Abstract
Background. Cervical dystonia (CD) is a neurological movement disorder characterized by involuntary contractions of the cervical musculature and is known to be associated with proprioceptive dysfunction in dystonic/nondystonic limbs. Objectives. We examined how neck botulinum neurotoxin (BoNT) injection affects wrist proprioception and the corresponding sensorimotor cortical activity in CD. Method. Wrist position sense acuity of the dominant (right) hand was evaluated in 15 CD and 15 control participants. Acuity measures were a psychophysical position sense discrimination threshold (DT; based on passive joint displacement) and joint position matching error (based on active movement). Cortical activity during the motor preparation period of the active joint position matching was examined using electroencephalography. Results. In their symptomatic state, patients demonstrated a significantly higher wrist proprioceptive DT, indicating an abnormal passive wrist position sense. Yet BoNT injections had no significant effect on this threshold. During active joint position matching, errors were significantly larger in patients, but this difference vanished after the administration of BoNT. Motor preparation of active wrist position matching was associated with a significantly higher rise of β-band (13-30 Hz) power over contralateral somatosensory-motor cortical areas in patients. This excessive cortical activity significantly declined post-BoNT. Conclusion. Wrist proprioceptive perception during passive/active movements is abnormal in CD. An excessive rise of premotor/motor cortical β-oscillations during motor planning is associated with this proprioceptive dysfunction. Neck BoNT injections normalized the cortical processing of proprioceptive information from nonsymptomatic limbs, indicating that local injections may affect the central mechanisms of proprioceptive function in CD.
Introduction
Cervical dystonia (CD) is a non-task-specific focal dystonia characterized by involuntary cervical muscle contractions, which may be sustained, intermittent, or accompanied by dystonic tremor. 1 CD is the most common form of focal dystonia with a prevalence of 28 to 183 cases per million. 2 Pharmacotherapy, neck botulinum neurotoxin (BoNT) injection, and deep brain stimulation are major treatment options for focal dystonia, including CD. 3 CD is associated with somatosensory dysfunction beyond the dystonic site as evidenced by elevated hand tactile spatial discrimination4,5 or finger position sense thresholds.6,7 Approximately three-quarters of all CD patients temporarily correct abnormal postures by touching areas of the head/neck. 8 Such successful geste antagoniste maneuvers indicate a close link between the underlying somatosensory and overt motor deficits in CD.
Abnormal somatosensory processing that affects proprioceptive as well as tactile function likely underlies the faulty motor control observed in movement disorders, such as CD. Two scenarios can be envisioned: first, mechanisms of afferent somatosensory feedback (external) or predicted sensory feedback (internal) could be defective. 9 Both mechanisms would affect the feedback-driven control of head posture. Second, processing of external and internal feedback is intact, but their integration is faulty.10,11 Such failure of sensory integration may affect the weighting of multimodal inputs, which, in turn, may lead to abnormal head motion or posture.
Neck BoNT injection, a symptomatic treatment for CD motor symptoms, alters the afferent signals from proprioceptive mechanoreceptors of the injected muscles because BoNT not only blocks spinal α but also γ motor neuron endings, consequently reducing the spindle inflow to the α motor neurons.12,13 These altered proprioceptive signals from the periphery are processed by higher cortical centers of the somatosensory system that consequently affect proprioceptive perception as well as somatosensory-motor cortical processing.
In conclusion, despite the existing evidence of abnormal proprioceptive function in CD, our knowledge of the degree of proprioceptive impairment in this disorder is still limited. In addition, the effect of neck BoNT injection as the most common symptomatic treatment for CD on the central mechanisms of somatosensory information processing has remained unclear. Thus, this study examined the effect of neck BoNT on proprioception and the associated somatosensory-motor cortical processing in CD. We applied 2 complementary methods to measure proprioceptive function in a nondystonic limb in patients with CD before and after neck BoNT injections. We evaluated wrist position sense acuity using (1) a psychophysical position discrimination threshold (DT) paradigm and (2) a joint position matching paradigm. Because our psychophysical paradigm involved passive movement, this method can determine if a primary somatosensory deficit was present. The joint position matching paradigm, however, required voluntary movements, which enabled us to examine abnormal sensorimotor integration processes in CD. 14 Electroencephalographic (EEG) evaluation of the motor preparation period during active joint position matching movement allowed us to explore the underlying electrocortical activity. Findings of this study provide a deeper understanding of the sensory aspects of dystonia and the effect of neck BoNT injection on proprioceptive function and somatosensory-motor cortical processing in CD. From a clinical perspective, they will also lay the foundation for a broader and more directed use of noninvasive neurorehabilitation therapies, such as transcranial alternating current stimulation for CD, by targeting and modulating those brain oscillations that are particularly altered in this disorder.
Methods
Participants
A total of 15 CD participants, all presenting with isolated dystonia (mean age ± SD: 54.3 ± 13.4 years) and 15 age- and gender-matched healthy controls participated in the study (Table 1). CD participants were recruited from the Clinics and Surgery Center, University of Minnesota. The experimental protocol was approved by the University of Minnesota Institutional Review Board. Participants gave their written informed consent prior to testing. Control participants attended the study only once, whereas CD participants repeated the study twice using the exact same protocol in both visits: (1) within 1 week prior to BoNT injection (symptomatic state), and (2) 3 to 4 weeks after BoNT injection (optimal state). For each CD participant, the order of attendance for the 2 data collection sessions was randomized. Specifically, 9 out of 15 CD participants attended the first session of data collection during their optimal state and completed the second session in their symptomatic state. The remaining 6 CD participants attended the 2 sessions in a reversed order.
