Abstract
Transcutaneous electrical stimulation (tES) is a new approach that aims to stimulate the brain. Recently, we have developed tES approaches to enhance plasticity that modulate cortical activity via the greater occipital nerve (ON) in a “bottom-up” way. Thirty subjects between the ages of 55 and 70 years were enrolled and tested using a double-blind, sham-controlled, and randomized design. Half of the participants received active stimulation, while the other half received sham stimulation. Our results demonstrate that ON-tES can enhance memory in older individuals after one session, with effects persisting up to 28 days after stimulation. The hypothesized mechanism by which ON-tES enhances memory is activation of the locus coeruleus–noradrenaline (LC-NA) pathway. It is likely that this pathway was activated after ON-tES, as supported by observed changes in α-amylase concentrations, a biomarker for noradrenaline. There were no significant or long-lasting side effects observed during stimulation. Clinicaltrial.gov (NCT03467698).
Introduction
Advancements in medicine and public health, improved standards of living, and increases in education and nutrition have lengthened the human life span. 1 This increase in longevity has made evident that cognitive development in adulthood and old age is both variable across and malleable within individuals. One crucial aspect of human cognition is memory. Memory impairments are most often expressed by older people, with the processes of encoding information showing the greatest declines due to aging. 2 The root causes of age-related memory decline are not entirely understood, though recent evidence suggests a strong role for downregulated neuromodulatory processes. 1
The lifelong potential of the human brain for neuroplasticity can be harnessed to maintain the viability of neural structures and postpone the onset of cognitive decline. One such way to achieve this is by transcutaneous electrical stimulation (tES). TES aims to stimulate the brain in ways that avoid the complications of trying to artificially upregulate these neurotransmitters using pharmacological methods. Yet, tES has received relatively little attention in aging research, even though the goals of selectively modulating brain activity and triggering the release of neuromodulators have been pursued for more than a century. Recently, bottom-up approaches to enhance cortical reorganization have been introduced. The bottom-up modality has been shown to modulate cortical activity via peripheral nerve stimulation (tES) further influencing the central nervous system and neuroplastic changes. 3 TES applies weak direct currents to the scalp to modify cortical excitability, inducing long-lasting changes with no significant adverse effects and low levels of discomfort for participants. 4 Moreover, combining tES with behavioral training can have an augmenting effect on learning.
Recent research has shown that invasively stimulating peripheral nerves such as the tenth cranial nerve, that is, the vagus nerve, in conjunction with presenting either tones or rehabilitative training, respectively can help patients suffering from tinnitus5,6 or patients undergoing motor rehabilitation following ischemic stroke. 7 Research implementing transcutaneous vagus nerve stimulation (tVNS) in healthy older individuals has been used to show the capability of non-invasive stimulation techniques to improve associative memory performance in older individuals, even after a single session.8-10 Furthermore, continued stimulation of the vagus nerve was shown to modulate memory formation in an undirected way. 11 This outcome was achieved by exerting the effects of stimulation via the ascending fibers of the vagus nerve that synapse with neurons in the nucleus of the solitary tract (NTS), which then project to the locus coeruleus (LC) and promote noradrenaline (NA) release that plays a key role in driving neuroplasticity and related memory processes.12,13 A recent functional magnetic resonance imaging (fMRI) study additionally confirms that it is possible to modulate the pathway from the NTS to the amygdala and hippocampus by using noninvasive stimulation. 14 Furthermore, it is known that the NTS, next to the vagus nerve, also receives inputs from the greater occipital nerve.15-17 Further evidence was revealed in generalized pain disorders such as fibromyalgia and chronic pain. 18-21 We showed that pain modulation goes through the LC-NA pathway, via ascending and descending LC projections, 22 further suggesting that stimulating the greater occipital nerve modulates the LC-NA pathway.
The objective of this study is to increase our understanding of the effects of transcranial direct current stimulation (tDCS) targeting the greater occipital nerve during an associative memory task, and to determine if tDCS may be used as a way to enhance brain plasticity during an associative memory task to optimize associative memory performance. We hypothesize that ON-tES can upregulate memory in healthy older persons via stimulation targeting the LC-NA pathway.
Methods
Participants
The study is double-blinded, sham-controlled, and uses randomized design. Participants were included if they met the following criteria: no evidence of cognitive deficits on neuropsychological screening; no presence of any neurological or psychiatric diseases; no cardiac diseases; no psychoactive or blood pressure medication use; no abuse of alcohol or drugs; English as the native language; no knowledge of the Swahili language; able to give informed consent. The study was in accordance with the ethical standards of the Helsinki declaration (1964) and was approved by the Institutional Review Board of the University of Texas at Dallas (#18-40). After consenting, each participant performed a wide range of tests covering mood, executive functioning and memory including Beck Depression Inventory (BDI), 23 Beck Anxiety Inventory (BAI), 24 Mini-Mental State Examination (MMSE), 25 California Verbal Learning Test (CVLT-II), 26 Trial Making Test A (TMT-A) and B (TMT-B), 27 WAIS-IV Digit Span and Coding, 28 Controlled Oral Word Association Test (COWAT), 29 or the Delis-Kaplan Executive Function System (D-KEFS). 30 The MMSE was used to screen for cognitive impairment. All participants included in the study scored above the single cutoff score, indicating no cognitive impairment and that their memory performance was within normal range for their age and education level. Their handedness was assessed by the Edinburgh Handedness Inventory. 31 The trial was registered on clinicaltrial.gov (NCT03467698).
