Abstract
Background
Habituation of postural response to perturbations is impaired in people with Parkinson’s disease (PD) due to deficits in cortico-basal pathways. Although transcranial direct current stimulation (tDCS) modulate cortico-basal networks, it remains unclear if it can benefit postural control in PD.
Objective
To analyze the effect of different intensities of anodal tDCS on postural responses and prefrontal cortex (PFC) activity during the habituation to the external perturbation in patients with PD (n = 24).
Methods
Anodal tDCS was applied over the primary motor cortex (M1) with 1 mA, 2 mA, and sham stimulation in 3 different sessions (~2 weeks apart) during 20 minutes immediately before the postural assessment. External perturbation (7 trials) was applied by a support base posterior translation (20 cm/s and 5 cm). Primary outcome measures included lower limb electromyography and center of pressure parameters. Measures of PFC activity are reported as exploratory outcomes. Analyses of variance (Stimulation Condition × Trial) were performed.
Results
Habituation of perturbation was evidenced independent of the stimulation conditions. Both active stimulation intensities had shorter recovery time and a trend for lower cortical activity in the stimulated hemisphere when compared to sham condition. Shorter onset latency of the medial gastrocnemius as well as lower cortical activity in the nonstimulated hemisphere were only observed after 2 mA concerning the sham condition.
Conclusions
tDCS over M1 improved the postural response to external perturbation in PD, with better response observed for 2 mA compared with 1 mA. However, tDCS seems to be inefficient in modifying the habituation of perturbation.
Introduction
Patients with Parkinson’s disease (PD) demonstrate impairments in postural responses to an external perturbation, which leads to increased risk of falling.1-4 For example, after external perturbation, they demonstrate an excessive muscular coactivation and greater displacement of the centers of mass (CoM) and pressure (CoP).1-4 Patients with PD also show delayed adaptation/habituation of the postural responses to the perturbation, requiring more trials to decrease CoM and CoP displacements. 5 Postural responses seem to be generated mainly from the subcortical structures (ie, pedunculopontine nucleus [PPN] and reticular formation [RF]). 6 Deficits in cortico-basal pathways are evidenced in patients with PD,7,8 affecting PPN and RF activity. Patients with PD have lower excitatory activity of the thalamus on primary motor cortex (M1) and higher inhibitory activity of the basal ganglia on PPN and RF, therefore resulting in hypoactivation of these regions involved in postural responses. 9 A growing body of evidence suggests the involvement of cortical areas such as M1, supplementary motor area (SMA) and prefrontal cortex (PFC) in the control of the postural responses.9-13 M1 is part of the direct postural pathway, which is activated in less challenging situations and reflects an automatic postural control.13,14 On the other hand, PFC and SMA are part of the indirect postural pathway, which is activated when automaticity is reduced (by pathologies, lower intrinsic subject balance ability or challenging perturbations/situations) and compensatory executive-attentional resources are required for postural control.13-17
Transcranial direct current stimulation (tDCS) increases excitability of cortical and subcortical areas.6,18 A single session of tDCS improved balance and functional mobility in patients with PD 19 and decreased the muscle onset latency in young adults after external perturbation. 6 In addition, although clinical, cognitive, and balance improvements were observed in PD following tDCS,19-21 the effects vary with the intensity of stimulation.19-22 Stimulation with 2 mA seems to be more effective than 1 mA in improving working memory.21 On the other hand, motor aspects and reaction time do not seem to be influenced by stimulation intensity.21,22 However, to our best knowledge, the effects of different intensities of tDCS on postural control in patients with PD in situations with external perturbation have not been investigated so far. Establishing the best parameters of tDCS to improve postural control in people with PD is highly relevant to both clinicians and patients since it might optimize/enhance the beneficial effects already observed for this cohort.23,24
Thus, the study aimed to analyze the effect of different intensities (1 and 2 mA) of anodal tDCS applied over M1 on postural responses and PFC activity during the habituation to the external perturbation in patients with PD. We hypothesized that tDCS with 1 or 2 mA will positively improve postural control of patients with PD, with shorter recovery time and onset latency, and lower range of CoP 6 compared with sham condition (placebo). In addition, we also expected that patients with PD will reach the habituation plateaus in fewer trials after tDCS application compared with sham condition. Since the stimulation protocol targeted the direct postural pathway (ie, tDCS applied over M1), we expected to observe decreased PFC activity in response to tDCS. 12 Superior positive effects on postural responses were also expected after 2 mA when compared with 1 mA stimulation condition 21 because 2 mA seems to be more effective in increasing motor excitability.25,26
Methods
Participants
Twenty-four patients with idiopathic PD diagnoses based on UK Brain Bank criteria participated in this crossover, randomized, double-blind, and sham-controlled study (Figure 1). Exclusion criteria were score above three in the Hoehn & Yahr scale (H&Y), musculoskeletal, vestibular, or visual impairments that affected balance, presence of any uncontrolled disease that could affect peripheral sensory function (eg, diabetes) and risk of receiving the stimulation (neural implants, pacemaker, history of seizures and epilepsy). Also, exclusion criteria included changes in the PD medication during the study period and not participating in all three days of assessment/stimulation. The research ethics committee from São Paulo State University approved this study (CAAE: 87653818.2.0000.5465). All participants signed the statement of consent.
