Abstract
Background. Many stroke survivors suffer from leg muscle paresis, resulting in asymmetrical gait patterns, negatively affecting balance control and energy cost. Interventions targeting asymmetry early after stroke may enhance recovery of walking. Objective. To determine the feasibility and preliminary efficacy of up to 10 weeks of gait training assisted by multichannel functional electrical stimulation (MFES gait training) applied to the peroneal nerve and knee flexor or extensor muscle on the recovery of gait symmetry and walking capacity in patients starting in the subacute phase after stroke. Methods. Forty inpatient participants (≤31 days after stroke) were randomized to MFES gait training (experimental group) or conventional gait training (control group). Gait training was delivered in 30-minute sessions each workday. Feasibility was determined by adherence (≥75% sessions) and satisfaction with gait training (score ≥7 out of 10). Primary outcome for efficacy was step length symmetry. Secondary outcomes included other spatiotemporal gait parameters and walking capacity (Functional Gait Assessment and 10-Meter Walk Test). Linear mixed models estimated treatment effect postintervention and at 3-month follow-up. Results. Thirty-seven participants completed the study protocol (19 experimental group participants). Feasibility was confirmed by good adherence (90% of the participants) and participant satisfaction (median score 8). Both groups improved on all outcomes over time. No significant group differences in recovery were found for any outcome. Conclusions. MFES gait training is feasible early after stroke, but MFES efficacy for improving step length symmetry, other spatiotemporal gait parameters, or walking capacity could not be demonstrated. Trial Registration. Netherlands Trial Register (NTR4762).
Introduction
Many stroke survivors suffer from limitations in functional mobility and an increased fall risk.1-4 Therefore, regaining independent walking is considered one of the primary goals in stroke rehabilitation.4,5 However, only 60% to 80% of the stroke survivors achieve this ability.3,5,6 After unilateral stroke, a limited walking capacity can be attributed to a variety of impairments on the paretic side, such as pes equinovarus, knee instability, and/or hip instability during the stance phase; and foot drop, limited knee flexion, and/or limited knee extension during the swing phase.2,7,8 These impairments are largely the result of paretic leg muscle weakness and disrupted timing of muscle activations during gait.3,4,7 The literature on gait restoration after unilateral stroke shows that, despite major improvements in gait independence, gait speed, and walking distance, the altered timing of muscle activity and the kinetic and kinematic gait abnormalities tend to persist,4,9-11 which is why stroke survivors greatly rely on compensatory mechanisms by the trunk and the nonparetic leg, especially moderately to severely affected individuals.2,4,9 As a result, their gait is often asymmetric and mechanically inefficient,4,10,12-14 even though it can be considered “optimal” given the neuromechanical consequences of stroke.12,15 Nevertheless, an asymmetric gait pattern forms a risk of overloading the trunk and the nonparetic side and is associated with limited gait adaptability, increased fall risk, fear of falling, and reduced physical activity.1,2,16
From this perspective, the ambition to restore gait symmetry is still an ultimate goal, which implies that the search for interventions to reduce gait asymmetry remains justified. Until now, several longitudinal studies reported persistent asymmetry during and after early rehabilitation,9,10,13,14 while there are no therapeutic interventions with established effectiveness for improving gait symmetry in persons after unilateral stroke.17,18 A possible reason is that controlled studies may not have focused specifically enough on improving gait symmetry by influencing kinetic and kinematic aspects of gait,17,18 for instance, by combining training with a medical-technical intervention such as neuromuscular electrical stimulation.13,19,20 Furthermore, many intervention studies measuring gait symmetry did not focus on the early time period (<12 weeks) poststroke,17,18 during which neurological recovery of the paretic leg is most pronounced and the “window of opportunity” to restore gait symmetry is believed to be optimal.3,9,20,21 Hence, there is a lack of studies focusing on gait training assisted by medical-technical applications in the early phase after stroke that aim to restore a symmetric gait pattern.
New techniques such as multichannel functional electrical stimulation (MFES) can facilitate gait training by supporting the activation of specific paretic leg muscles—adequately timed to the gait cycle—with the aim to promote a gait pattern that is as normal as possible.2,4 When applied in the subacute phase after stroke, gait training assisted by MFES (MFES gait training) may push the central nervous system toward more effective reorganization 20 and support the reacquisition of an adequate gait pattern22,23 by providing somatosensory input,23-25 maintain physical and physiological body characteristics,23-25 and prevent inappropriate compensations and secondary complications.23,25
Until now, there is little evidence for the feasibility and efficacy of MFES as an adjunct to conventional gait rehabilitation early after unilateral stroke to restore gait symmetry. Four small randomized controlled trials (RCTs) suggested that MFES, adequately timed to the gait cycle, early after stroke may improve motor function, balance control, gait speed and capacity, and functional activities of daily living.22,26-28 However, these studies had a high risk of bias by incomplete reporting of subject selection and results,22,26 unblinded outcome assessments,22,27 loss to follow-up, 28 and imprecision of effect estimates due to small sample sizes and poor statistical analyses.22,26-28 Moreover, three out of four studies were not dose-matched and investigated MFES in a supine position independent of gait rehabilitation.26-28 Only 1 small RCT (n = 13) investigated the efficacy of gait training assisted by MFES, but did not report on gait symmetry. 22 Therefore, it remains unknown whether conventional gait training assisted by MFES is feasible and effective for the restoration of gait symmetry early after unilateral stroke.
