Abstract
Background. Neurorehabilitation interventions to improve lower limb function and neuropathic pain have had limited success in people with chronic, incomplete spinal cord injury (iSCI). Objective. We hypothesized that intense virtual reality (VR)–augmented training of observed and executed leg movements would improve limb function and neuropathic pain. Methods. Patients used a VR system with a first-person view of virtual lower limbs, controlled via movement sensors fitted to the patient’s own shoes. Four tasks were used to deliver intensive training of individual muscles (tibialis anterior, quadriceps, leg ad-/abductors). The tasks engaged motivation through feedback of task success. Fourteen chronic iSCI patients were treated over 4 weeks in 16 to 20 sessions of 45 minutes. Outcome measures were 10 Meter Walking Test, Berg Balance Scale, Lower Extremity Motor Score, Spinal Cord Independence Measure, Locomotion and Neuropathic Pain Scale (NPS), obtained at the start and at 4 to 6 weeks before intervention. Results. In addition to positive changes reported by the patients (Patients’ Global Impression of Change), measures of walking capacity, balance, and strength revealed improvements in lower limb function. Intensity and unpleasantness of neuropathic pain in half of the affected participants were reduced on the NPS test. Overall findings remained stable 12 to 16 weeks after termination of the training. Conclusions. In a pretest/posttest, uncontrolled design, VR-augmented training was associated with improvements in motor function and neuropathic pain in persons with chronic iSCI, several of which reached the level of a minimal clinically important change. A controlled trial is needed to compare this intervention to active training alone or in combination.
Keywords
Introduction
In about 50% of patients, motor and sensory function is preserved below the lesion level resulting in an incomplete spinal cord injury (iSCI). 1 However, iSCI often leads to long-lasting lower limb motor dysfunction associated with a range of physical and psychosocial problems, including pain, which are challenging to treat. Motor dysfunction and associated neuropathic pain may share some underlying cortical mechanisms that are currently treated very differently. The present investigation tests the use of virtual reality (VR)–augmented movement training programs to provide interactive, multimodal sensory stimuli and biofeedback using representations of the patient’s lower limbs. VR-augmented neurorehabilitation exploits the idea of inducing activation in action observation, motor imagery, and processing systems, which in turn activate downstream cortical areas involved in movement execution, 2 a process that can be increased in intensity when the movement is toward an object.3-5 This system can still be activated in chronic complete SCI patients using motor imagery, attempted movements and observation of foot movements.6,7
In addition to motor impairments that affect walking in persons with American Spinal Cord Injury Association Impairment Scale (AIS) C and D, neuropathic pain affects 40% to 60% of patients.8,9 Long-term prognosis for neuropathic pain following SCI is poor 10 and the pain often continues or worsens over time. 11 The “virtual walking” illusion studies in SCI subjects by Moseley 12 and Soler et al 13 showed that when subjects see their upper body in a mirror above a video screen that plays a movie of walking legs, they reported analgesic effects. It has been demonstrated in several neurological disorders that perturbations of the somatosensory system associated with central pain can be reversed or modulated by employing motor imagery and related task execution combined with visual illusions.
Based on these findings, we hypothesized that iSCI patients using VR-augmented movement tasks should show improvements in motor function and reduction in neuropathic pain that outlasts the training sessions and transfers into daily life.
Material and Methods
Patients
Incomplete SCI outpatients (age 28-71 years) from the University Hospital Balgrist (Zurich, Switzerland) were included in the study between August 2010 and March 2012 (Table 1). Inclusion criteria were as follows: clinically incomplete, chronic SCI (time since injury >1 year), motor level of lesion below C4, able to sit in a chair without assistance and support systems (eg, securing belt), preserved motor functions below the level of lesion corresponding to AIS C or D at time of inclusion (C = sensorimotor incomplete, with an average strength of the muscles below the level of lesion <3; D = sensorimotor incomplete, but with average muscle strength ≥3). 14
Clinical and Demographical Characteristics of Patients With Spinal Cord Injury.
