates “good” quality, medium grey indicates “moderate” quality, and dark grey indicates “poor” quality.
Cost of Walking of All Studies Including pwMS
The cost of walking at multiple speeds of all pwMS is represented in Figure 2a including 29 studies. The minority of these studies (n = 6) presented data of the Cw at higher speeds (>1.44 m/s),30-34 compared with all 29 studies also reporting slower speeds (<1.44 m/s). The highest Cw values are related to the lowest walking speeds with Cw values ranging from 0.43 to 0.44 mL O2/kg/m at speeds between 0.28 and 0.56 m/s.35-38 These studies did not report disease disability scores in EDSS or PDDS; however, all participants were allowed to use a walking aid. The lowest Cw values ranging between 0.15 and 0.17 mL O2/kg/m concurred with walking speeds ranging from 0.95 to 1.78 m/s.30,32,34,39-41 Studies including persons with a moderate disease disability did not reach speeds faster than 1.4 m/s. A scatter plot can be consulted in Supplementary File 3 (available online). A second order polynomial fit (R2 = 0.65, P < .05) estimated the lowest Cw to be 0.16 mL O2/kg/m at a walking speed of 1.44 m/s.
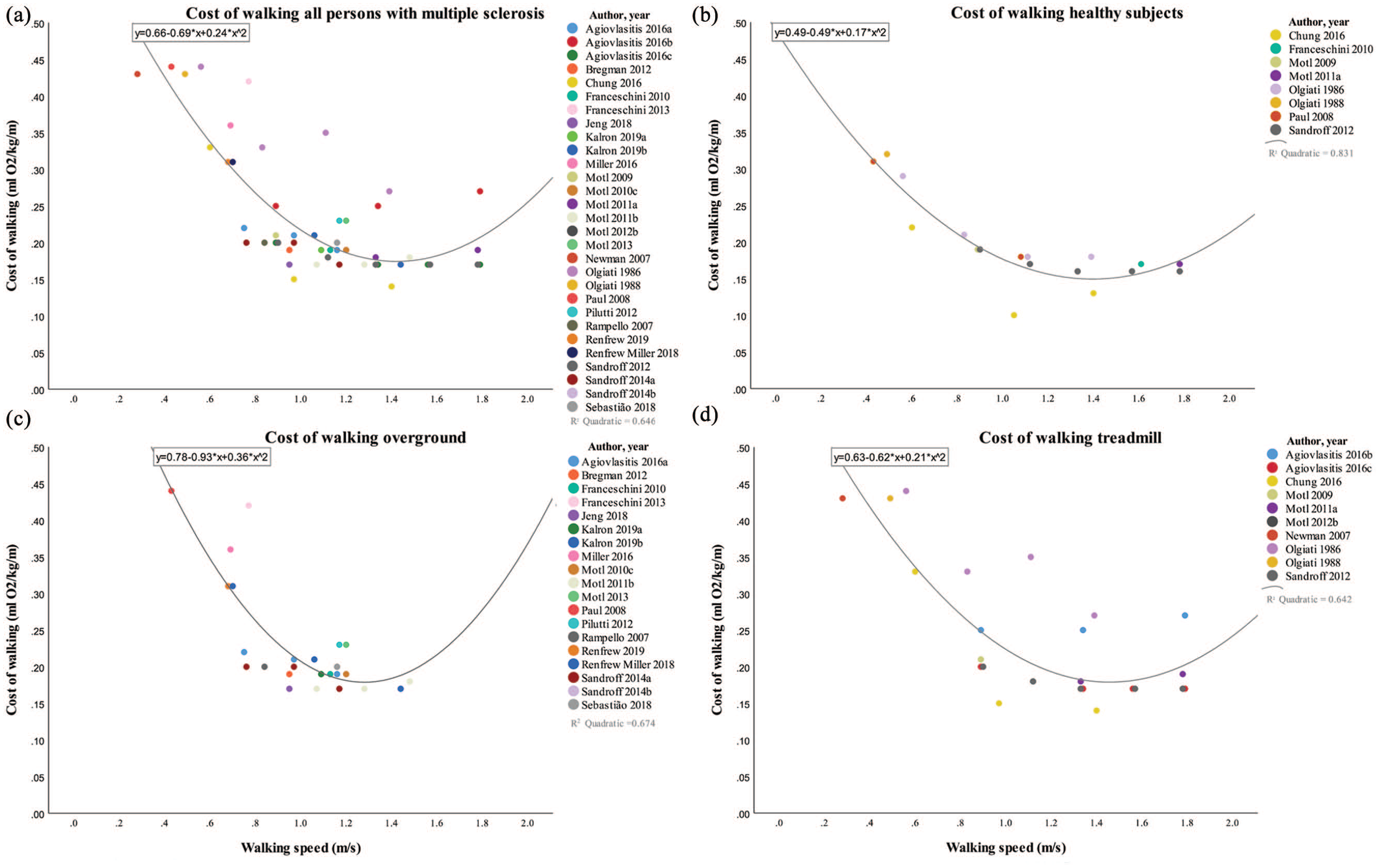
(a) Cost of walking for all studies including persons with multiple sclerosis. (b) Cost of walking for all studies including healthy subjects. (c) Cost of walking for all studies including persons with multiple sclerosis in overground walking conditions. (d) Cost of walking for all studies including persons with multiple sclerosis in treadmill walking conditions.
