Abstract
Background. People with Alzheimer disease (AD) are capable of new learning when cognitive support is provided, suggesting that there is plasticity even in a degenerating brain. However, it is unclear how a cognition-focused intervention operates on a neural level. Objective. The present study examined the effects of cognitive rehabilitation (CR) on memory-related brain activation in people with early-stage AD, as measured by functional magnetic resonance imaging (fMRI). Methods. A total of 19 participants either received 8 weeks of CR treatment (n = 7) or formed a control group (n = 12). We scanned participants pretreatment and posttreatment while they learned and recognized unfamiliar face-name pairs. Results. Following treatment, the CR group showed higher brain activation during recognition of face-name pairs in the left middle and inferior frontal gyri, the left insula, and 2s regions in the right medial parietal cortex. The control group showed decreased activation in these areas during recognition after the intervention period. Neither group showed an activation change during encoding. Behavioral performance on face-name learning did not improve for either group. Conclusions. We suggest that CR may have operated on the process of recognition through partial restoration of function in frontal brain areas that are less compromised in early-stage AD and that physiological markers may be more sensitive indicators of brain plasticity than behavioral performance.
Introduction
Alzheimer disease (AD) is clinically characterized in the early stages by progressive impairments in episodic memory but also affects other cognitive domains such as working memory and visuospatial ability. 1 On a neuronal level, the disease process is associated with neurofibrillary pathology and atrophy extending from the entorhinal cortex into the hippocampal area, temporal and insular regions, and the anterior and posterior cingulated.2,3 This causes deficits in the exchange of information between parietal secondary association areas and limbic and subcortical areas (hippocampus, amygdala, and thalamus) and leads to reduced input of the limbic loop to the prefrontal cortex (PFC). 4
Typically, people with early-stage AD are capable of some new episodic learning when appropriate cognitive support is given.5,6 Strategies such as mnemonic imagery, 7 semantic elaboration, 8 and spaced retrieval 9 have enhanced memory performance in people with AD. Building on these findings, a series of studies provided individualized interventions for people with AD, using combinations of strategies to teach personally relevant information or achieve changes in behavior. Some of these reports have focused on relearning of previously known information.10,11 Others have demonstrated successful new learning and suggested that this was facilitated by targeting residual explicit memory capability.12,13 This is significant because the ability to form an association between 2 previously unrelated items in memory is especially compromised in AD.14,15
The findings from these studies have led to the development of a cognitive rehabilitation (CR) approach for people with early-stage AD. 16 CR is a cognition-focused intervention where people with cognitive impairments and their families work together with a therapist to tackle individually relevant goals. 17 The application of this approach for people with dementia focuses primarily on individualized rehabilitation goals but also includes instruction and practice in the use of memory strategies and strategies to help maintain attention and concentration, stress management, and the implementation of practical compensatory strategies and memory aids. 16
The evidence that people with dementia can (re)learn information supports the possibility that neural plasticity can operate even in a brain affected by a degenerative disorder. Functional magnetic resonance imaging (fMRI) has been used to study the effects of neuroplasticity, for example in rehabilitation of reading ability in people with traumatic brain injury18,19 and working memory training in people with schizophrenia.20,21 Clare and colleagues 22 also reported changes in brain activity alongside behavioral improvement in a case study of CR for a patient with mild cognitive impairment. However, to our knowledge no study has yet reported neural changes following cognitive intervention in AD on a group level. The paucity of studies examining the relationship between neural change and therapy effects seems remarkable because the identification of biological markers of treatment is important in order to understand more about the neural mechanisms that underlie nonpharmacological interventions.23,24
Altered patterns of brain activity have been frequently reported in cross-sectional studies comparing people with AD to a healthy control group,15,25-30 but different explanations have been proposed to account for this finding. First, it has been proposed that the degree of damage to a neural system determines the behavior of this system. 31 A mildly dysfunctional neural system may be characterized by decreased processing efficiency coupled with (almost) intact processing capacity. In this scenario, the reduced activation in AD is a result of decreased processing efficiency, but partial restoration of neural function is possible. Conversely, several studies have suggested that altered patterns of brain activation in AD compared with healthy ageing compensate for loss of function in the medial temporal lobe.29,30,32 Indeed, Grady and colleagues 28 found that higher activation in the PFC was associated with better performance on an episodic retrieval task in people with AD. Finally, a third possible mechanism underlying the efficacy of cognitive intervention may be enhancing effective inhibition of task-irrelevant brain activation. Inhibition of task-irrelevant activation may be reduced in AD (and more generally in ageing). For example, older people with and without AD failed to inhibit effectively activity in the medial frontal and medial parietal areas that were deactivated during active task performance in young adults.33,34 Medial frontal and parietal areas are associated with a default-mode network that is active during internally focused tasks or “rest” relative to when a specific cognitive task is performed. 35 Failure to efficiently inhibit such brain areas during task performance may be a marker of preclinical AD and is present in cognitively normal individuals genetically at risk of AD.36,37
The current study focused on possible changes in brain activation patterns following a CR intervention in people with early-stage AD. We compared preintervention and postintervention neuroimaging data from a group of patients who received CR with data from a control group to detect the neural changes supporting CR.
Methods
Design
Participants in the current study were among those recruited to a randomized controlled trial (RCT) comparing the effects of CR with relaxation therapy (RT) and no treatment (NT). 38 Here, we present a detailed account of the neuroimaging data for those participants who consented to an fMRI scan prior to and following treatment (n = 19). To increase statistical power, the fMRI data from the RT and NT groups were collapsed to form 1 control group. The RT and NT groups did not differ significantly in demographic variables and behavioral performance.
Although RT is claimed to have a positive effect on behavioral difficulties in people with AD 39 and may even improve word fluency and performance on a short-term memory task, 40 it does not directly target memory function, and there is no clear-cut evidence that it improves memory function in daily life situations.
