Abstract
The most difficult clinical questions in stroke rehabilitation are “What is this patient’s potential for recovery?” and “What is the best rehabilitation strategy for this person, given her/his clinical profile?” Without answers to these questions, clinicians struggle to make decisions regarding the content and focus of therapy, and researchers design studies that inadvertently mix participants who have a high likelihood of responding with those who do not. Developing and implementing biomarkers that distinguish patient subgroups will help address these issues and unravel the factors important to the recovery process. The goal of the present paper is to provide a consensus statement regarding the current state of the evidence for stroke recovery biomarkers. Biomarkers of motor, somatosensory, cognitive and language domains across the recovery timeline post-stroke are considered; with focus on brain structure and function, and exclusion of blood markers and genetics. We provide evidence for biomarkers that are considered ready to be included in clinical trials, as well as others that are promising but not ready and so represent a developmental priority. We conclude with an example that illustrates the utility of biomarkers in recovery and rehabilitation research, demonstrating how the inclusion of a biomarker may enhance future clinical trials. In this way, we propose a way forward for when and where we can include biomarkers to advance the efficacy of the practice of, and research into, rehabilitation and recovery after stroke.
Keywords
Introduction
Stroke is a heterogeneous condition, making choice of treatment, and prediction of outcome and treatment response, difficult. Despite this, clinical trials are often designed with a ‘one size fits all’ point of view, which can make them vulnerable to patient heterogeneity, reduced statistical power, and thus failure. Biomarkers can greatly inform patient selection for trials in general medical research, and this is equally true for stroke recovery. A stroke recovery biomarker (SRB) can be defined as an indicator of disease state that can be used as a measure of underlying molecular/cellular processes that may be difficult to measure directly in humans, and could be used to understand outcome, or predict recovery or treatment response. 1
In practical terms, biomarkers should improve our ability to predict long-term outcomes after stroke across multiple domains. This is beneficial for: (a) patients, caregivers and clinicians; (b) planning subsequent clinical pathways and goal setting; and (c) identifying whom and when to target, and in some instances at which dose, with interventions for promoting stroke recovery. 2 This last point is particularly important as methods for accurate prediction of long-term outcome would allow clinical trials of restorative and rehabilitation interventions to be stratified based on the potential for neurobiological recovery in a way that is currently not possible when trials are performed in the absence of valid biomarkers. Unpredicable outcomes after stroke, particularly in those who present with the most severe impairment 3 mean that clinical trials of rehabilitation interventions need hundreds of patients to be appropriately powered. Use of biomarkers would allow incorporation of accurate information about the underlying impairment, and thus the size of these intervention trials could be considerably reduced, 4 with obvious benefits. These principles are no different in the context of stroke recovery as compared to general medical research. 5
Interventions fall into two broad mechanistic categories: (1) behavioural interventions that
Our goal is to provide a consensus statement regarding the evidence for SRBs that are helpful in outcome prediction and therefore identifying subgroups for stratification to be used in trials. 7 We focused on SRBs that can investigate the structure or function of the brain (Table 1). Four functional domains (motor, somatosensation, cognition, and language (Table 2)) were considered according to recovery phase post stroke (hyperacute: <24 h; acute: 1 to 7 days; early subacute: 1 week to 3 months; late subacute: 3 months to 6 months; chronic: > 6 months 8 ). For each functional domain, we provide recommendations for biomarkers that either are: (1) ready to guide stratification of subgroups of patients for clinical trials and/or to predict outcome, or (2) are a developmental priority (Table 3). Finally, we provide an example of how inclusion of a clinical trial-ready biomarker might have benefitted a recent phase III trial. As there is generally limited evidence at this time for blood or genetic biomarkers, we do not discuss these, but recommend they are a developmental priority.9 –12 We also recognize that many other functional domains exist, but focus here on the four that have the most developed science.
Summary of Possible Brain Biomarkers to Measure Brain Structure or Function.
