Abstract
Background. Although dopaminergic medication improves dual task walking in people with Parkinson disease (PD), the underlying neural mechanisms are not yet fully understood. As prefrontal cognitive resources are involved in dual task walking, evaluation of the prefrontal cortex (PFC) is required. Objective. To investigate the effect of dopaminergic medication on PFC activity and gait parameters during dual task walking in people with PD. Methods. A total of 20 individuals with PD (69.8 ± 5.9 years) and 30 healthy older people (68.0 ± 5.6 years) performed 2 walking conditions: single and dual task (walking while performing a digit vigilance task). A mobile functional near infrared spectroscopy system and an electronic sensor carpet were used to analyze PFC activation and gait parameters, respectively. Relative concentrations of oxygenated hemoglobin (HbO2) from the left and right PFC were measured. Results. People with PD in the off state did not present changes in HbO2 level in the left PFC across walking conditions. In contrast, in the on state, they presented increased HbO2 levels during dual task compared with single task. Regardless of medication state, people with PD presented increased HbO2 levels in the right PFC during dual task walking compared with single task. The control group demonstrated increased PFC activity in both hemispheres during dual task compared with single task. People with PD showed increases in both step length and velocity in the on state compared with the off state. Conclusions. PD limits the activation of the left PFC during dual task walking, and dopaminergic medication facilitates its recruitment.
Introduction
People with Parkinson disease (PD) present gait impairments that lead to increased risk of falling, disability, and reduced quality of life.1,2 The typical gait patterns in people with PD include shorter step length, slower gait velocity, and greater gait variability, indicating reduced automaticity.2-4 Behavioral studies suggest that people with PD recruit prefrontal cognitive resources (ie, executive function and attention) during walking as a compensatory mechanism for their basal ganglia dysfunction.5,6 This has been confirmed by studies using functional near infrared spectroscopy (fNIRS), which showed that people with PD present increased activity in the prefrontal cortex (PFC) during usual walking.7-9 It is possible that PD leads to a shift from automatic gait control (ie, direct locomotor pathway) to a more attentional, executive control (ie, indirect locomotor pathway), which involves the PFC. 3
Gait impairments in people with PD are exacerbated when a concomitant cognitive task is performed during walking, known as dual task walking. 10 Previous studies have consistently reported that dual task walking further reduces gait speed and step length, with adverse consequences in PD, such as an increased risk of falls.11-13 Although the mechanisms underlying dual task interference in PD are not yet fully understood, it is well accepted that basal ganglia dysfunction leads to reduced movement automaticity, which increases reliance on attentional and executive resources to control movements.7,11 Similarly, cognitive performance also depends on the PFC. 14 Therefore, both motor and cognitive tasks compete for the same circuitry, which is impaired in PD, increasing the difficulties of performing a dual task. 11
fNIRS studies on dual task walking in PD present inconsistent results. We found 4 fNIRS studies assessing PFC response to dual task walking in PD, 3 of them while on5,7,15 and 1 while off medication. 16 Two studies, both on medication, reported increased PFC activity during dual task walking compared with single walking,5,15 whereas the other 2 studies reported no significant change across walking conditions.7,16 Inconsistent findings might be explained, at least in part, by the different secondary tasks (ie, verbal vs nonverbal cognitive tasks, different difficulty levels, etc) used in these studies. For example, increases in fNIRS signal related to speaking do not represent neurovascular coupling alone and, hence, may not represent greater cortical activation and lead to false positive results. 17 Additionally, it should be noted that no study performed a direct comparison between medication states.
Dopaminergic medication has been shown to improve some gait parameters18-20 and cognitive function in PD.21,22 In addition, dual task walking impairments may also be benefited by antiparkinsonian medication,23,24 although contradictory results have been reported in the literature.25,26 Taken together, these results suggest that prefrontal activation may ameliorate under the effect of medication. However, the neural mechanism of dual task improvement after taking medication is not yet known, and assessing the PFC activity with and without levodopa is essential to understand the cortical correlates of this mechanism.
The hypothesis of compensation-related utilization of neural circuits (CRUNCH) postulates that processing inefficiencies cause the aging/impaired brain to recruit more neural resources to achieve computational output equivalent to that of a healthy younger brain. 27 This compensatory activation is relatively effective at lower levels of task demand, such as during usual walking. However, a resource ceiling may be reached with increased task demand (such as dual task walking) or pathology, leading to insufficient processing and behavioral decrements. Because dopaminergic medication improves gait parameters during usual walking and reduces dual task cost in people with PD, one may argue that dopamine ameliorates gait automaticity in PD. For example, Gilat et al 28 showed that people with PD in the on state, operating a foot pedal in a supine position (simulating walking), demonstrated lesser activation in the bilateral orbitofrontal cortex compared with the off state as well as lesser step time variability, which is indicative of increased gait automaticity. 3 These authors also showed that dopaminergic medication improved the connectivity between striatal and cortical areas (ie, cortical-striatal pathway). 28 The improved automaticity may reduce the reliance on the prefrontal compensatory mechanism for gait control in PD, 7 releasing cognitive resources to be used for a concomitant secondary task during dual task walking. In this context, we investigated the effect of dopaminergic medication on PFC activity and gait parameters during dual task walking in people with PD. We hypothesized that people with PD off medication would be unable to increase PFC activation during dual task walking compared with usual walking 16 because of a resource ceiling. Dopaminergic medication would then increase available resources and compensatory potential, leading to improved gait under the dual task condition (ie, off vs on).
