Abstract
Background
Current available therapies for Parkinson disease (PD) have strong limitations, and patients usually present with refractory symptoms despite all efforts. Deep brain stimulation (DBS), which has been used in PD patients for decades (since 1987), has best indications for symptoms like tremor, motor fluctuations, or dyskinesia. However, postural instability and gait disturbances (PIGD) have restricted benefits with DBS. In 2009, spinal cord stimulation (SCS), a well-established therapy for chronic pain, has emerged as a potential alternative therapy that may help control unresponsive symptoms such as bradykinesia, PIGD, and freezing of gait.
Methods
The main studies regarding SCS in PD are reviewed here from the first studies in animal models to the latest clinical trials.
Conclusions
Despite promising findings, the heterogeneity of methodologies used and small samples in human studies pose a challenging problem to be addressed in order to have robust clinical evidence to support SCS as a viable PD treatment
Introduction
Parkinson disease (PD), a common neurodegenerative disorder,1,2 has a significant impact on patients’ quality of life because motor and cognitive functions are impaired. 3 Current treatment options target only a limited spectrum of symptoms. Levodopa (L-dopa), the gold standard pharmacotherapy, 4 is currently known to be effective in treating initial appendicular PD symptoms (limb tremor, bradykinesia, and rigidity) but less effective in alleviating some axial symptoms (eg, postural instability and gait disturbances [PIGD]) as PD progresses. In addition, within 5 years of levodopa treatment, 30% of patients develop motor fluctuations and abnormal involuntary movements (dyskinesias) that are as debilitating as the disease symptoms.5-8 Similarly, the well-established deep brain stimulation (DBS) is not indicated as a treatment for PIGD and most axial symptoms when applied to the main targets—subthalamic nucleus (STN) and globus pallidus internus9,10—despite being able to ameliorate some gait parameters 11 and being consolidated as an effective treatment for resistant tremor, motor fluctuations, and dyskinesia. 12 In this context, dorsal column spinal cord stimulation (SCS) has emerged as a possible option to treat PIGD symptoms in PD, besides ensuring a less-invasive alternative to DBS.
The first report by Fuentes et al 13 showing locomotion restoration and rupture of β oscillations (aberrant basal ganglia-cortical-thalamic low-frequency oscillations,14,15 a hallmark finding of PD) in akinetic rodents paved the way for a series of studies investigating the underlying SCS mechanisms in animal models and its applicability in PD patients. 16 The present review aims to summarize and discuss the findings regarding the use of SCS in PD, highlighting important neurophysiological and clinical features, and to propose a roadmap to future investigations.
Spinal Cord Stimulation and PD
In 1967, SCS was used for the first time to treat chronic pain in a patient diagnosed with inoperable bronchogenic carcinoma. Shealy, Mortimer, and Reswick, in their pioneering work, 17 opened the way for a therapeutic approach that has, since then, been successfully used in patients with refractory neuropathic pain18,19 (for an extensive review see Verrills et al 20 ). SCS effects were thought to be exerted by mediating the electrical activation of large-diameter afferent fibers of the dorsal column, according to gate control theory.21,22 Following studies showed that other mechanisms, such as modulation of neural activity in the dorsal columns/supraspinal structures and interference on the balance between blood supply and demand, were also involved. 23 However, insights into its potential application on PD have only emerged with neural recordings during peripheral nerve stimulation in epilepsy studies, 13 showing that peripheral nerve stimulation was able to reduce the frequency and the duration of epileptic episodes by desynchronizing aberrant low-frequency neural oscillations.24-26 Because PD is also characterized by increased low-frequency neural oscillations,14,15 a natural question was raised: could the stimulation of afferent somatic pathways (such as dorsal columns in the spinal cord) induce similar results in PD? Despite pathophysiological differences among those diseases, this assumption was suitable as demonstrated by the following studies, represented by a timeline in Figure 1.