Experimental Paradigm
Participants performed a Swahili-English word association task, based on a well-known paradigm. 32 Participants were asked to learn a list of 50 Swahili-English word pairs (eg, mashua-boat) selected from previously published norms. 32 Participants learned the list of word pairs across a total of 3 blocks of study (S) and test (T) phases. The study phase of each block consisted of 50 word pairs followed by a test phase of the 50 words. During study trials, participants saw each Swahili word and its English translation on a computer screen simultaneously for 5 seconds and were told to study the pair so that they could recall the English word given the Swahili word. After every study period, there was a 30-second rest period (consolidation). During the test trials, subjects saw each Swahili word and a cursor, and their task was to type the correct English translation. Each test trial lasted 16 seconds, after which the computer program automatically advanced to the next item regardless of whether the participant had entered a response. If subjects fail to recall an item during testing, they were not given any feedback. Participants studied the entire list in each study period but only items that they had not yet recalled in the previous block were tested in the test. Participants were then dismissed and returned for the test 7 days and 28 days later (recall phase). During this test, subjects were shown each Swahili word for 16 seconds and were asked to type the correct English translation. This is identical to the test phase from the learning period. See Figure 1 for study design.
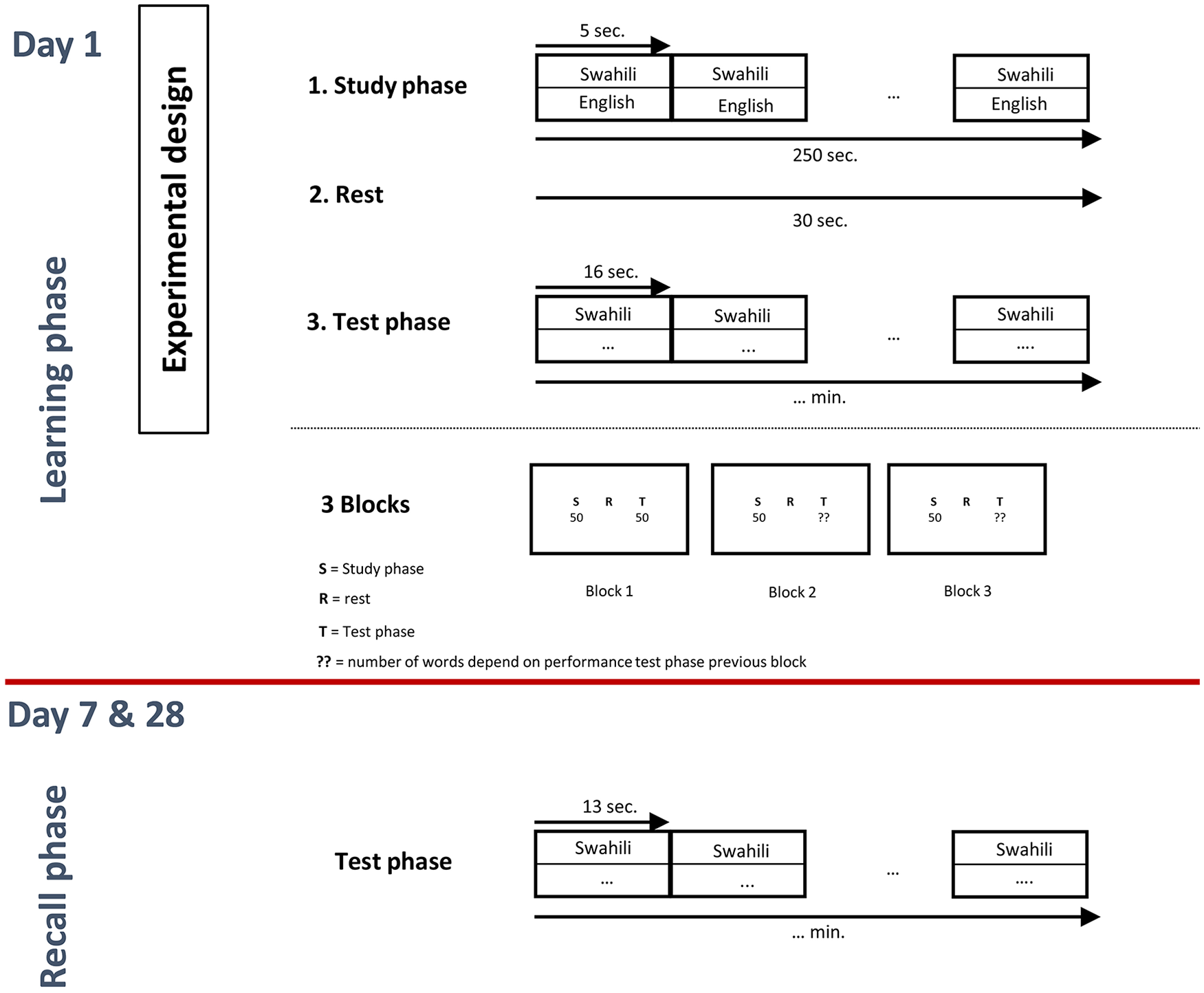
Study design for the word-association memory task.