Methods
Participants
A total of 20 individuals with PD and 30 healthy older people (control group) participated in this study. Participants were included if they met the following inclusion criteria: >60 years of age and able to walk without the use of any aids. People with PD were included if they were diagnosed with idiopathic PD, as defined by the UK Brain Bank criteria, in stages I to III of the Hoehn and Yahr Scale (H&Y), and taking PD medication. Participants were excluded if they presented a significant cognitive impairment (Mini Mental State Examination [MMSE] < 24) and any musculoskeletal, orthopedic, and/or visual impairments that could affect gait. This study was approved by the local research ethics committee. All participants provided written informed consent to participate in this study.
Procedures
People with PD were assessed in 2 visits to the lab (1 week apart). During one visit, they were assessed approximately 1 hour after taking their regular dopaminergic dose (on state). During the other visit, participants were tested after overnight withdrawal (at least 12 hours) from their regular dopaminergic medication (off state). The order of medication condition was counterbalanced across people with PD.
Demographic characteristics and cognitive status were obtained for both groups. Cognitive assessment was performed once when participants with PD were in their on medication state. The MMSE was used to assess global cognitive function. 29 The Movement Disorders Society—United Parkinson’s Disease Rating Scale (MDS-UPDRS) part III was used to assess motor symptom severity, 30 and the H&Y scale was used to determine the stage of PD. 31 The levodopa equivalent dose was calculated according to Tomlinson’s suggestions. 32
Gait and PFC activity were assessed in 2 walking conditions: single task and dual task. In both tasks, participants walked at a normal and comfortable pace. They performed 5 trials for each condition in a randomized order. Each trial started with 30 s of standing still, followed by the instruction “ready-go” for the participant to start 30 s of walking around a 26.8-m circuit, with two 7-m parallel straights. During the standing still period, participants were instructed to stand quietly, look straight ahead, and think of nothing in particular. 15 The dual task consisted of a digit vigilance task, which required participants to walk while listening to random numbers (from 1 to 9) played over a speaker for 30 s. 33 The intervals between numbers were randomized to prevent gait synchronization. Immediately before starting to walk, participants were given the class of numbers (odd or even) that they were required to count mentally and determine how many times the class occurred. They then called out the answer at the end of each trial. The performance (ie, error score) in the cognitive digit vigilance task was quantified by the absolute error (difference between the correct answer and the response given by the participant) and expressed in percentage (where 0% indicates that there is no error). The error score [(Absolute error/Correct response) × 100] was calculated for baseline and dual task walking. The participant performed the cognitive task while sitting in order to avoid overestimation of dual task interference. 12
Functional Near Infrared Spectroscopy
A mobile fNIRS system (OctaMon, Artinis Medical System, Netherlands) with continuous wave diodes and wavelengths of 760 and 850 nm was used to record changes in HbO2 at a sampling rate of 10 Hz. An 8-channel arrangement with 10 optodes, consisting of 8 transmitters and 2 detectors, covered Brodmann areas 9, 10, and 46 in both right and left hemispheres. The interoptode distance was 35 mm. After the Cz position was determined on the participant’s head, a neoprene headcap (fiber holder) marked with labels of the international 10–20 electroencephalography system was placed on the participant’s head. OxySoft version 3.0.52 (Artinis Medical Systems, Netherlands) was used for data collection.
