Abstract
Background
Pain of neuropathic origin in spinal cord injury (SCI) is unbearable and challenging to treat. Research studies conducted in the past have shown that mental imagery (MI) techniques have a significant impact on the reduction of symptoms of central neuropathic pain in people with SCI.
Objectives
The objective of this study was to evaluate the effect of MI training on pain intensity, neuropathic pain symptoms, and interference of pain with function in SCI.
Methods
A total of 42 SCI participants with central neuropathic pain (duration 6-12 months) were recruited and randomly allocated to MI or control groups. A MI training protocol was administered to MI group and for 30 min/d for 5 days. Outcome measures were assessed at baseline and at the end of 4 weeks.
Results
There was significant reduction in differences of mean [95% CI] scores of numeric rating scale (−2.1 [CI −2.78 to −1.41];
Conclusions
This study shows the effectiveness of the MI protocol developed as a rehabilitative approach in improving central neuropathic pain in SCI.
Keywords
Introduction
Neuropathic pain can be defined as an unpleasant sensory and emotional experience arising from a primary lesion or disease of the somatosensory nervous system. 1 Research shows that approximately 40 to 60% of people with spinal cord injury (SCI) develop neuropathic pain that commonly appears within the first year of injury. 2 This type of pain could be spontaneous or evoked (such as allodynia and hyperalgesia) and may be associated with abnormal sensations (such as dysesthesias and paresthesias). 3
In people with SCI, pain of neuropathic origin commonly occurs at or below the lesion as a result of posttraumatic changes in the spinal cord. 4 Typically chronic in nature, it may hinder sleep, participation in activities of daily living, and social functioning resulting in reduced life satisfaction and poor quality of life. 5
Pain management approaches include analgesics, surgery, and nontraditional approaches. 6 Nontraditional approaches such as mind-body therapies provide significant pain relief by modulation of pain processing pathways. 7 Techniques such as hypnosis, mirror therapy, guided imagery (GI), visual illusion, and mental imagery (MI) training are believed to alter cortical reorganization associated with central pain. 8 Of these, MI is the process through which we symbolize information in our minds in the absence of appropriate sensory inputs. 9
GI is a form of imagery that helps create multimodal sensory-rich images resembling the actual perception of some object, scene, or event in the absence of external stimuli. 10 It has been described as the interaction of mind and body to bring about changes in physical, emotional, or spiritual dimensions using the power of imagination. 10 In GI, the individuals use their power of imagination to bring about physical and emotional changes in them. They are “guided” with music and quiet narrations to create sensory awareness. The results of GI for chronic pain in patients with cancer, 11 nonmusculoskeletal conditions, 12 and postoperative pain 13 have been encouraging. Schwoebel et al 14 suggested that the severity of pain influences the mental representation of movements. Left/right judgments of body images particularly hands (or feet) known as laterality training has been used individually or as a part of graded motor imagery to reduce chronic pain in neurological conditions. 15
In people with SCI, disrupted body schema is known to result in sensorimotor mismatch leading to chronic pain. 16 Different forms of MI techniques have been used either alone17,18 or in combination with other interventions19,20 for chronic pain in SCI in the past. In most of these studies, pain was reported to be reduced.17,19 However, conflicting observations were reported by Gustin et al, 18 which could be due to the type of the injury, pain, and technique of imagery used. One of the more commonly used approach, graded motor imagery has been effectively used for treatment of central neuropathic pain in neurological conditions such as stroke, 21 phantom limb pain, 22 and complex regional pain syndrome. 23 This approach encompasses 3 stages (laterality training, imagined hand movements and mirror visual feedback). In the third stage of this approach (ie, mirror visual feedback stage) a patient is required to place the affected limb inside a mirror box and watch movements of their nonaffected limb in the mirror. 24 However, this has limited scope for tetraplegic patients due to their bilateral involvement. In this study, we developed a protocol consisting of laterality training which is a form of implicit motor imagery 21 followed by GI. Studies show that chronic pain results in altered representation of body parts, including right/left discrimination. 14 We believe that laterality training would contribute to reduce the effect of somatosensory dysfunction on imagery and subsequently help reduce maladaptive plasticity effects as a result of chronic pain. 25 As GI allows individuals to use their own imagination to affect their psychological and physiological states 10 ; we believe laterality training prior to guided imagery will improve precision in imagery representation hence reducing pain.
Previous studies utilizing MI in patients with SCI have used diverse techniques but the sample size was low and hence unconvincing. Moreover, neuropathic pain in SCI occurs due to central sensitization and there is a great potential for mind-body therapies as they influence the mind to affect the pain symptoms. Gard et al 26 studied brain mechanisms by which pain can be modulated by techniques that create awareness of symptoms. They found reduced activation in the lateral prefrontal cortex and enhanced activation in the right posterior insula and anterior cingulate cortex along with improved emotional regulation. 26 The pain in people with SCI is common and debilitating in nature. However, there is inadequate evidence regarding current treatments using mind-body interventions such as MI for neuropathic pain in SCI. Additionally, techniques such as MI have fewer side effects. So, in the first part of our study we developed a MI protocol consisting of guided imagery and laterality training to be used in patients with SCI having neuropathic pain. Hence, the primary objectives of this study were to (1) assess the effect of mental imagery protocol (consisting of laterality training and guided imagery) on neuropathic pain intensity scores and neuropathic pain symptoms and (2) to measure the effect on pain interference with function scores in SCI participants having neuropathic pain.
Methods
The study consisted of 2 phases: protocol development and protocol delivery.
