Abstract
Occipital stimulation in a small group of refractory chronic migraine and cluster headache patients has been suggested as a novel therapeutic approach with promising results. In an earlier study we have shown that a drug-induced block of the greater occipital nerve (GON) inhibits the nociceptive blink reflex (nBR). Now, we sought to examine the effects of low-frequency (3 Hz) short-time nociceptive stimulation of the GON on the trigeminal system. We recorded the nBR responses before and after stimulation in 34 healthy subjects. Selectivity of GON stimulation was confirmed by eliciting somatosensory evoked potentials of the GON upon stimulation. In contrast to an anaesthetic block of the occipital nerve, no significant changes of the R2-latencies and R2-response areas of the nBR can be elicited following GON stimulation. Various modes of electrical stimulation exist with differences in frequency, stimulus intensity, duration of stimulation and pulse width. One explanation for a missing modulatory effect in our study is the relatively short duration of the stimulation.
Keywords
Introduction
Functional interaction between the greater occipital nerve (GON) and the trigeminal system in the pathogenesis of primary headache has been discussed for > 50 years (1). The basis of this ‘referred pain’ is a convergence of trigeminal and occipital afferents in the trigeminocervical complex (2, 3). Electrical stimulation of the GON activates the ipsilateral dorsal horn of the upper cervical roots as well as of the ipsilateral trigeminal nucleus caudalis (TNC) (4), whereas trigeminal stimulation leads to activation of upper cervical structures in cats (5). Clinically, suboccipital stimulation significantly improved chronic migraine (6) and chronic cluster headache (7–9). As our group found that the nociceptive blink reflex (nBR) was inhibited after ipsilateral GON block with prilocain (3, 10), we were interested whether nociceptive electrical GON stimulation would also modulate the trigeminal system.
Subjects and methods
Subjects
Thirty-four healthy subjects (17 male, 17 female, age 18–37 years, mean age 25.1 years,
Design
Response areas (AUC) and onset latencies of the R2 components of the nBR were defined as the main outcome variables.
Electrophysiology
The nBR was elicited with a specific electrode by selective A-δ stimulation (11). A monopolar square-wave pulse of 200 µs was administered and the nBR was recorded six times every 5 s. The R2 latencies and AUC were calculated offline from averages of 6. Further details are given elsewhere (3). To stimulate selectively the GON, a bipolar electrode was placed halfway in the line between the mastoid process and the occipital protuberance. Single stimuli were administered moving the electrode along the previously defined line. When a maximum extension of a painful skin area to the ipsilateral forehead was perceived, two intracutaneous platinum electrodes were inserted. Repetitive GON stimulation was then performed with sub maximum intensity perceived as maximum discomfort but not yet unbearable (number of stimuli, mean 272,

Occipital somatosensory evoked potential (SEP). An example of the typical waveform after stimulation of the right greater occipital nerve is given (subject 29). The latencies are as follows: N13 13.0 ms; P19 22.4 ms; N27 28.4 ms.
Statistics
Results were stored digitally for off-line analysis. The differences of R2 latencies and R2 AUC before and after the intervention on both sides were compared by means of paired t-test using SPSS for Windows 11.0 (
Results
We found no significant change of R2 latencies or R2 AUC in either part of the study (occipital stimulation and no stimulation) (Table 1, Fig. 2). The evoked OSEP confirmed correct positioning of the electrode and thus specific stimulation of the GON.