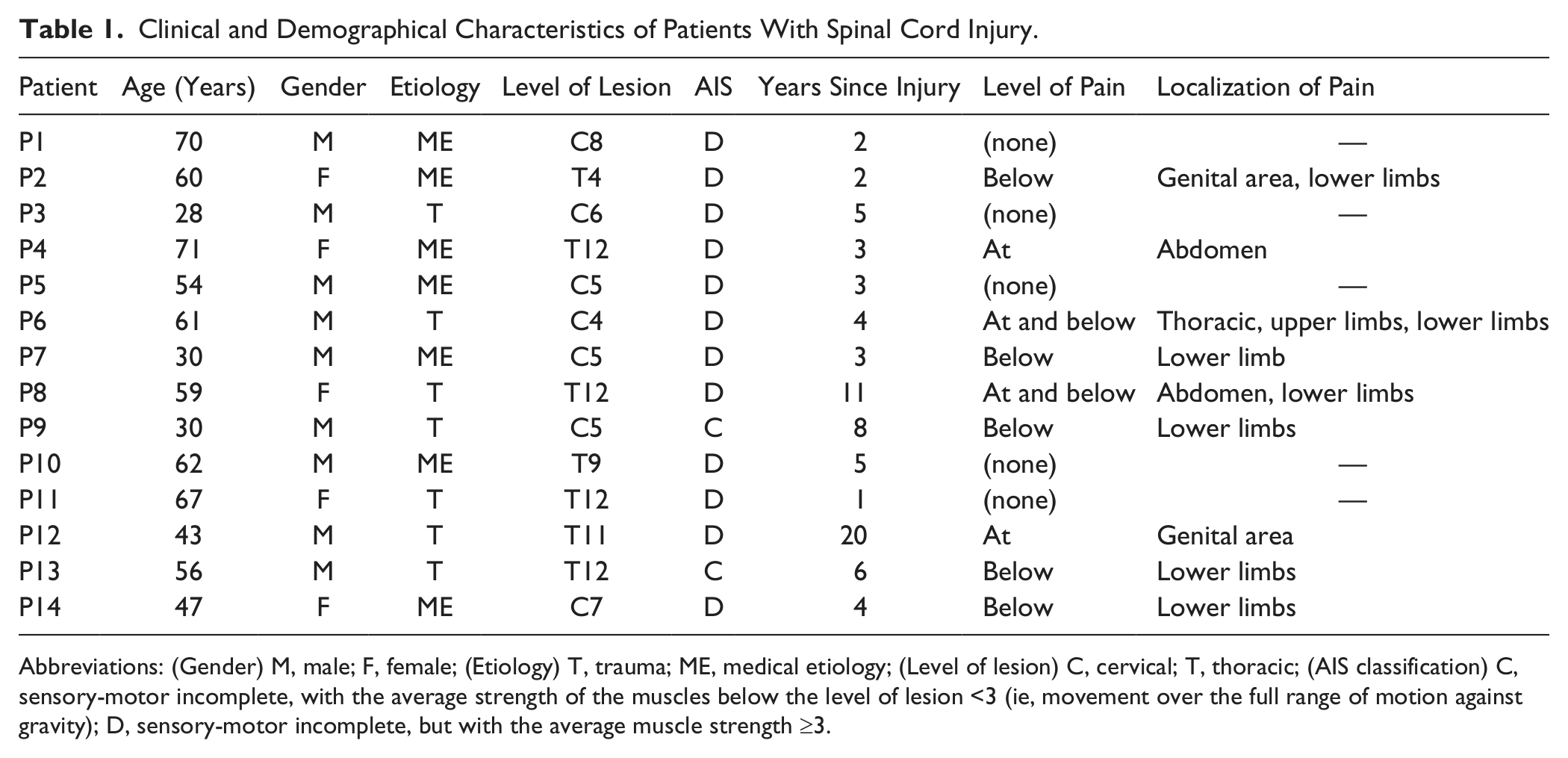
Abbreviations: (Gender) M, male; F, female; (Etiology) T, trauma; ME, medical etiology; (Level of lesion) C, cervical; T, thoracic; (AIS classification) C, sensory-motor incomplete, with the average strength of the muscles below the level of lesion <3 (ie, movement over the full range of motion against gravity); D, sensory-motor incomplete, but with the average muscle strength ≥3.
Nine patients were experiencing neuropathic pain, as assessed by a structured interview. Information was collected on pain intensity, pain descriptors, sensory deficits, and allodynia or hyperalgesia within the pain distribution. 15 Based on the most recent taxonomy for pain after SCI neuropathic pain was classified into at-level and below-level SCI (neuropathic) pain. 15 At-level pain is defined as pain located within the dermatome and 3 dermatomes below the lesion level, and not in any lower dermatomes, unless the pain is thought to be caused by damage to the cauda equine. Below-level pain is defined as pain present more than 3 dermatomes below the lesion level, and the lesion or disease must affect the spinal cord and that the pain is believed to arise as a result of this damage.
Exclusion criteria were psychiatric or other neurological disorders, head injuries causing cognitive or visual impairment, spasticity limiting performance of lower limb movements, and medication influencing ability to attend to therapy for 45 minutes. Furthermore, patients with depressive symptoms (score >14 on the Beck Depression Inventory) were excluded. 16
All patients were aware of the purpose of the study and informed consent was obtained from them. The experimental protocol was in accordance with the Declaration of Helsinki and performed with the approval of the local ethics committee.
Virtual Reality–Augmented Training
The training study used a VR-augmented therapy system for lower limbs combining action observation and execution (Figure 1).