The present pilot RCT aimed to fill this knowledge gap by determining the feasibility and preliminary efficacy of up to 10 weeks of gait training assisted by surface-based MFES applied to both the peroneal nerve and a knee flexor (biceps femoris brevis) or extensor (vastus medialis) muscle. MFES gait training was compared with dose-matched conventional gait training regarding the restoration of step length symmetry (primary outcome) and other spatiotemporal parameters and walking capacity (secondary outcomes) in an inpatient rehabilitation population. Step length symmetry was considered a good measure of gait symmetry, because adequate bilateral step length is determined by kinematic characteristics of the swing phase (eg, sufficient hip flexion, knee extension, and ankle dorsiflexion) and indirectly by kinetic characteristics of the stance phase (eg, sufficient stance stability, ankle power, and propulsive impulse).7,8,29 Moreover, this parameter has shown to respond to the beneficial effects of implanted peroneal nerve stimulation in chronic stroke survivors, particularly in those with relatively pronounced step length asymmetry at baseline.30,31
Methods
We performed a stratified single-center, assessor-blinded, pilot RCT and randomized 40 participants to either the experimental group, receiving MFES gait training, or the control group, receiving gait training as usual. Computer-generated 4-block randomization and allocation was effectuated by an independent researcher allowing concealment for the next allocation. Randomization was stratified by functional walking capacity at enrolment, that is, “dependent gait” (Functional Ambulation Categories 1 and 2) versus “independent gait” (Functional Ambulation Categories 3 to 5). The intervention period lasted 10 weeks or until discharge from inpatient rehabilitation, whichever came first. Until data analysis was completed, the randomization was concealed to the primary researcher (MVB) and the outcome assessors and the participants were instructed not to reveal their group allocation. To ensure integrity of blinding, data were analyzed by an independent statistician. The RCT was registered in the Netherlands Trial Register (NTR4762) and the study protocol of this RCT was described in detail previously. 32
The study protocol was approved by the Medical Ethics Committee of the Academic Medical Center of the Amsterdam University Medical Centers (protocol number NL50002.018.14) and conformed to the standards set by the Declaration of Helsinki. Participants provided written consent prior to enrolment. 32
Trial Setting and Participants
Participants were recruited from one Dutch rehabilitation center (Merem Medical Rehabilitation, Huizen). All stroke survivors admitted for inpatient rehabilitation from November 2014 to September 2017 were screened for eligibility by their physiatrist. 32 The inclusion criteria were: clinical diagnosis of unilateral stroke; being within 31 days since stroke onset; age 18 to 80 years; indication for gait training (indicated in case of restrictions in walking activities and impairments in gait and/or balance); able to walk with or without aids and physical assistance from one physical therapist; and range of motion on passive ankle dorsiflexion ≥0° with full knee extension. Exclusion criteria were as follows: subarachnoid hemorrhage or cerebellar or brainstem stroke; severe spasticity of knee or ankle flexors or extensors (ie, Modified Ashworth Scale ≥3); medical comorbidities that might significantly interfere with gait (including a previous stroke with persisting gait deficits); severe cognitive problems or aphasia that might interfere with the ability to comprehend test instructions (based on clinical judgment); medical conditions that might lead to inability to comply with the study protocol; demand-type cardiac pacemaker, defibrillator or electrical implant; metallic implant at the paretic leg; or cancerous lesion at the paretic leg.
Interventions
Both groups received individualized conventional gait training for 30 minutes on weekdays (dose-matched). Individualized conventional gait training included improving walking distance and endurance, walking on uneven and smooth surfaces, walking with dual tasks, improving gait speed and adaptability, improving standing and walking balance, ramp ascent and descent walking, obstacle avoidance, functional gait activities, all with attention to gait quality. Most gait training sessions were overground, but treadmill walking was performed as well. Walking aids, orthoses, orthopedic shoes, and medication could be used. Additionally, participants received gait training in groups, fitness training, sports, and hydrotherapy depending on their personal needs and interests. In the experimental group, individualized conventional gait training was assisted by MFES (MFES gait training). Physical therapists, specifically trained in the protocol and use of MFES, carried out the gait training.
The MFES device used in this study (NESS L300 Plus, Bioness) delivers electrical pulses to muscles in the paretic leg to promote ankle dorsiflexion in combination with knee flexion or extension during gait. The electrodes of the lower leg cuff are positioned over the common peroneal nerve and the tibialis anterior muscle to elicit active ankle dorsiflexion. The lower leg stimulation starts at “heel off” and gradually terminates after “heel contact”, as it can be extended beyond heel contact to control the first rocker. The electrodes of the thigh cuff are positioned over the vastus medialis muscle (ventral positioning) to promote knee extension or over the biceps femoris brevis muscle (dorsal positioning) to promote knee flexion. With this configuration, either paretic muscles can be stimulated or excessive stiffness of spastic muscles can be counterbalanced.23-25,32,33 Authorized clinicians observed the gait pattern, performed additional tests (eg, muscle strength and spasticity), and determined which gait aspect (ie, stance stability, step length, foot clearance, foot prepositioning, or energy conservation) was inadequate and had priority to be improved. The vastus medialis muscle was stimulated to influence excessive knee flexion during the stance phase, or reduced knee extension during the stance or swing phase; or the biceps femoris brevis muscle was stimulated to influence knee hyperextension during the stance phase, or reduced knee flexion during the swing phase. 32 The effect of the MFES settings on the impaired gait aspect were evaluated and fine-tuned by gait observation to achieve the best possible gait pattern. The thigh stimulation could start and end once or twice at any segment in the gait cycle. Further information about the MFES settings has been reported elsewhere. 32 The authorized clinicians (physical therapists and orthotists) were specifically trained to fit the MFES device and set the stimulation parameters. They fitted the device at baseline and fine-tuned the settings of the device every 2.5 weeks.
Outcomes
Adherence was assessed by recording the number of sessions per week the participant attended during the training period, as monitored by the therapists. Participant satisfaction was assessed postintervention. Satisfaction with the experimental or conventional gait training was assessed on a 10-point numeric rating scale from 0 (“most unsatisfied”) to 10 (“most satisfied”). Moreover, satisfaction of the participants in the experimental group with the MFES device was evaluated by a questionnaire with 5-point Likert-type scales (1 = very unsatisfied, 2 = unsatisfied, 3 = neutral, 4 = satisfied, 5 = very satisfied) addressing comfort of wearing, quality of the gait pattern, walking distance, gait speed, effort of walking, stability during walking, and walking stairs.
Spatiotemporal gait parameters were assessed at baseline, 6 six weeks intervention, postintervention (maximally after 10 weeks), and at 3-month follow-up after cessation of the intervention. Additional secondary outcomes were assessed every 2 weeks during the intervention period as well as at 3-month follow-up.
The primary outcome of the efficacy study was step length symmetry. Step length during comfortable gait speed was measured with a customized spatiotemporal gait analysis system (SGAS). 34 Participants walked along a 10-m walkway until 5 valid left and right foot steps were collected in which each foot landed within the 130-cm-wide video field-of-view. The primary condition was walking without shoes and orthosis with as minimal as possible use of walking aids or physical assistance. The secondary condition was shod walking with as minimal as possible support.
