Abstract
Background:
After an acquired injury to the motor cortex, the ability to generate skilled movements is impaired, leading to long-term motor impairment and disability. While rehabilitative therapy can improve outcomes in some individuals, there are no treatments currently available that are able to fully restore lost function.
Objective:
We previously used activity-dependent stimulation (ADS), initiated immediately after an injury, to drive motor recovery. The objective of this study was to determine if delayed application of ADS would still lead to recovery and if the recovery would persist after treatment was stopped.
Methods:
Rats received a controlled cortical impact over primary motor cortex, microelectrode arrays were implanted in ipsilesional premotor and somatosensory areas, and a custom brain–machine interface was attached to perform the ADS. Stimulation was initiated either 1, 2, or 3 weeks after injury and delivered constantly over a 4-week period. An additional group was monitored for 8 weeks after terminating ADS to assess persistence of effect. Results were compared to rats receiving no stimulation.
Results:
ADS was delayed up to 3 weeks from injury onset and still resulted in significant motor recovery, with maximal recovery occurring in the 1-week delay group. The improvements in motor performance persisted for at least 8 weeks following the end of treatment.
Conclusions:
ADS is an effective method to treat motor impairments following acquired brain injury in rats. This study demonstrates the clinical relevance of this technique as it could be initiated in the post-acute period and could be explanted/ceased once recovery has occurred.
Introduction
Traumatic brain injury (TBI) and other acquired brain injuries contribute substantially to disability and death worldwide. Survivors are often left with severe motor disabilities and prolonged medical complications resulting in a decreased quality of life and a significant burden on the health care system.1 -4 Once an injury has progressed beyond the acute phase, there are limited treatment options available, especially during the sub-acute phase of injury when they are likely to be most effective. 5 Generally, therapeutic strategies target the overt symptoms of brain injury rather than addressing the underlying causes of impairment, leading to recovery profiles that are often mixed in response and limited in scope. It is critical to develop treatment modalities that target the underlying damage and focus on restoration of function rather than mere accommodation of disability.
Immediately following a TBI, there is a cascade of damage to neurons, glia, and blood vessels followed by edema, inflammation, and mitochondrial dysfunction at the point of impact. There is also a global change in functional connectivity throughout the brain.6 -10 When such an injury occurs in primary motor cortex (M1), communication within the entire sensorimotor network is disrupted. Beyond the loss of descending motor output to the spinal cord, there is a disruption in the signals to and from premotor and somatosensory cortices, interfering with motor planning and movement kinematics11 -13 and the integration of proprioceptive and tactile information necessary for motor execution.14,15 This disruption of such a highly coordinated flow of information results in persistent impairment of the planning and execution of volitional movement.
Generally, long-term rehabilitative therapies are focused on compensating for or restoring lost motor function through repetitive practice on skilled volitional tasks. The efficacy of rehabilitation is thought to be dependent on the reorganization of spared tissue to facilitate motor recovery. A range of electroceutical strategies developed to promote and utilize this reorganization have been employed as adjunctive therapies but have shown only modest effects in functional recovery. Non-invasive cortical stimulation modalities that utilize an open-loop modulation of neural activity within a relatively large volume of cortical tissue, such as transcranial direct current stimulation (tDCS) and repetitive transcranial magnetic stimulation (rTMS) have been used in combination with rehabilitative therapy to promote behavioral recovery in stroke patients. These therapies enhance neural excitability in the injured hemisphere and decrease transcallosal inhibition.16 -21 More invasive and focal epidural stimulation has been shown to promote improvements in motor recovery after ischemic injury in pre-clinical animal studies but has failed to show efficacy in clinical trials.22 -28 While current therapeutic strategies show promise in improving motor function after cortical injury, they rarely result in a complete restitution of function and may be limited by their inability to directly target neural pathways or specifically shape neural communication between areas impacted by the injury.
Following injury, large-scale neuroplasticity occurs throughout the brain, including novel axonal projections developing between disconnected regions. This reorganization is likely advantageous for reestablishing communication and in turn improving motor outcomes. Using electrical stimulation, it is possible to artificially induce Hebbian plasticity by directly pairing the intrinsic activity of one neuron to an evoked response in a second, distant neuron. This technique, known as activity-dependent stimulation (ADS) was first described by Jackson and colleagues to alter the output properties of neurons in M1 29 and has been utilized in other motor-related contexts since. Using intrinsic neural activity as a pairing signal (rather than strategies such as paired-pulse paradigms) has several advantages including strengthening of neural connections, reinforcement of neuroplasticity, enhancement of signal synchronization, and activation of reinforcing reward-based circuits.30 -32 ADS can also induce alterations in neuronal activity, such as changes in firing rate of trigger neurons and increases in the correlation of activity within a region, indicators of greater synaptic coupling.29,31,33 -37 These prior studies support the use of ADS to synchronize activity and strengthen functional connections between discrete areas.
Given the efficacy of ADS to modify neural connectivity in vivo, Guggenmos et al, 35 developed its use as a tool for rehabilitative therapy. Within hours following a controlled cortical impact (CCI) injury to M1 in a rat model of TBI, ADS was initiated and delivered continuously for 4 weeks in a paradigm where each action potential at a single electrode site within premotor cortex (PM) triggered a single intracortical microstimulation (ICMS) current pulse to the forepaw area of primary somatosensory cortex (S1). Rats receiving ADS showed substantial behavioral improvements on a skilled reaching task within 8 days of injury and were indistinguishable from healthy uninjured rats by Day 14 post-injury. In contrast, rats that received random stimulation demonstrated modest behavioral recovery while rats that received no stimulation had persistent motor deficits. The behavioral benefits of ADS are likely the result of the facilitation in communication between the coupled areas through the coordination of neural activity.
