Abstract
Background. Intermittent hypoxia can induce respiratory neuroplasticity to enhance respiratory motor outputs following hypoxic treatment. This type of respiratory neuroplasticity is primarily mediated by the activation of Gq-protein-coupled 5-HT2 receptors and constrained by Gs-protein-coupled 5-HT7 receptors. Objective. The present study hypothesized that the blockade of 5-HT7 receptors can potentiate the effect of intermittent hypercapnic-hypoxia on respiratory function after cervical spinal cord contusion injury. Methods. The ventilatory behaviors of unanesthetized rats with midcervical spinal cord contusions were measured before, during, and after daily acute intermittent hypercapnic-hypoxia (10 episodes of 5 minutes of hypoxia [10% O2, 4% CO2, 86% N2] with 5 minutes of normoxia intervals for 5 days) at 8 weeks postinjury. On a daily basis, 5 minutes before intermittent hypercapnic-hypoxia, rats received either a 5-HT7 receptor antagonist (SB269970, 4 mg/kg, intraperitoneal) or a vehicle (dimethyl sulfoxide). Results. Treatment with intermittent hypercapnic-hypoxia induced a similar increase in tidal volume between rats that received SB269970 and those that received dimethyl sulfoxide within 60 minutes post-hypoxia on the first day. However, after 2 to 3 days of daily acute intermittent hypercapnic-hypoxia, the baseline tidal volumes of rats treated with SB269970 increased significantly. Conclusions. These results suggest that inhibiting the 5-HT7 receptor can transiently improve daily intermittent hypercapnic-hypoxia–induced tidal volume increase in midcervical spinal contused animals. Therefore, combining pharmacological treatment with rehabilitative intermittent hypercapnic-hypoxia training may be an effective strategy for synergistically enhancing respiratory neuroplasticity to improve respiratory function following chronic cervical spinal cord injury.
Introduction
Respiratory compromise is the main cause of mortality and morbidity in patients with cervical spinal cord injury due to limited spontaneous recovery of damaged bulbospinal respiratory pathway and cervical phrenic motoneurons.1-5 Although mechanical ventilation can temporarily maintain the essential ventilation, it is an urgent priority to develop potential therapy to restore the respiratory function following cervical spinal cord injury. Several studies have proved that neurorehabilitative training with intermittent hypoxia can significantly improve the respiratory function in preclinical animal model and human patients.6-11 Studies have established that intermittent hypoxia can induce respiratory neuroplasticity to enhance respiratory motor outputs by activating Gq-protein-coupled 5-HT2 receptors in normal animals. 12 Recent reports also indicate that modulating the 5-HT system can influence the effect of intermittent hypoxia on respiratory function in rats with cervical spinal cord injuries at the chronic injured stage. More specifically, Dougherty et al 9 demonstrated that broad-spectrum serotonin antagonist (ie, methysergide) attenuated daily acute intermittent hypoxia-induced respiratory recovery in high cervical hemisected rats. Moreover, our recent study revealed that methysergide blunted the acute intermittent hypercapnic-hypoxia–induced ventilatory long-term facilitation in chronically midcervical contused rats. 11 These studies suggest that activating the serotonin system may be a critical factor in inducing respiratory neuroplasticity in animals with cervical spinal cord injury.
While the 5-HT2 receptor is involved in inducing intermittent hypoxia-induced respiratory neuroplasticity, a series of studies have shown that the 5-HT7 receptor can also modulate respiratory activity. For example, the episodic intrathecal application of a 5-HT7 agonist into the cervical spinal cord can induce phrenic motor facilitation, a type of respiratory neuroplasticity similar to intermittent hypoxia-induced phrenic long-term facilitation that progressively augments phrenic burst amplitude after treatment.13,14 Interestingly, the blockade of the 5-HT7 receptor significantly enhanced intermittent hypoxia-induced phrenic long-term facilitation, suggesting that the activation of the 5-HT7 receptor during hypoxia may constrain the expression of respiratory neuroplasticity following intermittent hypoxia. 15 However, the above-mentioned studies were conducted in normal animals, and it remains unclear whether modulation of 5-HT7 receptors can influence respiratory neuroplasticity in animals with cervical spinal cord injury. Several reports demonstrated that cervical spinal cord injury is associated with alteration of serotonergic innervation and/or receptors,16-18 which may influence the modulatory role of the serotonergic system on respiratory function. 19 Accordingly, we hypothesized that inhibiting the 5-HT7 receptor may enhance the therapeutic effectiveness of intermittent hypercapnic-hypoxia on respiratory function following cervical spinal cord injury. The aim of this study is to evaluate whether pretreatment with a 5-HT7 receptor antagonist (ie, SB269970) improves respiratory function in midcervical spinal cord contused rats with daily acute intermittent hypercapnic-hypoxia treatment (dAIH).
Methods
Animals
Thirty-eight Sprague-Dawley male rats (aged 7-8 weeks) purchased from BioLasco Taiwan Co, Ltd, were used for this study. Five animals died after cervical spinal cord contusion surgery and were thus excluded from the following experimental procedures. All experimental procedures were approved by the Institutional Animal Care and Use Committee at National Sun Yat-sen University.
Midcervical Spinal Cord Contusion
Based on the database of National Spinal Cord Injury Statistical Center, the prevalence of midcervical (C3-C5) spinal injury is higher than high (C1-C2) and low (C6-C8) cervical spinal cord injury. Therefore, the midcervical spinal cord contusion model was used in the current study. The cervical spinal cord injury surgery was performed similar to what we published previously.10,11,20,21 Briefly, animals were anesthetized with xylazine (10 mg/kg, subcutaneous [sc], Rompun, Bayer) and ketamine (140 mg/kg, intraperitoneal [ip], Ketalar®, Pfizer) at 9 to 10 weeks of age. After an absence of the toe-pinch withdrawal reflex, an incision was made in the dorsal skin and muscle to expose the C3-C4 vertebral bones, which were then clamped using Cunningham spinal adaptors and transverse clamps (Stoelting Co). The rat was then placed in a prone position on the stage of Multicenter Animal Spinal Cord Injury Study (MASCIS) Impactor System (Model II, W.M. Keck Center for Collaborative Neuroscience) followed by dorsal C3-C4 laminectomies. Unilateral midcervical spinal cord contusion was induced by releasing a rat impact rod from a height of 6.25 mm above the left C3-C4 spinal cord surface. If the animal stopped breathing after the injury, mechanical ventilation with hyperoxic gas (50% to 60% O2, balance N2; Harvard Apparatus) was provided until spontaneous breathing recovery. The surgical skin and muscle wounds were sutured with 4-0 chromic sutures and 4-0 nylon sutures, respectively. A subcutaneous injection of yohimbine (1.2 mg/kg, Tocris) was given to reverse the effect of xylazine, and lactated Ringer’s solution (5 mL, Nang Kuang Pharmaceutical Co, Ltd) was subcutaneously injected to prevent dehydration. An analgesic agent (buprenorphine, 0.03 mg/kg, sc, Reckitt Benckiser Healthcare [UK] Limited) was provided after the animal woke up from anesthesia. A daily supplement of Nutri-cal (1-3 mL, EVSCO Pharmaceuticals) and an injection of lactated Ringer’s solution (5 mL, sc) were delivered until the rats regained their preinjury body weight.