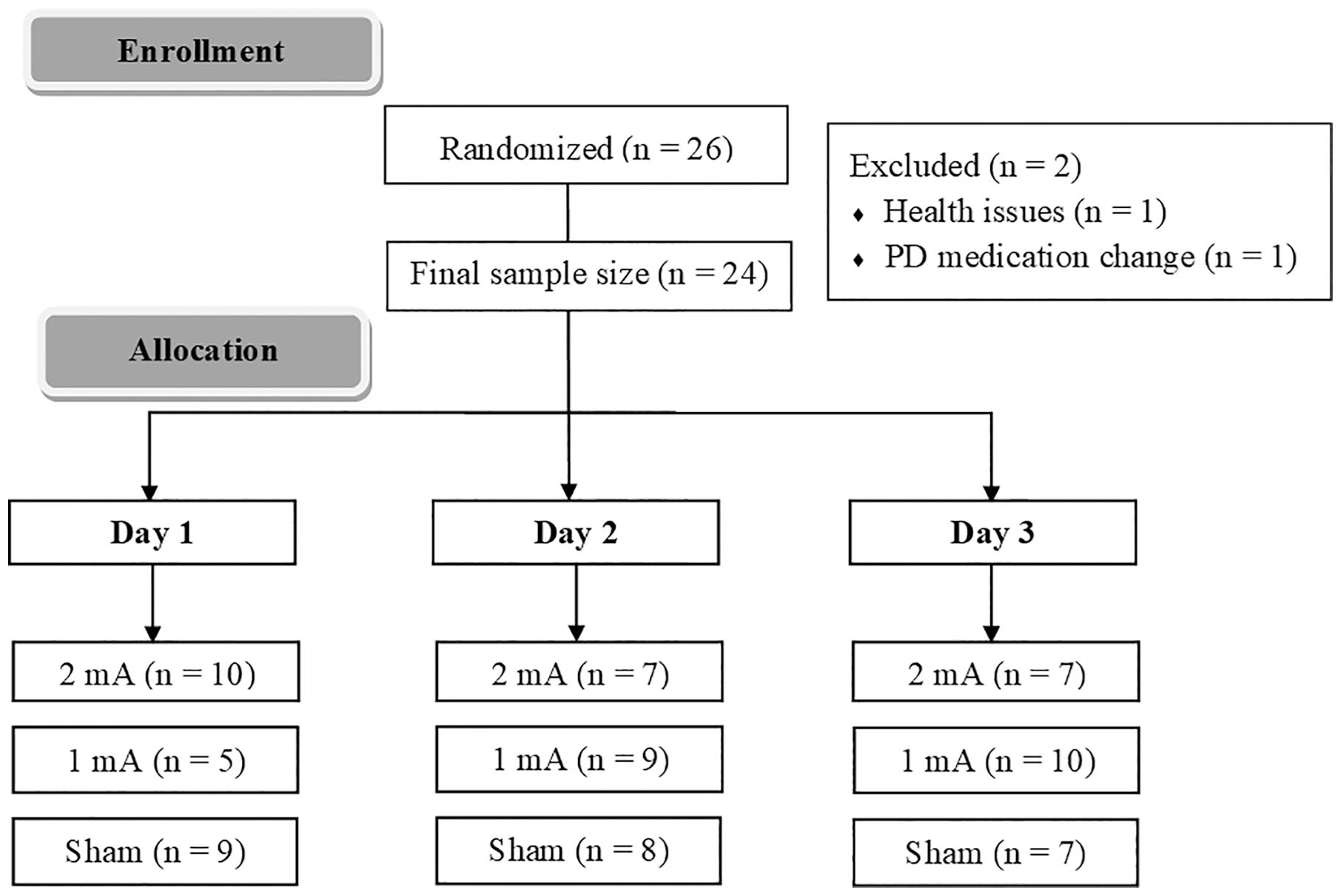
Distribution of the order of the stimulation condition across subjects.
Experimental Design
All evaluations were performed “ON” PD medication (approximately 1 hour after medication intake). Participants’ postural control was measured on three different sessions (separated by at least 2 weeks) in which they first received “1 mA, 2 mA, or sham.” The team member who applied the tDCS procedures randomly determined the order of the stimulation condition, thus guaranteeing blindness. This team member did not provide any instruction during postural control assessment and did not participate in the data analysis. The order was counterbalanced across subjects (Figure 1), and the 3 data collection sessions of each participant were scheduled at the same time of the day (in order to minimize the impact of motor fluctuations). Clinical and cognitive assessments were carried out by an experienced researcher using the motor section of the Movement Disorders Society–Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) 27 and the Mini-Mental State Examination (MMSE) 28 (Table 1). A structured questionnaire applied immediately after the end of the stimulation, assessed potential adverse effects of tDCS.29,30 In addition, after day 3, patients should report what type/intensity of tDCS they received at each day.29,30
Demographic, Cognitive, and Clinical Outcomes of the Patients With PD.
