Abstract
Background
Dopaminergic medication improves gait in people with Parkinson disease (PD). However, it remains unclear if dopaminergic medication modulates cortical activity while walking.
Objective
We investigated the effects of dopaminergic medication on cortical activity during unobstructed walking and obstacle avoidance in people with PD.
Methods
A total of 23 individuals with PD, in both off (PDOFF) and on (PDON) medication states, and 30 healthy older adults (control group [CG]) performed unobstructed walking and obstacle avoidance conditions. Cortical activity was acquired through a combined functional near-infrared spectroscopy electroencephalography (EEG) system, along with gait parameters, through an electronic carpet. Prefrontal cortex (PFC) oxygenated hemoglobin (HbO2) and EEG absolute power from FCz, Cz, and CPz channels were calculated.
Results
HbO2 concentration reduced for people with PDOFF during obstacle avoidance compared with unobstructed walking. In contrast, both people with PDON and the CG had increased HbO2 concentration when avoiding obstacles compared with unobstructed walking. Dopaminergic medication increased step length, step velocity, and β and γ power in the CPz channel, regardless of walking condition. Moreover, dopaminergic-related changes (ie, on-off) in FCz/CPz γ power were associated with dopaminergic-related changes in step length for both walking conditions.
Conclusions
PD compromises the activation of the PFC during obstacle avoidance, and dopaminergic medication facilitates its recruitment. In addition, PD medication increases sensorimotor integration during walking by increasing posterior parietal cortex (CPz) activity. Increased γ power in the CPz and FCz channels is correlated with step length improvements achieved with dopaminergic medication during unobstructed walking and obstacle avoidance in PD.
Introduction
Gait impairment is a disabling symptom in Parkinson disease (PD) 1 and is characterized by reduced step length and gait speed as well as increased step width, double support time, and step-to-step variability. 1 Previous studies have indicated an exacerbated hypometric and bradykinetic gait pattern during obstacle avoidance in people with PD, consisting of smaller step length, slower gait speed, and greater gait variability during the steps preceding obstacle avoidance (approach phase).2-5 Furthermore, tripping over obstacles has been identified as a major cause of falls in PD.6,7
Safe locomotion through cluttered environments requires the use of cognitive and motor resources to examine the environment, plan an action, and adapt the locomotor pattern to avoid obstacles.8,9 Studies involving functional near-infrared spectroscopy (fNIRS) reported increased oxygenated hemoglobin (HbO2) concentration (ie, a proxy for greater cortical activation) in the prefrontal cortex (PFC) during obstacle avoidance compared with unobstructed walking for both healthy older adults and people with PD.10-13 This suggests that obstacle avoidance requires greater cognitive demand for motor planning.10-12 Moreover, previous studies using electroencephalography (EEG) have shown that electrocortical activity varies according to the locomotor task.14-19 For example, α (9-13 Hz) and β power (14-30 Hz) in the motor/sensorimotor cortical area decreased when walking stability is reduced, such as walking on a balance beam versus treadmill walking, 18 active versus passive robot-assisted walking, 17 normal walking versus stabilized walking, 19 and precision stepping. 16 Additionally, inclined walking, 15 walking on a balance beam, 18 and gait adaptations to visual feedback 14 decreased γ band power (31-50 Hz) in sensorimotor areas. These findings suggest that changes in EEG spectral power in the motor/sensorimotor areas are related to motor adjustment to maintain stability during walking.15,18,19 Therefore, EEG is a relevant method to describe the involvement of motor/sensorimotor cortical areas in gait control.14,15
Recent technologies allow concomitant recordings of electrocortical (EEG) and hemodynamic (fNIRS) activities, representing suitable tools to advance our understanding of brain dynamics during locomotion. Although previous studies have observed a reduction of α, β, and γ power in motor/sensorimotor cortical areas during higher balance demand tasks,14-19 there is a lack of research on electrocortical activity during obstacle avoidance in PD. Moreover, studies on PFC hemodynamic response during obstacle avoidance in people with PD were conducted while on medication only. Dopaminergic therapy increases functional connectivity within regions of the cognitive corticostriatal pathway, which is impaired in PD, contributing to effective motor performance. 20 In fact, some gait parameters, such as step length and velocity, are improved by dopaminergic medication.21,22 However, the effect of dopamine on cortical activity during real walking remains poorly understood. In this context, we investigated the effects of dopaminergic medication on gait and cortical activity using both fNIRS and EEG simultaneously during unobstructed walking and obstacle avoidance in people with PD. In addition, to investigate the potential involvement of cortical areas in gait improvement achieved with dopaminergic medication in PD, we examined the association between gait and cortical responses to medication.
It is known that administration of dopaminergic medication restores functional connectivity across subdivisions of the striatum as well as communication across corticostriatal circuits in PD. 23 In addition, we recently demonstrated that dopaminergic medication facilitates PFC activation during dual-task walking in people with PD. 24 Thus, we hypothesized that people with PD in the off state would not increase PFC activity during obstacle avoidance (relative to unobstructed walking), whereas both people with PD in the on state and healthy older adults would present increased PFC activity. Because PD involves excessive GABAergic inhibition in thalamocortical neurons, which leads to slower cortical activity,25,26 we expected that people with PD in the off state would present higher power at lower EEG frequencies (α power) and lower power at higher EEG frequencies (β and γ) in motor/sensorimotor areas during walking when compared with healthy older adults. Furthermore, we expected that dopaminergic medication would increase EEG frequency power in motor/sensorimotor areas during walking. In addition, we hypothesized that both people with PD (regardless of the medication state) and healthy older adults would present reduced α, β, and γ band spectral power in motor and sensory areas during obstacle avoidance compared with unobstructed walking because these bands are involved in the control of stability and gait adaptation.18,19 Finally, we expected that changes in gait performance after medication intake would be related to alterations in cortical activity.