Overview of the virtual reality (VR) training setup (left) and the different interactions for training the various lower limb muscles and functions (right). Ankle flexion: Footbag—juggling a ball (1), Hamster Splash—launching hamsters into a swimming pool (2). Knee flexion: Star Kick—kicking balls toward stars (3). Leg ad-/abductors: Planet Drive—(avoiding) touching oncoming cars (4).
The system and the motivating training tasks have been described in Villiger et al. 17 The system used sensors based on a hardware architecture originally designed for data gloves used in an upper-limb rehabilitation system, 2 which was subsequently commercialized (YouGrabber, YouRehab AG, Zurich, Switzerland). The main hardware modifications were the addition of 2 extra 3-degrees of freedom accelerometer sensor nodes (same as the existing nodes, for a total of 4 nodes) and the removal of the finger bend sensors. The 4 sensor nodes were mounted on the back of each ankle and lower calf. A short cable connected each foot-to-calf sensor node pair, and longer cables connected each calf sensor node to a main relay board, which was connected in turn to a USB cable. The ankle sensor nodes were held in place using Velcro attached to modified elastic rubber overshoes worn over the patient’s own shoes, while the calf sensor nodes were attached to adjustable Velcro straps around each leg. Data from the sensors was transmitted over the USB connection to custom software written using the Unity 3-dimensional (3D) game engine (Unity Technologies, San Francisco, CA). The angles of the patient’s real lower leg and foot were mapped in real time to a 3D articulated virtual lower limb model and presented in a first-person perspective on a large monitor (132 cm diagonal). If desired for more training variety, the therapist could change the mapping to have the right real leg control the left virtual leg, and vice versa. Clinically relevant interactions for training foot and leg movements in a sitting or standing position were developed in cooperation with therapists. These were as follows:
Footbag: A simple exercise in which the patient juggles a ball between the left and right foot, using dorsal ankle flexion movements (approximately 35/min per leg depending on patient ability), a necessary exercise to prevent foot dragging. The trajectory of the ball is preset so that it always moves correctly through the air between the left and right feet. A juggle is registered when the foot moved through a fixed threshold angle of 3° above the horizontal. The juggling settings can be set by the therapist to either alternate between the left and right feet, or to randomly go to either foot (thus requiring more attention).
Hamster Splash: Hamsters run up to the patient’s toes. The patient’s task is to perform a dorsal flexion of the ankle (approximately 15/min per leg) to launch each hamster into a swimming pool. Launching the hamsters with a faster ankle movement leads to higher scores and triggers more elaborate hamster movements (somersaults, swimming patterns). The therapist can specify “heavy hamsters,” which doubles the required launch speeds, thus increasing difficulty.
Star Kick: The patient performs a knee extension (approximately 12/min per leg) by kicking a ball toward the displayed stars. For every hit, the patient receives a score reward. This exercise required the patient to tilt the lower leg and thus trained knee extension without requiring direct measurement of knee angle.
Planet Drive: Cars are moving on a highway toward the virtual feet. The patient’s task is to avoid touching the cars by tilting the lower leg sideways (approximately 4/min per leg). Note that because the sensors could not measure translation, the patient moved the legs sideways by moving the foot outward while keeping the knee in a more central position—an unusual movement when played in the sitting position, where it would be more natural to abduct the leg at the knee.
During a typical training session, each interaction was presented 3 times for 2 minutes each. Within a training session, a typical patient executed around 300 repetitions of ankle movements (Footbag, Hamster Splash) and 75 knee movements (Star Kick) with each leg. Planet Drive was less emphasized because of its low intensity and the less physiologically suitable movements that it required.
In each training application, scoring was based on the number of successfully completed movements. The tasks were always presented in the same order (Footbag, Planet Drive, Star Kick, Hamster Splash) during a training session, providing alternation of muscle groups to prevent fatigue. Task difficulty was adjusted by the therapist when a subject scored either the same and/or higher number of repetitions in 3 successive sessions (increase in difficulty) or the number of repetition was less in 3 successive sessions (decrease in difficulty). An increase in task difficulty resulted in higher speed (ie, in more repetitions) for Footbag und Hamster Splash, whereas for Planet Drive the number of cars was increased. For Star Kick there was only one level of difficulty. Two therapists were involved in the training and assessment. Therapist MV conducted the clinical assessments, whereas therapist DB performed the Vicon analysis. Both therapists participated equally in the training sessions.
Clinical Study Design
The iSCI patients were trained with the 4 VR tasks during a period of 4 weeks in 16 to 20 sessions of 45 minutes (4-5 per week). At entry to the study, general patient information was collected—age, gender, height, weight, etiology, and level of lesion, AIS, time since injury, medication, pain presence, pain intensity, and pain location. After each training session, patients rated their enjoyment, motivation and attention level on an 11-point Numeric Rating Scale (NRS) from 0 (worst) to 10 (best), as well as their mood and pain level before and after each session.