Step length was calculated as the distance between the position of initial contact of one foot and the position of initial contact of the opposite foot in the sagittal plane (determined by the position of the heels or, in case of mid- or fore-foot landing, the toes). Step length symmetry was expressed as an index of asymmetry: the absolute difference between the step length of the paretic and nonparetic leg divided by the mean step length of both legs, multiplied by 100%. 35 An index with an inter-limb difference score as numerator was used, because a simple symmetry ratio might easily be inflated and show a skewed distribution.15,35,36 In addition, the absolute difference score was used, because step length asymmetry after stroke may go in either direction and gait training was aimed to restore each type of asymmetry toward symmetric gait. One-hundred percent was imputed for indexes exceeding this percentage (in cases where the swinging foot landed next to the opposite standing foot, giving a negative or small positive step length of one leg in contrast to a relatively large step length of the opposite leg). A value of 0% indicates perfect symmetry, while 100% indicates maximal asymmetry. Normative data are available for stroke survivors as well as for healthy adults.35,36 We applied a cut-off point for the step length asymmetry index of <7.6% as normal. 36 To our knowledge, the minimal clinically important difference and minimal detectable change for step length asymmetry have not been reported.
The secondary outcomes of the efficacy study were additional spatiotemporal gait parameters (step time asymmetry index, single-leg stance time asymmetry index, stride length, and stride time) for the barefoot and shod conditions,34-36 walking capacity measures (walking balance measured with the Functional Gait Assessment37,38 and comfortable gait speed calculated from the 10-Meter Walk Test 37 ), and balance control measured with the Berg Balance Scale. 39 Furthermore, fear of falling was evaluated by the Falls Efficacy Scale I (score range 16-64) and assessed only at 3-month follow-up. 40
Adverse Events
All adverse events were documented, regardless of their relationship to the MFES gait training, by the primary researcher or staff at each visit. 32
Statistical Analysis
Sample size was based on the feasibility of recruitment in one center (yearly admission rate of 80 stroke survivors) within 2 years and set at 40 participants (20 in each group). 32 In the absence of data on variance in step length symmetry, a sample size calculation could not be performed. However, we expected that a sample size of 40 participants would be large enough to inform the inclusion needed in a larger trial. 41
In order to assess the feasibility of the intervention, we evaluated adherence (based on the proportion of participants in the experimental group who attended ≥75% of the protocolled gait training sessions) and satisfaction with gait training (based on a score ≥7 on the numeric rating scale for participant satisfaction).
Primary efficacy analysis was performed on an intention-to-treat basis, including all participants who were randomized. In addition, a per-protocol analysis was performed for the primary outcome and was based on the participants with an intervention period of at least 5 weeks and who completed all assessments. Furthermore, subgroup analysis was performed for the primary outcome and was based on the participants with step length asymmetry at baseline (step length asymmetry index ≥7.6%).
Linear mixed models were used to assess the effect of treatment on the primary and secondary outcomes.42,43 All longitudinal analyses modeled the change from baseline as a function of time since randomization, adjusted for treatment, baseline value, and the stratification factor (Functional Ambulation Categories). All linear mixed models incorporated a random intercept per subject to account for the within-subject clustering. Time since randomization was modeled nonlinearly using natural splines with two degrees of freedom. 43 Treatment effect was assessed by adding an interaction term between time since randomization and treatment. The likelihood ratio test was used to determine the significance of the interaction. 43 The assumption on normality of the residuals was visually checked.
Considering the nonnormally distributed data of the participant satisfaction outcomes and Falls Efficacy Scale I, group comparisons postintervention and at follow-up were performed using Mann-Whitney U tests. In all analyses, statistical uncertainty was expressed by means of 95% confidence intervals. Significance was set at P < .05. All statistical analyses were performed in R version 3.5.1. 44 Linear mixed models were fitted using the lmer function in the R package lme4 (version 1.1-19). 45 Plots were generally constructed by using the ggplot function in the package ggplot2 (version 3.1.0). 46
Results
Of 188 persons consecutively admitted to inpatient stroke rehabilitation, 40 (21%) met the study criteria and agreed to participate. Figure 1 details the participant flow through the trial and reasons for dropout. Before baseline measurement, 2 randomized participants dropped out. A third participant dropped out during baseline assessment because of incorrect inclusion (unable to walk with aids and physical assistance from 1 physical therapist and severe cognitive problems). This participant was therefore excluded from analysis.
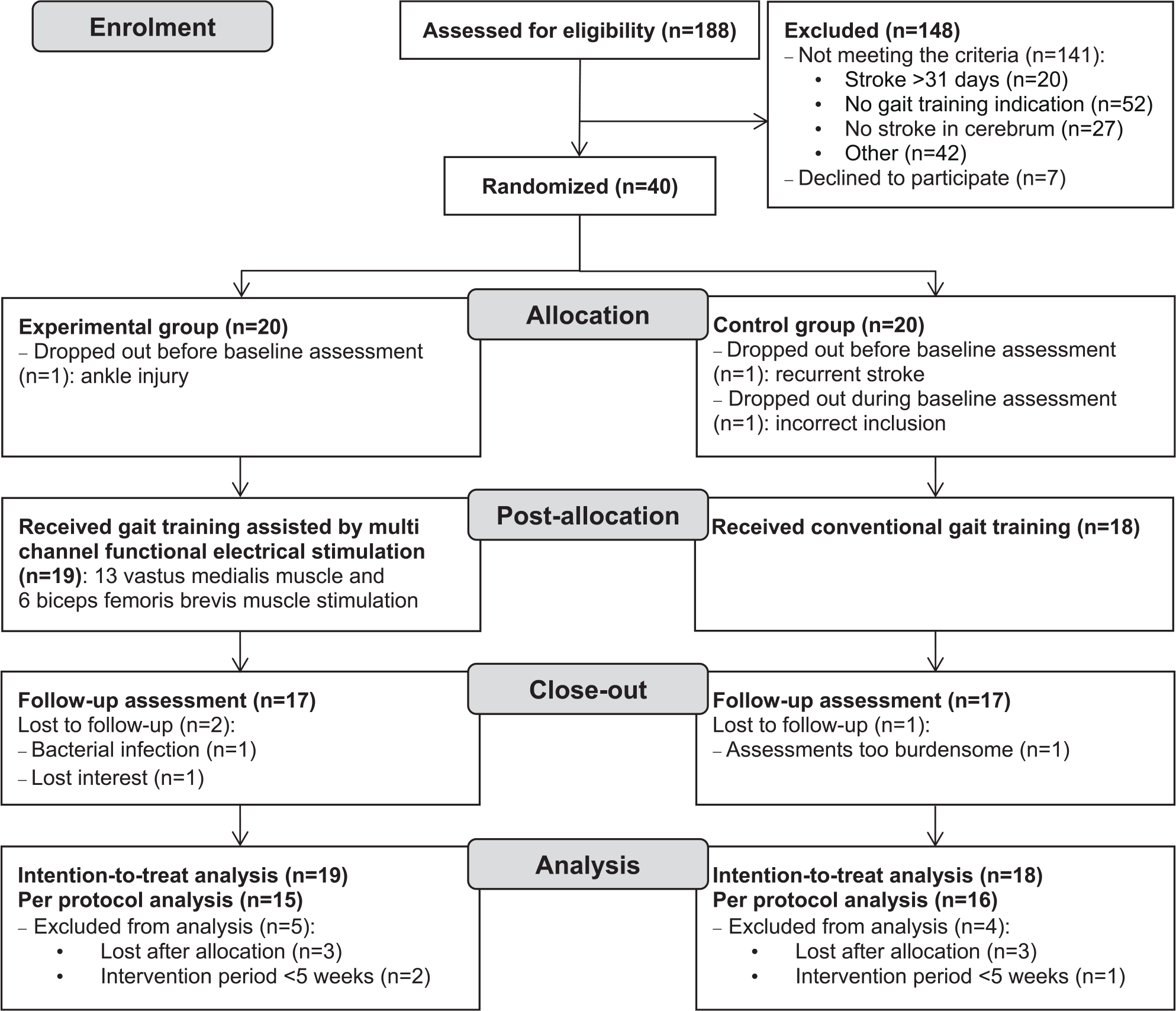
The flow of participants through the study.