Cost of Walking of pwMS and Healthy Subjects
Results of 8 studies including healthy subjects and pwMS are presented in Figure 2b. Seven studies compared the Cw of healthy subjects at matched walking speeds as pwMS with an average walking speed of 1.31 m/s with a Cw of 0.23 mL O2/kg/m for pwMS and 0.18 mL O2/kg/m for healthy subjects.30-32,36-38,40 The self-selected walking speed was measured in 3 studies, resulting in a walking speed of 0.84 m/s for pwMS and 1.25 m/s for healthy subjects with Cw values of 0.26 and 0.15 mL O2/kg/m, respectively.36,40,42
By fitting a second-order polynomial equation (R2 = 0.83, P < .05) over the data of all included studies assessing the Cw in healthy persons, the lowest Cw was estimated to be 0.14 mL O2/kg/m at a walking speed of 1.44 m/s. The energetically most optimal walking speed of pwMS (n = 29) was similar to healthy subjects (n = 8) but with a higher energetic cost (0.16 vs 0.14 mL O2/kg/m for healthy subject).
Cost of Walking of Overground Versus Treadmill Walking in pwMS
Results of overground and treadmill walking studies are presented in Figure 2c and Figure 2d, respectively. The range in walking speeds in overground conditions28,30,39,41,43-55 was lower, compared with treadmill walking,33-35,56 with speeds between 0.43 and 1.48 m/s with Cw values between 0.16 and 0.44 mL O2/kg/m. The majority of the treadmill speeds were set at a predetermined velocity, ranging from 0.28 to 1.79 m/s, with concurring Cw values from 0.14 to 0.44 mL O2/kg/m. The use of a walking aid was specifically allowed in 74% (14/19) of all overground studies, versus 50% (5/10) of the treadmill walking trials. Walking aids were specifically not allowed in 16% (3/19) of the overground and in 50% (5/10) of the treadmill walking studies. The remaining studies did not specify. A second-order polynomial fit (R2 = 0.67, P < .05; R2 = 0.64, P < .05) computed an energetically most optimal walking speed for overground and treadmill walking of 1.29 m/s and 1.48 m/s with concurring Cw values of 0.18 mL O2/kg/m and 0.17 mL O2/kg/m, respectively.
Discussion
The primary aim of this review was to determine the relationship between walking speed and Cw in pwMS, thereby assessing the walking speed with the lowest energetic cost. It was expected that pwMS walked slower and with a higher energetic cost, compared with healthy subjects. Our results confirmed that pwMS walk with an elevated Cw at all speeds while showing a similar energetically most optimal walking speed compared with healthy subjects. In addition, walking was energetically more costly at slower speeds in pwMS.
A higher Cw could be the result of an inefficient energy transfer between legs (ie, step-to-step transition) since the double support phase entails 60% to 70% of the energy transfer between legs at a given speed. 13 The prolonged double support time, as found in pwMS, is related to altered spatiotemporal step parameters such as decreased step length and cadence and increased step width. 57 This could imply that energy is lost due to an altered walking strategy resulting in an increased Cw in pwMS.
Besides altered spatiotemporal parameters, pwMS walk with a higher kinematic variability, 58 which is related to an increased Cw in general. 55 In addition, the inverse relationship between gait variability and walking speed could explain the elevated Cw at slower speeds in pwMS compared with healthy subjects, as found in our results. The similar increase in Cw at speeds faster than energetically most optimal could be the result of similar strategies for increasing joint power at higher speeds in pwMS and healthy subjects, as suggested by Brincks et al. 59 Other kinematic gait alterations that are related to an increased Cw in pwMS such as decreased arm swing60,61 and increased forward-backward trunk range of motion 62 are also related to a decreased dynamic balance. Balance deficiency is present in all impairment levels of pwMS63-65 and presents itself in the majority of the gait alterations as described above. Improving balance might decrease the Cw in pwMS 66 and could be considered an overall goal in gait training.