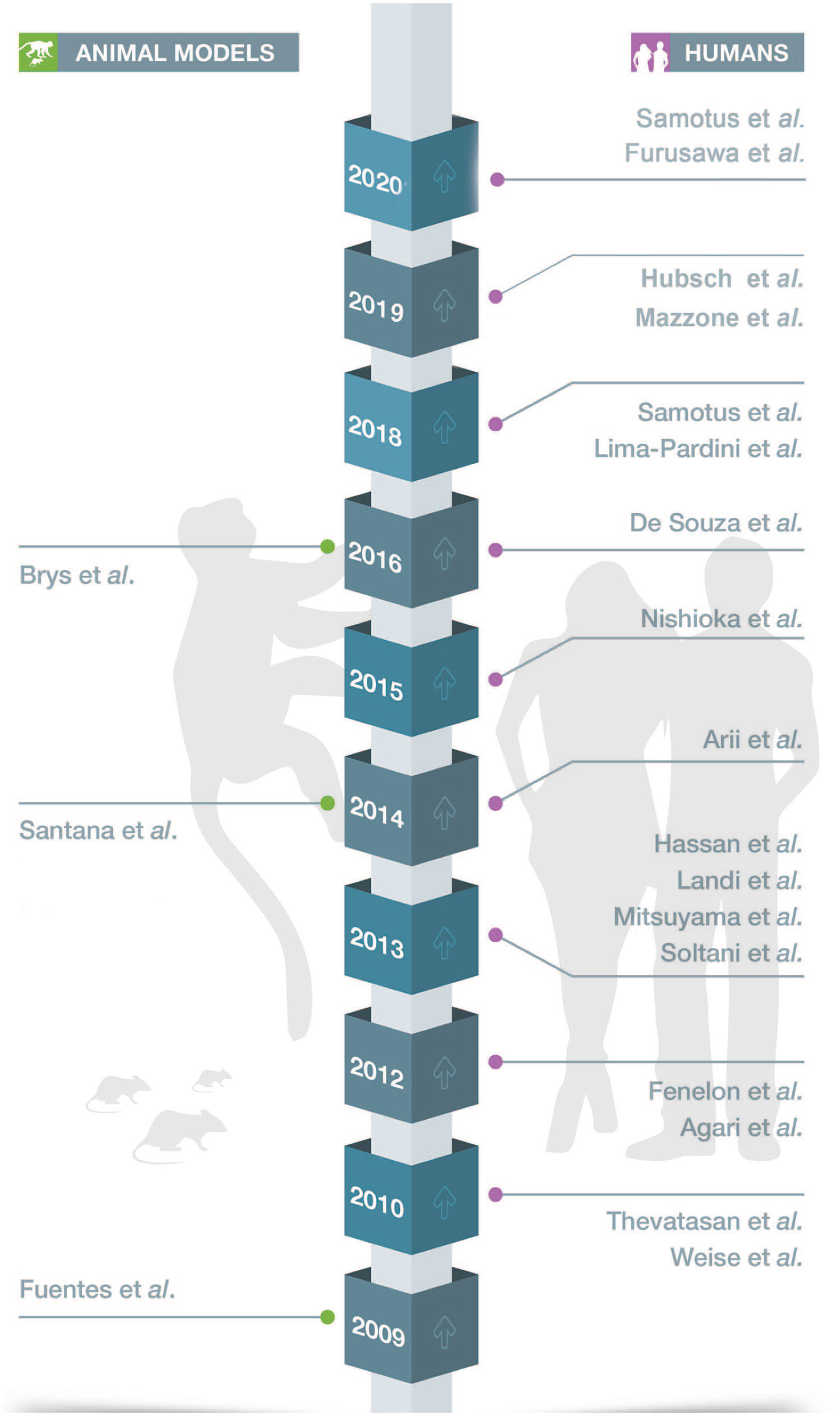
Timeline of studies exploring spinal cord stimulation on Parkinson disease (PD). Left: articles in animal models (rodents and marmosets); right: articles in PD patients.
Animal Model Studies: From Synchrony Disruption to Neural Triggering Pathways
Fuentes et al 13 used acute and chronic rodent models of PD in a pioneer attempt to answer the previous question. In all experiments, a platinum electrode was implanted epidurally above the dorsal columns of the spinal cord at the upper thoracic level. Interestingly, SCS induced an improvement in motor symptoms (increased amount of locomotion and decreased bradykinesia, especially at 300 Hz SCS) that was strongly correlated to the suppression of β-oscillations (synchrony disruption). Electrophysiological changes were detected almost instantly after SCS onset (44 ± 5 ms) in opposition of delayed motor improvements, suggesting that SCS may create a brain state permissive of locomotion instead of directly inducing the initiation of locomotion. Also, Fuentes et al 13 showed impressive reduction on L-dopa injected dosage required to restore movement under SCS in severely dopamine-depleted animals (reduction of 80%).
The above results raised enthusiasm regarding SCS as a suitable treatment for PIGD symptoms in PD and were followed by a series of studies (9 studies) approaching SCS efficacy in PD patients, as shown in Figure 1 and described later. Despite promising results in those initial clinical studies, the mechanisms underlying SCS needed to be further explored. Which pathways trigger desynchronization? How does long-term SCS affect PD pathophysiology? Would the human corticobasal ganglia-thalamic loop have a similar response to SCS? These questions provided the impetus to explore SCS effects in a nonhuman primate PD model.