Characteristics of the Cervical Dystonia Participants.
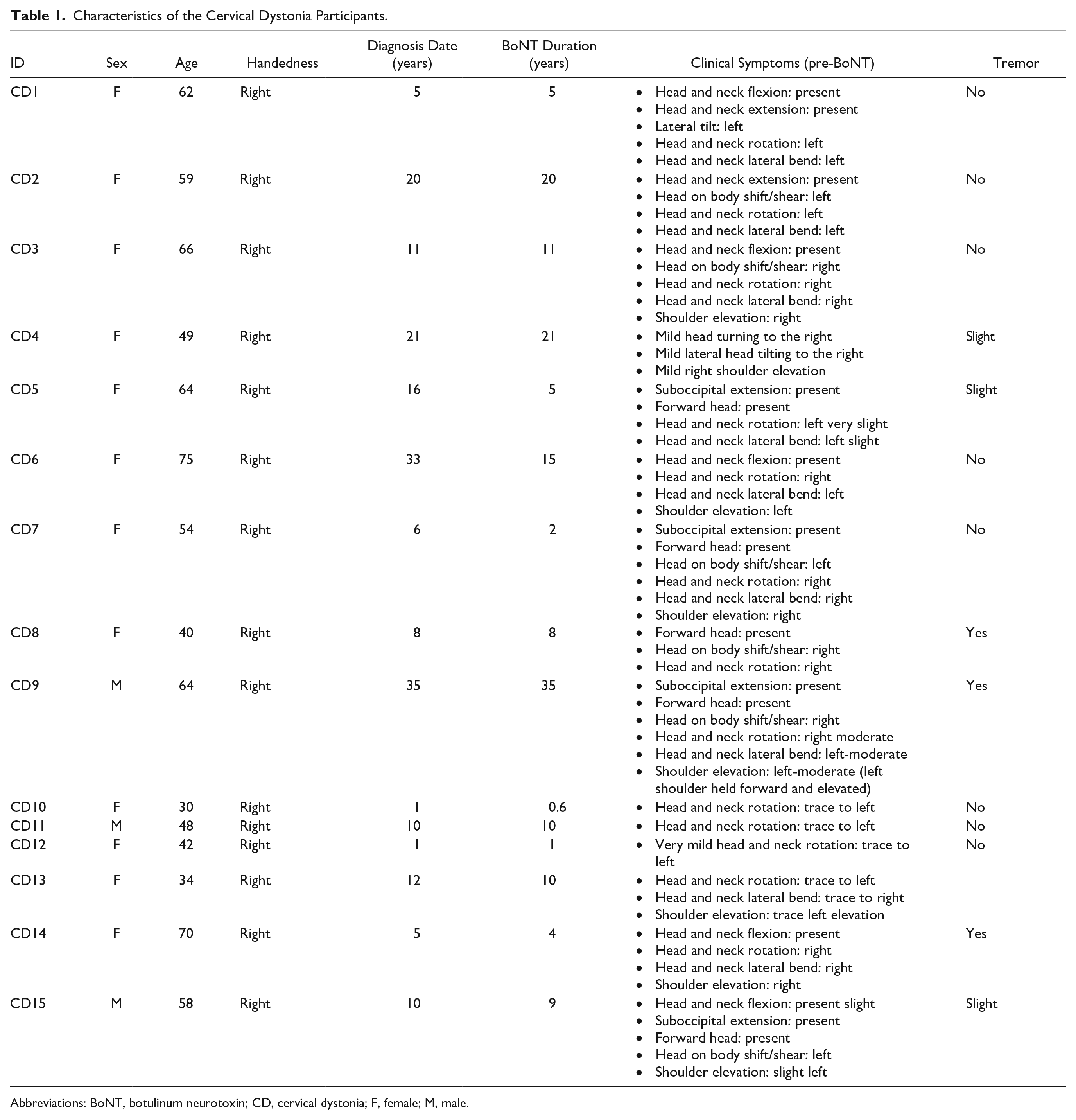
Abbreviations: BoNT, botulinum neurotoxin; CD, cervical dystonia; F, female; M, male.
Apparatus
A custom-built, wrist bimanual manipulandum with 1 degree of freedom in the horizontal plane was used to assess wrist position sense acuity (Figure 1A). Optical encoders (spatial resolution: 0.036°; US Digital H6), housed at the rotating point of the lever arms, recorded the angular position of each hand at a sampling rate of 200 Hz. To allow for the testing of distinct joint positions across the joint range of motion, the device had a pegboard with holes in a semicircular arrangement. By inserting a metal pin, one could select from 140 different positions across 0° to 70° range of motion of the wrist in 0.5° increments (Figure 1A).

Experimental device: the wrist manipulandum used for the assessment of wrist joint proprioception. A pegboard base panel housed under the lever arms allowed the insertion of a metal pin into a specific hole to mark a distinct reference position. Electroencephalographic data were recorded simultaneously with the joint position matching task via a 64-channel EEG cap.