Phase I: Protocol Development
In the first phase of the study, a comprehensive literature review was conducted with the aim to identify types of MI interventions used to treat central neuropathic pain in neurological conditions. Studies pertaining to the use of imagery techniques for management of neuropathic pain in SCI were explored individually in depth.17-20,27 Articles were searched from databases such as MEDLINE, Embase, Cumulative Index to Nursing and Allied Health Literature (CINAHL), Scopus, Academic Search Premier, Web of Science, Allied and Complementary Medicine, the Cochrane Collaboration, and the Physiotherapy Evidence Database (PEDro). The primary search was conducted using combinations of keywords such as “mental imagery,” “GI,” “hypnosis,” “central neuropathic pain,” “pain relief,” “visual illusion,” “spinal cord injury,” “mental imagery techniques,” “laterality training,” “visual imagery,” and “kinesthetic imagery.” A protocol was then developed after reviewing the literature thoroughly. It consisted of 2 parts: laterality training and GI.
Laterality Training
Laterality training involves right/left discrimination of body parts such as hands and feet. For the purpose of providing laterality training, an interface was developed. Realistic pictures of hands and feet were clicked and presented in the software. Sixteen pictures of hands (2 hands × 2 views × 4 orientations) at the following angles of rotation: 0°, 90°, 180°, 270°, and 8 depictions of feet (2 feet × 4 orientations) making it a total of 24 stimuli were used. 28 The software randomized the pictures in such a manner that the participants would be presented with a total of 192 stimuli in 8 sets. The participant was given 5 seconds to respond to each stimulus. If no response were made within 5 seconds, the next image would appear. The software recorded the time as 5 seconds and a blank response for that image.29,30
Guided Imagery
The second part of the protocol consisted of GI session. For this purpose, a GI script was developed in English. The script was created based on findings of previous professional literature. An audiotape of the script was recorded with the background of soothing music (meditation) by professionals. 13 The 15-minute instruction audio consisted of suggestions to help subjects to relax, then slowly increase their awareness of all body parts especially those experiencing neuropathic pain symptoms and gradually eliminating them from the body. The process of validation of script was done by a focus group consisting of 5 individuals on script content and sound quality. The script once approved was then translated into local language by professional translators and recorded in the native language of subjects for better understanding.
Phase II: Protocol Delivery
A clinical trial was designed to see the efficacy of the protocol on SCI participants having central neuropathic pain.
Study Design
A prospective, assessor blinded, randomized, controlled, parallel group design was undertaken. The participants were randomly assigned to mental imagery (MI) group or a control (CONT) group. The total duration of study was 4 weeks. The period of recruitment was from May 2018 to June 2019. The experimental design summarized in Figure 1 is consistent with current Consolidated Standards of Reporting Trials (CONSORT) schematic.
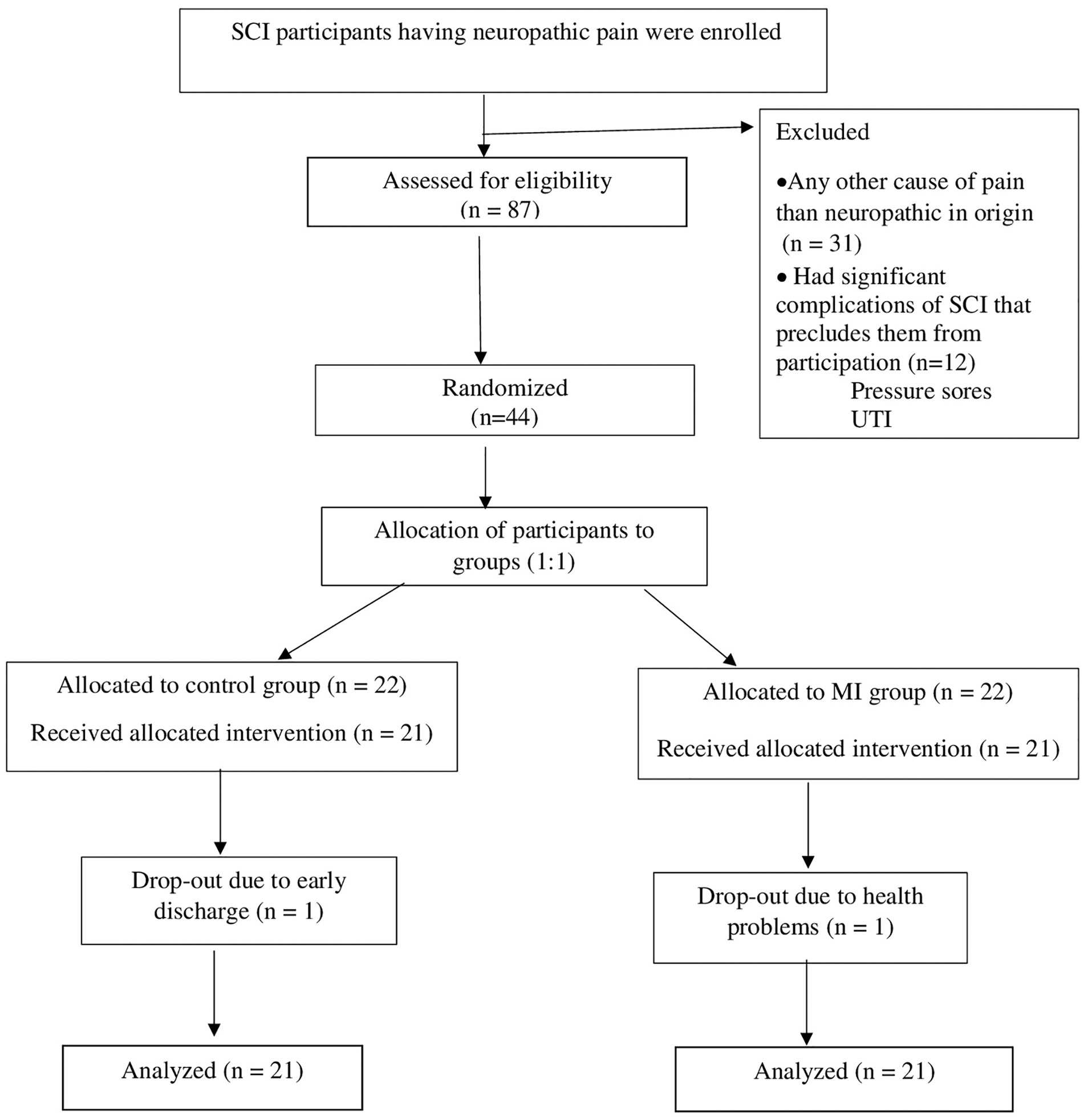
CONSORT (Consolidated Standards for Reporting of Trials) flow diagram showing eligible participants recruitment through each stage of the randomized, clinical trial. SCI, spinal cord injury; MI, mental imagery; UTI, urinary tract infection.