Santana et al, 27 in 2014, investigated whether SCS would affect the cortico-basal ganglia-thalamic loop synchrony in a primate model of PD. The study used 5 adult male common marmosets (Callithrix jacchus) in a PD animal model with dopamine depletion secondary to 6-OHDA injections at the medial forebrain bundle. After PD induction, a spinal electrode was implanted at the T3-T4 level. Stimulation parameters were as follows: 4 - 300 Hz/100, 1600 µA/200 or 400 µs, separated by 100 µs. In this study, SCS improved akinesia and restored the activity of the corticobasal ganglia-thalamic loop to the normal state (reduced β-band synchronization in each structure and reduced coherence between pairs of structures). Also, Santana et al 27 suggested that SCS directly activates primary somatosensory areas (ventral posterolateral thalamus [VPL] and the primary somatosensory cortex [S1]) through the medial dorsal column–medial lemniscal pathway and that a phase-reset mechanism is likely involved in the β-desynchronization observed with SCS.
Also, the study by Santana et al 27 corroborates prior studies13,28 that hypothesized that disruption of synchronized oscillatory activity rather than changes in neuronal firing is the main underlying mechanism in SCS. This conclusion was based on motor improvement (freezing [31%], hypokinesia [23%], posture [23%], and bradykinesia [21%]) and β-band desynchronization regardless of the SCS frequency applied, in contrast to alterations of neuronal firing: low-frequency induced excitability, whereas high-frequency induced inhibitory effects. Therefore, firing rate patterns of neurons in the loop could not explain the improvements observed.
Later, Brys et al, 29 in 2016, tested the efficacy of SCS in a progressive and chronic PD model for the first time. The authors injected adeno-associated viral vectors encoding human α-synuclein in the right substantia nigra of rats and presented results of a 10-week follow-up. Stimulation parameters were as follows: 300 Hz/400 µs/25 µA. SCS induced an improvement in forelimb asymmetry in a weekly 5-minute session of cylindric test, from the sixth week (the first week SCS was applied) until the end of the experiment at week 10. However, no significant difference in locomotion activity was found between time and groups, nor in TH immunoreactivity.
Human Studies: From Conflicting Results to Enthusiastic Clinical Trials
The hallmark results in the rodent pioneer study brought enthusiasm into its application in humans. Since then, several human studies have been published. It is extremely fundamental to consider 2 factors when analyzing these studies: anatomy of the spinal cord and individual characterization of patients enrolled in each study. In the former, it is important to highlight that the high thoracic spinal cord has a lower diameter when compared with the cervical or low-thoracic/lumbar, which allows a current to be less selective to dorsal columns (which is also deeper in this topography), influencing other structures and tracts. 30 In fact, each topography may have a different setting of best stimulation parameters for inducing a motor improvement. 31 These aspects need to be addressed considering the latter factor because the individualization of patient condition is primordial to strengthen the conclusions on the SCS efficacy in PD.
In this context, human studies go from an initial controversial short-term trial to more robust clinical trials, passing through several small case reports/series. Despite the initial skepticism, the latest clinical trials had promising results toward clinical application.
Pioneering Trial and Conflicting Results
In contrast to the rodent study, the first human study in 2010 (Thevathasan et al 32 ) showed no motor improvement (Unified Parkinson’s Disease Rating Scale [UPDRS] motor subscale) with SCS in PD. This study used an electrode (Medtronic models 3487a or 3898) implanted into the high cervical epidural space of 2 patients (patient 1 received a 130-Hz [common parameter in DBS applications]/3V/240 µs stimulation, whereas patient 2 received a 300-Hz [highlighted results in the rodent pioneer study]/4 V/200 µs stimulation).
Fuentes et al 28 brought to light possible explanations for the conflicting results (rodents vs humans) when exploring the mechanistic approach involved in this study, suggesting as major factors the electrode geometry and spinal cord level of stimulation. According to Fuentes et al, 28 in the rodent study, the flat electrode was in a longitudinal position, resulting in an electrode/spinal cord contact area from 4 to 7 times greater than that in the cylindrical and transversal electrode used in the Thevathasan et al 32 study. Also, differences in neuron recruitment between high thoracic (rodent study) and high cervical (human study) stimulation were proposed to play a role. Despite the logical reasoning pointing to an apparent recruitment of more neurons in cervical stimulation, findings from chronic SCS studies33,34 demonstrated a proportionally smaller recruitment in sensory neurons from lower limbs when compared with thoracic stimulation (assumed as a key role factor for SCS efficacy in PIGD). The authors argued that such findings could partially explain the minimal improvement in gait and posture in the pioneer human study.
Case Reports and Case Series
After the initial human study, several other studies were performed in a small number of patients with chronic pain syndromes (especially in the leg/back) who later developed PD and were under treatment with SCS (Weise et al, 35 Fénelon et al, 36 Agari and Date, 37 Landi et al, 38 Soltani and Lalkhen, 39 Mitsuyama et al, 40 and Nishioka and Nakajima 41 ).