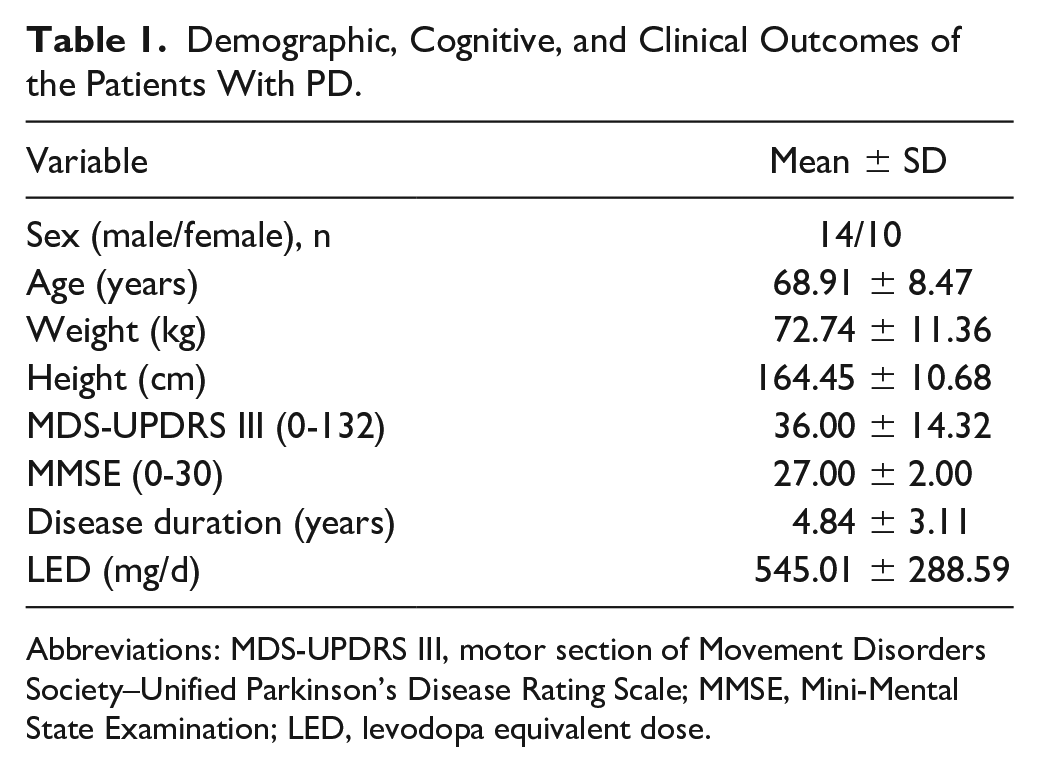
Abbreviations: MDS-UPDRS III, motor section of Movement Disorders Society–Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; LED, levodopa equivalent dose.
tDCS Protocol
The participants remained seated on a comfortable chair during the stimulation protocol, which was performed just before the postural assessment. tDCS was applied using a Microestim GENIUS (NKL Electronic Products) via conductive-rubber electrodes, placed in 2 saline-soaked sponges (35 cm2). Anodal electrode was positioned over M1 (C3/C4 on the 10-20 international electroencephalogram system), and the reference electrode was positioned over the contralateral supraorbital region.6,31 The anodal stimulation was performed in the cerebral hemisphere contralateral to the most affected body side of the PD, 32 which was determined through the MDS-UPDRS items. 33 The active tDCS stimulations (1 and 2 mA) were applied for 20 minutes with a 30-second ramp-up at the beginning and 30-second ramp-down at the end of the stimulation period. For the sham, the same 30 seconds of ramp up/ramp down was performed, but the stimulation remained active (2 mA) between these periods only for 10 seconds 19 (Figure 2).
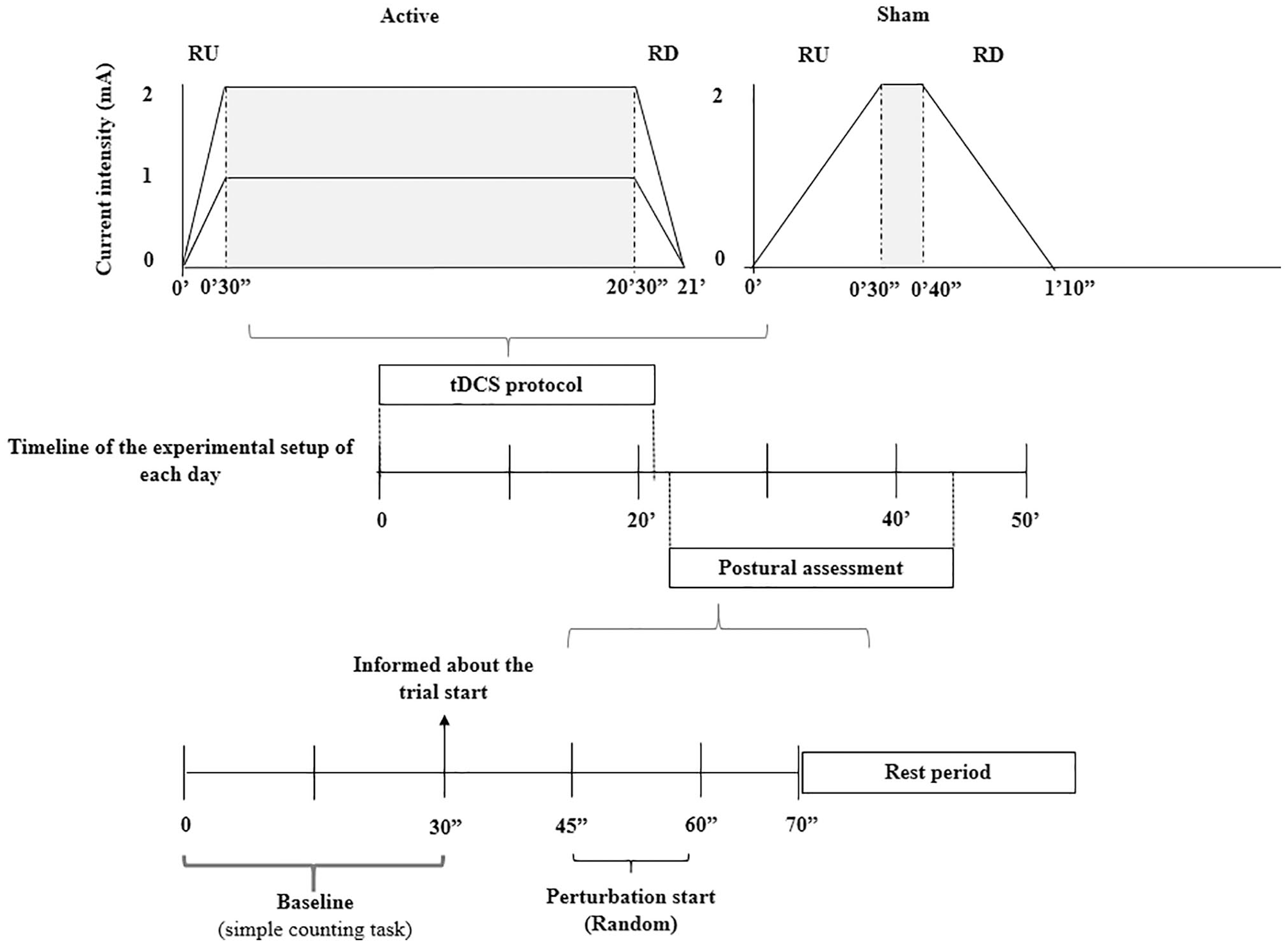
Experimental setup of the transcranial direct current stimulation (tDCS) protocol and the postural assessment of each day. Note: The gray area in the tDCS protocol represents the period of active stimulation. RU = ramp-up (30 seconds); RD = ramp-down (30 seconds).
