Abstract
Introduction
Huntington’s disease (HD) is associated with a range of motor and cognitive deficits, as well as psychiatric symptoms.1,2 Individuals with symptomatic HD (symp-HD) eventually experience impaired mobility and balance, which can lead to limited functional ability. 3 As the disease progresses, falls may become more pronounced and result in serious injuries that increase risk of mortality. 4 The specific underlying pathophysiology relating to mobility and balance is poorly understood; however, there is evidence of an association between basal ganglia dysfunction and increaseed motor deficits in symp-HD. 5
Several studies have provided anecdotal evidence suggesting tripping over obstacles is one of the major causes of falls in symp-HD. 6 Gait abnormalities are common in the early stages of the disease and worsen with disease progression. 7 Previous research has shown that the prominent motor symptoms of chorea, stride variability, bradykinesia, and cognitive decline, as well as behavioral changes, may also contribute to a disturbance in balance leading to falls. 8 Bradykinesia has been associated with reduced step-length and gait velocity in symp-HD, 9 as well as increased step-time, all of which increase the risk of tripping. 10 Moreover, a previous study, with an aged population, reported that underlying deterioration in visual, orthopedic, and cognitive systems may contribute to a decline in obstacle crossing performance, 11 which could also be a contributing factor in symp-HD.
To our knowledge, no previous research has investigated the characteristics of gait features in HD while navigating around obstacles. Most previous studies have been limited to examining gait variables during self-paced (or baseline) walking. We extended previous research to investigate how participants navigate
The overall aim of this study was to identify deficits in underlying spatiotemporal gait characteristics during obstacle navigation, in the hope that results would enable new insights into adaptive gait patterns in symp-HD. Given the exploratory nature of the study, we limited our investigation to 3 key spatiotemporal gait variables (step-length, step-time, and step-width) during 2 navigation obstacle conditions.
Methods
Participants
The study involved a total of 32 participants; 16 symp-HD and 16 age- and sex-matched healthy controls aged 41 to 65 years. Based on G-power analysis, with power set at 80%, α at .05, and a prior effect size of .76,
3
a sample of 44 (22 for each group) participants was needed. However, HD is a rare neurodegenerative condition and we experienced some difficulty in recruting further individuals for this study. Subsequently, recruitment of 16 participants for each group, equating to 72.7% of the estimated total sample size, was achieved. Participants were recruited from the “ENRU-STOUT HD Participant Database” located at Monash University, Melbourne, Australia. Symp-HD participants were screened according to inclusion criteria—confirmed CAG length >39 on 1 of the 2 alleles (on which the HD gene is located), as well as a motor score of >5 on the Unified Huntington’s Disease Rating Scale (UHDRS). For all participants, exclusion criteria included the presence of brain injury, other neurological or severe diagnosed psychiatric illness, pregnancy, dementia, the inability to understand instructions in English, and the inability to walk independently without a walking aid. Ten symp-HD participants were taking psychotherapeutic medications. Three were taking medication for high blood-pressure, 2 for heart disease, and 2 for diabetes. None of the control participants were taking medications at the time of the study. For each symp-HD participant, a disease burden score (DBS) was calculated using the formula: (CAG − 35.5) × age.
12
Participants were assessed for premorbid IQ (NART),
13
psychiatric disturbance (Beck Depression Inventory, Second Edition [BDI-II])
14
and anxiety levels (Beck Anxiety Inventory [BAI]).