EEG data were recorded with the ActiveTwo system (Biosemi B V, Amsterdam, Netherlands) at a sampling rate of 512 Hz. Brain potentials were captured with active gel-based electrodes using a Biosemi 64-channel EEG cap with an equiradial system of electrode placement. Participants were guided throughout the experiment by a series of 200-ms-long auditory cues (1000 Hz, 98 dB) generated by the RPvdsEx software (Tucker-Davis Technologies Ltd, Alachua, FL) presented via bilateral speakers. The time stamps of auditory cues were recorded simultaneously by the ActiveTwo system.
Experimental Procedure
Participants sat in an electrically and acoustically shielded chamber and placed their right, dominant arm on the lever of the wrist manipulandum. Chair height, lever arm length, and handle placement were adjusted to the anthropometrics of each participant, so that the approximate wrist joint axis was aligned with the rotating axis of the optical encoder. Visual input was masked (Figure 1B).
For psychophysical threshold testing, the participant’s dominant hand was passively flexed to 2 different positions from the neutral position: a reference position at 30° flexion and a comparison position at a particular angular position ≤29.5°. Using a 2-alternative forced choice method, participants judged which position was farther from the neutral position. Based on the participant’s verbal response, an adaptive algorithm using the psi-marginal method 15 generated the comparison position of the following trial to guarantee a fast convergence toward the proprioceptive perception threshold within 30 trials. Between trials, the order of presentation of reference and comparison positions was randomized to account for possible order effects.
For joint position matching, the experimenter flexed a participant’s dominant hand to a 30° reference position, held it there for 2 s, and returned it to the neutral position. After a few seconds (<5 s) a 200-ms-long auditory “Warning” cue was given, and within a time interval (randomly changing between 2, 3, or 4 s to prevent any learning effects) a “Go” cue was presented that informed the participant to actively move the dominant hand to the previously experienced joint position. During active joint position matching, EEG data were recorded simultaneously.
The idea behind performing a delayed position matching task guided by a pair of auditory cues was to obtain access to the movement preparation stage in the recorded EEG data and to evaluate the spectral characteristics of this period. Because between-subject variabilities in reaction time and movement speed influenced the execution of the voluntary matching movement, any EEG analysis occurring around the Go cue was necessarily confounded by these factors. To guard against this, we focused on the initial 2 s of the time interval between the Warning and Go cues. This 2-s long period was in common among all trials. It is known to contain the early component of the contingent negative variation, called the orienting wave, known to peak 0.5 to 1.5 s after the Warning cue.
Measures
Proprioceptive Measures
The psychophysical measures included the following: (1) wrist position DT for assessing proprioception under passive displacements and (2) wrist position matching error (ME) as a marker of proprioceptive perception during active movements. To obtain each participant’s DT, the correctness of his/her judgment in the 2-alternative forced choice task (correct: 1; incorrect: 0) for all trials were fitted to a logistic Weibull function. DT corresponded to the difference between a pair of reference and comparison positions located at the 75% correct response level. ME was computed as the mean absolute difference between the matched and target positions across all trials. This was interpreted as a bias or systematic error in position matching. Proprioceptive function assessments were conducted on the dominant hand, which for all study participants was the right hand.
Electrophysiological Measures
The EEGLab toolbox (Delorme and Makeig, 2004) of MATLAB (The MathWorks, Natick, MA) was used for the offline analysis of EEG data. Electrodes were referenced to the average of the 2 external electrodes over the left and right mastoid bones. Baseline drifts were addressed by 1-Hz high-pass filtering, and the power line noise was removed via zero-phase notch filtering. Channels were also re-referenced to the common average of all electrodes to reduce the effect of noncortical sources that may have been captured by all electrodes. Segments of recording from 1000 ms before the Warning cue to 2000 ms afterward were derived as data epochs. 16 Subsequently, we performed independent component analysis on all 64 channels using the runica algorithm and applied an automated multiple artifact rejection algorithm SASICA 17 on the generated components to remove contaminated ICs. The SASICA algorithm uses spatiotemporal criteria to distinguish cortical components from artifactual ICs. The remaining components were linearly added, and the resultant data set was used for EEG feature extraction.
The derived measures included the following: (1) event-related spectral perturbation (ERSP); and (2) event-related coherence (ERCOH). ERSP reflects the logarithm of the mean event-related deviation in spectral power relative to the resting state (spontaneous EEG) at each frequency bin. 18 ERCOH calculates the event-related coherence between 2 channels as an indicator of the level of synchronous activity between 2 cortical locations. 19 EEG epochs were prewhitened prior to coherence computations to remove potential autocorrelations and trends that might have interfered with the results.
ERSP was computed for EEG electrodes nearby the left and right neck and hand somatosensory-motor areas: CP1, C1, FC1, CP3, C3, FC3, CP2, C2, FC2, CP4, C4, and FC4. CP or centroparietal electrodes are nearby somatosensory areas; C or central electrodes are nearby motor cortical areas; and the FC or frontocentral electrodes are nearby premotor cortical areas (Figure 1C). Odd indices represent the left hemisphere, and even indices represent the right side. ERCOH measures were derived for the following pairs of electrodes: (1) between somatosensory and motor cortical regions in 1 hemisphere: CP1-C1, CP1-FC1, CP3-C3, CP3-FC3, CP2-C2, CP2-FC2, CP4-C4, CP4-FC4; and (2) between bilateral somatosensory-motor cortical areas: CP1-CP2, CP3-CP4, C1-C2, C3-C4, FC1-FC2, and FC3-FC4. EEG measures were computed for the physiologically relevant frequency bands (ie, <50 Hz): θ (4-8 Hz), α (8-13 Hz), β (13-30 Hz), and the low γ (30-49 Hz). Feature extraction was focused on the preparatory stage of the position matching movement.