Methods
Participants
A total of 23 individuals with PD and 30 healthy older adults (control group [CG]) participated in this study. All participants were >60 years of age, independent walkers, and free of cognitive impairment (Mini Mental State Examination [MMSE] >24 27 ), neurological disorders (other than PD), and musculoskeletal, orthopedic, and/or visual impairments that could affect their performance. People with PD had a clinical diagnosis of idiopathic PD, according to the UK Brain Bank criteria, at Hoehn and Yahr stage I to III, and were taking PD medication (levodopa). This study was approved by the local research ethics committee. All participants provided written informed consent to participate in this study.
Experimental Setup
People with PD were tested on 2 separate days, 1 week apart, and at the same time. They were tested once after taking their regular antiparkinsonian medication (on state: approximately 1 hour) and once after overnight withdrawal (at least 12 hours) from their PD medication (off state). The order of testing (on/off) was randomized and counterbalanced across participants. The CG was assessed once.
The MMSE was used to assess global cognitive function 27 and was performed once (for people with PD, when on medication). The motor section of the Movement Disorders Society United Parkinson’s Disease Rating Scale (MDS-UPDRS-III) was obtained from people with PD in both on and off medication states. 28 In addition, the levodopa equivalent daily dose (LED) was calculated. 29 The MMSE was used to characterize both people with PD and the CG. The MDS-UPDRS-III and LED were used to characterize the people with PD.
Gait and cortical activation were assessed in 2 self-paced walking conditions: (1) unobstructed walking and (2) obstacle avoidance. Each condition started with 30 s in a quiet stance, during which participants were required to look straight ahead and think about nothing. A verbal sign indicated the participant should start walking along a 26.8-m circuit for 30 s, with two 7-m parallel straights. In the obstacle avoidance condition, 4 foam obstacles were evenly spaced along the walking path (Figure 1). Five trials for each condition were performed in random order. All participants completed the 26.8-m circuit lap in 30 s at least once in each trial.
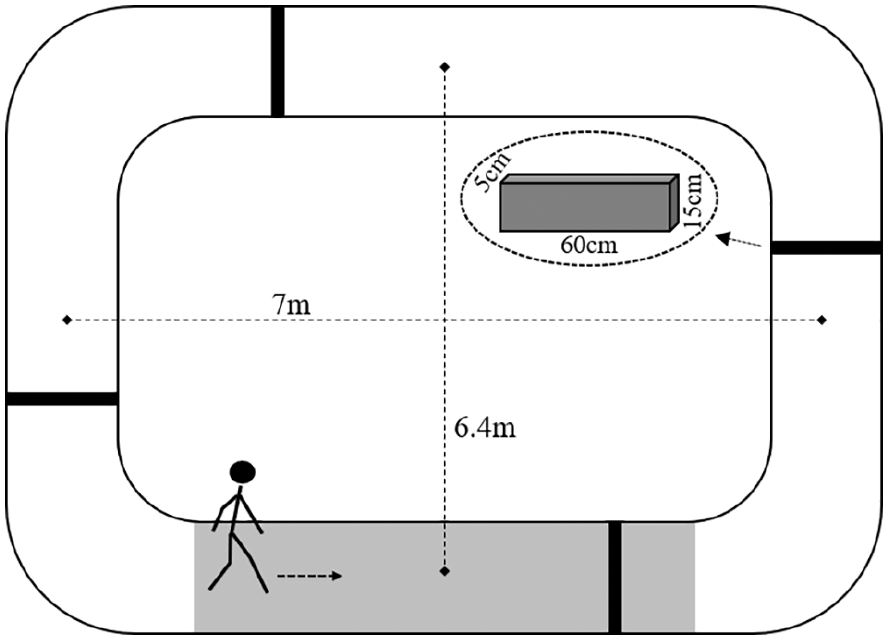
Experimental setup of obstacle avoidance condition. Black bars represent the obstacle position on the walkway and the gray area shows the GAITRite position. The arrow indicates the walking direction.
fNIRS Recordings and Processing
Changes in HbO2 concentration in the PFC were measured with a wireless 8-channel (35 mm of interoptode distance) OctaMon fNIRS system (Artinis Medical Systems, Netherlands), with a 10-Hz sampling rate. The Oxysoft 3.0.52 software (Artinis Medical Systems, Elst, Netherlands) was used for data collection. The cap was positioned on the left and right sides of the forehead following the international 10-20 system. This location roughly targets left and right Brodmann’s areas 9 and 10, which represent the dorsolateral and anterior PFC. Oxysoft 3.0.52 software (Artinis Medical Systems, Elst, Netherlands) was used for data collection.