Processing of fNIRS followed previous recommendations. 34 We chose the HbO2 concentration because it is the most used indicator of walking-related changes in cortical activity.35-37 Miyai et al 36 observed that HbO2 is more sensitive than deoxygenated hemoglobin in locomotor tasks. HbO2 concentration was calculated using the modified Beer-Lambert law. The differential path-length factor (DPF) was set to 6.61 for all participants, because the DPF for adults older than 50 years is not known. The fNIRS data were preprocessed using NIRS-SPM open source toolbox for MATLAB. 38 A low-pass filter (cutoff 0.15 Hz) based on the canonical hemodynamic response function was used to reduce the high-frequency noise.38,39 A wavelet-minimum description length detrending algorithm was applied to decompose NIRS measurements into global trends, hemodynamic signals, and uncorrelated noise components as distinct scales.40-42 Preprocessed data were then exported to MATLAB (MATLAB and Statistics Toolbox Release 2015, The MathWorks, Inc, Natick, MA), in which further data processing was performed using a customized script. HbO2 concentration signals were averaged per region of interest (4 channels for the left PFC and 4 channels for the right PFC) and normalized by dividing them according to corresponding signal amplitude of the whole experiment.43,44 Because PD is usually asymmetrical 45 and our sample showed clinically important differences between the right and left limbs, we opted for considering left and right PFC as separate/different regions of interest. Trials were divided into 2 phases: (1) a control task period (referred to as the baseline), corresponding to the final 15 s before the walking bout (the 5 s immediately before the walk were removed to exclude potential anticipatory changes by the participants; therefore, the baseline period consisted of 10 s), and (2) an experimental task period, walking from 5 to 25 s after the instruction “go” (Figure 1). The normalized HbO2 concentration was averaged (in time) over the baseline period and experimental task for each region of interest and each trial. Baseline HbO2 concentration was subtracted from the experimental task to evaluate the relative change in HbO2 concentration in each experimental condition.7,8,15,46-48
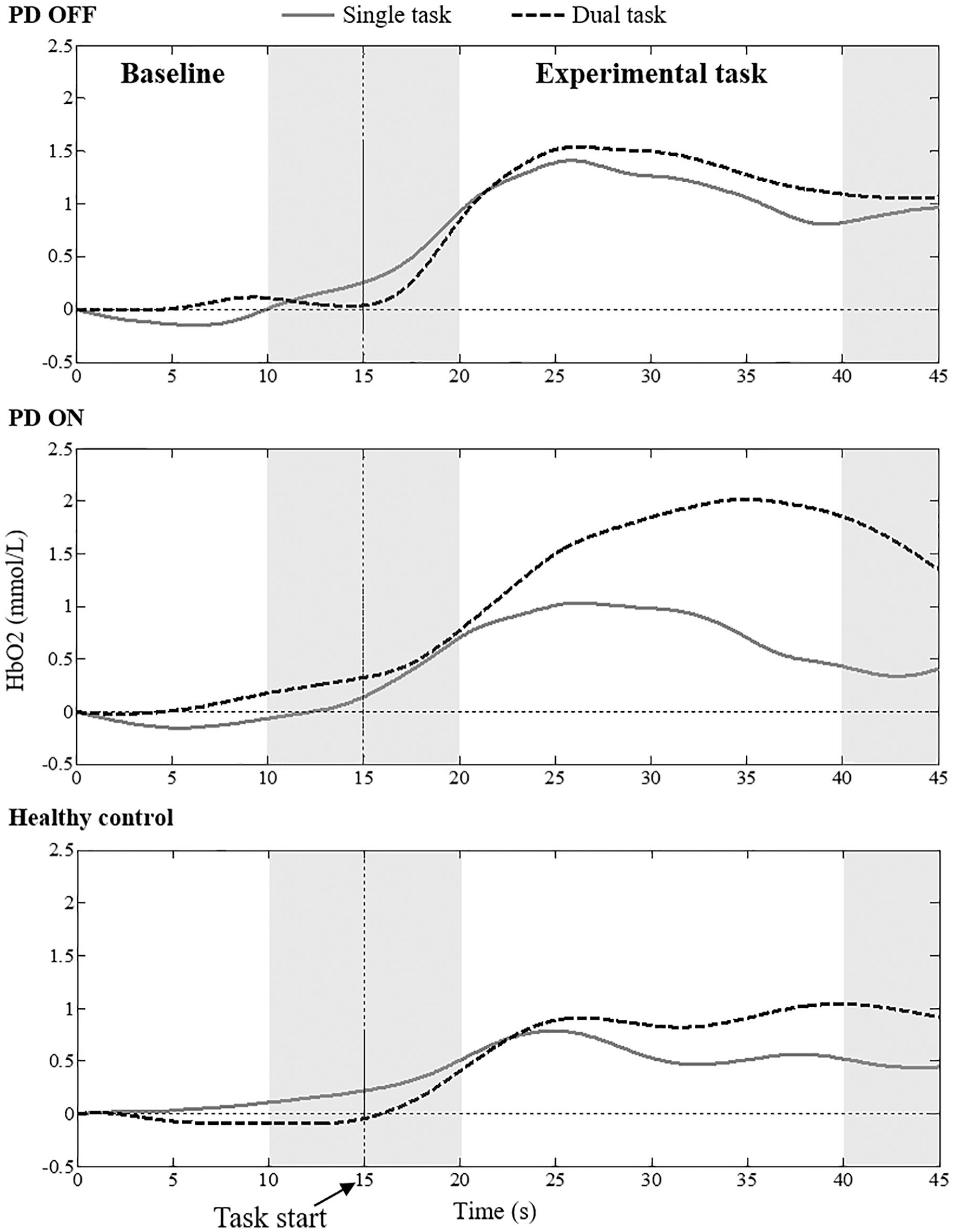
Filtered HbO2 level (averaged signal of 5 trials) of 2 representative participants (1 participant with PD in off and on medication state and 1 healthy control) during single and dual task conditions. The gray area represents the excluded signals of analysis.