Participants
Forty-four participants with SCI having neuropathic pain were recruited from Indian Spinal Injuries Centre, New Delhi, India. Those interested in participating had to undergo a detailed screening against the eligibility criteria for enrollment in the study. The demographic details were obtained and after thorough neurological examination participants were selected based on inclusion and exclusion criteria. SCI participants were included if they (1) had neuropathic pain diagnosed by Douleur Neuropathique en 4 questionnaire (≥4 cutoff), 31 (2) belonged to the age group of 18 to 60 years, (3) C3 and below neurological level of injury, (4) had both complete and incomplete injury (American Spinal Cord Injury Association Impairment Scale grading A-D), (5) had an ability to imagine (assessed by visual analogue scale for vividness), 32 (6) were medically stable, (7) were willing to give informed consent, and (8) were able to understand the instructions. Participants were excluded if they (1) had any pain other than neuropathic in origin (like visceral pain or musculoskeletal pain); (2) had significant impairment or disability, including physical, neurological or psychological impairments, additional to the spinal cord injury that precludes them from participation in the study; (3) had auditory or visual impairment; (4) had been diagnosed with severe depression (scores on the Beck Depression Inventory–II ≥29); (5) had an existing stage 3 or 4 pressure ulcer according to the National Pressure Ulcer Advisory Panel classification; (6) had severe spasticity (scores of ≥3 on the modified Ashworth Scale); or (7) had recently changed their pain medications in the past 2 weeks.
Pain medications taken by participants were noted. They were instructed to inform and refrain from any change in medications, 2 weeks before and after completion of the study.
Ethical Considerations
Prior to screening, all potential trial participants were informed verbally and in writing about the purpose of this trial, its potential risks, costs involved, benefits of participation, and the right to withdraw. A written informed consent was taken from all the participants who were interested in participating in the study. The participants signed the informed consent and in case of tetraplegics who were unable to sign; thumb impressions were taken. The study protocol was approved by the Institutional Research Review Committee and the Institutional Ethical Committee (ISIC/RP/2018/091) of Indian Spinal Injuries Centre, New Delhi. The trial was registered with the Clinical Trials Registry–India under Indian Council of Medical Research (CTRI/2018/07/014884).
Randomization
The samples were randomized by a computer-generated randomization sequence by a blinded person other than the principal investigator and researchers associated with the study. The allocation schedule was sequentially numbered and sealed in opaque envelopes. A person not associated with the study opened the numbered envelopes sequentially to reveal the participant’s group allocation. Participants were allocated to one of the following 2 groups: mental imagery (MI) group and control (CONT) group.
Intervention
MI Group
Participants were made to sit comfortably with hands resting on armrest of the chair/wheelchair and feet on footrest of the wheelchair/floor. A laptop containing the software developed for laterality training (described previously) was placed at a distance of 60 cm from the participants’ eyes. 33 The participants were asked to sit comfortably with their hands resting palm down on the keyboard of the laptop. Before starting the session, they were instructed that they would be presented with pictures of (right and left) hands and feet in different directions, and they have to differentiate and respond quickly but accurately to each stimulus as right or left. The means to respond could be either pressing a computer key (“a” key for “left” and “l” key for “right”) or through eye gaze which has to be selected prior to training. 28 The training involved 8 sets of 24 stimuli presented in random order. The responses appeared on the computer screen as left/right and “no” if no response was made. Response times were taken from all responses whether they were correct or not. The total duration of laterality training was 15 minutes. Thereafter, the participants took part in the GI session. They were made to wear headphones and close their eyes. Participants were told to relax and listen to prerecorded audio script. They had to follow the script as closely and as accurately as possible in order to get the maximum effect.13,34,35 GI session was be administered for 15 minutes. The total duration of the intervention was 30 minutes, 5 days in a week for 4 weeks.
Control Group
The CONT group was asked to perform random addition task of 1-digit numbers through the software. There were one right and the other wrong answer and they had to select the correct response. They responded by either pressing the key or gazing of eyes in the direction of correct response. The duration of arithmetic task was 15 minutes. After 15 minutes, participants were asked to select songs out of a range of music CDs available to them and listen to their choice of music for 15 minutes.
Outcome Variables
The measures selected as outcomes were selected in accordance with the IMMPACT guidelines. 36 Outcome measures were assessed preintervention and after 4 weeks postintervention. For measuring pain intensity, visual analogue scale and numeric rating scale were used. 1 Neuropathic Pain Symptom Inventory, a self-administered questionnaire, was used to evaluate different symptoms of neuropathic pain. 37 The effect of pain on daily activities was measure by Pain interference measure of Brief Pain Inventory. 38 For this scale, the term mobility in was used as walking ability item was not relevant for most of the participants. 19 Patients’ Global Impression of Change is a single-item rating used to assess improvement with treatment. 39
Statistical Methods
For phase II of the study, sample size estimation was done to test the improvements in the outcome measures in MI group over CONT group. An a priori sample size estimation was performed using software G*Power 3 (v3.1.2-3.1.9) (Heinrich Heine University). Using an α level of .05, a 1 − β of 0.8 and an allocation ratio of 1:1, the total sample size was calculated as 42.
