Abstract
Background. Neurorehabilitation efforts have been limited in their ability to restore walking function after stroke. Recent work has demonstrated proof-of-concept for a functional electrical stimulation (FES)–based combination therapy designed to improve poststroke walking by targeting deficits in paretic propulsion. Objectives. To determine the effects on the energy cost of walking (EC) and long-distance walking ability of locomotor training that combines fast walking with FES to the paretic ankle musculature (FastFES). Methods. Fifty participants >6 months poststroke were randomized to 12 weeks of gait training at self-selected speeds (SS), fast speeds (Fast), or FastFES. Participants’ 6-minute walk test (6MWT) distance and EC at comfortable (EC-CWS) and fast (EC-Fast) walking speeds were measured pretraining, posttraining, and at a 3-month follow-up. A reduction in EC-CWS, independent of changes in speed, was the primary outcome. Group differences in the number of 6MWT responders and moderation by baseline speed were also evaluated. Results. When compared with SS and Fast, FastFES produced larger reductions in EC (Ps ≤.03). FastFES produced reductions of 24% and 19% in EC-CWS and EC-Fast (Ps <.001), respectively, whereas neither Fast nor SS influenced EC. Between-group 6MWT differences were not observed; however, 73% of FastFES and 68% of Fast participants were responders, in contrast to 35% of SS participants. Conclusions. Combining fast locomotor training with FES is an effective approach to reducing the high EC of persons poststroke. Surprisingly, differences in 6MWT gains were not observed between groups. Closer inspection of the 6MWT and EC relationship and elucidation of how reduced EC may influence walking-related disability is warranted.
Keywords
Introduction
Stroke is a leading cause of long-term disability. 1 Marked physical inactivity—with its increased risk of second stroke, heart disease, and diabetes, 1 and relation to hypertension, depression, 2 and a reduced health-related quality of life2,3—is a concerning sequela of stroke.4,5 For persons after stroke, rehabilitation is the cornerstone for recovery; however, current efforts are unable to resolve the motor impairments contributing to walking-related disability,6-11 which continues to worsen over the first year after stroke. 12 A critical need thus exists for innovative rehabilitation approaches capable of improving poststroke walking ability.
A recent critical review of poststroke walking therapies demonstrated similar (and limited) outcomes following rehabilitation efforts of varying sophistication 13 ; however, this assessment was based solely on the gains in walking speed observed following training. As a gross measure of walking ability, walking speed provides poor resolution for identifying the mechanisms underlying walking recovery, 14 and thus offers only a limited understanding of an intervention’s effects. Evaluating the effects of poststroke gait interventions solely on improvements in gross measures of walking function, such as speed, may be problematic because the biomechanical mechanisms underlying the ability to walk faster after intervention may be just as critical to facilitating community walking activity as the magnitude of improvement in walking speed. For example, recent work has suggested that if the ability to walk at faster speeds following gait intervention is not accompanied by more symmetric walking, a higher energy cost of walking may result. 15 Considering the relationship between high energy costs of poststroke walking16,17 and deficits in community participation 18 and walking function, 19 reducing the energy cost of poststroke walking may be critical to reducing walking-related disability.
Functional electrical stimulation (FES), which is the application of electrical stimulation for the generation of functional movement, is a common neurorehabilitation adjunct used in the treatment of walking dysfunction after stroke.20-32 Primarily, FES has been used for its orthotic effects with recent evidence demonstrating its equivalence,20-22 and in some cases superiority, 23 to ankle-foot orthoses. Although FES has recently gained popularity as a therapeutic aide,24-29 it remains unclear if FES produces therapeutic gains (ie, long-lasting effects observed when FES is not being delivered) superior to those produced by non-FES interventions.21,25,26,30-32 To date, the study of FES-based walking interventions in persons poststroke has focused mostly on the application of FES to the paretic dorsiflexors to reduce foot-drop. Consistent with previous work,33,34 our laboratory has shown that factors beyond foot-drop limit poststroke walking function. Indeed, we have recently shown that the propulsive force generating ability of the paretic limb (paretic propulsion) explains more of the variance in long-distance walking ability than measures related to ground clearance (eg, knee flexion or ankle dorsiflexion). 33 Because individuals in the chronic phase of stroke recovery identify deficits in their ability to walk farther distances as a key factor limiting their community engagement, 35 the study of FES-based locomotor therapies that target paretic propulsion by stimulation of the ankle plantarflexor muscles during late stance phase is warranted.
Early work from our laboratory demonstrated that the orthotic application of FES to both the paretic ankle dorsiflexors during swing phase (ie, targeting foot-drop) and plantarflexors during stance phase (ie, targeting propulsion) outperformed the conventional FES approach of stimulating only the dorsiflexors. 36 Subsequently, we demonstrated that combining this novel FES approach with fast treadmill walking yielded larger within-session improvements in gait mechanics than when FES was combined with comfortable speed treadmill walking. 37 The safety and feasibility of a 12-week intervention combining fast walking and functional electrical stimulation (FastFES) for the treatment of poststroke walking dysfunction was subsequently demonstrated. 28 In that preliminary study, meaningful improvements across the body structure and function, activity, and participation domains of the International Classification of Function, Disability and Health 38 were observed following training. Specifically, therapeutic gains in the propulsive force generated by the paretic limb during walking, as well as gains in walking speed, walking distance, balance, and self-perceived participation were observed in 11 of the 12 participants. 28 However, because FastFES combines fast-speed training, which is a popular and effective training approach,39-41 with FES, the particular contribution of the FES to the outcomes observed is not clear. These findings thus prompted the present study, which evaluates the efficacy of FastFES locomotor rehabilitation.