Primary Outcomes
To control for possible transfer effects, primary outcomes for both motor functions and pain were assessed at 4 different time points: Pre-baseline assessments 4 to 6 weeks before treatment, baseline assessments immediately before starting the treatment, post-assessments after completing the training program, and follow-up assessments 12 to 16 weeks after treatment (Table 2). Furthermore, we added the minimal detectable change (MDC—a statistical estimate of the smallest amount of change that can be detected by a measure that corresponds to a noticeable change in ability) and the minimal clinically important difference (MCID—the smallest amount of change in an outcome that might be considered important by the patient or clinician) when established for the primary outcomes in SCI patients. Published estimates for the MDC and MCID are available for a number of different outcome measures and indications, including SCI. The primary outcome measures were as follows:
Assessment Time Points and Primary Outcome Measures. a
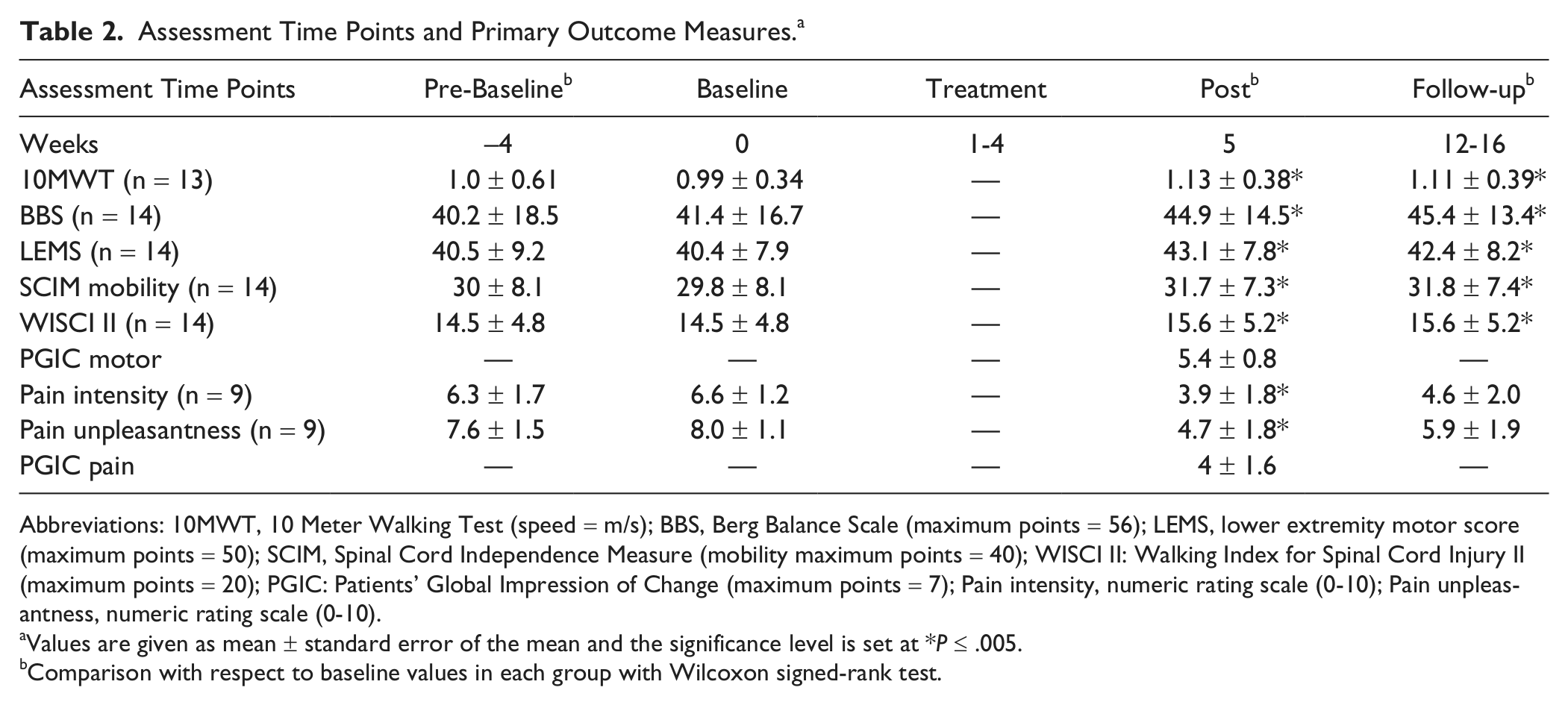
Abbreviations: 10MWT, 10 Meter Walking Test (speed = m/s); BBS, Berg Balance Scale (maximum points = 56); LEMS, lower extremity motor score (maximum points = 50); SCIM, Spinal Cord Independence Measure (mobility maximum points = 40); WISCI II: Walking Index for Spinal Cord Injury II (maximum points = 20); PGIC: Patients’ Global Impression of Change (maximum points = 7); Pain intensity, numeric rating scale (0-10); Pain unpleasantness, numeric rating scale (0-10).