Gait Assessment
A 5.74-m GAITRite system (CIR Systems Inc, Clifton, NJ) with a frequency of 200 samples/s, placed over one 7-m straight segment of the circuit, was used to measure gait parameters. This system was connected to a computer using GAITRite software version 4.7.5 (CIR Systems Inc, Clifton, NJ) for processing and storing data. All steps registered by the system were considered for the analysis (average of footsteps: PD off: 58.25 ± 11.60 steps; PD on: 56.37 ± 10.97 steps; control group: 51.47 ± 9.53 steps). The gait variables included step length, width, time, and velocity as well as the percentage of step time spent in the swing and double support phases. In addition, step length variability, step time variability, and step velocity variability were calculated by the coefficient of variation, defined as follows: (SD/Mean) × 100.
Statistical Analysis
Statistical analysis was performed using SPSS 22.0 for Windows. The level of significance was set at 5% for all analyses. The Pearson χ2 test and t-test were performed to compare groups for demographic and cognitive data. The paired-samples Wilcoxon test was used to compare disease severity between the on and off medication states. Differences in levels of HbO2 and gait parameters were analyzed by 2-way ANOVA, with group (PD off × Control group; PD on × Control group) and condition (Single task × Dual task) as factors, with repeated measures for the second factor. In addition, to verify the effect of medication, a 2-way ANOVA was performed, with medication (PD off × PD on) and condition as factors and repeated measures for both factors. The Bonferroni post hoc test, corrected for multiple comparisons (P value/Number of comparisons), was used to localize the differences where ANOVA revealed significant interactions. The partial η2 squared (pη2) statistic provided estimates of the effect sizes.
Results
The characteristics of the participants are summarized in Table 1. As expected, the Wilcoxon test indicated improvement in motor symptoms (MDS-UPDRS-III scores) after medication intake (Z = −3.924; P < .001).
Participant Characteristics (Mean ± SD).
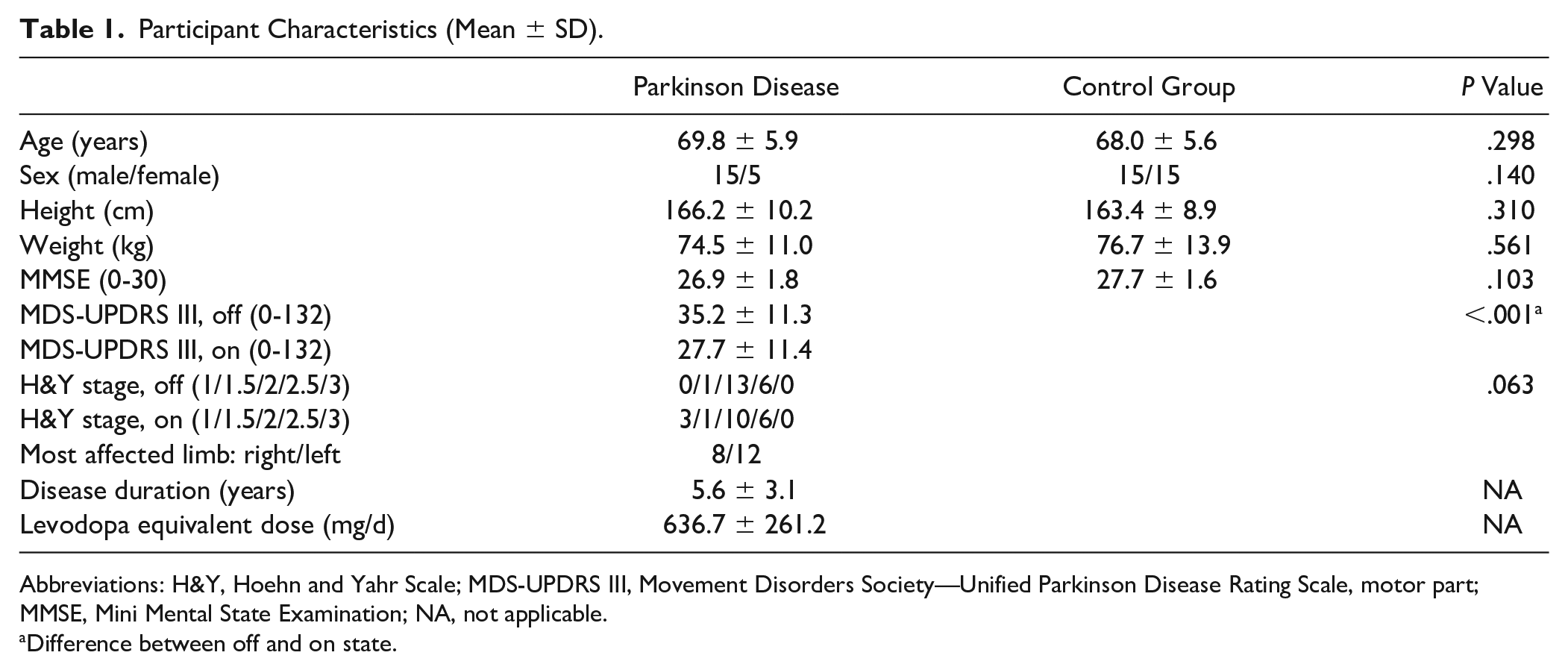
Abbreviations: H&Y, Hoehn and Yahr Scale; MDS-UPDRS III, Movement Disorders Society—Unified Parkinson Disease Rating Scale, motor part; MMSE, Mini Mental State Examination; NA, not applicable.