The primary objective of this investigation was to determine the effects of 12 weeks of FastFES locomotor training on the energy cost of poststroke walking. We hypothesized that, compared with conventional locomotor training at fast or comfortable speeds, 12 weeks of FastFES training would produce a larger reduction in the energy cost of walking. To determine the effects of FastFES training on participants’ gross walking function, this investigation also evaluated changes in participants’ long-distance walking ability. This study also addressed the heterogeneous nature of poststroke motor impairments. Because the FastFES program is centered on treadmill training at a fast walking speed, we posited that participants’ baseline fast walking speed would influence outcomes. Indeed, previous work has shown that walking at faster speeds improves the biomechanical positioning of the paretic trailing limb, 42 which is an important determinant of paretic propulsion.43,44 As such, comfortable speed treadmill training in faster participants is inherently different, and presumably more effective, than comfortable speed training in slower participants. Thus, we hypothesized that in participants with baseline maximum walking speeds slower than 1.2 m/s, FastFES training would be more effective than training at comfortable walking speeds. The proposed walking speed cutoff value of 1.2 m/s is based on previous work that has suggested that this value may be meaningful within the context of the energy cost of poststroke walking. 28 Indeed, members of our team have shown that in persons with baseline maximum walking speeds faster than 1.2 m/s, walking even faster does not produce a reduction in the energy cost of walking, but does for individuals that walk at slower speeds. 11
Methods
Participants
Participants (see Table 1) were recruited from a registry of individuals who had sustained a stroke and had expressed interest in participating in research studies. Individuals in this registry were recruited over 24 months from local health facilities and patient support groups. Figure 1 depicts participant flow from recruitment through analysis. Data collected from the participants in this study have been included in previous work15,33,45,46; however, the present study is the first to test the efficacy of FastFES on participants’ energy cost of walking and long-distance walking ability. Inclusion criteria included a single cortical or subcortical stroke, observable gait deficits, the ability to walk without orthotic support or the support of another individual for 6 minutes, passive ankle dorsiflexion to neutral with the knee extended, 10° of passive hip extension, and the ability to communicate with investigators and follow instruction. Participant exclusion criteria included cerebellar stroke, conditions other than stroke that limit walking ability, neglect or hemianopia, or unexplained dizziness during the prior 6 months. Written informed consent, physician medical clearance, and cardiac clearance based on a submaximal stress test were obtained for each participant prior to enrollment. All procedures were approved by the institutional review board of the University of Delaware. Testing was conducted pretraining, following 12 weeks of training (posttraining), and at a 3-month follow-up evaluation.
Participant Baseline Characteristics.
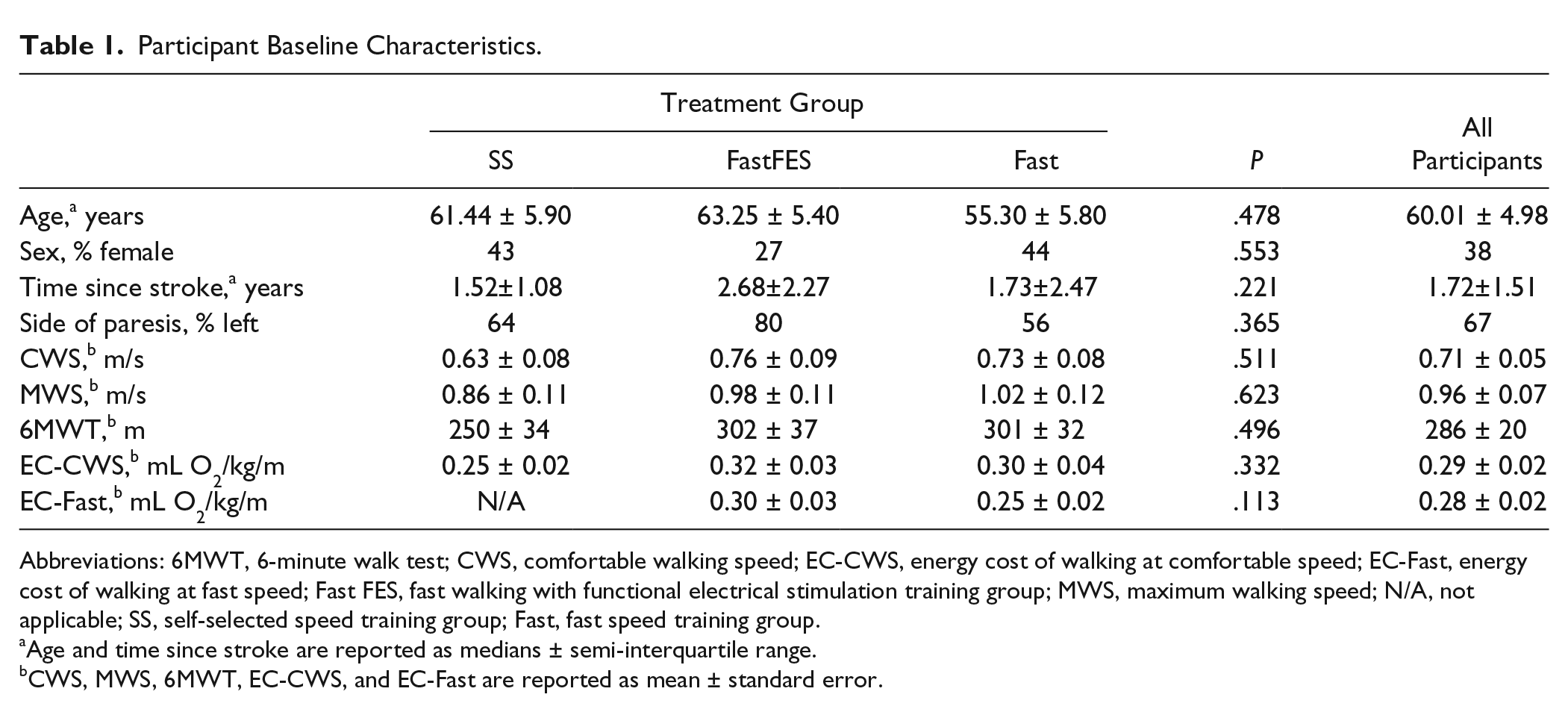
Abbreviations: 6MWT, 6-minute walk test; CWS, comfortable walking speed; EC-CWS, energy cost of walking at comfortable speed; EC-Fast, energy cost of walking at fast speed; Fast FES, fast walking with functional electrical stimulation training group; MWS, maximum walking speed; N/A, not applicable; SS, self-selected speed training group; Fast, fast speed training group.