Drug Preparation
The 5-HT7 receptor antagonist (SB269970, #17081, Cayman Chemical) was dissolved in dimethyl sulfoxide (DMSO; Fisher Scientific) at 5 mg/mL and stored at −20°C. SB269970 is a potent 5-HT7 receptor antagonist and can penetrate the blood-brain barrier to exert function in the central nervous system.22,23 On the day of the experiment, the drug is diluted to 2 mg/mL in phosphate-buffered saline (PBS; UniRegion Bio-Tech). SB269970 (4 mg/kg) or DMSO vehicle (40% in PBS) was intraperitoneally injected 5 minutes before AIH or normoxia (ie, time control) treatment after 60 minutes of baseline respiratory measurements.
Whole-Body Plethysmography
Ventilatory behaviors (eg, respiratory frequency, tidal volume, and minute ventilation) of unanesthetized rats were measured using the whole-body plethysmography system (Buxco Whole Body Plethymography, Data Sciences International).10,11,24,25 for 5 consecutive days beginning at 8 weeks (57 ± 1 days) postinjury. Rats were divided into 4 groups—DMSO + dAIH (n = 10); SB269970 + dAIH (n = 10); DMSO + Normoxia (n = 7); SB269970 + Normoxia (n = 6)—for the following experimental protocols. The rat was placed in a Plexiglas chamber (#PLY4213, volume: 3.9 L) with compressed air flow (2.5 L/min) for 60 minutes and then received an intraperitoneal injection of SB269970 or DMSO. Daily acute intermittent hypercapnic-hypoxia (dAIH; 10 episodes of 5 minutes of hypoxia [10% O2, 4% CO2, balance N2; 2.5 L/min] with 5-minute intervals of normoxia) or 100 minutes of continuous normoxia (ie, time control) were applied 5 minutes postinjection followed by 60 minutes of normoxic recording. This intermittent hypercapnic-hypoxia protocol has been proven to be able to induce long-term facilitation of the tidal volume in midcervical contused rats in our previous studies.10,11
Spinal Cord Histology
Animals were anesthetized with urethane (1.6 g/kg, ip, Sigma) and systemically perfused transcardially with heparinized saline (7.5 IU/mL, 250 mL, Nang Kuang Pharmaceutical Co Ltd) followed by 4% paraformaldehyde (250 mL, Alfa Aesar) and 10% sucrose (Bio Basic Inc) in 4% paraformaldehyde (250 mL) after the final day of whole body plethysmography measurement. The cervical spinal cord was dissected and placed in 30% sucrose (Sigma) in PBS (UniRegion Bio-Tech) until the tissue sank. The spinal cord was then transversely sectioned into 40-µm slices (CM 1850, Leica), mounted on glass slides (S7441, Matsunami Glass Ind, Ltd), and stained with cresyl violet (Acros Organics). The spinal cord morphology was evaluated and imaged by an upright microscope (BX43, Olympus) connected to a charge-coupled device (CCD; DS-5Mc, Nikon). The spinal cord area and lesion area at the lesion epicenter were manually outlined and quantified using Image J software, as previously described.20,24
Data Analysis and Statistics
Animals were randomly assigned to groups before the lesion; however, some animals died during or shortly after the injury. Thus, we had to use additional animals to complete the whole experiment. In addition, the experiments were not blinded, because the first author (MJW) needed to know which animals would be receiving vehicle or 5-HT7 receptor antagonist when preparing the drug and injection procedure. However, the analysis script is written by the corresponding author (KZL), and this script can automatically analyze the respiratory response before, during, and after intermittent hypercapnic-hypoxia when the raw data were imported into the analysis script. Therefore, the present results were not biased by the experimental protocol since the analysis was done automatically by a software.
FinePointe software (Data Sciences International) was used to analyze ventilatory parameters, which were then exported to an Excel file in a 1-minute bin as previously described.10,11 A stable parameter of 10 minutes during normoxic breathing before drug/vehicle treatment was averaged and defined as the baseline value (BL). The baseline data of each day were presented as an absolute value and normalized by the baseline value at the first day (ie, % BLDay1). The acute hypoxia response at the first hypoxic treatment was defined as the averaged data during the last 2 minutes of hypoxia. The respiratory response after dAIH was presented as the averaged data between 1 and 60 minutes after intermittent hypercapnic-hypoxia. These data were expressed as an absolute value and normalized by a percentage of the baseline at each day (% BL). The normalized data (% BLDay1 and % BL) can enable us to evaluate the effectiveness of 5-HT7 receptor antagonist on modulation of ventilatory behavior (eg, a percentage change from the baseline or the first day value).
The individual data points of the ventilatory parameter before, during, and after hypoxic treatment are shown in Supplementary Figures 1 to 3. These data were analyzed using a 2-way mixed design ANOVA followed by a Dunnett’s post hoc test (factor 1: drug effect [DMSO vs SB269970]; factor 2: time points [first day to fifth day]). If data did not pass normality and equal variance test, nonparametric Mann-Whitney rank sum test and repeated-measures ANOVA on ranks would be used to evaluate the drug (DMSO vs SB269970) and time (first day to fifth day) effect, respectively. The right and left sides of the spinal cord and lesion cavity were compared using a paired t test.
All data are expressed as the mean ± standard error. A P value lower than .05 was considered statistically significant.
Results
Breathing Pattern During the Baseline
Representative examples of respiratory airflow recorded from the whole-body plethysmography during dAIH treatment are shown in Figure 1. The daily baseline respiratory frequency was similar in contused animals that received DMSO or SB269970 (Figure 2A). However, the tidal volume in animals that received SB269970 was significantly elevated to 1.77 ± 0.07 mL by the second day from its first day value of 1.54 ± 0.09 mL (P < .05; Figure 2B). The tidal volumes of rats treated with DMSO were comparable across the 5 days of dAIH administration. When the data were normalized by the baseline on the first day, tidal volumes in SB269970-treated animals significantly increased to 116.0 ± 3.7% BLDay1 and 108.9 ± 4.0% BLDay1 on the second and third days, respectively (P < .05; Figure 2E). While the tidal volume of rats treated with DMSO maintained at ~100% to 102% BLDay1 on the second to third day of dAIH. Despite the fact that the tidal volume was enhanced with SB269970, the minute ventilation was not significantly different between DMSO- and SB269970-treated rats from the first to fifth days of dAIH (Figure 2C and F).