19
Potential loss to attrition was estimated as 5% based on researches; the total sample size was 44. The demographic data and the outcome measures’ data were normally distributed. Levene’s test was used to test the equality of variance and data normality. Pearson’s chi-square and chi-square for significance analysis of proportional data were used for nominal and ordinal variables, respectively. Parametric analyses were used in the study because the outcome measures’ data were normally distributed and the sample size was adequate. Mean change scores were calculated as the difference between post- and pretest scores and an independent
Results
Demographic Details and Neuropathic Pain Characteristics of Participants
Out of 44 participants recruited; there were 2 dropouts. The remaining 42 participants were randomized into MI group and CONT group having 21 participants each. The demographic details of participants are summarized in Table 1. The neuropathic pain characteristics of participants are elaborated in Table 2.
Demographic and Clinical Characteristics of Participants of the MI Group (n = 21) and the Control Group (n = 21). a
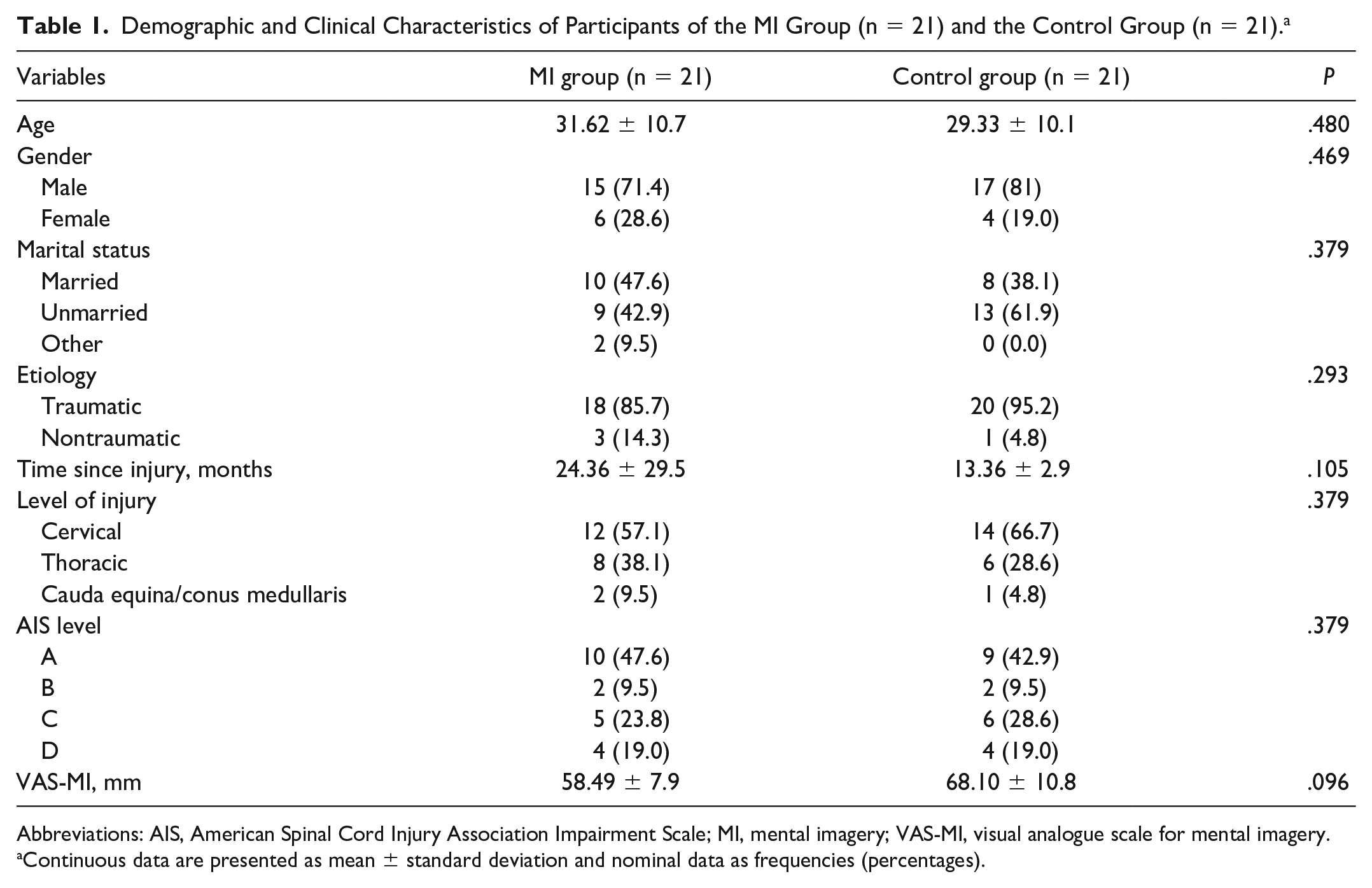
Abbreviations: AIS, American Spinal Cord Injury Association Impairment Scale; MI, mental imagery; VAS-MI, visual analogue scale for mental imagery.
Continuous data are presented as mean ± standard deviation and nominal data as frequencies (percentages).
Neuropathic Pain Characteristics of Participants.