Of the 37 participants, 19 were randomized to the experimental intervention and 18 to conventional gait training. Sociodemographic and stroke characteristics are shown in Table 1. In the experimental group, 13 participants received stimulation of the vastus medialis muscle and 6 of the biceps femoris brevis muscle. Supplementary File 1 provides information about the individual MFES device settings and training duration. For the per-protocol analysis, 31 participants (15 in the experimental group and 16 in the control group) were included (Figure 1). At baseline, 10 participants in the experimental group and 11 participants in the control group used walking aids (quad cane, cane, or rolling walker). Four participants in each group required an ankle-foot orthosis. Over time, the use of walking aids and orthoses changed in both groups, but this did not differ between groups (detailed information in Supplementary File 2). Low-dose benzodiazepines were used by 1 participant in the experimental group and by 2 participants in the control group. Low-dose antispasmodics were used by one participant in the experimental group. No antipsychotics were used.
Baseline Characteristics and Scores.
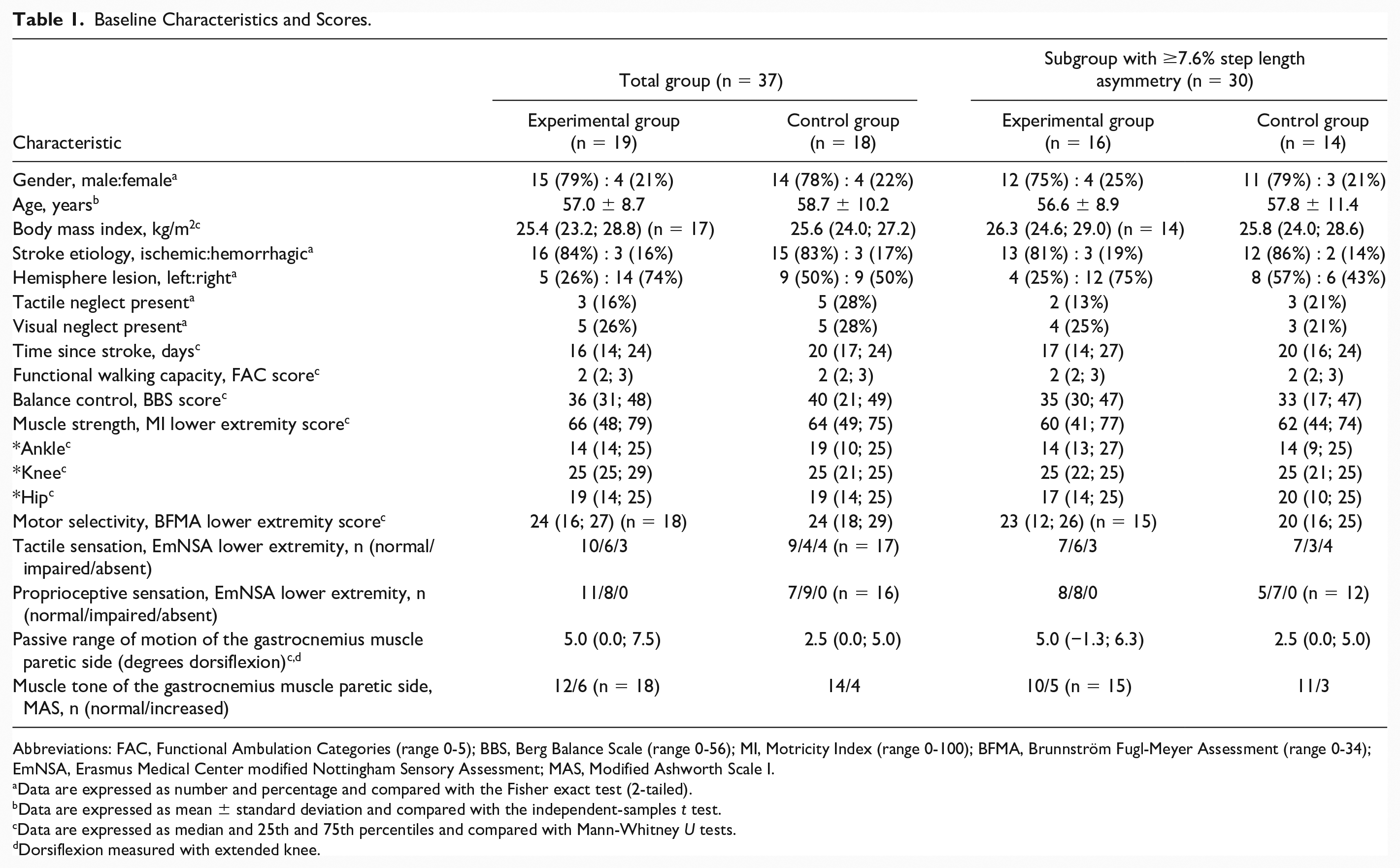
Abbreviations: FAC, Functional Ambulation Categories (range 0-5); BBS, Berg Balance Scale (range 0-56); MI, Motricity Index (range 0-100); BFMA, Brunnström Fugl-Meyer Assessment (range 0-34); EmNSA, Erasmus Medical Center modified Nottingham Sensory Assessment; MAS, Modified Ashworth Scale I.
Data are expressed as number and percentage and compared with the Fisher exact test (2-tailed).