Participants
Participants were recruited through memory clinics of the National Health Service in North Wales and were diagnosed by independent clinicians as meeting NINCDS-ADRDA criteria for probable AD. 41 All participants were on a stable dose of acetylcholinesterase-inhibiting medication for at least 3 months prior to participation (donepezil 5-10 mg/d, galantamine 16-24 mg/d, or rivastigmine 6-12 mg/d). Inclusion criteria for the RCT were a diagnosis of AD or mixed dementia (vascular and AD) in the absence of any other neurological disease and a score of 18 or above on the Mini Mental State Examination (MMSE). 42 Participants were excluded if there was evidence of current or past history of psychiatric illness or stroke. For the fMRI part of the study, further exclusion criteria comprised dyslexia, poor vision that could not be corrected to normal, and any other conditions generally known to be incompatible with fMRI.
In all, 19 participants were both willing and able to have a scan before and after the treatment period; 7 participants received an 8-week individualized CR intervention. A control group comprising 12 participants received either the placebo RT (n = 5) or were in the NT group (n = 7). Experimental procedures for the RCT and for the fMRI part of the study were approved by the research ethics committees of the School of Psychology, Bangor University, and of the North West Wales National Health Service Trust.
Measures
Behavioral measures
The Canadian Occupational Performance Measure (COPM) 43 served as the primary behavioral outcome measure. Participants identified up to 5 personally relevant goals relating to self-care, household tasks, and leisure. Participants rated their performance and their satisfaction on each goal prior to and following the intervention period. Prior to the intervention period, participants also completed the Hospital Anxiety and Depression Scale (HADS). 44
fMRI paradigm
In a blocked design, participants studied and recognized unfamiliar face-name pairs. They also indicated whether or not a written sex label (male/female) correctly matched a face presented concurrently (see van Paasschen et al 24 ). Two task versions were created; in each version, for encoding and recognition, 12 gray scale pictures of white faces (6 men and 6 women) were paired with common British first names and shown over 6 runs. In each run, 2 stimuli (either 2 male or 2 female faces) were presented 3 times, resulting in 6 encoding and 6 recognition trials per run. For the control task, 12 other faces (6 men and 6 women) were shown with the word “Male” or “Female” printed below each picture. One male and 1 female stimulus were presented 3 times, so that there were 6 control trials per run. Each stimulus was presented for 5800 ms with a 200-ms interstimulus interval. Each condition was separated by a fixation cross shown for 16 000 ms. Instructions were given prior to each of the 3 conditions and presented for 18 000 ms. All participants received a 15-minute practice session (using different stimuli) outside the scanner prior to the start of the experiment. During encoding, participants were instructed to try to learn which faces and names belonged together. In the recognition phase, participants were asked to indicate via a yes/no button press whether the face and name had been paired during the encoding phase. During the control task, participants indicated via a yes/no button press whether the combination of face and sex was correct. The control task was always administered last to reduce task switching effects. Figure 1 illustrates the paradigm.
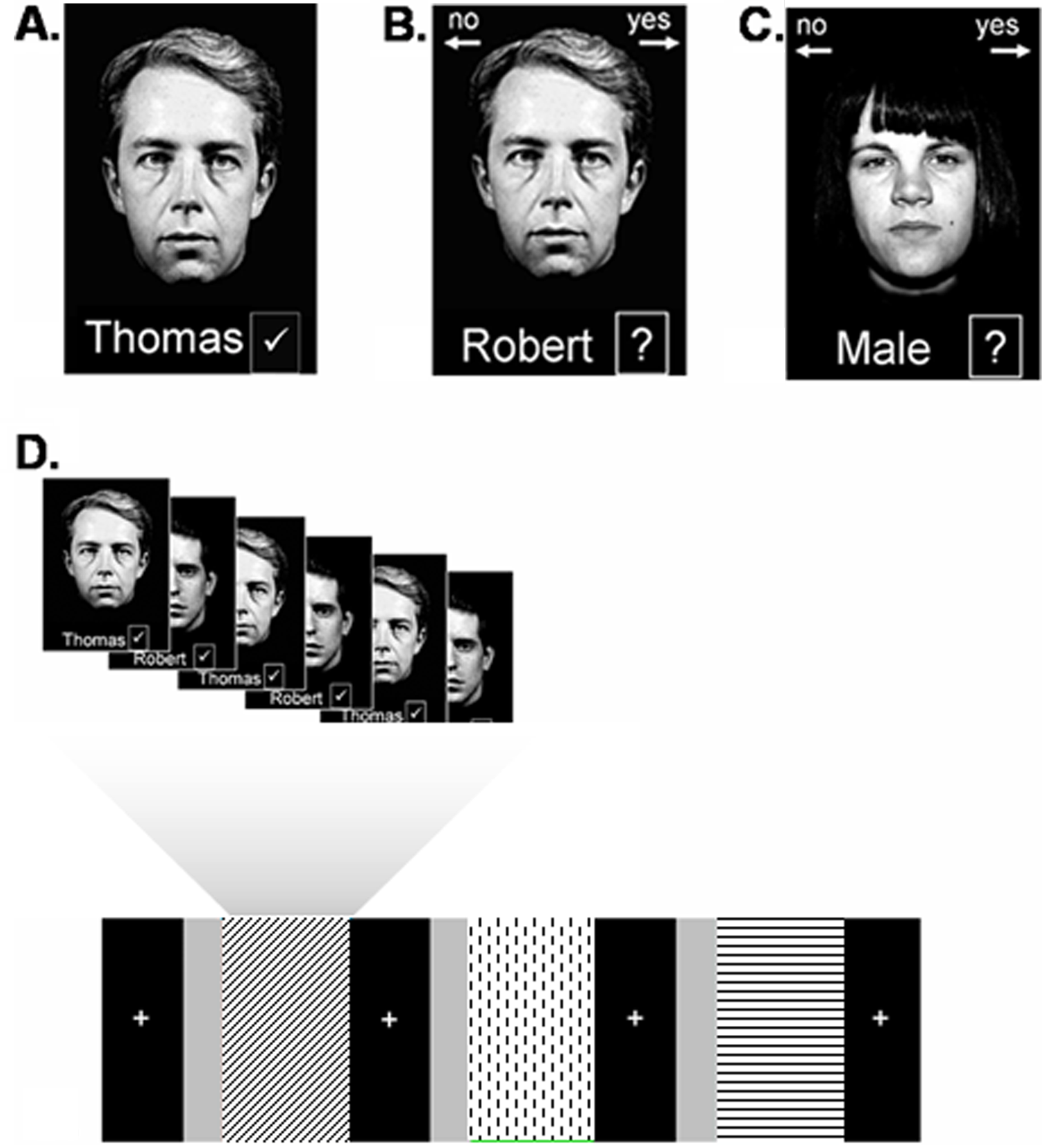
The experimental paradigm: the top row shows stimuli as shown during encoding (A), recognition (B), and the control task (C). Below is a schematic representation of 1 run (D), including encoding (diagonal stripes), recognition (dotted), and the control task (horizontal stripes). The uniform gray bars represent the instructions given prior to each condition. The black bars represent blocks where a fixation cross was shown.