15
Informed consent was obtained from all participants and the study was approved by the Monash University Human Research Ethics Committee. The demographic, clinical, and psychiatric characteristics of all participants are summarized in Table 1, along with effect sizes (
Group Difference With Independent
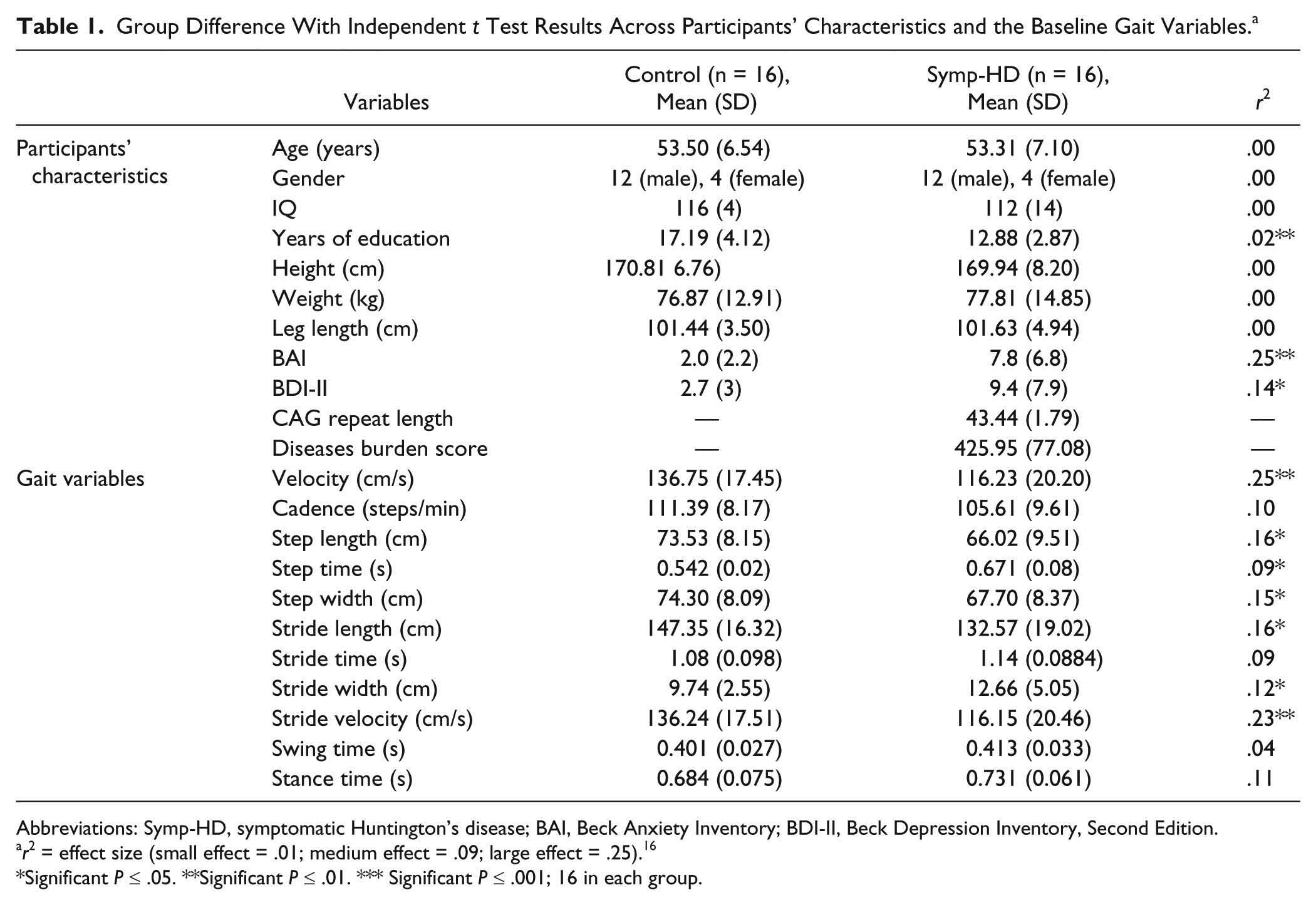
Abbreviations: Symp-HD, symptomatic Huntington’s disease; BAI, Beck Anxiety Inventory; BDI-II, Beck Depression Inventory, Second Edition.
Significant
Apparatus
The GAITRite system
The GAITRite electronic walkway mat (CIR Systems Inc, Clifton, NJ, USA) examined spatiotemporal gait characteristics while participants walked at their preferred speed. The GAITRite system is a valid and reliable method of measuring gait, and has demonstrated high validity when compared with the “gold standard” 3-dimensional motion in older adults with and without neurodegenerative disease.17,18 It has also previously been used in HD research.3,7,19 The gait mat measured 593 cm × 89 cm, contained 18 432 sensors in a horizontal grid pattern, and was connected to a Windows-based computer via a USB cable.
The Berg Balance Scale
The Berg Balance scale (BBS) is a performance-based measure that is validated and commonly used in elderly and clinical settings. The BBS has also been used in HD research.4,20,21 It assesses functional balance over time, evaluates a person’s response to treatment and predicts falls.
22
The BBS determines 2 dimensions of balance: (
The Timed Up and Go Test
The Timed Up and Go (TUG) test is also commonly used withing elderly and clinical settings to measure balance, mobility, locomotor performance, and risk of falls in people with balance disorders. 23 The TUG has also been used in HD research.4,20,21 It consists of a series of tasks—rising from a chair, walking 3 m, turning around, walking back, and sitting down. The total standard time expected to complete these tasks is 10 seconds, with a time >20 seconds indicating impairment in mobility and the need for a gait aid.