Processing of fNIRS followed previous recommendations. 30 We chose the HbO2 concentration because it is the most sensitive indicator of walking-related changes in cortical activity.31-33 The concentration of HbO2 was calculated using the modified Lambert-Beer law. The differential path factor was set to 6.61 for all participants because no data are available for adults aged 50+ years. The fNIRS data were preprocessed using NIRS-SPM open source toolbox for MATLAB (MathWorks, Natick, MA). 34 A low-pass filter (0.14 Hz) based on canonical hemodynamic response function was used to reduce the high-frequency noise.34,35 A wavelet-minimum description length detrending algorithm was applied to decompose NIRS measurements into global trends (ie, breathing, cardiac, vasomotion), hemodynamic signals (ie, HbO2 concentration), and uncorrelated noise components (ie, movement artifacts) as distinct scales.36-38 Preprocessed data were exported to MATLAB, where further data processing was performed using a customized algorithm. HbO2 concentration signals were averaged per region of interest (left and right PFC) and normalized by dividing them by corresponding signal amplitude (from minimum to maximum) value of the whole experiment.24,39 We averaged left and right PFC signals for subsequent analyses.10-13 Each of the 5 trials was divided into 2 phases: (1) a baseline period, 15 s before the instruction to start walking (the 5 s immediately before the instruction were excluded), and (2) experimental task, from 5 to 25 s walking. In this selected period, participants crossed approximately 4 obstacles (PD medication off state [PDOFF] range: 4-5; PD medication on state [PDON] range: 4-5; CG range: 4-6) in each obstacle trial. The baseline HbO2 concentration was subtracted from the HbO2 concentration during the experimental task.10,12,24
EEG Recordings and Processing
EEG and trunk accelerometry were recorded using a wireless Mobita 32-channel system (TMSi, Netherlands) sampled at 500 Hz. This is a gel-based system with passive electrodes. All processing and analysis were performed in MATLAB using an open-source toolbox for processing EEG data (EEGLAB 13.0.1b). 40 Initially, EEG data were segmented into gait cycles (epochs), according to the accelerometer data, which were processed following previous recommendations. 41 Briefly, acceleration data were transformed to a horizontal (fore-aft) and vertical coordinate system and low-pass filtered (20 Hz) with a fourth-order Butterworth filter. To estimate the initial and final contact events within the gait cycle, we used a continuous wavelet transform (CWT) of vertical acceleration, which was first integrated and then differentiated using a Gaussian CWT. The initial and final contacts were detected as the local minima and maxima of the CWT, respectively. Both right and left heel strike were identified as well as the crossing step (in obstacle avoidance condition).
Individual EEG data sets from both conditions were merged into a single data set, which were band-pass filtered (1-100 Hz) and had line noise removed (60 Hz). Channels with substantial artifacts throughout the recordings were removed using the following methods: (1) channels with magnitude <30 or >2000 µV, (2) channels with kurtosis >5 SDs from the mean, and (3) channels with SD substantially greater compared with neighboring channels.16,42 After rejecting channels, independent component analysis (RUNICA, from EEGLAB) was performed on the data set to identify and remove eye blinks and artifacts related to muscle activity.43-45 In addition, epochs were removed by visual inspection. These methods of rejecting motion-related artifacts during walking have been tested in previous studies and showed efficacy.42,46
Four steps were selected before obstacle crossing, and a matching number of epochs from the unobstructed walking condition was randomly created for comparison with the obstacle condition (26.47 ± 5.76 epochs). Three channels across all participants were selected (FCz, Cz, and CPz). These channels were chosen because they may capture information from brain regions related to motor planning (supplementary motor area [SMA]), motor execution (primary motor cortex; M1), and sensorimotor processing (posterior parietal cortex), respectively. The power spectrum from the selected EEG channels was computed across the time series representing the 4 steps prior to obstacle crossing (Hanning windowing, 1024 ms FFT; 512-ms point window). The absolute power of the power spectrum from each channel was averaged in the α (9-13 Hz), β (14-30 Hz), and low γ (31-50 Hz) frequency bands across the epoch time course.
Gait Assessment
Spatiotemporal gait parameters were measured using a 5.74-m GAITRite system (CIR Systems Inc, Clifton, USA), sampled at 200 Hz, placed over one straight segment of the circuit. This system was connected to a computer using GAITRite software version 4.7.5 (CIR Systems Inc, Clifton, USA) for recording, initial processing, and storing data. A customized MATLAB algorithm was used to calculate gait parameters. The final 4 steps before the obstacle (approach phase) and 4 steps of unobstructed walking were considered. Means and variability, calculated by the coefficient of variation ([SD/Mean] × 100 47 ) of step length, step duration, and velocity were calculated.
Statistical Analysis
The Kolmogorov-Smirnov test was used to examine data distribution. Variables that violated the assumption of normal distribution (EEG data and gait variability parameters) were log-transformed to improve normality of distribution. Variables were checked for the presence of outliers (defined as any value lower than (3× interquartile range) + first quartile or greater than (3× interquartile range) + third quartile).
Results
There was an expected improvement in motor symptoms (MDS-UPDRS-III scores) after medication intake. The PD group presented a lower MMSE score and fewer women than the CG (Table 1). However, no PD participants showed cognitive decline according to the MMSE score. All participants successfully avoided the obstacles, and people with PD did not present freezing of gait during the experimental task.
Participants’ Characteristics (Mean ± SD).