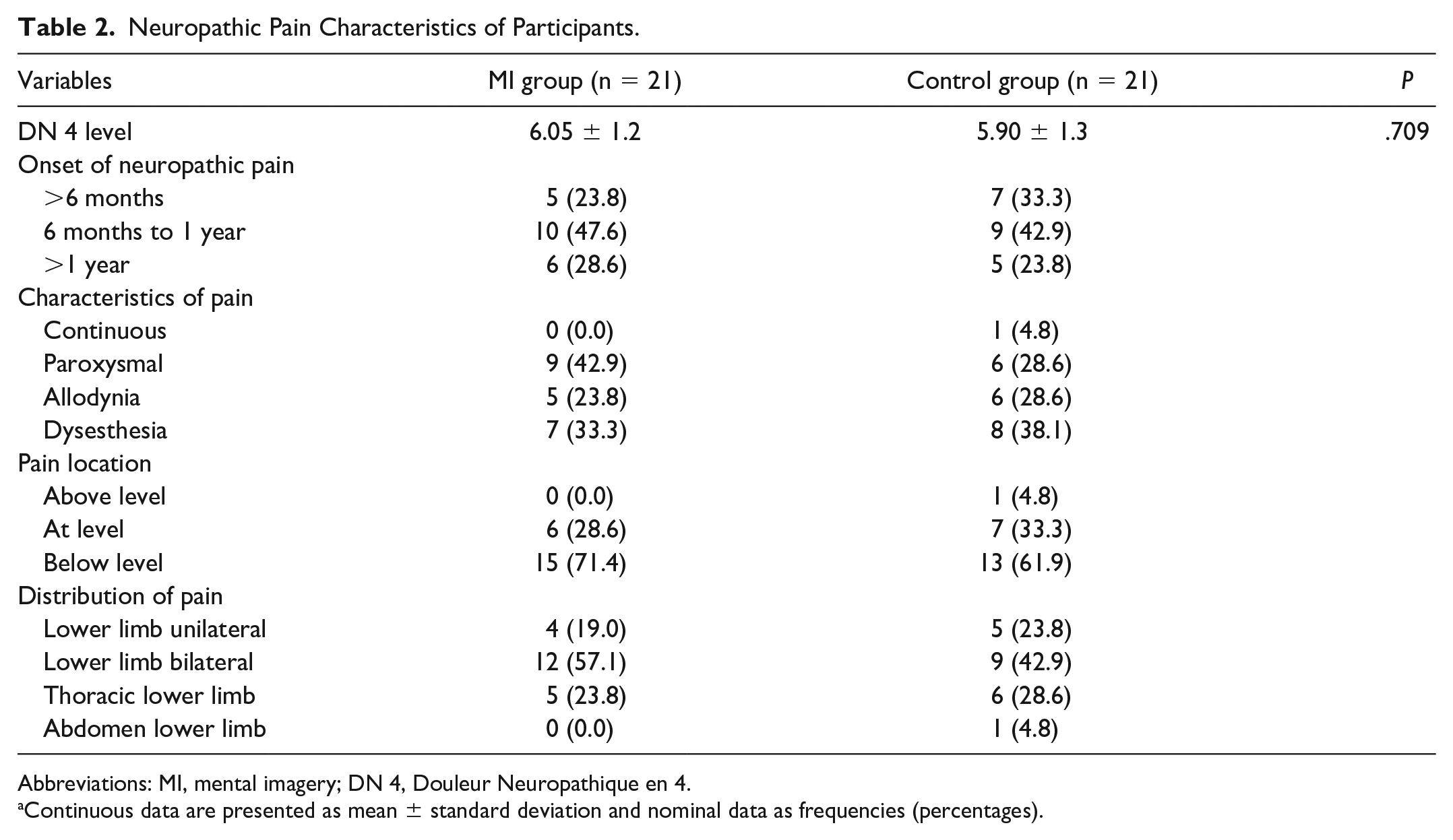
Abbreviations: MI, mental imagery; DN 4, Douleur Neuropathique en 4.
Continuous data are presented as mean ± standard deviation and nominal data as frequencies (percentages).
Comparison of Pain Scores of Numeric Rating Scale and Visual Analogue Scale in the MI and CONT Groups
After 4 weeks of training; significant reduction was seen in pain scores of numeric rating scale (
Comparison of Mean Change Scores of NRS, VAS, and BPI and Its Components in the MI Group (n = 21) and the Control Group (n = 21).
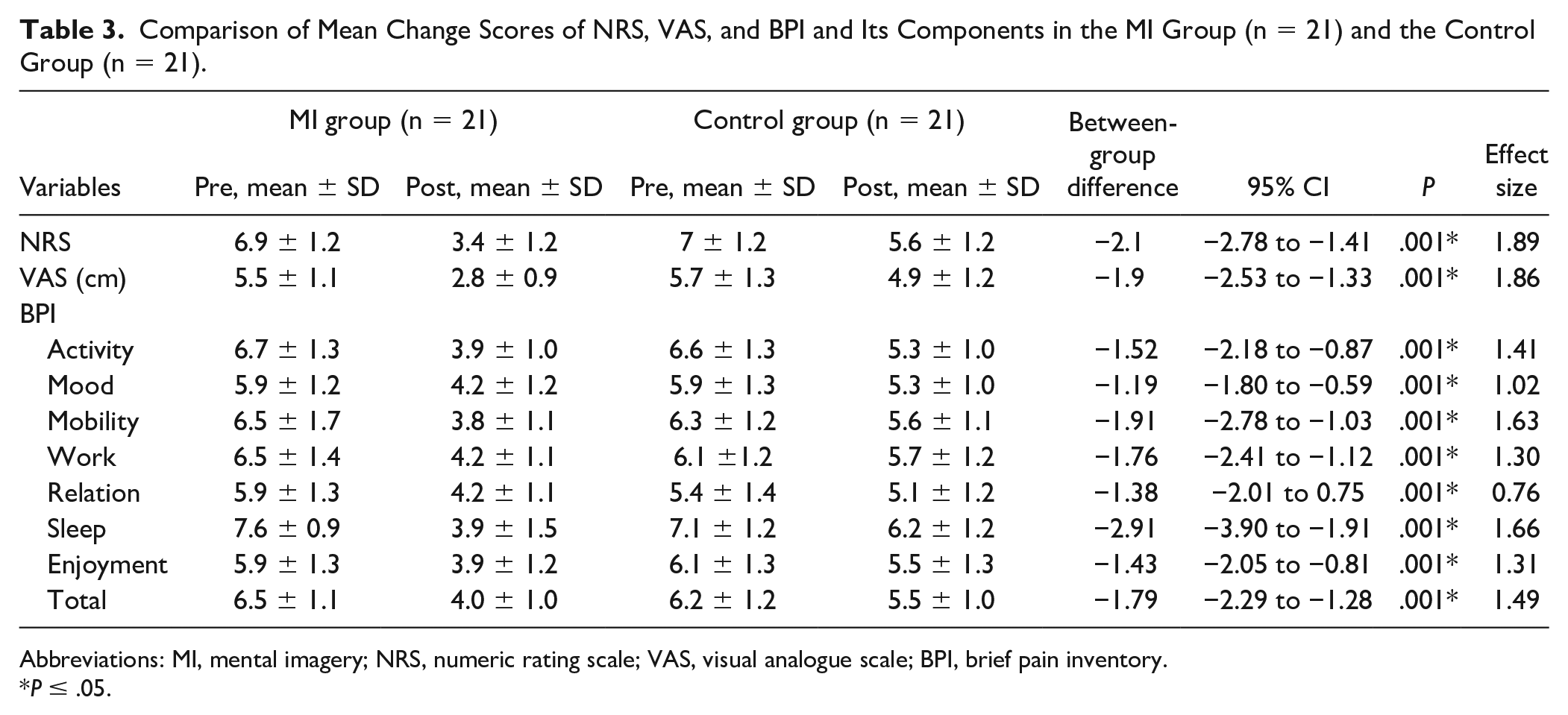
Abbreviations: MI, mental imagery; NRS, numeric rating scale; VAS, visual analogue scale; BPI, brief pain inventory.