Intervention
The CR intervention was tailored to each participant’s personal difficulties in daily life as identified by the COPM. One or 2 rehabilitation goals were selected to work on during the intervention. Participants received 8 weekly 1-hour sessions of CR with a therapist and practiced between sessions. Participants were introduced to strategies for acquiring new information, including verbal and visual mnemonics, semantic elaboration, and expanding rehearsal. All participants initially learned and practiced all 3 strategies and then chose 1 preferred strategy to implement in daily life. A detailed account of the CR intervention can be found in Clare 16 and Clare et al. 38
Image Acquisition
All participants were scanned using a 1.5-T Philips scanner with a head coil for parallel imaging. Foam padding was used to reduce head motion. Functional images were obtained with a T2*-weighted gradient echo sequence (TR [repetition time] = 2000 ms, TE [time to echo] = 40 ms, flip angle = 90°, FOV [field of view] = 192, 20 axial slices, 64 × 64 in-plane matrix, voxel dimensions = 3 × 3 × 5 mm3). A 3D anatomic T1 scan consisting of 150 slices (resolution 1 × 1 × 2 mm3) was obtained for coregistration with functional data. The time per run was 3 minutes 46 s for the experimental task (113 volumes).
Data Analysis
Functional MRI data were preprocessed and analyzed using BrainVoyager QX (Brain Innovation, Maastricht, Netherlands). We discarded the first 2 volumes of each run to avoid differences in T1 saturation. Motion correction was carried out on all images. We applied spatial smoothing using a 4-mm FWHM (full width at half maximum) Gaussian kernel and temporal smoothing with a Gaussian kernel of 2.8 s FWHM to remove high-frequency fluctuation. We then manually coregistered the functional data with the 3-dimensional anatomic scans. These were resampled to isometric 3 × 3 × 3 mm3 voxels with trilinear interpolation. A Talairach transformation was applied to the 3D scans. 45 We then applied the coordinates resulting from this transformation to the coregistered functional data and resampled these to 1 × 1 × 1 mm3 voxels. We modeled correlates for encoding and recognition by convolving all blocks with a standard 2γ model of the hemodynamic response function. The exact shape of this function is provided by the “hrf” function, available in the BrainVoyager MATLAB toolbox (BVQXtools). We used a random-effects analysis to create whole-brain group activation maps to compare the 2 groups. Because our main interest was in the effects of treatment on memory-related brain activity, we entered the fMRI data into a 2 × 2 × 2 mixed analysis of variance (ANOVA) with Time (preintervention; postintervention) and Task (encoding, recognition) as within-subject factors and Group (CR, control) as a between-subject factor. Voxels that exceeded a height threshold of F(1, 17) = 10.384, P < .005, and an extent threshold of 135 mm3 were considered significant. Activated clusters were corrected for multiple comparisons at the cluster level P < .05 using a Monte Carlo simulation with 1000 permutations 46 as implemented in BrainVoyager QX.
We carried out an additional exploratory event-related analysis comparing brain activity for hit and miss responses during recognition (see supplementary data).
Behavioral data were analyzed using SPSS version 15.0 for Windows. Changes in COPM scores and in scores on the face-name task before and after the intervention were explored using a repeated-measures ANOVA. Differences were considered to be significant at P < .05.
Results
To increase statistical power, in the present report, data from the RT and NT groups are collapsed to form 1 control group because neither group showed a change in score on the main outcome measure (COPM) following the treatment period. RT group: COPM performance, t(4) = 0.398, P = .711; COPM satisfaction, t(4) = 0.433, P = .687. NT group: COPM performance, t(6) = 0.540, P = .609; COPM satisfaction, t(6) = 2.145, P = .076.
Group characteristics for all participants, scores on behavioral outcome measures, and mean scores/standard deviations on the experimental tasks are presented in Tables 1 and 2.
Demographic Characteristics.