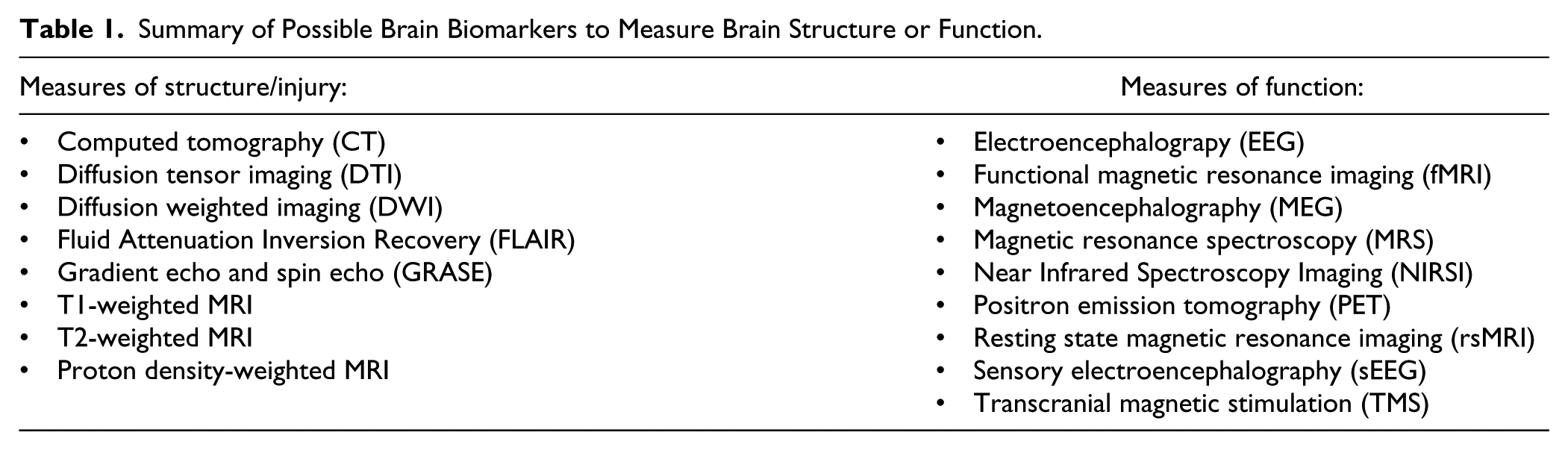
Scope of Functional Domains Considered Given Existing Literature.
Scope for Expert Consensus Biomarker Recommendations.

A challenge across the reviewed literature was to determine where the biomarker data explained recovery beyond that denoted by clinical outcome measures, such as the proportional recovery rule that has been demonstrated using motor,3,13 visuospatial neglect 14 and language 15 outcomes. Given the recency of these models, we were unable to address this in this paper and urge people to consider this in their future trial design. Further, to fully understand the predictive capacity of biomarkers, we need to move beyond cross-sectional studies, which constitute the bulk of the biomarker literature at present, and conduct mechanistic studies that go beyond simple correlations, as well as conduct longitudinal studies that provide data useful for predicting outcome or treatment response.
Motor
Neuroimaging biomarkers include quantitative characterisation of the stroke lesion itself, as well as the structure and function of non-lesioned brain areas. There is little consensus regarding the usefulness of characterizing the ischemic penumbra at the hyper-acute stage in relation to predicting motor outcomes. Recent data suggest that the site of ischemic penumbra, rather than volume, could predict outcome or treatment response (i.e. to thrombolysis) and affect motor recovery. 16 Acute infarct volume correlates with motor outcome (National Institute of Health Stroke Severity (NIHSS)), but this relationship is attenuated with increasing leukoaraiosis severity.17,18 The extent of existing white matter disease (i.e. leukoaraiosis) has been associated with acute lesion size, degree of lesion expansion and stroke severity indicated by initial NIHSS score. 19 These findings underscore the point that biomarker performance varies across different stroke subgroups.