While these previous results demonstrated the efficacy of this approach, implementation of ADS immediately after cortical injury may not be feasible from a clinical perspective. Stimulation was initiated immediately, in the acute phase of injury, which is impractical as the patient would need to become medically stable and post-TBI edema resolved prior to the implantation of invasive electrodes, a process which may take days to weeks. It is unknown whether a delay of ADS treatment would lead to similar, significant recovery profiles. If so, this would lead to an increased likelihood of translating this technology to clinical usage. To this end, we expanded on our previous results by investigating (1) the effect of delaying the onset of stimulation on the behavioral recovery profile of rats with a traumatic injury to M1 and (2) the persistence of recovery after ADS is ceased.
Methods
Animals
Adult, male Long-Evans rats (n = 24, weight: 330–450 g) were procured at 10 weeks of age. Male rats were used exclusively in this study to reduce the variability introduced by the time of impact related to the estrus cycle. 38 In an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) accredited facility, rats were singly housed in transparent cages, provided with food and water ad libitum, and kept at an ambient temperature of 22°C on a 12h:12h light/dark cycle. All procedures were in accordance with standards outlined by the Guide for the Care and Use of Laboratory Animals and approved by the local Institutional Animal Care and Use Committee.
Timeline
Prior to behavioral training on a skilled reaching task, rats were randomly assigned to four groups: Week1, Week2, Week3, and Control with reference to the time of initiation of treatment. Once rats achieved a mean behavioral score of 70 (percentage of successful reaches in 60 trials over three assessments) on the task, they underwent surgery to receive a controlled cortical impact (CCI) injury to M1 forelimb area, microelectrode implant (PM and S1), and microdevice connection. Week1, Week2, and Week3 rats received ADS continuously 24 h/day for 4 weeks beginning post-injury day 7 (Week1), day 14 (Week2), or day 21 (Week3). Control rats did not receive ADS. Post-injury behavioral assessments were conducted weekly with the final assessment 1 week after ADS ceased (Figure 1A). A subset of randomly assigned Week1 rats was assessed for an additional 8 weeks after ADS ceased (Figure 1B).
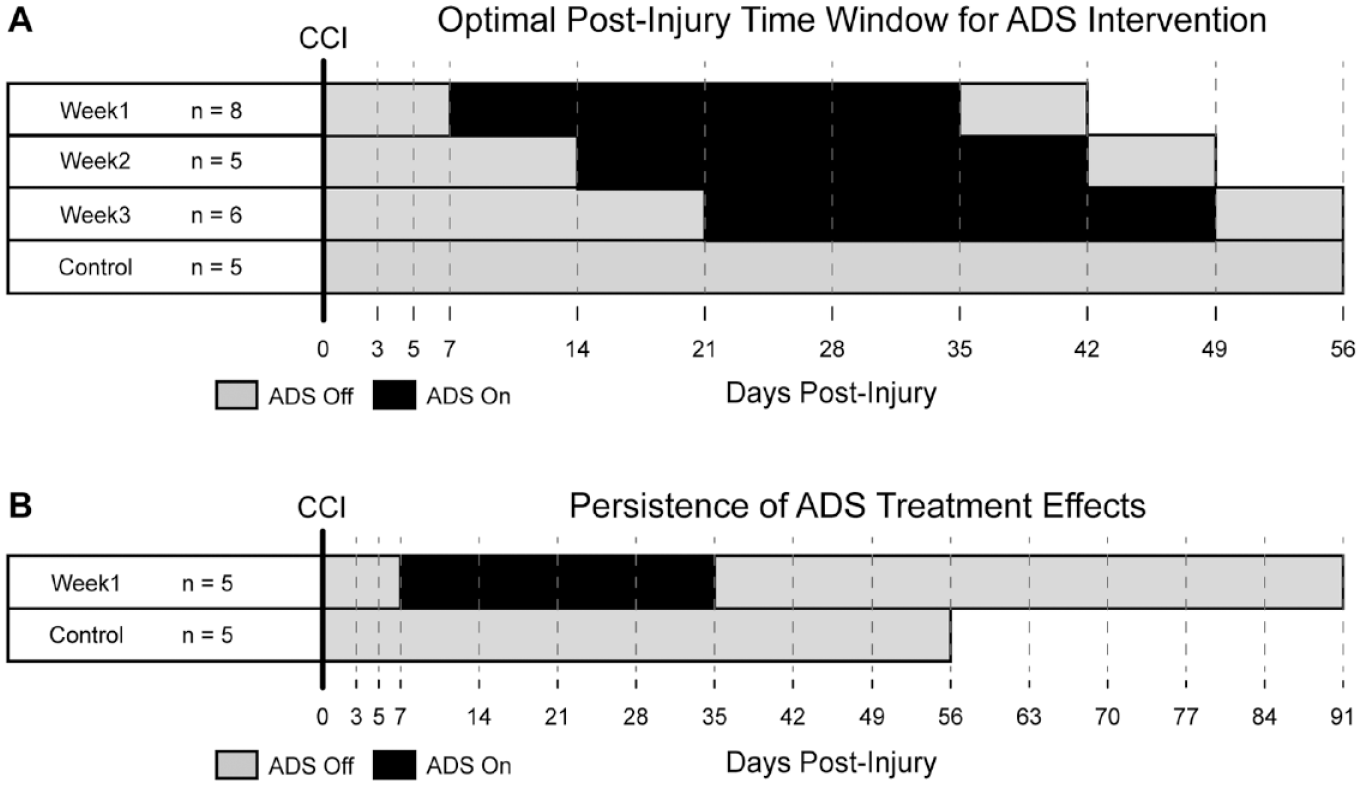
Experimental timeline. (A) Rats were randomly assigned to one of four groups (Week1, Week2, Week3, or Control). After successful training on a skilled reach task, all rats underwent a surgery to receive a CCI to M1 forepaw area and were implanted with a chronic recording microelectrode array in PM and a chronic stimulating microelectrode array in S1 (Day 0). ADS was delivered continuously 24 h/d for 4 weeks beginning post-injury day 7 (Week1), day 14 (Week2), or day 21 (Week3). Control rats did not receive stimulation. Behavioral assessments were conducted on post-injury days 3, 5, and 7 then weekly for the remainder of the study (gray dashed lines). (B) Behavioral assessments on a subset of randomly assigned Week1 rats were conducted for an additional 8 weeks after ADS was ceased.
