Abstract
Background. Mild intermittent hypoxia has been considered a potential approach to induce respiratory neuroplasticity. Objective. The purpose of the present study was to investigate whether mild acute intermittent hypoxia can improve breathing function in a clinically relevant spinal cord injury animal model. Methods. Adult male rats received laminectomy or unilateral contusion at the C3-C4 spinal cord using a MASCIS Impactor (height: 6.25 or 12.5 mm). At 4 weeks postinjury, the breathing patterns of unanesthetized rats were measured by whole body plethysmography before, during and after 10 episodes of 5 minutes of hypoxia (10% O2, 4% CO2, balance N2) with 5 minutes of normoxia intervals. Results. The results demonstrated that cervical contusion resulted in reduction in breathing capacity and number of phrenic motoneurons. Acute hypoxia induced significant increases in frequency and tidal volume in sham surgery and contused animals. In addition, there was a progressive decline in the magnitude of hypoxic ventilatory response during intermittent hypoxia. Further, the tidal volume was significantly enhanced in contused but not sham surgery rats at 15 and 30 minutes postintermittent hypoxia, suggesting intermittent hypoxia can bring about long-term facilitation of tidal volume following cervical spinal contusion. Conclusions. These results suggest that mild acute intermittent hypoxia can elicit differential forms of respiratory plasticity in sham surgery versus contused animals, and may be a promising neurorehabilitation approach to improve respiratory function after cervical spinal cord injury.
Introduction
Cervical spinal cord injury (SCI) usually causes respiratory complications1-4 based on interruption of respiratory bulbospinal pathways and compromise of spinal respiratory motoneurons.5-8 The most extensively studied cervical SCI model is unilateral hemisection at the second cervical spinal cord (ie, C2 hemisection). This injury model usually induces a rapid shallow breathing pattern and inactivation of phrenic and/or diaphragmatic outputs during an acute injury state.9-11 However, both respiratory outputs (eg, phrenic activity) and respiratory behaviors (eg, tidal volume) can partially recover weeks to months after SCI.10,12-14 It is documented that intermittent hypoxia can influence many physiological functions. Although severe intermittent hypoxia (<9% O2, >50 episodes per day) leads to deleterious effects on cardiorespiratory functions, mild intermittent hypoxia (9%-16% O2, 3-15 episodes per day) has been utilized to improve endogenous recovery following high-cervical SCI.15-19 For example, Golder and Mitchell 20 demonstrated that mild acute intermittent hypoxia (mAIH, 3 × 5 minutes 10%-12% O2) can induce long-term facilitation (LTF) in phrenic motor output ipsilateral to the lesion in rats at 8 weeks post-C2 hemisection. Both respiratory and somatic motor function was improved after daily mAIH (10 × 5 minutes 10.5% O2 for 7 days) in C2 hemisected rats. 21 Moreover, a daily mAIH paradigm can elicit functional recovery of tidal volume in unanesthetized rats at 2 weeks following high-cervical hemisection. 11 This evidence suggests that mAIH is an effective neurorehabilitative approach to promote respiratory function in neurological disorders.
The majority of human SCI-affected regions and types is midcervical contusion.22,23 Unlike a C2 hemisection that only interrupts respiratory bulbospainal respiratory pathways, though preserves intact phrenic nuclei, 24 midcervical contusions directly damage phrenic motoneuron pools and a number of bulbospinal pathways.25,26 Therefore, phrenic outputs are unable to appropriately respond to respiratory challenge following cervical contusion.25,27 Although previous studies have shown that mAIH can improve respiratory recovery subsequent to high-cervical spinal hemisection, it remains unclear what the impact of mAIH is on respiratory function in a more clinically-relevant midcervical contusion animal model. Notably, recent reports demonstrated that mAIH can induce ventilatory long-term facilitation in spinal cord injured patients,1,28 suggesting it may be feasible to apply mAIH to the patient population. However, the mechanism underlying mAIH-induced respiratory neuroplasticity in incomplete SCI model remained unclear. Moreover, it has been shown that combination of mAIH and other therapies (eg, cellular transplantation and pharmacological intervention) can synergistically improve the breathing function in the spinal hemisected animal model,15,29 but whether combinatorial therapies were more effective when the phrenic motoneurons were lost (ie, contusion model) is unclear. Accordingly, it is essential to establish a clinically relevant spinal cord contusion animal model as a proof-of-principle platform to evaluate the therapeutic effectiveness of mAIH on respiratory recovery.
Methods
All experimental protocols were approved by the Institutional Animal Care and Use Committee at National Sun Yat-sen University.
Animals
Fifty-seven male Sprague-Dawley rats (aged 7-8 weeks) purchased from BioLasco Taiwan Co, Ltd, were used in the present study and divided into sham surgery (ie, C3-C4 laminectomy, n = 18), moderate (n = 18), and severe (n = 21) contusion groups.
Spinal Cord Contusion
Animals were anesthetized with xylazine (10 mg/kg, subcutaneous [s.c.], Rompun, Bayer) and ketamine (140 mg/kg, intraperitoneal [i.p.], Ketalar, Pfizer) at 8 to 9 weeks of age and placed in the prone position. The cervical vertebrate was exposed by incisions in dorsal skin and muscle after confirming absence of the toe-pitch withdrawal reflex. The C3-C4 vertebral bones were immobilized by a Cunningham spinal adaptor and transverse clamps (Stoelting Co), which were mounted on a stereotaxic instrument (David Kopf Instruments). The animal was placed on a stage of Multicenter Animal Spinal Cord Injury Study (MASCIS) Impactor System (Model II, W.M. Keck Center for Collaborative Neuroscience) followed by dorsal C3-C4 laminectomies. Contusion injury was induced by releasing a rat impact rod at height of 6.25 mm (moderate injury) or 12.5 mm (severe injury) above the surface of left C3-C4 spinal cord. The animal was ventilated with hyperoxic gas (50% O2, balance N2) (Harvard Apparatus) if breathing ceased after contusion until the animal was able to spontaneously breath again. The surgical wound was closed with 4-0 chromic sutures for the muscle and 4-0 nylon sutures for the skin. Subcutaneous injections of yohimbine (1.2 mg/kg, Tocris) and lactated Ringer’s solution (5 mL, Nang Kuang Pharmaceutical Co, Ltd) were provided to reverse action of xylazine and to prevent dehydration, respectively. An analgesic agent (buprenorphine, 0.03 mg/kg, s.c., Shinlin Sinseng Pharmaceutical Co, Ltd) was given after the animal woke from anesthesia. An oral supply of Nutri-cal (1-3 mL, EVSCO Pharmaceuticals) and lactated Ringer’s solution injection (5 mL, s.c.) were supplied daily until the animal recovered their appetite.