Procedure
To obtain baseline gait data, participants first completed 6 unobstructed free walking trials at their preferred speed. Participants subsequently completed 2 different conditions for navigating around obstacles: (
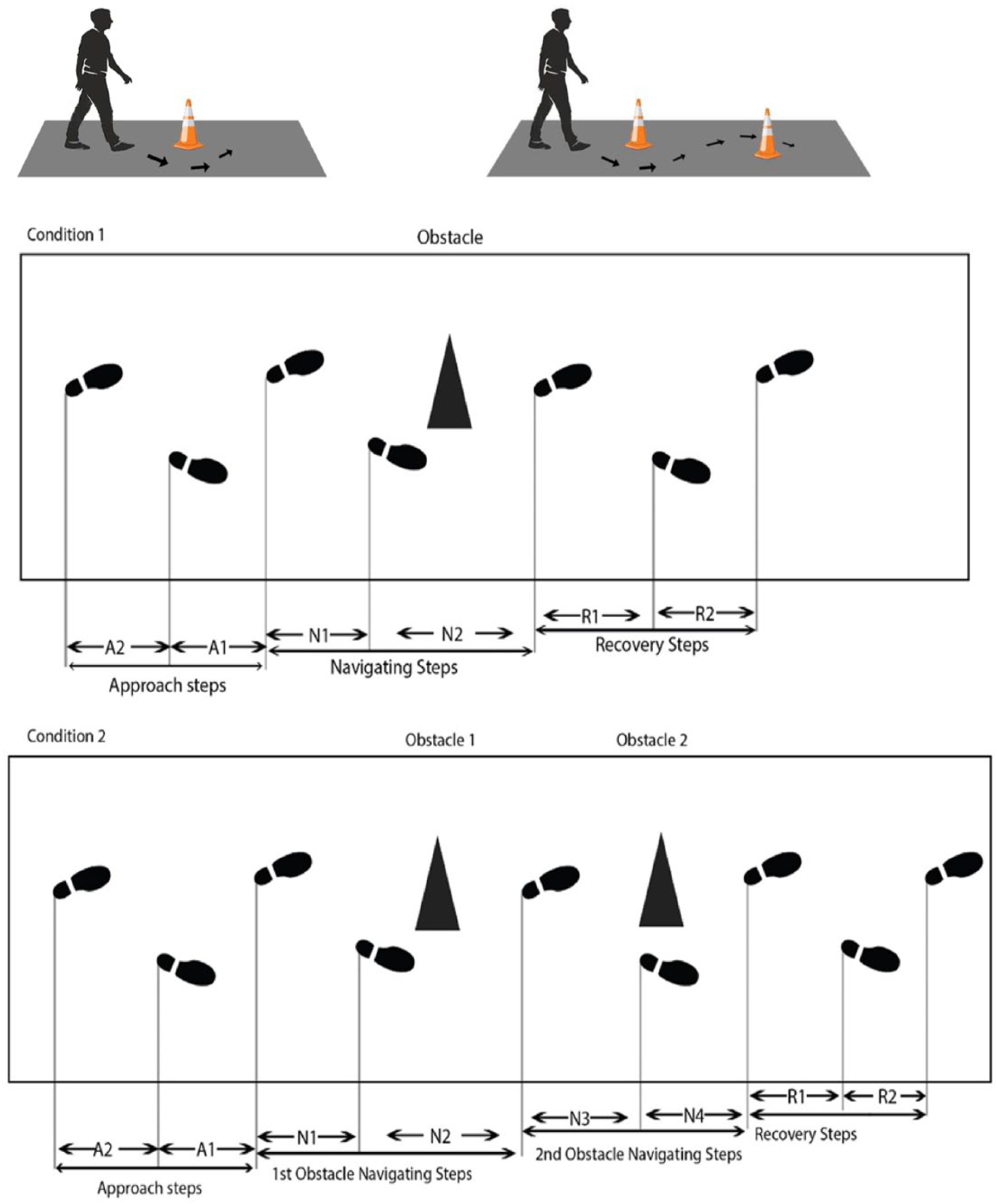
Diagram of the experimental plan for each step across the entire trial while navigating 1 or 2 obstacles.
Data Analysis
Gait data were analyzed using GAITRite software. Mean gait values were obtained from the average of the six experimental trials from the baseline and navigation conditions. For baseline walking, we analyzed the following spatiotemporal gait variables: velocity, cadence, step (length, time, and width), stride (length, time, and width), swing-time, and stance-time. For the navigation conditions, we examined three key gait variables for each step: step-length (measured on the horizontal axis of the walkway from heel point of current footfall to the heel point of the previous footfall on the opposite foot), step-time (time elapsed in seconds from ground contact of one foot to the ground contact of the opposite foot), and step-width (measured on the vertical axis of the walkway from heel point of the current footfall to the heel point of the previous footfall on the opposite foot).