Behavioral Task
A skilled reaching task 35 was used to measure motor behavioral recovery following brain injury. Training. Rats were placed inside a 30 cm × 30 cm × 52 cm clear Plexiglas chamber. On an external shelf positioned 3 cm from the bottom of the chamber, a single food pellet (45 mg; Bioserv) was placed in a shallow well 2 cm from the front wall of the chamber (oriented so that only the preferred forelimb could perform the task). Rats were required to reach through a narrow slot in the front wall of the chamber, grasp the pellet with the forepaw, and retrieve the pellet to the mouth. During each assessment, a reach trial was counted when the rat made paw-to-food pellet contact. Success rate was measured as the percentage of trials in which the rat successfully grasped, retrieved, and delivered the pellet to the mouth (60 trials per assessment). In some cases, the severity of the injury, especially Days 3 and 5 post-injury, impaired the rat’s ability to perform a reach that met our criteria to be qualified as a reach attempt, thereby preventing the rat from completing 60 trials. In the event the rat did not complete 60 trials within 1 hour, the assessment was ended and scores were assessed based on the number of trials completed. Prior to inclusion on study, each rat was required to achieve a 70% or greater success rate averaged over three behavioral assessments. Assessment. Following the injury, behavioral assessments (60 trials) were conducted on post-injury days 3, 5, 7 and then weekly for the remainder of the study (Figure 1) with the final assessment occurring 1 week after ADS ceased. On the day that ADS was initiated, the behavioral assessment was conducted prior to activating the microdevice and commencing the stimulation. During the 4-week window of ADS, rats received stimulation during the assessment. When ADS was to be ceased, the behavioral assessment was conducted before the stimulation was turned off. All behavioral assessments were recorded for offline analysis by an investigator blinded to date, subject, and group identifiers.
Surgical Procedures
After behavioral training, all rats underwent surgery to receive a CCI injury to M1 forelimb area, microelectrode implant (PM and S1), and microdevice connection. Rats were anesthetized with an initial inhalation of isoflurane and injections of ketamine (80–100 mg/kg, IP) and xylazine (5 mg/kg, IM) and maintained throughout the surgery by supplemental ketamine injections (10–20 mg/kg/h, IM). A systemic dose of prophylactic antibiotic was administered (Penicillin, 45K IU, SC). With the rat’s head secured in a stereotaxic frame, a midline incision of the scalp was made and extended caudally to allow for a laminectomy (to control brain edema after the craniectomy). The temporalis muscles were resected, and four burr holes were drilled in the skull for later placement of skull screws and the microdevice anchoring rod (Figure 2A).
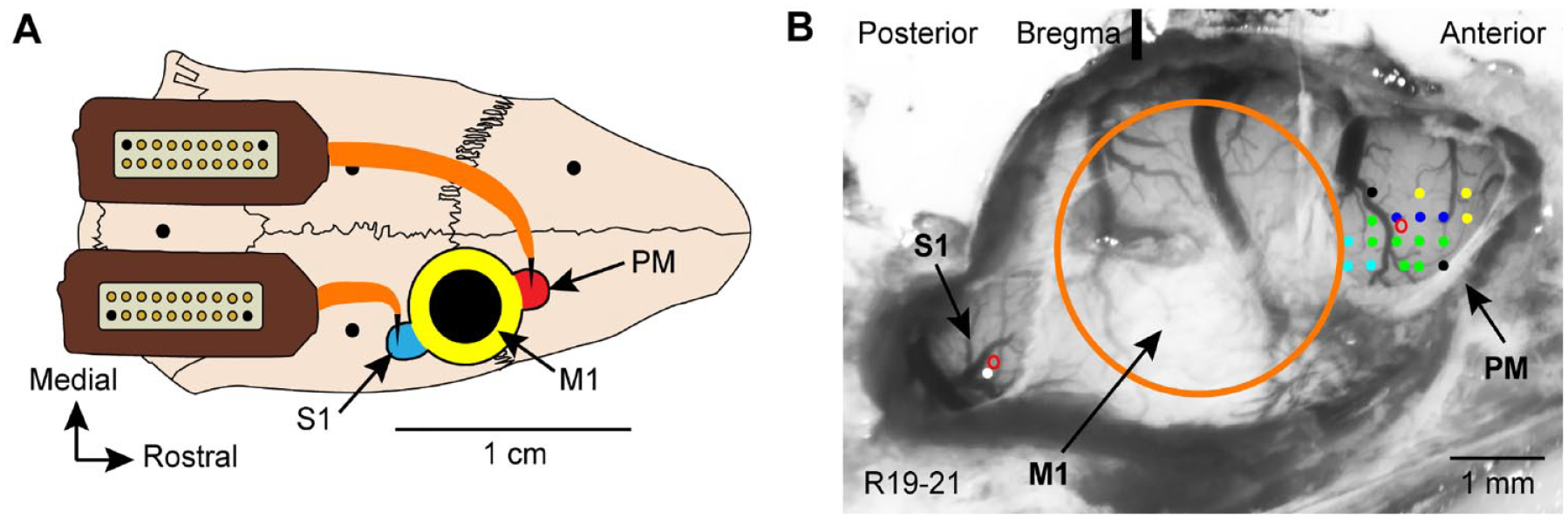
CCI and microelectrode implant. (A) Schematic of the rat skull with the approximate location of the craniectomy over M1 (yellow), S1 (blue), and PM (red). Large black circle indicates location of CCI. Four small black circles indicate locations of skull screws and microdevice anchoring rod. A chronic recording probe was implanted in PM. A chronic stimulating probe was implanted in S1. Omnetics connector bases of each probe were affixed to the skull. (B) Cortical surface vasculature following a craniectomy. ICMS (colored dots indicate electrode penetrations) was used to identify microelectrode implant target location in PM (green, distal forelimb; dark blue, proximal forelimb; light blue, trunk; yellow, face; black, no response). ICMS parameters: 40 ms train of thirteen 200 μs monophasic cathodal pulses delivered at 350 Hz repeated at 1/s was generated by a constant-current stimulus isolation unit. Single unit neural recordings were used to determine evoked sensory activity in the forepaw area of S1 (white dot). Small red circles, target sites of microelectrode implants in S1 and PM. Large orange circle, target site of CCI over M1.