One moderate contused and 5 severe contused animals did not survive at 1 day after injury. The survival rate was 94% (17/18) in moderate contused group and 76% (16/21) in severe contused group.
Whole-Body Plethysmography
Four weeks after spinal cord surgery, ventilatory behaviors (eg, respiratory frequency, tidal volume and minute ventilation) of unanesthetized rats (sham surgery: 448 ± 9 g; moderate contusion: 430 ± 10 g; severe contusion: 360 ± 13 g) were measured using a whole-body plethysmography system (Buxco Whole Body Plethymography, Data Sciences International) before, during and after mAIH. Animals were placed in a Plexiglas chamber (#PLY4213, volume: 3.9 L) with compressed air flow (2.5 L/min) for 1 hour. Ten episodes of 5-minutes hypoxia (10% O2, 4% CO2, balance N2; 2.5 L/min) with 5-minute normoxia intervals (sham surgery, n = 11; moderate contusion, n = 11; severe contusion, n = 11) or 10 episodes of 5-minute normoxia (21% O2, balance N2; 2.5 L/min) with 5-minute normoxia intervals (sham surgery, n = 7; moderate contusion, n = 6; severe contusion, n = 5) were introduced to the chambers followed by 1 hour of normoxic recording. Hypoxia can induce an increase in the ventilation, which further causes a reduction in PaCO2 and may attenuate the hypoxia-induced ventilatory response. A previous report indicated that ventilatory LTF could not be induced following intermittent hypoxia unless a slightly inhaled CO2 was included in awake human. 30 In addition, intermittent isocapnic hypoxia (8%-9% O2 + 4% CO2) has been effectively induced ventilatory LTF in awake rats. 31 Accordingly, the present study used 10% O2 + 4% CO2 as the stimuli parameter of intermittent hypoxia.
Spinal Injury Severity
Thirty animals (sham surgery, n = 10; moderate contusion, n = 10; severe contusion, n = 10) were employed to evaluate spinal cord morphology and injury severity. After ventilatory behavior measurements, animals were anesthetized with urethane (1.6 g/kg, i.p., Sigma) and systemically perfused with heparinized saline (5.5 IU/mL, 250 mL), followed by 4% paraformaldehyde (250 mL, Alfa Aesar) and then 10% sucrose in 4% paraformaldehyde (250 mL). The C2-C5 spinal cord was dissected and immersed in 30% sucrose in phosphate-buffered saline. The spinal cord was cryoprotected and transversely sectioned into 40-µm slices (CM 1850, Leica) after the tissue sank. The spinal cord sections were mounted on glass slides and stained with cresyl violet (Acros Organics). The spinal cord section was imaged using a microscope (BX43, Olympus) connected to a charge-coupled device (CCD, DS-5Mc, Nikon). The lesion epicenter was defined as the section with the largest lesion cavity relative to total spinal cord area in the same section. The area of total spinal cord section and lesion cavity of the right and left spinal cord was manually outlined and quantified using Image J software. The data are presented as mm2 and as a percentage of ipsilateral hemicord area (ie, % hemicord).
Phrenic Motoneuron Labeling
A subset of animals (sham surgery, n = 5; moderate contusion, n = 6; severe contusion, n = 5) was used to evaluate the impact of contusion on the number of phrenic motoneurons. After ventilatory behavior measurements, animals were anesthetized with xylazine (10 mg/kg, s.c.) and ketamine (140 mg/kg, i.p.) and laparotomy was performed to expose the diaphragm. A 40 µL monosynapic retrograde tracer (0.05%, cholera toxin subunit B conjugated with Alexa Fluro 594 [#C34777, ThermoFisher Scientific]) was painted on the bilateral diaphragm as previously described.32,33 The surgical wound was closed with sutures and postsurgical care protocols were the same as already mentioned. 3 days following tracer application, the animals were perfused under urethane anesthesia and the spinal cord tissue was horizontally sectioned into 40 µm slices using a Cryostat or vibratome. The spinal cord sections were mounted on glass slides and cover-slipped with mounting medium containing 4′,6-diamidino-2-phenylindole (DAPI, Sigma). The tracer-labeled phrenic motoneurons were detected by a microscope (BX43, Olympus) with a fluorescence filter cube (U-FMCHE, Olympus). Microphotographs were taken with a CCD (DS-5Mc, Nikon) and the number of tracer-labeled phrenic motoneurons were counted manually.
Data Analysis and Statistics
The ventilator parameters were calculated using FinePointe software (Data Sciences International) and exported to an Excel file in a 1-minute bin. The data during normoxic breathing before mAIH treatment were averaged over 10 minutes as a baseline value (BL). The first acute hypoxic responses were represented by the data averaged during the last 2 minutes of hypoxia and the 4 to 5 minutes after hypoxia (ie, last 2 minutes of normoxia after first hypoxia). To evaluate whether mAIH induced LTF in ventilation, the data were averaged over 5 minutes at 15, 30, and 60 minutes after the last hypoxic treatment. The baseline breathing patterns between groups were analyzed by a 1-way analysis of variance (ANOVA) followed by a Student-Newman-Keuls (SNK) post hoc test. The acute hypoxic responses were assessed using a 2-way mixed design ANOVA followed by a SNK post hoc test (factor 1: groups; factor 2: time point [ie, baseline, during, and following hypoxia]). A 1-way repeated measures ANOVA followed by a SNK post hoc test was employed to examine the alteration of ventilation in response to successive hypoxic treatments. A 2-way mixed design ANOVA followed by a SNK post hoc test was used to compare ventilatory behaviors between animals that received mAIH and those that were supplied with intermittent normoxia (factor 1: treatment; factor 2: time point).
The spinal cord section area, injury area, and number of phrenic motoneurons between groups was evaluated with a 2-way mixed design ANOVA followed by a SNK post hoc test (factor 1: groups; factor 2: right vs left side).
All data are expressed as the mean ± standard error of the mean. A P value less than .05 was considered statistically significant.
Results
Spinal Cord Histology
Representative examples of transverse cervical spinal sections are provided in Figures 1A-C. The area of the left-side (ie, injured side) spinal section was 1.3 ± 0.2 mm2 in moderate and 1.2 ± 0.1 mm2 in severely contused group, significantly lower than the area in sham surgery group (2.5 ± 0.1 mm2) (P < .01, Figure 1D). Although the right spinal cord was not directly contused by the impactor rod, the area of the right spinal section was also lower in contused animals versus sham surgery animals (P < .01, Figure 1D). The contusion injury predominantly disrupted the gray matter, and the lesion area of the left spinal cord was 0.35 ± 0.08 mm2 (27.5% ± 3.9% hemicord) and 0.59 ± 0.11 mm2 (48.4% ± 5.6% hemicord) in moderately and severely contused groups, respectively (Figures 1E and F). The injury severity of the left spinal cord was greater in severe contusion than moderate contusion group (P < .05, Figures 1E and F). The contusion injury also extended to the right spinal cord in severe contusion group. The injury area in the right spinal cord of severely contused animals was 0.12 ± 0.04 mm2 (8.9 ± 2.8% hemicord).