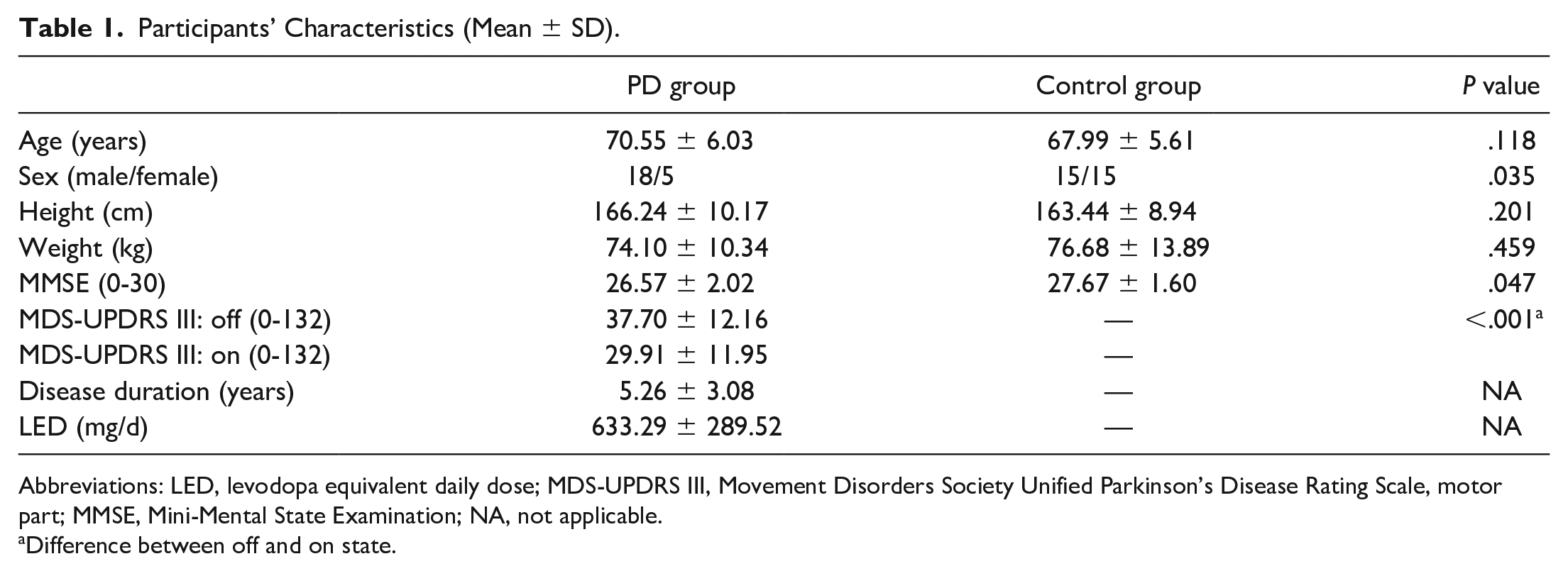
Abbreviations: LED, levodopa equivalent daily dose; MDS-UPDRS III, Movement Disorders Society Unified Parkinson’s Disease Rating Scale, motor part; MMSE, Mini-Mental State Examination; NA, not applicable.
Difference between off and on state.
Changes in PFC Activation: fNIRS Data
PDOFF Versus Control Group
There was a significant group versus condition interaction for HbO2 levels (
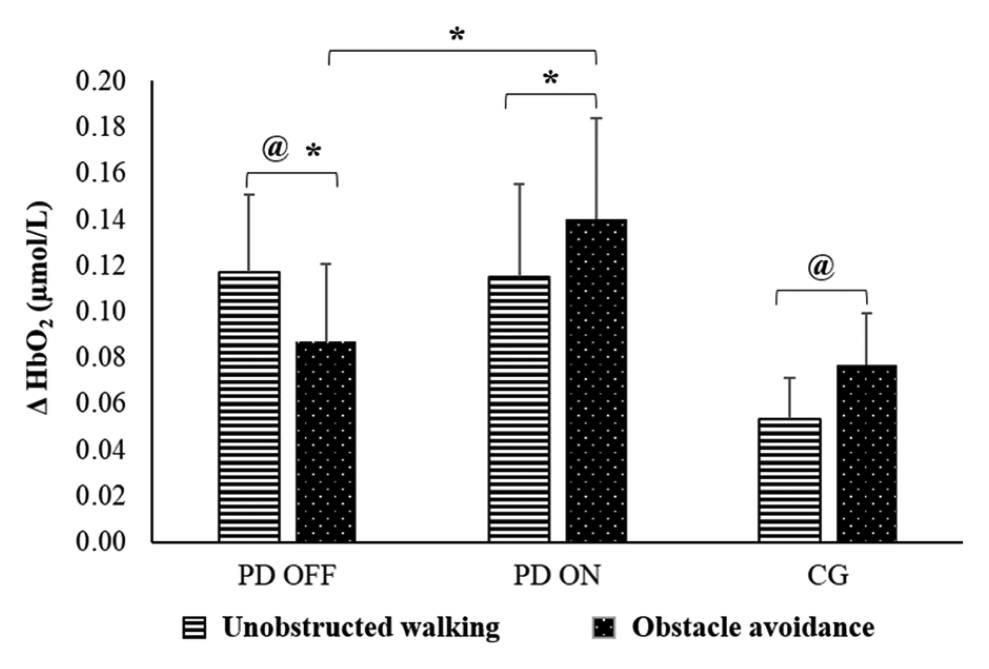
Means and standard errors of relative oxygenated hemoglobin (HbO2) during unobstructed walking and obstacle avoidance for people with PD in the off and on states and in the control group.a
PDOFF Versus PDON
There was a significant medication versus condition interaction for HbO2 levels (
EEG Absolute Power During Unobstructed Walking and Obstacle Avoidance
Data from 3 participants of the PD group and 1 of the CG were removed because of artifacts. Therefore, EEG data from 20 and 29 participants from the PD group and CG, respectively, were included in the analyses. Figure 3 presents the log-transformed EEG data. Greater negative values represent lower absolute power. Nontransformed EEG data are detailed in Supplementary Figure 1.