Measures of corticospinal tract (CST) white matter integrity in the acute stage may predict motor outcome. Early measurement of CST fiber number via diffusion tensor imaging (DTI), a reflection of white matter integrity, predicts motor outcome (Fugl–Meyer score) at 12 months, especially for patients with initially more severe impairment. 20 Other data, 21 though not all, 22 also suggest that fractional anisotropy (FA) of the ipsilesional and contralesional CST at the acute stage is higher in individuals who achieve better motor recovery after stroke. Data also support the potential utility of the asymmetry between the ipsilesional and contralesional CST FA to predict treatment gains in the chronic stage. 23
Measures of the extent of CST injury in the
Other regions distant from the lesion influence motor recovery. While measures related to distant regions require further development to reach the stage of valid biomarker, several useful observations have been published in the
There is broad consensus that the presence of an upper limb motor evoked potential (MEP) in response to transcranial magnetic stimulation (TMS) at the hyperacute and acute stages strongly predicts good motor outcome37,38 and that shorter MEP latencies and central motor conduction times are associated with better outcome. 39 The presence of a MEP has been found to identify which patients will follow the proportional recovery rule. 40 Similarly, in the leg, the presence of a MEP indicates that an individual is more likely to be independently mobile 12 months post-stroke,41–43 yet this measure does not relate to walking recovery. 13 Prediction of recovery is more challenging for patients without an MEP40,44 and combining TMS with MRI biomarkers may be useful in this context 38 TMS at the chronic stage helps explain the relationship between corticomotor function and motor performance in cross-sectional studies, and those who have an MEP are more likely to benefit from physical interventions.23,45,46
Resting state functional connectivity (rsFC) findings in the early and late subacute phases converge on the conclusion that interhemispheric connectivity is of particular importance to motor control. Cross-sectional studies have demonstrated decreased rsFC correlates with the degree of motor impairment.47,48 The are positive associations between acute/early subacute rsFC (ipsilesional primary motor cortex [M1] to contralesional thalamus, supplementary motor area (SMA), and medial frontal gyrus) and motor outcomes at six months (Fugl–Meyer score). 49 In late subacute patients, the amount of CST damage combined with interhemispheric M1 rsFC best predicted therapy-induced gains. 27 Fan et al. 50 found that in late subacute patients, change in interhemispheric M1-M1 rsFC predicts improvements in the Wolf Motor Function Test. Results from a ridge regression machine-learning algorithm analysis of a large sample of early subacute stroke patients suggest that rsFC may explain a smaller amount of the behavioral variance observed than the amount of structural damage to the CST. 51 As rsFC can be performed in patients with severe deficits after stroke and can interrogate all brain networks simultaneously, it represents a priority for development.
Quantitative indices extracted from functional MRI (fMRI) in the early and late subacute stage, such as the laterality index from M1, and the study of its change over time, show that stroke is associated with a less lateralized pattern of activation as compared to healthy subjects, a finding that is exaggerated among patients with poorer outcomes.52,53 One unifying conclusion across studies is that the best motor outcomes are associated with the greatest shift towards the normal state of brain function. 54 The laterality index has been used as a judgment criterion of efficacy in the chronic stage in trials testing mirror therapy, 55 constraint-induced therapy 56 and robotic intervention protocols, 57 and can predict treatment response. 58 Other fMRI metrics such as activation volume59,60 or percent signal change 61 within key motor network nodes can predict response to treatment in the chronic stage. As there are fewer long-term studies of the laterality index, and it often shows significant biological associations, this is an area of priority for development.
Magnetoencephalography (MEG) or electroencephalography (EEG), non-invasive measures of cortical neuronal oscillations, are sensitive to alterations in both GABAergic and glutamatergic signalling that are important for plasticity and recovery after stroke.62–64 Changes in cortical excitation and inhibition represent novel therapeutic targets, but cannot be measured directly in humans. Stroke patients with poorer outcomes have persistent, increased low-frequency oscillations at the acute, and early/late subacute stages,
65
suggesting predominant inhibitory mechanisms in the peri-lesional cortex. Acutely, lower beta-rebound in response to tactile finger stimulation (which indicates increased early post-stroke sensorimotor excitability)
66
and increased somatosensory map size
67
predict good recovery after stroke. Also, in a single stroke patient, zolpidem
Combining neuroimaging and neurophysiology biomarkers may be useful for predicting motor outcomes and therapy response. 70 Upper limb outcomes at three months can be predicted at the early sub-acute stage by measuring first clinical, then TMS and finally MRI biomarkers in a stepdown approach, as in the PREP algorithm.71,72 Stoykov and Stinear 73 treated chronic stroke patients using active-passive bilateral arm training and discovered that fractional anisotropy asymmetry between the two CST tracts accounted for 40% variability in clinical improvement. Factoring in whether patients were MEP+/− improved the predictive model. One recent study emphasized that combining neuroimaging measures of neural injury and neural function was key to best predicting response to a standardized robotic therapy in the chronic stage. 27
In summary, neuroimaging and neurophysiology CST biomarkers can predict motor outcome and response to therapy after stroke, and are recommended for use in clinical trials, e.g. for stratifying patients. The evidences for rsFC, fMRI and MEG/EEG biomarkers are promising and are developmental priority areas (Table 4a).
Expert Consensus Biomarker Recommendations.

CST: corticospinal tract; CT: computed tomography; DTI: diffusion tensor imaging; FC: functional connectivity; fMRI: functional magnetic resonance imaging; MEP+: motor evoked potential present; MEG: magnetoencephalography; MEP-: motor evoked potential absent; MRI: magnetic resonance imaging; PLORAS: predicting language recovery and outcome after stroke; rsFC: resting state functional connectivity; sEEG: sensory electroencephalography.