Studies assessing dynamic balance in pwMS often focus on muscle strength, 67 which is one of the disease symptoms in pwMS affecting gait. 18 The ability of producing sufficient muscle force for walking at the energetically most optimal walking speed could be impaired due to muscle weakness. This is supported by the inverse relationship between walking speed on the one hand and strength of knee flexors and extensors 18 and ankle dorsiflexors 68 on the other. Improving muscle strength increases walking speed69,70 and might contribute to a decreased Cw.
Besides muscle weakness, altered muscle activation patterns leading to muscle fatigue could also be related to an increased Cw. In a recent study assessing muscle fatigue in pwMS, it was found that the ankle plantar flexors exhibited muscle fatigue after prolonged walking. 71 This is in line with previous studies describing a reduced ankle push off power for forward propulsion, which consequently decreases walking speed. 72 The inability to generate sufficient muscle power for forward propulsion could explain the finding that the majority of the included studies assessing the Cw at slower speeds in pwMS. In addition, since muscle contraction is the main source of energy, the energetic demand increases as a result of muscle co-activation. 73 PwMS were found to walk with increased co-activation of the knee and ankle muscles, compared with healthy subjects. Slower walking speed, higher impairment level, and decreased balance were related to this increased co-activation. 74 One should note that slower gait speed and increased Cw are related to increased levels of fatigue in pwMS. 40 This relationship was not found at higher speeds, which could indicate walking faster is not only energetically more beneficial but also in terms of level of fatigue.40,47
A recent systematic review by Stella et al 75 assessed the Cw at comfortable walking speed in pwMS of 19 included studies. They found that pwMS walk at a comfortable walking speed of 1.12 m/s with a gross Cw of 0.19 mL O2/kg/m in overground conditions. This Cw is similar to our results when calculating the Cw with the second-order polynomial function from our data. Although the aim of the study by Stella et al was to report the Cw at comfortable walking speed, they included some studies in which subjects were instructed to perform a 6-m walking test by walking as fast as possible. 76 It is therefore expected that the actual comfortable walking speed in pwMS might be lower. Our results describing the Cw at the energetically most optimal walking speed by including speeds ranging from slow to fast is considered a broader clinical relevant investigation, compared with the comfortable speed assessed by Stella et al. 75
The results of our secondary aim to compare the Cw between overground and treadmill walking indicates that the lowest energetic cost is similar in both conditions. However, the optimal walking speed is higher in treadmill conditions with an optimum at 1.44 m/s, compared with 1.29 m/s in overground walking. Previous literature suggested that treadmill walking requires a higher energetic cost due to the more constraint environment compared with overground walking. The lack of optic flow could lead to a higher regulation of motor output and thus increase the Cw.21,22 This is supported by the study of Berryman et al, 23 suggesting that the Cw is higher in treadmill walking, compared with overground walking in healthy older adults. However, our results show no differences in Cw between both conditions. The large range in Cw in the treadmill conditions could suggest an inclusion of heterogeneous groups in our review. This could be the result of sampling bias of many studies including pwMS with lower disability levels and by restricting the use of walking aid in the treadmill walking conditions. A possible explanation for the higher energetically most optimal walking speed in treadmill walking as found in this systematic review could be related to an increased balance control. 77 Since pwMS are known to experience balance impairments, walking at higher speeds might increase stability in treadmill conditions. 74 This is supported by Boudarham et al, 74 suggesting that pwMS walking at higher speeds decreased muscle co-activation in treadmill walking, compared to slower walking.20,78
With regard to the quality of the studies on the different domains, it is found that most studies clearly reported their Study objective. In the domains Accuracy of outcome and exposure and Risk of bias, study quality was more often reported as moderate and poor quality. Specifically, the items on sample size calculation including power description, variance, and effect estimates was often not reported, except for 2 studies.28,45 Both the expected and measured power of a certain outcome would improve transparency of the study results. In addition, the item on blinding was not reported correctly in most studies. It is of note that study designs in this systematic review do not necessarily require blinding. However, strictly taken, assessors might increase risk of bias when not blinded for patient/controls or for disability levels due to biased interpretation of the results.
Although sufficient articles were included in this systematic review, several limitations could influence the results on the relationship between Cw and walking speed. First of all, it should be noted that daily life gait speed might be slower compared to laboratory-based (overground) gait speed. 79 Given that the focus of this review was on laboratory-based overground and treadmill gait, one should be cautious by translating our findings to daily life gait.