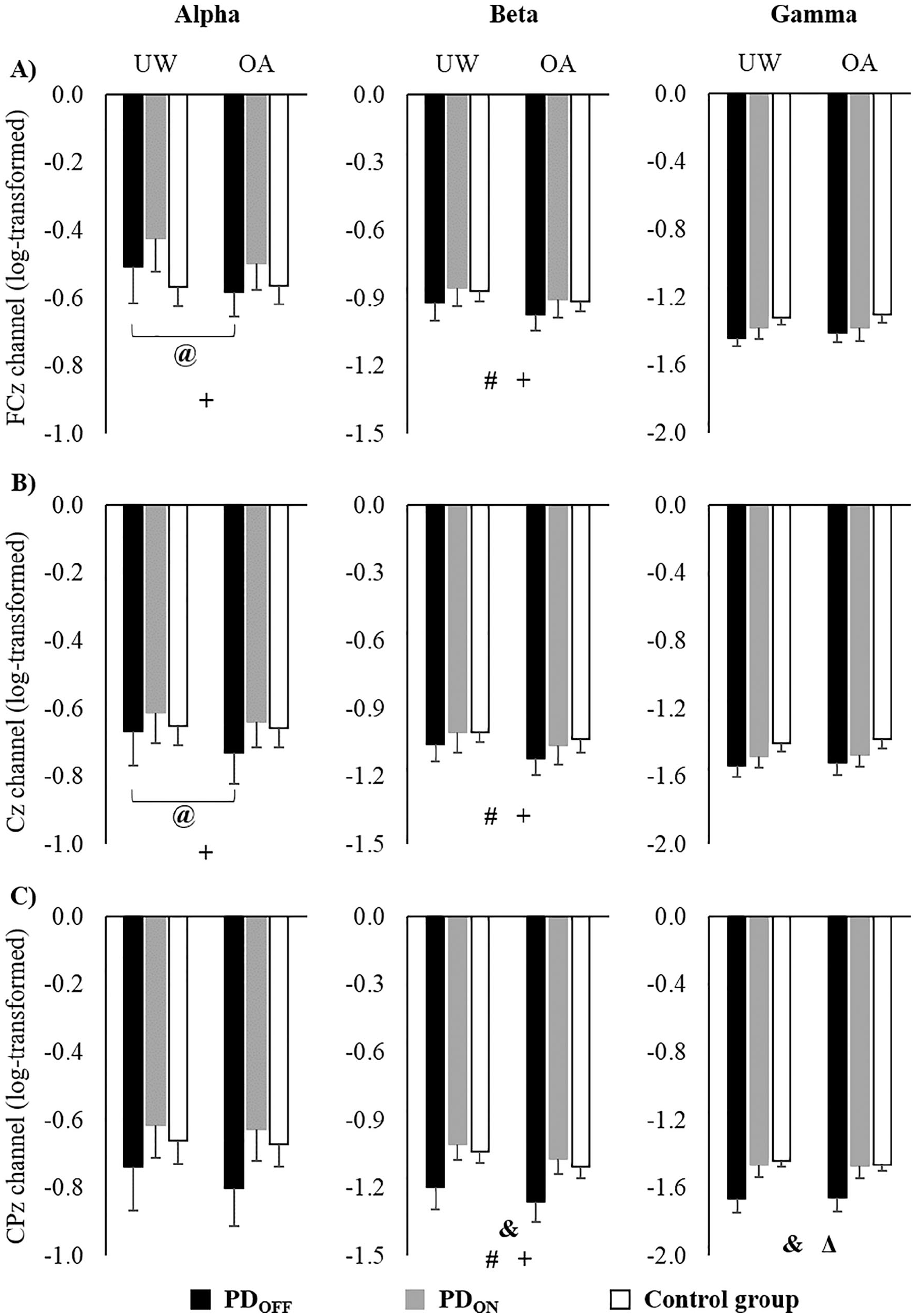
Means and standard errors of log-transformed EEG data of (A) FCz, (B) Cz, and (C) CPz channels during unobstructed walking (UW) and obstacle avoidance (OA).a
PDOFF Versus Control Group
The α Band
There was a significant interaction between group and condition for absolute power in the FCz (
The β Band
A main effect of condition was observed for FCz (
The γ Band
A main effect of group was found for CPz (
PDOFF Versus PDON
The α Band
A main effect of condition was observed, showing that in both medication states, people with PD presented decreased α power in the FCz (
The β Band
ANOVA revealed a medication effect for CPz (
The γ Band
A medication effect was observed for CPz (
Gait Behavioral Performance
PDOFF Versus Control Group
A main effect of group revealed that people with PD in the off state had smaller step length (
Gait Parameters of UW and Approach Phase of OA, by Group (Mean ± SD).