Data are expressed as mean ± standard deviation and compared with the independent-samples t test.
Data are expressed as median and 25th and 75th percentiles and compared with Mann-Whitney U tests.
Dorsiflexion measured with extended knee.
Feasibility
The percentage of completed gait training sessions was 79% (mean total number of sessions 26 ± 9, range 7-39) for the experimental group and 79% (mean total number of sessions 27 ± 10, range 11-40) for the control group. Of the participants in the experimental group, 90% attended ≥75% of the MFES gait training sessions. The duration of gait training did not differ between the experimental group (mean 7.0 ± 2.0 weeks) and control group (mean 7.9 ± 2.5 weeks, P = .25). Per session, participants in the experimental group received approximately 20 minutes of MFES gait training due to donning and doffing of the device. Participants in the control group received approximately 25 minutes of gait training per session.
Satisfaction of the experimental group with MFES gait training ranged from 7 to 10 (median score 8 [P25; P75 8; 10]) and did not differ from the scores in the control group ranging from 7 to 10 (median score 8 [P25; P75 8; 9], P = .58). The participants were generally satisfied with the MFES device (comfort of wearing, walking distance, gait speed, and walking stairs median score 4 [P25; P75 3; 5]; quality of the gait pattern median score 4 [P25; P75 4; 5]; and effort of walking and stability during walking median score 4 [P25; P75 3; 4]).
Step Length Symmetry
Step length symmetry improved in both groups over time (Table 2). Step length symmetry was not normally distributed, therefore log-transformation was applied for statistical analyses. No group difference in step length symmetry was found on an intention-to-treat basis (log-transformed % estimated group difference of −0.07 with 95% confidence interval −0.50 to 0.36, P = .99; Table 2 and Figure 2 panel A) nor on a per-protocol basis (log-transformed % estimated group difference −0.17 with 95% confidence interval −0.64 to 0.31, P = .87; Supplementary Files 3 and 4 panel A). Natural splines were included in the models. Individual trends are shown in Figure 3. Similarly, subgroup analysis based on participants with step length asymmetry at baseline (16 participants in the experimental versus 14 participants in the control group) revealed no group differences (log-transformed % estimated group difference −0.03 with 95% confidence interval −0.53 to 0.48, P = .95; Supplementary Files 3 and 4 panel B).
Effects of Gait Training Assisted by Multichannel Functional Electrical Stimulation in the Experimental Group Compared With the Control Group (Intention-to-Treat Analysis).
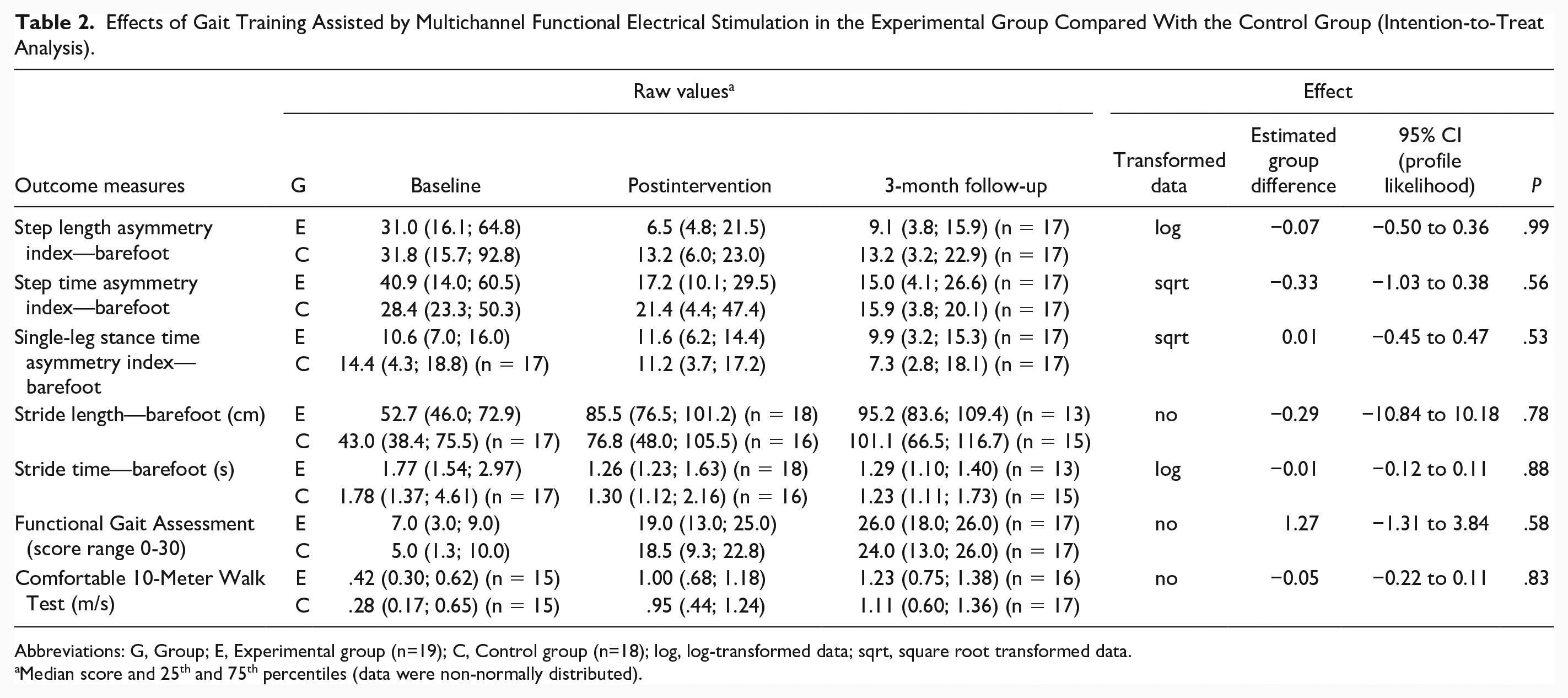
Abbreviations: G, Group; E, Experimental group (n=19); C, Control group (n=18); log, log-transformed data; sqrt, square root transformed data.
Median score and 25th and 75th percentiles (data were non-normally distributed).