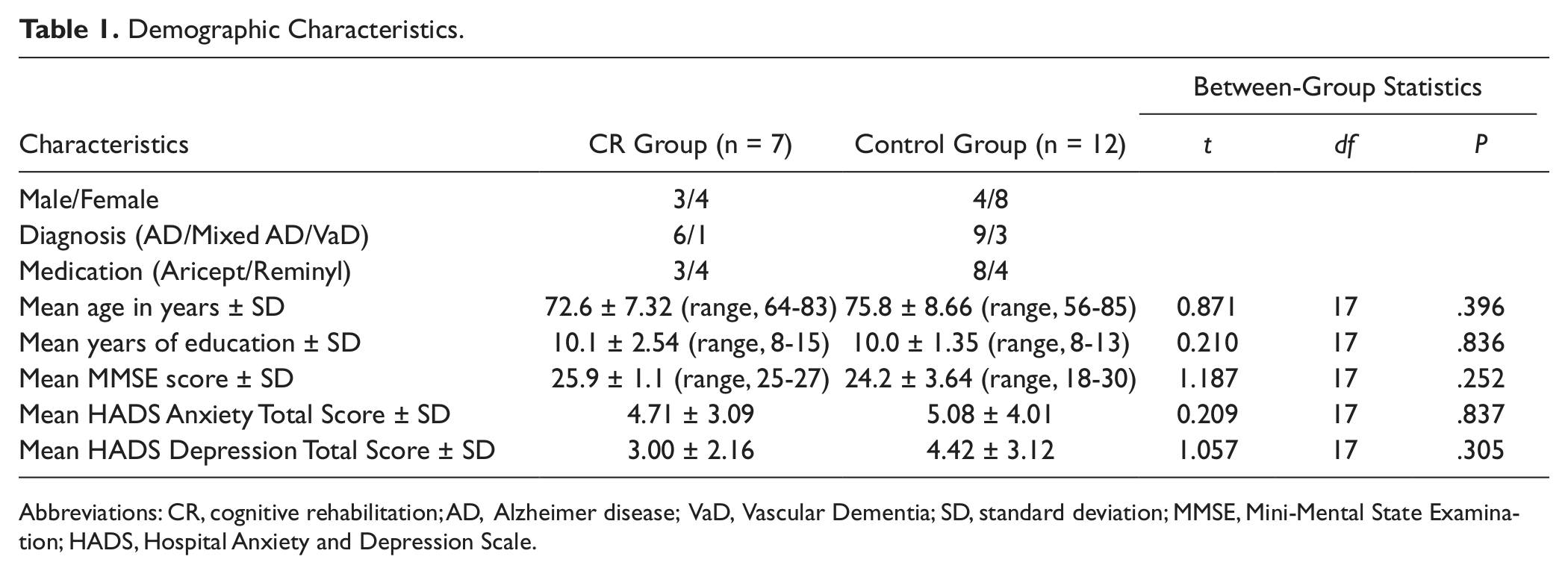
Abbreviations: CR, cognitive rehabilitation; AD, Alzheimer disease; VaD, Vascular Dementia; SD, standard deviation; MMSE, Mini-Mental State Examination; HADS, Hospital Anxiety and Depression Scale.
Mean Scores on the Behavioral Outcome Measures and the Experimental Paradigm.
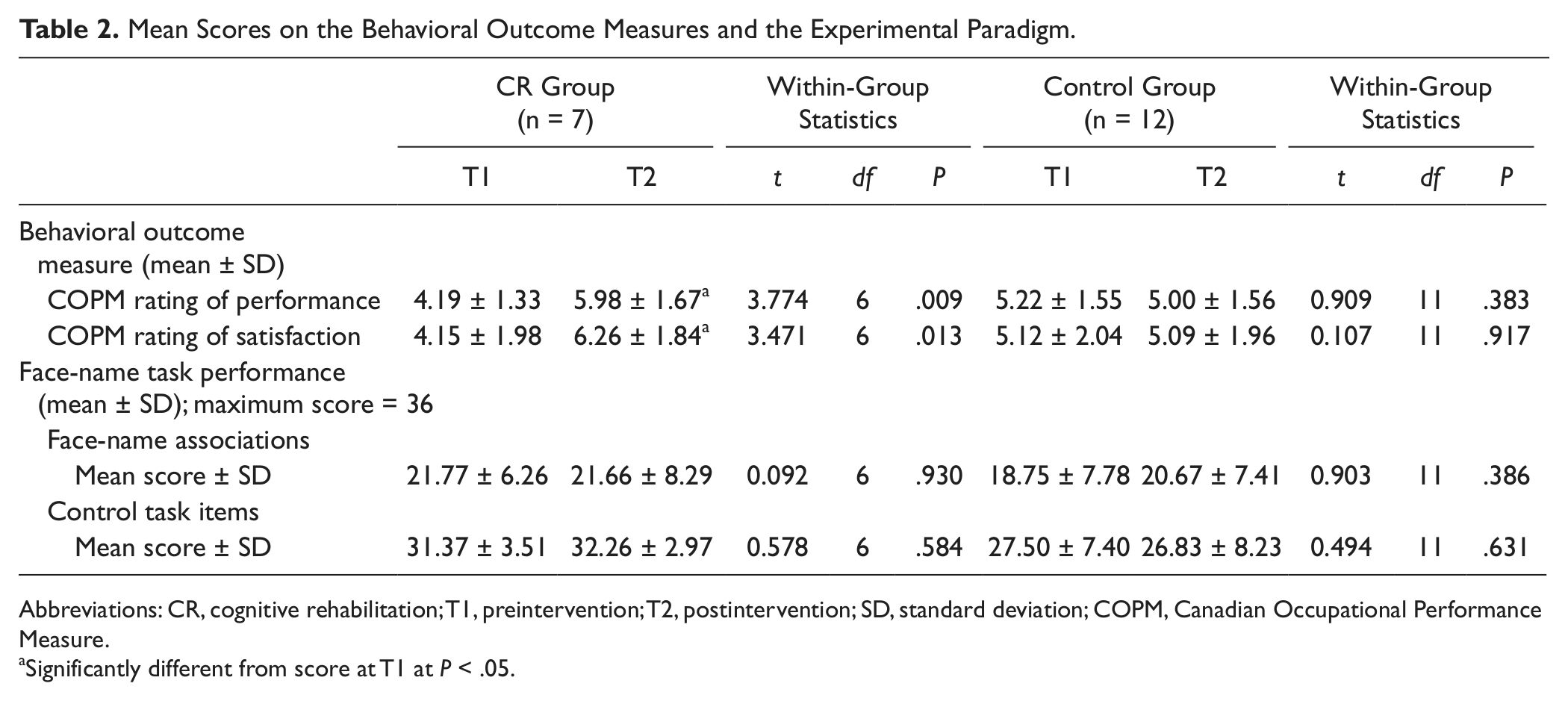
Abbreviations: CR, cognitive rehabilitation; T1, preintervention; T2, postintervention; SD, standard deviation; COPM, Canadian Occupational Performance Measure.
Significantly different from score at T1 at P < .05.
Group Characteristics
Independent samples t tests revealed no significant difference between the CR group and the control group in educational level—t(17) = 0.210, P = .836—MMSE score—t(17)=.1.187, P = .252—HADS depression and anxiety scores—t(17)=.1.057, P = .305 and t(17)=.209, P = .837, respectively—or age—t(17)=.871, P = .396.