Although the GaitRITE system provides composite data of all footfalls in each trial, the system can also provide data for each individual footfall (using pressure sensor points) within a particular trial. The location of obstacles were identified in the GaitRITE system, as the obstacles that could be detected by pressure sensors in the mat before participants commenced the trials. The required “footfalls” for obstacle navigation walking were identified respectively in relation to and around the location of the specific obstacles (using pressure sensor points) at the GAITRite system.Therefore, the whole walk was divided into 3 phases (approach, navigation, and recovery) using the respective footfalls. Specifically, for the one obstacle navigation trials, footfalls 1 and 2 were classified as
Data were analyzed using IBM SPSS Statistics 24.0 version. To understand the underlying impairment mechanism of spatiotemporal gait variables and specific changes in each step during navigation, the mean was calculated separately for each step’s gait variable throughout each trial. To compare group differences in gait performances at baseline, independent-measure
Pearson correlations were conducted to determine any association between baseline gait variables using the BBS and TUG tests. Other measures, including the UHDRS, BDI, and BAI, were also correlated with gait variables for the HD group only.
Results
There were no significance differences in gender, IQ score, height, weight, or leg length between the participant groups (see Table 1).
Group Comparison of Baseline Walking
Results from
Navigation Around 1 Obstacle
The ‘level hypothesis,” which provides the between-subject differences for multiple dependent variables, was the first stage of profile analysis. Table 2 shows that there were significant differences between the symp-HD and control groups, and large effect sizes, with all key gait variables (ie, step-length, step-time, and step-width) when navigating around one obstacle. At the second stage of analysis, the “flatness hypothesis” provided data on within-subject effects across the steps. Results demonstrated a significant difference between groups for step-length and step-time with large and medium effect sizes, respectively. Though the step-width variable was not significant when navigating around 1 obstacle, it was associated with a small effect size. The final stage of profile analysis, the “parallelism test,” explored interaction across steps by group. For this stage, step-length (