Age and time since stroke are reported as medians ± semi-interquartile range.
CWS, MWS, 6MWT, EC-CWS, and EC-Fast are reported as mean ± standard error.
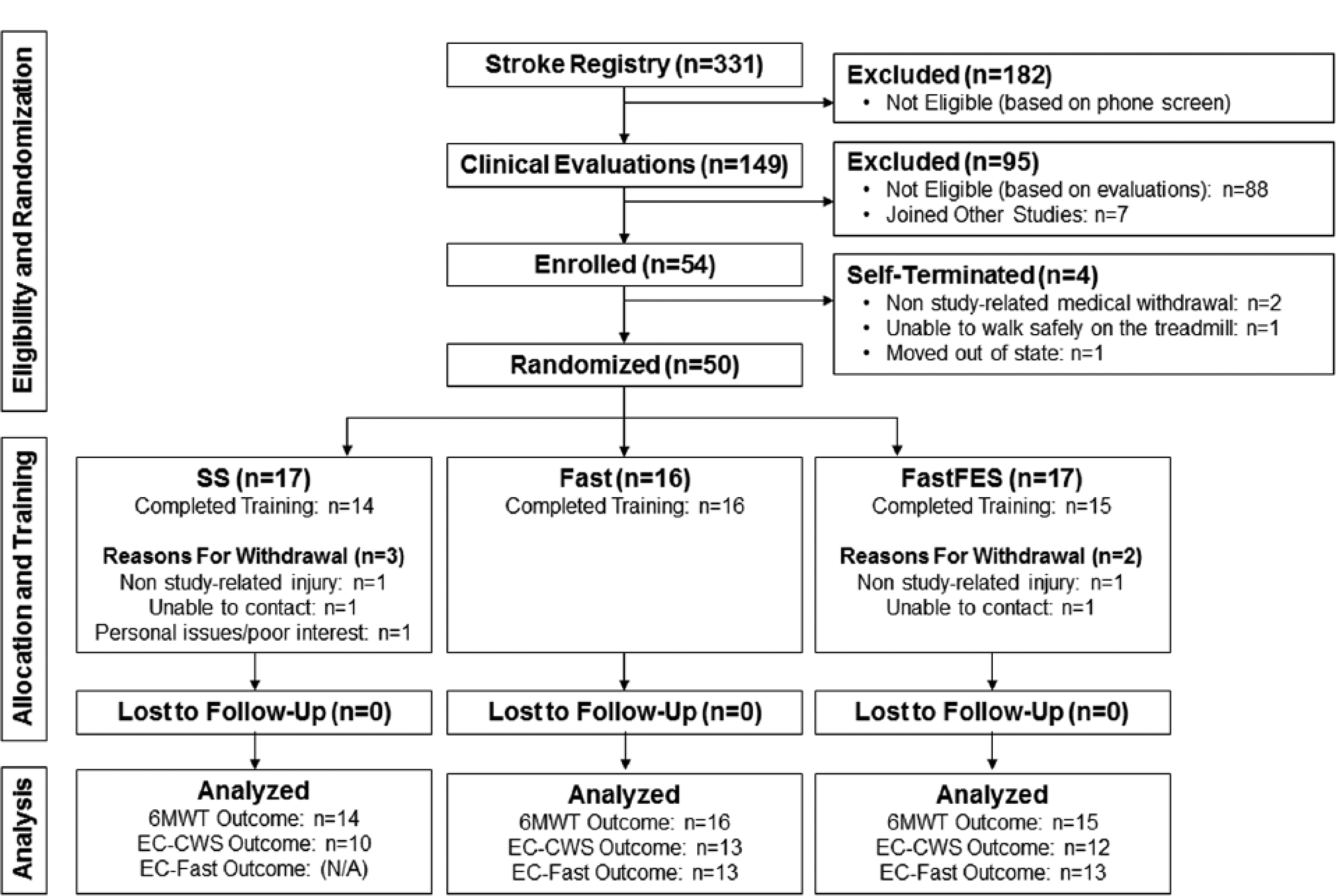
Schematic of participant flow from recruitment through analysis. 6MWT, 6-minute walk test; CWS, comfortable walking speed; EC-CWS, energy cost of walking at comfortable speed; EC-Fast, energy cost of walking at fast speed; Fast FES, fast walking with functional electrical stimulation training group; SS, self-selected speed training group; Fast, fast speed training group.