Transcutaneous Electrical Stimulation
We utilized tDCS delivered by a specially developed, battery-driven, constant current stimulator with a maximum output of 10 mA (http://www.neuroconn.de) via a pair of saline-soaked surface sponges (35 cm2) on the scalp. One electrode each was placed over the left and right C2 dermatomes. A constant current of 1.5 mA intensity was applied during each of the 3-study phases (ie, 250 seconds × 3 blocks). For sham tES, placement of the electrodes was identical to active tES. TES was first switched on in a ramp-up fashion over 30 seconds. Current intensity (ramp down) was gradually reduced (over 5 seconds) as soon as tES reached a current flow of 1.5 mA. Hence, sham tES only lasted 10 seconds for each block. The rationale behind this sham procedure was to mimic the transient skin sensation at the beginning of active tES without producing any conditioning effects on the brain. Previous findings by our group show that participants are unable to tell whether they received real or sham stimulation following this protocol. 18
Saliva Collection
Participants’ saliva was collected 4 times during the experiment: before stimulation, immediately after stimulation, 7 days after stimulation, and 28 days after stimulation. The participants were asked to refrain from dental work at least 48 hours before each saliva collection. Participants were further requested to refrain from foods and drinks with high sugar, acidity, caffeine content, or alcohol 1 day prior to saliva collection. Energy drinks, nicotine consumption, nonapproved prescription drugs, steroidal/anti-inflammatory drugs were also avoided by participants. All participants were asked to have a good night’s rest the day before the experiment, as well. Participants were asked to avoid a major meal 60 minutes before the experiment, avoid brushing their teeth 45 minutes before the experiment, avoid indulging in any rigorous exercise and were asked not to drink water or rinse their mouth 10 minutes before saliva collection. If the study was scheduled for the afternoon, participants were requested to avoid taking naps during the day. When the participants were ready to collect saliva, they were requested to gently tilt their head backward and collect saliva on the floor of their mouth and when ready, passively drool into the mouthpiece of the tube provided by Salimetrics. The participants were requested to collect 2 mL of saliva in one straight flow and avoid breaks between drool as much as possible. The length of time to collect 2 mL of saliva was noted and the timer was started only when participants began to passively drool into the tube. The flow rate was calculated using the formula given by Salimetrics:
On completion of the collection procedures, a total of 80 saliva samples were packed in dry ice and sent to the Salimetrics laboratory for analysis. The Salimetrics analysis protocols and determination techniques for each of the 2 targeted biomarkers are described below.
The amount of α-amylase in the sample is directly proportional to the increase in absorbance at 405 nm. Ten microliters of the sample are diluted and well mixed. Eight microliters of the diluted samples are then pipetted into individual wells of 96-well microtiter plate. A volume of 320 µL of preheated chromagenic substrate solution is added to each well and the plate is rotated at 500 to 600 RPM at 37 °C for 3 minutes. Optical density of the sample is determined at the 1-minute mark and again at the 3-minute mark.
To determine cortisol levels, a highly sensitive enzyme immunoassay was used, which uses 25 µL of saliva per determination and has a lower sensitivity of 0.007 µg/dL, a standard curve range from 0.012 to 3.0 µg/dL, an average intra-assay coefficient of variation of 4.6%, and an average inter-assay coefficient of variation of 5.9%.
Blinding
At the end of the experiment, participants were asked after the study if they thought they were assigned to the sham or active group.
tES Adverse Events
We used the tES exit questionnaire developed by Brunoni et al 33 to assess potential side effect (headache, neck pain, scalp pain, tingling, itching, burning sensation, skin redness, sleepiness, trouble concentrating, mood changes).
Procedure
All participants were randomly assigned to the active or sham tES group at the beginning of the study such that half of them were in each group. All participants received (active or sham) tES during the study phase, but not during the rest or test phases, at visit 1. Participants in the Swahili-English word association task must come back 7 and 28 days after their first visit to perform one test phase (no tES). The person controlling the tES device was not involved in instructing the participant; this was done by a second person that was blind to the stimulation protocol. A third person will conduct the second and third visits (7 and 28 days later).
Statistics
Chi-square (χ2) tests were calculated to compare if there is a difference between the active and sham group for sex, Hispanic background, race, and handedness. In addition, 1-way analyses of variance (ANOVAs) were computed to see if there is a difference between the active and sham group for BDI, BAI, MMSE, CVLT-II, TMT-A and -B, Digit Span, Coding, COWAT, or the D-KEFS.
A repeated-measures ANOVA with stimulation (active vs sham) as the between-subjects variable and correctly recalled words as well as speed of processing immediately after learning, 7 and 28 days after learning as within-subjects variables were used. If a significance was obtained a simple contrast analysis was applied to see the effect of correctly recalled words at 7 and 28 days after learning, respectively, for active versus sham stimulation.
Pearson correlations were obtained between correctly recalled words at day 7 and day 28, corrected for the words the remember immediately after the training.
A multivariate analysis of variance (MANOVA) was calculated with stimulation (active vs sham) as the independent variable and α-amylase levels immediately after stimulation, 7 and 28 days after stimulation as dependent variables and α-amylase levels at baseline at covariate. If a significance was obtained a univariate ANOVA was applied to see the effect of α-amylase levels immediately after stimulation, 7 and 28 days after stimulation between active and sham stimulation. A similar analysis was applied for cortisol levels.
To see if there was an effect for blinding, we applied χ2 tests to compare what participants receive (active vs sham stimulation) versus what they think they receive (active vs sham stimulation). In addition, we applied a MANOVA with stimulation (active vs sham) as the independent variable and the different side effects as dependent variable.
