Abstract
Background. Mesenchymal stem cell (MSC)-derived exosomes play a critical role in regenerative medicine. Objective. To determine the dose- and time-dependent efficacy of exosomes for treatment of traumatic brain injury (TBI). Methods. Male rats were subjected to a unilateral moderate cortical contusion. In the dose-response study, animals received a single intravenous injection of exosomes (50, 100, 200 µg per rat) or vehicle, with treatment initiated at 1 day after injury. In the therapeutic window study, animals received a single intravenous injection of 100 µg exosomes or vehicle starting at 1, 4, or 7 days after injury. Neurological functional tests were performed weekly after TBI for 5 weeks. Spatial learning was measured on days 31 to 35 after TBI using the Morris water maze test. Results. Compared with the vehicle, regardless of the dose and delay in treatment, exosome treatment significantly improved sensorimotor and cognitive function, reduced hippocampal neuronal cell loss, promoted angiogenesis and neurogenesis, and reduced neuroinflammation. Exosome treatment at 100 µg per rat exhibited a significant therapeutic effect compared with the 50- or 200-µg exosome groups. The time-dependent exosome treatment data demonstrated that exosome treatment starting at 1 day post-TBI provided a significantly greater improvement in functional and histological outcomes than exosome treatments at the other 2 delayed treatments. Conclusions. These results indicate that exosomes have a wide range of effective doses for treatment of TBI with a therapeutic window of at least 7 days postinjury. Exosomes may provide a novel therapeutic intervention in TBI.
Keywords
Introduction
Traumatic brain injury (TBI) affects more than 10 million people annually, leading to mortality and hospitalization worldwide. 1 To date, no effective neuroprotective agent has been identified from clinical TBI trials. 2 Multipotent mesenchymal stem cells (MSCs) are self-renewing stem/progenitor cells from the bone marrow, adipose tissue, skin, umbilical cord blood, and peripheral blood as well as other organs. 3 Previous studies show that a small proportion of transplanted MSCs migrate into the injured brain, of which only a small number displayed neural-like markers, indicating that functional recovery is not linked to neural differentiation from the transplanted MSCs.4,5 MSC-induced brain remodeling and functional recovery are associated with their secretion-based paracrine role.6,7 These therapeutic paracrine effects are likely mediated by MSC-generated exosomes. 8 In contrast to MSCs, nanosized exosomes can easily pass the blood-brain barrier and deliver their cargos, including genetic materials, lipids, and proteins to recipient cells 9 without vascular obstructive effect and risks of tumor formation. 10
MSC-derived exosomes significantly improve functional recovery in rats after TBI. 11 However, the dose- and time-dependent effects of MSC-derived exosomes on functional recovery after TBI remain elusive, which ultimately will determine their clinical utility. The present study was designed to investigate the dose- and time-dependent efficacy of exosomes after TBI in improving cognitive and sensorimotor functional recovery, amplifying neurovascular remodeling, and reducing neuroinflammation after TBI.
Materials and Methods
Animals
Adult (3-month-old) male Wistar rats weighing 339.2 ± 13.6 g (Charles River, Wilmington, MA) were used. All experimental procedures were approved by the Henry Ford Health System Institutional Animal Care and Use Committee. The persons who performed the experiments, collected data, and assessed outcome were blinded throughout the course of the experiments (see Supplemental Material for details).
Exosome Isolation and Characterization
Human MSCs were purchased from Theradigm (Bethesda, MD) and expanded, as previously described. 12 MSC-derived exosomes were collected according to the published methods. 13 Exosome size and particle number were characterized using a NanoSight (Merkel Technologies) instrument. Exosome morphology and exosomal markers (Alix and Hsp70) were examined by means of transmission electron microscopy and Western blot, respectively. Please see Methods in the Supplemental Material for details.
Animal Model of TBI
A well-established controlled cortical impact (CCI) rat model of TBI was utilized for the present study. Animals were anesthetized intraperitoneally with ketamine (80 mg/kg) and xylazine (13 mg/kg). Rectal temperature was maintained at 37 ± 0.5°C using a feedback-regulated water-heating pad. Rats were placed in a stereotactic frame. Two 10-mm diameter craniotomies were performed adjacent to the central suture, midway between lambda and bregma. The second craniotomy was bilaterally symmetrical to the first and performed in the contralateral skull allowing for lateral movement of cortical tissue. The dura mater was kept intact over the cortex. Cortical injury was delivered by impacting the left parietal cortex (ipsilateral cortex) with a pneumatic piston of a 6-mm diameter tip at a rate of 4 m/s and 2.5 mm of compression, as described in our previous study. 11 The cranial defect was not filled after the impact. This moderate TBI induced by CCI targets the parietal cortex and causes consistent cortical tissue loss (lesion cavity) and significant neuronal cell loss in the ipsilateral hippocampus as well as white matter damage. 2
Experimental Groups
In the dose-response experiment, the study animals were randomly divided into 5 groups (n = 8/group): (1) sham; (2) TBI + vehicle phosphate-buffered saline (PBS); (3) TBI + 50 µg exosomes per rat (Exo-50); (4) TBI + 100 µg exosomes per rat (Exo-100); and (5) TBI + 200 µg exosomes per rat (Exo-200). Exosomes generated from MSCs (50, 100, and 200 µg total protein of exosome precipitate in 0.5 mL PBS per rat or an equal volume of PBS [0.5 mL]) were administered intravenously over 5 minutes via the tail vein, starting 1 day after injury. TBI animals treated with PBS served as a treatment control group. Sham animals underwent surgery without injury and treatment.
In the therapeutic window experiment (n = 8/group), TBI rats received a single dose of 100 µg total protein of exosome precipitate in 0.5 mL PBS per rat via the tail vein, starting at 1, 4, or 7 days postinjury, respectively (designated as Groups D1, D4, and D7). TBI animals receiving 0.5 mL PBS served as a control group. Sham animals underwent surgery without injury and treatment.
For labeling proliferating cells, 5-bromo-2′-deoxyuridine (BrdU, 100 mg/kg) was injected intraperitoneally into all animals daily for 10 days, starting 1 day after TBI, as described in our previous study. 14
Evaluation of Neurological Outcome
Modified Neurological Severity Score (mNSS) 15 and foot faults 16 were performed on all rats preinjury and at 1, 7, 14, 21, 28, and 35 days after TBI. To measure spatial learning and memory impairments, the Morris water maze (MWM) test was performed for 5 consecutive days on days 31 to 35 after TBI, as described in our previous TBI study. 11 All neurological functional tests were performed by observers blinded to the treatment groups. Please see Methods in the Supplemental Material for details.