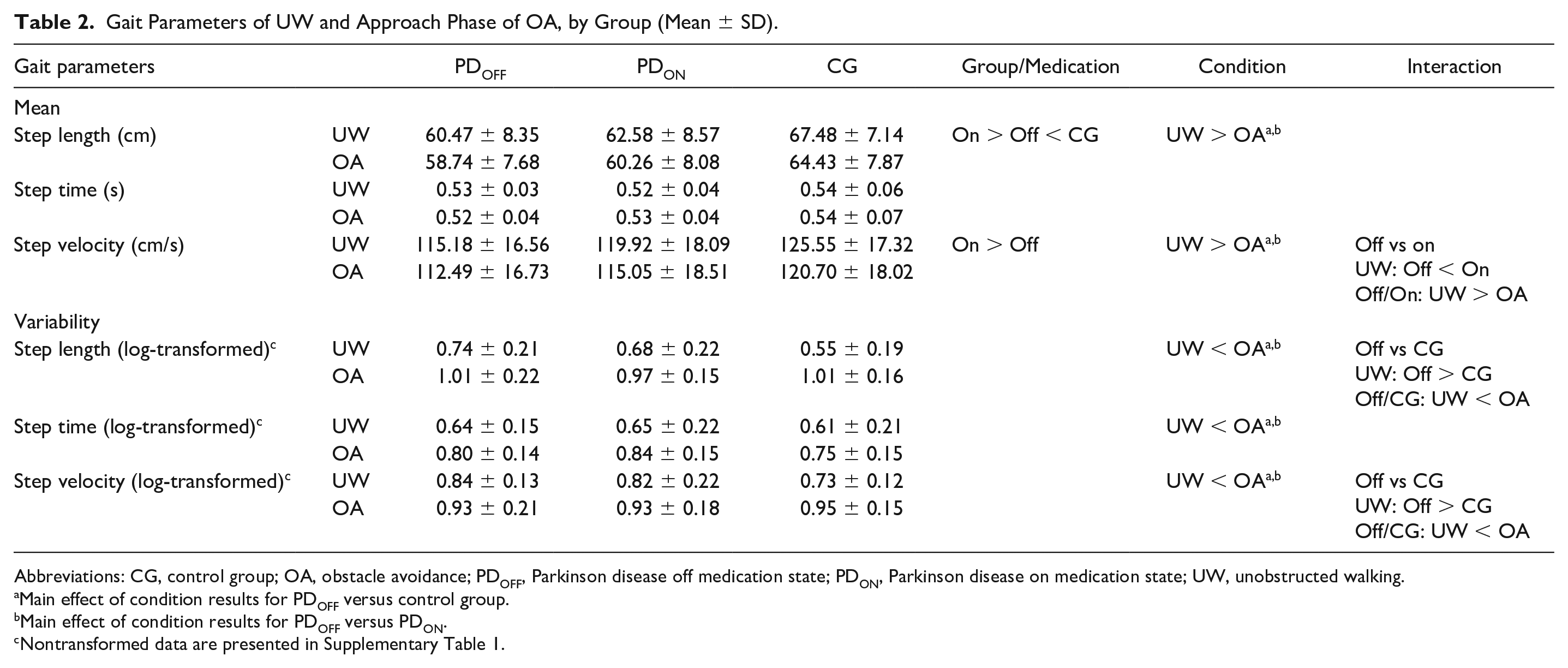
Abbreviations: CG, control group; OA, obstacle avoidance; PDOFF, Parkinson disease off medication state; PDON, Parkinson disease on medication state; UW, unobstructed walking.
Main effect of condition results for PDOFF versus control group.
Main effect of condition results for PDOFF versus PDON.
Nontransformed data are presented in Supplementary Table 1.
PDOFF Versus PDON
A main effect of medication showed that people with PD presented increases in both step length (
Associations Between Cortical Activity and Gait Parameters: Response to Medication (ΔON-OFF)
In the unobstructed walking condition, changes in HbO2 levels were negatively associated with changes in step length variability (
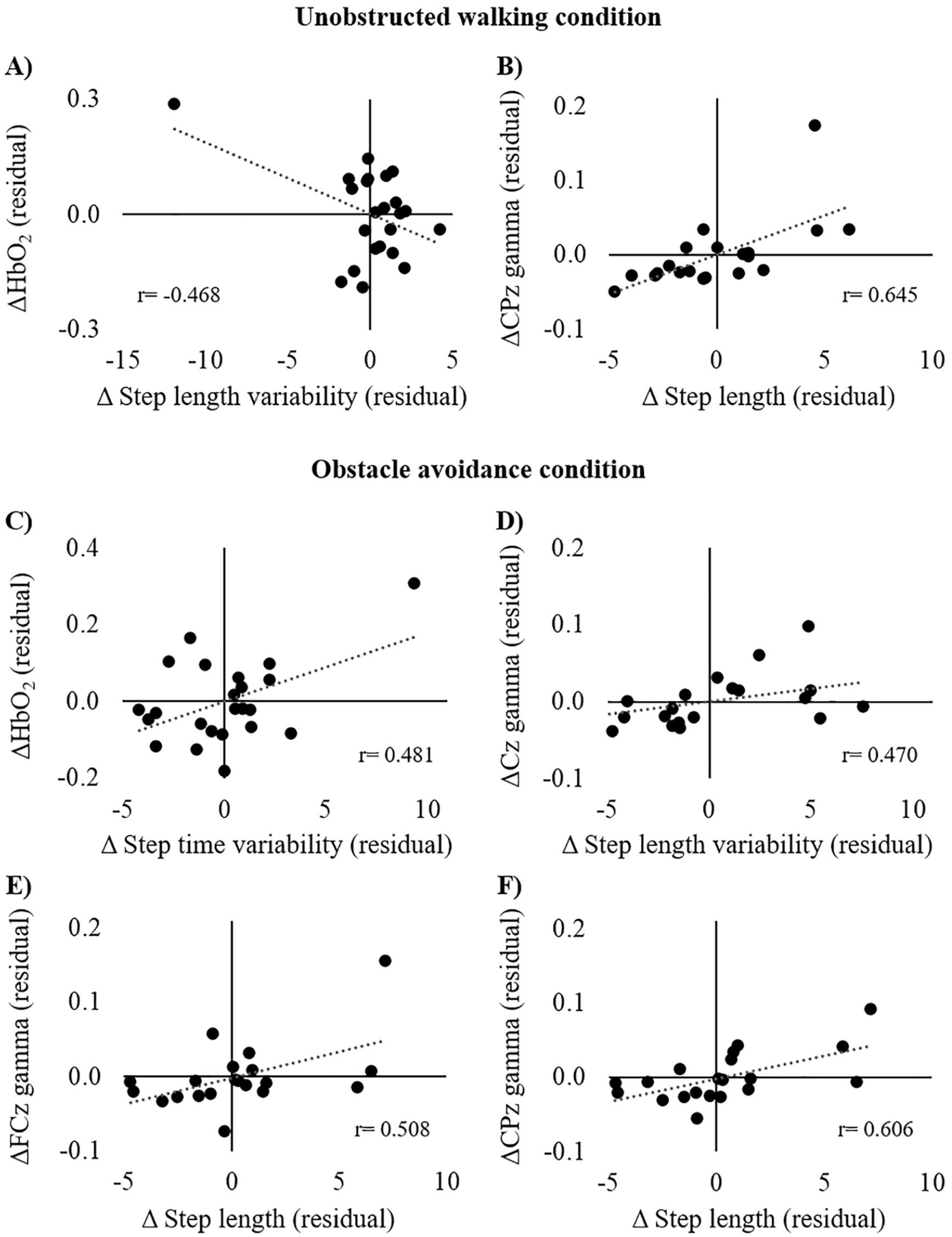
Partial correlation analysis between changes in cortical activity and gait parameters (ΔON-OFF) during (A-B) unobstructed walking and (C-F) obstacle avoidance condition adjusted for levodopa equivalent daily dose.
Discussion
This study investigated the effects of dopaminergic medication on cortical activity during unobstructed walking and obstacle avoidance in people with PD. Overall, our results demonstrated that dopaminergic medication increases the activation of the PFC during obstacle avoidance. Regardless of the medication state, people with PD presented reduced α and β power in FCz, Cz, and CPz during obstacle avoidance compared with unobstructed walking. We also observed increased β and γ power in CPz after levodopa intake. Moreover, alterations in the cortical activity after taking medication were associated with changes in gait in both unobstructed walking and obstacle avoidance conditions.
Effect of PD Medication on PFC HbO2
Dopaminergic medication facilitated the activation of the PFC during obstacle avoidance. Increased PFC activity while walking seems to be required because of reduced movement automaticity, 49 which is affected by ageing12,13,50 and to a greater extend by PD, 10 or increased cognitive demand of the task, such as obstacle avoidance.10-13 Our results suggest that PD (off state) affects the ability to activate the PFC to deal with obstacles placed in the path. People with PD in the off state reduced PFC activity from unobstructed walking to the obstacle condition. In contrast, increased PFC activation was observed during obstacle avoidance compared with