Outcomes
Licensed physical therapists blinded to treatment group conducted all evaluations. A reduction in the energy cost of walking at participants’ comfortable walking speed (EC-CWS) was the study’s primary outcome. The energy cost of fast walking (EC-Fast) and the distance (m) traveled during the 6-minute walk test 47 (6MWT) served as secondary outcomes.
Other Measures
Participants’ comfortable (CWS) and maximum (MWS) overground walking speeds (m/s) were measured using the 10-meter walk test 48 to provide an understanding of participants’ baseline level of function and to facilitate setting the speeds used for metabolic testing and training (see “Locomotor Training” section). Assistive devices were allowed during all evaluations, if necessary. Participants who used an assistive device at their pretraining evaluation also used the same assistive device during subsequent evaluations.
Measuring the Energy Cost of Walking (EC)
The methods utilized to measure EC have previously been described.15,19 Briefly, oxygen consumption (mL O2/min) was measured by a ParvoMedics TrueOne 2400 Metabolic Measurement System (Sandy, UT) as participants walked on a treadmill for 5 minutes at their overground CWS. Data from the fifth minute of walking were subsequently normalized to body weight (kg) and speed (m/min) to yield EC (mL O2/kg/m). For participants who trained at a fast walking speed (see “Locomotor Training” section), EC was also measured at their fast walking speeds.
Locomotor Rehabilitation
Participants were randomized to either 12 weeks of nontargeted or targeted locomotor training. The nontargeted walking programs were composed of walking training at either participants’ fast (Fast) or self-selected, comfortable (SS) walking speeds. The targeted walking program combined Fast with FES to the paretic ankle plantarflexors during late stance phase and dorsiflexors during swing phase (FastFES). Stimulation was triggered by 2 compression-closing foot switches attached to the sole of the paretic limb’s shoe. FES was provided in an alternating pattern of 1 minute on and 1 minute off during treadmill walking. Details regarding the FastFES system, parameters, and training have been described previously.19,28,36,37,49-51
Participants in all 3 groups received an equivalent dose of walking practice with training occurring at a frequency of 3 sessions per week, for a total of 36 sessions. Each session was composed of 5 bouts of 6 minutes of treadmill walking followed by 1 bout of 6 minutes of overground walking, for a total of 36 minutes of walking per session. Rest breaks were provided between bouts. An overhead harness was attached for safety, but body weight was not supported.
The targeted training speed for participants randomized to Fast and FastFES was their overground MWS. However, if their MWS was not a safe training speed or their MWS could not be maintained for at least 4 minutes on a treadmill, participants were allowed to start training at slower speeds. This initial training speed, however, was never slower than a participant’s overground CWS. Fast and FastFES training differed only in that 15 of the 30 minutes of treadmill walking practice were assisted by FES. The training speed for SS participants was their overground CWS. Participants’ overground speeds were regularly measured and training speeds were progressed accordingly over the 12 weeks of training (see Table 2 for initial and final training speeds).
Participants’ Overground and Training Speeds a .
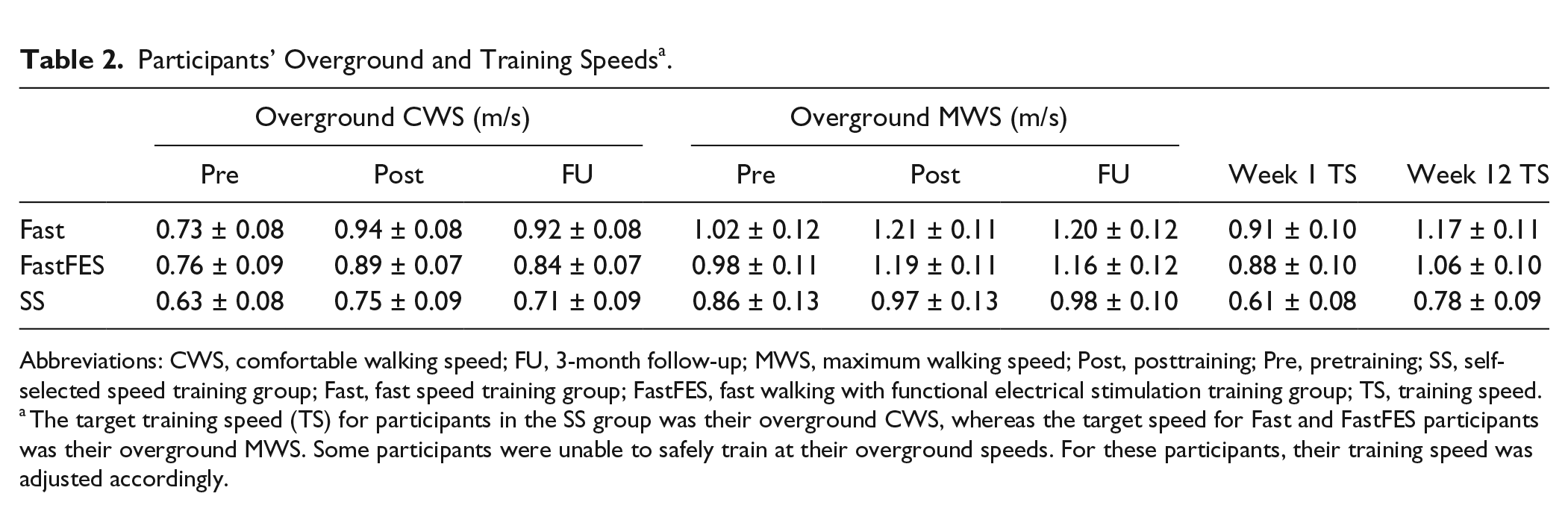
Abbreviations: CWS, comfortable walking speed; FU, 3-month follow-up; MWS, maximum walking speed; Post, posttraining; Pre, pretraining; SS, self-selected speed training group; Fast, fast speed training group; FastFES, fast walking with functional electrical stimulation training group; TS, training speed.