Tissue Preparation, Measurement of Lesion Volume, and Immunohistochemistry
On day 35 after TBI, anesthetized rats were sacrificed for collecting brains. Brains were embedded in paraffin, and a series of 6-µm-thick slides were cut. The cortical lesion volume was measured. CD 68 (marker for microglia/macrophages), glial fibrillary acidic protein (GFAP; marker for astrocytes), and neuronal nuclei (NeuN, for neurons) immunostainings were performed. Please see Methods in our previous study 12 and the Supplemental Material for details.
Immunofluorescent Staining and Cell Counting
Newly generated endothelial cells and newborn mature neurons 35 days after TBI were identified by double labeling for BrdU with endothelial barrier antigen (EBA) or NeuN, respectively. 11 EBA+ endothelial cells, CD68+ microglia/macrophages, GFAP+ astrocytes, and EBA/BrdU-colabeled cells were counted in the lesion boundary zone (LBZ) and the dentate gyrus (DG). 11 The LBZ is defined as the region of cortical tissue between the lesion cavity and the intact tissue (S1 Figure). For analysis of neurogenesis, we counted NeuN/BrdU-colabeled cells in the subgranular zone and granular cell layer of the DG. The fields of interest were digitized under the light microscope (Nikon, Eclipse 80i) at a magnification of either 200 or 400 using CoolSNAP color camera (Photometrics) interfaced with MetaMorph image analysis system (Molecular Devices). In brief, 5 fields of view in the LBZ from the epicenter of the injury cavity (bregma −3.3 mm) and 9 fields of view in the ipsilateral DG were counted in each section, as described in detail previously. 17 Please see Methods in the Supplemental Material for details.
Data Analysis
Data are presented as means with SDs and were tested for normality. When data were not normally distributed, ranked data or a nonparametric Kruskal-Wallis test was used. Analysis of variance (ANOVA) was used for repeated measurements of spatial performance and sensorimotor function. For cell counting, a 1-way ANOVA followed by post hoc Tukey tests were used to compare the differences between the exosome-treated, PBS-treated, and sham groups. Differences were considered significant if the P value was <.05.
Results
Identification of Exosomes From MSC Culture Medium
Using a qNano nanopore-based exosome detection system, we demonstrated that MSCs generate exosome-enriched particles (147.4 ± 62.6 nm in diameter, with a mode diameter at 122 nm in size), which is consistent with MSC exosomes identified with transmission electron microscopy (S2 Figure A). Western blot confirmed that exosomes contained high levels of exosome proteins markers Alix and Hsp70 compared with the supernatant (S2 Figure B).
MSC Exosome Administration Significantly Enhances Spatial Learning in Rats After TBI
In the dose-response study (Figure 1A), compared with the PBS group, post hoc Tukey testing demonstrated significantly increased time spent by TBI rats treated with exosomes in the correct quadrant of the MWM test in the 50-µg exosome group on day 35 (P < .05), 100-µg exosome group from days 32 to 35 (P < .05), and 200-µg exosome group on days 34 and 35 (P < .05). There was a statistically significant improved therapeutic effect with 100 µg compared with 50 µg on day 34 (P < .05). There was a significantly reduced latency for TBI rats treated with exosomes in the correct quadrant in the 50-µg exosome group from days 33 to 35 (Figure 1B, P < .05), 100-µg exosome group from days 32 to 35 (P < .05), and 200-µg exosome group from days 32 to 35 (P < .05). There was a statistically significant reduction in latency with 100 µg compared with 50 µg from days 32 to 35 (P < .05) and 100 µg compared with 200 µg from days 33 to 35 (P < .05). There was no significant difference in latency between the 50- and 200-µg groups at all time points (P > .05).
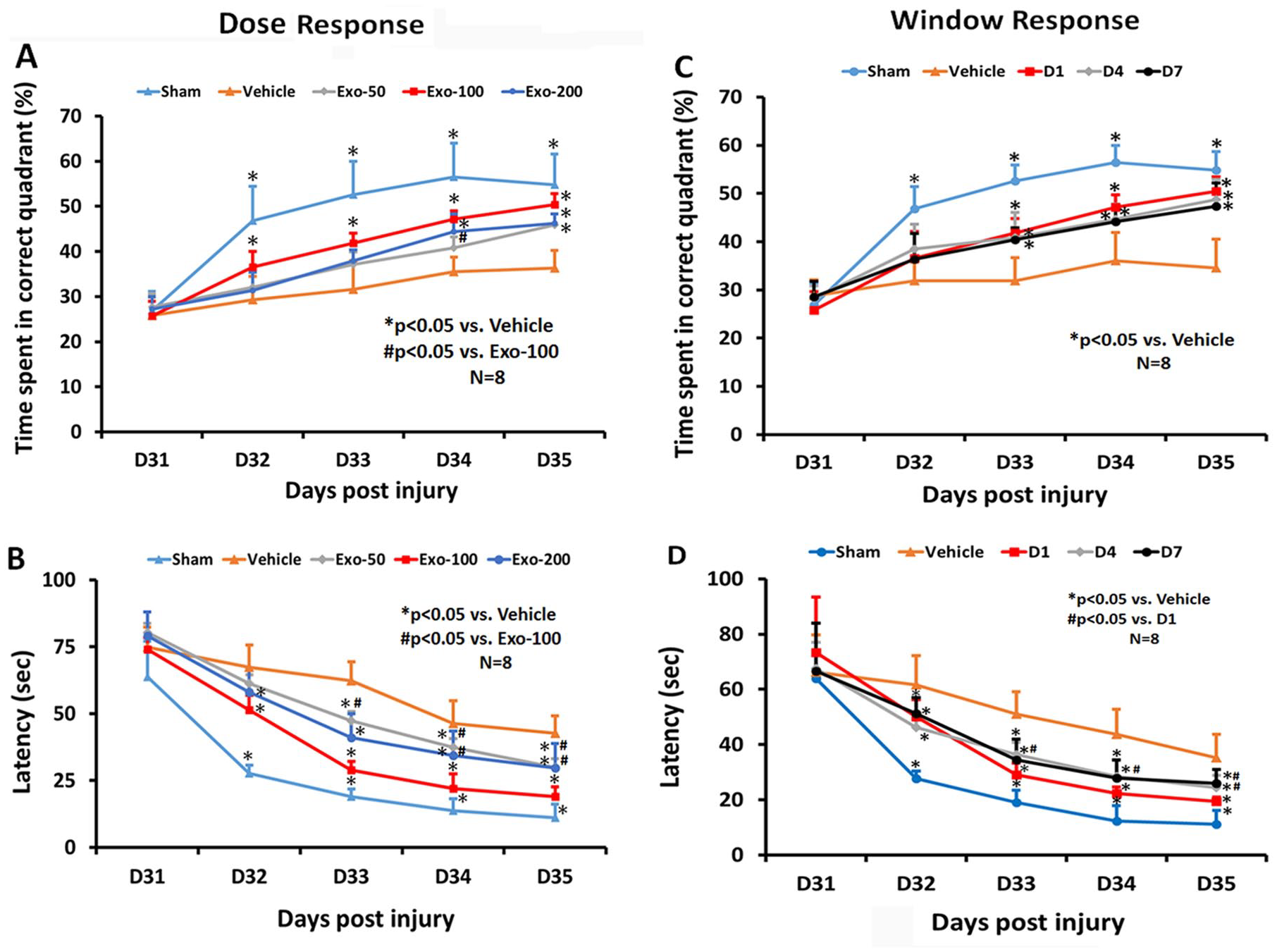
Treatment with exosomes significantly improves spatial learning after traumatic brain injury: The dose-response (A, B) and window response (C, D) studies show the percentage of time spent by animals in the correct quadrant where the hidden platform was located (A, C) and latency for animals to find the hidden platform (B, D). Data represent mean ± SD (n = 8 per group).