Behavioral Outcome Measures
For the purpose of readability, in the following section, data collection prior to the start of the intervention will be referred to as T1, whereas data collection following treatment will be referred to as T2.
A multivariate ANOVA comparing the CR group and the control group on initial COPM scores showed no significant differences between the groups at baseline (performance score: F(1, 17) = 2.144, P = .161; satisfaction score: F(1, 17) = 1.019, P = .327). A repeated-measures ANOVA was carried out to detect changes in COPM performance and satisfaction scores over time, using group (CR group, control group) as the between-subjects factor and time (T1, T2) as the within-subjects factor. A Group × Time interaction showed that at T2, performance, and satisfaction ratings on the COPM had increased for the CR group while remaining unchanged in the control group: F(1, 17) = 16.467, P = .001. Follow-up paired t tests showed that in the CR group, scores for both COPM performance—t(6) = 3.77, P = .009—and satisfaction—t(6) = 3.47, P = .013—improved following treatment, whereas no change was observed in scores in the control group for either performance—t(11) = 0.909, P = .383—or satisfaction—t(11) = 0.107, P = .917—following the intervention period. Moreover, the CR group scored significantly higher than the control group on postintervention ratings for both COPM performance—F(1, 16) = 11.35, P = .004—and satisfaction—F(1, 16) = 9.79, P = .007. These results are in keeping with the outcome of the whole trial, which showed improvement on the COPM in the CR group only. 38
Behavioral Data From the fMRI Paradigm
Overall, participants performed significantly above chance level (50%, or 18/36) on the face-name association task—mean score = 20.4 ± 7.3, t(37) = 2.064, P = .046—and on the control task—mean score = 28.9 ± 6.7; t(37) = 9.977, P < .001. This demonstrates that participants were able to concentrate, understand the written instructions, and give meaningful “yes” and “no” answers.
To investigate whether there was an improvement in performance on the face-name association task, scores for the face-name task at T1 and T2 were entered into a 2 × 2 mixed, repeated-measures ANOVA with Time (T1, T2) as a within-subjects factor and Group (CR group, control group) as a between-subjects factor. No significant interactions were found: F(1, 17) = 0.761; P = .395.
fMRI Data
A 3-way interaction between Time, Group, and Condition was found in 8 regions, including the left PFC and the left insula, and small areas in the right parietal cortex and right basal ganglia (see Figure 2 and Table 3). To further investigate the nature of this interaction, mean β values were extracted from these clusters and entered into SPSS. Paired-samples t tests were conducted to compare the difference between the β values for T1 and T2 in each area, under each condition, for each group separately.
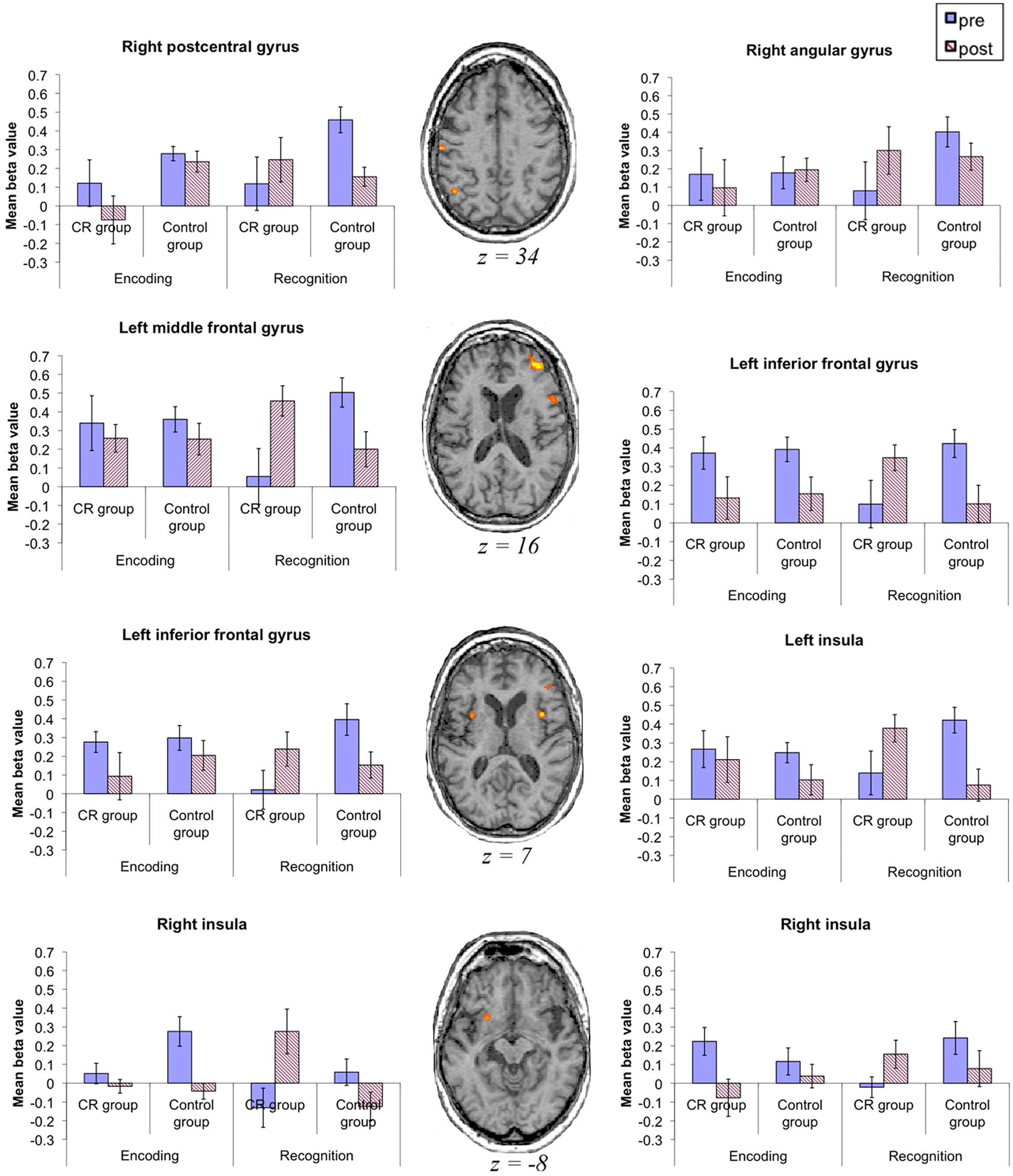
Areas showing a Time × Group × Task interaction: all brain slices are shown in the transversal plane using radiological convention (ie, right side of the brain is on the observer’s left and vice versa). Graphs show β values for each group in encoding and recognition preintervention and postintervention. Error bars indicate the standard error of the mean. Abbreviation: CR, cognitive rehabilitation.