The target training speed (TS) for participants in the SS group was their overground CWS, whereas the target speed for Fast and FastFES participants was their overground MWS. Some participants were unable to safely train at their overground speeds. For these participants, their training speed was adjusted accordingly.
Statistical Analyses
Time × group mixed design analyses of variance (ANOVAs) were used to test within- and between-group effects. Based on our a priori hypothesis that participants’ baseline maximum walking speed (MWSbaseline) would influence treatment effects, a time × group × MWSbaseline interaction was added to each model. If an interaction with MWSbaseline was observed, post hoc moderated regression (as previously described15,33,52,53) was planned to evaluate the interaction. If an interaction with MWSbaseline was not observed, a model testing the main effects of time, group, and the time × group interaction was interpreted. Additionally, based on prior work demonstrating that simply walking faster can reduce EC, 11 the EC-CWS and EC-Fast models adjusted for the posttraining change in participants’ comfortable and fast walking speeds, respectively. These analyses thus evaluate training-induced changes in EC independent of changes in walking speed, and can therefore speak to the unique effects of FastFES training. With N = 45 and α = .05, the EC ANOVAs were 80% powered to detect a medium effect (Fcrit = 2.75, Cohen’s f = 0.25). Additionally, the number of participants that surpassed the 6MWT minimal detectable change (MDC) score of 54.1 m 54 after training was compared between groups using a chi-square test. With N = 45 and α set to .05, this analysis was 80% powered to detect a medium effect, χ2(2)crit = 5.99, φ = 0.46.
All analyses were performed using SPSS version 22. All descriptive statistics are reported as mean and standard error (M ± SE). All statistical assumptions were tested. Specifically, Box’s M was used to test for homogeneity of covariance matrices and Maulchy’s W tested the assumption of sphericity with a Huyn-Feldt adjustment applied if violated. The Shapiro-Wilk test was run on model residuals to test the assumption of normality with outliers removed as indicated. Homoscedasticity in the models was examined through residual plots. Bonferroni adjustments were used for all post hoc multiple comparisons.
Results
Sixty-one (18.4%) of the 331 individuals screened were eligible based on the study’s inclusion and exclusion criteria (see Figure 1). Seven participants joined other ongoing studies, 1 was unable to walk safely on the treadmill and was therefore also not eligible, and 3 withdrew prior to randomization. The remaining 50 individuals were randomized into the 3 training groups. Five of these individuals withdrew during the training period for reasons not related to the study. No participants who completed training were lost to follow-up. The 3 groups did not differ in baseline characteristics (see Table 1).