Difference between off and on state.
Medication Effects on Prefrontal Activity
Interaction between medication and condition was found for HbO2 level in the left PFC (F1,19 = 4.373, P = .050, pη2 = 0.187; Figure 2). A post hoc test showed that people with PD in the off state did not present changes in HbO2 level across walking conditions. On the other hand, people with PD in the on state presented increased HbO2 level during dual task compared with single task (P = .008). A main effect of condition was observed for HbO2 level in the right PFC (F1,19 = 8.425; P = .009; pη2 = 0.307), with higher HbO2 level during dual task compared with the single task (Figure 2). There was no main effect of medication.
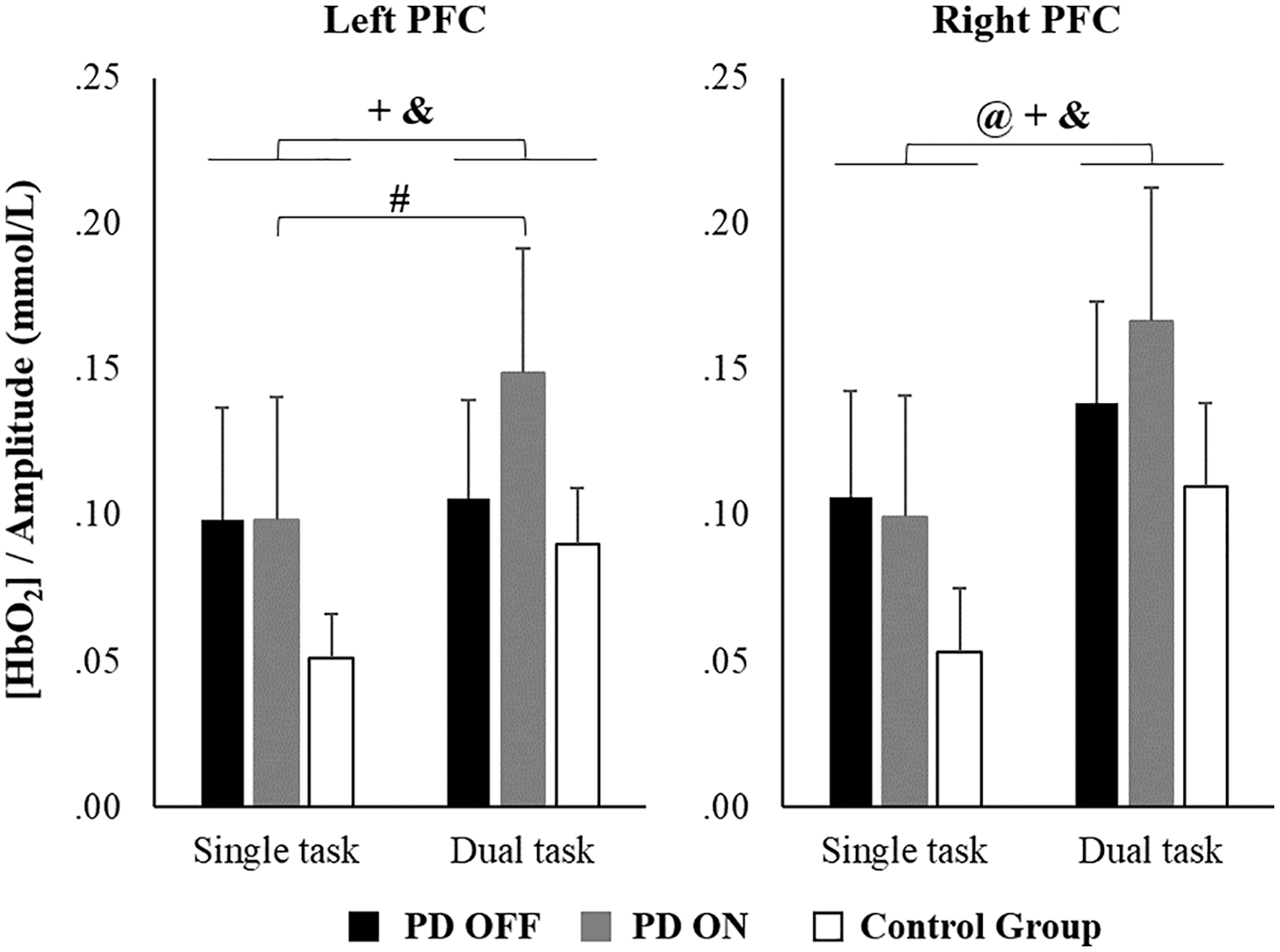
Means and standard errors of normalized oxygenated hemoglobin (HbO2) levels during single and dual tasks for PD off, PD on, and control groups.a
Difference Between PD Group and Control Group in Prefrontal Activity
PD Off Versus Control Group
There was no interaction between group and condition or main effect of group. A main effect of condition was observed for HbO2 level in the left PFC (F1,48 = 4.573; P = .038; pη2 = 0.087) and in the right PFC (F1,48 = 11.303; P = .002; pη2 = 0.191). Participants demonstrated increased HbO2 levels during dual task compared with single task (Figure 2).