Profile Analysis of Spatiotemporal Gait Variables While Navigating Around 1 and 2 Obstacles Showing Differences Between Patients With Symptomatic Huntington’s Disease and Controls for Results on Level, Flatness, and Parallelism Analyses With
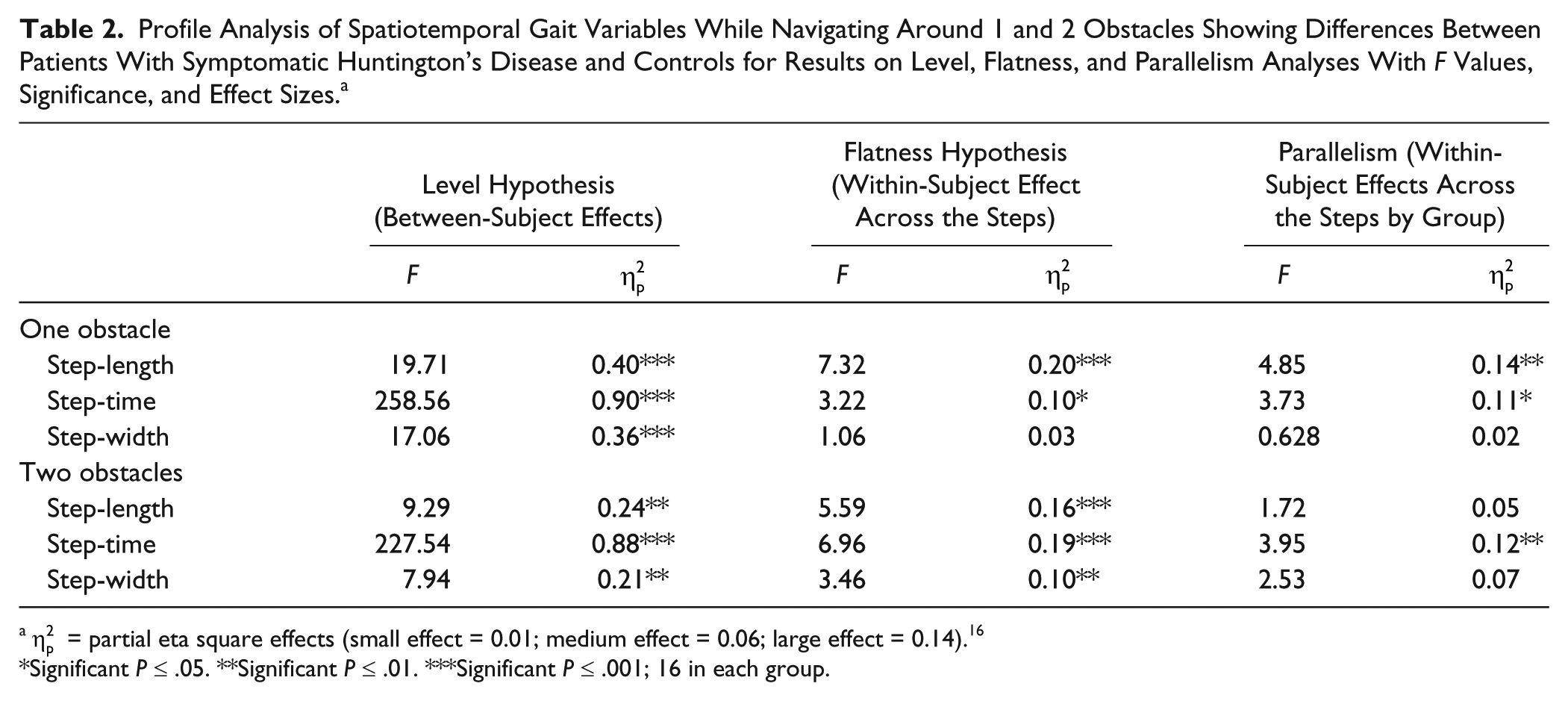
Significant
Navigation Around 2 Obstacles
At the “level hypothesis” stage, there were significant differences between the symp-HD and control groups with large effect sizes for all key gait variables when navigating 2 obstacles (see Table 2). The “flatness hypothesis” revealed significant group differences for step-length, step-time, and step-width gait variables with medium to large effect sizes. The “parallelism test” indicated that only step-time (
See Table 2 for results of profile analyses on step-length, step-time, and step-width for both 1- and 2-obstacle navigation conditions, including
Step-Length Changes Across Each Step
The mean and standard deviation of gait variables across the approach, navigation, and recovery phases for navigating around 1 and 2 obstacles are shown in Supplementary Tables 1 and 2. Figure 2A provides gait patterns when navigating around 1 obstacle. As shown in Figure 2A, symp-HD participants significantly (
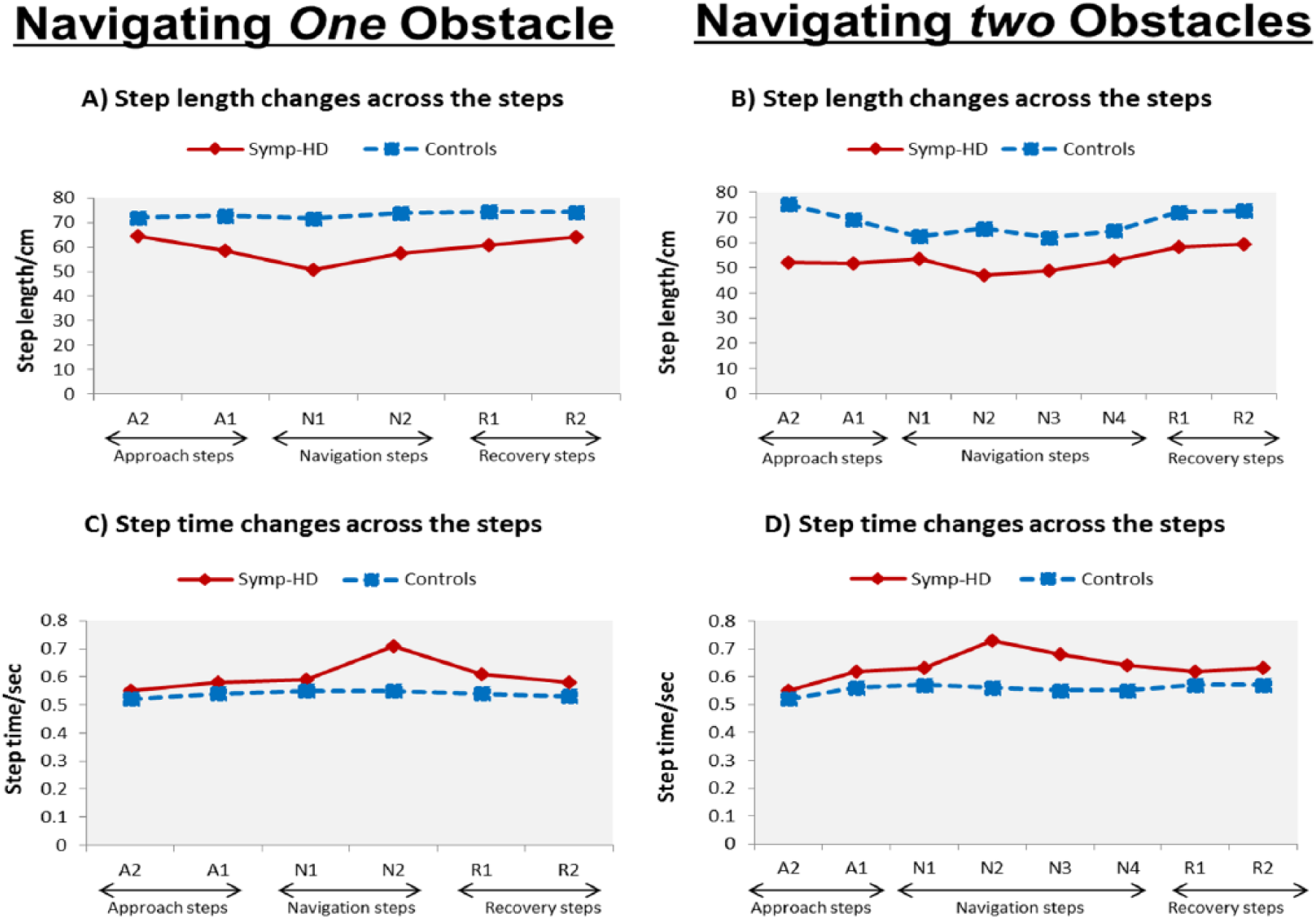
Spatiotemporal gait characteristics during navigating around 1 and 2 obstacles at approach, navigating and recovery phase.
Step-Time Changes Across Each Step
Figure 2C reveals that when navigating around
Changes in step-width were not analyzed further, as this gait variable was not significantly different between the groups across steps during the “parallelism” test of the profile analysis.
BBS and TUG Correlations With Gait Variables
Pearson correlations were conducted separately in both groups as baseline gait variables differed across the groups. The threshold for significance was not adjusted due to the exploratory nature of the experiment. UHDRS and BAI correlated with the BBS, but not with the TUG tests. Most of the baseline gait variables correlated with both the BBS and the TUG tests. For the
BBS and TUG Correlations With Key Gait Variables.
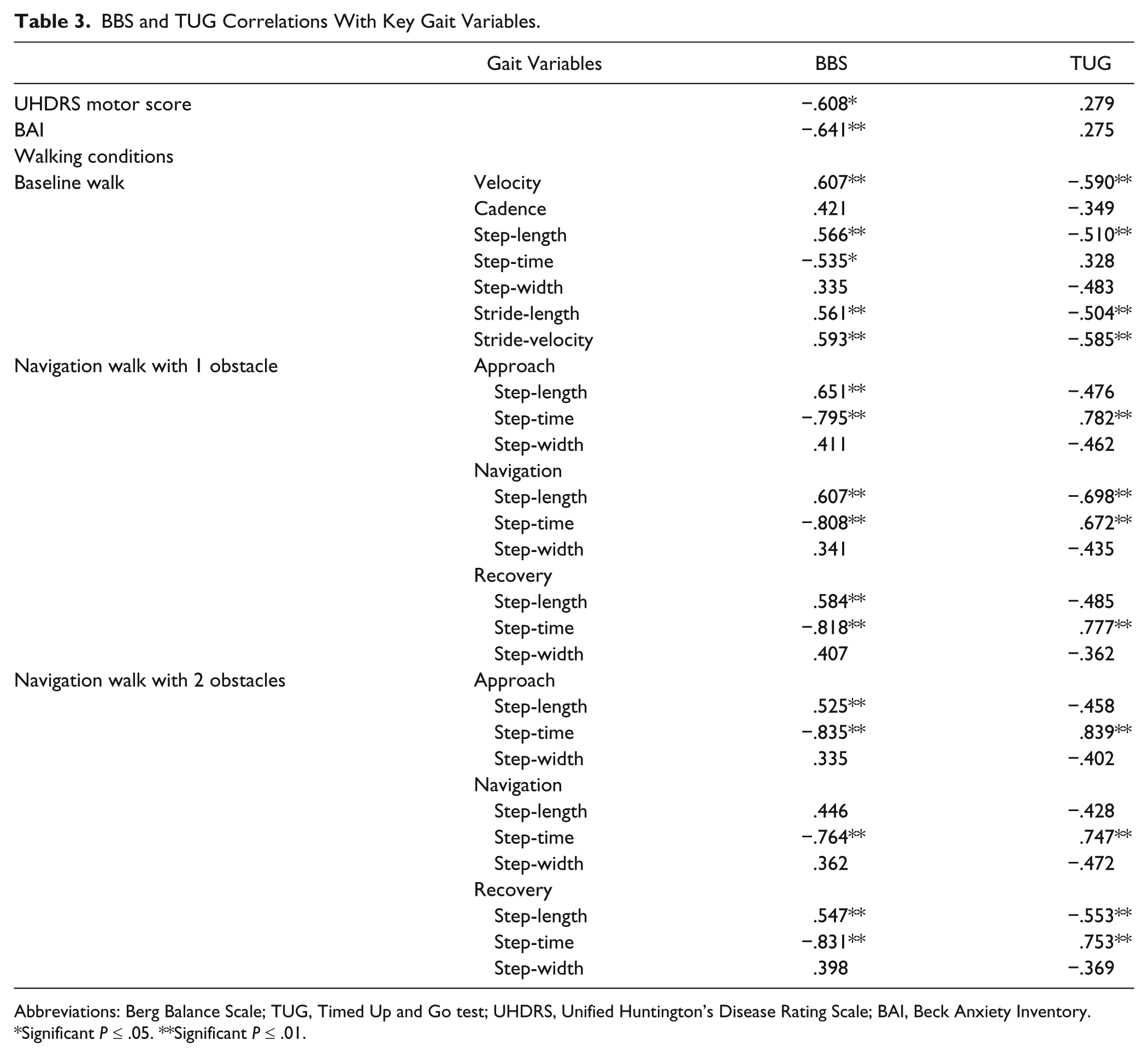
Abbreviations: Berg Balance Scale; TUG, Timed Up and Go test; UHDRS, Unified Huntington’s Disease Rating Scale; BAI, Beck Anxiety Inventory.
Significant
Discussion