The position matching task and the corresponding EEG recording were repeated in 3 blocks of 20 trials, but the ME measure was computed based on the first 5 trials. Both the ERSP and ERCOH measures were computed for the 0- to 2000-ms interval after the presentation of the Warning cue. This delay or the anticipatory period before the execution of movement is known to correspond to brain processes associated with motor planning.20,21 Assessments were only performed on the dominant right hand.
Statistical Analysis
Because based on the Kolmogorov-Smirnov test, the behavioral and EEG data were not normally distributed, we report the median (MED) and median absolute deviation (MAD). MAD is a measure of statistical dispersion that is resilient to outliers in a dataset. We applied Mann-Whitney U tests to determine statistical group differences. Because the patient data pre-BoNT versus post-BoNT are considered a paired dataset, the Wilcoxon signed rank test was used. P values were adjusted for multiple comparisons using the Benjamini-Hochberg false discovery rate correction method. 22 The significance level was set at .05. Effect size was computed using Cohen’s d (d values of 0.2, 0.5, and 0.8 correspond to small, medium, and large effect sizes).
Results
Evidence for Abnormal Passive and Active Wrist Position Sense in CD
The MED and MAD of DT and ME represented the sensory outcome measures. We first assessed the wrist position sense acuity based on passive joint flexion. As a group, CD participants exhibited a 51% larger DT (MED: 6.61°; MAD: 1.94°) compared with healthy controls (MED: 4.38°; MAD: 0.63°; P = .006). The effect size was d =1.23, which is considered very large. 23 In addition, we investigated wrist position sense acuity based on active joint position matching. We found that the MED ME of the CD group (MED: 3.64°; MAD: 0.48°) was significantly larger compared with those of controls (MED: 2.37°, MAD: 0.93°; P = .009), indicating a systematic difference of bias in proprioceptive acuity between the two. The corresponding effect size was large (d = 1.00).
Evidence for Abnormal Sensory-Motor Cortical Activity During Active Joint Position Matching in CD
When CD participants prepared to actively match the remembered joint position, they exhibited a significantly larger β-band ERSP over premotor/motor cortical areas in the left hemisphere: C1 (P = .009; d = 1.24), FC1 (P = .009; d = 1.16), and FC3 (P = .008; d = 1.34). All effect sizes were large (d > 0.8) or very large (d > 1.2). This indicates that during the symptomatic state (pre-BoNT), the synchronized deviation from baseline neuronal activity in motor cortical areas controlling the wrist joint was excessively elevated in the β-band (Figure 2).
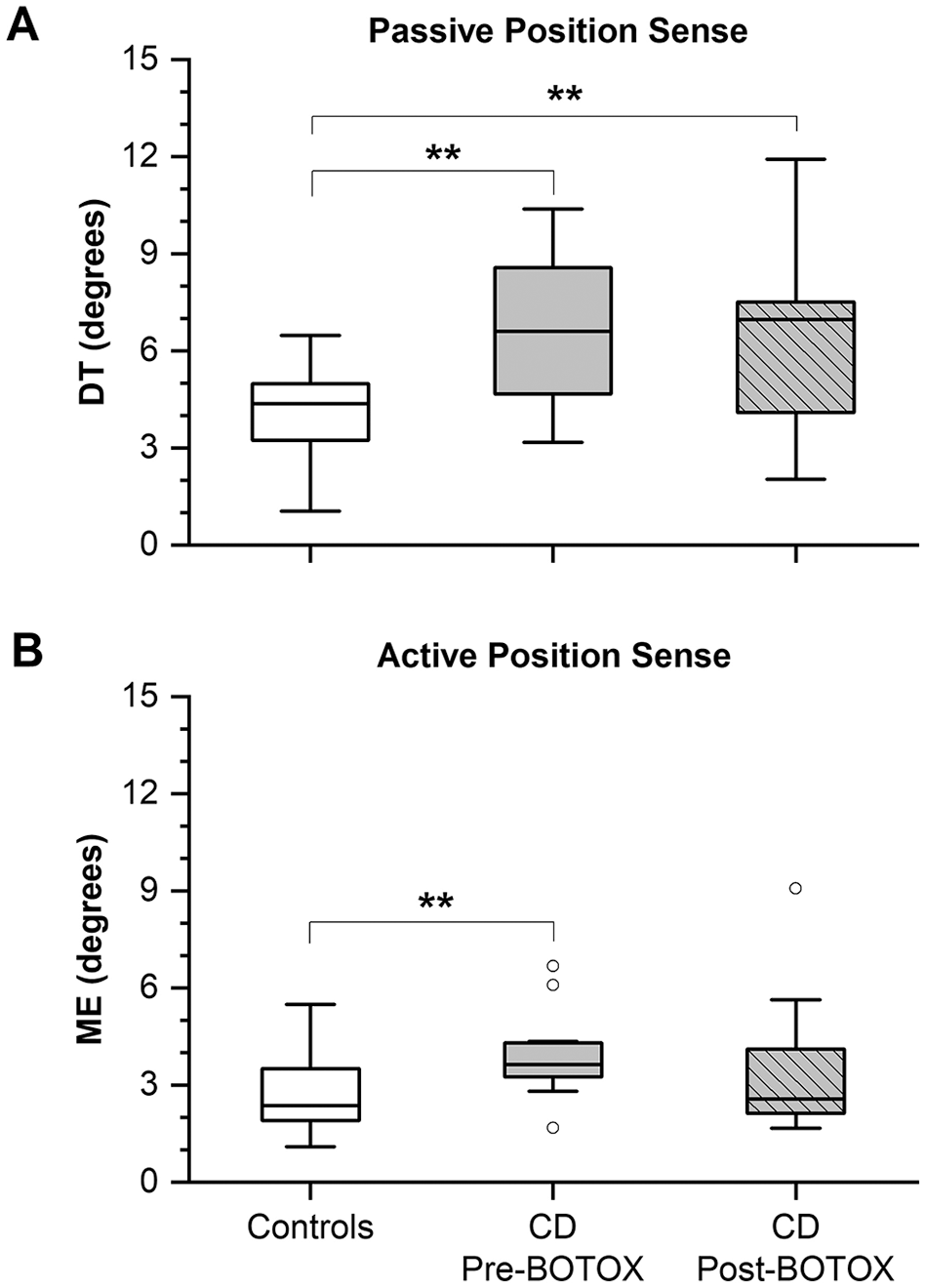