PD On Versus Control Group
There was no interaction between factors or main effect of group. A main effect of condition was revealed for HbO2 level in the left PFC (F1,48 = 16.932; P < .001; pη2 = 0.261) and right PFC (F1,48 = 17.137; P < .001; pη2 = 0.263). Participants presented increased HbO2 levels during dual task compared with single task (Figure 2). There was no main effect of group or interaction between factors.
Medication Effects on Gait Parameters
PD Off Versus PD On
Gait parameters are presented in Table 2. Interaction between group and condition was not observed. A main effect of medication was found for step length (F1,19 = 6.228; P = .022; pη2 = 0.247) and step velocity (F1,19 = 4.137; P = .056; pη2 = 0.179; trend). People with PD increased both step length and velocity in the on state compared with the off state. A main effect of condition was revealed for step length (F1,19 = 19.446; P < .001; pη2 = 0.506), step velocity (F1,19 = 17.291; P = .001; pη2 = 0.476), swing time (F1,19 = 17.676; P < .001; pη2 = 0.482), and double support time (F1,19 = 17.729; P < .001; pη2 = 0.483). People with PD presented decreased step length, velocity, swing time, and increased double support duration during dual task compared with single task.
Gait Parameters by Groups and Conditions (Mean ± SD). a
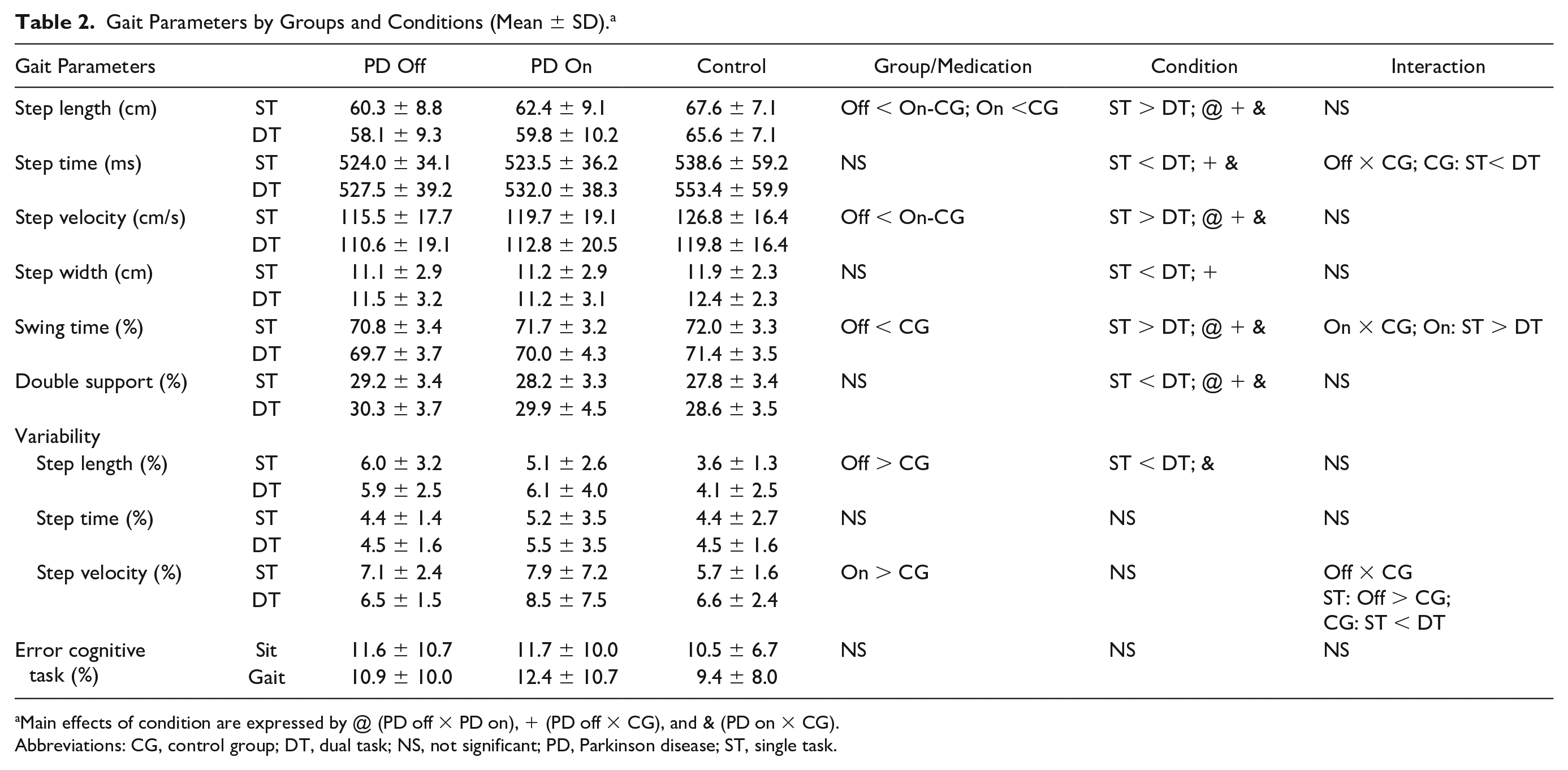
Main effects of condition are expressed by @ (PD off × PD on), + (PD off × CG), and & (PD on × CG).