Latencies and response areas (AUC) of the R2 component of nociceptive blink reflexes in 34 healthy subjects ipsi- and contralaterally
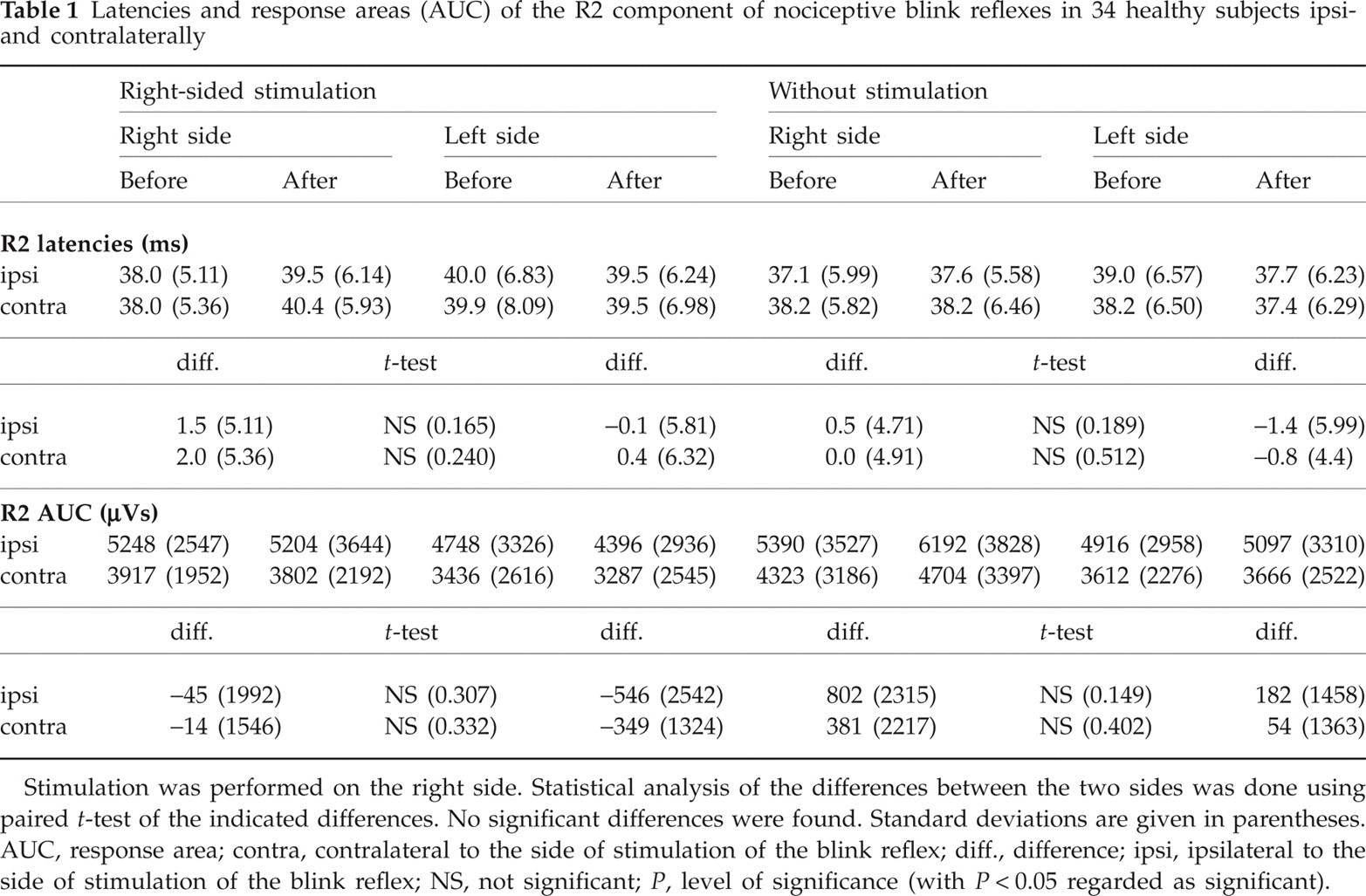



Stimulation was performed on the right side. Statistical analysis of the differences between the two sides was done using paired t-test of the indicated differences. No significant differences were found. Standard deviations are given in parentheses. AUC, response area; contra, contralateral to the side of stimulation of the blink reflex; diff., difference; ipsi, ipsilateral to the side of stimulation of the blink reflex; NS, not significant; P, level of significance (with P < 0.05 regarded as significant).

(a) Box plots demonstrate right-sided R2 latencies (ms) of nociceptive blink reflex (ipsilateral to the side of blink reflex stimulation) in 34 healthy subjects before and after low-frequency occipital stimulation on the right side or without stimulation. The results did not change significantly. (b) Box plots demonstrate right-sided R2-response areas (AUC) (relative units) of nociceptive blink reflex (ipsilateral to the side of blink reflex stimulation) in 34 healthy subjects before and after low-frequency occipital stimulation on the right side or without stimulation. The results did not change significantly. Box plots: the horizontal black line gives the median, the box the interquartile range (between upper and lower quartile). The whisker gives the maximum and the minimum (P < 0.05 regarded as significant).
The thresholds for sensory perception (I
S) and sensation of a pinprick-like pain (I
P) remained stable throughout the examination. Mean intensities were as follows: I
S on the right 0.29 mA (
In the second part of the study (no stimulation), mean intensities were as follows: I
S on the right 0.30 mA (
Although no serious side-effects occurred, two subjects reported a brief unilateral stabbing headache persisting for approximately 6 h, which started with a latency of some hours after the stimulation without any accompanying symptoms. In both subjects, the headaches remitted without need for pain medication. One subject recalled that he had suffered from approximately six migraine attacks with visual aura during adolescence with a positive family history, but had been headache free for > 12 years.