Measures of passive and active position sense for the control group and CD patients at the pre-BoNT and post-BoNT injection stage. (A) Wrist joint DT based on passive joint displacement (passive position sense). (B) Wrist joint position ME based on active joint displacement (active position sense). Lower and upper boundary of each box indicates the 25% and 75% quartiles, respectively. The horizontal line within the box depicts the median. The upper and lower whiskers extend to +1.5 and −1.5 interquartile range, respectively. Outliers are shown as white circles.
Effects of BoNT on Perceptual and Neural Measures of Proprioception
Neck BoNT injections had no systematic effect on wrist DT. Patients still showed a significantly larger DT (MED = 6.97°, MAD = 1.90°; P = .007; d = 1.10) in comparison to controls (MED = 4.38°, MAD = 0.63°; P = .006; see Figure 3A), indicating that BoNT did not alter abnormal passive position sense in this nondystonic motor system. For active joint position matching, the CD group showed no significant difference in ME between their symptomatic and optimal states (pre-BoNT: MED = 3.64°, MAD = 0.48°; post-BoNT: MED = 2.57°, MAD = 0.82°; Figure 3B). However, in their optimal state (post-BoNT), the MED ME of the CD group was no longer significantly different from that of corresponding healthy controls MED ME (CD: MED = 2.57°, MAD = 0.82°; controls: MED = 2.37°, MAD = 0.93°; see Figure 3B).
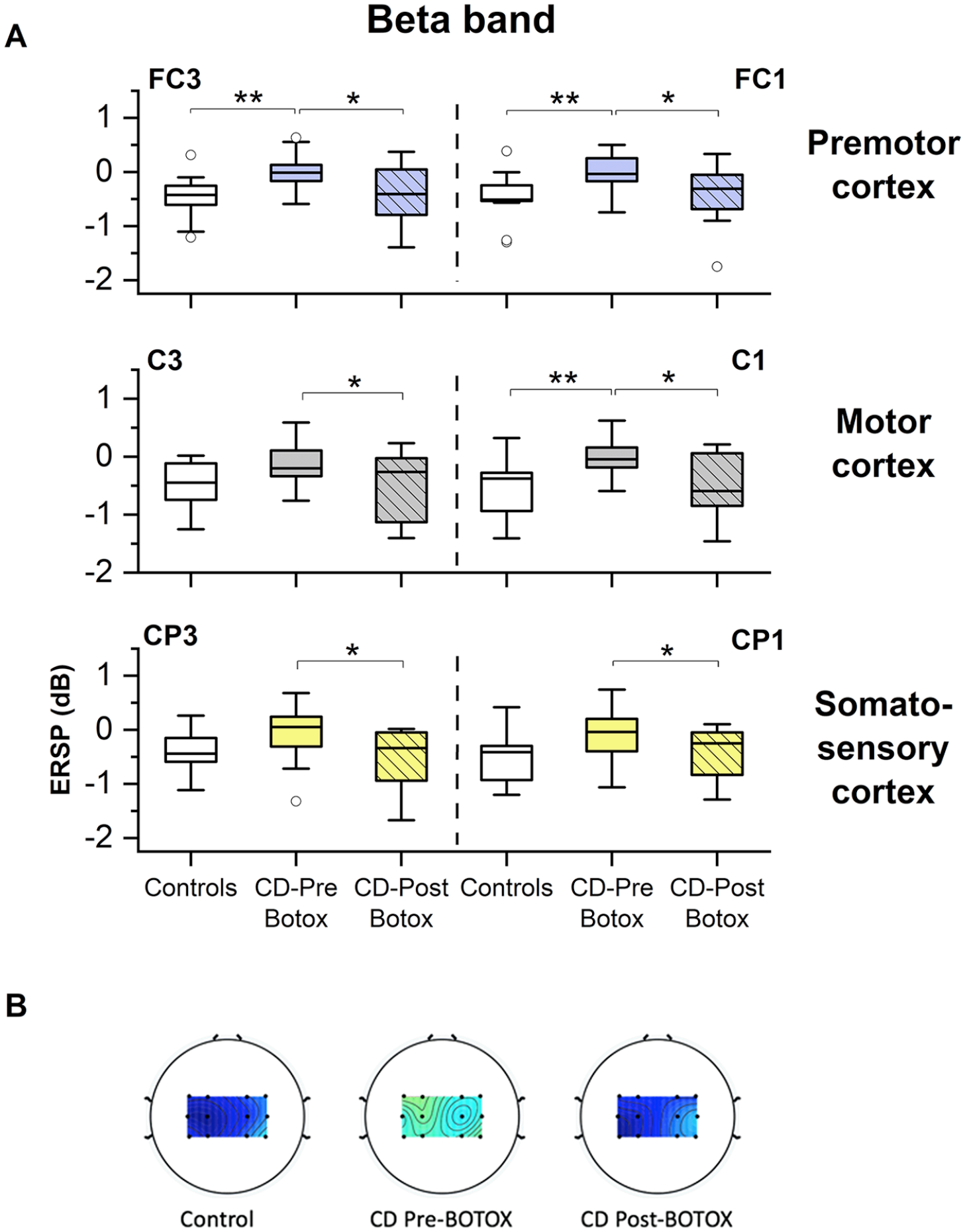
Comparison of β-band ERSP between the control group and CD patients, pre-BoNT and post-BoNT injection, during the anticipation period prior to the execution of the position matching task. (A) β-band ERSP over the left hemisphere. Patients at pre-BoNT had significantly larger spectral power over the left motor cortex in the β-band when compared with healthy participants. Within the patient group, BoNT injection was associated with the significant decline of β-band spectral power over the left somatosensory-motor cortical areas. Lower and upper boundary of each box indicates the 25% and 75% quartiles, respectively. The horizontal line within the box depicts the median. The upper and lower whiskers extend to +1.5 and −1.5 interquartile range, respectively. Outliers are shown as white circles. (B) Comparison of the group-averaged β-band ERSP over bilateral somatosensory-motor cortical areas during the preparatory stage of the wrist position matching between the control group, CD group pre-BoNT, and CD group post-BoNT injection. The dark blue color indicates suppression of β-band ERSP, and green/yellow indicates the rise of β-oscillations in relation to the resting condition. As a group, CD participants had a higher mean β-band ERSP over somatosensory-motor cortical regions in their pre-BoNT state when compared with controls. This excessive activity was suppressed in the CD group after receiving neck BoNT injections.
Following BoNT injection, the left hemisphere of CD patients exhibited a significant suppression of β-band ERSP over somatosensory and motor cortical areas (Figure 2): C1 (P = .0183; d = 1.06), CP1 (P = .0201; d = 0.88), FC1 (P = .0361; d = 0.81), C3 (P = .0361; d = 0.83), CP3 (P = .0201; d = 0.92), and FC3 (P = .0201; d = 0.95).