Abbreviations: CG, control group; DT, dual task; NS, not significant; PD, Parkinson disease; ST, single task.
Comparison Between PD and Control Group on Gait Parameters
PD Off Versus Control Group
A significant interaction between group and condition was revealed for step time (F1,48 = 6.608; P = .013; pη2 = 0.121) and step velocity variability (F1,48 = 5.256; P = .026; pη2 = 0.099). Post hoc tests showed that people with PD in the off state presented greater step velocity variability (P = .015) than the control group in the single task. In addition, the control group demonstrated increased step time (P < .001) and step velocity variability (P = .034) during dual task compared with single task. Main effect of condition showed that participants decreased step length (F1,48 = 50.692; P < .001; pη2 = 0.514), velocity (F1,48 = 59.426; P < .001; pη2 = 0.553), and swing time (F1,48 = 14.083; P < .001; pη2 = 0.227) and increased step time (F1,48 = 17.968; P < .001; pη2 = 0.272), step width (F1,48 = 8.518; P = .005; pη2 = 0.151), and double support duration (F1,48 = 17.096; P < .001; pη2 = 0.263) during dual task compared with single task. A main effect of group was observed for step length (F1,48 = 10.767; P = .002; pη2 = 0.183), step velocity (F1,48 = 4.345; P = .042; pη2 = 0.083), and step length variability (F1,48 = 11.462; P = .001; pη2 = 0.193). People with PD in the off state presented shorter and slower steps and greater step length variability than the control group.
PD On Versus Control Group
Two-way ANOVA revealed a trend for interaction between group and condition for swing time (F1,48 = 3.733; P = .059; pη2 = 0.072). Post hoc tests showed that only people with PD significantly decreased swing time during dual task compared with single task (P < .001). Main effect of condition revealed that both people with PD in the on state and the control group decreased step length (F1,48 = 37.986; P < .001; pη2 = 0.442), step velocity (F1,48 = 59.844; P < .001; pη2 = 0.555), and swing time (F1,48 = 17.445; P < .001; pη2 = 0.267) and increased step time (F1,48 = 37.661; P < .001; pη2 = 0.440), double support duration (F1,48 = 18.148; P < .001; pη2 = 0.274), and step length variability (F1,48 = 6.931; P = .011; pη2 = 0.126) during dual task compared with single task. A main effect of group demonstrated that people with PD in the on state presented shorter step length (F1,48 = 5.629; P = .022; pη2 = 0.105) and greater step length variability (F1,48 = 6.240; P = .016; pη2 = 0.115) than the control group.
Discussion
To our knowledge, this is the first study to investigate the effects of antiparkinsonian medication on PFC activation during single and dual task walking in people with PD. As expected, people with PD in the off state presented an insufficient compensatory mechanism, not showing increased (left) PFC activity during dual task walking. However, after taking medication, people with PD presented increased (left) PFC activity in the dual task condition. These findings are in line with the CRUNCH framework. 27 It is possible that walking off medication is so demanding that people with PD reach the ceiling of available neural resources in the left PFC, leading to insufficient processing and poor behavioral performance. In contrast, dopaminergic medication, which ameliorates the connectivity between striatal and cortical areas, 28 may increase available resources and compensatory potential 27 and also improve gait parameters.