Because those studies involved patients with previous pain, 2 factors need to be considered. First, the patients were normally submitted to the stimulation parameters and spinal cord topography commonly used on SCS for pain control. 42 In that sense, all these studies performed a low-frequency SCS (7-130 Hz) with a wide pulse width. The other factor is that the motor results could be biased by SCS action in pain control because the pain may act as a confounding variable for conclusions regarding PD symptoms. Despite that, motor improvements were constantly described.
Among those studies, the only one that demonstrated no motor improvement was the study by Weise et al 35 in 2010. In agreement with Thevathasan et al, 32 this study found no clear improvement in UPDRS III subscale (including bradykinesia) and in the Timed Up and Go (TUG) test in a cervicothoracic SCS performed in a 72 year-old woman with advanced PD and chronic back pain. A previous 2-year DBS treatment had been unsuccessfully applied for gait abnormalities. No further information regarding stimulation parameters, electrode design, and follow-up were provided; therefore, no relevant inference could be made.
Meanwhile, the other studies described a broad spectrum of motor improvements after SCS. Those studies performed a low-thoracic, low-frequency stimulation (common in SCS for pain control 42 ) in patients with chronic leg/back pain syndrome and later-developed PD disease. Fénelon et al, 36 in 2012, described a 50% reduction in UPDRS scores and reduction in bradykinesia/tremor subscore with SCS (130 Hz/3.5 V/410 µs/T9-T10). However no improvement in locomotion (evaluated by 7-m turn–walk back) was obtained in a 74-year old man with back failure surgery syndrome and tremor-predominant PD. Agari and Date 37 described the effects of SCS (5-20 Hz/0-4 V/210-330 µs/T7-T12) on PIGD in 15 PD patients with chronic refractory lower back/leg pain, demonstrating a significant improvement in posture, gait, and bradykinesia subscores (UPDRS) at 3 months (reduced at 12 months) and improvement in locomotion with gait scores. Landi et al 38 showed improved locomotion and balance in a 65-year-old woman with advanced PD (who later developed chronic leg pain) under long-term low-thoracic level (T9-T10) SCS (stimulation parameters: 30 Hz/1.8-2.5 V/250 µs). Then, 3 brief reports of SCS were published, with no extensive descriptions (Soltani and Lalkhen, 39 Mitsuyama et al, 40 and Nishioka and Nakajima 41 ), showing benefits, despite some conflicting results. For a more detailed review of these studies, see Cai et al. 43 Table 1 summarizes these findings.
Description of Human Studies.
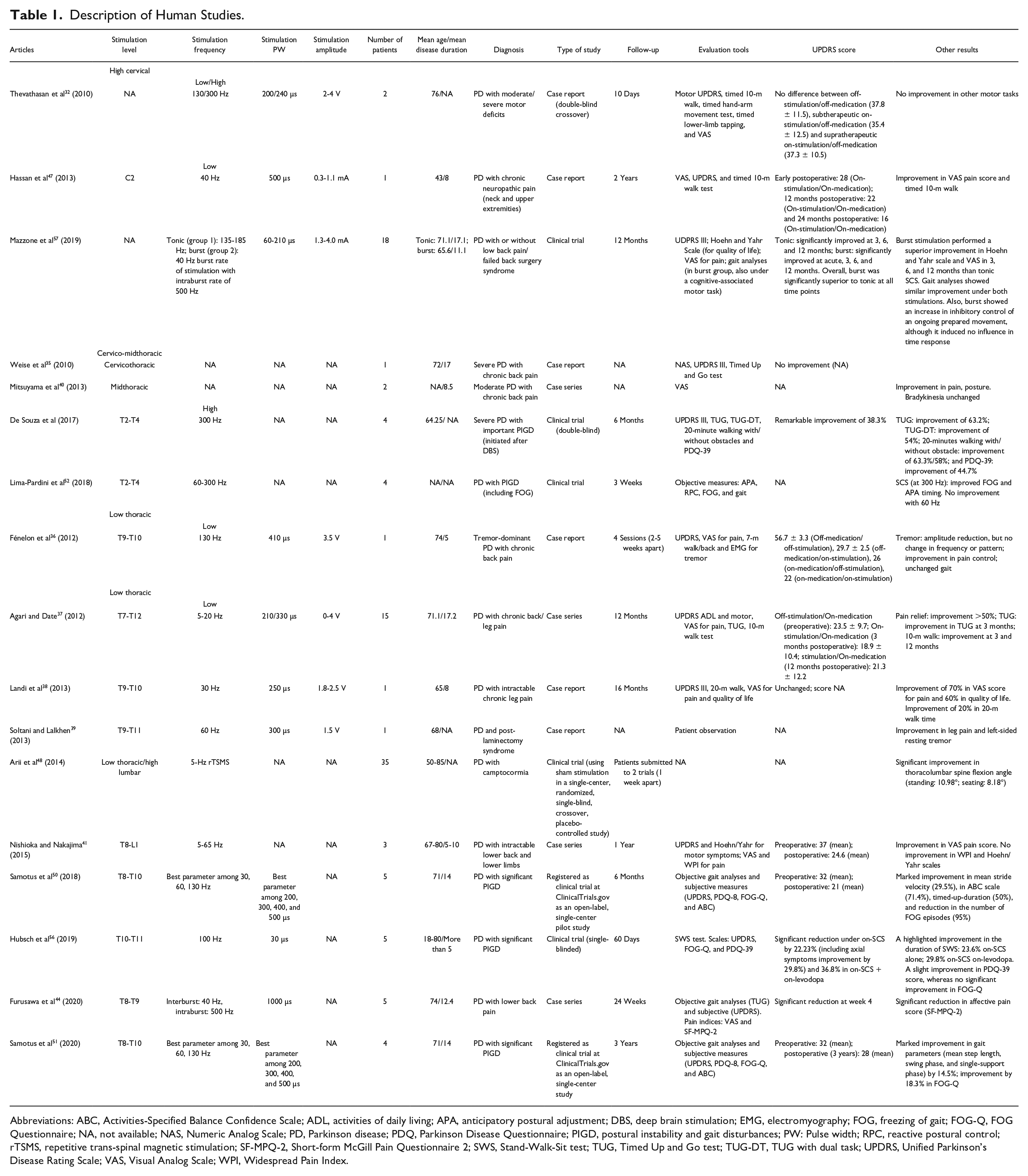