Second, with regard to the Cw curve, no studies included walking at higher speeds than 1.8 m/s and only few above 1.44 m/s (n = 6). This could be the result the inability of walking at higher speeds due to disease characteristics. When assessing disability levels at different walking speeds, pwMS with higher disability levels walk at slower speeds. The relationship between level of impairment and walking speed and thus Cw could explain the higher energetic demand at slower speeds in pwMS. Ideally, studies should include a wider range in walking speeds in which the Cw curve is reconstructed per subject. However, the walk-to-run transition should be kept in mind when including higher speeds. In this review, the absence of walking at higher speeds in overground conditions decreases the fitted curve of the polynomial regression. Although the relationship between walking speed and Cw is known to be quadratic, the Cw at higher speeds should be interpreted with caution.
A third methodological consideration of this systematic review is the conversion of oxygen consumption, bodyweight, and walking speed to Cw. Although this enables a comparison of Cw between studies, reliability would be higher in case this was reported in the included studies. Considering the second-order polynomial fit, the variance in Cw can be explained with higher certainty in healthy subjects, compared with pwMS. Future studies should account for the big variance in Cw in pwMS by increasing sample size or within group homogeneity.
Last, considering the quality assessment checklist, a note should be made that the authors adjusted the checklist to fit the research objective. Although it is suggested that quality checklists should fit the study objective, this could affect the outcome of the overall quality assessment.
Clinical Implications
The energetically most optimal walking speed in pwMS is similar to healthy subjects but pwMS walk with an increased Cw. The majority of the studies included walking at (self-selected) slower speeds, with a higher Cw compared to the energetically most optimal speed (1.44 m/s). Therefore, our results indicate that the majority of the pwMS included in the studies energetically benefit from walking faster. The outcome of gait training in pwMS focusing on increasing the self-selected walking speed might decrease the Cw. From recent literature, it is expected that not one specific but a combination of parameters explain the elevated Cw in pwMS. Increasing self-selected walking speed can be achieved by improving balance control, muscle strength, and muscle performance since these are known to be related to a decreased walking speed or Cw. This is confirmed by experts recommending task-specific training for improving walking ability. 80 By optimizing walking ability, the benefit of increased physical fitness and decreased levels of fatigue and risk for other comorbidities are expected to result in a higher quality of life. The design of such interventional studies should account for potential differences in overground versus treadmill walking.
Conclusion
Overall, the Cw is elevated in pwMS but with a similar energetically most optimal walking speed, compared with healthy subjects. Although the optimal energetic cost is similar between overground and treadmill walking, the energetically most optimal walking speed is greater in treadmill walking.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683211005028 – Supplemental material for The Relationship Between Walking Speed and the Energetic Cost of Walking in Persons With Multiple Sclerosis and Healthy Controls: A Systematic Review
Supplemental material, sj-docx-1-nnr-10.1177_15459683211005028 for The Relationship Between Walking Speed and the Energetic Cost of Walking in Persons With Multiple Sclerosis and Healthy Controls: A Systematic Review by Kyra Theunissen, Guy Plasqui, Annelies Boonen, Bente Brauwers, Annick Timmermans, Pieter Meyns, Kenneth Meijer and Peter Feys in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683211005028 – Supplemental material for The Relationship Between Walking Speed and the Energetic Cost of Walking in Persons With Multiple Sclerosis and Healthy Controls: A Systematic Review
Supplemental material, sj-docx-2-nnr-10.1177_15459683211005028 for The Relationship Between Walking Speed and the Energetic Cost of Walking in Persons With Multiple Sclerosis and Healthy Controls: A Systematic Review by Kyra Theunissen, Guy Plasqui, Annelies Boonen, Bente Brauwers, Annick Timmermans, Pieter Meyns, Kenneth Meijer and Peter Feys in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-3-nnr-10.1177_15459683211005028 – Supplemental material for The Relationship Between Walking Speed and the Energetic Cost of Walking in Persons With Multiple Sclerosis and Healthy Controls: A Systematic Review
Supplemental material, sj-pdf-3-nnr-10.1177_15459683211005028 for The Relationship Between Walking Speed and the Energetic Cost of Walking in Persons With Multiple Sclerosis and Healthy Controls: A Systematic Review by Kyra Theunissen, Guy Plasqui, Annelies Boonen, Bente Brauwers, Annick Timmermans, Pieter Meyns, Kenneth Meijer and Peter Feys in Neurorehabilitation and Neural Repair
Footnotes
All coauthors contributed equally for this work. Additionally, authors Bente Brauwers and Annelies Boonen assisted during the quality assessment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is funded by special research fund 2017 call for doctoral grants in the framework of BOF UHasselt—Maastricht UMC+ cooperation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