Comparison of Neuropathic Pain Symptom Inventory and Its Subtypes in the MI and CONT Groups
The results show that there were significant reductions in the mean [95% CI] total scores of neuropathic pain symptom inventory in the MI group as compared with the CONT group (−4.52 [CI −5.86 to −3.18];
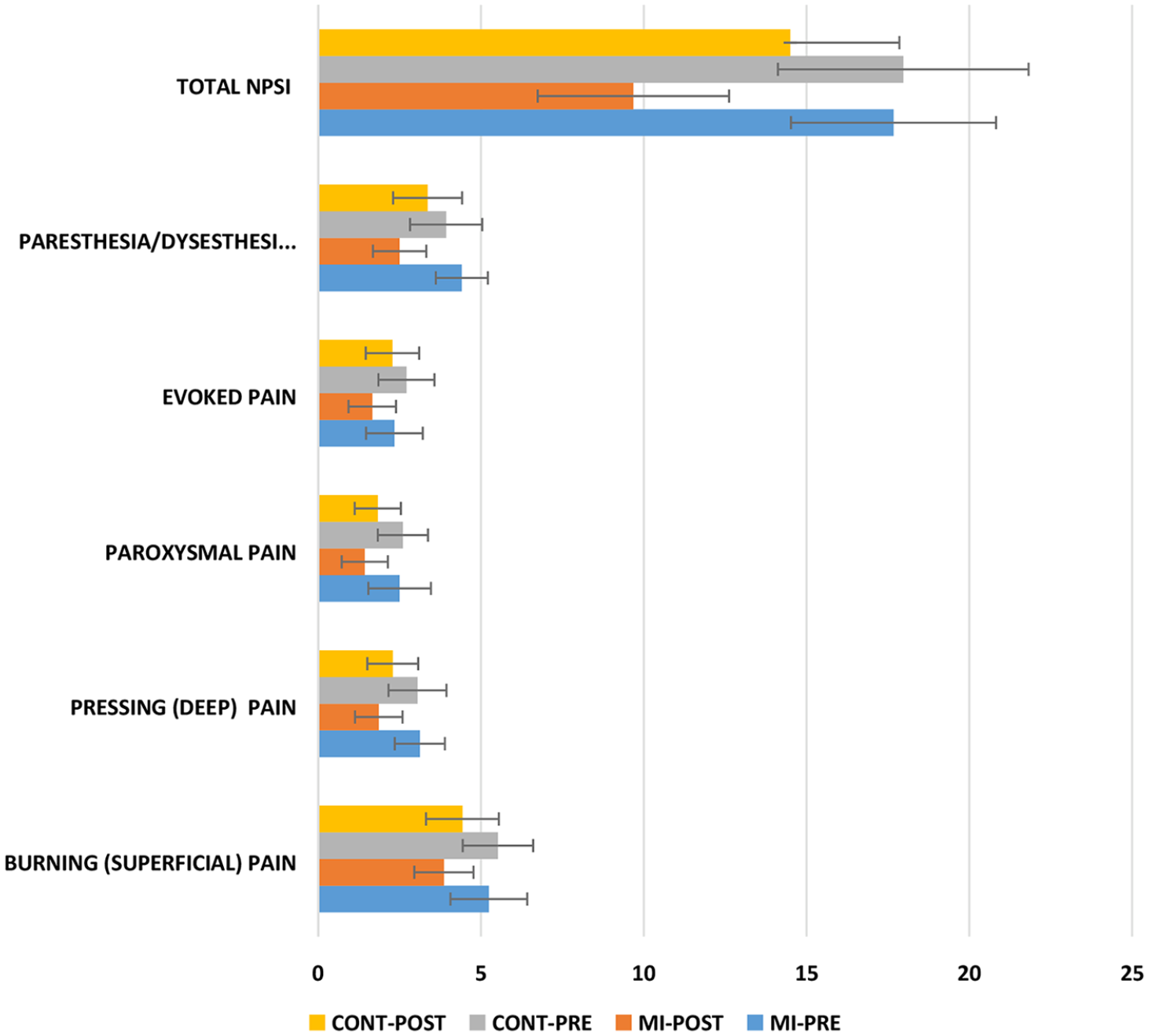
Frequency distribution for scores of Neuropathic Pain Symptom Inventory (NPSI) and its subtypes for the mental imagery (MI) and control (CONT) groups.