In the therapeutic window study (Figure 1C), among the 5 groups, a statistically significant between-group effect on the time spent in the correct quadrant was noted in the MWM test from days 32 to 35 (P < .001). Relative to the PBS group, there was a significant increase in time spent in the correct quadrant by TBI rats treated with exosomes in the D1, D4, and D7 exosome treatment groups from days 33 to 35 (P < .05). There was no statistically significant difference in improvement among the 3 exosome groups (D1, D4, D7) from day 31 to day 35 (P > .05). A statistically significant between-group effect on the latency was noted in the MWM test from days 32 to 35 (Figure 1D, P < .001). Relative to the PBS group, from days 32 to 35, a significantly reduced latency in the correct quadrant was detected in TBI rats treated with exosomes at different time points (D1, D4, D7; P < .05). A significant difference in reducing latency was detected between D1 and D4 treatments on days 33 and 34, between D1 and D4 treatments on day 35, as well as between D1 and D7 treatment groups on day 35 (P < .05), suggesting that the D1 treatment is most effective. There was no significance difference in reducing latency between D4 and D7 treatment groups (P > .05).
MSC Exosome Significantly Promotes Sensorimotor Functional Recovery in Rats After TBI
The mNSS score was identical in the PBS and exosome groups on day 1 post TBI, indicating neurological functional deficits were comparable in all TBI rats before treatment (Figure 2A; F3,28 = 0.24, P = .87). A significant reduction in the mNSS score was found over time in the PBS-treated animals starting from day 7 and up to day 35 compared with day 1 postinjury (F5,42 = 76.37; P < .01), suggesting a significant spontaneous sensorimotor functional recovery occurring after TBI. In the dose-response study (Figure 2A), compared with the PBS treatment, functional recovery was significantly improved in the 3 exosome-treated groups (50, 100, 200 µg per rat) on days 7 to 35 after TBI (P < .01, post hoc Tukey test). There was a significant reduction in mNSS in the 100-µg group compared with the 50- and 200-µg groups from days 7 to 35 (P < .05). There was a significant effect of 200 µg treatment on mNSS compared with the 50-µg treatment group from days 14 to 35 (P < .05). There was no significant difference in foot fault tests among TBI groups on day 1 postinjury before assignment of treatments (PBS or exosomes). A significant reduction in the foot fault number was found over time in the PBS-treated animals starting from day 7 and up to day 35 compared with day 1 postinjury (F5,42 = 88.95; P < .01), suggesting the presence of a significant spontaneous recovery in foot fault after TBI. Exosome treatment (50-200 µg per rat) also significantly reduced the frequency of forelimb foot fault occurrence as compared with PBS controls (Figure 2B; from days 7 to 35, P < .01). There was a significant reduction in foot fault in the 100-µg group compared with the 50-µg group from days 7 to 28 (P < .05).
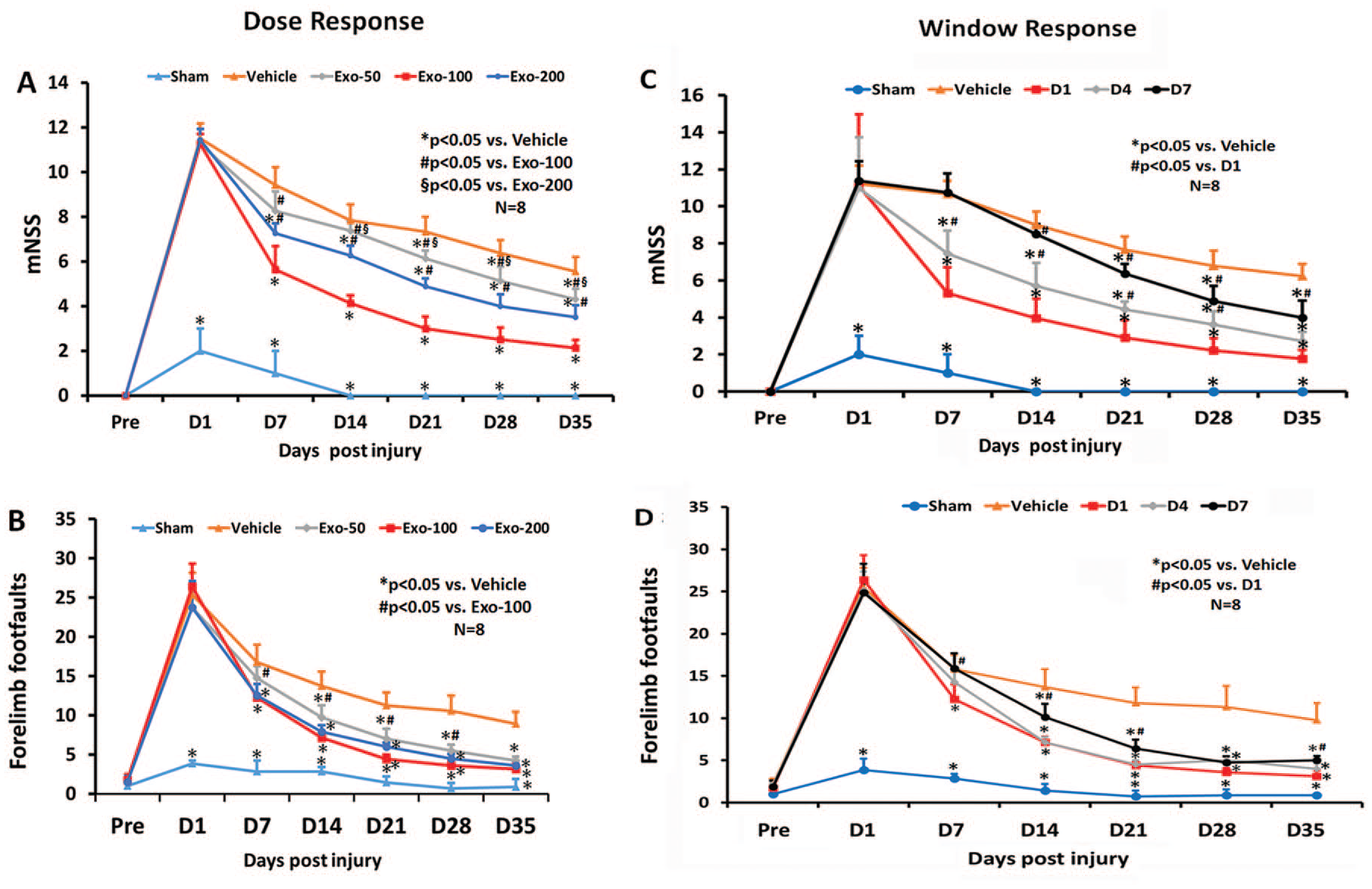
Treatment with exosomes significantly improves sensorimotor function after traumatic brain injury. The dose-response (A, B) and window response (C, D) studies show the mNSS score (A, C) and frequency of foot fault occurrence (B, D). Data represent mean ± SD (n = 8 per group).