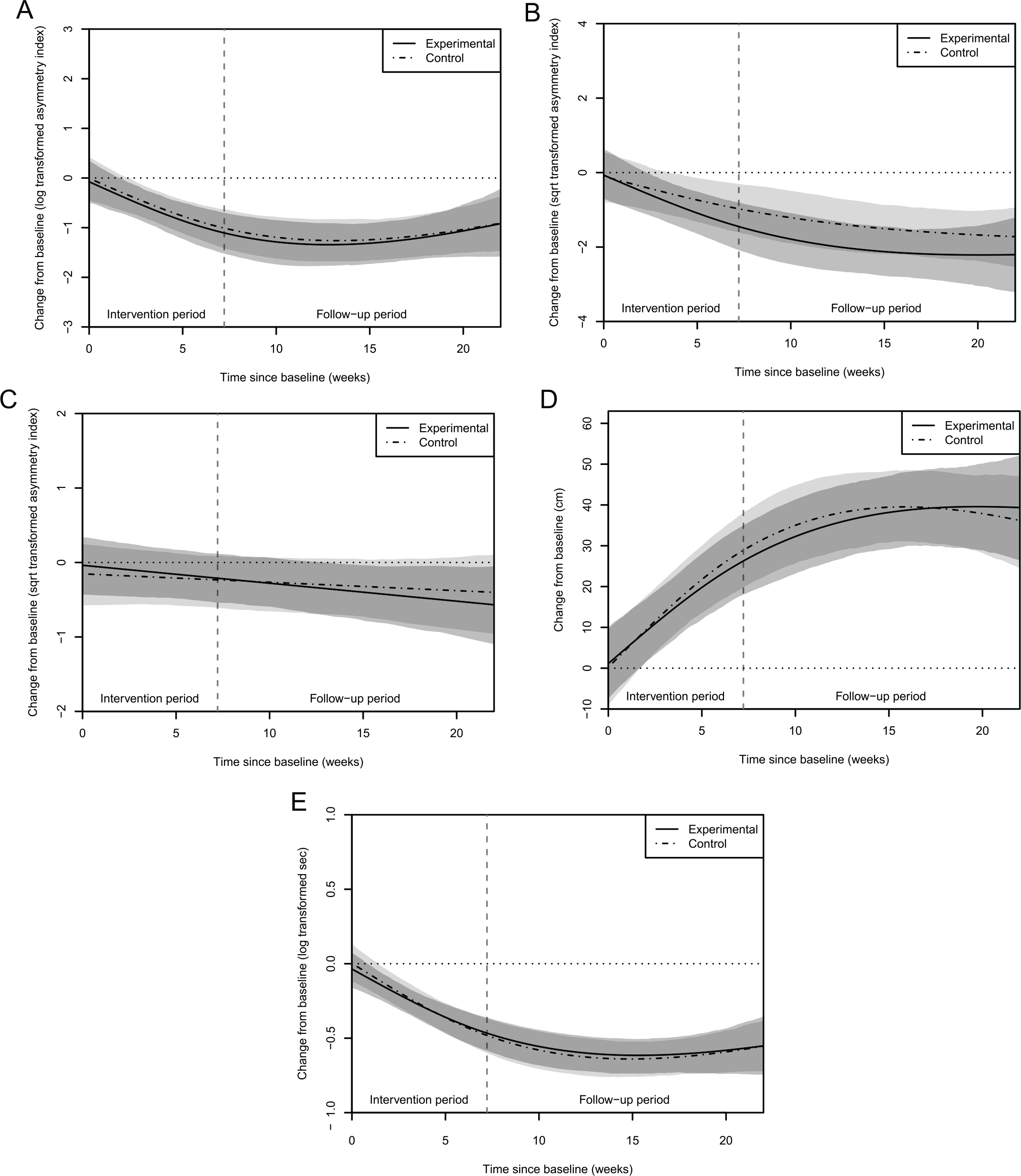
Time trends of asymmetry indexes and strides for both groups: the shaded areas indicate the 95% confidence intervals of the group means (light gray is control group, middle gray is experimental group, and dark gray is overlap between both groups). The vertical gray dashed line marks the end of the intervention period (overall mean). Panel A: Time trend of step length asymmetry. Panel B: Time trend of step time asymmetry. Panel C: Time trend of single-leg stance time asymmetry. Panel D: Time trend of stride length. Panel E: Time trend of stride time.
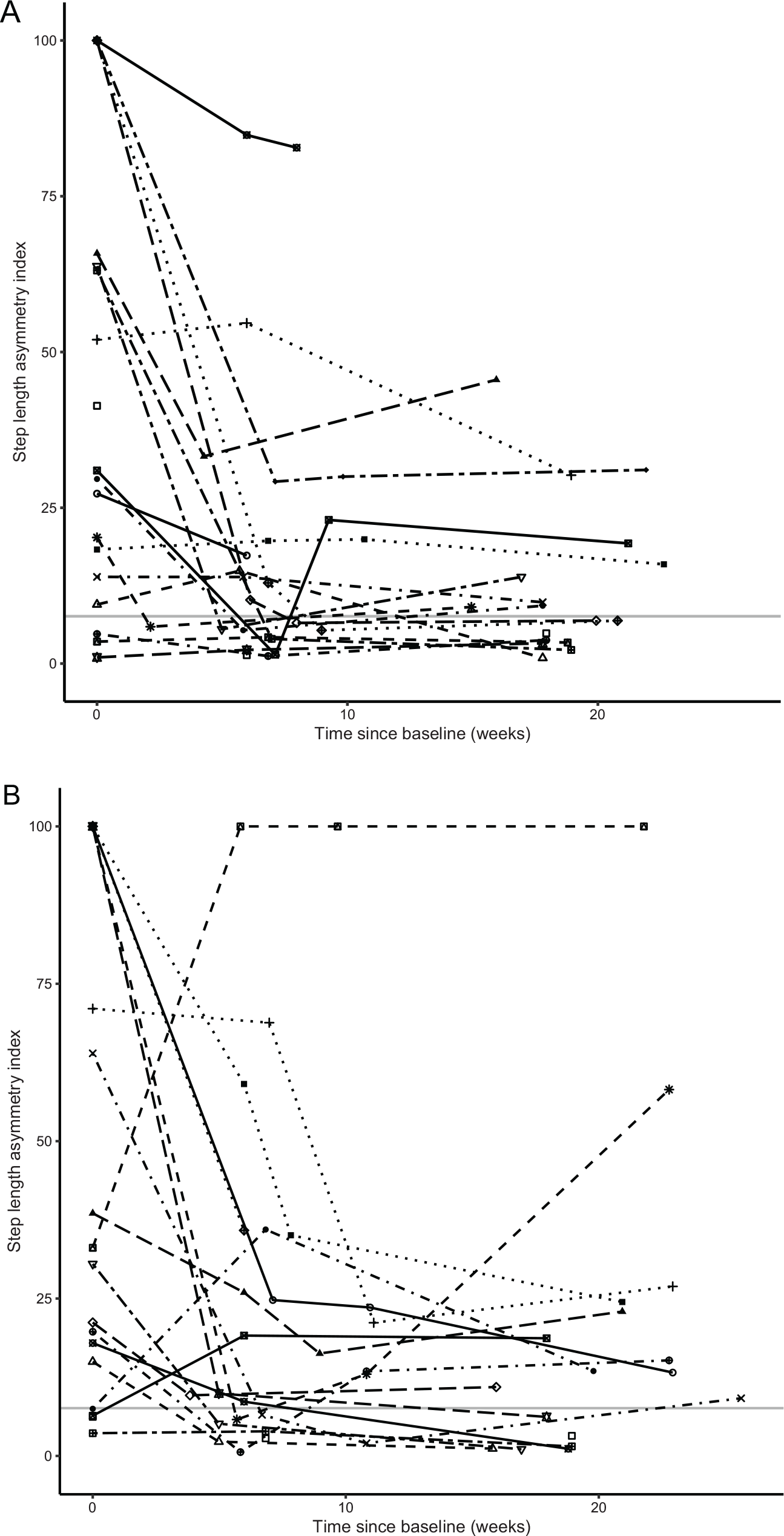
Individual time trends of step length asymmetry (indicated with unique symbols per individual) for both groups: the horizontal gray line represents the normative cutoff point. The symbols indicate the assessments (baseline, if applicable after 6 weeks intervention, postintervention (maximally after 10 weeks), and at 3-month follow-up). Panel A: Experimental group. Panel B: Control group.