Abbreviations: ABC, Activities-Specified Balance Confidence Scale; ADL, activities of daily living; APA, anticipatory postural adjustment; DBS, deep brain stimulation; EMG, electromyography; FOG, freezing of gait; FOG-Q, FOG Questionnaire; NA, not available; NAS, Numeric Analog Scale; PD, Parkinson disease; PDQ, Parkinson Disease Questionnaire; PIGD, postural instability and gait disturbances; PW: Pulse width; RPC, reactive postural control; rTSMS, repetitive trans-spinal magnetic stimulation; SF-MPQ-2, Short-form McGill Pain Questionnaire 2; SWS, Stand-Walk-Sit test; TUG, Timed Up and Go test; TUG-DT, TUG with dual task; UPDRS, Unified Parkinson’s Disease Rating Scale; VAS, Visual Analog Scale; WPI, Widespread Pain Index.
Recently, other studies were published regarding the influence of low-thoracic SCS on parkinsonian symptoms. A brief case series of 5 PD patients (Furusawa et al 44 ), with prominent nonmotor PD symptoms (lower back pain), showed significant pain and motor improvements (in UPDRS subscores) under SCS (interburst: 40 Hz; intraburst: 500 Hz/1000 µs/T8-T9), as shown by improvements in gait at week 24 and lower-limb akinesia at week 4 when compared with baseline. This study was important for demonstrating that the burst (a paresthesia-free stimulation) SCS was also efficient in mediating motor improvements similarly to tonic SCS, in agreement with other clinical trials (described later). Additionally, 2 case reports were recently published, demonstrating the efficacy of low-thoracic SCS in alleviating parkinsonian symptoms, such as improvement of FOG in multiple system atrophy with parkinsonism (Zhang et al 45 ) and tremor in restless legs syndrome (De Vloo et al 46 ).
Later, SCS was tested in a high-cervical level (Hassan et al 47 ). Differently from the pioneer human Thevathasan et al 32 study (also cervical stimulation), improvements in PIGD, tremor, and rigidity were observed following SCS (40 Hz/0.3-1.1 V/500 µs/C2) in a 43-year-old woman with PD and chronic neck/upper extremities pain secondary to trauma. Importantly, the positive results of the Hassan et al 47 study were assessed throughout a long-term evaluation of 24 months (vs acute assessment Thevathasan et al 32 study), showing a sustained improvement.
Overall, the lack of consolidated concepts on the best settings of SCS (parameters and spinal cord level of stimulation) and inconsistent patient selection (different stages of PD assessment) may partially explain the heterogeneity of results found by those studies. Also, the majority of studies were performed in patients with chronic pain, and the benefits of SCS could be partially explained by interference in those pain mechanisms. Nonetheless, the long-term improvement in PIGD demonstrated by Agari and Date 37 (especially), Landi et al, 38 and Hassan et al 47 was important in highlighting the potential of SCS for posture control in PD.
Recent Clinical Trials
After important findings regarding the potential of SCS in improving PD motor symptoms in animal models and conflicting results in humans, more rigorous study designs were performed to better understand its applicability in PD patients. In this context, a single-blind clinical study showing significant immediate motor improvements with SCS in PD was published in 2014 (Arii et al 48 ).