Values are given as mean ± standard error of the mean and the significance level is set at *P ≤ .005.
Comparison with respect to baseline values in each group with Wilcoxon signed-rank test.
Walking speed: 10 Meter Walking Test (10MWT) assessing gait speed.18,19 The MDC for the 10MWT is 0.13 m/s 20 and the MCID is >0.05 m/s. 21
Balance: Berg Balance Scale (BBS), which assesses balance during functional activities with 14 balance items from 0 (no balance) to 4 (good balance). 22 No estimates of the MDC or MCID are available for SCI.
Muscle strength: Lower extremity motor score from 0 (complete paralysis) to 50 (normal strength). 14 No estimates of the MDC or MCID are available for SCI.
Mobility: The transfer, indoors, and outdoors mobility parts of the Spinal Cord Independence Measure (SCIM—hereinafter referred to as “SCIM mobility”) from 0 (no mobility) to 40 (normal mobility) were assessed together with the Walking Index for Spinal Cord Injury II (WISCI II) from 0 (unable to walk) to 20 (able to walk without assistive devices).23,24 For the SCIM mobility, no estimates of the MDC and MCID are available for SCI. WISCI II has no estimates of the MCID but MDC: A change of one WISCI level can be considered a real difference in a clinical context. 25
Neuropathic pain: The items pain “intensity” and “unpleasantness” were measured on an NRS from 0 (no pain) to 10 (worst pain imaginable) from the Neuropathic Pain Scale (NPS). 26 The MDC for lower back pain is 2 points, 27 whereas the MCID for the NRS has been reported for chronic pain as 1.74 points or a reduction of 27.9%. 28
Self-reported relieving effect: At the end of the treatment (post-assessment), the Patients’ Global Impression of Change (PGIC) was evaluated according to the method of Farrar et al 28 for both motor and pain with no change (score 0-1), minimally improved (score 2-3), much improved (score 4-5), and very much improved (score 6-7). The patients answered the following question: “Since beginning treatment at this program, how would you describe the change (if any) in activity limitations, symptoms, emotions, and overall quality of life related to your condition?”
Locomotion: To control for transfer effects, locomotion was assessed at baseline and at post-assessment. Gait patterns were recorded and quantified with a 3D motion analysis system (Vicon MX, Oxford, UK).
Data Analysis
For statistical analysis (PASW 19.0 software, SPSS, Chicago, IL), variables were represented as mean ± standard error of the mean (SEM). The differences between the time points (within-group differences) were analyzed using Friedman’s test and pairwise comparisons were performed with the Wilcoxon signed-rank test to assess significant differences in the group for 2 time points. The significance of percentage changes at each assessment time point compared with the baseline was also performed by pairwise comparisons. To address the problem of multiple comparisons, Bonferroni correction was used for the 9 primary outcomes and the significance level was set at P ≤ .005. In addition, Spearman rank correlations were computed to reveal relationships between demographic characteristics such as age and years since injury and primary outcomes.
Locomotion
To control for transfer effects, locomotion was assessed at baseline and at post-assessment. Gait patterns were recorded and quantified with a 3D motion analysis system (Vicon MX, Oxford, UK). Patients walked with their preferred speed on a treadmill and a sufficient number of gait cycles were recorded. Statistical analysis for locomotion was performed individually. Gait parameters obtained from 15 gait cycles were presented as mean ± standard deviation (SD). The Plug-in gait model was used to calculate angular displacements of hip, knee, and ankle joints. 29 All signals were analyzed off-line using Matlab (Matlab 7.10_R2010a, Mathworks Inc, Natick, MA), which calculated the kinematic parameters. For each patient, the 15 consecutive walking cycles were normalized to the actual gait cycle duration (heel strike to next heel strike of same foot = 100%). The time-normalized gait cycles were then used to average the mean angular displacement profiles of the lower limb joints. To check whether the data were comparable to that of healthy subjects, a control group was incorporated. Baseline and post-assessment angular values at specific points of the gait cycle were compared using t tests.
Results
Patient Characteristics and Motivational Factors
All 14 patients completed the training (mean age 52.7 ± 14.9 years). The clinical characteristics are summarized in Table 1. Two of the patients were AIS C and 12 AIS D. Nine of the patients reported neuropathic pain. The patients were quickly familiarized with the VR system. The patients reported high enjoyment (9.6 ± 0.7), motivation (8.8 ± 0.3), and attention (8.7 ± 1.2) levels during training. Seven of them reported a better mood after the training in more than 50% of the sessions. One patient (P8) mentioned a transient occurrence of musculoskeletal pain in the right leg because of its increased use during the sessions. Four patients reported less pain sensation in at least 50% of all training sessions, combined with sensations of lightness or positive tingling in the legs.