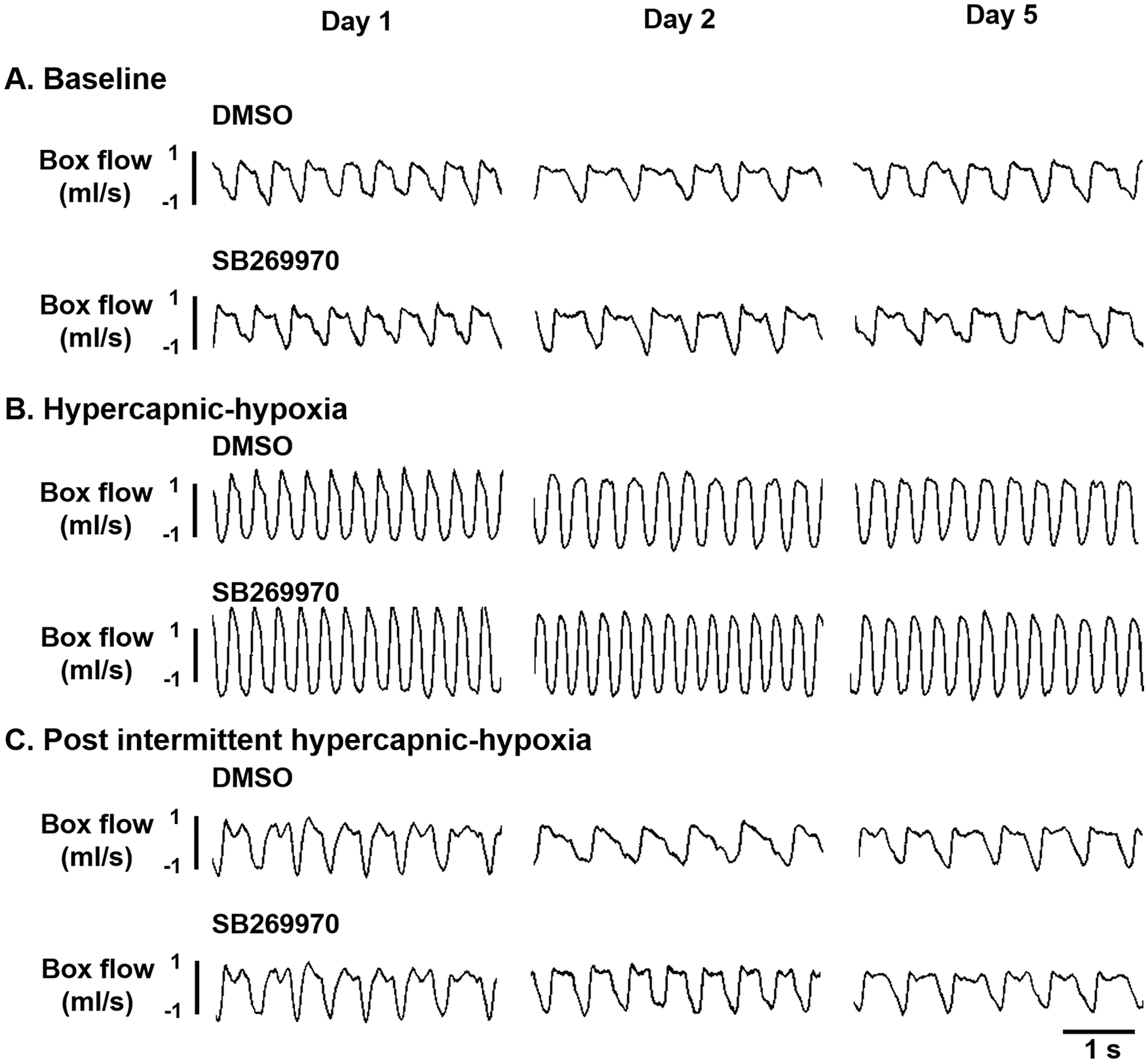
Representative examples of the respiratory airflow recorded by the whole-body plethysmography during the baseline (A), first hypercapnic-hypoxic treatment (B), and 30 minutes after acute intermittent hypercapnic-hypoxia (C) in contused rats who received dimethyl sulfoxide (DMSO) or SB269970 at the first, second, and fifth days of daily intermittent hypercapnic-hypoxia.
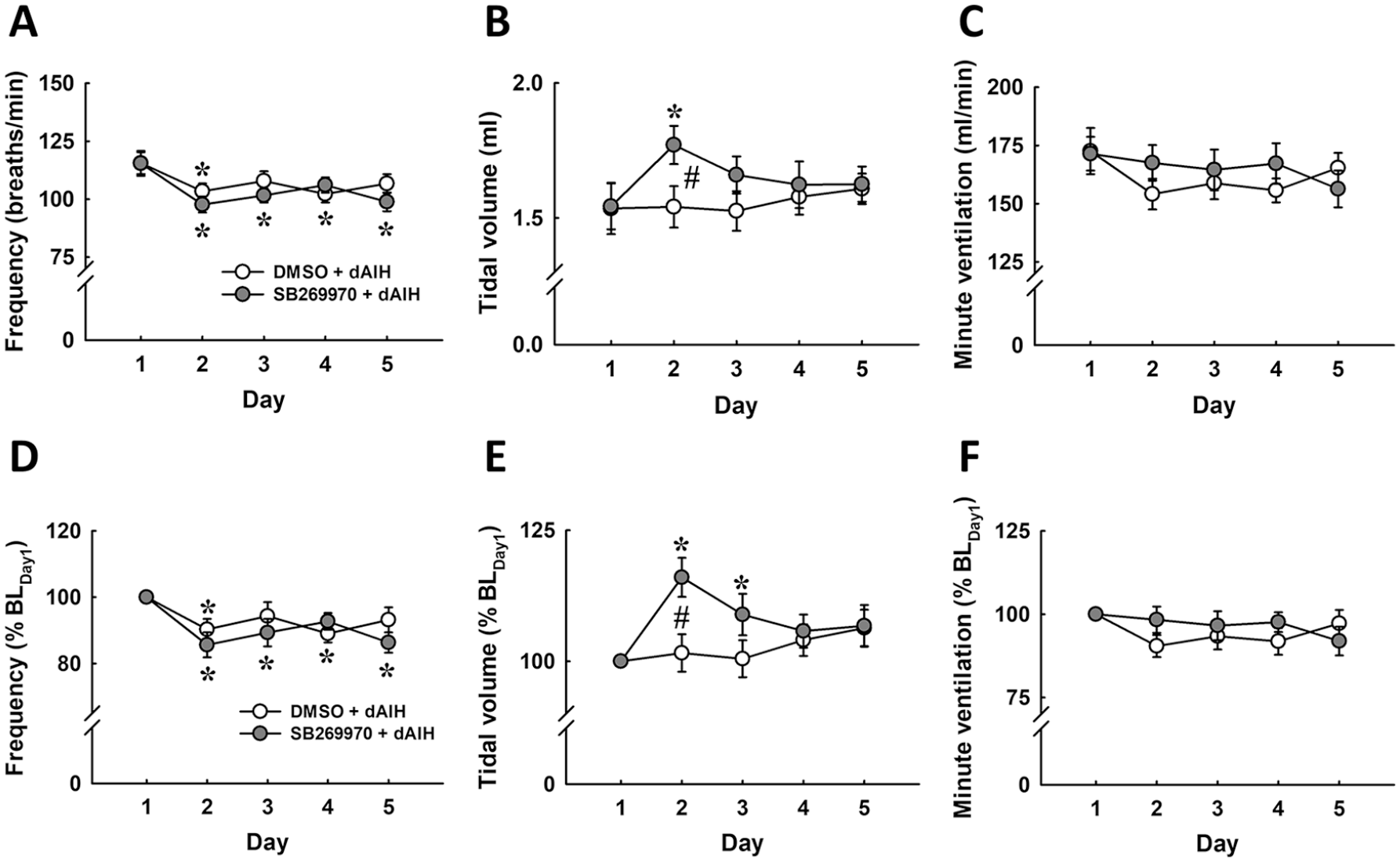
The baseline breathing pattern (frequency, tidal volume, and minute ventilation) of contused animals in response to daily acute intermittent hypercapnic-hypoxia and the administration of dimethyl sulfoxide (DMSO) or SB269970. The data were presented as an absolute value (A-C) and normalized by a percentage of the value on the first day (% BLDay1; D-F). *P < .05 versus Day 1; #P < .05 significant difference between DMSO and SB269970.
Breathing Pattern During Acute Hypercapnic-Hypoxia
The respiratory frequency, tidal volume, and minute ventilation during acute hypercapnic-hypoxia were significantly enhanced relative to the baseline parameters in DMSO- and SB269970-treated animals (P < .01; Figures 1 and 3D-F). While the hypoxic ventilatory response was similar in DMSO- and SB269970-treated groups on the first day of intermittent hypercapnic-hypoxia, the administration of