In this study, SCS was tested under a different setting, called repetitive trans-spinal magnetic stimulation (rTSMS) in 37 PD patients with camptocormia (anteroflexion of trunk in standing position). The patients were randomly divided into 2 groups and submitted to a single session of low thoracic 5 Hz rTSMS or sham stimulation in a crossover study. rTSMS immediately induced a reduction in thoracolumbar spine flexion angle in both standing and sitting positions, but no sustained effects were detected after 1 week.
This was followed by a hallmark study by de Souza et al, 49 who evaluated SCS influence in gait disorders of PD patients under a double-blind clinical trial in 2016. Differently from Arii et al, 48 this study performed a long-term evaluation of SCS in PD, and the positive results raised enthusiasm about the chronic, sustained effects of SCS. The inclusion criteria allowed selecting patients to prospectively evaluate SCS regarding PIGD improvement as the primary clinical effect, consequently achieving a more accurate design to test SCS efficacy. For that, 4 patients with advanced PD and significant PIGD were submitted to a midthoracic (300 Hz/supine [2.0 ± 0.5 V] or upright position [4.6 ± 1.9 V]/90 µs/T2-T4) SCS. The 300-Hz frequency was selected because it showed optimal results in the initial rodent study.
Importantly, the experiments were performed in patients with chronic STN DBS (common scenario in clinical practice), in accordance with Landi et al. 38 In that sense, SCS induced a main improvement in all gait tests and motor scales at 6 months: time and number of steps to accomplish 20-m walking with or without obstacles were significantly reduced (63.3% and 70.1%; 58% and 65.7%), along with improvement in stride length (170%), TUG (63.2%), and TUG test with dual task (54%) scores, in accordance with Agari and Date. 37 In terms of motor scales, UPDRS-III was 50% lower (on SCS/on DBS/on medication) when compared with baseline (off SCS/on DBS/on medication), and PDQ-39 (quality of life outcome) ameliorated by 44.7%. Furthermore, SCS significantly reduced the risk of falls in 8% (Berg Balance Scale) and freezing (Freezing of Gait Questionnaire [FOG-Q]) in 56.4% after 6 months.
Finally, because the 300-Hz SCS elicited good results, the authors decided to evaluate SCS under a different setting to exclude placebo effects because it was an open-label design and patients could self-assess electrical stimulation. At the beginning of month 4, patients were evaluated through 20-m walking test and TUG under SCS of 2 different frequencies to mask patients from stimulation and its clinical effects: 60 Hz and 300 Hz (both induce similar paresthesia, but only 300 Hz was assumed to be therapeutic). Interestingly, motor improvements could not be replicated when using 60-Hz stimulation.
Until this point, the studies adopted a restricted preselected pattern of stimulation parameters to be used throughout the experiment. In 2018, Samotus et al 50 incorporated an innovative method instrument (gait framework), ensuring an optimization of stimulation parameters for each participant’s characteristics while performing a meticulous objective computation of the influence of SCS on different gait domains and in PD-related FOG. This open-label study (not double-blind) was performed in 5 male PD patients with significant PIGD under low thoracic (T8-T10) SCS. Patients were evaluated (clinical scales and gait analysis) preoperatively and afterward at 2-week interval sessions, in which SCS was performed with a random selection of pulse widths (200, 300, 400, and 500 µs) and frequencies (30, 60, 130 Hz) until the end of week 16. Then, the best combination of parameters was selected for each participant to be used in the last session at week 24. Between sessions, SCS was performed daily with the optimized parameters established in the previous visit and between 16 and 24 weeks with the best parameters overall.
It is worth noting that the best parameters for those patients were achieved when large pulse widths were applied in SCS. As already known, this type of SCS is less selective, allowing a long time of stimulation to induce depolarization of less-sensitive structures and deeper structures (other than the dorsal column itself). 30 Thus, these results may indicate other mechanisms for SCS action rather than VPL/S1 modulation (mediated by dorsal columns), as suggested by Santana et al. 27 This effect can be even more prominent when applied to the upper thoracic spinal cord because its diameter is smaller. 30
After detection of individual best parameters, gait analysis demonstrated that SCS promoted a marked improvement in mean stride velocity at week 24 (29.5%) when compared with preoperative assessment, whereas mean step length, percentage swing, single and double support, step time, and stance phase showed nonsignificant improvements. Timed-up duration (sit-to-stand) was also significantly improved by 50%, corroborating similar findings in other studies.37,38,49 Importantly, SCS showed a significant reduction (95%) in the number of episodes of FOG (but not in the duration) at week 24, despite the nonsignificant improvement in FOG-Q (questionnaire), which was inferior to that demonstrated by de Souza et al. 49 Finally, SCS showed a significant improvement in UPDRS (including bradykinesia) and the Activities-Specified Balance Confidence (ABC) Scale by 33.5% and 71.4%, respectively.