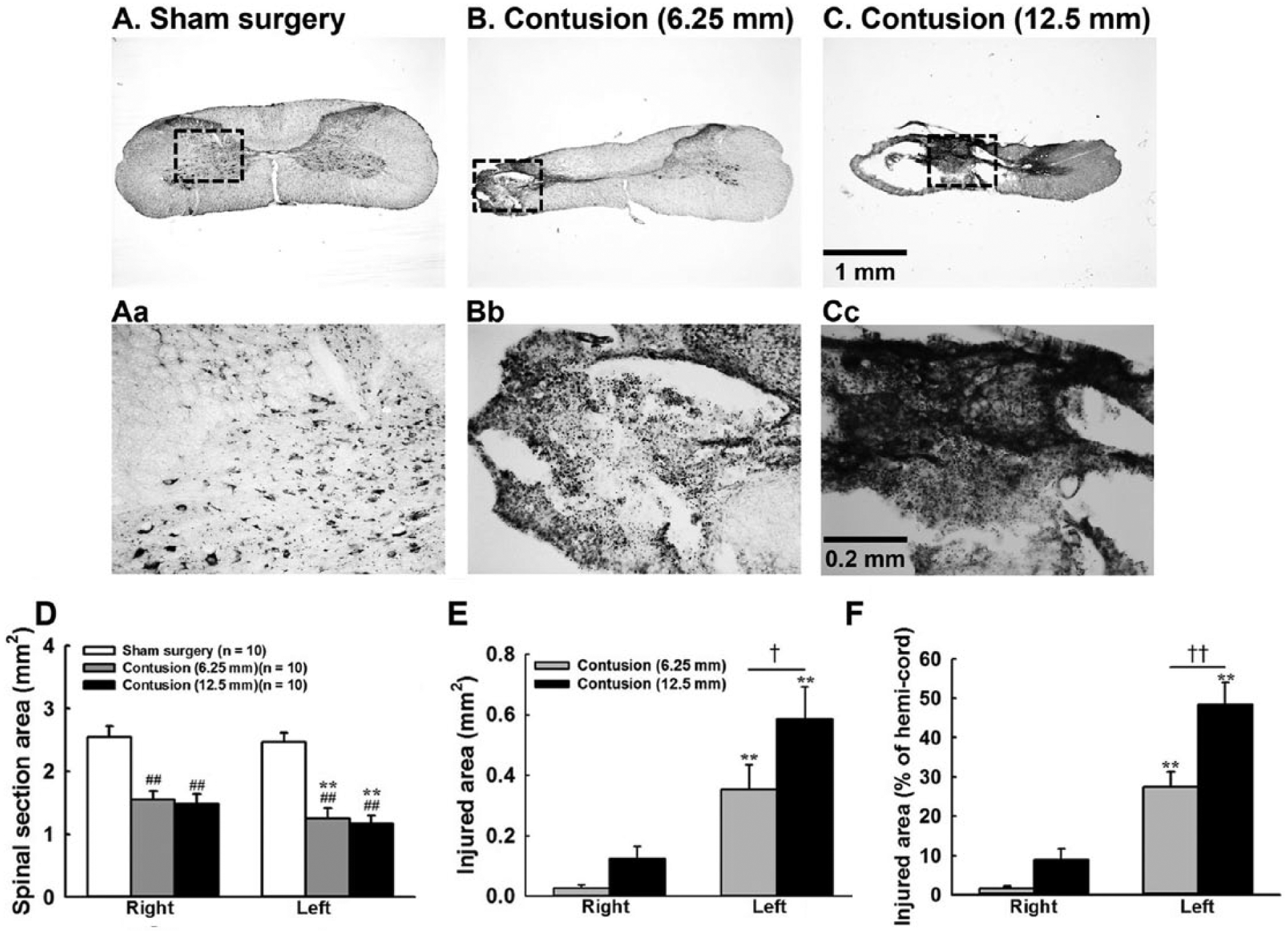
Representative examples of transverse midcervical spinal cord sections in sham surgery (A), moderately contused (B), and severely contused (C) animals at 4 weeks postinjury. Aa, Bb, and Cc represented the high magnificent image labeled by the dash square in panels A-C. Panel D represents the averaged value of total spinal cord section area. Panels E and F depict the averaged injured area expressed in mm2 and percentage of hemicord, respectively. **P < .01 significant difference between right and left sides. ##P < .01 significant difference from sham surgery animals. †P < .05; ††P < .01 significant difference between moderately (6.25 mm) and severely (12.5 mm) contused animals.
Phrenic Motoneuron Labeling
Representative examples of horizontal cervical spinal sections are seen in Figures 2A-C. The contusion injury caused a significant cavitation in the left spinal cord (Figures 2B and C, left panel). The number of tracer-labeled phrenic motoneuron of the right spinal cord was lower in moderately (154 ± 18) and severely contused groups (154 ± 24) than sham surgery group (220 ± 28); however, this difference did not reach significance (P = .112, Figure 2D). On the contrary, animals that received contusion had a significantly lower number of tracer-labeled phrenic motoneurons on the left side (moderate contusion: 101 ± 17; severe contusion: 115 ± 18) compared to sham surgery animals (201 ± 22) (P < .01, Figure 2D).