In the therapeutic window study, a statistically significant between-group effect on reducing mNSS was noted from days 7 to 35 (Figure 2C; P < .001). Relative to the PBS group, a significantly reduced mNSS was found in TBI rats treated with 100 µg per rat exosomes (D1, D4, and D7 groups) from days 21 to 35 (P < .05). In addition, early treatments (D1 and D4 groups) significantly reduced mNSS on days 7 and 14 postinjury compared with the PBS group (P < .05). There was no statistically significant effect on mNSS in the D7 exosome group on day 14 compared with the PBS groups (P > .05). The earlier the exosome treatment started, the better the effect observed on reducing mNSS (D1 > D4 > D7 groups; P < .05). There was a statistically significant between-group effect on foot faults noted from days 7 to 35 (P < .05). Relative to the PBS group, from days 14 to 35, a significantly reduced foot fault occurrence was found in TBI rats treated with exosomes at different time points (D1, D4, D7; P < .05). There was no significantly improved therapeutic effect on foot fault occurrence between D1 and D4 groups (P > .05). However, the D1 and D4 groups had a significant reduction in foot fault occurrence compared with the D7 group on day 14 (P < .05).
MSC Exosome Administration Significantly Reduces Hippocampal Neuronal Cell Loss in Rats After TBI
In the dose-response study (Figure 3A), compared with the PBS treatment, exosome treatments (50, 100, 200 µg per rat) significantly reduced the NeuN+ neuronal cell loss in the CA3 (F4,35 = 47.00; P < .05) and DG regions (F4,35 = 328.91; P < .05) detected on day 35 postinjury. A statistically significant effect with higher doses of exosomes on reducing neuronal cell loss was found in the DG (100 vs 50 µg, 200 vs 50 µg; P < .05). There was no significant effect on neuronal cell number in the CA3 among 3 exosome doses (P > .05).
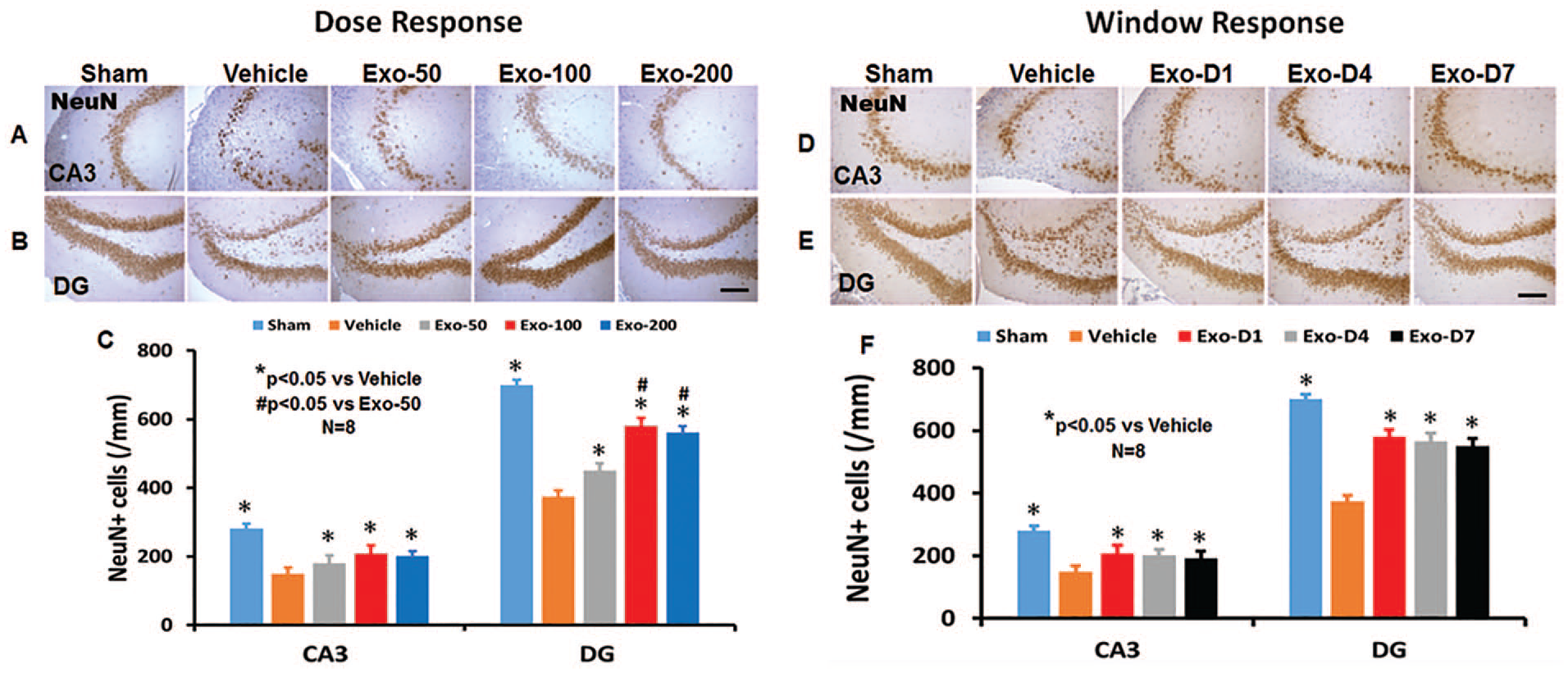
Treatment with exosomes significantly reduces hippocampal neuronal cell loss after traumatic brain injury (TBI). Neuronal nuclei (NeuN) staining was performed for detection of mature neurons on day 35 after TBI in the CA3 region and dentate gyrus (DG). Representative images show NeuN+ cells in the dose-response (A, B) and window response (D, E) studies. Scale bars = 100 µm. Quantitative data shown in bar graphs (C, F) represent mean ± SD (n = 8 per group).