SB269970 has the potential to augment the hypoxic frequency response and attenuate hypoxic tidal volume response during the second dAIH (P < .05; Figure 3D and E). Additionally, hypoxic ventilatory responses were gradually attenuated during dAIH. For example, the minute ventilation reached 545.6 ± 40.5 mL/min in DMSO-treated animals and 518.5 ± 16.2 mL/min in SB269970-treated animals on the first day (Figure 3C). By the fifth day of dAIH, the minute ventilation only increased to 413.7 ± 27.1 mL/min in DMSO-treated rats and to 405.8 ± 16.0 mL/min in SB269970-treated rats (P < .05; Figure 3C). The adaption of hypoxic minute ventilation during dAIH was similar between the 2 groups (Figure 3C and F) and was primarily caused by a significant reduction in the hypoxic tidal volume response (P < .05; Figure 3B and E).
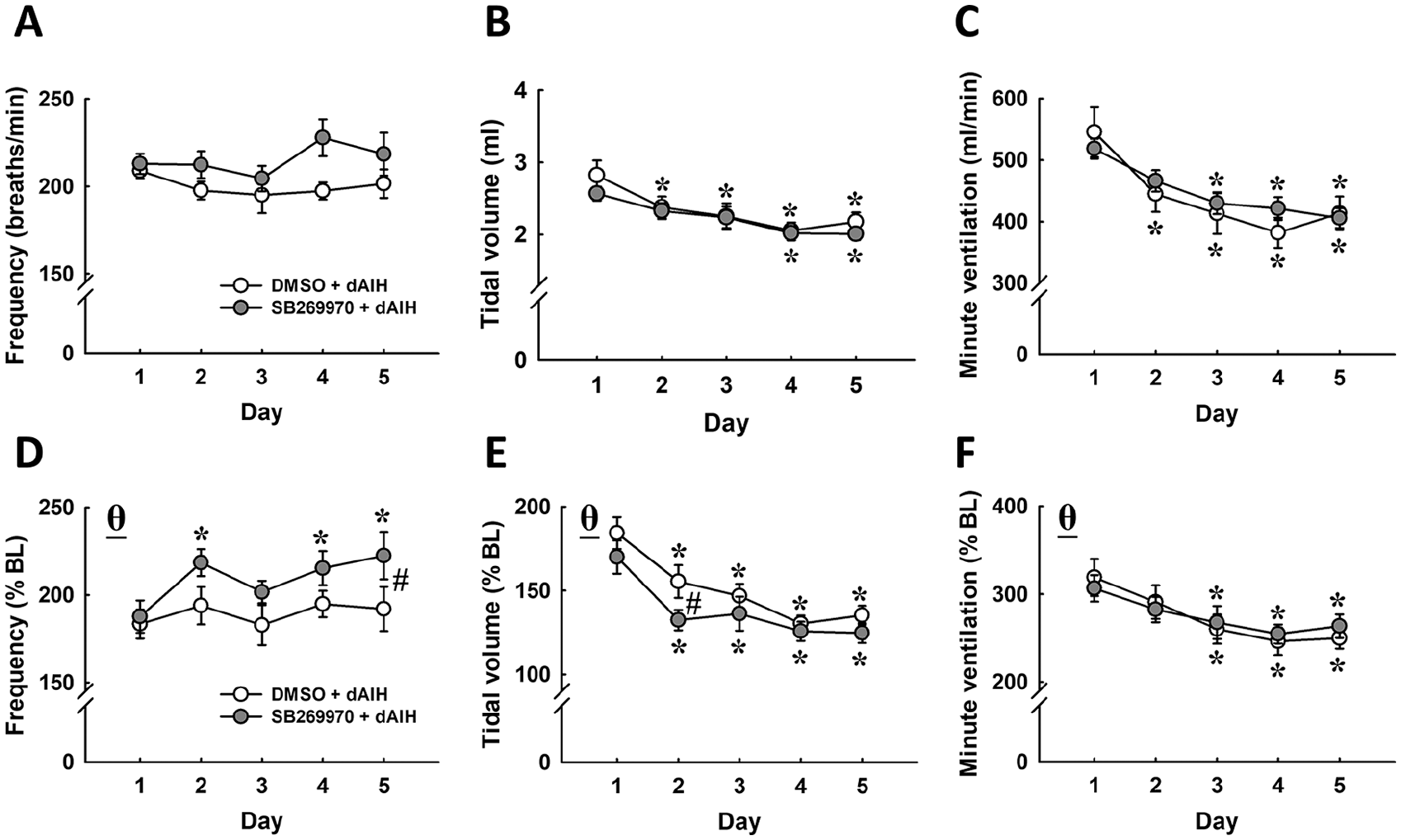
The hypercapnic-hypoxic breathing pattern (frequency, tidal volume, and minute ventilation) of contused animals in response to daily acute intermittent hypercapnic-hypoxia and the administration of dimethyl sulfoxide (DMSO) or SB269970. The data were presented as an absolute value (A-C) and normalized by a percentage of the value during the baseline (% BL; D-F). *P < .05 versus Day 1; #P < .05 significant difference between DMSO and SB269970. θP < 0.01 versus the baseline.