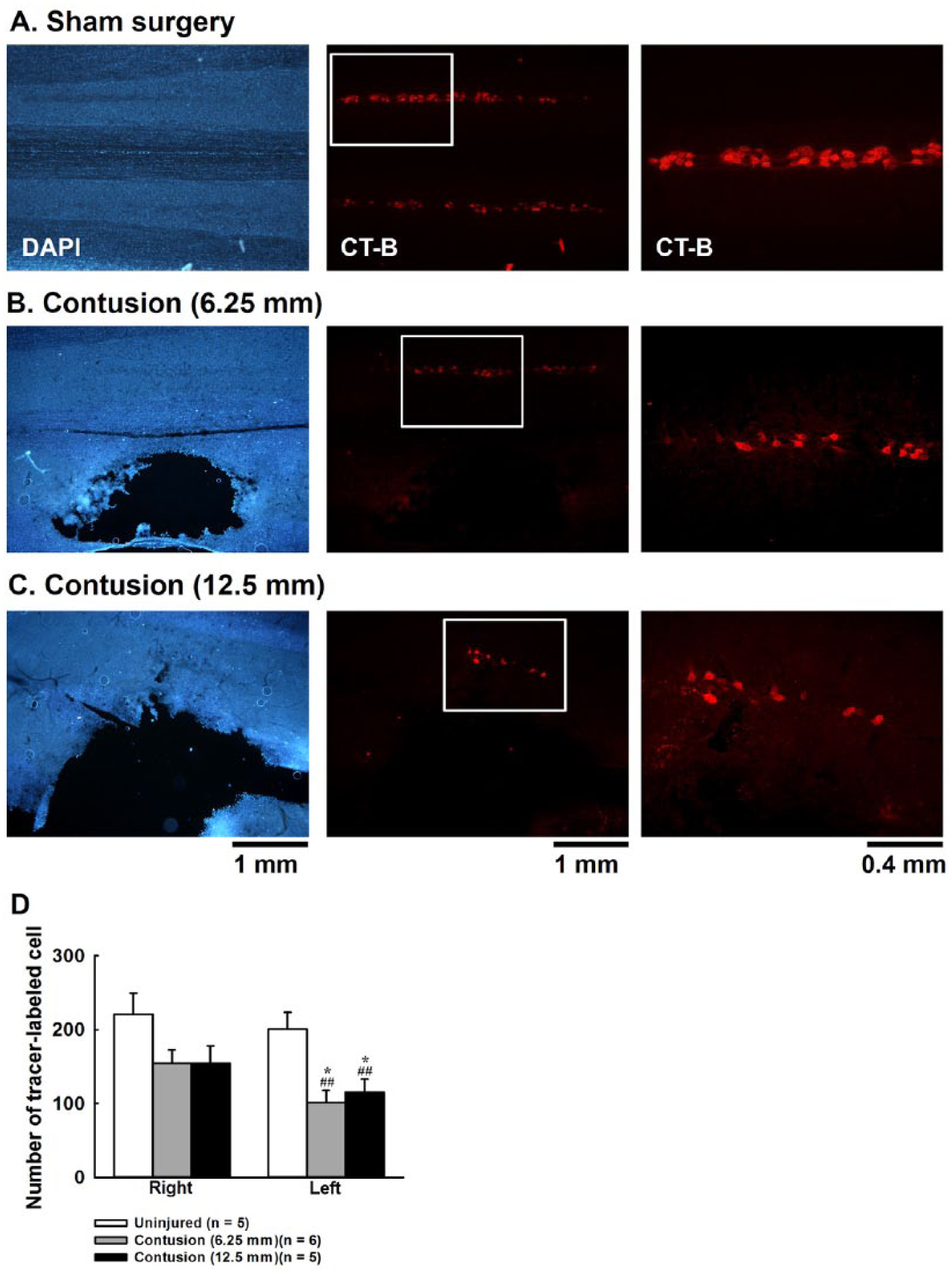
Representative examples of horizontal cervical spinal cord sections in sham surgery (A), moderately contused (B), and severely contused (C) animals at 4 weeks postinjury. The left column in panels A-C outlines the sections stained with DAPI (4′,6-diamidino-2-phenylindole). The middle column in panels A-C is the image showing retrograde tracer (ie, cholera toxin subunit B) labeled phrenic motoneurons. The right column in panels A-C is the high magnification view from the region labeled by the white box in the middle column. Panel D portrays the average number of tracer-labeled neurons in sham surgery and contused animals. *P < .05 significant different between right and left side. ##P < .01 significant difference from sham surgery animals.
Baseline Breathing Pattern
Representative examples of respiratory airflow recorded from whole-body plethysmography are depicted in Figure 3. During baseline, respiratory frequency was similar between groups (Figure 4A). However, tidal volume of severely contused animals (1.51 ± 0.07 mL) was significantly lower than moderately contused (1.84 ± 0.06 mL) and sham surgery animals (1.96 ± 0.05 mL) (P < .01, Figure 4B). Similarly, minute ventilation of severely contused animals was also lower than the other 2 groups (P < .01, Figure 5C).
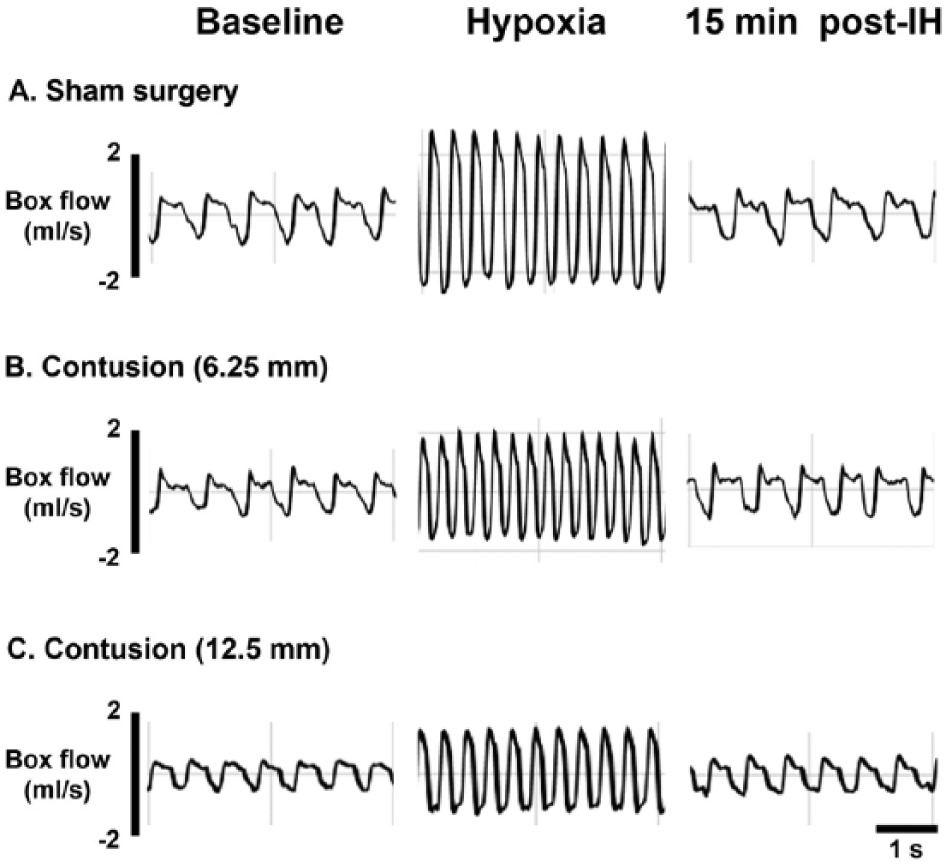
Representative examples of respiratory airflow recorded by whole-body plethysmography during baseline (left panel), first hypoxia (middle panel) and 15 minutes after intermittent hypoxia (IH).