Results
We screened 49 participants. Six participants withdrew prior to the trial procedure due to loss of interest or time conflict. Thirteen were excluded due to ineligibility. Thirty subjects between the ages of 55 and 70 years were enrolled (see Figure 2). No differences were obtained for sex, χ2(1) = 1.53, P = .22; Hispanic background, χ2(1) = 0, P = 1.00; race, χ2(2) = 4.00, P = .26; or handedness, χ2(1) = 1.03, P = .31; between the active and sham stimulation condition. Furthermore, no difference was demonstrated for age (U = 111, P = .95), years of education (U = 87.5, P = .29), BDI (U = 102, P = .64), BAI (U = 98, P = .52), MMSE (U = 95.5, P = .47), CVLT (U = 91, P = .37), TMT-A (U = 80, P = .18), TMT-B (U = 102, P = .77), Digit Span (U = 105.5, P = .78), Coding (U = 74.5, P = .12), COWAT (U = 106, P = .79), and D-KEFS (U = 91, P = .37) between the active and sham stimulation condition. See Figure 3 for an overview.
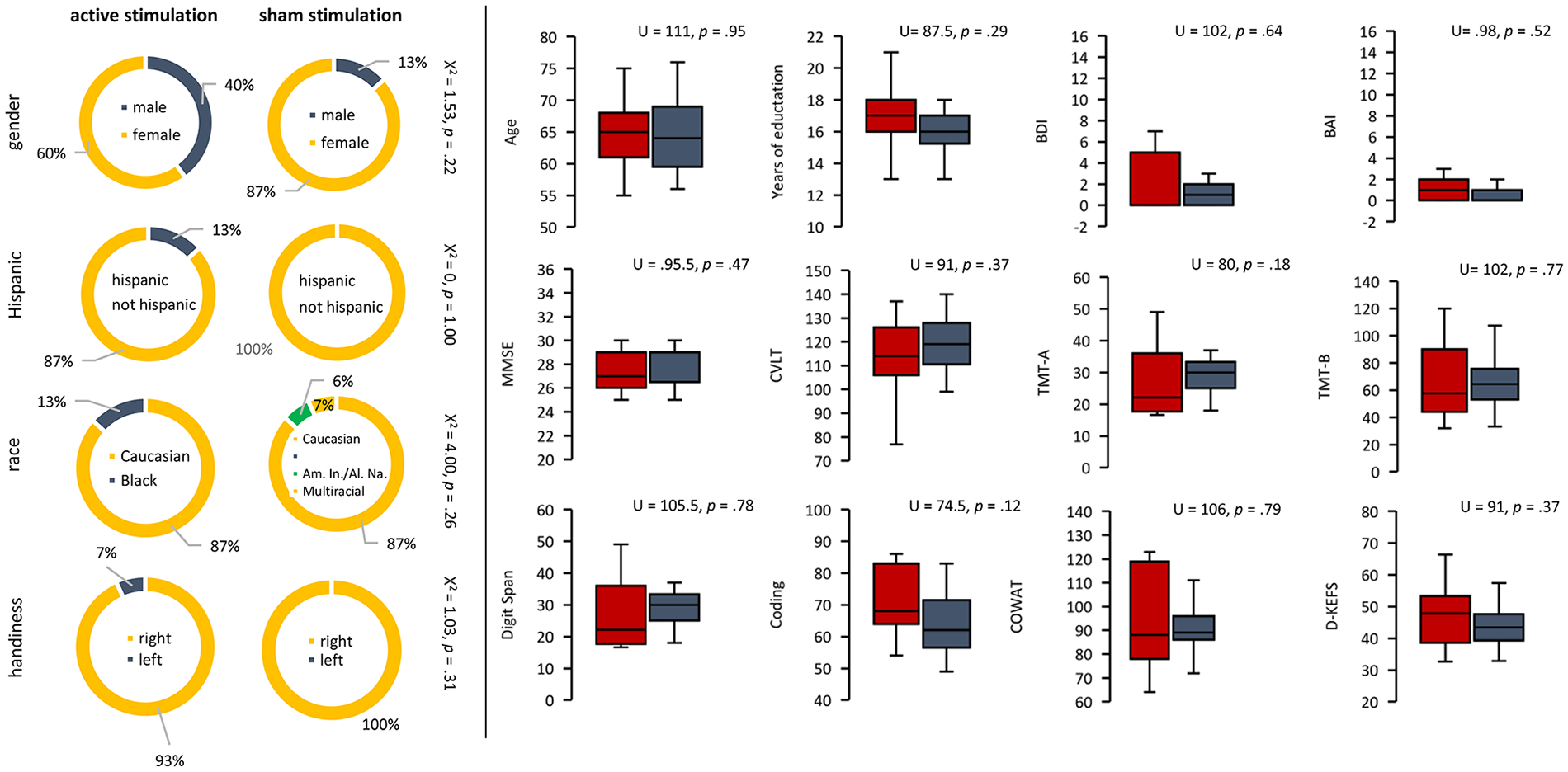
No differences were obtained for sex, Hispanic background, race, or handedness between the active and sham stimulation conditions. Furthermore, no difference was demonstrated for age, years of education, Beck Depression Inventory (BDI), Beck Anxiety Inventory (BAI), Mini-Mental State Examination (MMSE), California Verbal Learning Test (CVLT), Trial Making Test A (TMT-A), Trial Making Test B (TMT-B), Digit Span, Coding, Controlled Oral Word Association Test (COWAT), or the Delis-Kaplan Executive Function System (D-KEFS) between the active and sham stimulation conditions.