Somatosensory
Currently, there are few studies of structural or functional biomarkers conducted to understand outcome, predict recovery or predict treatment response in the somatosensory functioning domain in the hyperacute or acute phases post-stroke. Most work on structural biomarkers involving the non-lesioned brain has focused on understanding outcome by mapping the structural integrity of residual pathways. Feasibility of visualisation of sensorimotor systems by tracking fibres has been demonstrated in hyperacute, acute and early subacute phases for somatosensory symptoms. 74 There are changes in morphology of the somatosensory cortex of chronic stroke patients, 75 with co-localized structural (cortical thickness) and functional (brain activation, tactile stimulation) effects. Only one study noted associations between the structure of somatosensory regions and motor outcome in the chronic phase. 76
Failure to activate the somatosensory cortex during median nerve stimulation in the acute stage predicts poor clinical recovery at three months. 77 Using MEG, reduction in interhemispheric asymmetries of activity at chronic compared to acute phases was associated with a worse clinical state. 78 Studies using MEG in early and late subacute phases show that changes in source strength of the primary somatosensory cortices correlate with the extent of recovery of sensorimotor functions as determined by neurological exams (e.g. graphesthesia test). 79 Yet, MEG can be complicated to employ, and so broader evaluation of these findings might benefit from use of less complex electrophysiological methods such as EEG. In the subacute phase, differences in brain activity measured with task-related fMRI correlated with touch impairment in patients with thalamus /internal capsule lesions compared to those with lesions of primary (SI) or secondary (SII) somatosensory cortex. 80 Similarly, responsiveness of SI at 1–15 days post-stroke is associated with improvement of two-point discrimination three months post-stroke. 81 Resting-state FC studies of touch impairment and recovery in the chronic phase demonstrated a correlation between changes in connectivity from contralesional SII and contralesional inferior parietal and middle temporal gyrus with changes in a tactile discrimination score that were absent in a control group. 82
Associations are observed between somatosensory function (using the Hand Active Sensation Test) and a left/right FA ratio from the sensory component of the superior thalamic radiation in the chronic stage 83 and the frontoparietal tracts in the acute 84 and chronic 85 phases. In addition, somatosensation function in the chronic phase correlates with activity in the ipsilesional and contralesional primary sensorimotor cortex 86 and a more distributed pattern of activity involving parietal cortex. 83 Improvement in touch discrimination at six months was associated with increased rsFC between seeds in the contralesional hemisphere and distributed regions, including cerebellum. 82 Using MEG, involvement of ipsilesional primary hand representation areas positively contributed to clinical recovery. 87
Changes have also been reported in association with training of touch discrimination, 88 passive proprioception 89 and sensorimotor function, 90 with a focus on tracking outcomes and mechanisms, rather than prediction. For example, touch discrimination training of patients with somatosensory loss in the chronic stage post-stroke was associated with different patterns of change in activation with thalamic/capsular compared to SI/SII cortical lesion. 88 This area of research is a priority for development.
There is insufficient evidence to recommend the use of any specific biomarkers of somatosensory system function in clinical trials; however, several candidates are suggested. The recovery of somatosensation is often overlooked despite well-documented observations that impaired sensation is an impediment to optimal recovery.91–93 Functional biomarkers, including task-related activation and rsFC are a developmental priority (Table 4b).
Cognition
Mapping executive/cognitive functions to specific brain regions is problematic because these functions are distributed widely across broad brain networks, and their relationships are complex. Indeed, studies that readily identify structure–function relationships for phonology and semantic processing, and often fail to find an equivalent for executive function. 94 The most consistent relationships were found in white matter. Frontal and basal ganglia region microbleeds were associated with executive dysfunction outcome in the chronic phase, 95 and another study found that mean diffusivity of normal appearing white matter (whole brain) in non-lesioned areas correlated with outcomes for executive function among individuals with ischaemic leukoaraiosis (plus a previous lacunar stroke).96,97 EEG changes in frontal lobes paralleled behavioral gains across multiple cognitive domains in one study that used intensive video gaming in health adults 98 ; the same may extend to patients with stroke.