Breathing Pattern After Intermittent Hypercapnic-Hypoxia
Intermittent hypercapnic-hypoxia induced an immediate long-term facilitation of the tidal volume on the first day of its daily administration (P < .05; Figures 1 and 4E). Specifically, the tidal volume following intermittent hypercapnic-hypoxia increased to 114.9 ± 3.5% BL and 111.6 ± 1.5% BL in DMSO- and SB269970-treated animals, respectively (P < .05; Figures 1C and 4E). While the absolute value of the ventilatory parameters was similar for both groups from the second to fifth days of dAIH (Figure 4A-C), the ability to increase tidal volume declined in rats who received SB269970 instead of DMSO on the second day (SB269970: 100.6 ± 2.4% BL; DMSO: 113.4 ± 2.4% BL) and third day (SB269970: 107.9 ± 2.5% BL; DMSO: 118.9 ± 4.0% BL; P < .05; Figure 4E). This phenomenon may be caused by a higher baseline tidal volume in SB269970-treated rats on the second and third days of dAIH (Figures 2B and E).
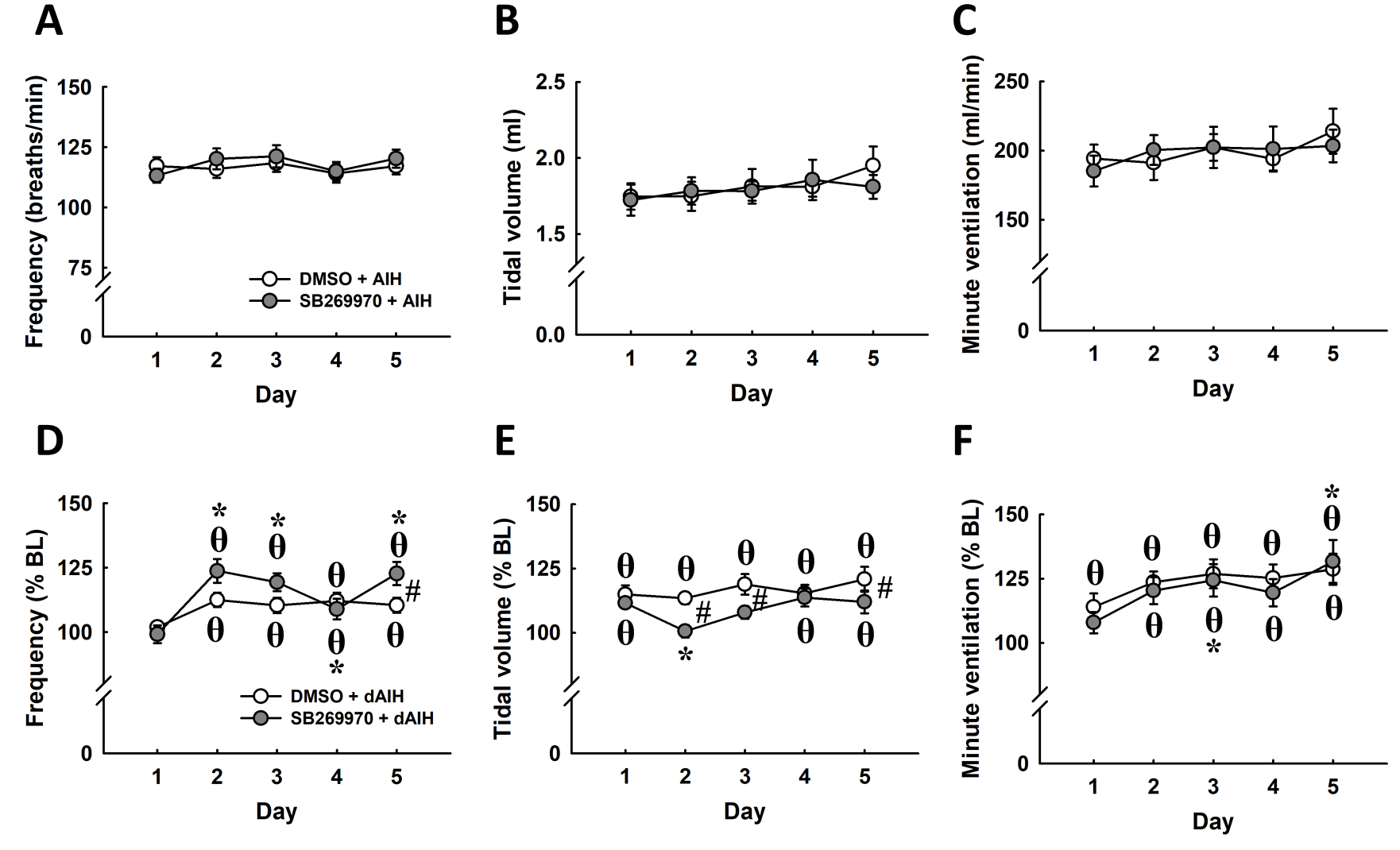
The breathing pattern (frequency, tidal volume, and minute ventilation) of contused animals in response to the administration of dimethyl sulfoxide (DMSO) or SB269970 after acute intermittent hypercapnic-hypoxia. The data were presented as an absolute value (A-C) and normalized by a percentage of the value during the baseline (% BL; D-F). *P < .05 versus Day 1; #P < .05 significant difference between DMSO and SB269970. θP < 0.05 versus the baseline.
Long-term facilitation of respiratory frequency was noticed in both DMSO- and SB269970-treated rats from the second to fifth days of dAIH (P < .05; Figure 4D). The magnitude of frequency of long-term facilitation was generally enhanced by SB269970 and proved significantly greater than the effect of DMSO on the second day (SB269970: 123.7 ± 4.6% BL; DMSO: 112.5 ± 2.8% BL) and the fifth day (SB269970: 122.7 ± 4.5% BL; DMSO: 110.4 ± 2.9% BL) of dAIH (P < .05; Figure 4D).