Areas Showing a Time × Group × Task Interaction. a
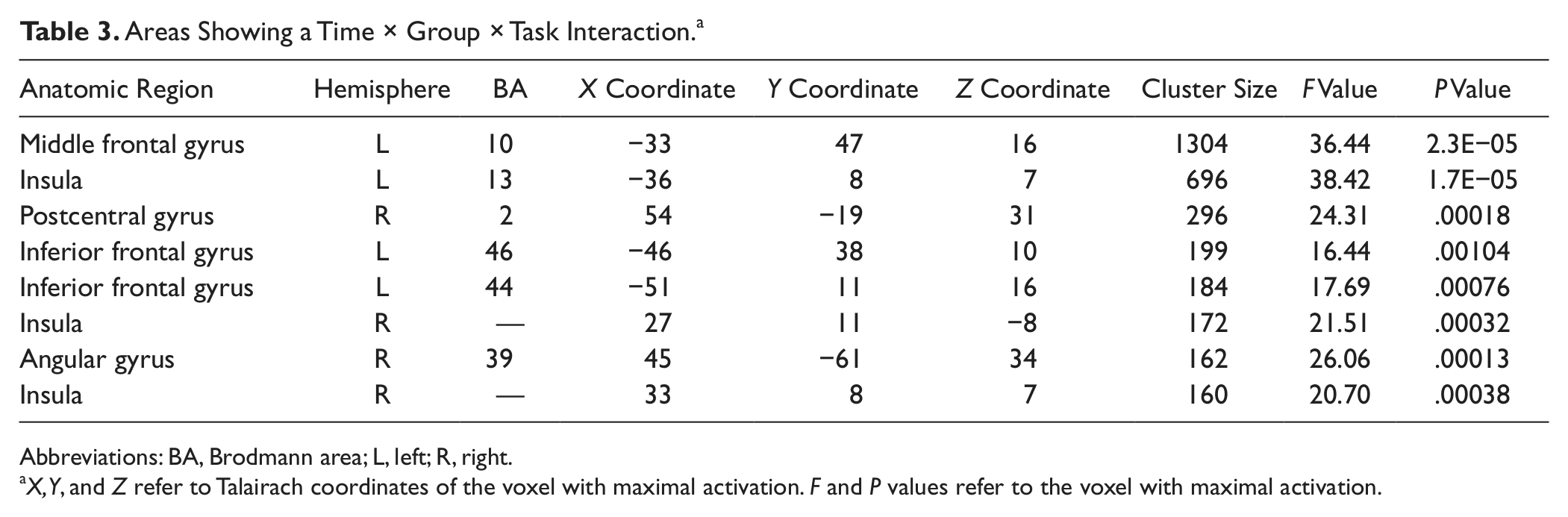
Abbreviations: BA, Brodmann area; L, left; R, right.
X, Y, and Z refer to Talairach coordinates of the voxel with maximal activation. F and P values refer to the voxel with maximal activation.
During encoding, the CR group showed no significant change in brain activation in any of the relevant brain regions—t(6) < 1.788, P > .124 for all—apart from a decrease in activity at T2 in the right insula—t(6) = 5.047, P = .002. Similarly, there were no significant alterations in brain activation in the control group—t(11) < 1.643, P > .129 for all relevant areas—except for a decrease in the left inferior frontal gyrus (BA 44) at T2: t(11) = 2.83, P = .016.
However, during recognition, the CR group showed a significant increase in activation at T2 compared with T1 in the left middle frontal gyrus, bilateral insula, and angular gyrus—t(6) ≥ 2.587, P < .041 for all—and there was a trend toward this pattern in 2 areas in the left inferior frontal gyrus: BA 44/46, t(6) = 2.268, P = .064; t(6) = 2.376, P = .055). The only exception was the right postcentral gyrus, where no significant activation change was observed: t(6) = 1.035, P = .341, NS. The control group, on the other hand, showed a significant activation decrease at T2 for all areas—t(11) > 2.218, P < .049—apart from 1 region in the right insula—t(11) = 1.77, P = .104, NS.
We also detected a Group × Time interaction (ie, irrespective of task) in bilateral prefrontal areas, the left basal ganglia, right posterior cingulated, and left cuneus (BA 7/31); see Figure 3 and Table 4. The interaction was mainly driven by a significant (P < .012 for all) decrease of activation at T2 in the control group in all areas presented in Table 4, whereas the CR group showed significant activation increase at T2 in the left superior frontal gyrus (BA 10) and right medial prefrontal and right middle frontal gyrus (BA 10/32), a cluster in the lentiform nucleus, and the left cuneus (BA 7; P < .040 for all).