Dual task walking leads to increased contribution of the indirect locomotor pathway to the control of locomotion in healthy older adults. Our results showed that older adults presented increased PFC activity during dual task walking compared with single task. This finding is in line with previous studies, which demonstrated recruitment of additional prefrontal cognitive resources to maintain performance during dual task walking in older adults.46-49 It has been postulated that the indirect locomotor pathway is activated when the automatic execution of walking is reduced and compensatory mechanisms are necessary, such as in dual task walking.50,51 In this case, the neuronal commands are transmitted via PFC and premotor areas to the basal ganglia and subthalamic as well as mesencephalic locomotor regions. 50
Interestingly, 2 studies reported contradictory results with regard to PFC activation during dual task performance in people with PD. Whereas Maidan et al 7 showed no difference between single and dual task (serially subtracting 3 s from a given 3-digit number), Nieuwhof et al 15 found that people with PD increased bilateral HbO2 concentrations of the PFC during walking while serially subtracting and reciting digit span. A possible explanation for these differences could be the “posture first/second” strategy. Maidan et al 7 showed that people with PD had a success rate of 89.4% in the cognitive performance, as compared with 96.2% in healthy older people (who had greater cortical activity during the dual task), suggesting that people with PD used the “posture first” strategy, which is to sacrifice performance in the cognitive task in order to optimize their gait. Nieuwhof et al 15 showed that people with PD hit 98.4% in the cognitive task, demonstrating that they used the “posture second” strategy. In our study, people with PD had a similar score in the cognitive task while sitting and during walking; they also presented worse gait parameters in the dual task than in the single task condition. These findings suggest that our participants also used a “posture second” strategy. Therefore, it is possible that PFC activation is influenced by the strategy used during dual task walking in PD, with the “posture second” strategy leading to greater PFC activation.
In people with PD, PFC cortical response to dual task is influenced by dopaminergic medication only in the left hemisphere. Similar to healthy older adults, people with PD in both off and on states increased right PFC activity in response to a dual task. However, people with PD only increased left PFC activity in response to the dual task while on medication. Our findings are in line with previous studies showing that people with PD on medication present increased PFC activity in both hemispheres during dual task. Nieuwhof et al 15 found bilateral PFC activation during dual task in people with PD in the on state. Al-Yahya et al 5 observed increased left PFC activation under dual task condition in people with PD in the on state; this study did not assess the right PFC. In contrast, Stuart and Mancini, 16 using the average of both hemispheres, did not observe difference in PFC activity between single and dual task in people with PD in the off state. The left PFC might be more affected by PD than the right PFC (the right PFC responded to the dual task even while off medication, whereas the left PFC only responded to the dual task while on medication). The left PFC has been shown to play an essential role in the performance of tasks involving working memory, 52 which is particularly affected by PD.53,54 Because patients with PD have impaired prefrontal dopamine signaling, 55 it is possible that the levodopa intake improved prefrontal dopamine signaling, which in turn led to increased left PFC activity in response to the dual task. Our limited sample size and mix of patients with right/left most-affected hemisphere limit the possibility for further explanations. Therefore, future studies should target the laterality of current findings.
PD is caused by a dysfunction in a complex neural network. A pathophysiological hallmark of PD is degeneration of dopaminergic neurons in the substantia nigra.56,57 Hence, people with PD show disrupted balance between excitatory and inhibitory activity in the basal ganglia. In PD, the output nuclei of the basal ganglia send excessive inhibitory signaling to the thalamus, which in turn sends reduced excitatory signaling to cortical areas, including motor and cognitive areas.58,59 However, dopaminergic replacement increases the availability of dopamine in the system and reestablishes the thalamocortical pathway by improving functioning of the basal ganglia. 58 Thus, the findings of the current study suggest that during the on state, people with PD are able to recruit both left and right PFC when the task demands, increasing the availability of the cognitive resources during dual task. In addition, after taking their regular medication, people with PD presented longer and faster steps in both walking conditions, demonstrating that dopaminergic medication improves bradykinetic and hypometric characteristics of gait.19,20 It is possible that gait got better because of improved automaticity and/or more efficient prefrontal compensatory mechanism (which is more required for dual task walk).
This study has some limitations. First, because we did not control for superficial hemodynamic or systemic changes in the skull, we cannot rule out the potential effect of superficial contamination. However, we used filters to minimize these aspects according to suggestions of review studies.34,60 Second, we analyzed only PFC. The assessment of other cortical areas, such as premotor areas and the primary motor cortex, is required for a more complete understanding of the cortical mechanisms involved in the control of locomotion in PD. Therefore, these limitations should be considered in future studies. Despite this limitation, our article brings relevant new information to the literature. For example, we used an ecological approach (walking overground instead of on a treadmill or mitigating gait while lying in a scanner) to study the effects of antiparkinsonian medication on PFC activity during dual task walking. It is worth mentioning that the cognitive task used is purely cognitive, not involving the motor component of speech, which may affect the fNIRS signals.
In conclusion, PD limits the activation of the left PFC during dual task walking, which suggests a neural inefficiency to recruit the additional cognitive areas in order to compensate for PD-related deficits in automaticity. However, levodopa facilitates the recruitment of the left PFC when the task demands it. Therefore, PD medication may increase the available cognitive resources during walking and also improve gait parameters.
Footnotes
Acknowledgements
The authors would like to thank Vinicius Cavassano Zampier, MSc, Vinicius de Belli, and Lucas Meira Fiorio for their assistance with data collection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Brazilian Federal Agency for Support and Evaluation of Graduate Education (CAPES; Finance Code 001), the São Paulo Research Foundation (FAPESP; Grant Numbers: 2016/21499-1; 2014/22308-0), and the National Council for Scientific and Technological Development (CNPq; Grant Number: 429549/2018-0).