Breathing Pattern During Daily Normoxic Treatment
The breathing pattern of contused animals during the daily time-controlled periods of normoxia is shown in Table 1. Similar ventilatory parameters exist between contused rats who received DMSO or SB269970 from the first to fifth day of normoxic treatment. This suggests that a daily injection of SB269970 did not significantly modulate the breathing patterns of contused animals.
The Baseline Breathing Pattern of Contused Animals in Response to Normoxic Treatment (ie, Time Control).
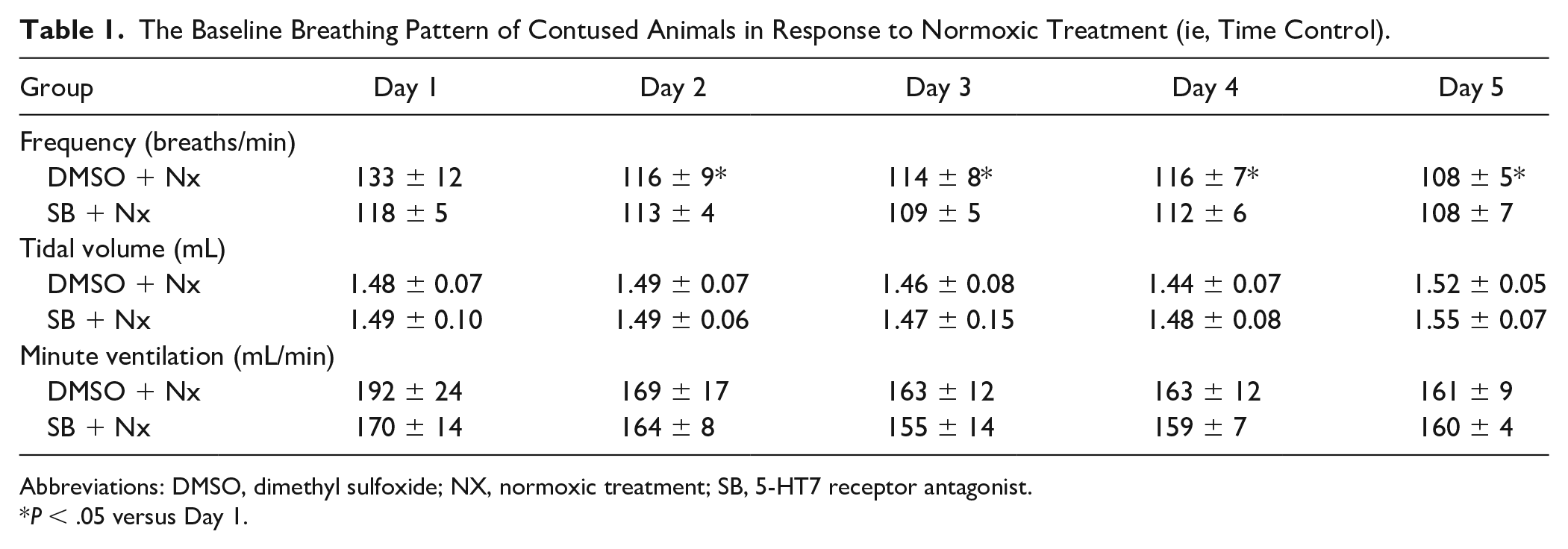
Abbreviations: DMSO, dimethyl sulfoxide; NX, normoxic treatment; SB, 5-HT7 receptor antagonist.
P < .05 versus Day 1.
Spinal Cord Histology
Three representative examples of the transverse cervical spinal section are provided in Figure 5A, B, and C, with magnification of injured areas (Figure 5D, E, and F). The area of the lesion cavity by side (left and right) was not significantly different between the 4 groups in terms of spinal area in mm2 (Figure 5G), lesion area in mm2 (Figure 5H), and lesion area (expressed in %; Figure 5I), we combined the spinal cord histological data by side in order to show the impact of unilateral cervical spinal cord contusion on spinal cord morphology. Notably, the spinal cord area is substantially smaller on the left (0.86 ± 0.04 mm2) compared with the right (1.83 ± 0.09 mm2) side (Figure 5G). Gray and white matter in the left spinal cord were significantly lesioned by the contusion injury. The lesion cavity of the left spinal cord at the lesion epicenter is 0.27 ± 0.04 mm2 (Figure 5H; corresponding to 28.5 ± 3.2% hemi-cord; Figure 5I), which is significantly greater than the right spinal cord lesion (0.05 ± 0.02 mm2, 2.9 ± 1.2% hemi-cord; P < .01; Figure 5H and I).