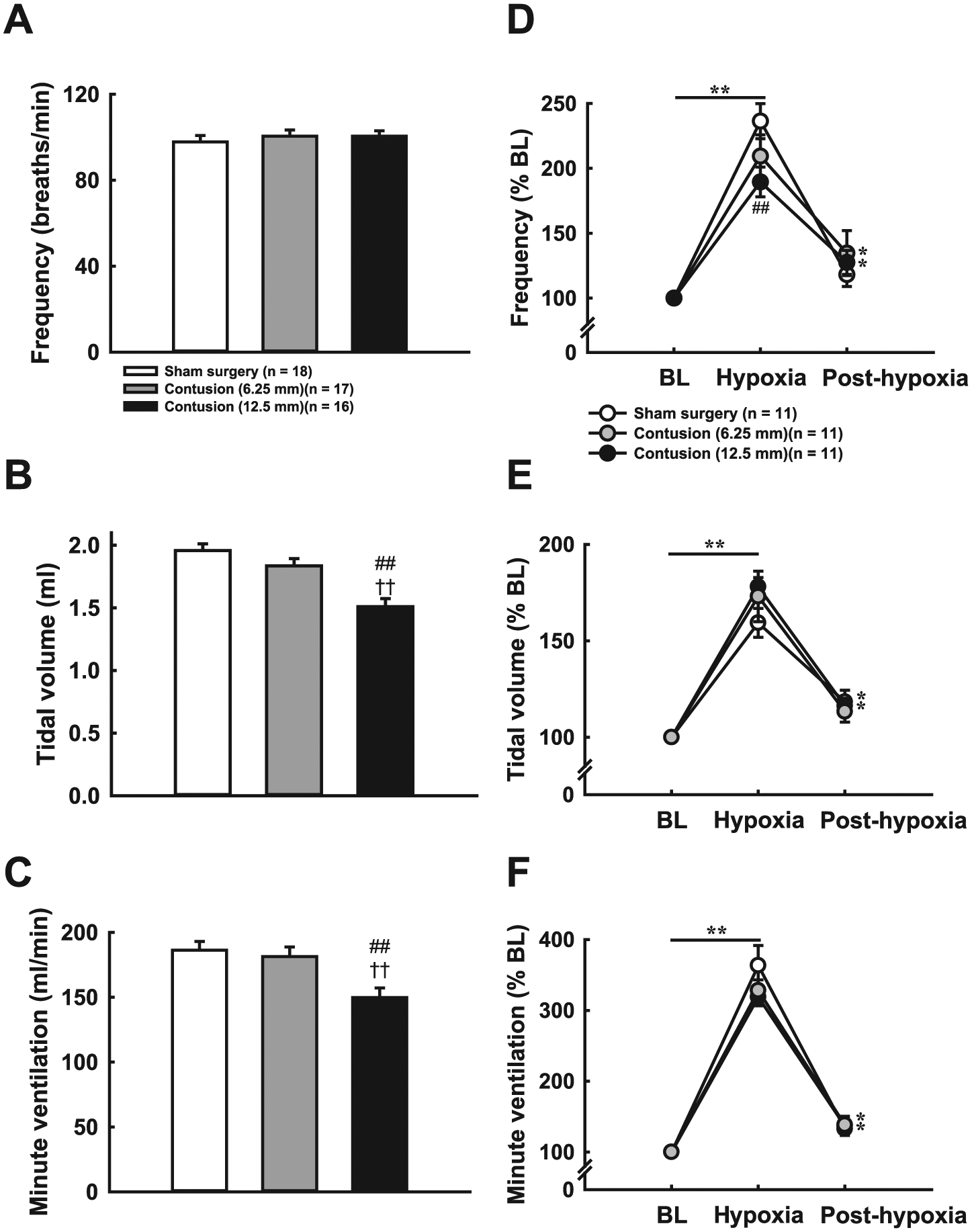
Baseline breathing patterns (A-C) and hypoxic responses (D-F) of sham surgery and contused animals at 4 weeks postinjury. Respiratory frequency (A) was similar between groups; however, both tidal volume (B) and minute ventilation (C) in severely contused animals were significantly lower than in sham surgery and moderately contused animals. Hypoxia elicited a significant increase in respiratory frequency (D), tidal volume (E) and minute ventilation (F) in all three groups. At 5 minutes posthypoxia, the respiratory frequency and minute ventilation of contused animals remained higher than baseline. The tidal volume was greater than baseline after hypoxia in sham surgery and severely contused animals. ##P < .01 significant difference from sham surgery animals. ††P < .01 significant difference from moderately contused animals. **P < .01 significant difference from baseline (BL) value.
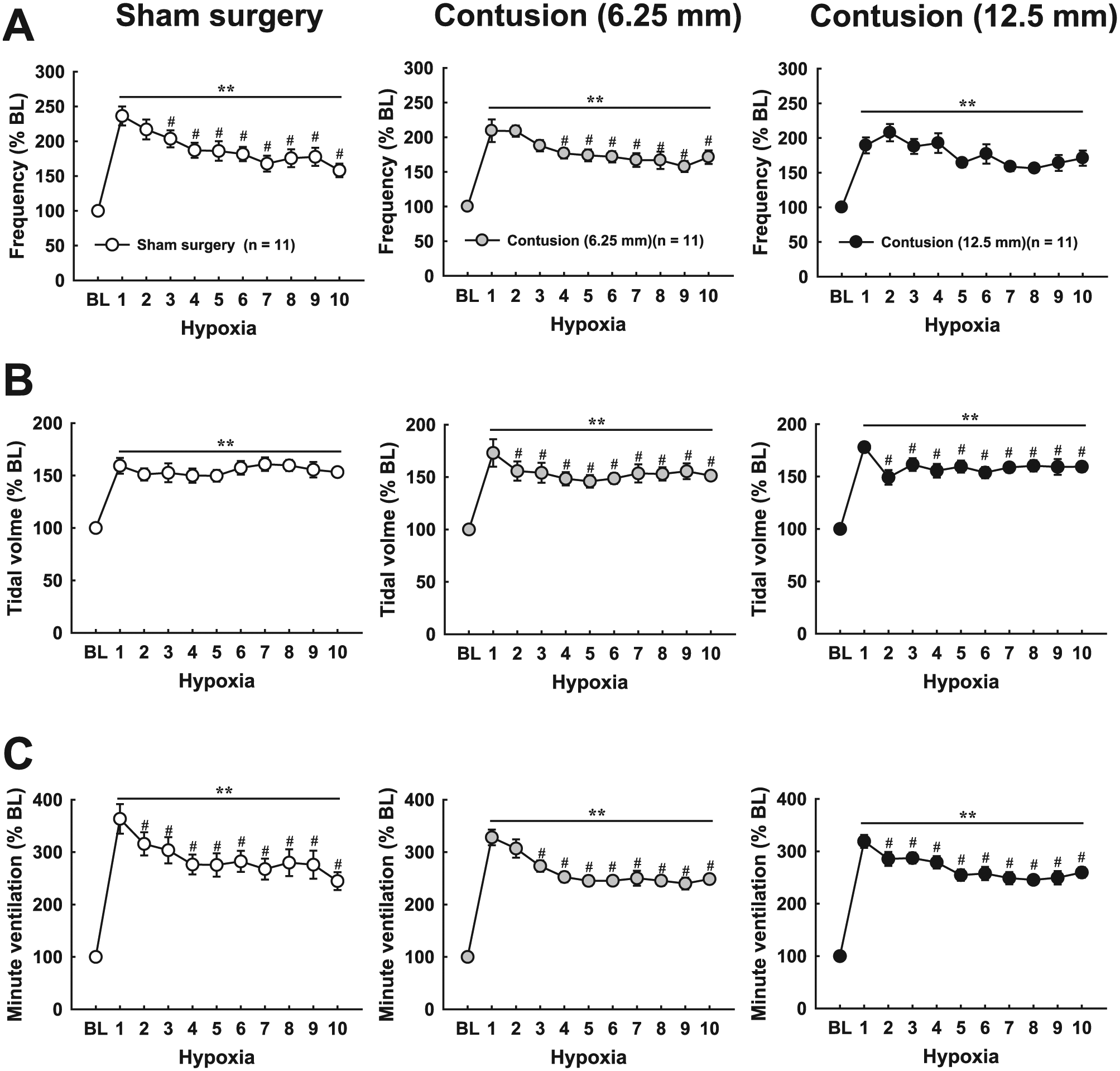
Progressive hypoxic responses of respiratory frequency (A), tidal volumes (B), and minute ventilations (C) during intermittent hypoxia. Ventilatory parameters were enhanced during all trials of hypoxia in all groups; however, there was a progressive decline in minute ventilation. This attenuation of hypoxic ventilatory response was because of a gradual reduction in frequency response in sham surgery animals and tidal volume in severely contused animals. In moderately contused animals, both frequency and tidal volume were progressively decreased with successive hypoxia. **P < .01 significant difference from baseline (BL) value. #P < .05 significant difference from the first hypoxic response.