unobstructed walking in the on state as well as in healthy participants. Previous studies have also reported greater PFC involvement and the allocation of cognitive resources to achieve satisfactory motor performance levels in obstacle conditions.10-12 Likewise, the higher gait variability in both healthy individuals and people with PD when avoiding obstacles suggests reduced automatic control of walking, making it necessary to implement compensatory mechanisms to complete the motor task.49,51
Increased PFC activity with medication intake may be related to reduced corticostriatal disruption because people with PD present disrupted balance between excitatory and inhibitory activity in the basal ganglia.52,53 GABAergic outputs of the basal ganglia in thalamocortical neurons may decrease cortical excitation, such as at the PFC.26,54 However, dopaminergic medication increases the availability of dopamine in the system, reestablishing functional connectivity across subdivisions of the striatum and improving communication across corticostriatal circuits.20,23,54 Taken together, our findings show that dopaminergic medication may facilitate PFC activation during challenging walking tasks. 24
Electrocortical Activity During Obstacle Avoidance
The reduced α and β power in FCz, Cz, and CPz, mainly for people with PD during obstacle avoidance, suggests greater cortical involvement of motor/sensorimotor areas in postural control. These channels may represent activity in the SMA, M1, and posterior parietal cortex, respectively. Previous studies have demonstrated that the power in either α or β bands in the sensorimotor cortices is reduced during complex motor actions, such as active walking compared with robot-assisted walking, 17 walking on a balance beam, 18 and normal walking compared with stabilized walking. 19 In addition, precision stepping tasks decrease electrocortical activity in sensorimotor areas. 16 Although the CG presented electrocortical modulation during obstacle avoidance (reduced β power in FCz, Cz, and CPz), greater electrocortical modulation was observed in people with PD regardless of medication state (reduced α power in FCz and Cz and reduced β power in FCz, Cz, and CPz). Taken together, our findings suggest that people with PD present reduced α and β power from the SMA, M1, and posterior parietal cortex to safely avoid obstacles during walking.