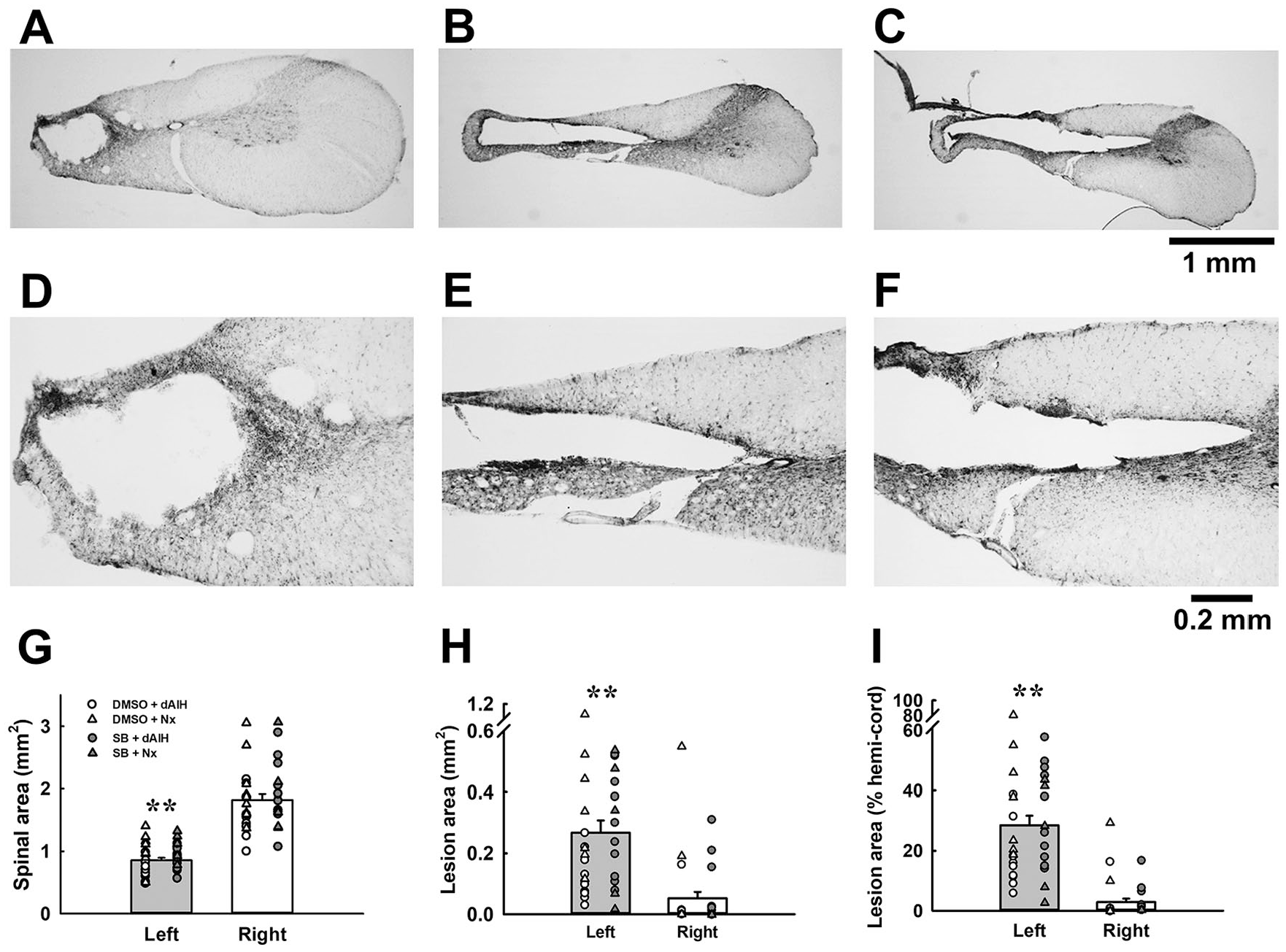
The morphology of the cervical spinal cord section following midcervical contusion. Panels A to C are representative examples of the transverse spinal cord section showing different lesion severity. Panels D to F present the enlarged view of lesion site in panels A to C. Panels G and H demonstrate the absolute value of the area of the bilateral spinal cord and the lesion, respectively. Panel I presents the area of the lesion normalized by the unilateral spinal cord area (% hemi-cord). Individual data points of all groups were included in panel I. **P < .01 significant difference between left and right side.
Discussion
The present study demonstrates that dAIH can induce long-term facilitation of minute ventilation following hypercapnic-hypoxia treatment in chronic contused animals (Figure 4F). In addition, the administration of a 5-HT7 receptor antagonist (ie, SB269970, 4 mg/kg, ip) may prolong the facilitatory effect of acute intermittent hypercapnic-hypoxia on tidal volume (Figure 4B and F). These results indicate that dAIH may be a feasible rehabilitative strategy for improving the respiratory function at chronic injured stage; moreover, a pretreatment with a 5-HT7 receptor inhibitor may strengthen the respiratory neuroplasticity and therefore augment the therapeutic effectiveness of intermittent hypercapnic-hypoxia.
Critique of Methods
Several concerns about our experimental approach must be addressed to responsibly interpret our findings. First, this study administered the 5-HT7 receptor antagonist (ie, SB269970) via an intraperitoneal injection. 5-HT7 receptors are widely distributed in the central nervous system (ie, hypothalamus, thalamus, cortex) and peripheral tissue (ie, smooth muscle and intestine). 26 The wide distribution of 5-HT7 receptors means that we remain unsure of which specific area mediates the modulatory effect of SB269970 on the breathing patterns observed with dAIH. Fields et al 27 have demonstrated that phrenic motoneurons exhibit 5-HT7 receptors. Moreover, acute intermittent hypoxia-induced phrenic long-term facilitation could be enhanced after the blockade of the spinal 5-HT7 receptor in anesthetized and ventilated rats. 15 Accordingly, we speculate that SB269970 may at least inhibit the activation of 5-HT7 receptors in the phrenic motoneurons. Second, 4 isoforms of 5-HT7 receptors (ie, 5-HT7a, 5-HT7b, 5-HT7c, and 5-HT7d) exist, all of which exhibit similar functional and pharmacological profiles.28,29 It thus remains to be determined which specific isoform of 5-HT7 receptor mediated the influence of SB269970 on the breathing pattern following dAIH. Third, Krobert and Levy 30 have demonstrated that 5-HT7 receptors can display constitutive activity to enhance adenylyl cyclase activity in the absence of 5-HT. Furthermore, because SB269970 exhibits both antagonistic and inverse agonistic functions,31,32 the effect of SB269970 on the dAIH-induced respiratory function may be due to the blockade of endogenous 5-HT acting on 5-HT7 receptors and/or the attenuation of constitutive 5-HT7 receptor activity. Fourth, a previous report demonstrated that SB269970 can also block noradrenaline-induced inhibitory post-synaptic potential and hyperpolarization in submucosal neurons of the guinea pig. 33 Therefore, we cannot exclude that the effect of SB269970 in the present study could be partially mediated by inhibition of α2 adrenoreceptor.
5-HT7 Receptor Inhibition Improves dAIH-Induced Tidal Volume
Baseline tidal volume did not significantly change following dAIH, suggesting that 4 continuous days of acute intermittent hypercapnic-hypoxia may not be sufficient to progressively enhance the baseline respiratory function in midcervical contused rats at the chronic injured stage. Previous studies have shown that the tidal volume of high-cervical hemisected rats can be improved with 7 days of daily acute intermittent hypoxia.8,9,34 These results suggest that at least 1 week of repetitive acute intermittent respiratory stimulus is necessary to improve baseline tidal volume a few days after treatment despite the fact that a single AIH can significantly elevate tidal volume within 60 minutes treatment.
The current results demonstrate that the tidal volume of contused rats that received SB269970 were significantly greater than those of the rats that received DMSO on the second and third days of dAIH. This phenomenon indicates that inhibiting the 5-HT7 receptor may potentiate the effect of dAIH on tidal volume in midcervical contused animals. Notably, the absolute value of tidal volume during hypercapnic-hypoxia treatment was similar between contused rats who received DMSO and those who received SB269970. This suggests that the 5-HT7 receptor may not be involved in central/peripheral chemosensitivity and the modulation of respiratory drives to spinal respiratory motoneurons during respiratory challenge following chronic spinal contused injury. In addition, we noticed that the tidal volume following acute intermittent hypercapnic-hypoxia was also similar between DMSO- and SB269970-treated groups despite the fact that baseline tidal volume on the second and third days of dAIH was greater in contused rats that received SB269970. These results suggest that the potentiated effect of SB269970 on the baseline tidal volume may be due to the prolongation or maintenance but not the augmentation of the magnitude of intermittent hypercapnic-hypoxia–induced tidal volume long-term facilitation at the chronic injured stage.