We also explored the relation between β-band ERSP and ME in the CD group post-BoNT versus pre-BoNT (Figure 4) to understand if there is a consistent EEG signature of the observed perceptual-motor error in CD. Figure 4 illustrates that for the majority of study participants, reduction in ME was associated with a suppression of β-band ERSP during the motor planning phase of the matching movement, although there was no clear sign that the magnitude of cortical suppression was closely related to the magnitude of the decrease in perceptual-motor error.
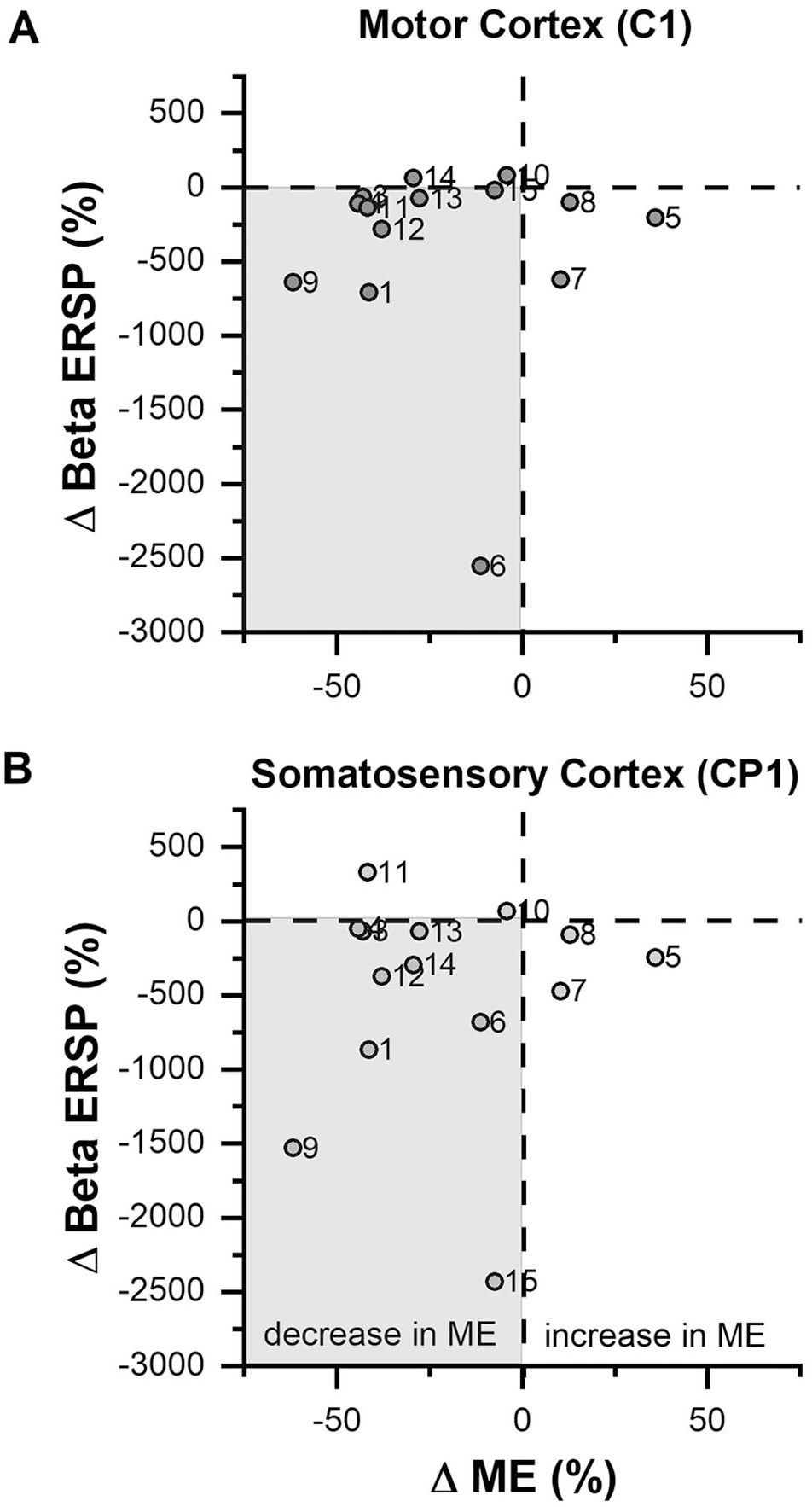
Relationship between the relative percentage change in ME and the relative percentage change in ERSP within the CD group over the left motor (C1) and somatosensory (CP1) cortex, post-BoNT versus pre-BoNT injection. It can be observed that for the majority of participants, reduction of active wrist joint position ME after the injection of neck BoNT was concurrent with the reduction of β-band power over the contralateral somatosensory-motor cortical areas.
Discussion
This study evaluated the behavioral and neural aspects of wrist proprioceptive perception in CD pre-BoNT and post-BoNT injection. The main findings were as follows: first, in their symptomatic state, patients showed a significantly higher DT compared with controls, reflecting a primary somatosensory deficit in CD. BoNT injection had no effect on DT. Second, in their symptomatic state, the active position sense of the CD group was abnormal, as indicated by a significantly larger ME compared with controls. In the optimal state, this error was no longer significant, reflecting that neck BoNT injections did affect active joint position sense. Third, CD patients in their symptomatic stage (pre-BoNT) exhibited a reduced level of β-band desynchronization during the wrist movement preparation phase when compared with controls. In their optimal state (post-BoNT), this significant difference was no longer present in the CD group, meaning that, as a group, the excessive β-band activity during motor preparation was significantly reduced after BoNT injection.
Abnormal Wrist Proprioception: Evidence for a Primary Generalized Sensory Deficit in CD
There is increasing evidence that faulty processing of somatosensory information originating from tactile and proprioceptive mechanoreceptors is common in focal dystonia.9,24 Numerous reports documented that dystonia patients have impaired abilities in discriminating tactile stimuli in the spatial and temporal domains.5,25,26 Moreover, finger position sense thresholds and the perception of arm motion were found to be abnormal in CD or blepharospasm,6,7 which reflects proprioceptive abnormalities across multiple body regions. Detecting such abnormalities in nonaffected body segments indicates a widespread neurophysiological trait for focal dystonia.