Comparison of Brief Pain Inventory Interference Scale and Its Components in the MI and CONT Groups
Significant reduction in mean [95% CI] scores of total interference scores (−1.79 [CI −2.29 to −1.28];
Comparison of Number of Patients’ Ratings of Patients’ Global Impression of Change Scale in the MI and CONT Groups
At the end of treatment, most individuals in MI group with SCI having neuropathic pain reported that pain had improved on patients’ global impression of change. The results are presented in Figure 3.
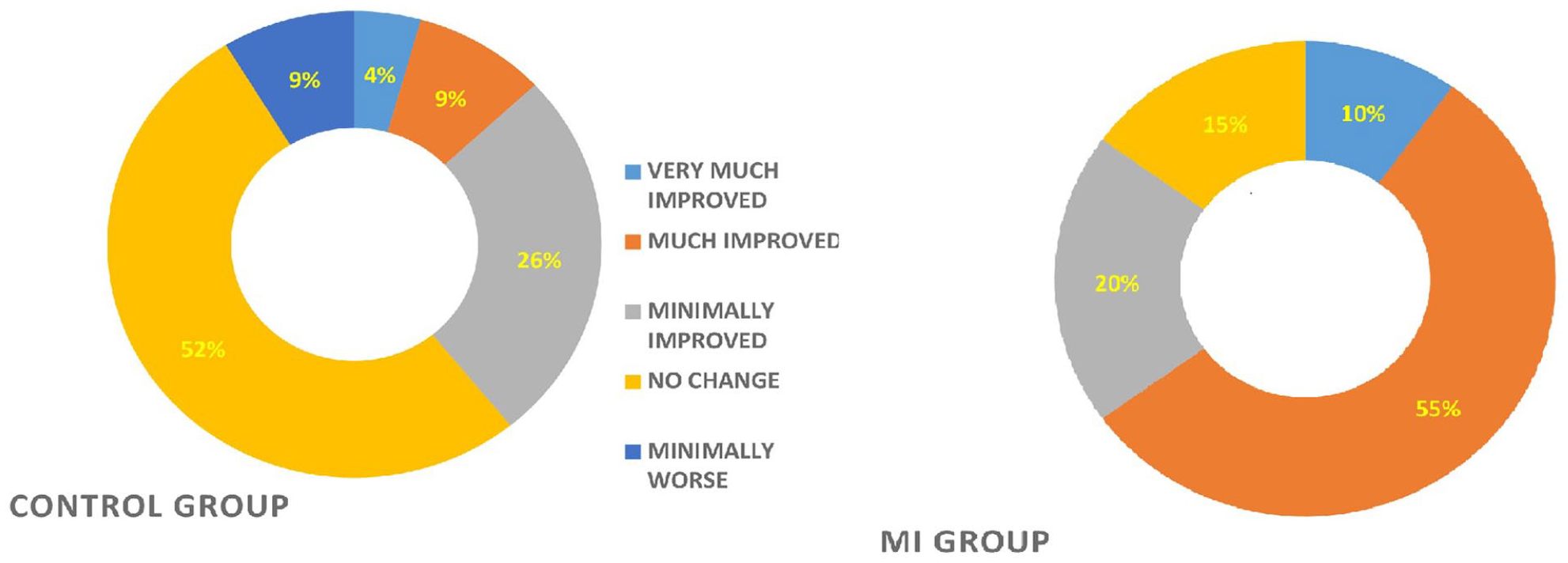
Patient Global Impression of Change (PGIC) ratings of study participants of the mental imagery (MI) group (n = 21) and the control (CONT) group (n = 21). Values are expressed in percentages (%).
Discussion
The results of this study show that there was significant reduction in the scores of numeric rating scale and visual analogue scale in the MI group as compared with the control group. Participants in the MI group demonstrated a significant decrease in the scores of pressing (deep) spontaneous pain, evoked pain, dysesthesias/ paresthesia, and total scores of the Neuropathic Pain Symptom Inventory. However, there were no between-group differences in burning (superficial) spontaneous pain and paroxysmal pain scores. There was also a significant decline in the scores of Brief Pain Inventory–pain interference scale and its subtypes with MI training. This is one of the few studies in which MI protocol had highly significant effect on chronic neuropathic pain.
Chronic pain is often accompanied by altered mental representation of the affected body part, 40 probably due to the additive effect of distorted afferent signal processing 41 and the consequently weak reliance on somatosensory input from the affected body part. 42 This could explain the (impaired) status of pain perception before the imagery training found in the present study. Similarly, the improvements after the imagery training could be interpreted as signs of the importance of restoring somatosensory perception 43 and sensory reweighting of the signals coming from the affected body part. 44
Effect of MI Protocol on Pain Intensity
In this study, pain intensity was reduced after MI training in SCI participants. SCI alters mental representation of the body resulting in distortion of body representation. 45 Persons with SCI are reported to have corporeal illusions such as body loss, body part misperception, disownership-like feelings and somatoparaphrenia-like sensations. 46 Research findings from fMRI have shown remapping of the somatosensory cortex particularly primary motor cortex in central neuropathic pain in SCI that is proportional to pain intensity. 16 Imagery techniques such as visual illusion can alter cortical excitability. 19 MI is now considered as a potent cognitive tool affecting modulation of pain matrices. In a study by Scandola et al, 47 it was seen that mental rotation of foot images had improved significantly in individuals with SCI receiving physiotherapy. This clearly suggests the cognitive benefits associated with such interventions in people with SCI. 47 We believe that participation in laterality training may have enhanced body representations in these patients, which in turn augmented their participation in GI sessions. Post laterality training, participants could focus on each part of the body in a better manner 24 and gradually eliminate pain and other sensations. Another reason behind reduced pain intensity can be explained on the basis of pain gate theory, which states that if the route of the pain carrying pathway is blocked by pleasant stimulus the perception of pain is reduced. 48 Techniques such as GI use the power of imagination to help break the pain pathway resulting in reduction in pain threshold and perception over time. It is also believed that modification of somatosensory pain memories may be useful in reducing neuropathic pain. GI is also believed to cause endorphin release, 49 which in turn decreases the pain sensitivity.