Postural Control Assessment
Participants stood in a bipedal standing on a force plate fixed to a translating platform for seven trials. The resting period lasted 30 seconds between each trial to reset the platform and to allow the participants to move, to avoid prolonged periods of static standing. Each trial lasted 70 seconds, in which the initial 30 seconds were used for the cortical activity baseline measures. For that, the participants were asked to maintain a quiet standing and conduct a simple counting task (in steps of 1) to avoid mind wandering 34 and were informed that the perturbation would not occur in this period. After that, the participants were asked to stop counting and were informed about the trial start. The temporally unpredictable perturbation was applied by the translation of the support base in the posterior direction 3 (20 cm/s and 5 cm), and it was initiated randomly (between 15 and 25 seconds after informed the trial beginning) in all trials. For security, the participant used a harness during all postural assessments. The evaluation did not exceed 30 minutes to guarantee the peak effect of the tDCS6,35,36 (Figure 2).
Equipment
A Trigno Wireless System (Delsys, Inc, 2000 Hz) was used to acquire the electromyogram (EMG), and the sensors were positioned in medial gastrocnemius (MG) and tibialis anterior (TA) of the most affected limb following the SENIAM recommendations. A force plate (AccuGait, Advanced Mechanical Technologies, 200 Hz) was positioned on the equipment that induced the perturbation and it was used to assess CoP behavior. For the cortical activity during the postural evaluation, the patients were equipped with a mobile functional near-infrared spectroscopy (fNIRS) system (OctaMon, Artinis Medical Systems, 10 Hz). The system consisted of a headband with 8 light emitters and 2 light detectors (wavelengths 760 and 850 nm) with 35 mm between them positioned at the PFC according to the 10-20 system. fNIRS detects the cerebral hemodynamic activity (changes in deoxy and oxygenated hemoglobin concentration [HbO2] due to altered neural activity). 37 PFC was chosen because of its involvement in postural control mechanisms, mainly in PD, also due to its connection with the PPN and the thalamus 16 as well as to assist in posture control through attentional and anticipatory perturbation processes. Still, previous study with tDCS suggested a possible neural network between the PFC and M1. 31 Besides, one active marker of the optoelectronic capture system (Optotrack, NDI, 200 Hz) and an accelerometer (Trigno Wireless System, Delsys, 148.15 Hz) were positioned in the force plate to synchronize the perturbations start, EMG, and CoP activity.
Data Analysis
To analyze the EMG and the CoP, data were collected for a period of 5 seconds before the perturbation until the end of the trial, and during the whole trial for the fNIRS. The reactive adjustments of the CoP behavior were analyzed for a period between the perturbation start and 800 ms. 2 For the EMG, a period between the onset of muscle activity and 250 ms was considered. The onset of muscle activity was determined by a semiautomatic algorithm and corresponds to the first point where the activity value was higher than the mean plus 4 standard deviations of the baseline (400 to 200 ms before the perturbation) by at least 50 ms. For the cortical activity, the last 10 seconds before the instructions of the trial beginning was considered as the baseline period. Reactive adjustments were considered for a period from 3 to 10 seconds after the perturbation start. 16 This period was chosen considering the physiologic hemodynamic delay16,38 and the peak of the response that occurs approximately 6 seconds after the stimulus. 38
Analysis of EMG Parameters
EMG signals were filtered with a band-pass filter (20-300 Hz), and after rectification, data were again low-pass filtered (15 Hz). In addition, the system was calibrated with a rejection rate of 80 db, and a 16 bits A/D resolution. Onset latency was analyzed only for MG. The magnitude of muscle activation was calculated by the area below the curve (integral, iEMG). The iEMG was determined by
Analysis of CoP Parameters
Recovery time to a stable position was determined by the CoP behavior analysis. For this, a semiautomatic sliding window algorithm was developed in a Matlab environment (Mathworks). The recovery time was determined by the time (first frame) at which CoP variability (standard deviation during 1 second) after perturbation was less or equal than to CoP variability in the baseline period (determined over a period of 1 second before the perturbation). 3 Also, the range of CoP and the peak of the CoP velocity in the anterior-posterior direction were analyzed after the perturbation. 3 These parameters were chosen because they indicate the ability of the individual to control the posture and the behavior of the postural response after external perturbation.2,3
Analysis of Cortical Activity
HbO2 concentration was calculated using Oxysoft software using the modified Beer-Lambert law. Furthermore, after the filtering processes described below, the raw data from all channels of all individuals in all trials were visually inspected to ensure signal quality and reliability. After that, 2 participants were excluded from the fNIRS analysis due to the signal noise. The data processing was conducted through NIRS-SPM (NIRS-SPM, http://www.nitrc.org/projects/nirs_spm) 41 with filtering and detrending steps following the current recommendation. 42 A low-pass filter (0.14 Hz) based on canonical hemodynamic response 43 was used to reduce/remove high-frequency noise. The signal was decomposed by a wavelet algorithm into a global trend due to heart rate, blood vessel tone, breath, and other possible experimental errors (including movement artifacts and drift of the signal).42,44,45 Then the processed signal was analyzed by a custom algorithm developed in the Matlab environment for further processing steps. The peak of the [HbO2] in the reactive adjustment was normalized by the mean of [HbO2] at the baseline for each individual (delta – Δ). Changes in [HbO2] on PFC in the stimulated and nonstimulated hemispheres were analyzed separately by the mean of the channels for each cerebral hemisphere.