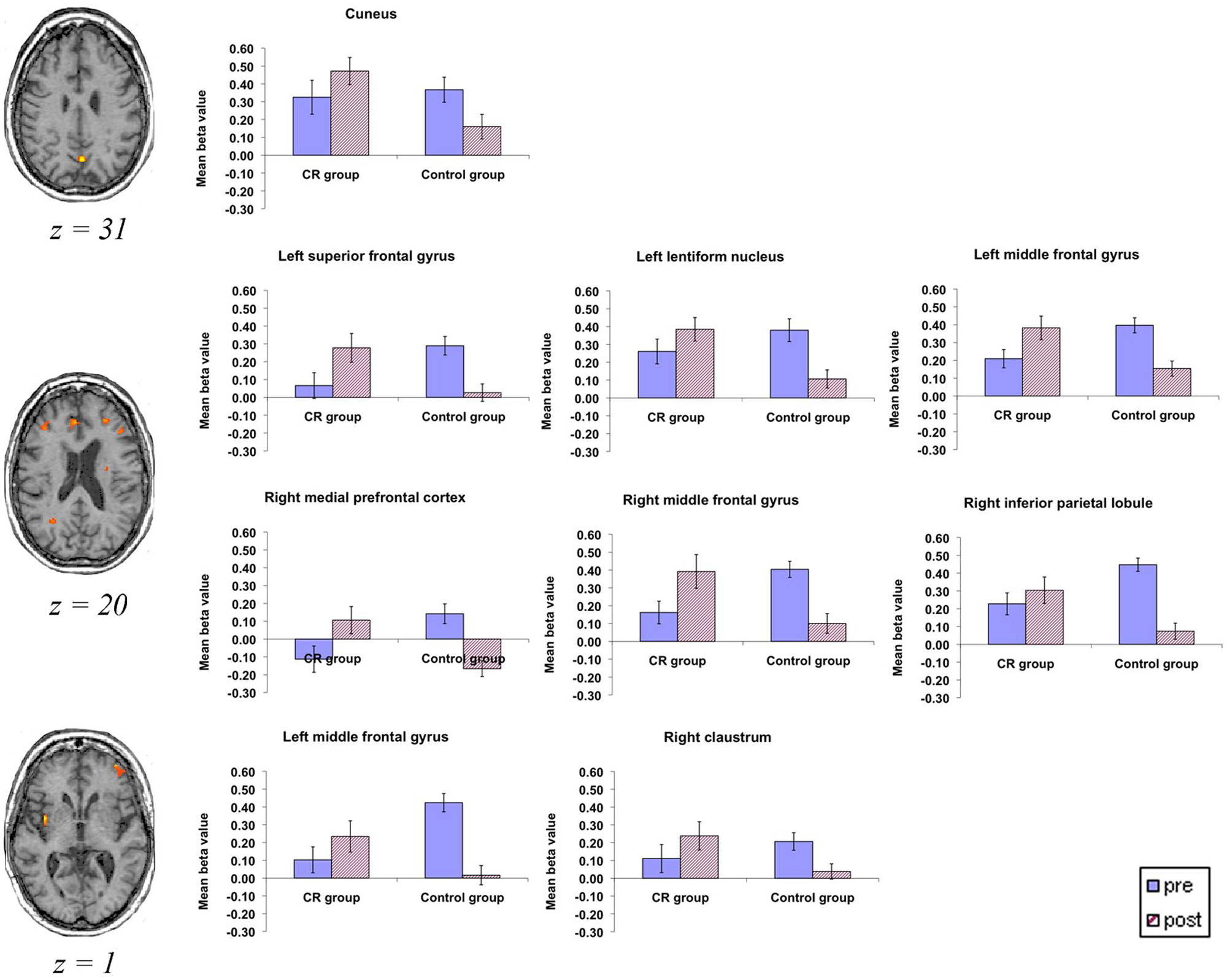
Areas showing a Time × Group interaction: all brain slices are shown in the transversal plane using radiological convention (ie, right side of the brain is on the observer’s left and vice versa). Graphs show β values for each group preintervention and postintervention in each area. Error bars indicate the standard error of the mean. Abbreviation: CR, cognitive rehabilitation.
Areas Showing a Time × Group Interaction. a
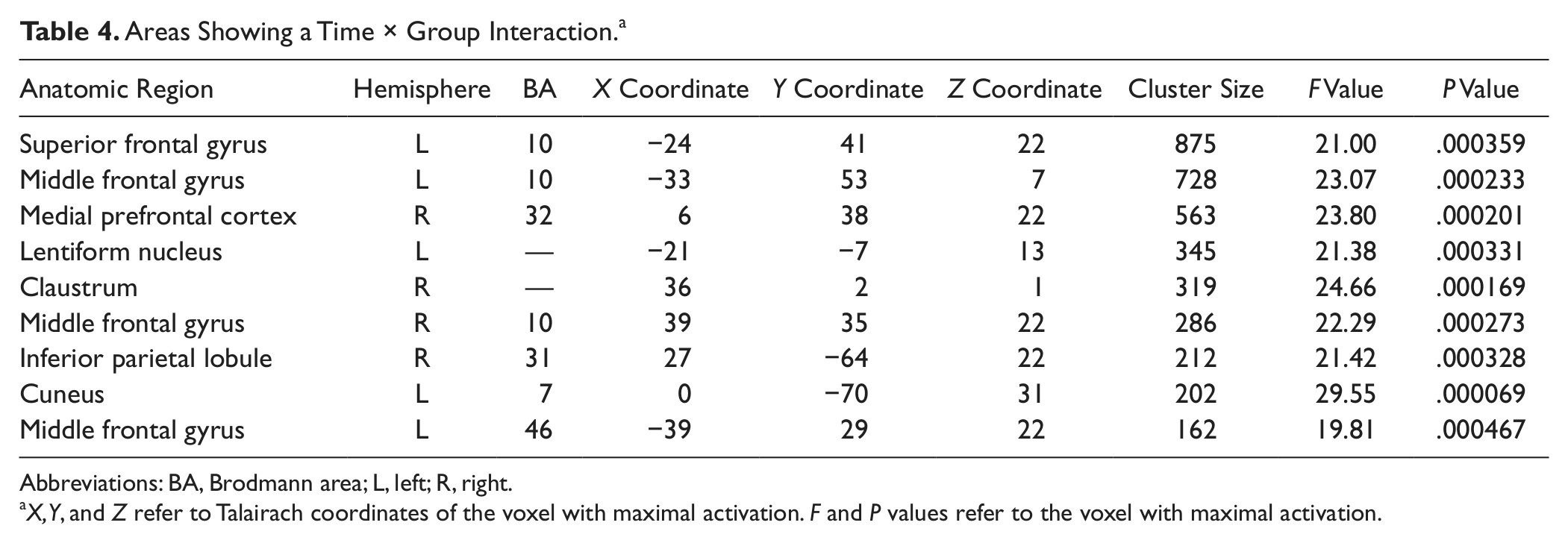
Abbreviations: BA, Brodmann area; L, left; R, right.