Primary Outcomes
Table 2 summarizes the primary outcome measures across the various assessment time points (pre-baseline, baseline, post-assessment, and follow-up). No significant differences between pre-baseline and baseline measurement were found, indicating stability of all the measured variables. With respect to the assessment of motor functions, only one patient was unable to perform the walking tests (P9). For the other patients, walking speed was increased in comparison with the baseline assessment for both post- and follow-up assessments (post P ≤ .001, follow-up P ≤ .003). At post-assessment, 8 out of 13 patients showed increased speed that met or exceeded the MDC, and all patients met the limits for the MCID. At the follow-up assessment, 7 out of 13 patients reached the MDC and 11 out of 13 reached the MCID. Significant increases were found for all patients in BBS score (P ≤ .001), muscle strength (P ≤ .001), and mobility (SCIM mobility, P ≤ .001; WISCI II, P ≤ .004) at both post- and follow-up assessments. At the post- and follow-up assessments, 8 out of 14 patients reached the MDC of WISCI II. With respect to pain, in the post- and follow-up assessments, decreases were found in intensity (P ≤ .004, P ≤ .031) and unpleasantness (P ≤ .004, P ≤ .016).
In the neuropathic pain group, at post-treatment assessments 6 out of 9 patients had improved scores that reached the MDC, and 5 out of 9 patients reached the MCID. Five out of 9 patients retained both limits of clinical improvement in the follow-up.
The percentage changes after treatment compared to baseline are shown in Figure 2 for the post- and follow-up assessments. Increases were found in walking speed with an increase of 11.9% at post-assessment persisting at 9.8% at follow-up (P ≤ .001 in both cases). Balance scores improved by 16.5% at post-assessment (P ≤ .021), which persisted at 13% at follow-up (P ≤ .002). Muscle strength increased by 7.3% and 6.6% (P ≤ .002 in both cases) at the post- and follow-up assessments, respectively. Mobility as measured by the SCIM mobility improved by 8.5% (P ≤ .002) at post-assessment and remained at 9.6% at follow-up (P ≤ .004), whereas the WISCI II improved by 12% (P ≤ .004) in both measurement points. NRS decreases were also found in pain intensity: 38.9% at post-assessment and 36.3% at follow-up (P ≤ .008 in both cases). Pain unpleasantness decreased by 37.9% at post-assessment and 29.6% at follow-up (P ≤ .016 in both cases). No significant relations were found between age and years since injury and primary outcomes in at post-assessment and follow-up.