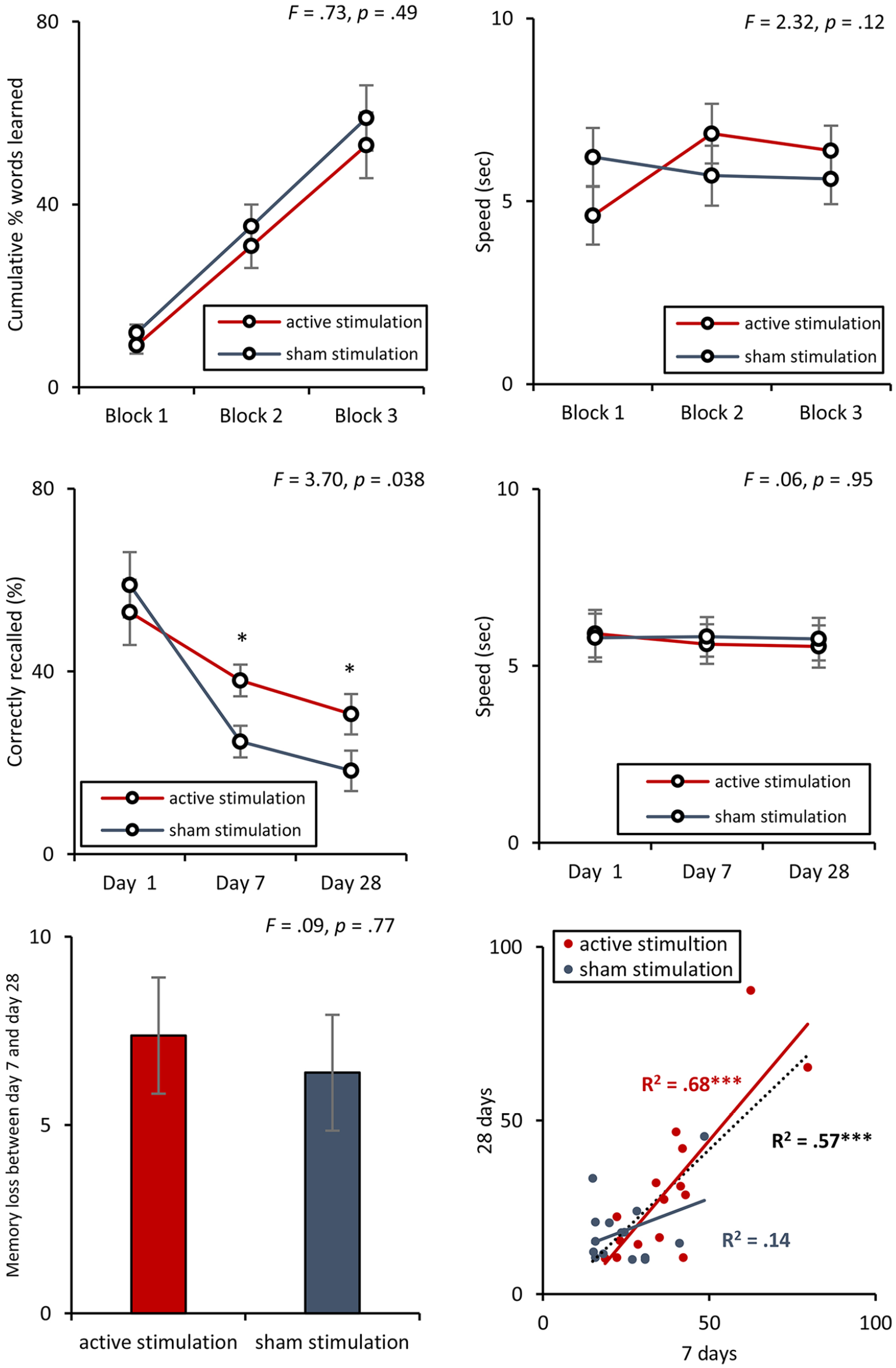
No difference between the active and sham stimulation groups was obtained for how many words were learned and processing speed during day 1. A significant effect was observed for correctly recalled words 7 and 28 days after learning. occipital nerve transcutaneous electrical stimulation (ON-tES) did not have effect on processing speed when comparing active and sham simulation group 7 and 28 days after learning the words A positive correlation was obtained between correctly recalled words at day 7 and day 28.
Effect of ON-tES on Memory Task
No difference was obtained between how many words were learned during day 1 between the active and sham stimulation groups, F(2, 27) = 0.73, P = .49. This goes together with no difference between the active and sham simulation group for processing speed during day 1, F(2, 27) = 2.32, P = .12. See Figure 3.
A repeated-measures ANOVA with stimulation (active vs sham) as the between subjects variable and correctly recalled words at the first day, and 7 and 28 days after learning showed a significant interaction effect, F(2, 27) = 3.70, P = .038, η2 = .22; see Figure 3. A simple contrast analysis revealed that for day 1 no significant effect, F(1, 28) = 0.70, P = .41, was obtained between the active (M = 52.93, SD = 25.87; 29.47 words out of 50) and sham stimulation group (M = 58.93, SD = 24.88; 26.47 words out of 50). Both at days 7 and 28, a significant effect (day 7: F(1, 28) = 7.49, P = .011 η2 = .21; day 28: F(1, 28) = 4.31, P = .041; η2 = .14) was obtained, indicating that the active stimulation group (day 7: M = 38.04, SD = 16.06, 19.02 words out of 50; day 28: M = 24.65, SD = 10.04, 15.33 words out of 50) correctly recalled more than the sham stimulation group (day 7: M = 30.66, SD = 12.12, 12.33 words out of 50; day 28: M = 18.27, SD = 9.90, 9.13 words out of 50).