Interestingly, levodopa intake increased β and γ power in the CPz (related to posterior parietal cortex), which is involved in sensory integration and drives decision-related activities. 55 Activation in the parietal cortex has also been observed during visually guided gait modifications, suggesting that this may contribute to locomotor control.56,57 An EEG study demonstrated that the parietal cortex plays a role in planning limb trajectories and foot placement next to the obstacle. 58 Therefore, the higher β and γ spectral power in people with PD in the on state in our study may imply increased sensorimotor integration to control locomotion and successfully avoid obstacles. Thus, our results suggest that levodopa increases cortical activity in the parietal areas and ameliorates the impaired sensorimotor integration during walking in people with PD.
Cortical Activity Underlying Gait Improvements Achieved With PD Medication
It has been well documented that dopaminergic medication improves pace parameters during walking, such as step length and step velocity.21,22 In fact, we have also found longer and faster steps during walking with dopamine replacement in people with PD. However, these results may represent neither a reduced risk of falls nor an improvement of postural instability because these factors seem unresponsive to dopaminergic treatment. 22 For example, there were no improvements in gait variability, which has been related to increased risk of falls. 1 In addition, previous studies have reported that gait variables related to postural instability, such as step width and double support time, do not respond to dopaminergic medication.21,22 Thus, dopamine replacement may improve pace but not stability during walking. It has been suggested that cholinergic dysfunction, and not dopaminergic dysfunction, is associated with gait variability and instability (increased risk of falls). In support to such suggestion, complementary interventions with cholinergic augmentation (ie, rivastigmine and donepezil) have been shown to reduce gait variability and improve instability in people with PD.22,59 Future studies should further explore the effects of different treatments on gait and cortical activity in PD.
Our findings suggest that changes in cortical activity observed with dopaminergic medication play a role in dopaminergic-related improvements in gait. An increased HbO2 level after medication intake was related to a reduction in step length variability during unobstructed walking, suggesting that people with PD with higher PFC activity presented more consistent gait patterns during walking in the regular environment. This result indicates that dopaminergic medication may facilitate cognitive recruitment even in a regular environment, making the compensatory mechanism more efficient during walking. In addition, patients with a greater increase in step length after medication intake presented a greater increase in γ power in the CPz channels during unobstructed walking. This finding suggests that the posterior parietal cortex plays an important role in gait improvement achieved with dopaminergic medication.
Likewise, increased step length was associated with higher γ power in the CPz as well as FCz channels during obstacle avoidance as a consequence of medication intake. Therefore, improved gait performance in obstacle avoidance is related not only to posterior parietal cortex activation, but also SMA activation. Interestingly, while approaching the obstacle, increased γ power in the Cz channel and increased HbO2 levels after taking levodopa are associated with step length variability and step time variability, respectively. In environments with obstacles, individuals need to continuously perform adjustments during the approach phase to safely avoid the obstacle, which results in increased gait variability but without decreasing efficiency. 5 Therefore, activation of cognitive (PFC) and motor (M1) areas achieved with dopaminergic medication play an important role in performing gait adjustments while approaching obstacles in people with PD.
This study presents some limitations. First, we did not use short-separation channels to control the peripheral hemodynamic response. However, we applied Wavelet-MDL detrending to remove unknown global trends from our data.30,36 Additionally, the low number of EEG channels may impose a limitation to our conclusions. The use of high-density EEG (64 channels or more) would have allowed the extraction of independent components from participant data sets.15,19,58
Conclusion
In conclusion, PD compromises PFC activation during obstacle avoidance. In contrast, dopaminergic medication facilitates its recruitment, increasing the use of a compensatory mechanism to avoid obstacles. PD medication helps improve sensorimotor integration during walking by increasing posterior parietal cortex (CPz) activity. Dopaminergic-related changes in cortical activity, particularly increased γ power in the CPz and FCz channels, are involved in step length improvements achieved with dopaminergic medication during unobstructed walking and obstacle avoidance in PD.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683211000736 – Supplemental material for Cortical Activity Underlying Gait Improvements Achieved With Dopaminergic Medication During Usual Walking and Obstacle Avoidance in Parkinson Disease
Supplemental material, sj-docx-1-nnr-10.1177_15459683211000736 for Cortical Activity Underlying Gait Improvements Achieved With Dopaminergic Medication During Usual Walking and Obstacle Avoidance in Parkinson Disease by Diego Orcioli-Silva, Rodrigo Vitório, Priscila Nóbrega-Sousa, Victor Spiandor Beretta, Núbia Ribeiro da Conceição, Anderson Souza Oliveira, Marcelo Pinto Pereira and Lilian Teresa Bucken Gobbi in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank Ellen Lirani-Silva, PhD, Vinicius Cavassano Zampier, MSc, Vinicius de Belli, and Lucas Meira Fiorio for their assistance with data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES; Finance Code 001), Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP; Grant Numbers: 2017/19845-1; 2016/21499-1; 2014/22308-0), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; Grant Number: 429549/2018-0).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