The Potential Mechanism Underlying the Effect of 5-HT7 Receptor Inhibition on dAIH-Induced Tidal Volume Improvement
Our recent study, together with several others, show that the beneficial effect of acute intermittent hypoxia or hypercapnic-hypoxia–induced respiratory function could be attenuated by the administration of a broad-spectrum 5-HT receptor antagonist (eg, methysergide) in chronic cervical spinal injured rats. This suggests that 5-HT is a primary mediator contributing to AIH-induced respiratory neuroplasticity at the chronic injured stage.9,11 However, Komnenov et al 35 demonstrate that AIH can still promote respiratory function in tryptophan hydroxylase 2–deficient mice with cervical spinal cord injuries. This finding indicates that other mediators may also contribute to acute intermittent hypoxia-induced improvement of respiratory function following spinal cord injury. Indeed, the episodic or single administration of several molecules (eg, α1 adrenergic receptor agonist, adenosine 2A receptor agonist, vascular endothelial growth factor, and erythropoietin) into the spinal cord have also been shown to induce phrenic motor facilitation.36-39 Therefore, the above-mentioned factors could also contribute in the augmentation of tidal volume following dAIH in chronic injured animals.
The molecular mechanisms underlying intermittent hypoxia-induced phrenic long-term facilitation in anesthetized rats have been extensively investigated by Mitchell and his colleagues. Briefly, the 5-HT2A receptors of phrenic motoneurons were activated by 5-HT released from the raphe during acute intermittent hypoxia. The activation of 5-HT2A receptors triggers a series of signal cascades (ie, the activation of protein kinase C, the synthesis of the brain-derived neurotrophic factor, the activation of tyrosine kinase receptors, and the activation of the phosphor-extracellular signal regulated kinase) to enhance phrenic motoneuronal excitability and thus increase phrenic burst amplitude.40,41 Interestingly, the blockade of 5-HT7 receptors and the inhibition of the 5-HT7-associated protein kinase A can enhance acute intermittent hypoxia-induced phrenic long-term facilitation in anesthetized rats, suggesting that 5-HT7 receptors constrain hypoxia-induced phrenic neuroplasticity through PKA- and cAMP-mediated signal transduction.14,15 Our current study provides compatible evidence, demonstrating that inhibiting the 5-HT7 receptors can improve tidal volume in animals with midcervical contusions during dAIH. These results lead us to speculate that the daily administration of a 5-HT7 receptor antagonist may reduce spinal PKA level and subsequently potentiate dAIH-induced respiratory neuroplasticity through the 5-HT2 signal cascade in the present experimental model. In addition, alteration of phrenic motoneuronal excitability following cervical spinal cord injury may also potentiate the effect of dAIH and/or 5-HT7 receptor antagonist.42-44 However, since our injury leads to a phrenic motoneuronal loss on the injured side,10,11 we cannot rule out that the phrenic neuroplasticity that sustains the tidal volume increase in response to dAIH could happen into the phrenic motoneurons in the intact side, 45 as well as other respiratory-associated muscles such as the intercostals.21,46,47 In addition, supraspinal respiratory-related nucleus and/or spinal interneurons may be also involved in modulating the tidal volume in our current experimental model.2,48-50
While rats that received SB269970 had a greater baseline tidal volume compared to that observed on the first day of dAIH, this improvement in tidal volume was gradually attenuated and was no longer observed by the fourth and fifth days of dAIH. These results suggest that SB269970 only had a transient effect and was unable to persistently improve dAIH-induced tidal volume at the chronic injured stage. Krobert et al 51 have demonstrated that the pre-incubation of SB269970 for 24 hours (but not for 1 hour) significantly attenuated basal adenylyl cyclase activity in 5-HT7 receptors expressed in HEK293 cells. Moreover, the 5-HT-induced activation of adenylyl cyclase activity was also reduced in the presence of SB269970. 51 In addition, the density of the 5-HT7b receptor was reduced in response to SB269970 treatment under in vitro conditions. 31 Accordingly, a daily injection of SB269970 may reduce the expression of the 5-HT7 receptor and/or associated intracellular signaling, which further attenuate the effect of SB269970 on tidal volume during dAIH. Investigation of the mechanism underlying the attenuated SB269970 effect is needed for the future drug development.
Physiological Significance
Both animal and preclinical studies have shown that acute intermittent hypoxia can significantly enhance respiratory function following chronic cervical spinal cord injury.6,7,10,11,46 However, Tester et al 7 indicate that the administration of daily acute intermittent hypoxia for 10 days did not enhance the magnitude of ventilatory long-term facilitation in humans living with chronic spinal injuries. In addition, the baseline tidal volume of these patients with spinal cord injuries was similar on the initial and final days of daily intermittent hypoxia. These findings suggest that other factors exist that limit the augmentation of respiratory motor outputs following daily acute intermittent hypoxia. Our current experiments in the preclinical spinal injured model indicate that the inhibition of the 5-HT7 receptors can potentiate the therapeutic effectiveness of acute intermittent hypercapnic-hypoxia, suggesting that a combination of intermittent hypercapnic-hypoxia training and pharmacological administration may be a potential strategy to enhance intermittent hypercapnic-hypoxia–induced respiratory neuroplasticity. 52 Moreover, several reports already demonstrated that intermittent hypoxia can also improve nonrespiratory motor function.53-57 Therefore, it will be of great value to evaluate whether 5-HT7 inhibition can generally improve the intermittent hypoxia-induced respiratory and locomotor function in the future study.
Supplemental Material
Supp._Figure – Supplemental material for 5-HT7 Receptor Inhibition Transiently Improves Respiratory Function Following Daily Acute Intermittent Hypercapnic-Hypoxia in Rats With Chronic Midcervical Spinal Cord Contusion
Supplemental material, Supp._Figure for 5-HT7 Receptor Inhibition Transiently Improves Respiratory Function Following Daily Acute Intermittent Hypercapnic-Hypoxia in Rats With Chronic Midcervical Spinal Cord Contusion by Ming-Jane Wu, Stéphane Vinit, Chun-Lin Chen and Kun-Ze Lee in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by grants from the Ministry of Science and Technology (MOST 105-2628-B-110-002-MY3, 109-2636-B-110-001 and 108-2636-B-110-001), Higher Education Sprout Project (07C030111), and NSYSU-KMU Joint Research Project (108-I005 and 109-I007).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