ON-tES did not have an effect on processing speed when comparing active and sham simulation group immediately after learning the word, or 7 and 28 days after learning the words, F(2, 27) = 0.06, P = .95; see Figure 3.
We did not find a significant difference between the active and sham simulation group in terms of how many words participants forgot between day 7 and day 28, F(2, 27) = 0.09, P = .77; see Figure 3).
A positive correlation was obtained between correctly recalled words at day 7 and day 28 (R2 = .57, P < .001). Including only the participants that received active stimulation, a positive correlation was obtained between correctly recalled words at day 7 and day 28 (R2 = .68, P < .001), while for the sham stimulation group no effect was obtained (R2 = .14, P = .18). A comparison between the correlation outcome for the active and sham stimulation group shows a significant effect (Fisher Z = 1.96, P = .025). See Figure 3.
Saliva Samples
A comparison of α-amylase levels immediately after stimulation, 7 and 28 days after stimulation corrected for baseline showed a significant effect, F(3, 25) = 9.62, P < .001, η2 = .53. We saw a significant increase immediately after stimulation, F(1, 27) = 19.14, P < .001, η2 = .41; 7 days after stimulation, F(1, 27) = 18.56, P < .001, η2 = .41; and 28 days after stimulation, F(1, 27) = 8.44, P = .007, η2 = .23; for the active stimulation group in comparison to the sham stimulation group. No effect was obtained for cortisol, F(3, 25) = 0.91, P = .45, immediately after stimulation, 7 and 28 days after stimulation corrected for baseline. See Figure 4.
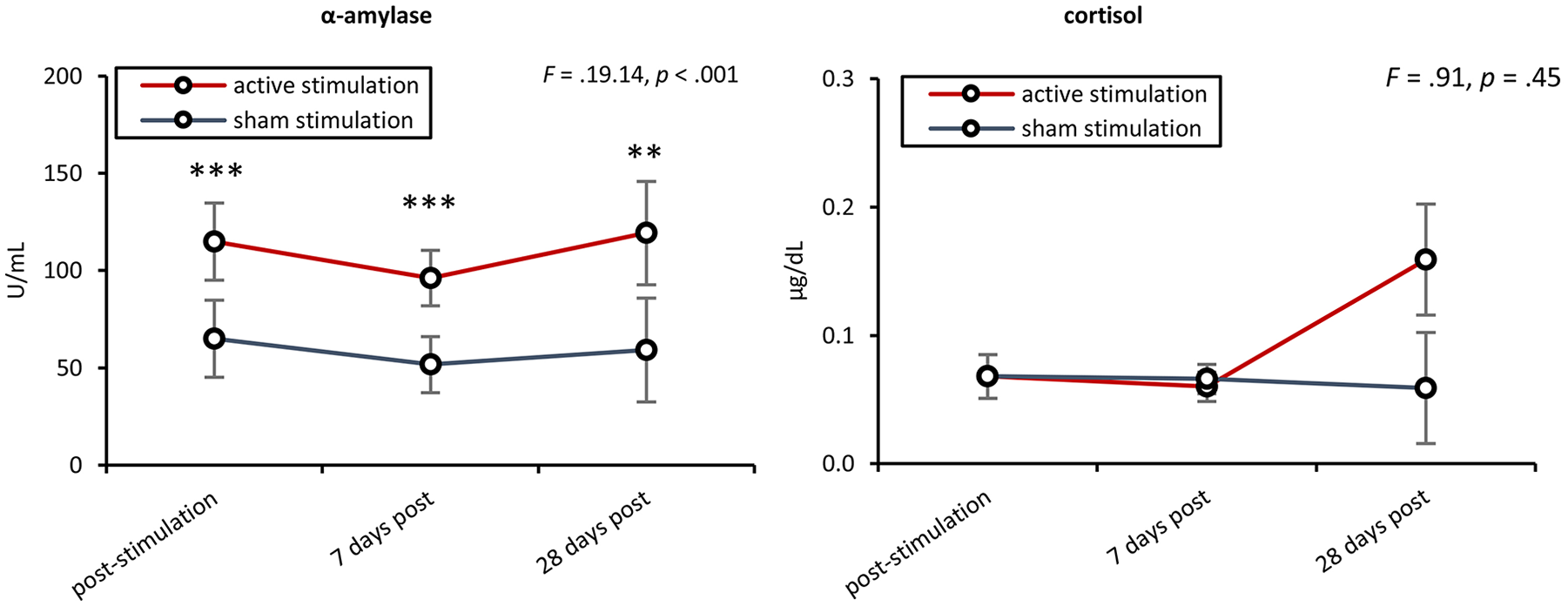
A comparison of α-amylase levels before stimulation with immediately after stimulation, 7 and 28 days after stimulation showed a significant effect. After baseline correction, we see a significant increase immediately after stimulation, 7 and 28 days after stimulation for the active stimulation group in comparison to the sham stimulation group. No effect was obtained for cortisol when comparing before stimulation with immediately after stimulation, 7 and 28 days after stimulation.
Blinding
Participants were not able to tell if they were assigned to the active or sham stimulation group, χ2(1) = 0.56, P = .81. In the active group, 36% of the participants anticipate perceiving sham stimulation, while 64% expected active stimulation. For the sham group, 40% of the participants anticipate perceiving sham stimulation, while 60% expected active stimulation. See Figure 5.