In the window study (Figure 3B), compared with the PBS treatment, exosome treatments (D1, D4, and D7) significantly reduced the NeuN+ neuronal cell loss in the CA3 (F4,35 = 49.05; P < .05) and DG regions (F4,35 = 235.01; P < .05) detected on day 35 postinjury. No statistically significant difference in the effect of exosomes on reducing neuronal cell loss in the DG and CA3 regions among the 3 exosome treatment groups was detected (P > .05).
MSC Exosome Administration Significantly Increases Vascular Density and Angiogenesis in Rats After TBI
TBI alone significantly increased the density of vessels measured on day 35 postinjury in the LBZ (Figure 4; F4,35 = 363.55, P < .001) and DG (F4,35 = 219.12, P < .001) of the ipsilateral hemisphere compared with sham controls. Exosome treatments significantly increased the vascular density in the LBZ and DG compared with the PBS treatment (Figures 4A and 4C; P < .01). Exosome treatment at 50 to 200 µg per rat significantly increased angiogenesis identified by EBA/BrdU+ double labeling for newborn endothelial cells in the LBZ (Figures 4B and 4D; F4,35 = 264.91, P < .001) and DG (F4,35 = 300.63; P < .001) compared with PBS treatment. No significant increases in vascular density and angiogenesis in the injured brain were detected between 100- and 200-µg groups (P > .05). However, the 100- and 200-µg treatment groups exhibited significantly increased vascular density and angiogenesis compared with the 50-µg group (P < .05).
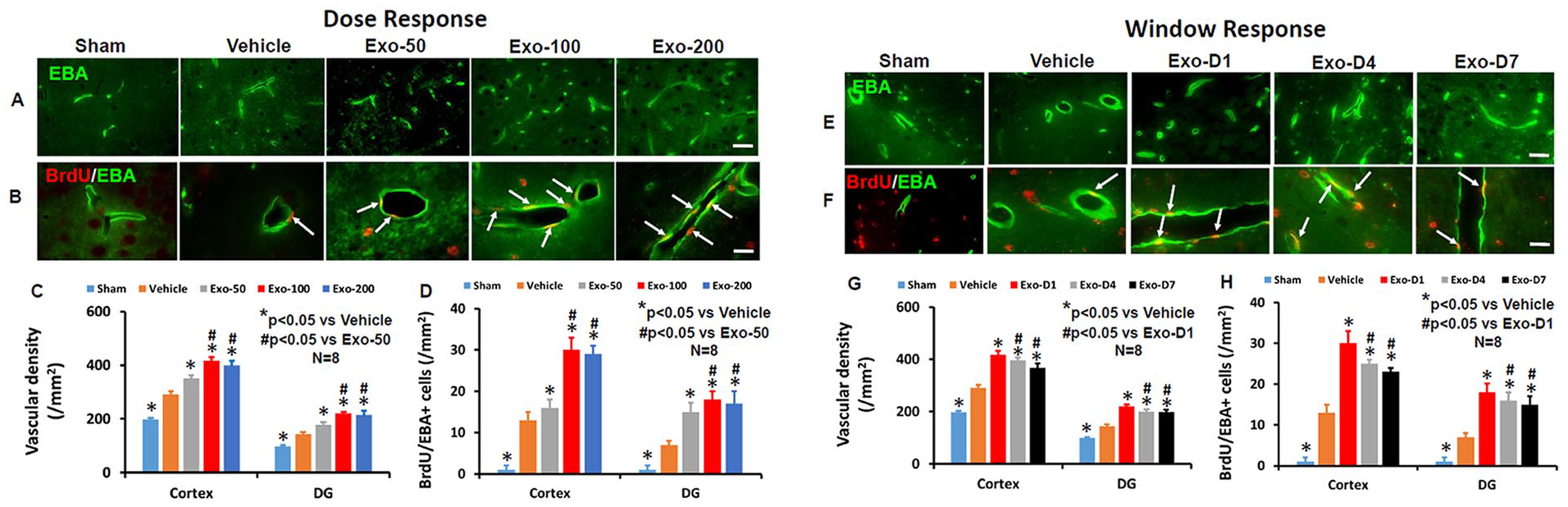
Treatment with exosomes significantly increases vascular density and angiogenesis after traumatic brain injury (TBI). EBA staining was performed for detection of mature vasculature on day 35 after TBI in the lesion boundary zone (LBZ) and DG. Representative images from the injured brain show EBA+ endothelial cells in the dose-response (A) and window response (E) studies. Scale bars = 50 µm. Representative images show BrdU+/EBA+ newly generated endothelial cells in the dose-response (B) and window response (F) studies. Scale bars = 25 µm. Quantitative data on vascular density (C, G) and angiogenesis (D, H) shown in bar graphs represent mean ± SD (n = 8 per group).
In the therapeutic window study (Figures 4E-4H), exosome treatment initiated on days 1, 4, and 7 postinjury at 100 µg per rat significantly increased the vascular density (the number of EBA+ cells) in the LBZ (Figures 4E and 4G; F4,35 = 423.47, P < .001) and DG (F4,35 = 385.53; P < .001) compared with the PBS treatment. Exosome treatments (days 1, 4, and 7) significantly increased angiogenesis identified by EBA/BrdU+ cells in the LBZ (Figures 4F and 4H; F4,35 = 332.00, P < .001) and DG (F4,35 = 146.57; P < .001) compared with the PBS treatment (P < .01). There was a significant increase in both vascular density and angiogenesis in the LBZ and DG in the D1 group compared with the D4 or D7 groups, P < .05). There were no significant differences in the vascular density and angiogenesis in the LBZ and DG between the D4 and D7 exosome groups (P > .05).