Our data clearly corroborate these earlier findings by showing that wrist proprioceptive acuity decreases in CD. Given that we tested passive and active position sense, we can now extend on this previous work and document that both types of position sense are affected in CD. We know from PET imaging work that passive movement mainly activates contralateral primary and secondary somatosensory regions, whereas active movement is associated with a much wider network with additional activation in the primary and premotor cortex, the supplementary motor area, the basal ganglia, and the cerebellum. 27 We also know that proprioceptive mechanoreceptors are intact in CD, 28 excluding the possibility that the faulty proprioception function is a result of altered afferent proprioceptive signals but, foremost, a primary sensory deficit likely associated with abnormal processing of proprioceptive afferent information reaching the somatosensory cortex. Neurophysiological evidence consistent with this view comes from primate models of focal hand dystonia reporting “smeared” overlapping and highly enlarged receptive fields in area 3b, 29 which could explain the finding of abnormal proprioceptive perception in nondystonic limbs.
In contrast, our data are not in support of the notion that focal dystonia reflects a primary deficit in sensorimotor integration. In such a scenario, the processing of proprioceptive information in the somatosensory system is intact, but the motor system is unable to use this information to control posture and voluntary movement. It would imply that proprioceptive perception during passive limb movement is intact in CD, whereas it is impaired during active motion. Instead, our data provide evidence for impairments in both active and passive limb position sense. Moreover, the magnitude of proprioceptive error for both types of position sensing was nearly identical. Sensing wrist position using passive joint displacement yielded, on average, a 51% larger threshold for the CD group when compared with healthy controls and a 53% larger error when CD patients attempted to actively match a remembered joint position (Figure 3). This implies that CD does not impair mechanisms of sensorimotor integration to the extent that proprioceptive signals increase the magnitude of the error (Figure 4). Finally, it is noteworthy that Parkinson’s disease, another disorder affecting basal ganglia-cortical sensorimotor networks leads to similar deficits in active and passive somatosensory perception. 10 Given the somatotopic organization that is present within the basal ganglia-thalamocortical circuitry, it is plausible that the subcortical nuclei of this circuitry also exhibit altered mechanisms of sensorimotor processing during motor preparation. Consequently, neck BoNT injection is expected to have an impact on the neural activity within these nuclei.