Primary Motor Cortex (M1)
A 5 mm diameter craniectomy was centered over the forelimb area of M1 (AP, +0.5; ML, +3.25) for the subsequent CCI. Previous mapping studies have shown that stereotaxic coordinates are reliable and effective in estimating the center of M1.35,39 -41 Location was verified by visual inspection of cortical vasculature. The dura was left intact (Figure 2B).
Premotor Cortex (PM)
The craniectomy was extended rostrally exposing PM (AP, +3.5; ML, +2.5) and the dura was resected. The location of the forepaw area was confirmed using standard ICMS techniques. 39 Briefly, a pulled glass microelectrode (containing 3.5M NaCl and a platinum wire) was inserted to a depth of ~1700 µm. A 40 ms train of thirteen 200 µs monophasic cathodal pulses was delivered at 350 Hz repeated at 1/s. Current was increased until either a movement was observed, or the current reached a maximum of 70 µA. Using this technique, the forepaw area of PM was identified (Figure 2B).
Primary Somatosensory Cortex Forepaw Area (S1)
The craniectomy was extended caudally exposing S1 (AP, -1.25; ML, +4.25) and the dura was resected. The location of the forepaw area was confirmed by identifying forepaw receptive fields. Briefly, a 16-channel, single shank Michigan electrode (NeuroNexus, Ann Arbor MI) was lowered into layers III-IV of S1. Neural activity was amplified, digitized, filtered, displayed on a computer monitor, and emitted through a speaker using commercial equipment (Tucker-Davis Technologies, Alachua, FL). Receptive fields were identified by noting the extent of the skin surface that evoked neural activity with light touch and/or palpation (Figure 2B).
Controlled Cortical Impact
A commercial impactor device (Leica Microsystems) with a 3 mm flat-tipped rod was centered over M1 forelimb area. An impact was delivered at 1.5 m/s to a depth 2 mm below the dura surface, with a dwell time of 200 ms. This produces highly reproducible lesions to all cortical layers within M1 but minimal damage to adjacent cortical areas and underlying white matter tracts. 40
Microelectrode implant and microdevice attachment
After the CCI, recording and stimulation microelectrodes were implanted in PM and S1 as described in detail by Guggenmos et al. 35 Figure 2B illustrates the location of the microelectrodes. Briefly, skull screws were implanted into the parietal bones, an anchoring rod was implanted into the intraparietal bone, and dental acrylic was applied to reinforce the screws and create a base for the microelectrode connectors. In PM, a 16-channel, single shank, chronic Michigan electrode was inserted to a depth of ~1700 µm below the cortical surface using a micropositioner with recording sites situated at ~1500 to 1700 µm below the cortical surface (presumptive cortical layer V). The probe was secured in place with a thin coat of silicone polymer on the surface of the cortex (Kwik-Cast, World Precision Instruments, Sarasota, FL) and the base of the probe connector was fixed in place to the dental acrylic. In S1 forepaw area, an activated 16-channel, single shank, chronic Michigan electrode (typical pre-activation impedance, 600–800 kΩ; typical post-activation impedance, 70–100 kΩ) was inserted to a depth of ~1700 µm below the cortical surface using a micropositioner with stimulating sites situated at ~800 to 1200 µm below the cortical surface (presumptive cortical layers III-IV). The probe was secured in place similarly to above. Ground wires for both probes were attached to the anchoring rod. Any remaining exposed cortex was coated with silicone polymer and the incision was sutured. The microdevice was then attached to the anchoring rod and the recording and stimulating probes were connected to the microdevice.
Following surgery, wound edges were treated with lidocaine cream and triple antibiotic ointment. A second systemic dose of prophylactic antibiotic was administered (Penicillin, 45K IU, SC). The animal was given postoperative analgesics (acetaminophen, 20–40 mg/kg, oral; buprenorphine, 0.05–0.10 mg/kg, SC) over the next 48 hours and was not tested while recovering.