MSC Exosome Administration Significantly Increases Neurogenesis in the DG in Rats After TBI
TBI alone significantly increased the number of newborn mature neurons in the DG of the ipsilateral hemisphere compared with sham controls measured on day 35 postinjury (Figure 5; F4,35 = 230.50, P < .001). Colocalization between BrdU and NeuN was verified using a Zeiss LSM 510 META confocal laser scanning microscope (S3 Figure). However, exosome treatments at doses of 50, 100, and 200 µg per rat significantly increased the number of newborn neurons compared with the PBS control (Figures 5A and 5B; P < .01). A significant increase in neurogenesis was detected in the 100- and 200-µg groups compared with the 50-µg group (P < .05). There was no significant difference between the 100- and 200-µg groups (P > .05).
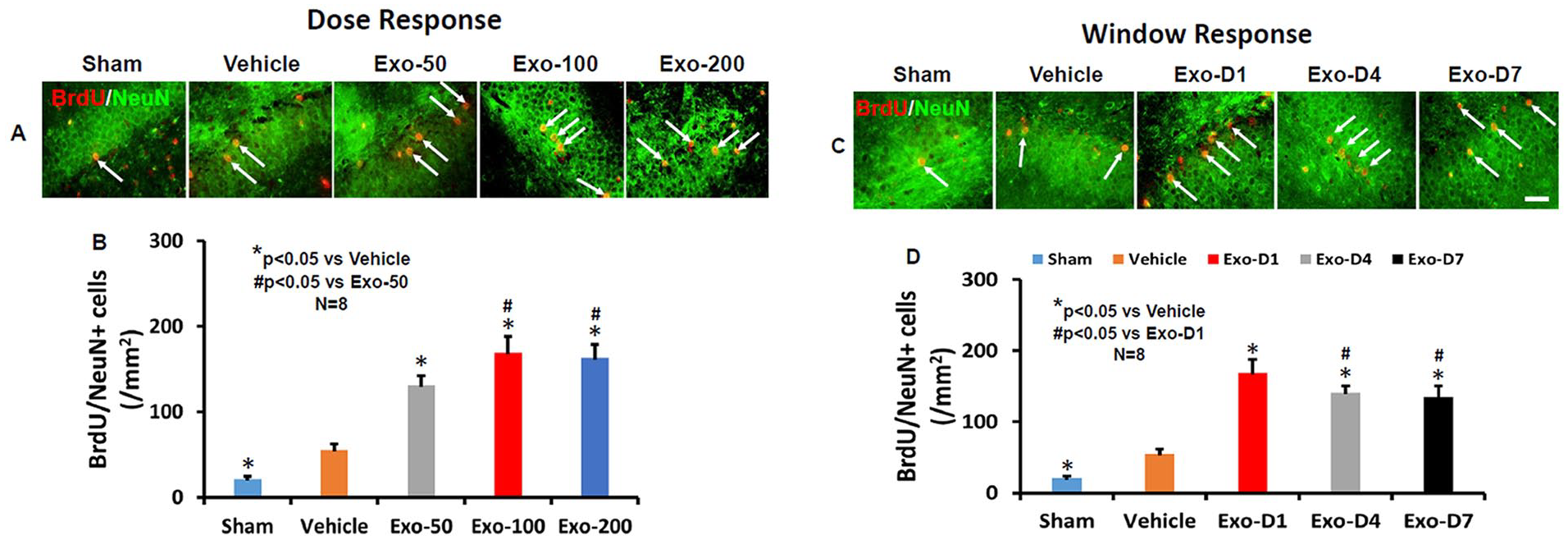
Treatment with exosomes significantly increases neurogenesis 35 days after TBI. Double staining with BrdU (red)/NeuN (green) was performed to identify newborn mature neurons in the DG (yellow arrows). Scale bar = 25 µm. Representative images show BrdU+/NeuN+ newly generated neurons in the dose-response (A) and window response (C) studies. Scale bars = 25 µm. Quantitative data shown in bar graphs (B, D) represent mean ± SD (n = 8 per group).
In the therapeutic window study, exosome treatments on days 1, 4, and 7 postinjury significantly increased the number of newborn neurons compared to the PBS control (Figures 5C and 5D; F4,35 = 209.54, P < .01). A significantly increased effect was detected in the D1 group compared with the D4 or D7 group (P < .05). There was no significant difference between the D4 and D7 groups (P > .05).
MSC Exosome Administration Significantly Reduces Brain Inflammation in Rats After TBI
TBI alone significantly increased the density of CD68+ macrophages/microglia in the LBZ (Figures 6A and 6C; F4,35 = 1100.10, P < .01) and DG (Figures 6B and 6C; F4,35 = 751.50, P < .01) of the ipsilateral hemisphere compared with sham controls. Exosome treatments at doses of 50, 100, and 200 µg per rat significantly reduced the CD68+ cell density in the LBZ and DG compared with the PBS treatment (Figure 6C; P < .01). Compared with the 50-µg group, the 100- and 200-µg groups exhibited significantly reduced CD68+ cell number in the LBZ and DG areas (P < .01). There were no significant differences detected in CD68+ cells between the 100- and 200-µg groups (P > .05). In the therapeutic window study, exosome treatments on days 1, 4, and 7 postinjury significantly reduced the number of CD68+ cells (F4,35 = 826.72 for the cortex and 1023.30 for the DG; P < .01) compared with the PBS controls (Figures 6D-6F). The D1 and D4 groups exhibited a significant reduction of CD68+ cells in comparison to the D7 group in the LBZ and DG (P < .05, with ANOVA followed by the post hoc Tukey test).