Despite its impact, this study demonstrated only a short-term evaluation of SCS effects because only a period of 24 weeks was assessed. Aiming to fill this gap, the same study group recently published a new study (Samotus et al 51 ) describing the effects of the long-term SCS on PD. For that, 4 patients of the previous study remained under observation, and a 3-year SCS effect (during 4 passes of self-paced ambulatory walking, after 1-1.5 hours of L-dopa) was assessed. The data were compared with the baseline results from the initial study (Samotus et al 50 ), using the same instruments of evaluation. Importantly, the number of FOG episodes were reduced at 3 years of SCS when compared with baseline, and 2 participants did not freeze during the evaluation. This result was followed by improvement by 18.3% in the FOG-Q. Additionally, the long-term SCS improved the temporal gait asymmetry as well as gait parameters (mean step length, swing phase, and single support phase) by 14.5%. Furthermore, subjective analyses also followed this trend, as demonstrated by improvement of rigidity and axial symptoms UDPRS III subscores, respectively, by 23.1% and 20.4% and by a smaller improvement in bradykinesia subscore (9.4%). No improvement was observed in the ABC scale.
Based on the above findings (initial results from Samotus et al 50 ), de Lima-Pardini et al 52 hypothesized that SCS-mediated PIGD improvement in PD patients could be a result of balance improvement. To test this hypothesis, they analyzed movement mechanics of 4 PD male patients with PIGD (including FOG) under midthoracic (T2-T4) SCS. The impact of postural adjustment in predictable (called anticipatory postural adjustment [APA]) and in unpredictable (called postural reactive control) situations was tested in different settings. In addition, FOG detection was assessed through an accelerometer during a 10-m walk task, in 3 trials. Patients were evaluated in the following conditions: SCS off, 60 Hz SCS, and 300 Hz SCS with and without medication for at least 12 hours, with each session at least 1 week apart.
Overall, this study showed that 300-Hz SCS significantly improved FOG and APA duration (but not APA amplitude), which is in accordance with previous studies that showed that deficient APA is associated with FOG episodes. 53 Because APA timing (not amplitude) is thought to be modulated by the supplementary motor area (SMA),54,55 the authors hypothesized that SCS may modulate SMA activity, secondary to the disruption of thalamic-basal ganglia-cortex aberrant oscillations. In addition, this study showed no improvement in postural control, demonstrating that this control has no major influence on FOG episodes, after initial studies showing conflicting results (Agari and Date 37 and Landi et al 38 ). However, those initial studies performed a qualitative measurement analysis assessed through the pull test, depending on a subjective evaluation.
Finally, 2 studies were recently published. First, Hubsch et al 56 evaluated the influence of tonic (100 Hz/30 µs/T10-T11) SCS in 5 patients with prominent axial symptoms, primarily through the Stand-Walk-Sit Test (SWS) assessed by a blinded neurologist (this was not a double-blind study because patients could feel paresthesia). Patients were assessed 60 days poststimulation, both off and on levodopa. An improvement in the duration of SWS was shown under SCS (23.6% on-SCS alone and 29.8% on-SCS on-levodopa; whereas on-levodopa reduced in 19.3%), besides the reduction in the number of steps (12.4% on-SCS alone and 20% on-SCS on-levodopa, whereas on-levodopa reduced in 18%). Also, this study showed a significant reduction under on-SCS in the motor subscale UPDRS III by 22.23% (including axial symptoms that improved 29.8%). Such improvement was similar to the on-levodopa improvement but lower than the one observed with on-SCS + on-levodopa (36.8%). A slight improvement in PDQ-39 scores (especially in mobility scores) was obtained in on-SCS, whereas no significant improvement in FOG-Q was achieved. For more details of the influence of SCS in FOG, see Fonoff et al. 42
Lastly, Mazzone et al 57 performed a high-cervical SCS in PD patients, approaching an interesting aspect: differences in motor improvement and pain relief of PD patients under different types of SCS (tonic vs burst). For that, the study separated 2 groups: (1) 6 PD patients with pain conditions (low back pain and failed back surgery syndrome), with pain relief as the primary SCS outcome, submitted to tonic stimulation (135-185 Hz/60-210 µs/1.3-4.0 mA/electrode tip at C1-C2) SCS; and (2) 12 PD patients submitted to burst (trains of high-frequency stimuli: 40-Hz burst rate of stimulation with intraburst rate of 500 Hz/1000 µs/0.3-0.7 mA/electrode tip at C1-C2) SCS to treat PD motor symptoms. The study performed an acute and long-term follow-up (12 months).