Statistical Analysis
Statistical analyses were performed by SPSS 21.0 (IBM Corp) software, and the significance level was maintained as 0.05. Normality and homogeneity were verified by Shapiro-Wilk and Levene test. Friedman’s analysis of variance (ANOVA) was conducted to analyze the tDCS side effects. Reactive adjustments analysis was determined by 2-way ANOVA with repeated measures for the factors Stimulation Condition (1 mA × 2 mA × sham) and Trial (1 × 2 × 3 × 4 × 5 × 6 × 7). Bonferroni correction post hoc was applied when differences between the Stimulation Conditions were indicated in the analysis. Two simple planned contrasts were performed when interaction and/or Trial effect were revealed in the analysis. Contrast 1 was performed to analyze the capacity to change the postural response after the first trial (trial 1 vs trials 2, 3, 4, 5, 6, and 7). Contrast 2 was performed to analyze when individuals reached the habituation plateaus, representing the last change concerning trial 7 (trial 7 vs trials 2, 3, 4, 5, and 6). Previous studies indicated that the habituation plateaus in patients with PD occurs before trial 7. 5
Results
Side Effects and Stimulation Condition Perception
Friedman’s ANOVA indicated that there was no significant difference for sensations and discomfort between cortical stimulation with 2 mA, 1 mA, and sham. In addition, the main sensations reported were tingling (52.78%) and itching (22.22%) (Table 2). However, the scores of each stimulation condition in both sensations were mild from the adapted questionnaire score.29,30 Regarding the ability to perceive the stimulation condition, 63% of the participants were not able to perceive any difference between them. When considering the total number of sessions performed in the experiment (72 sessions), there was only 12% accuracy to the type of stimulation performed in a given session.
Means and Standard Deviations of Side Effect Score After Each Stimulation Condition. a
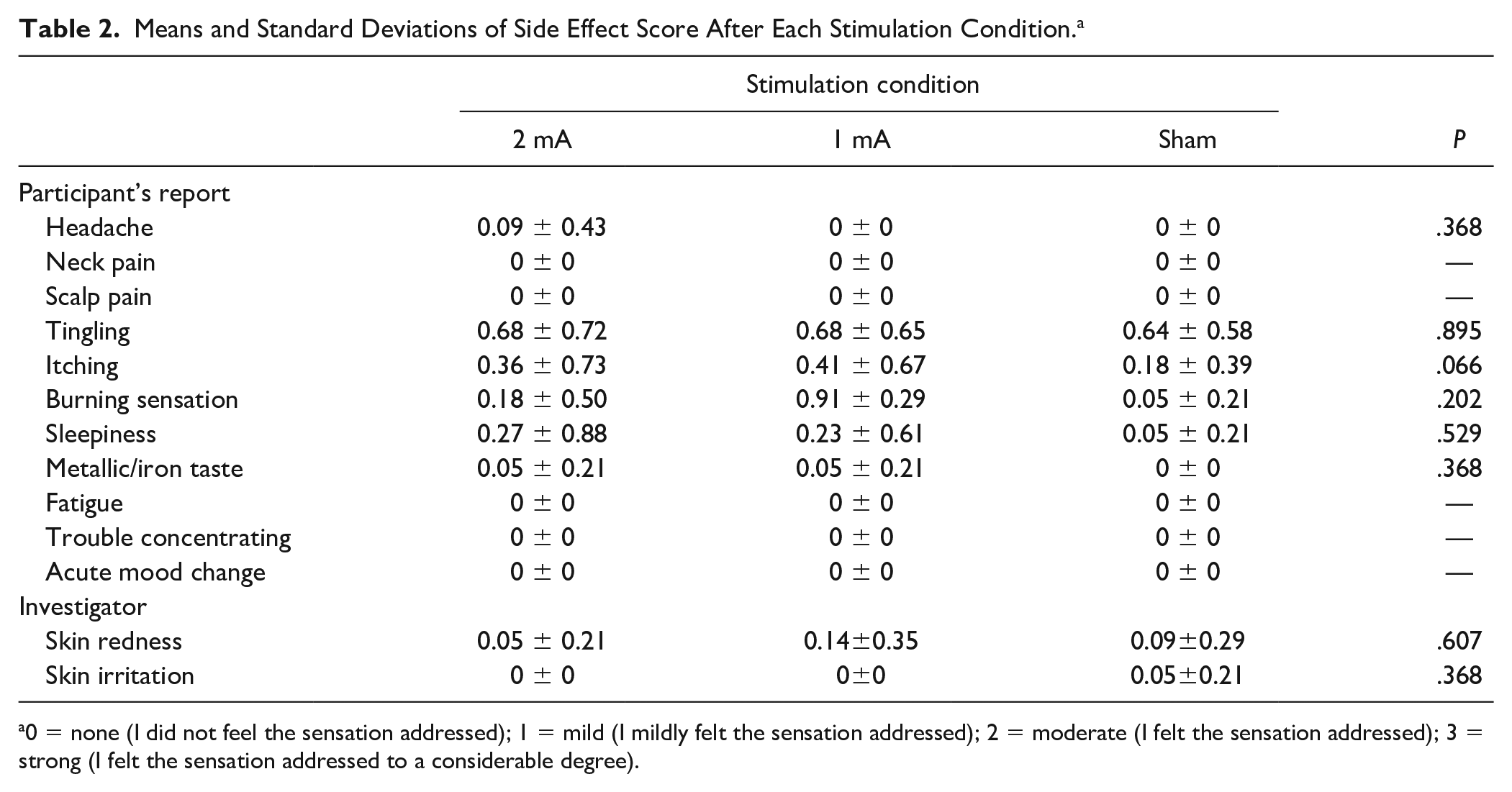
0 = none (I did not feel the sensation addressed); 1 = mild (I mildly felt the sensation addressed); 2 = moderate (I felt the sensation addressed); 3 = strong (I felt the sensation addressed to a considerable degree).