Reduced β-Band Desynchronization During Motor Preparation in CD
While our psychophysical data quantified the magnitude of wrist proprioceptive deficits in CD, our EEG data allowed us to examine the underlying cortical dynamics of limb position sensing during the active joint position matching task. Previous research confirmed a reduced movement-related desynchronization of β-band activity in cervical and segmental dystonia,30,31 writer’s cramp, 32 and in α-band activity for laryngeal dystonia 33 . This study documents that cortical β-wave desynchronization in CD is significantly reduced when preparing the active joint position matching movement—that is, during a sensorimotor preparation stage characterized by the processing of afferent somatosensory information, the integration of remembered and current joint position information, and the mapping of such sensory information onto motor commands (sensorimotor integration).
It is widely known that α (8-12 Hz) and low β (13-20 Hz) are the predominant frequency components of local field potentials in the human sensorimotor cortex 34 and that these oscillations are reduced in amplitude (“desynchronized”) during voluntary movements. 35 Rhythmic β activities are hypothesized to contribute to 2 main functional roles in the human sensorimotor system: the recalibration/integration of sensory-motor mechanisms 36 and the presentation of an essential state for the maintenance of steady-state muscle contractions.37,38 The desynchronization of β-band power relative to the resting state is considered as an indicator of the onset of movement preparation and the anticipation of somatosensory tasks.39-41 Therefore, our finding of excessive β-band cortical activation before movement onset is another piece of evidence for abnormal somatosensory processes in CD.
Botulinum Injection Modulates Somatosensory-Motor Cortical Oscillations in CD
When comparing the position sense DTs or joint position MEs of the CD group in their symptomatic versus the optimal state (pre-BoNT vs post-BoNT), no significant differences in both measures of proprioceptive function emerged. However, compared with controls, CD participants no longer showed significantly larger wrist joint position MEs after neck BoNT injection. Given that the effect of BoNT injection on the modulation of central neural processes is a result of the cholinergic blockade of extrafusal and intrafusal muscle fibers, BoNT may (1) act like a sensory trick in CD by altering the inputs of somatosensory afferents from intrafusal muscle spindles or (2) create a form of short-term plasticity via transsynaptic neurotoxin migration. 13 The altered peripheral somatosensory signals would reach the somatosensory cortex via afferent pathways, ultimately affecting the different cortical/subcortical components of the sensorimotor network. 42
Limitations of the Study
The fact that participants in this study did not receive identical levels of BoNT during injection constitutes a confounding factor that could have affected the level of change in β-band ERSP in CD participants. We could not control for this factor, which implies that part of the response variability was a result of the presence of different levels of BoNT. In addition, our analysis cannot establish a causal relationship between the reduction in β-band cortical synchronization of the CD group after BoNT and the related improvement in proprioceptive acuity. One could argue that the improvement of wrist joint position MEs and the observed reduction in cortical synchronous activity over the somatosensory-motor cortex are simply 2 unrelated phenomena, albeit both are associated with the administration of BoNT. Although this is a valid argument, we would contend that we observed this abnormality in cortical processing not during an unrelated motor task but in a perceptual task that relied on an active movement. That is, the task itself and the cortical activity prior to initiating a matching movement contained a proprioceptive processing component.
Conclusions
Abnormal passive and active proprioceptive perception of the nonsymptomatic wrist joint is a sensory manifestation of CD. An excessive rise of β-band cortical synchronizations during motor planning corresponds to this somatosensory deficit. Neck BoNT injections led to a significant decline of abnormal β-oscillations and the improvement of active wrist joint position MEs in patients, indicating that local injections did affect the central mechanisms of proprioceptive function in CD. These findings provide a better understanding of cortical abnormalities associated with motor planning in CD and may set the basis for proposing novel biomarkers for the disease. From a clinical perspective, studying the cortical oscillatory behavior that corresponds to abnormal motor preparation in CD would also open an avenue for a broader and more directed use of noninvasive neurorehabilitation therapies for CD, such as transcranial alternating current stimulation. This can be achieved by targeting and modulating those brain oscillations that are particularly altered in this disorder and by bringing them into the range of heathy individuals.
Footnotes
Acknowledgements
We greatly appreciate Dr Dennis Dykstra, Associate Professor of Rehabilitation Medicine, University of Minnesota, Mrs. Tanya Baxter from M Health Fairview Clinics and Surgery Center, University of Minnesota, and Dr Joshua Aman, Assistant Professor of Neurology, University of Minnesota, for their invaluable support in recruiting the study participants. We sincerely thank all attendees who kindly and generously dedicated their time to this research. We also extend our gratitude toward the Center for Applied and Translational Sensory Sciences (CATSS) for providing the recording facilities.
Authors’ Note
Conceptualization and experimental design: JK and SK. Data acquisition: SK and JB. Data analysis and interpretation: SK, JK, MDJ. Preparation of the first draft of the article: SK and JK. Critical review of the article: JK, MDJ, JB. Supervision of the work: JK.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially funded by the Center for Applied and Translational Sensory Science at the University of Minnesota via a graduate student grant awarded to SK.