Effect of MI Protocol on Neuropathic Pain–Related Symptoms
There was also a reduction in the mean change score on the Neuropathic Pain Symptom Inventory in MI group as compared with the CONT group. Paresthesia and dysesthesia were the most common symptoms reported by participants in both groups. The cause of these abnormal sensations associated with neuropathic pain is central sensitization, which occurs as a result of chronic pain. 50 These abnormal sensations occur even in cases of complete injuries. 2 Mind-body interventions such as GI use multisensory stimulation and creates a conscious experience of the symptoms experienced by the patients. 51 The reduction of scores of Neuropathic Pain Symptom Inventory may have resulted due to increased awareness of symptoms during both laterality training and conscious elimination of these symptoms during imagery session. However, the reduction in the scores of burning or paroxysmal pain was not statistically significant. The causes maybe related to patient characteristics (such as type of pain, chronicity, level of injury among others) and needs to be explored further.
Effect of MI on Pain Interference Scores
There was a decline in pain interference scores in the MI group as compared with the CONT group. Participants reported better sleep, better mood, and increased participation in daily activities. Significant reductions in pain scores may have been responsible for substantially lower brief pain inventory scores. According to a study by Jensen et al, 52 there is a strong associations between the average intensity of pain and interference in activities of daily living. Some of our participants expressed that after the session they felt comfortable throughout their daily routine tasks.
Effect of MI on Patients’ Global Impression of Change Ratings
According to the guidelines for conducting clinical trials for pain, use of patients’ global impression of change is recommended as it comprises of only one question regarding patients’ perception of improvement in their condition since the inception of treatment. 36 In the MI group 55% of the patients reported that pain had much improved, 10% of patients reported that their pain had improved maximally and 15% reported no change in pain after treatment. However, in the CONT group, majority of the participants (52%) reported no change in the pain scores and 9%reported that their pain had become worse.
CONT Group
There were changes seen in outcome measures in CONT group also. These could be due to redirection of attention away from pain with the use distraction strategies like addition of numbers 53 or listening to music. Distraction techniques are commonly used in pain studies particularly in control conditions. However, the results of a meta-analysis suggest that its use should not be encouraged among chronic pain patients. 54 Another study suggested that the use of distraction might result in increased pain on disengaging from distraction. 55
The clinical implication of this study is that it offers mental imagery as a potentially useful rehabilitation technique for neuropathic pain in SCI. It further corroborates the relationship between mental imagery and pain. This in turn, would improve our understanding on pain perception, MI, and their relationships with corticospinal excitability. 56
Our study had some limitations. SCI is a multi-faceted condition with a number of associated conditions so differences in variables such as the level of injury, chronicity and degree of impairments can influence the results. 57 Furthermore, for most of the participants’ chronicity of neuropathic pain was between 6 months to 1 year so future studies can be done to assess the effect of the protocol on suffering from pain for many years. Another limitation was that self-assessment measures were used to assess pain. With regard to outcome measures for any pain treatment; patient’s perception of pain and his expectations from treatment have a significant role. 58 However, pain is a subjective phenomenon, so scales were used as outcome measures for measuring pain. Furthermore, we followed the IMMPACT recommendations for clinical trials for pain to assess all domains related to pain. Additionally, pain medications taken by the patients may have affected the pain scores though the participants did not change their medications or dosage throughout the study. Last, we could not do a follow up of the intervention, as this was not a part of the study. Future studies may be conducted to assess the carryover effect of the intervention.
However, this is one the few protocols that has been developed specifically for individuals with SCI. Despite the limitations, the protocol is the first to show a highly significant effect on neuropathic pain and has remarkable rehabilitative potential to treat patients having neuropathic pain with great efficiency and minimal side effects. Furthermore, due to its simplicity and affordability, it can be recommended as a home exercise program. Future research can be carried out to assess its effectiveness in neuropathic pain in multiple sclerosis, stroke, and other conditions. Studies can also be conducted to see the carryover effect of this treatment. The study may contribute to new evidence-based protocol for SCI participants with complaints of neuropathic pain.
Supplemental Material
PARTICIPANTS_DATA_2.1 – Supplemental material for Mental Imagery as a Rehabilitative Therapy for Neuropathic Pain in People With Spinal Cord Injury: A Randomized Controlled Trial
Supplemental material, PARTICIPANTS_DATA_2.1 for Mental Imagery as a Rehabilitative Therapy for Neuropathic Pain in People With Spinal Cord Injury: A Randomized Controlled Trial by Jaskirat Kaur, Shampa Ghosh, Asish Kumar Sahani and Jitendra Kumar Sinha in Neurorehabilitation and Neural Repair
Supplemental Material
supplementary_GUIDED_IMAGERY_SCRIPT_sample_only – Supplemental material for Mental Imagery as a Rehabilitative Therapy for Neuropathic Pain in People With Spinal Cord Injury: A Randomized Controlled Trial
Supplemental material, supplementary_GUIDED_IMAGERY_SCRIPT_sample_only for Mental Imagery as a Rehabilitative Therapy for Neuropathic Pain in People With Spinal Cord Injury: A Randomized Controlled Trial by Jaskirat Kaur, Shampa Ghosh, Asish Kumar Sahani and Jitendra Kumar Sinha in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We wish to thank Siddharth Hans (GaitON) and Jaideep Singh for their technical support in development of software for laterality training. The authors would also like to thank all the study participants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