Temporal Parameters of EMG and CoP
ANOVA revealed Stimulation Condition main effect for the onset latency of the MG (
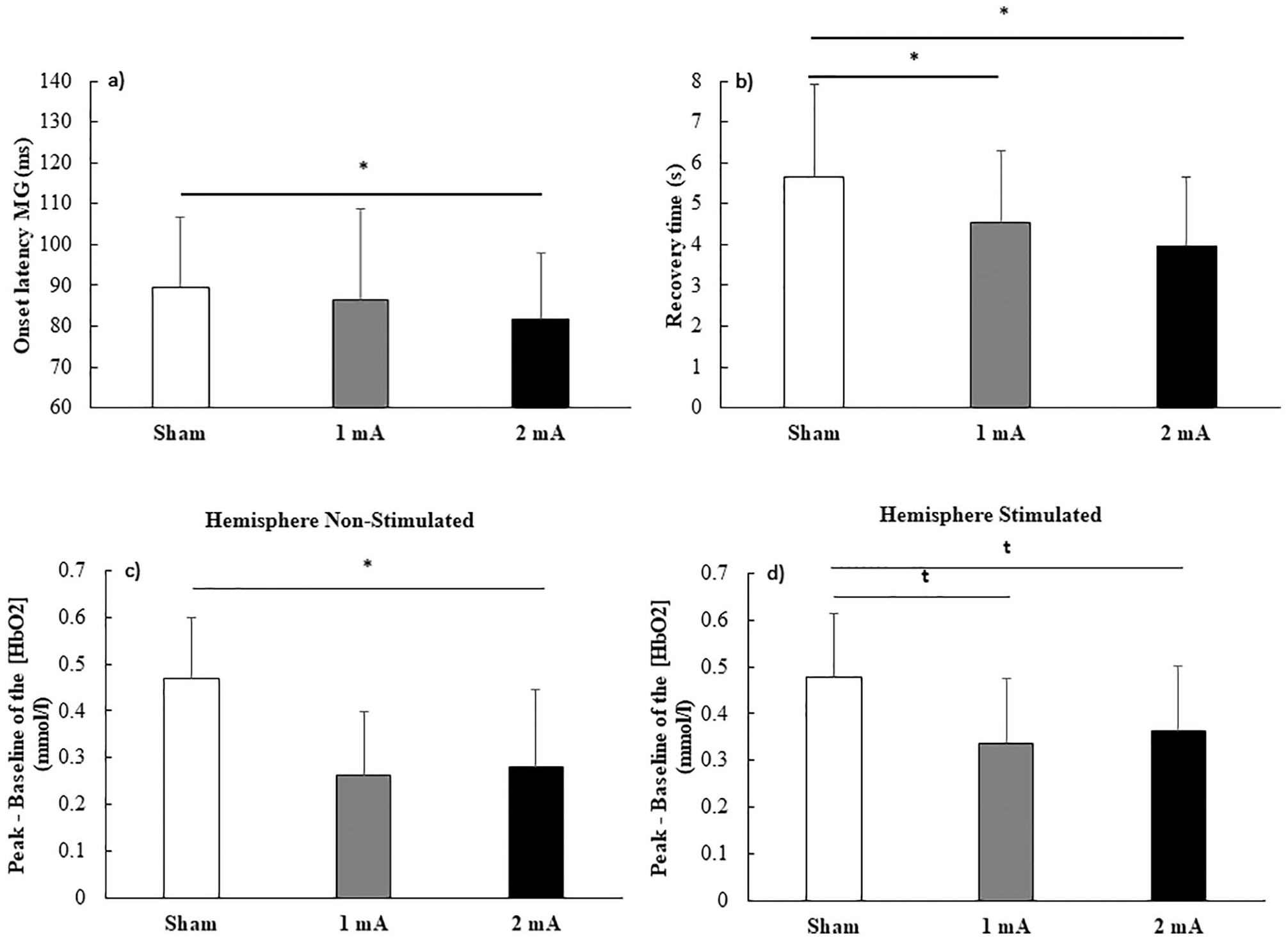
Stimulation Condition main effect. Values correspond to the means and standard deviations (a, b) and standard errors (c, d). (a) Onset latency of medial gastrocnemius (MG); (b) recovery time; (c) [ΔHbO2] in hemisphere nonstimulated; and (d) stimulated. Note: *Significant difference between Stimulation Conditions;
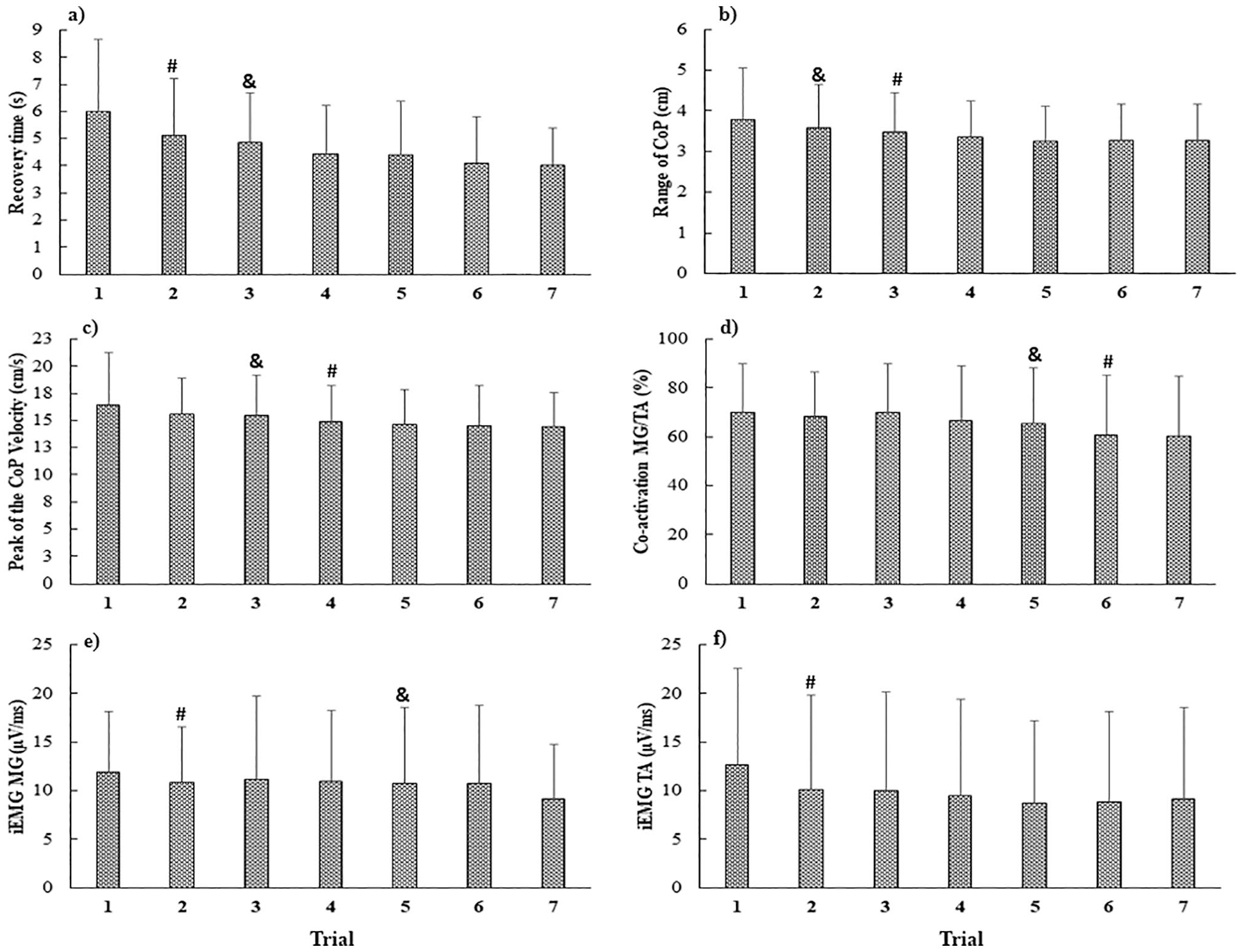
Trial main effect. Values correspond to the mean and standard deviations of the: (a) recovery time; (b) range of CoP; (c) peak of the CoP velocity; (d) coactivation of medial gastrocnemius/tibialis anterior (MG/TA); (e) iEMG of MG; (f) iEMG of TA. Note: #Significant difference for trial 1. &Significant difference for trial 7.
Amplitude of EMG and CoP Parameters
ANOVA revealed Trial main effect for the iEMG of the MG (
Cortical Activity
ANOVA revealed Stimulation Condition main effect for the PFC [ΔHbO2] in the nonstimulated (
Discussion
The present study analyzed the effect of different intensities of tDCS over M1 on the habituation of the postural responses to external perturbation in patients with PD. As expected, a single session of tDCS was able to improve postural control. Both active tDCS intensities reduced the time to recover balance after the perturbation as compared to the sham condition. Additional benefits were observed after the 2-mA stimulation, such as the shorter onset latency of MG. The lower PFC activity observed after active stimulations suggests that anodal tDCS applied over M1 reduces the involvement of the indirect executive postural pathway in the control of posture. However, unexpectedly, tDCS was unable to induce habituation of postural responses to perturbation in fewer trials.
tDCS seems to be a promising technique to improve postural control in patients with PD.19,46 However, protocols with different intensities, duration, and electrode positioning have been used, which makes it difficult to develop specific protocols with this technique.23,46 This study demonstrated that a positive effect of tDCS could also be achieved for postural responses in situations with external perturbations. Our results suggest a positive effect of anodal tDCS over M1 on postural control in patients with PD, especially with 2-mA intensity evidenced by the decrease in recovery time, MG onset latency and PFC activity. These changes could be explained by 2 mechanisms that are not mutually exclusive.