Microdevice
Recording
The microdevice has been described in technical detail by Azin et al42,43 and used in awake, behaving rats by Guggenmos et al. 35 The microdevice was powered by a 1.55 V coin battery which was replaced every 24 hours to maintain a consistent operating voltage. Using a flexible, multi-conductor cable, the microdevice could be connected to a custom controller board for programming and signal monitoring and to a personal computer. When connected to the controller board, the microdevice could record, amplify, digitize, and filter neural activity from up to 4 of the 16 channels on the recording probe in PM. The microdevice also had the ability to wirelessly, via an RF signal, transmit neural activity from one channel. ADS programming. Spike discrimination and ADS programming has been described in detail previously. 35 For initial programming, rats were connected using a wired connector and 4 channels of neurophysiological data were observed in real-time. A single channel within PM was selected based on signal-to-noise ratio for spike detection. A snippet of recorded data from this channel was piped to a custom MATLAB (Mathworks) script for spike discrimination of a single unit using thresholding and a dual time-amplitude window discriminator (Figure 3). The parameters for this discrimination were recorded and used for device programming. Stimulation parameters were set to deliver single 60 µA, 192 µs, pseudo-biphasic (initial cathodal phase with a charge-balancing capacitive discharge) current pulses with a 7.5 ms delay after spike detection using the programmed time-amplitude discriminator followed by a 28 ms blanking interval through the S1 stimulating probe. Spike discrimination and stimulation parameters were uploaded to the microdevice for online spike discrimination. Neural activity from PM was observed and recorded daily beginning Day 3 post-injury from all animals. ADS was delivered continuously 24 h/day (excluding brief periods to adjust spike discrimination parameters or to change the battery) for 4 weeks beginning Day 7, 14, or 21 (group Week1, Week2, Week3, respectively; Figure 1). In general, spike detection parameters were stable across days, typically requiring only single sample alterations in timing or voltage level. In the rare case that signal was lost on a particular channel, reassessment and reprogramming occurred as above. Neurophysiological data collection. Microdevice performance was monitored daily by recording neurophysiological data (either wirelessly or wired) from the PM microelectrode for ~30 minutes while the rats were awake and in their home cages. During the 4-week window of ADS treatment, neurophysiological data were recorded during stimulation. Weekly neural recordings (acquired on behavioral assessment days but during spontaneous behavior in the home cage) were imported into MATLAB and processed using a custom MATLAB script to identify stimulation rates per session in relation to duration of ADS treatment. All spiking rates were log-normalized prior to statistical analysis.
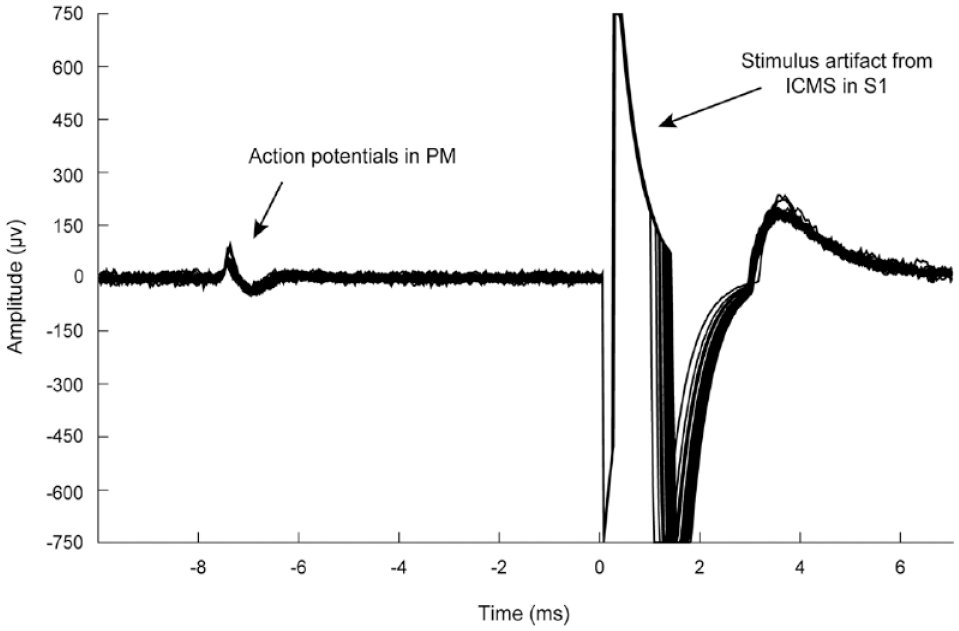
Display of activity-dependent stimulation (ADS). Upon spike detection in PM, the microdevice delivered a pseudo-biphasic stimulus pulse to S1 (7.5 ms delay, 28 ms blanking period, 60 μA, 200 μs). Detected spikes and stimulus artifact were overlaid, time 0 was aligned with stimulus pulse delivery.
Post-Mortem Analysis of Lesion Size
Animals were humanely euthanized with Beuthanasia-D (390 mg pentobarbital, 50 mg phenytoin sodium, 50–100 mg/kg, IP) and transcardially perfused (0.2% heparin/lidocaine in 0.9% saline followed by 3% paraformaldehyde, pH 7.4). The brains were removed, and the hemispheres were separated and post-fixed in 20% glycerol/4% paraformaldehyde. A series of 50 µm thick sections were cut tangentially through the cortex with a freezing microtome for future tract-tracing analysis. Several sections were stained with cytochrome oxidase (CO) to visualize the cortical sensory areas. One section from each animal was identified for lesion analysis as the lesion was induced perpendicularly to the tangentially sectioned brain slice and all impacts were to the same depth (Supplemental Figure 1). Lesion area was calculated and averaged for each group with MBF Bioscience StereoInvestigator software.