A comparable result as for barefoot walking was found for step length symmetry in the shod condition (log-transformed % estimated group difference −0.03 with 95% confidence interval −0.56 to 0.52, P = .46; Supplementary Files 3 and 4 panel C).
Secondary Outcomes
Both groups showed improvements on all outcomes over time, except for single-leg stance time symmetry in the control group (Figure 2). No significant group differences were found for any of the secondary outcomes (Table 2 and Figure 4). The outcomes of the shod condition and Berg Balance Scale are presented in Supplementary Files 3 and 4. Fear of falling, measured with the Falls Efficacy Scale I, at 3-month follow-up did not differ significantly (P = .72) between the experimental (median score 20 [P25; P75 19; 22], n = 17) and control group (median score 20 [P25; P75 17; 26], n = 15).
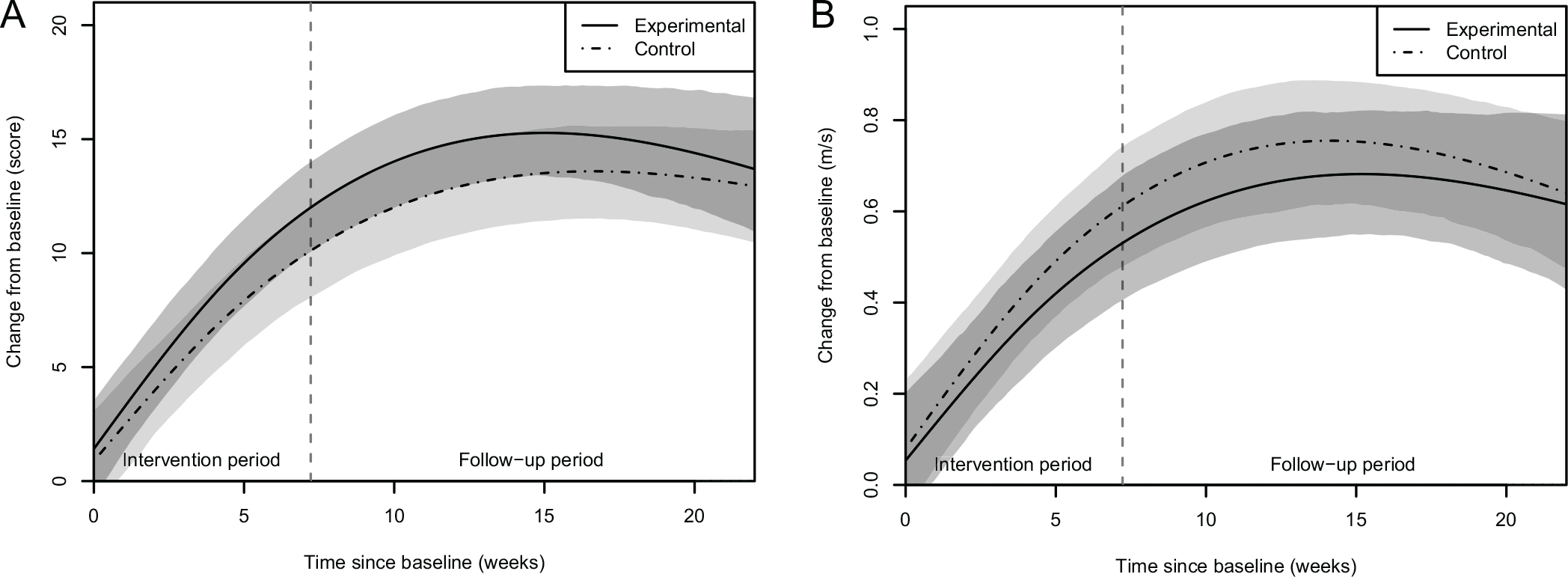
Time trends of clinical outcomes for both groups: the shaded areas indicate the 95% confidence intervals of the group means (light gray is control group, middle gray is experimental group, and dark gray is overlap between both groups). The vertical gray dashed line marks the end of the intervention period (overall mean). Panel A: Time trend of walking balance (Functional Gait Assessment). Panel B: Time trend of gait speed (10-Meter Walk Test).
Adverse Events
One adverse event was reported concerning MFES gait training. A participant developed a small wound localized in the popliteal fossa due to movement of the thigh cuff, which did not require medical treatment. Tighter attachment of the cuff solved the skin irritation. One adverse event was reported concerning the assessments. A participant in the control group perceived the assessments as too burdensome and discontinued study participation (Figure 1).