First, tDCS applied over M1 has been shown to improve sensorimotor integration, 47 which is essential for postural control. 48 Sensory inputs from external perturbation are sent to brainstem, thalamus, and cerebral cortex structures for integration and processing of this information. 8 However, patients with PD show a decreased sensory signal-to-noise ratio in these structures, which makes it difficult to use sensory information in the planning and programming of adequate postural responses, especially in small perturbation intensity. 49 A previous study indicated that tDCS could modulate somatosensory perception. 47 Furthermore, anodal tDCS over M1 seems to increase the activity of the somatosensory cortex, SMA, and caudate nucleus after peripheral stimulation by pressure on feet soles. 47 These cortical regions are involved with the integration and interpretation of somatosensory signals and hence, with postural control due to their dependence on the somatosensory information to maintain the balance.8,12,47 Thus, the tDCS seems to contribute to the perception and integration of sensory information, possibly by increasing their signal-to-noise ratio (in this study from the perturbation). This improvement may help in the velocity of corticospinal pathways motor program output, which could improve the onset latency of MG observed in patients with PD demonstrated in our study.
Second, tDCS applied over M1 reduced the involvement of the indirect pathway for postural control, as observed by the decreased PFC activity.6,14 tDCS may increase the blood flow and the amount of oxygen available in the subcortical areas involved with the direct pathway (more automatic movement control).24,50,51 The facilitation of subcortical, and of direct pathway structures after tDCS, may be explained by the change in cortico-reticular drive and the direct change in the excitability of these structures.6,18 RF and PPN are structures involved in the preparation and generation of postural responses, which are altered in PD, and may be affected by stimulation of M1 with 2 mA. 6 Some studies suggest that stimulation with 2 mA seem to be more efficient to increase the motor excitability than 1 mA due to more robust neurophysiological changes. This may be associated with increased firing rates in the membrane, prolonged changes in membrane potential and decreased inhibitory interneural activity.25,26,52
Despite the positive effects on postural responses of patients with PD, tDCS did not promote habituation in fewer trials. The delayed habituation in patients with PD may be related to the deficits in the basal ganglia function that are important to modulate the postural adjustments.10,53 Although delayed, patients can change their responses due to the involvement of other brain areas, such as the cerebellum and motor cortex seeking to compensate for the pathways deficits caused by the disease.16,54,55 tDCS did not promote habituation in fewer trials, but patients with PD changed the postural responses after the first trial and fully habituated those responses. This behavior was observed by the range of CoP (trials 3 and 2, respectively). A possible explanation is because the effects of tDCS on postural adjustments were mainly observed on onset latency, 6 and this parameter seems to be poorly responsive to habituation.5,56 Another explanation can be that the effect of tDCS on postural responses are present since the first trials. Previous studies suggested that the ability to change the postural response in patients with PD is because they demonstrate a higher postural response in the first trials, which enables more potential to present change in their response to the subsequent trials. 57 Thus, the effect of tDCS in the cortical and subcortical pathways could improve the postural responses since the first trials decrease the room for further improvement. Also, PFC activity did not change throughout the trials. Payne et al 58 indicated that the rate of change in muscle and cortical activities during the repetition of perturbation is not equal. These modifications do not seem to occur at the same time and are dissociated. 58 This could explain the habituation to the perturbation observed by the CoP and EMG parameters even with the lack of change in cortical activity following the trials present in this study.
A limitation of the present study is the lack of motor cortex behavior analysis, which could confirm our hypothesis of the greater involvement of direct pathway after anodal tDCS. However, the decrease of the PFC activity suggests this behavior due to its involvement in postural control. In addition, the ability of tDCS to change the cortical excitability is well established.25,26,59 Another limitation is the lack of comparison between patients with PD and healthy older adults, which could provide further evidence around the underlying pathology. Therefore, future studies should consider the analysis of the activity and excitability of the motor cortex and should investigate the relationship between the improvements of the postural response and cognition mainly when tDCS is applied over cognitive areas (ie, PFC). Furthermore, we did not control how long the tDCS effects remain on postural responses, and we did not analyze the postural responses habituation in situations with external perturbation after several sessions of tDCS at the 2-mA intensity. These features should be considered in future studies. Despite such limitations, our results suggest that a single session of tDCS, mainly with 2 mA, can improve the postural responses to perturbation in patients with PD. Such findings encourage further development of tDCS protocols for a potential future clinical use. Future studies should focus on determining the optimal stimulation parameters (eg, number of sessions, stimulation duration, montage, how long the effects remain, etc) and identifying potential predictors of response to tDCS. 24
Conclusion
Anodal tDCS over M1 improves postural response to external perturbation in patients with PD, with 2 mA leading to benefits in more parameters of postural control than 1 mA. However, the studied tDCS protocol seems to be inefficient in modifying the habituation to perturbation. Additionally, anodal tDCS over M1 led to reduced involvement of the prefrontal cortex in postural response to perturbation.
Footnotes
Acknowledgements
We gratefully thank all patients for their participation in our study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the São Paulo Research Foundation (FAPESP) (Grant Number #2018/07385-9, #2016/21499-1, #2014/22308-0); National Council for Scientific and Technological Development (CNPq) (Grant Number #142057/2017-7, #309045/2017-7); and financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior–Brazil (CAPES) (Finance Code 001).