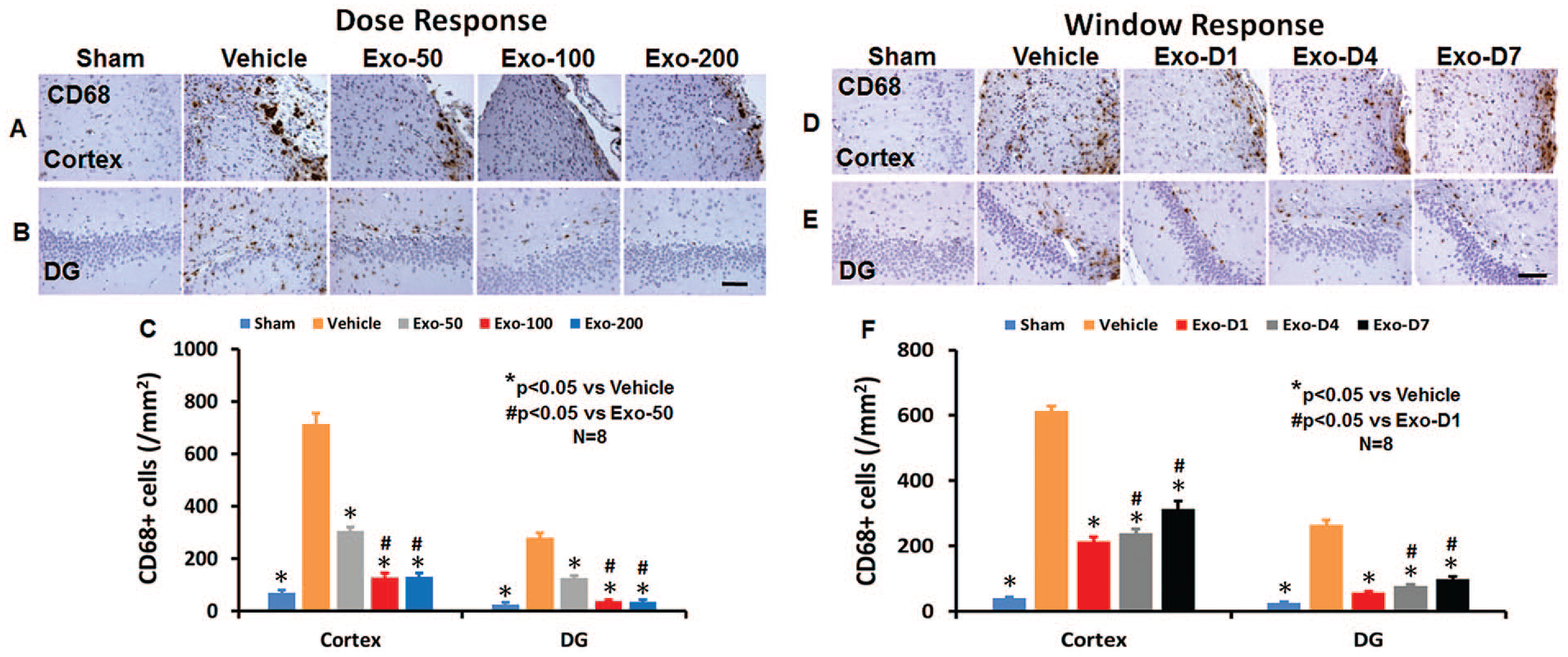
Treatment with exosomes significantly reduces the number of activated CD68+ microglia/macrophages after TBI. CD68 staining was performed to identify activated microglia/macrophages in the dose-response (A for cortex, B for DG) and window response (D for cortex, E for DG) studies. Scale bar = 100 µm. Quantitative data shown in bar graphs (C, F) represent mean ± SD (n = 8 per group).
TBI also significantly increased the density of GFAP+ astrocytes compared with sham controls (S4 Figures A-C; P < .01). Exosome treatments (50-200 µg per rat) significantly reduced the GFAP+ astrocyte density in the LBZ and DG groups compared with the PBS treatment (S4 Figures A-C; P < .01). The 100- and 200-µg treatment groups exhibited significantly reduced GFAP+ cell number in the injured brain compared with the 50-µg treatment group (P < .05). There was no significant difference between the 100- and 200-µg treatment groups. There was no significant difference in GFAP+ cells among the D1, D4, and D7 groups (S4 Figures D-F; P > .05).
Exosome Administration Does Not Alter the Volume of Total Brain Tissue Loss in Rats After TBI
No significant differences in the lesion volume were observed 35 days postinjury (S5 Figures A-D; P > .05).
Discussion
The present study demonstrates that intravenous administration of MSC-derived exosomes significantly (1) improves cognitive and sensorimotor functional recovery in rats after TBI; (2) increases the number of BrdU+/NeuN+ newborn mature neurons in the DG (neurogenesis); (3) increases the number of BrdU+/EBA+ newborn endothelial cells in the LBZ and in the DG (angiogenesis); (4) reduces neuronal cell loss in the CA3 and in the DG (neuroprotection); and (5) reduces the number of CD68+ microglia/macrophages and GFAP+ astrocytes in the LBZ and DG (neuroinflammation). The levels of neuroprotection and neurorestoration are affected in a dose-response manner, with a greater effect detected at 100 µg per rat exosomes administered 1 day after injury. In addition, the therapeutic window of exosomes can be extended up to at least 7 days post-TBI. These therapeutic effects of exosomes are not confined to a single cell type or region of the injured brain after TBI. Exosomes showed significant effects in both the injured cortex and hippocampus in terms of neuroinflammation and angiogenesis and neurogenesis in the DG.
Among stem cells, MSCs, a mixed-cell population of stem and progenitor cells, are a promising source of cell-based therapy for TBI because they can be easily isolated from many tissues and expanded in culture from patients without ethical and immune rejection problems.7,18 TBI induces acute and chronic immune response in the brain. 8 Our recent studies show that intravenous injection of 100 µg total protein of exosomes collected from culture media of approximately 2 × 106 MSCs improves functional recovery in rats with stroke 19 and TBI. 11 Treatment with exosomes derived from MSCs improve functional recovery and neuroplasticity by reducing long-term neuroinflammation in TBI rats.11,12,20 In this study, we first determined a dose-response efficacy for this novel mode of exosome treatment for TBI. Our data indicate that a higher dose of exosomes (200 µg per rat) does not provide additional beneficial effects compared with the 100-µg per rat group. However, a lower dose of exosomes (50 µg per rat) provides less beneficial effects than 100 µg per rat. Our dose-response study is in line with a recent report showing that intravenous administration of exosomes from adipose tissue–derived human MSCs (50, 100, and 200 µg per rat, 1 day postinjury) improves functional recovery, brain repair, and neural connectivity in rats with subcortical stroke induced by injecting endothelin-1. 21 Our data suggest that 50 to 200 µg of exosomes enhance brain repair and recovery in TBI rats, whereas 100 µg of exosomes is the most effective dose when treatment is initiated 1 day post–CCI-TBI.