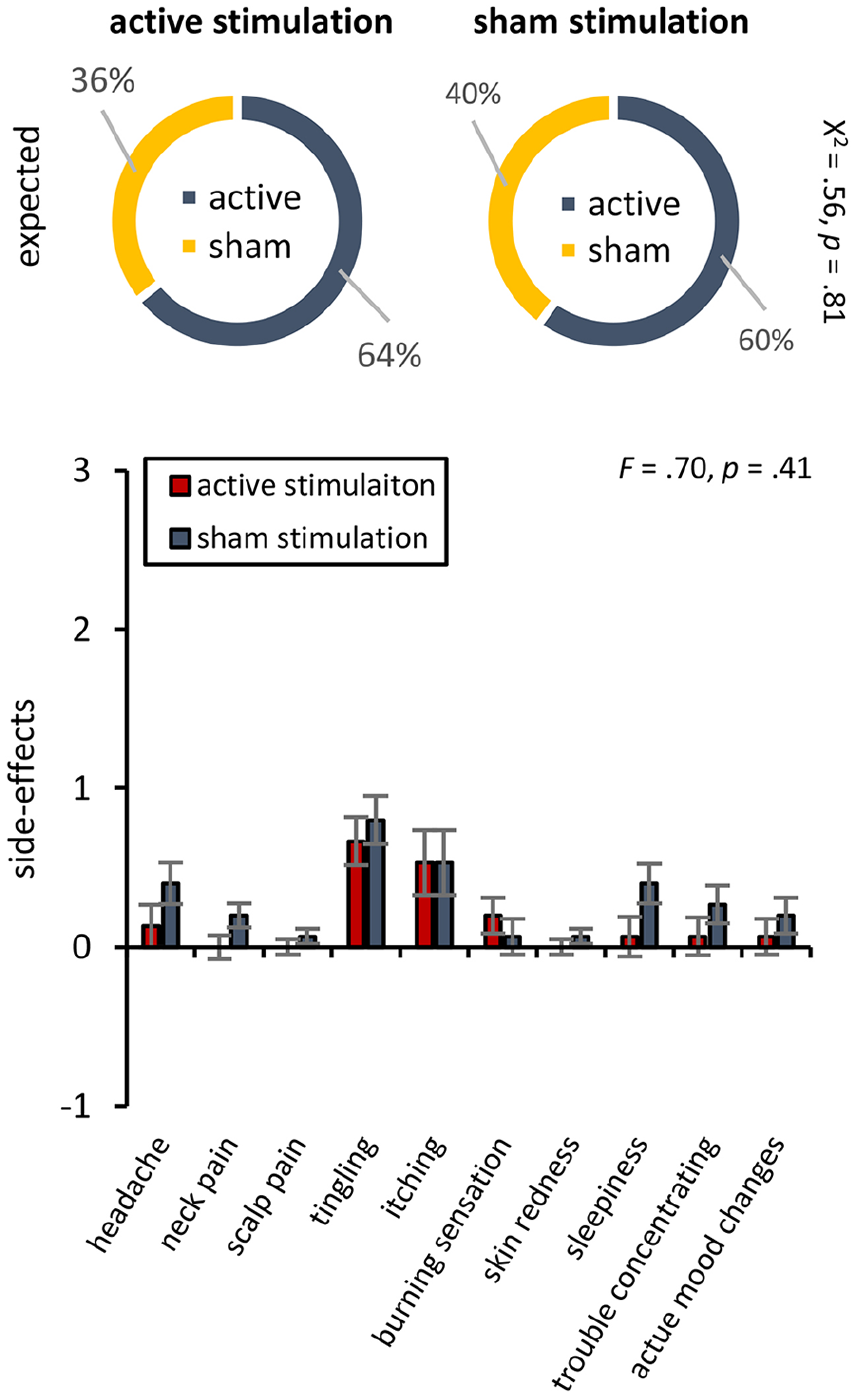
Participants were not able to tell if they were assigned to the active or sham stimulation group. No significant differences were demonstrated between active and sham stimulation group for side effects.
Adverse Events
No significant differences were demonstrated between the active and sham stimulation groups for side effects, F(11, 17) = 0.70, P = .40). No major adverse events were reported.
Discussion
The objective of this study was to increase our understanding of the effects of tDCS targeting the greater occipital nerve during an associative memory task, and to determine if tDCS may be used as a way to upregulate memory in healthy older persons via activation of the LC-NA pathway. Our results provide evidence that ON-tES can enhance memory in older individuals after one session with an effect up to 28 days after stimulation.