Statistical Analysis of Behavioral Performance
Linear mixed models (LMMs) were used to assess differences in groups pre-injury and analyze the effect of stimulation on recovery of motor function. SAS version 9.4 (SAS Institute, Inc.) was used for analysis. The primary outcome was the percentage of successful reaches in 60 trials. Models that assessed differences in groups pre-injury were adjusted for the fixed effects of time (continuous linear), treatment group, and their interaction. Models used to analyze recovery of motor function were adjusted for the fixed effects of time (continuous linear, quadratic, and cubic terms), stimulation group, and their interactions (Supplemental Figure 2). Third-order polynomials accounted for the nonlinear curvature in behavioral score over time based on forward selection, with P values <.05 required for inclusion in corresponding F-tests adhering to the hierarchy principle for lower order terms. Each animal was allowed a random intercept to account for repeated measures. Model assumptions were assessed visually by examining plots of residuals versus observed values, quantile–quantile plots and residual histograms. A linear pattern in the plot of residuals versus observed values was present since no animal could have a negative behavioral score.
Linear contrasts were utilized to test research hypotheses. Testing for treatment differences in the percentage of successful reaches pre-injury and post-injury (but pre-stimulation) was compared using F-tests at different timepoints. Testing recovery across treatments used change in each treatment groups’ mean percentage of successful reaches using global F-tests. Treatment differences following completion of stimulation were compared on post-injury Day 57 using an F-test. Pairwise treatment group differences were assessed using t-tests. Lastly, a t-test was used to test for a decline in behavioral score after stimulation using change in post-stimulation behavioral score (Day 35 vs Day 90) in the Week1 stimulation group.
Results
Behavioral Recovery After M1 Injury
Optimal Post-Injury Time Window for ADS Intervention
No differences were detected between groups in pre-injury behavior (F(6,44) = 0.54; P = 0.776). All groups demonstrated a profound behavioral impairment during the first week post-injury (Figure 4A). Of the 24 rats on study, 7 were unable to perform a reach that met our criteria to be qualified as a reach attempt on Day 3 post-injury and 1 was unable to perform a reach on Day 5 post-injury. Comparing the LMM-estimated mean scores for all groups, no differences were detected on post-injury days 3, 5, and 7 (F(3,218) = 0.39; P = 0.761, F(3,218) = 1.23; P = 0.300, and F(3,218) = 2.44; P = .065 respectively).
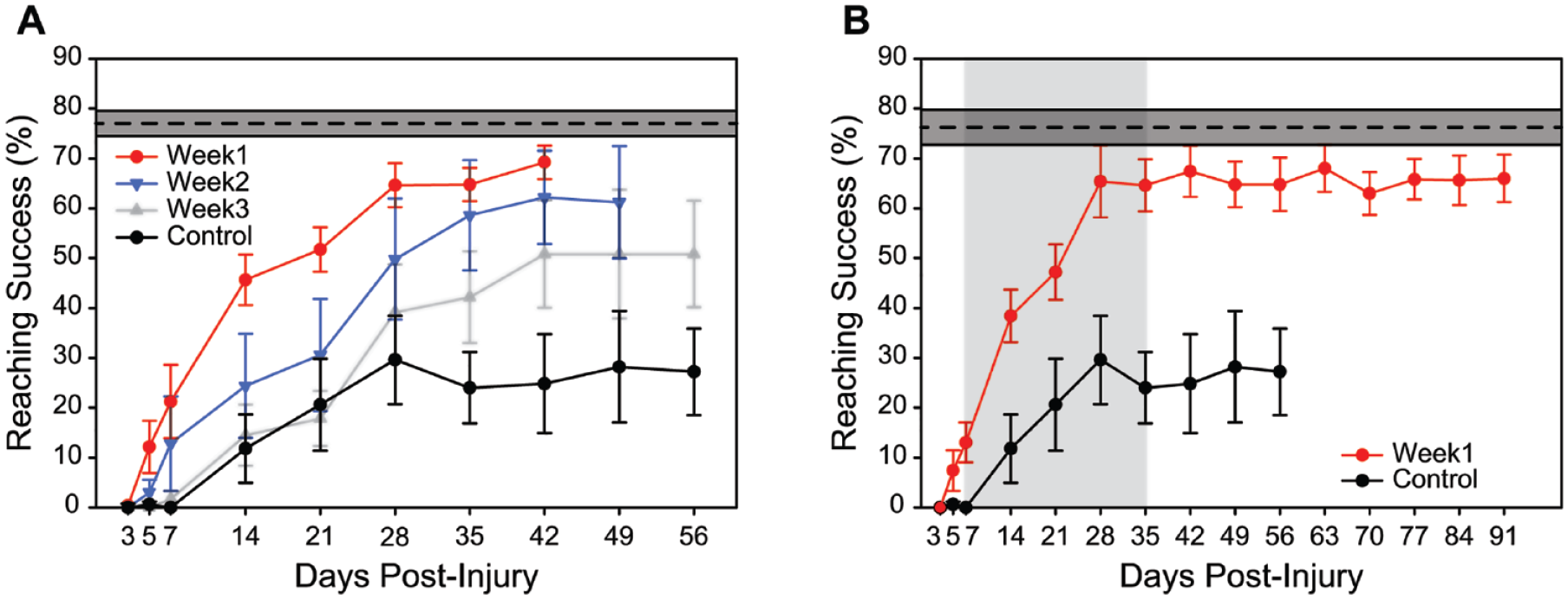
Performance on a skilled reach task following a CCI to M1 (successful reaches/60 trials ± SEM). (A) ADS was initiated at three time points relative to time of injury (post-injury day 7, Week1 group/red; day 14, Week2 group/blue; day 21, Week3 group/gray) and was delivered continuously for 24 h/d for 4 weeks in each treatment group. Control animals received no stimulation. The average pre-injury baseline behavioral success rate of all animals on study is indicated by the dotted line with a 95% confidence interval in gray. (B) Behavioral performance was assessed on a randomly assigned subset of Week1 animals for an additional 8 weeks after ADS ceased to determine persistence of ADS treatment effects. Light gray block indicates ADS treatment window for Week1 animals. Control animals (same animals from Figure 4A) did not receive stimulation. The average pre-injury baseline behavioral success rate of Week1 and Control animals is indicated by the dotted line with a 95% confidence interval in gray.