Acute Hypoxic Response
Hypoxia induced a significant increase in respiratory frequency, tidal volume, and minute ventilation in all groups, just as expected (Figure 4D). However, severely contused animals had a lower capability of increasing respiratory frequency than sham surgery animals (P < .01, Figure 4D). Specifically, the frequency response reached 236% ± 14% BL in sham surgery animals, but the response in severely contused animals was only 189% ± 11% BL. The respiratory frequency of both contused groups remained higher than baseline at 4 to 5 minutes after hypoxic treatment (P < .05, Figure 4D). The increase of tidal volume was insignificantly higher in contused animals (moderate contusion: 173% ± 13% BL; severe contusion: 178% ± 5% BL) than sham surgery animals (159% ± 7% BL) (Figure 4E). The minute ventilation was significantly enhanced by more than 300% BL during hypoxia in all groups. After termination of hypoxia, the minute ventilation of contused animals was still greater than baseline (moderate contusion: 138% ± 12% BL; severe contusion: 137% ± 7% BL).
Progressive Hypoxic Response
Successive hypoxia induced a significant increase in ventilator parameters in all groups (P < .01, Figure 5); however, it was noticed that there was a progressive reduction in respiratory frequency and tidal volume. In particular, the hypoxic frequency response during the 3rd to 10th hypoxia session was weaker than the response induced by the first hypoxia episode in sham surgery animals (P < .05, Figure 5A). This phenomenon was observed in moderately contused but not severely contused animals. Conversely, the hypoxic tidal response in all trials of hypoxia was similar in sham surgery animals, but contused animals exhibited a weaker tidal volume response during the 2nd to 10th hypoxia sessions compared with the first hypoxic response (P < .05, Figure 5B). This reduction in hypoxic frequency and/or tidal volume response induced a progressive decline in minute ventilation during successive hypoxia in all groups (P < .05, Figure 5C).
Ventilatory Response After Intermittent Hypoxia
The ventilatory parameters were not significantly different at 15 to 60 minutes postintermittent normoxia in all groups, suggesting the alteration of ventilation after mAIH was resultant of the effect of intermittent respiratory challenges, but not the fluctuation of airflow in the chamber or time-dependent changes in ventilation. Respiratory frequency was not significantly changed in sham surgery animals at 15 to 60 minutes after mAIH (Figure 6A). However, the facilitation of tidal volume was revealed at 15 minutes postintermittent hypoxia in moderately (110% ± 3% BL) and severely (116% ± 4% BL) contused animals (P < .05, Figure 6B). At 30 minutes postintermittent hypoxia, both contused groups still exhibited a larger tidal volume than baseline (moderate contusion: 112% ± 5% BL; severe contusion: 113% ± 4% BL) (P < .01, Figure 6B). Although the minute ventilation of contused animals was generally higher than baseline at individual time points post intermittent hypoxia, this difference did not reach a significant level (Figure 6C).