Our study revealed that ON-tES has no effect on learning as there was no difference between the active and sham condition during the learning phase, but differences did appear 7 and 28 days after learning the word-association task. Furthermore, we do not see changes in processing speed between the active and sham ON-tES during the leaning phase at day 1 or during the test phase on day 7 or 28. Taking these findings together, suggests that ON-tES works on the memory system, rather than generating a more general arousal effect, however, a more direct measure of attention and arousal should be utilized in the future to further validate our hypothesis. Previous research in animals and humans has already shown that ON-tES performed immediately after the training/encoding phase can be used to avoid learning-/performance-related issues such as sensation, or motivation, while also generating a memory effect. This is in line with our findings that ON-tES has an effect on memory consolidation and fits with the idea that neural processes underlying memory become consolidated after learning. 34
The neural mechanism by which ON-tES modulates memory is believed to include the LC-NA pathway. The locus coeruleus (LC) is a small nucleus in the dorsal pons that exerts powerful effects on neural processing via secretion of the neuromodulator noradrenaline (NA). The LC-NA system is thought to act at the synaptic, cellular, microcircuit, and network levels in addition to playing an important role in facilitating cognitive functions such as learning and memory. 35 This neuromodulatory system is involved in task engagement and, based on neuronal properties revealed in animal models, LC is thought to act as a “network reset” signal that allows flexible reconfiguration of brain networks to adapt and respond appropriately to the current context. 36 Recent investigations have shown the modulatory influence of the LC-NA projection on synaptic plasticity in the hippocampus and basolateral amygdala, which are both involved in storage.35,37,38 Neuroimaging research has further shown that successful encoding is associated with enhanced functional connectivity between the amygdala and the hippocampus. 39 Cranial and spinal nerve activity modulate the nucleus of the solitary tract (NTS) and are responsible for bottom-up regulation of cortical gain, psychological arousal, as well as the neurobiological responses to environmental stimuli and stressors.17,35 Specific afferent activity can modulate noradrenergic neurons via direct projections from the vagus or greater occipital nerve to the NTS. 40 In response to this activation, NTS neurons influence central noradrenergic activity through direct synapses on neurons in the LC and from the LC to the hippocampus and basolateral amygdala. 40 Activation of the LC induces the release of NA activity both directly, via synapses on neurons in the LC, and indirectly, via connections linking LC to the amygdala and hippocampus. 40 Consistent with this, direct stimulation of the vagus nerve in patients with epilepsy augments memory formation through the LC-NA pathway.11,41 Less well known is the fact that another peripheral pathway, the greater occipital nerve, has similar effects to those of the vagus. 42 Both pathways influence bottom-up regulation of cortical gain, psychological arousal, and neurobiological responses to environmental stimuli and stressors via the LC-NA system.35,38 Importantly, the occipital nerve can be targeted noninvasively using tES. 43
Evidence suggests that the LC-NA pathway was activated through observing the changes in α-amylase immediately after ON-tES. α-Amylase levels have been shown to co-vary significantly with circulating NA levels. Human fMRI showed LC activity rising concomitantly with α-amylase levels during the viewing of emotionally arousing slides. 44 Although we assumed increased levels of α-amylase immediately after ON-tES, we were not expecting to see a change 7 and 28 days after ON-tES. A recent study stimulating the vagus nerve paired with specific stimuli activating the LC-NA pathway showed a generalized effect to other stimuli associated with the specific experience. 45 Furthermore, research has showed the arousal associated to a context can be modulated using vagus nerve stimulation. 46 Based on these findings, it is possible that ON-tES also generates a more generalized effect related to the context. Bringing subjects back to this context elevates NA levels, which is in turn reflected by an increase in α-amylase.
Interestingly, we do not see any change in cortisol levels over the different time points. α-amylase is a biomarker for the LC-NA system while cortisol changes are directly related to the hypothalamic-pituitary-adrenal axis (HPA). Both systems are related to each other, as corticotrophin releasing factor neurons of the hypothalamus sending fibers to the brainstem and LC represents a connection site between the HPA axis and the LC-NA system. 47 Previous research has already shown a positive association between cortisol and α-amylase. 47 However, this association was only obtained when a different time course was considered. It is known that α-amylase reacts more rapidly than cortisol to psychological stressors. 47 Changes in HPA axis activity are not detectable for several minutes after inducing a stress response. 48
This is the first study examining the memory enhancing potential of ON-tES in an older adult population; however, this study has some limitations. An important variable to examine in future studies are the length of stimulation, the number of sessions, and the long-term cognitive effects. It is also possible that some participants might have rehearsed the information between visits, influencing their performance. Additionally, a future study utilizing a larger sample size will be needed to further test our hypothesis as well as in a clinical population. Future research could explore the possibility of the involvement of other pathways including the serotoninergic, cholinergic and the dopaminergic pathway. Recent research already showed that vagus nerve stimulation activates the serotonergic, 49 cholinergic 50 and dopaminergic 51 pathway, in addition to the LC-NA pathway.
In conclusion, our results provide, for the first time, evidence that ON-tES can enhance associative memory performance in well-controlled older individuals after one session up to 28 days later. There were no significant or long-lasting adverse side effects observed during stimulation. While our data provide some evidence of LC-NA pathway activation, as seen in related alpha-amylase concentration changes, future research utilizing advanced imaging techniques need to be completed to further determine the mechanism by which ON-tES enhances memory and to establish if it is truly through the activation of the LC-NA pathway.
Supplemental Material
participant_flow_chart_figure – Supplemental material for Greater Occipital Nerve Stimulation Boosts Associative Memory in Older Individuals: A Randomized Trial
Supplemental material, participant_flow_chart_figure for Greater Occipital Nerve Stimulation Boosts Associative Memory in Older Individuals: A Randomized Trial by Alison M. Luckey, S. Lauren McLeod, Ian H. Robertson, Wing Ting To and Sven Vanneste in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors thank Dr David Pruitt for writing the software program related to the behavioral tasks; and all members of the Lab for Clinical and Integrative Neuroscience (Lab Clint) for their support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was sponsored by the Defense Advanced Research Projects Agency (DARPA) Biological Technologies Office (BTO) TNT program under the auspices of Dr. Doug Weber and Tristan McClure-Begley through the Space and Naval Warfare Systems Center, Pacific Grant/Contract No. N66001-17-2-4011, Darrell Royal Foundation, and the UT Dallas Faculty Research Initiative.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