The primary outcome measurement was motor performance on the reaching task after treatment, as measured by the percentage of successful reaches in 60 trials. Behavioral performance in each of the three treatment groups compared with the control group is shown in Figure 4A. We observed a significant difference in the mean percentage of successful reaches on Day 57 between the treatment groups that received ADS (combined) and the control group after stimulation treatment (t(218) = 2.95; P = .004) (Supplemental Figure 2). Pairwise comparisons showed significant differences between Week1 and Week3 treatment groups versus the controls (t(218) = 4.63; P < .001, t(218) = 2.11; P = .036, respectively) but not between the Week2 treatment group versus the controls (t(218) = 1.33; P = 0.186). The estimated variance for the Week2 treatment group behavioral score was larger than for other groups.
A secondary outcome measurement was the change in mean number of successful trials between the last day prior to stimulation and the last day with stimulation on. Rats from groups Week1, Week2, and Week3 had model-estimated increases in behavioral scores of 48.96, 39.72, and 30.02, respectively, over the course of stimulation. These changes during stimulation were significantly different across these three groups (F(2,218) = 6.09; P = .003), with pairwise comparisons indicating Week1 and Week3 delays having significantly different changes from pre- to post-stimulation (t(218) = 3.43; P = .001), while Week1 versus Week2 (t(218) = 1.40; P > 0.16) and Week2 versus Week3 (t(218) = 1.28; P > 0.16) were not significant. Further, we tested whether there was an overall difference in the change in mean percentage of successful reaches over the entire duration of the study (i.e., Day 57 vs Day 3) across all four groups (F(3,218) = 5.72; P = .0009). Comparisons to control were t(218) = 4.02); P < .0001 for Week1, t(218) = 1.05; P = 0.294 for Week2, and t(218) = 1.96; P = .051 for Week3.
Persistence of ADS Treatment Effects
A randomly assigned subset of 5 Week1 rats were assessed for an additional 8 weeks after ADS ceased (Figure 4B) to identify any changes in behavior after the ADS treatment period ended. We tested whether there was a post-treatment decline in motor performance following cessation of stimulation and found no significant difference in the mean percentage of successful reaches between Day 90 and Day 35 (t(218) = 0.59; P = 0.556), indicating performance was maintained after stimulation was terminated. As illustrated in Figure 4B, during the 4-week course of ADS treatment, motor performance recovered to near baseline levels after 3 weeks, and this improved performance persisted after treatment. To evaluate whether effects were maintained following completion of treatment, we then tested whether the differences in the final behavioral assessments with stimulation and the subsequent week performance for all delay conditions equaled zero. This test resulted in P = .055 (F(3,218) = 2.57). Further examination of pairwise comparisons of these differences demonstrated similar performance between final week of stimulation and first week following its discontinuation for the Week2 stimulation (t(218) = −0.13; P = 0.898) and Week3 stimulation (t(218) = −0.71; P = 0.477) behavioral scores.
Post Hoc Neurophysiological Analysis
An analysis of variance (ANOVA) was performed to examine the effect of delay condition and week of treatment on stimulation rates. No significant difference in the interaction of delay condition or week of treatment on stimulation rate was observed (F(6,64) = 0.3247; P = 0.325). No main effects of delay condition (F(2,64) = 0.673; P = 0.672) or week of treatment (F(3,64) = 0.265; P = 0.851) were observed.
Lesion Analysis
A one-way ANOVA found no significant difference in lesion area between groups (F(3,17) = 0.61; P > 0.61). Examples of the lesion extent can be seen in Supplemental Figure 1.
Discussion
In all delay conditions (Week1, Week2, and Week3), ADS improved motor performance rapidly, with reaching success significantly greater than control animals after 1 week of stimulation and approaching baseline, pre-injury levels after 2 weeks. Therefore, this closed-loop treatment is effective at least when applied during the sub-acute (days to weeks) phase after injury. Despite having significantly higher motor performance than controls, we observed a significant drop in the speed and overall level of recovery in animals in the 3-week delay condition compared to the 1-week and 2-week delay conditions. Additional studies will be required to determine if recovery using ADS can extend beyond the sub-acute stage of injury into the chronic period. While ADS-driven recovery does not depend on the expression of acute-stage neuroplastic mechanisms, it may be more effective earlier in the sub-acute stage when the environment is more amenable to larger scale neural reorganization. Importantly, its implementation as treatment can be delayed while the immediate clinical concerns of the injury (edema, inflammation, excitotoxicity, etc.) are stabilized.