Moderate TBI induced by CCI not only causes cortical tissue loss but also neuronal cell loss in the CA3 region and DG of the hippocampus. 11 The hippocampus is an important brain structure for learning and memory. 11 Therefore, we analyzed cell loss, angiogenesis and neurogenesis, and inflammation in the hippocampus in addition to the LBZ. Emerging preclinical studies indicate that posttraumatic prolonged and progressive neuroinflammation is associated with neurodegeneration, which may be treatable long after the initiating brain injury. 22 Neuroinflammation is a key component of the secondary injury cascades following TBI. 22 In the present study, exosomes significantly suppressed activation of GFAP+ astrocytes and CD68+ microglia/macrophages compared with the PBS controls. This anti-inflammatory effect is similar to that of MSC therapy in animal models of strok23,24 and TBI. 25 One of the main functions of astrocytes and microglia is to monitor and sustain neuronal health by releasing proinflammatory and anti-inflammatory cytokines, free radicals, antioxidants, and neurotrophic factors. 26 Activated astrocytes and microglia contribute to both neuronal death and survival in neurodegeneration 27 and TBI.28,29 Our data support that suppression of activated microglia/macrophages by exosomes may contribute to increased angiogenesis and neurogenesis, and subsequent improvement in functional recovery after TBI.
To explore the therapeutic window for MSC-derived exosome treatment of TBI, onset of exosome treatment was initiated at different times (1 day, 4 days, and 7 days postinjury). There was a significant time effect on reducing neuroinflammation and neurovascular remodeling in both the injured cortex and hippocampus. Our present data demonstrate that MSC-derived exosomes improve functional recovery in rodents with a wide range of therapeutic window from 1 day to 7 days at a dose of 100 µg after TBI, which is consistent with the therapeutic window of parental MSCs for treatment of TBI rats.30-35 A recent study in a large animal model of TBI and hemorrhagic shock demonstrates that early (1 hour postinjury) treatment with a single dose of MSC-derived exosomes significantly reduces brain swelling and lesion size, lowers levels of blood-based TBI biomarkers, and improves blood-brain barrier integrity. 36 However, we note that we do not exclude the possibility of therapeutic effects with exosome treatments initiated beyond 7 days post-TBI.
Exosomes contain complex molecular cargo.37,38 The benefit and potential strength of exosome treatment, as with stem cell therapy, result from targeting multiple injury and repair mechanisms mediated by delivery of exosomal complex cargo to recipient cells. One single microRNA can regulate many genes at the posttranscriptional level. 39 Exosome therapy may transfer miRNA to elicit a multitargeted effect, rather than the traditional single molecular pathway approach.8,40,41 The precise mechanisms of exosome therapeutic effects on functional recovery after TBI are not clear. Exosomes can incorporate into recipient parenchymal cells in rats with stroke 42 and TBI. 8 Intravenous administration of cell-free MSC-generated exosomes improves functional recovery and enhances neurite remodeling, neurogenesis, and angiogenesis after brain injuries in rats,11,12,19 pigs, 43 and monkeys. 44 Further studies are warranted to identify the molecular constituents of the exosomes, including specific miRNAs and growth factors that promote angiogenesis and neurogenesis, and reduce neuroinflammation after TBI. When the specific molecules necessary for a therapeutic effect are identified, selective manipulation of expression of those molecules in the parent MSCs may lead to an enhancement of the therapeutic efficiency of isolated exosomes.
Neurovascular units in the central nervous system consist of endothelial cells, pericytes, neurons, glial cells, and extracellular matrix proteins. 45 The generation of new vasculature facilitates highly coupled neurorestorative processes, including neurogenesis and synaptogenesis, which in turn lead to improved functional recovery. 45 New neurons are generated from neural stem/progenitor cells occurring in mammals during adulthood and in the pathology of different neurological disorders, and thus, neurogenesis may be a potential target area for treatments. 46 Exosome-induced angiogenesis may contribute to motor functional recovery by promoting neurite growth and synaptogenesis in the injured brain. 19 Our data in the DG suggest that exosome-induced angiogenesis and neurogenesis may play an important role in improving learning and memory after brain injury, which is consistent with previous studies.47-50 In addition to exosomes, MSCs also release other extracellular particles and soluble factors, which may contribute to therapeutic effects underlying MSC therapy.37,38 We do not exclude their effect in MSC-mediating TBI recovery.
In the present study, the cell counting was performed using a profile-based method on a computer monitor to improve visualization and in 1 focal plane to avoid oversampling.51,52 This method, although suboptimal, provides a meaningful comparison of differences in cell numbers among groups after brain injuries.51,52 Because many investigators seek to determine relative and significant differences in cell numbers between treatment and control groups rather than attempting to estimate absolute cell numbers, it has been argued that, in many cases, the systematic bias will be canceled out, and thus conventional methods suffice for this task. 53 Collecting comparably reliable physical dissector data is very time-consuming requiring considerably more sophisticated equipment than profile counts. 54 Because we are interested in detecting a significant difference in relative cell numbers among groups, profile counting is the more rational choice. It should be emphasized that when information about absolute cell numbers is required, major biases should be ruled out by calibration, or the unbiased stereology method should be used.
Conclusions
MSC-derived exosomes administered intravenously following moderate CCI-TBI in rats improve function recovery with a wide therapeutic window (at least up to 7 days) and offer substantial neuroprotective and neurorestorative effects by reducing neuronal cell loss and neuroinflammation and amplifying angiogenesis and neurogenesis. Our data and literature suggest that intravenous administration of exosomes may represent a novel therapeutic approach for treatment of acute and subacute TBI.
Supplemental Material
Supplemental_materials-revised-clean_copy – Supplemental material for Mesenchymal Stem Cell–Derived Exosomes Improve Functional Recovery in Rats After Traumatic Brain Injury: A Dose-Response and Therapeutic Window Study
Supplemental material, Supplemental_materials-revised-clean_copy for Mesenchymal Stem Cell–Derived Exosomes Improve Functional Recovery in Rats After Traumatic Brain Injury: A Dose-Response and Therapeutic Window Study by Yanlu Zhang, Yi Zhang, Michael Chopp, Zheng Gang Zhang, Asim Mahmood and Ye Xiong in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors thank Sutapa Santra and Qinge Lu for their technical assistance.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Authors’ Note
Author contributions to the study and article preparation include the following: conception and design: YX, MC; acquisition of data: Yanlu Z, Yi Z, YX; analysis and interpretation of data: YX; drafting of the manuscript: YX, Yanlu Z; critical review of the article: YX, MC, ZG Zhang, AM; review of final version of the article and approval for submission: all authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institutes of Health Grant 1R01NS100710-01A1 to YX.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