Discussion
The main finding of our study is that, in contrast to an anaesthetic block of the occipital nerve, stimulation of this nerve did not alter R2 AUC or R2 latencies of the nBR. Stimulation was performed only on the right side, as preceding studies had demonstrated no significant side differences (3). Since we recorded the cortical responses during occipital stimulation, we can exclude incorrect positioning of the stimulation electrode.
We intended to examine the effects of stimulating nociceptive occipital afferents on the trigeminal system (as seen in clinical practice, where neck pain and frontal pain often coincide). Raymund et al. have reported that in nociceptive afferents the conduction velocity slows with increasing stimulus frequencies (2 Hz up to 10 Hz), showing a tendency to conduction failure with increasing frequencies (12). To prove selective stimulation of the GON, we elicited an OSEP upon stimulation, for which a low frequency of 3 Hz is most suitable. The stimulus intensity was similar to that applied by Willer et al. (13), who examined the effect of painful but tolerable low-frequency (2 Hz) electroacupuncture (EA) of the hand on the blink reflex with intensities reaching up to 12 mA. Therefore, a frequency of 3 Hz was chosen.
One could argue that the relatively short duration of stimulation could explain the missing modulatory effect. EA in an inflammatory pain model in rats had better analgesic effects at 3 Hz than at 15 or 100 Hz. Fifteen and 60 min of stimulation at 3 Hz induced long-lasting analgesia, whereas 1 min failed to do so (14). In humans, Melzack (15) used a duration of 20 min, which has been used frequently since then. However, our primary goal was to elicit an immediate response. Willer et al. (16) used high-frequency low-intensity transcutaneous nerve stimulation (TENS) and observed modulatory effects on the BR within 2 min of stimulation. In the first exploratory tests prior to our study, the majority of participants indicated that they were reluctant to sustain much longer stimulation. Therefore, we chose a relatively short stimulation duration of approximately 90 s. It is known that low-frequency stimulation at 1 Hz can induce long-term depression (LTD) at the level of the spinal cord (17). However, given the time course and the experimental set-up, our study was not designed to examine LTD. Furthermore, at least on a spinal level, LTD is generally thought to be mediated homosynaptically and homosegmentally.
Electrical stimulation is used in TENS, EA as well as suboccipital stimulation in primary headache disorders (7–9). However, direct comparison between these studies and our experimental set-up is problematic, as we examined healthy volunteers. A general disposition to migraine or chronic pain may well influence the physiological reaction to presented stimuli (18). Apart from these constitutional differences, temporal alterations in nociception (e.g. allodynia in migraine) have been described (19, 20), which may have an influence on the therapeutic outcome. Furthermore, the majority of therapeutic studies used much higher frequencies (partly > 100 Hz). Although pain is reported as a side-effect, establishment of paraesthesia is usually intended to prove that suboccipital stimulation is effective. We can only speculate that therapeutic stimulation with its higher frequencies and less painful intensity is mediated by neuroplastic changes of the trigemino-nociceptive system, rather than exhibiting a direct effect on trigeminal transmission. However, as stimulation of structures other than the GON has been shown to modulate the BR also (13, 16, 21), it would be interesting to know whether the therapeutic effects of electrical stimulation are confined to the occipital system, or whether the trigeminal system should be the primary target.
In this context, it is noteworthy that in cluster headache (7, 8), but not in migraine (6), stimulation had to be maintained for weeks to months before clinical effects were seen. In one pilot study of eight patients with chronic cluster headache the nBR response area increased significantly after 1 month of suboccipital stimulation (8). Whereas effects of short-term stimulation would rather favour adjacent structures at a similar level (thus afferent nerves and their nuclei in the brainstem or spinal cord), long-term stimulation could modulate more central structures (e.g. those belonging to the ‘pain matrix’).
Interestingly, two of our volunteers suffered from short episodes of unilateral headache ipsilateral to the side of stimulation with delayed onset after the stimulation. The hemicranial location of the headaches points to an effect on both the occipital and the trigeminal system. One subject recalled that he had suffered from approximately six migraine attacks with aura > 12 years ago. One could hypothesize that stimulation could evoke some central wind-up mechanisms that eventually lead to these headaches in subjects with a predisposition to primary headache.
Footnotes
Competing interests
None to declare.
Acknowledgements
The authors thank T. Schneider, E. Schoell and H. Kaube for technical support. The study was supported by a grant of the University of Regensburg (ReForM-A) to V.B.