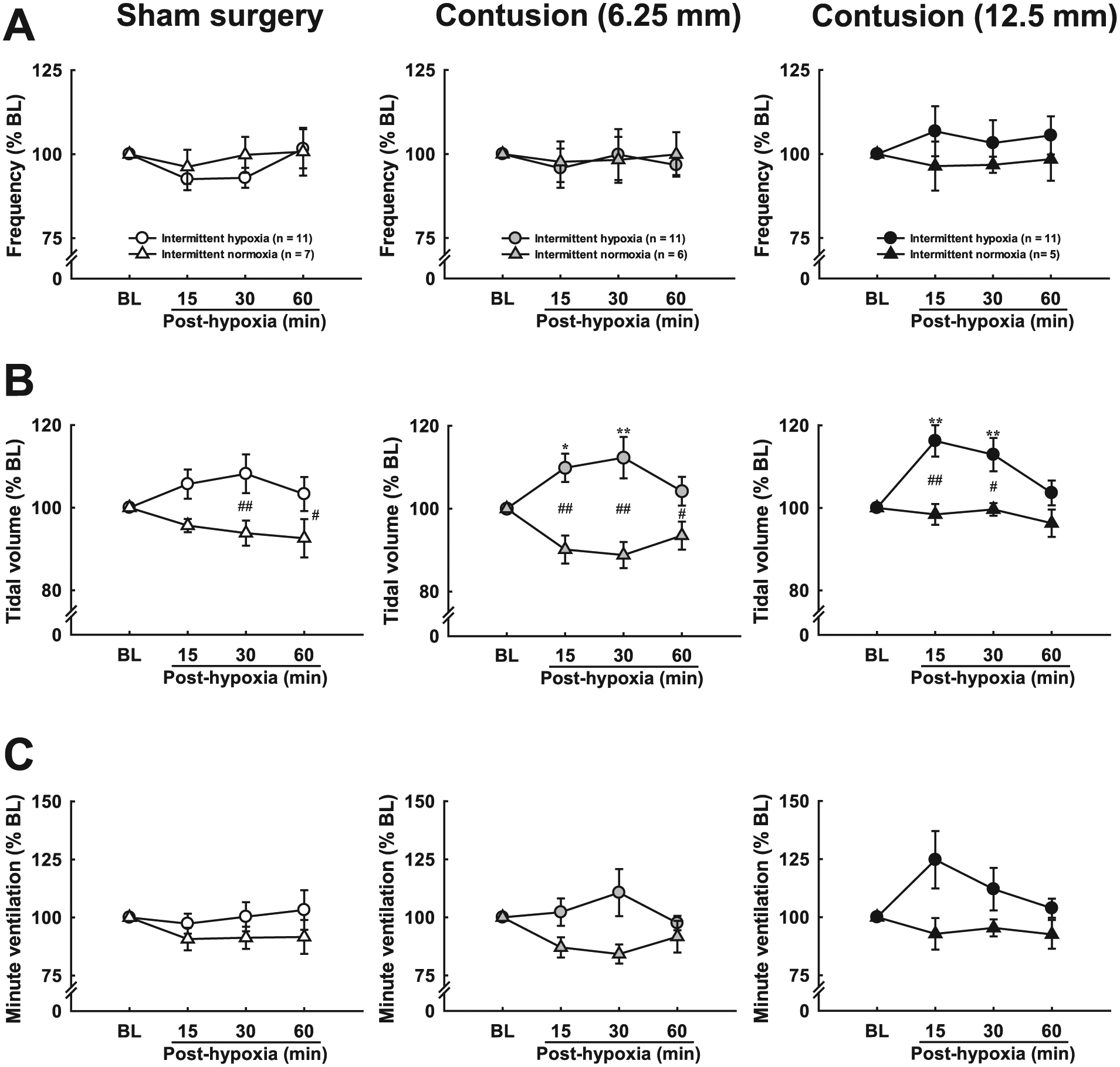
Ventilatory responses following intermittent hypoxia. Respiratory frequency and minute ventilation were not significantly modified after intermittent hypoxia in all groups. However, tidal volume in contused animals was significantly augmented at 15 and 30 minutes following hypoxia, suggesting intermittent hypoxia can induce long-term facilitation (LTF) of tidal volume following midcervical contusion. *P < .05; **P < .01 significant difference from baseline (BL) value. #P < .05; ##P < .01 significant difference from intermittent normoxia.
Discussion
The present study has demonstrated that acute hypoxia caused significant increases in respiratory frequency and tidal volume in sham surgery and contused animals; however, there was a gradual reduction in frequency response in sham surgery animals and decreased tidal volume response in contused animals with successive hypoxic episodes. Furthermore, it was shown that contused animals, but not sham surgery animals, exhibited an augmentation in tidal volume at 15 to 30 minutes following mAIH. These results indicate that hypoxia-induced respiratory neuroplasticity is differentially expressed in sham surgery versus contused animals. Induction of LTF in respiratory motor outputs in contused animals suggested that mAIH could be an effective neurorehabilitation approach to improve respiratory function after cervical SCI.
Cervical Spinal Cord Contusion
The present study used a MASCIS Impactor system combined with the stereotaxic Cunningham spinal adaptor and transverse clamps to induce unilateral midcervical contusion in adult rats. This configuration can firmly stabilize cervical vertebrate during drop of an impact rod. The histological data demonstrated that contusion injury caused a significant reduction in spinal cord section area and created an obvious lesion cavity on the left side. The lesion cavity was larger and extended to right side in severely contused animals. Moreover, the number of phrenic motoneurons ipsilateral to the lesion was significantly diminished in contused animals, suggesting contusion injury from the current study can effectively cause damage of the phrenic nuclei.
One technical consideration needs to be mentioned. Because both sham surgery and contused animals received C3-C4 laminectomy, and the spinal cord of these animals was clamped on the stereotaxic using transverse clamps. In addition, the spinal cord histology was evaluated at 4 weeks after spinal surgery. Our histological data showed that this spinal surgery procedure may cause a slightly compression on the spinal cord. However, the cell morphology and respiratory behavior of the sham surgery animals were normal, and the data of contused animals were compared with sham surgery animals. Therefore, the differential effect of intermittent hypoxia on the ventilatory response between uninjured and contused animals was primarily due to the influence of the contusion injury.
Breathing Patterns Following Cervical Contusion
Respiratory frequency was similar between groups, and tidal volume was significantly reduced in severely contused animals at 4 weeks postinjury. The reduction of tidal volume was possibly a consequence of loss of phrenic motoneurons and interruption of the bulbospinal respiratory pathway. However, tidal volume was not significantly lessened in moderately contused animals despite there being a significant decrease in the number of phrenic motoneurons. As the moderately and severely contused groups displayed a similar extent of phrenic motoneuron loss, it was suspected that maintenance of tidal volume in moderately contused animals could be attributed to residual neural tissue. The histological data indicated there was sparing of tissues preserved in the dorsal and ventral spinal column in moderately contused rats (Figure 1B). Lipski et al 34 demonstrated that phrenic motoneurons are primarily innervated by bilateral premotor neurons within the brainstem. The ipsilateral premotor neurons send projections to the phrenic nucleus through the lateral and dorsal-lateral funiculus, while the contralateral bulbospinal pathway was located within the ventral and ventral-medial funiculus. With this, unilateral contusion injury in moderately contused animals may interrupt the ipsilateral bulbospinal pathway but preserve the contralateral descending pathway to activate residual phrenic motoneurons. Awad et al 35 provided evidence for diaphragm electromyographic activity ipsilateral to unilateral midcervical contusion being completely eliminated after ipsilateral C2 hemisection at 7 days postinjury, suggesting the crossed spinal pathway remained inactive in contused animals. However, the breathing pattern was measured at 4 weeks postinjury in the present study, which may enable gradual activation of the crossed spinal pathway. In addition, Ghali and Marchenko 36 described the crossed spinal pathway becoming spontaneously active within hours following C1 hemisection in decerebrate rats. Therefore, here, recovery of tidal volume in moderately contused animals under unanesthetized conditions could be partially explained by activation of the crossed spinal pathway.