For the first time, we sought to characterize spatiotemporal gait measures and adaptive gait patterns in symp-HD during obstacle navigation. Findings indicated that when compared with controls, symp-HD participants demonstrated significant deficits in spatiotemporal gait parameters during both free walking and obstacle navigation conditions. Spatiotemporal gait measures also correlated significantly between the 2 balance measures (BBS and the TUG tests), and provided some evidence that difficulties navigating around obstacles are related to poor balance. Previous research has revealed the usefulness of these 2 clinical tools in measuring balance and risk of falls fair and moderately in HD.4,20,21 However, previously another research also found that the Tinetti Mobility Test is a valid tool for measuring the fall risk in HD, but has restricted utility for assessing the balance status due to a floor effect. 25
During obstacle navigation, some noteworthy results were observed. In particular, when navigating around 1 obstacle, symp-HD individuals decreased step-length and increased step-time during the obstacle navigation phase, in contrast to their performance during the approach and recovery phases. One possible explanation could be a different cognitive approach in symp-HD. That is, the participant’s perception of the obstacle location may have led to a different cognitive strategy that enabled continuation of the walk during obstacle navigation. Conversely, during the baseline walk (free walking without an obstacle), walking may not require prior planning to change strategy or divide attention to multiple stimuli. It is also possible that symp-HD participants chose a conservative walking pattern as a possible strategy to modulate their step-length and step-time. For example, previous research in older adults has suggested that shortened step-length might be a conservative obstacle-crossing strategy that could in turn increase risk of falls. 26
Results also showed that when participants navigated around 2 obstacles, step-time was increased significantly for symp-HD participants, while step-length was not significantly different. The findings are in contrast to previous research that has suggested there is a deficit in the regulation of footstep timing in HD. 27 In this particular reference, 27 the authors explained that HD participants have an impairment in generating steps at regular time intervals with cue synchronization due possibly to difficulty in perceiving time intervals for whole