Complete 6MWT datasets were available; however, due to technical and logistical issues during data collection, EC-CWS and EC-Fast data were not available for some participants. Specifically, complete EC-CWS data were available for n = 35 (FastFES n = 12, Fast n = 13, and SS n = 10). EC-Fast data, which were only collected for FastFES and Fast participants, were available for n = 26 (FastFES n = 13 and Fast n = 13). See Figure 2 for unadjusted data for each of these 3 outcomes at each time point.
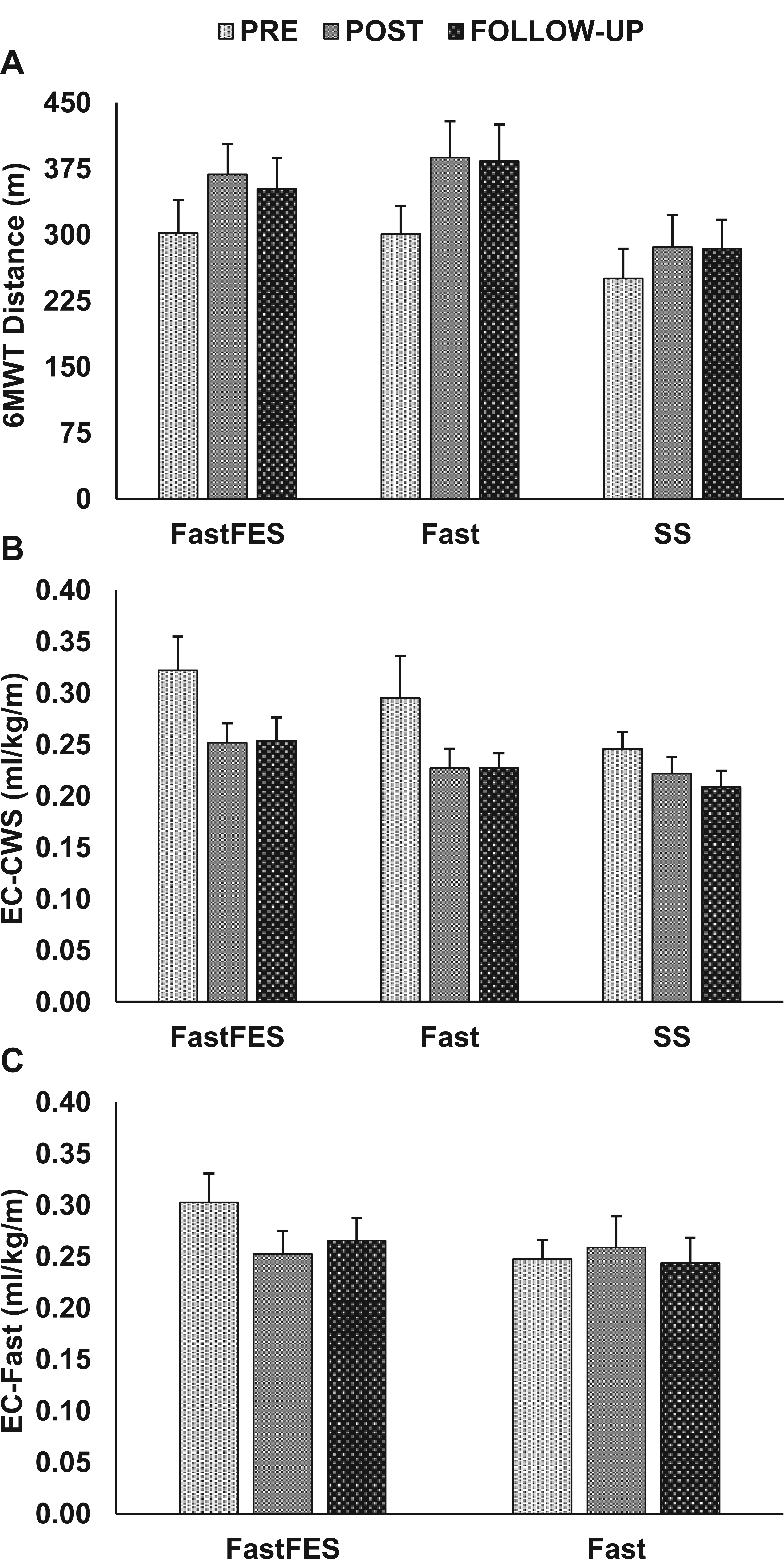
Pretraining (PRE), posttraining (POST), and 3-month follow-up data are presented for the 3 outcomes of interest: (A) 6-minute walk test (6MWT) distance, (B) energy cost of walking at a comfortable walking speed (EC-CWS), and (C) energy cost of walking at a fast walking speed (EC-Fast). Means ± standard error are presented for each time point. See text for results of statistical analyses.
Energy Cost at Comfortable Walking Speed (Primary Outcome)
The model evaluating the effect of treatment group over time on participants’ EC-CWS, while adjusting for changes in comfortable walking speed, revealed a significant group × time interaction, F(3.57, 53.59) = 2.93, P = .03, and main effect of group, F(2, 30) = 5.11, P = .01, but no main effect of time, F(1.79, 53.59) = 2.06, P = .14. Evaluation of the group × time interaction revealed that only FastFES produced a posttraining improvement in EC-CWS (24% average reduction, 0.08 ± 0.02 mL O2/kg/m, P < .001, see Figure 3A). The SS and Fast groups produced nonsignificant reductions in EC-CWS of 0.03 ± 0.02 mL O2/kg/m (P = .30) and 0.02 ± 0.02 mL O2/kg/m (P = .71), respectively. Posttraining changes in EC-CWS were retained at follow-up (posttraining to follow-up Ps >.05). The model evaluating moderation of these results by participants’ pretraining walking speed failed to find a significant interaction, F(4, 56) = 2.37, P = .06.
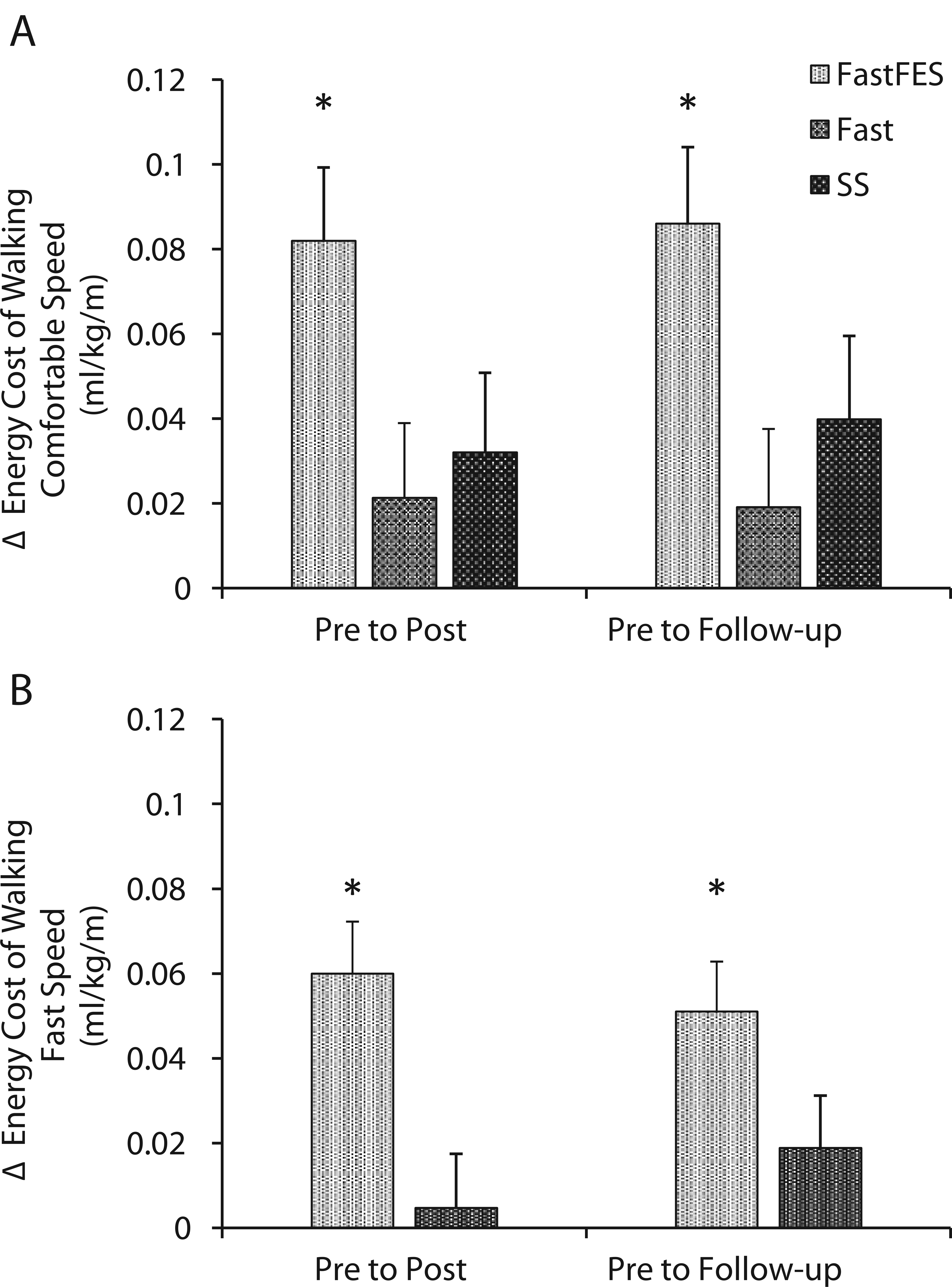
Changes in the energy cost of walking at (A) comfortable and (B) fast speeds, after adjusting for changes in walking speed, are presented (y-axis) for each training group. Only FastFES (fast walking with functional electrical stimulation) training reduced the energy cost of walking independent of changes in walking speed. *P < .05.
Energy Cost at Fast Walking Speed and 6MWT Distance (Secondary Outcomes)
Similarly, the model evaluating the effect of treatment group over time on participants’ EC-Fast, while adjusting for changes in fast walking speed, revealed a group × time interaction, F(1.70, 37.40) = 6.32, P = .006. Main effects of group, F(1, 22) = 2.36, P = .14, and time, F(1.70, 37.40) = 0.61, P = .52, were not significant. Similar to EC-CWS, only FastFES reduced EC-Fast (19% average reduction, 0.06 ± 0.01 mL O2/kg/m, P < .001, see Figure 3B). This reduction was retained at follow-up (posttraining to follow-up P = .58). The model evaluating moderation of these results by participants’ pretraining walking speed failed to find a significant interaction, F(1.50, 31.40) = 1.04, P = .35.