For all delay conditions, cessation of stimulation following 4 weeks of treatment did not negatively impact behavioral performance on the reaching task the following week. In a subset of the 1-week delay group, we monitored performance for 8 weeks following the end of the stimulation treatment and found no decrease in performance over the entire period. Presumably, at some point during the 4-week treatment reliance on artificial reinforcement of PM-S1 communication is reduced or eliminated and intrinsic pathways are utilized to maintain behavioral performance. While it is unknown if this persistence would also occur during a more prolonged delay between injury and treatment onset, the similarity in recovery of the three delay groups would suggest that similar results could be expected. Clearly, ADS facilitates intracortical communication that improves performance on the skilled reaching task rather than merely acting as a passive bridge and this persistent reinforcement leads to long-lasting structural and functional changes that supersede the dependence on ADS. Taken together, ADS was able to induce rapid and persistent recovery on the skilled reaching task without any additional behavioral or therapeutic interventions.
ADS utilizes Hebbian plasticity mechanisms by artificial pairing of two populations of neurons to alter functional connectivity within and between brain regions. In intact animals, this type of stimulation paradigm can lead to alterations in firing rates, patterns of neuronal firing, shifts in spiking correlations, and changes in output properties.29,33,34,36,44 While other stimulation paradigms such as paired-pulse and theta burst have been used to evoke neuroplasticity, their efficacy in promoting recovery after brain injury has been limited. We did not specifically compare these or other open-loop stimulation techniques to ADS in this study. However, using the intrinsic neural activity to drive stimulation rather than a forced, externally generated pattern allows the utilization of innate mechanisms of neuroplasticity and should result in a much more salient response. This becomes even more pronounced in our brain injury model where these alterations had clear and profound benefits for motor performance.
While we have demonstrated the effectiveness of ADS in inducing motor recovery in the acute and sub-acute phases of recovery, several mechanistic questions related to both the treatment and recovery remain. Skilled reaching relies on the coordination of proprioception, somatosensation, and motor planning; using S1 activity to trigger stimulation in PM is potentially the more physiologically relevant direction rather than PM to S1 used here. However, concerns that microstimulation in PM may disrupt motor output while performing the task dictated the decision to trigger on PM activity. Further, there are direct and indirect reciprocal connections between PM and S1.45 -47 Historically, Hebbian plasticity was described as occurring in one direction, with the post-synaptic cell facilitated into a higher likelihood of firing. Recent modeling papers have suggested that this is an overly simplistic view, and multisynaptic pathways offer a pathway for bidirectional strengthening of connections between neurons.48,49 While ADS is triggered by the activity of a single neuron within PM, it is likely that there is high synchronicity between this and other neurons within PM, and the stimulation pulse directly activates a small population of neurons. The pairing and facilitation of a single pair of neurons would not likely induce the behavioral responses that we observed; rather the coordinated activity of PM neurons and the subsequent activation of a population of S1 neurons and any reciprocal connections reinforcing these connections over time led to the rapid motor recovery.
In the sub-acute period following brain injury, the cellular environments in both peri-infarct and distant areas undergo a number of changes that allow for greater amounts of neuroplasticity, including increased synaptogenesis, dendritic arborization, motor map reorganization, and novel axonal sprouting.50 -55 In conjunction, alterations in neural activity occur, with a depressed response in peri-infarct areas, but an increase in cortical synchronization that seems to be critical for recovery to occur.51,56,57 The advantage of using ADS is then twofold: it can synchronize the activity of two disconnected brain regions which could strengthen recovery and it does this using Hebbian mechanisms that can utilize the amenable neuroplastic environment after injury.
ADS can be part of an effective solution for promoting motor recovery in the sub-acute period after injury. This period is the most amenable to neuroplasticity; by definition, the chronic injury phase is associated with a drop of the expression of neuroplastic mechanisms and a reduction in the ability for spontaneous or therapy-driven recovery to occur.58,59 Future investigation is necessary to incorporate both sexes in our experimental design to ensure that there are not sex-related differences in this response and is a limitation of the current study. In intact animals, we have demonstrated that ADS applied over time can increase the expression of synaptophysin, a marker for synaptic plasticity in the area stimulated. Other studies have suggested that, irrespective of injury, electrical cortical stimulation can induce neuroplasticity. 60 Further investigation is needed to assess whether ADS, either alone or paired with rehabilitative therapy, can overcome these limitations and provide a path for recovery in the chronically impaired.
Supplemental Material
sj-png-1-nnr-10.1177_15459683221145144 – Supplemental material for Broad Therapeutic Time Window for Driving Motor Recovery After TBI Using Activity-Dependent Stimulation
Supplemental material, sj-png-1-nnr-10.1177_15459683221145144 for Broad Therapeutic Time Window for Driving Motor Recovery After TBI Using Activity-Dependent Stimulation by Heather M. Hudson, PhD, David J. Guggenmos, PhD, Meysam Azin, PhD, Nicholas Vitale, MS, Katelyn A. McKenzie, PhD, Jonathan D. Mahnken, PhD, Pedram Mohseni, PhD, and Randolph J. Nudo, PhD, in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-2-nnr-10.1177_15459683221145144 – Supplemental material for Broad Therapeutic Time Window for Driving Motor Recovery After TBI Using Activity-Dependent Stimulation
Supplemental material, sj-tif-2-nnr-10.1177_15459683221145144 for Broad Therapeutic Time Window for Driving Motor Recovery After TBI Using Activity-Dependent Stimulation by Heather M. Hudson, PhD, David J. Guggenmos, PhD, Meysam Azin, PhD, Nicholas Vitale, MS, Katelyn A. McKenzie, PhD, Jonathan D. Mahnken, PhD, Pedram Mohseni, PhD, and Randolph J. Nudo, PhD, in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by W81XWH-16-1-0503 (PI Mohseni/Co-PI Nudo), R01NS030853 (PI Nudo), and the Landon Center on Aging.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