walk. However, the current study showed that symp-HD participants only increased their step-time during the obstacle navigation phase in both navigation conditions. That means, symp-HD participants consistently increased their step-time particularly at navigation phase for one or two obstacle navigation condition, but not for the whole walk. This finding indicates that symp-HD adopt an increased time spent strategy at the navigation phase as a possible compensatory strategy for postural instability. It may also be possible that under higher cognitive loads (ie, 2 obstacles in a pathway), walking/locomotion patterns might be influenced by cognitive impairment in HD. Studies have shown that cognitive ability for the prior planning of locomotion, 28 or before starting to walk, is required to complete a successful walk. 29 However, given that cognitive performance was not measured this interpretation remains speculative. Moreover, research has also indicated that accurate information about the position of an obstacle in the pathway, other surrounding environmental features, 30 or visual information, is required for cognitive processing during self-regulation of walking patterns. 31 Therefore, nonsignificant changes in step-length, when navigating around 2 obstacles, may be related to a poorer ability to execute prior planning of complex locomotion. 32
The BBS is a very accurate and sensitive measure of balance and mobility in individuals with neurodegenerative diseases.20,21 Consistent with previous studies, the current findings revealed that most baseline gait variables were highly correlated with the BBS and TUG test, suggesting that gait abnormalities observed in symp-HD participants during free walking are associated with a higher risk of falling. 4 Therefore, symp-HD participants have a balance impairment which produces significant functional disability, likely leading to reduced mobility in daily life. 33
This study also revealed that step-length was correlated with the BBS across the approach, navigating and recovery phases in symp-HD participants during navigation of 1 obstacle. However, during navigation of 2 obstacles, BBS was associated with step-length only across the recovery phase; not across the approach or navigating phases. Consequently, these findings demonstrate that the association between the BBS and gait variables may be dependent on the perceived balance of the individual and the direct result of the motor impairment.