The model evaluating the effect of treatment group over time on participants’ 6MWT distance revealed a main effect of time, F(1.81, 75.89) = 26.22, P < .001, but no main effect of group, F(2, 42) = 1.55, P = .22, or group × time interaction, F(3.61, 75.89) = 1.64, P = .18. Across the 3 training groups, a mean difference of 63 ± 9 m was observed posttraining (P < .001) and retained at follow-up (P = .99). Despite the lack of a group × time interaction, it should be noted that only the Fast and FastFES groups produced average changes larger than the MDC (see Figure 2A). Moreover, the chi-square test comparing the number of 6MWT responders after training—that is, those with a 6MWT change larger than the MDC—between groups revealed that only 35% (5/14) of SS participants were responders, whereas 73% (11/15) of FastFES and 68% (11/16) of Fast participants were responders. However, this difference did not reach significance, χ2(2) = 5.06, P = .08.
Discussion
This study evaluated the effects of a poststroke locomotor intervention targeting deficits in paretic propulsion through the novel combination of fast walking training with functional electrical stimulation to the paretic ankle musculature (FastFES). As hypothesized, compared with gait training at comfortable or fast speeds without FES, FastFES produced markedly larger reductions in the energy cost of poststroke walking that were independent of changes in walking speed. Despite FastFES’ unique effects on walking efficiency, there were no differences amongst groups in the ability to walk farther distances during the 6MWT. A major implication of this finding is that in persons in the chronic phase of stroke recovery, targeting specific locomotor impairments during rehabilitation may produce changes in walking function that are fundamentally different from the changes that are produced by nontargeted rehabilitation. We have previously posited that by targeting deficits in the paretic limb’s ability to generate propulsive force, the FastFES intervention would improve paretic limb neuromotor function during walking, ultimately reducing the metabolic expense of walking after stroke.28,36,55 The present results thus provide evidence validating the FastFES theoretical framework and support further study of the therapeutic effects of FES-based gait training.
Prior to training, participants in this study consumed approximately 61% more oxygen per meter ambulated during walking at their comfortable pace than healthy older adults (0.29 ± 0.02 vs 0.18 ± 0.002 mL O2/kg/m). 56 The 24% average reduction in the energy cost of walking at comfortable speed observed following FastFES training was thus a substantial change. Moreover, considering that the functional resources used by persons poststroke to increase walking speed are characterized by metabolically expensive compensatory strategies,57,58 the fact that FastFES also produced a 19% reduction in the energy cost of fast walking, whereas an equivalent dose of training without FES did not, provides additional evidence supporting its use during gait retraining. A follow-up study of the biomechanical mechanisms underlying the changes in EC observed following FastFES training is warranted to inform the development of other targeted gait interventions.