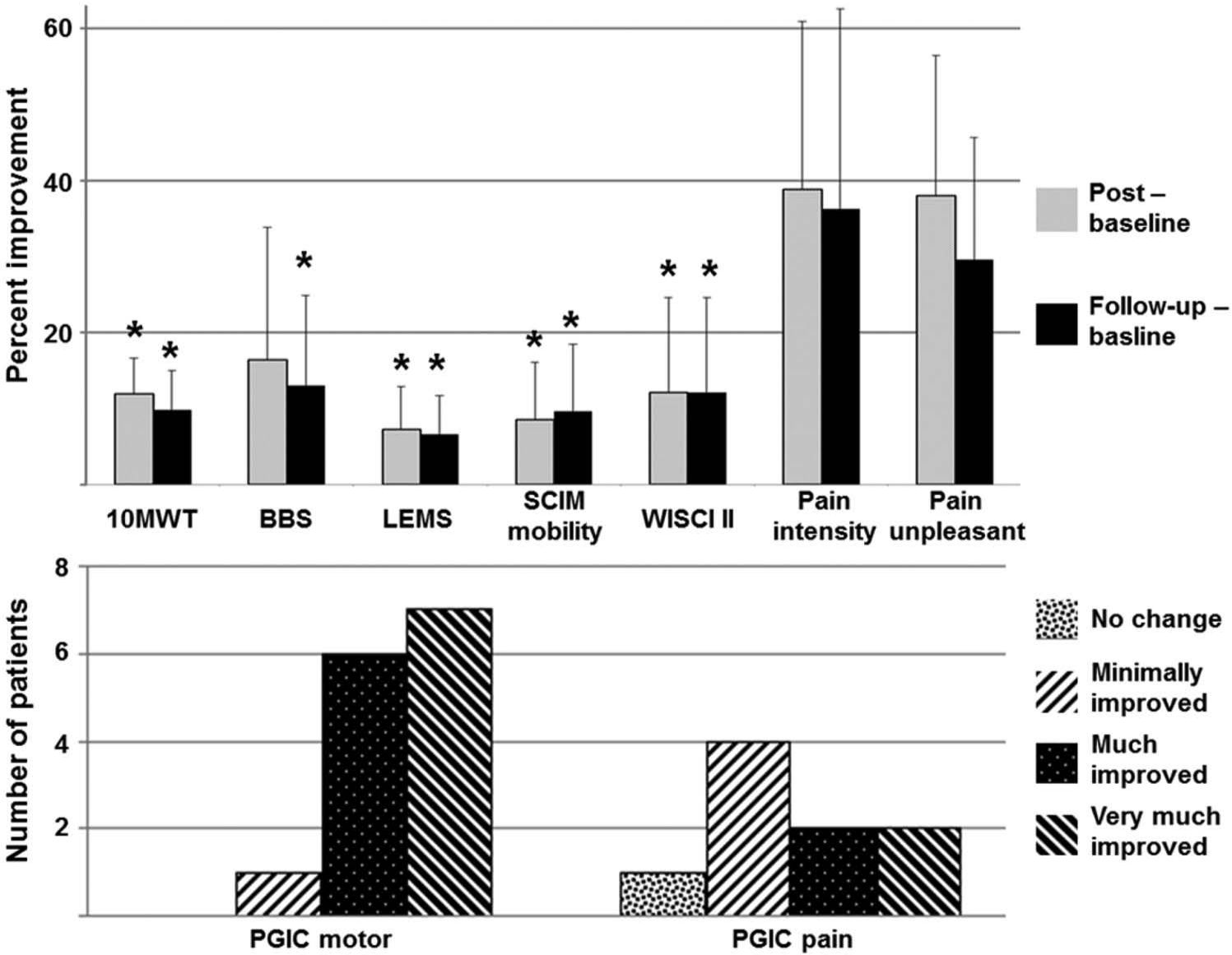
(Top) Percentage changes in primary outcome measures in post- and follow-up assessments versus baseline. Data are shown as mean ± standard error of the mean and the significance level is set at *P ≤ .005. (Bottom) Patients’ Global Impression of Change (PGIC) of motor function and pain after the last day of intervention, rated on a 7-point scale. For explanation of abbreviations, see Table 2.
All patients rated the motor and pain relieving effects of the treatment using the PGIC after the last day of treatment (Figure 2). For the PGIC motor, 13 out of 14 patients rated motor function as markedly improved (much improved or very much improved). For PGIC pain, 4 of the 9 patients rated their pain as markedly reduced, 4 as minimally and only 1 reported no change.
Locomotion
Seven of the patients (mean age 48.3 ± 14.7 years, range 28-62 years) with a mean height of 1.73 ± 0.10 m were able to participate in the locomotion assessment (P3, P5-P8, P10, P12). For the control group, 7 healthy subjects were recruited (mean age 31.9 ± 6.2 years, range 24-42 years) with a mean height of 1.71 ± 0.08 m. Because of the variability in the patients’ individual response patterns, no group analysis was performed. Every iSCI patient was compared individually with the whole control group. The range of ankle dorsiflexion increased at least unilaterally in 6 of 7 patients (P3, P5-P8, P10) after treatment. As an example, P3 increased ankle dorsiflexion during swing after treatment around 90% of the gait cycle: P < .01 (Figure 3). Additionally, an increased knee flexion during early stance at baseline due to knee extensor weakness, which was also evident in 4 subjects (P3, P5, P6, P7), was improved after treatment in P3 and P5. Knee extensor weakness was also compensated in 2 subjects (P10, P12) with a hyperextended knee joint during weight bearing. Slight reduction in hyperextension was evident at post-assessment suggesting an increased reliance on their knee extensors.
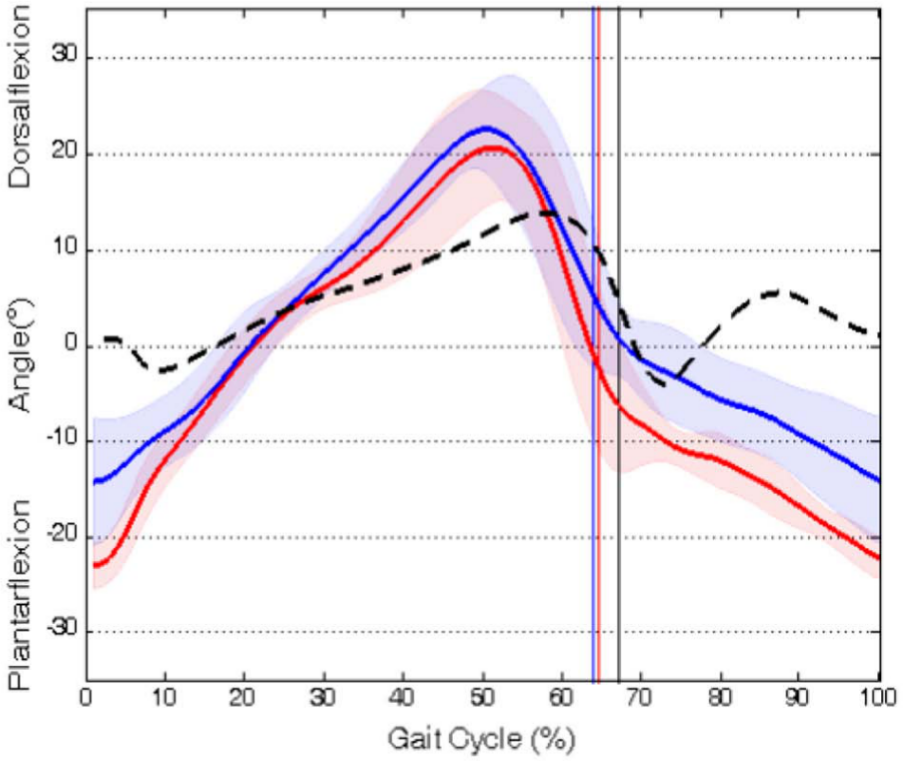
Angular displacement profiles of left ankle dorsiplantar flexion (baseline, lower solid line - red; post, upper solid line - blue) of patient 3 compared with the control group (black dashed line). X-axis: Percentage of the gait cycle, 0% = heel strike, toe-off indicated by a vertical line. Y-axis: Joint angle.