Discussion
Our study showed that MFES gait training, initiated in the first month after stroke and continued for up to 10 weeks during inpatient rehabilitation, was feasible and well appreciated. However, our results suggest that MFES gait training was not superior to conventional gait training for improving gait symmetry, other spatiotemporal gait parameters, or walking capacity. Both groups demonstrated similar improvements following the intervention period. The plateaus in the recovery trends seen at follow-up (Figures 2 and 4) were in accordance with previously reported recovery patterns for walking and activities of daily living in stroke survivors receiving rehabilitation.9,14,47-49
Our hypothesis was that MFES gait training started early after stroke—within the presumed critical time window for neuroplasticity—would improve step length symmetry and prevent inadequate compensatory motor strategies, thereby facilitating a more normal gait pattern. However, MFES gait training appeared to be no more effective than conventional gait training. In both groups gait symmetry improved, except for single-leg stance time symmetry in the control group (Figures 2 and 4 and Table 2). On an individual level, the time trends were diverse, while 19% of the participants already had a symmetric step length at baseline (Figure 3). Because a ceiling effect in these participants could have biased the results, we performed a post-hoc subgroup analysis including only the 30 participants with step length asymmetry at baseline. This analysis showed similar results as the primary analysis of the entire study sample.
In the literature, there is conflicting evidence about the persistence of gait asymmetry after stroke. Previous small, short-term studies in diverse stroke populations have shown increases in gait symmetry over time.17,19 Other studies, however, reported no improvement in step length or swing time symmetry during inpatient rehabilitation after stroke up to 6 months follow-up.9,10,13,14 Given the potential long-term consequences of persisting gait asymmetry (ie, increased risk of falls and injuries, reduced gait efficiency, poor aesthetics, risk of muscle shortening, joint deformation, and pain complaints), restoration of gait symmetry remains an important clinical issue. However, based on the available evidence, it is still questionable whether restitution of motor function after stroke can be influenced by training interventions beyond the influence of spontaneous neurological recovery. Although step length symmetry must be regarded as a surrogate outcome for restitution of motor function after unilateral stroke, it is most likely strongly influenced by the restoration of motor control of the paretic leg in terms of its kinematic and kinetic characteristics.7,8,29 Until now, neither animal nor human studies have been able to show that leg motor impairments after stroke can be restored by specific training interventions to improve the quality of motor performance.48,50,51 This is supported by the growing body of evidence suggesting that the degree of leg motor recovery after stroke is highly predictable in terms of synergism and muscle coordination, showing an almost invariant proportional relationship between leg motor impairment early after stroke and after 6 months.3,9,47,48,51 Hence, currently there is insufficient evidence to recommend the clinical use of MFES in early stroke rehabilitation.25,52-54 This is in line with the outcome of our study and the lack of evidence from earlier studies investigating the efficacy of daily 30 to 45 minutes of isolated MFES applied to the knee and ankle flexors and extensors early after stroke.22,26-28
The current study has several limitations, some of which are inherent in our aim to investigate feasibility and preliminary efficacy. First, this pilot trial may have been underpowered for efficacy assessment. Nevertheless, we do not suspect a false negative outcome based on the small, nonsignificant estimated treatment effects (Table 2) and the extensive overlap between the recovery trends in the models of the experimental and control group (Figures 2 and 4). Second, the inclusion criteria for selecting participants may not have been optimal, as 19% of the participants already had a symmetrical step length at baseline. Although subgroup analysis including only the participants with step length asymmetry at baseline showed similar results as the primary analysis, we were unable to take into account initial leg motor function given the small sample size. The differential response to training (Figure 3) suggests that future MFES studies with the aim to restore gait symmetry should focus on individuals with initial gait asymmetry and with a fair potential for restoration of leg motor function.12,15,18,19,48,51 Third, using quantitative gait analysis instead of visual analysis to determine the position and parameter settings of MFES would have increased standardization and precision, but we preferred to use a standardized clinical approach from a pragmatic, clinical perspective. Fourth, as argued above, step length symmetry must be considered a surrogate outcome measure for restoration of motor control of the paretic leg, which is why future studies should also incorporate kinematic and kinetic gait characteristics and muscle activation patterns. Fifth, the spatiotemporal gait data were characterized by a high stride-to-stride variability, which might have been due to a short warming-up period and a small number of repetitions. 55 The number of 5 steps per leg was chosen to minimize the burden on participants. The impact of this choice on our results remains unknown. For future studies, we recommend to extend the warming-up period and increase the number of strides to assess gait symmetry. Sixth, the intensity levels (amplitudes) of MFES turned out to differ per training session, but this was not accurately logged by the therapists. In future studies, MFES settings should be logged during the entire period of gait training to better control for treatment intensity. Seventh, the applied dose (approximately 20 minutes MFES gait training per working day) may have been insufficient. Moreover, the intervention period and therefore the total dose differed substantially between participants (Supplementary File 1). Yet, we provided the largest feasible dose that could be administered during regular gait training at our rehabilitation center given the participants’ length-of-stay. Intensifying MFES gait training may not be feasible unless the training is combined with daily “orthotic” application of MFES during the period of (in- and outpatient) gait rehabilitation. Indeed, there is preliminary evidence for the notion that such “orthotic” use of functional electrical stimulation is able to promote gait symmetry after unilateral stroke.30,31,56
Conclusions
This pilot RCT showed that MFES gait training initiated in the subacute phase after unilateral stroke is feasible. Nevertheless, efficacy for improving step length symmetry, other spatiotemporal gait parameters, or walking capacity in a fairly unselected group of typical rehabilitation inpatients seems similar to conventional gait training. Our results, however, do not preclude beneficial effects of MFES gait training in a more targeted population with a better potential for the restoration of gait symmetry.
Supplemental Material
sj-docx-10-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-docx-10-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-1-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-1-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-11-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-11-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-12-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-12-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-13-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-13-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-2-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-2-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-4-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-4-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-5-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-5-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-6-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-6-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-7-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-7-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-8-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-8-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-9-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-9-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Supplemental Material
sj-xlsx-3-nnr-10.1177_1545968320981942 – Supplemental material for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-xlsx-3-nnr-10.1177_1545968320981942 for Feasibility and Preliminary Efficacy of Gait Training Assisted by Multichannel Functional Electrical Stimulation in Early Stroke Rehabilitation: A Pilot Randomized Controlled Trial by Maijke van Bloemendaal, Sicco A. Bus, Frans Nollet, Alexander C. H. Geurts and Anita Beelen in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We gratefully acknowledge all participants and colleagues of Merem, Amsterdam UMC, and ProReva involved in this study. We would like to thank W. Bout, MSc, and P. Buitelaar, MSc, for their support in the data collection. Particularly, we would like to thank R.P.A. van Eijk, MD, PhD, for his statistical expertise.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the foundation “Stichting Vrienden van Merem,” Amsterdam, The Netherlands. The funding source has no influence on the trial design, collection, analyses, interpretation, and reporting of results.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