It is interesting that the unique effects of FastFES training on EC did not translate to FastFES participants walking farther distances during the 6MWT compared with Fast and SS participants. This disconnect suggests that the 6MWT may not be sensitive to changes in EC. That is, similar to walking speed (see Introduction), persons poststroke may improve 6MWT performance through metabolically expensive compensatory mechanisms. Indeed, previous work from our laboratory has shown that posttraining changes in 6MWT distance highly relate to changes in maximum walking speed 45 and previous investigators have concluded that the 6MWT may be redundant to measures of short-distance walking performance, such as the 10-meter walk test, for classifying patients.59,60 Ultimately, the findings of this investigation suggest that EC measures an aspect of poststroke walking different from the 6MWT and that both may be important to measure when trying to understand the effects of a targeted gait intervention.
The value of making poststroke walking more efficient may be questioned given the finding that more efficient walking does not necessarily translate to farther distances walked during the 6MWT. However, it is important to note that the 6MWT only moderately correlates to community participation after a stroke.61-63 As such, investigating the effects of gait training that influences factors not captured in the measured distance walked during the 6MWT, but are associated with community participation, is worthwhile. EC has been posited to be a major determinant of community participation in individuals with neurologically-based walking deficits.18,64,65 Moreover, in older adults with walking deficits, EC has been shown to explain variance in self-reported function even after adjusting for comorbidity and walking speed. 66 Prior work suggests that substantial effort during functional tasks will yield reduced physical activity67-69; however, this phenomenon may not be reflected in a timed walk test that instructs individuals to walk as far as possible with the only qualification being to do so safely. That is, walking performance in ecological contexts may be markedly different than what is observed during the 6MWT because individuals may not engage in long-distance walking-related activities that would necessitate frequent rests. EC, in contrast, has been posited to be a variable able to bridge the disconnect between clinical measures of walking function and real-world walking performance, 18 and may do so through its sensitivity to the interaction between walking speed and gait parameters. 15 The substantially larger reduction in EC observed after FastFES training is thus noteworthy despite FastFES training not having a differential effect on 6MWT performance. A better understanding of how a reduction in EC after a stroke translates to changes in community walking participation will be important before the full value of interventions capable of reducing EC can be appreciated. More specifically, evaluating the effects of FastFES training on aspects of daily walking activity that are likely to benefit from reduced physiological effort during walking—for example, the number of long-distance walks taken per day and self-perceived walking-related disability—would be a worthwhile direction for future study.
For a heterogeneous population such as those poststroke, a better understanding of how baseline characteristics influence the effects of intervention will be critical for the translation of hypothesis-driven interventions, such as FastFES, to clinical practice. In the present study, we observed that participants’ baseline walking speed did not influence the effects of the 3 training programs studied. Further study of how other baseline characteristics—such as self-efficacy or gait mechanics—influence targeted gait training would advance individualized rehabilitation after stroke.
It is important to note that all 3 of the interventions studied produced improvements in the 6MWT that were comparable to, and often larger than, the gains reported by recent intervention studies. Indeed, a recent randomized controlled trial studying the effects of 3 months of fast overground training reported a 6MWT gain of only 34.5 m. 70 Similarly, recent systematic reviews of treadmill and body weight support training 71 and mixed cardiorespiratory and strength training 72 reported pooled mean differences of only 30.6 and 41.6 m, respectively. Likewise, a meta-analysis examining the effects of strength training after stroke reported a 28-m gain in the 6MWT. 73 Even the top performing group in the STEPS randomized controlled trial, which received body-weight supported locomotor training combined with a lower extremity strength program, achieved only a 45.3-m average gain following training. 74 In contrast, a 36-m average gain was observed in the SS group, an 87-m gain was observed in the Fast group, and a 67-m gain was observed in the FastFES group. It is also important to note the 89-m average gain observed in a preliminary study of the FastFES intervention. 28 Our failure to demonstrate between-group differences in the average 6MWT gain resulting from training thus appears to be the result of the superiority of the control interventions tested, not the ineffectiveness of the FastFES intervention. The rehabilitation promise of the FastFES intervention is strongly supported by its ability to reduce the energy cost of poststroke walking at both comfortable and fast walking speeds.
Limitations
This study had several limitations. First, although evaluators were blinded to participants’ training group, participants were not blinded. Additionally, factors other than participants’ baseline walking speed may influence the effects of training and will need to be explored in future work. Moreover, the chi-square test on the frequency of 6MWT responders was likely underpowered. A study with a larger sample size may be needed to observe a significant effect. Finally, to validate fully the FastFES theoretical framework, the biomechanical mechanisms underlying the changes in energy cost observed must be determined.
Conclusions
Combining fast locomotor training with functional electrical stimulation is an effective rehabilitation approach for reducing the energy cost of poststroke walking. The lack of commensurate between-group difference in participants’ 6MWT performance suggests that for individuals in the chronic phase of stroke recovery, the 6MWT may not measure the same aspects of poststroke walking impairment that the energy cost of walking does. A closer inspection of the relationship between 6MWT performance and the energy cost of walking is thus warranted. Moreover, clinical trials investigating the efficacy of targeted poststroke gait interventions may not be well-served by gross measures of walking function, such as the 6MWT. A better understanding of how reductions in the energy cost of walking contribute to improved community walking participation is needed.
Footnotes
Acknowledgements
We would like to thank Margaret Roos, PT, PhD, and Tamara Wright, PT, DPT for assistance with training and data collection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the following National Institutes of Health grants: R01NR010786, K01HD050582, U54GM104941, P30GM103333, and T32HD007490, as well as an American Heart Association fellowship: 15POST25090068.