X, Y, and Z refer to Talairach coordinates of the voxel with maximal activation. F and P values refer to the voxel with maximal activation.
In our previous report, 38 we briefly discussed 4 areas in which Group × Time interactions were observed. Of note, these results were obtained using a liberal voxel-based threshold and without including Task as a factor in the statistical model. In the current report, we used the more conservative method of cluster correction and included Task as a factor in our ANOVA. Apart from the right medial PFC, the brain areas reported here as showing a Group × Time interaction differ somewhat from those discussed in the earlier article (ie, the right fusiform face area, right parahippocampal gyrus, and right temporo-parietal junction). However, the main pattern of findings is consistent with our previous report: there were more pronounced effects at recognition than encoding, with the CR group showing an activation increase and the control group showing a signal decrease during recognition posttreatment.
Third, we found a main effect of Time in several brain areas, including the bilateral PFC, basal ganglia, medial parietal cortex, middle temporal gyrus, and visual regions. The main effect of Time was driven by overall higher activation at T1 (mean β = 0.336 ± 0.120) compared with T2 (mean β = 0.118 ± 0.103). These activation decreases following the intervention conform to the changes observed in most training studies with younger adults. 47 It is important to note that in such studies, participants are not cognitively trained to perform a task differently; they are merely allowed to perform the task multiple times, so that they gain practice in the task at hand. Consequently, the expected pattern of activation is that of a decrease, thought to reflect the reduced demand for cognitive resources.
Finally, there was a main effect of Group in the left anterior cingulate and bilateral cerebellum. This effect was caused by generally higher activation in the CR group (mean = 0.182 ± 0.153) than in the control group (mean = 0.078 ± 0.420).
Discussion
The current study investigated brain activation changes during face-name learning in people with early-stage AD following an 8-week CR program. We found a differential response following the treatment period in each group: on the recognition task only, we observed increases in the CR group and decreases in the control group following treatment in bilateral prefrontal areas and in the bilateral insula. Behaviorally, performance on the face-name association task remained unchanged over time in both groups. However, the CR group but not the control group rated their performance and satisfaction on a range of everyday activities higher posttreatment.
The increased brain activity that we observed in the CR group following the intervention fits well with findings from previous studies that included participants with compromised memory function. We demonstrated enhanced brain activation in prefrontal and temporo-parietal regions in a single case study involving a patient with mild cognitive impairment (MCI) following 8 weeks of CR.22,24 Hampstead and colleagues 48 showed increased brain activity during face-name encoding in medial frontal, parietal, and occipital areas in 6 people with MCI following 3 sessions of explicit memory training. Task performance in both these studies also improved following the intervention. The sites showing increased activation form part of a default-mode network involved in self-initiated cognitive processes 49 that include episodic memory 50 and are also among regions typically affected in MCI and AD. 51 Such self-initiated processes are likely to occur when participants make use of the taught learning strategies to encode and recall information. Our finding of enhanced activation in predominantly left frontal cortices during the recognition task is suggestive of partial restoration of function. The left PFC has been associated with the successful use of a mnemonic to learn words in a group of older and young adults 52 and is thought to play a role in successful verbal recognition in young adults. 53
The clear differences in brain activation patterns and subjective ratings in the absence of objective performance differences between groups after the intervention suggest that physiological markers may be more sensitive indicators of brain plasticity than behavioral effects. Along similar lines, it has been argued that neurophysiological differences, as measured with functional imaging, may occur in early AD before the cognitive decline31,51,54,55 or in unaffected individuals with high genetic risk.56,57
Dissociation Between Encoding and Recognition
One important finding was the dissociation between encoding and recognition: there were small differences in brain activity during encoding (CR group: an activation decrease in the right insula at T2; control group: an activation decrease at T2 in the left inferior frontal gyrus). However, during recognition, the CR group showed an activation increase at T2 in a number of areas, including the left frontal gyrus, bilateral insula, and right angular gyrus. The opposite pattern was found in the control group. It is widely acknowledged that encoding processes are severely disrupted in early-stage AD, whereas recognition is only mildly affected in the first instance. 58 The lack of change in brain activity during encoding may indicate that CR was able to operate on recognition processes but did not reach the threshold needed to have an effect on encoding. It would be of interest whether a more focused, in-depth, or lengthy intervention would improve brain activity during encoding. Our study may thus be an example of a situation where imaging findings inform clinicians about the mechanisms of action of psychological interventions and help toward their refinement.
An exploratory event-related analysis (see supplementary data) suggested that in the left parahippocampal gyrus, the right insula, and the left superior temporal gyrus, the CR group showed increased activation for miss responses coupled with somewhat reduced brain activity for hit responses. These areas are all involved in recognition and task monitoring to some extent. Possibly, increased brain activity in the CR group for misses reflects greater conflict/increased effort to recall the original stimulus, perhaps while using a memory strategy participants learned during the intervention period.
Conclusion
This is the first study to use fMRI to directly compare memory-related brain activation pretreatment and posttreatment with CR in people with predominantly mild AD. Our results suggest that the CR intervention may have been able to operate on a partially intact recognition memory process; however, a more focused treatment may be required to target encoding. The use of functional imaging to monitor cognitive interventions may have a general benefit in detecting subclinical changes and providing cues for the further refinement of CR strategies.
Footnotes
Acknowledgements
We are grateful to Rhiannon Whitaker and Zoe Hoare for statistical advice, Tony Bedson for radiographer support, and Martijn van Koningsbruggen for help with programming.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by a grant from the Alzheimer’s Society (UK) to LC (PI), DEJL, RTW, and MDR. JvP was supported by a PhD studentship from the School of Psychology, Bangor University.