Discussion
The study assessed the effectiveness of first-person VR for intensive lower limb training in chronic iSCI patients by combining action observation and execution. The training was well accepted by the patients and the results revealed that (a) patients performed significantly better in walking speed, balance, mobility, and locomotion with increased lower limb muscle strength, and for the walking speed all subjects exceeded the MCID. In addition, 8 out of 14 patients met the MDC for the WISCI II. (b) Almost half of the patients experiencing neuropathic pain reported significant decreases in pain intensity and unpleasantness: For chronic pain intensity, 6 out of 9 patients met the MDC and 5 out of 9 met the MCID.
Motor Outcomes
Gait patterns, speed, muscle strength, and balance improved significantly after the training sessions. The improvements were clinically meaningful for the 10MWT (62% of patients met the MDC; all met the MCID). Improvements were also found in ankle dorsiflexion, reducing foot drag, in 6 of 7 tested. In combination with stronger knee extensor or general improvements in muscle strength, this increase may have enabled the patients to walk faster and more stably (improvement in 10MWT and BBS). Muscle strength is thought to be correlated with walking speed, 30 and the BBS is correlated with mobility measures, muscle strength, 31 and is important for postural control. 32 Significant improvements in functional assessments (SCIM mobility and WISCII II) also confirmed the improvements in ambulatory functions, with the MDC of the WISCI II met by 57% of patients. In concordance with the objective assessments, 93% of the patients rated their motor functions as markedly improved—in contrast with a recent study, which found that measured improvements were not similarly perceived in patients. 33
Repetitive practice, motivation and performance feedback are key concepts for motor learning. 34 Patients using the VR system performed more than 5000 ankle movements per leg during a month of training while maintaining high motivation, a higher dosage than that reported in a previous study. 35 The high dosage and thus high stimulation of motor brain areas may have induced plastic cortical and subcortical changes. In a recent study with stroke patients, effective cortical reorganization could be achieved with a mirror therapy setup. 36 Unlike locomotor task-specific training, the visual feedback in VR might activate top-down observation, motor imagery, and execution of sensorimotor networks to support functional recovery.
Pain Outcomes
Two thirds of the patients with central pain clinically improved, both immediately after the treatment and 12 to 16 weeks after treatment. The underlying mechanisms of neuropathic pain are still not well documented, but 2 recent investigations have reported beneficial effects of visual illusions on pain.12,13 In line with these studies, our prolonged, repetitive, and extensive training, may be one of the reasons for the positive effects of the treatment, as pain relief seemed to increase with 3 to 4 weeks of training on weekdays. However, the other studies were different in that their patients had complete motor SCI and the presentation of the virtual limbs was from a third-person perspective. An advantage of first-person perspective may be kinesthetic matching to support illusory effects, that is, moving avatars as in our setting.37,38
Study Limitations
The study is limited by the small and heterogeneous group of 14 participants (AIS C/D with various lesion and pain levels), the uncontrolled design, and nonblinded assessments. The pre-baseline testing helped ensure that each patient was stable prior to the intervention.
Conclusion
This study of VR-augmented training revealed short-term benefits in motor function and pain reduction. These effects may have been because of the customizable training dosage and intensity, the activation of top-down cortical processes via simultaneous action observation, motor imagery and execution, or a combination of both.
Footnotes
Acknowledgements
Thanks to Rob Labruyère for assisting in patient recruitment and Michael Brogioli for developing tests.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
There is a commercial interest of 3 authors (DK, PP, and KE) relevant to the subject of the article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
This research was supported by the International Foundation for Research in Paraplegia (IRP), Clinical Research Priority Program (CRPP) Neuro-Rehab UZH, Swiss National Science Foundation (SNF; Grant Number 51NF40-144619), OPO Foundation Zurich, and the Neuroscience Center Zurich (ZNZ).