Step-time was also highly correlated with the BBS across the approach, navigation, and recovery phases during both obstacle conditions. Furthermore, the TUG test was correlated with step-time across all phases with both obstacle conditions. As increased step-time was negatively correlated with the BBS and TUG test, these findings suggest greater impairment in gait pattern and increased risk of falls during obstacle navigation in symp-HD participants. Collectively, these findings suggest that symp-HD participants demonstrate a higher risk of falling during the obstacle navigation walk, particularly at approach and navigation phases.
The current study also found a highly negative association between the BBS and the BAI, which indicates that lower balance ability could lead to higher general anxiety in symp-HD participants. It is noted, however, that the anxiety data were self-reported. We did not investigate “anxiety related to falls,” which warrants further investigation.
Although HD is associated with basal ganglia dysfunction, several studies have claimed that the cerebellum is also involved in controlling spatiotemporal gait characteristics.34,35 It is well known that the basal ganglia, an area of early dysfunction in HD, are primarily responsible for the control of automatic well-learned movements. 36 More recent evidence also indicates that the basal ganglia control the execution of precise voluntary movements via inputs from the motor network outflow in the brainstem. 37 On the other hand, cerebellar dysfunction is associated with a wide-based ataxic gait, motor incoordination and impaired gait, which are all exacerbated by basal ganglia atrophy and disrupted frontostriatal circuits. 38 In addition, recent evidence also suggests that gait abnormalities could coexist with chorea, and symptoms may originate from distinct networks in the basal ganglia and downstream connections. 39 These findings highlight the neural network that plays a vital role in the execution of well-learned motor skills, including the initiation, preparation, execution and maintenance of desired movements known to be compromised by HD.
Limitations
There are some limitations which should be acknowledged. For example, the gait mat adopted in our study was short in length and limited in the range of data it could collect. To compensate for this limitation, a standard set of 6 trials was recorded for each condition, in order to generate enough steps for meaningful analyses. The use of 3-dimensional motion sensors was not possible with the equipment adopted, which would have enhanced the profiling of the gait in more detail. Future research should include individuals at different stages of disease to investigate whether changes in gait patterns, while crossing over obstacles, occur early in HD and how they decline with disease progression. A longitudinal assessment may offer insights into new strategies that could be developed to improve walking and reduce risk of falls in symp-HD. Finally, assessment of brain abnormalities using neuroimaging, including neurobiological and cognitive performance measures, would add a new dimension and allow exploration of whether specific gait deficits are related to basal ganglia and cerebellum dysfunction.
Conclusion
In summary, the current study demonstrated that symp-HD participants have particular gait impairments at the obstacle navigation phase. These could contribute to a higher risk of falls in environments with obstacles. This study also provided significant evidence that symp-HD participants use shorter step-length and longer step-time, thus suggesting more conservative walking strategies during obstacle navigation, possibly as a compensatory response. These findings could be used by clinicians to develop more targeted and effective management strategies to reduce risk of falls in people with HD.
Footnotes
Acknowledgements
We gratefully acknowledge the participation of individuals diagnosed with Huntington’s disease, as well as healthy adult volunteers. We are also very thankful to Bethlehem Hospital and Melbourne professionals working in the field who provided their input into the design of this experiment.
Authors’ Note
This research formed part of a PhD thesis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available on the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