Acute Hypoxic Response
Powell et al 37 demonstrated there is a short-term potentiation (STP) of respiratory outputs induced by a single bout of hypoxic treatment. Hypoxia-induced respiratory STP represents a progressive augmentation during hypoxia (ie, onset STP) followed by a slow recovery of ventilation after hypoxia (ie, offset STP). 37 It was observed in this work that severely contused animals had a lower capacity for increasing respiratory frequency during hypoxia despite the baseline frequency being similar between sham surgery and contused animals, alluding to onset STP being attenuated in contused animals. Previous studies in anesthetized animals have shown that phrenic burst frequency of C2 hemisected animals had a blunted response during changes of end-tidal CO2 at 2 and 4 weeks postinjury. 38 Moreover, prolongation of the expiratory duration during activation of pulmonary stretch receptors and bronchopulmonary C-fibers was also attenuated in cervical spinal injured rats.9,39-41 These data imply that cervical SCI may restrict the alteration of respiratory frequency in response to respiratory stimuli. Interestingly, the respiratory frequency of contused animals, but not sham surgery animals, remained higher than baseline at 5 minutes after first hypoxic treatment, suggesting offset STP of respiratory frequency was evident in contused animals. Collectively, hypoxia-induced onset STP of respiratory frequency was decreased but offset STP was elicited following cervical contusion. However, such differential expression of hypoxia-induced onset and offset STP was not reflected in the tidal volume response. This may be because cervical contusion has a differential impact on onset versus offset STP and that distinct mechanisms give rise to hypoxia-induced neuroplasiticity with respect to respiratory frequency and tidal volume in unanesthetized rats.
Progressive Hypoxic Response
Successive hypoxia has been demonstrated to induce progressive augmentation of respiratory outputs. For instance, Harris et al 30 demonstrated that episodic hypoxia induced progressive augmentation in breathing frequency under elevated end-tidal partial pressure of CO2 in awake humans. Additionally, progressive augmentation of phrenic burst amplitude could be produced in anesthetized rats during severe intermittent hypoxia. 42 However, progressive augmentation of hypoxic ventilatory response was not seen in unanesthetized rats in response to intermittent isocapnic hypoxia or poikilocapnic hypoxia. 43 Unexpectedly in the current study, there was a progressive decline in ventilatory response during mAIH. Several potential explanations for discrepancies between the present and other studies exist. First, the present study aimed to examine the impact of mAIH on respiratory function during the chronic injury state, thus, the age of animals used to measure ventilation were relatively older than those from other studies. McGuire and Ling 44 showed that mAIH-induced ventilatory LTF was weaker in 2- versus 1-month-old awake rats. Consequently, progressive decline in the hypoxic ventilator response may be, in part, the influence of aging effects on respiratory neuroplasticity. Second, Cummings and Wilson 45 suggested that modulation of carotid body afferent inputs was also involved in modulation of ventilatory response during and following hypoxia. They observed that the peak response of carotid sinus nerve activity was diminished during successive hypoxic bouts. 45 This occurrence indicated that the attenuation of hypoxic ventilatory response during mAIH in this study may be the result of lower peak response of peripheral chemoreceptors during successive hypoxia.
All groups showed a progressive drop in the magnitude of hypoxic ventilator response during mAIH based on alteration of respiratory frequency in sham surgery animals and tidal volume in severely contused animals. Previous studies have shown that cervical SCI caused diaphragm atrophy and a reduction in specific force of diaphragm.46,47 Thus, tidal volume generated from contused animals may not be able to be maintained at a consistent magnitude during successive respiratory challenges, leading to progressive deterioration of the tidal volume response.
Long-Term Facilitation
The present study demonstrated that mAIH can induce LTF of tidal volume in contused but not sham surgery animals. McGuire and Ling 44 demonstrated that ventilatory LTF could be elicited in 1-month-old, but not 2-month-old, rats following mAIH. The age of animals used to measure respiratory behaviors was ~3 months, therefore, the reason why mAIH could not evoke LTF in sham surgery animals may be because of aging effect.
The impact of mAIH on respiratory recovery has been studied in rats with C2 hemisections. Golder and Mitchell 20 provided evidence to show that mAIH can evoke LTF in phrenic outputs ipsilateral to injury in anesthetized rats at 8 weeks postinjury. Navarrete-Opazo et al 29 uncovered that contralateral, but not ipsilateral, diaphragm and intercostal activity exhibit LTF following mAIH in unanesthetized rats at 8 weeks postinjury. In addition, daily mAIH for 7 days can improve breathing capacity in C2 hemisected rats. 11 In the current study, a more clinically relevant SCI model (ie, cervical contusion) provided a case for mAIH inducing respiratory LTF at 4 weeks postcontusion. Collectively, these data indicate that mAIH was able to induce respiratory neuroplasticity in order to improve respiratory motor outputs following interruption of the bulbospinal pathway (ie, high-cervical hemisection) and/or damage of phrenic motoneurons (ie, midcervical contusion).
The mechanism underlying improvement of tidal volume after mAIH in contused animals remains unclear. Previous work has shown that improvements in ventilation after daily mAIH was mainly contributed by recovery tidal volume, not respiratory frequency, in unanesthetized C2 hemisected rats. 11 The magnitude of LTF in phrenic burst amplitude was usually greater than that of phrenic burst frequency in anesthetized injured rats.20,48 The results here also demonstrate that tidal volume, but not respiratory frequency, was significantly elevated at 15 and 30 minutes following mAIH. This supports the notion that functional benefits of mAIH on respiratory recovery are mainly from improvements in magnitude of respiratory outputs, not respiratory frequency. Based on the fact that only contused, and not sham surgery animals, exhibited LTF, it was suspected that cervical contusion enhanced excitability of spared phrenic motoneurons, which can amplify modulatory effect of mAIH. This concept was fostered by reviewing previous literature on the matter, indicating distribution of phrenic motoneurons shifted to late-inspiratory and silent motoneuron-type after C2 hemisection, and these phrenic motoneurons were capable of expressing greater hypoxia-induced neuroplasticity.38,49,50 It is worth noting that activation of ipsilateral diaphragm activity by supplementation with 5-HT precursors or activation of 5-HT2 receptors was not associated with improvement of tidal volume. 12 Indeed, Navarrete-Opazo et al 29 showed that mAIH can only induce LTF in an uninjured diaphragm and intercostals following C2 hemisection. Hence, compensatory plasticity 51 in uninjured spinal respiratory motoneurons may also play a role in enhancement of tidal volume following mAIH.
Physiological Significance
The present study demonstrated that mAIH can induce LTF in tidal volume in unanesthetized rats with midcervical contusion during a chronic injury state. This finding indicates that mAIH can not only improve respiratory function in an experimental hemisection model, but that it also has a beneficial impact on respiratory recovery in a clinically-relevant spinal contusion model. Several studies have investigated the potential therapeutic effectiveness of mAIH on motor function in spinal cord injury populations.1,16,28,52,53 Here, the current study provided a proof-of-concept animal model for the future repetitive mAIH treatment for improving breathing in spinal cord injury subjects.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Health Research Institutes (NHRI-EX105-10223NC), Ministry of Science and Technology (Most 102-2320-B-110-004-MY3 & Most 105-2628-B-110-002-MY3), and NSYSU-KMU Joint Research Project (105-I006).