Interestingly, results showed that burst stimulation yielded a superior improvement in UDPRS III (including PIGD, bradykinesia, and tremor), quality of life (Hoehn and Yahr Scale), and VAS for pain score (at 3, 6, and 12 months), when compared with tonic SCS. It is important to highlight, however, that both types of stimulation induced significant improvements when compared with baseline and that the scores after both treatment regimens had a tendency to equalize at 12 months, with the exception of VAS. In addition, acute burst SCS (2 days) was able to induce significant improvement in those tests/scales, which was not found with acute tonic SCS (in accordance with tonic SCS results in the Thevathasan et al 32 study). This finding may indicate different mechanisms of action between tonic and burst stimulation, which may result from distinct local/suprasegmental neural mechanisms, neurotransmitters, and neuroactive substances.58,59
Furthermore, gait analysis (performed at 6 months) showed improvement under both tonic and burst stimulation, especially in stride length, step length, and cadence. In the burst SCS group, a cognitive-associated motor task was also performed, showing no influence in time response (moving the joystick after a Go signal), despite increase in inhibition of an ongoing movement (not moving the joystick after the stop signal, with a Go signal thereafter). The authors suggested that it may result from increased action of burst SCS in the frontal lobe and cortico-subthalamic-palliday inhibitory pathway. 57
Overall, because those human studies applied a more rigorous study design, their positive SCS results raised expectations regarding its future application. In this context, the results of de Souza et al 49 for long-term PIGD improvement under a double-blind clinical trial were substantial and not caused by the self-assessment of the electrical stimulation (exclusion of placebo effect, not controlled in the other previous human studies). Also, SCS impact on FOG, a clinical aspect with severe impairment in PD, was rigorously investigated under an individual selection of the best parameters of SCS by Samotus et al, 50 which demonstrated an important reduction in FOG episodes. In agreement, Lima-Pardini et al 52 showed improvement of FOG under SCS and went further suggesting that this improvement is related to balance control improvement in anticipatory posture adjustment (not in reactive control, as shown through subjective analyses by Agari and Date 37 and Landi et al 38 ), modulated by SMA. Finally, Mazzone et al 57 performed and compared different types of stimulation (tonic vs burst) SCS for PD and suggested that burst stimulation may increase its clinical benefits by interfering in different neural mechanisms.
More than a decade after the Fuentes et al study, 13 many questions are yet far from being answered despite all the human studies. These questions rely on the complexity of how an electrical stimulation influences the local and suprasegmental neural circuitry, considering that its influence may not be completely tuned to individual neurons or tracts. In that sense, establishing the ideal parameters of stimulation (frequency, pulse width, and current amplitude) and topography of stimulation remains an important gap before moving to its clinical application.
Conclusion
The literature on SCS as a therapeutic option for PD has developed at a steady and fast pace, as shown by the time lapse between the first study in rodents 13 in 2009 and the many subsequent studies in humans (n = 15) by 2020. Animal studies have already revealed underlying SCS mechanisms that were unknown to the scientific community. The Santana et al 27 study in marmosets, for example, yielded strong evidence for the “permissive state” proposed by Fuentes et al, 13 further establishing that the β-band synchronous oscillatory behavior of the cortico-basal ganglia-thalamic circuit might be disrupted via a VPL/S1 phase-reset mechanism.
But most important are the potential benefits that patients refractory to other forms of treatment may obtain from this less-invasive therapy that yields promising results in PD. Despite negative initial results in the first human studies, following studies49-52 provided preliminary evidence supporting SCS as a viable therapeutic option to improve PIGD, bradykinesia, and rigidity. These effects on unresponsive symptoms may strongly influence disease management and patients’ quality of life in the future. Yet many questions remain. How to choose adequately patients who will benefit from SCS? Which stimulation parameters should be used? Where are the ideal electrode implantation sites?
It is hoped that this review will assist researchers to understand adequately the shortcomings of previous studies and the interesting methodological tools already at our disposal. In that sense, future studies should target the remaining gaps necessary for translating SCS into a daily practice for PD. Specifically, long-term evaluation studies with more enrolled patients (especially randomized double-blind studies) should focus on 2 important aspects: (1) prior characterization of the most adequate stimulation parameters for each individual and (2) implementing novelties in objective motor measures alongside established clinical scales to assess SCS effects on PD.
Footnotes
Acknowledgements
The authors acknowledge the financial support by CAPES Foundation (Coordination for the Improvement of Higher Education Personnel); The Michael J. Fox Foundation for Parkinson’s Research; Brazilian Innovation Agency FINEP 01.06.1092.00;INCEMAQ—Program of National Institutes of Science and Technology of CNPq/MCTIC; Graduate Program in Neuroengineering, Santos Dumont Institute; and Brazilian Ministry of Education (MEC). The authors also acknowledge Phillip Cesar Lima da Silva and Danth Andrade da Silveira for their contributions in the figure design.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support by CAPES Foundation (Coordination for the Improvement of Higher Education Personnel); The Michael J. Fox Foundation for Parkinson’s Research; Brazilian Innovation Agency FINEP 01.06.1092.00; INCEMAQ—Program of National Institutes of Science and Technology of CNPq/MCTIC; Graduate Program in Neuroengineering, Santos Dumont Institute; and Brazilian Ministry of Education (MEC).