Though functional imaging methods may offer the best hope of generating robust biomarkers for executive function, there is little published work. Available associations are correlative and from cross-sectional studies, rather than predictions of outcomes or more complex evaluation of biological hypotheses. In the late subacute phase, executive functioning correlates with alpha band functional connectivity between the left fronto-opercular cortex and the rest of the brain. 99 Yet, it is possible that the task-dependent changes observed with functional imaging data have less to do with new domain-specific areas being generated, and more to do with cognitive control networks improving residual performance. 100 In individuals with chronic post-stroke aphasia, a positive correlation between task-dependent activity in midline frontal cortex and language recovery was interpreted as reflecting domain-general cognitive control systems, 101 a finding that is consistent with training effects in healthy elderly subjects. 98 Development of biomarkers in this context would likely foster advances in therapeutic techniques to train executive function, a key priority that is feasible. 102
The default mode network (DMN) has emerged as a key biological substrate in the context of cognitive functioning. 103 Studies in the subacute and early chronic phases report altered rsFC in the DMN correlated with cognitive performance after stroke104–107 Re-emergence of the anticorrelation between the DMN and task-positive networks, such as the dorsal attention network (DAN), 108 is associated with behavioural recovery of cognitive functions. Resting state studies have provided robust examples of disruption of interhemispheric connectivity associated with domain-specific cognitive deficits47,51,109,110 and recovery. 111 In a small longitudinal study of stroke patients compared to healthy controls, He et al. 109 showed a robust correlation between left-right posterior intraparietal sulcus rsFC and accuracy of detection of targets presented in the left neglected visual field. Multiple studies have confirmed this connectivity is much depressed in the acute stage after stroke in persons with USN and returns toward normal in association with the resolution of symptoms, with the largest current longitudinal study retaining 64 stroke patients at 12 months. Similar relationships have been reported for the DMN and other networks.47,51,109,110 While correlational analyses cannot establish causality and do not provide the predictive functions required of an effective SRB, the finding that a change in rsFC correlates with behaviour lends support to the idea that measures of network connectivity have the potential to serve as useful biomarkers across multiple behavioural domains, a possibility that requires further studies.
In the domain of spatial cognition, multiple moderately sized studies of right hemisphere injury confirm that damage to different major long range white matter tracts may predict chronic persistence of unilateral spatial neglect. Two well-designed longitudinal studies implicate the inferior occipitofrontal fasciculus and uncinate fasciculus 112 and decreased FA in the left and right superior longitudinal fasciculus II, and forceps major of the corpus callosum with neglect scores. In cross-sectional studies, linear regression shows an association of unilateral spatial neglect with damage to the fronto-parietal segment of the arcuate fasciculus, and that 78.9% and 81.6% of patients with neglect had damage to the superior longitudinal fasciculus II and superior longitudinal fasciculus III, respectively, compared to only 15% and 30% in patients without neglect. 113 While damage to superior longitudinal fasciculus III, arcuate fasciculus, frontal aslant, and frontal inferior longitudinal fasciculus are increasingly implicated in abnormal spatial cognition. 114 Whether damage to any of these white matter structures as measured in the acute phase of stroke can serve as a biomarker for persistence of USN in the chronic phase, or in stratifying or selecting patients for interventions is yet to be determined.
Based on this evidence, biomarkers of cognitive function, including executive functioning, are not ready for immediate broad implementation in clinical trials. Thus, further study and validation of biomarkers that explain current state and future chante in cognitive functions are a significant priority area for development. Resting state FC is a promising candidate biomarker (Table 4c), and study of its utility as a biomarker of recovery is emphasized here.
Language
There are a number of studies identifying a relationship between lesion site and aphasia, 115 anatomical findings that suggest potential metrics to evaluate as biomarkers. In the hyperacute period, perfusion-weighted MRI showed that word comprehension deficits are strongly correlated with blood flow within Wernicke’s area. 116 A related study demonstrated that lexical processing was more strongly related to the volume of hypo-perfused tissue than the volume of lesion . 117 Imaging illustrates that recovery of word comprehension from the hyperacute to acute phase (three days) is associated with reperfusion of Wernicke’s area. 118 Recovery of naming in the hyperacute period is predicted by reperfusion of left posterior middle temporal/fusiform gyrus, Broca’s area, and/or Wernicke’s area.119–121 There are no established predictors of long-term (>3 days) recovery from biomarkers assessed in the hyperacute period (<24 h); thus, this is a developmental priority.
Impaired repetition in the acute phase was associated with structural damage to the arcuate fasciculus and Broca’s area as well as tissue dysfunction (hypoperfusion and frank damage) in the inferior portion of the left supramarginal gyrus and temporal-parietal junction. 122 Kummerer et al., 123 also observed that impaired repetition at this phase was associated with posterior temporal-parietal lesions and damage to the dorsal superior longitudinal and arcuate fasciculus, while comprehension deficits were associated with ventral extreme capsule fibre damage. Measures of functional connectivity may also be useful in this phase, as a recent study of acute lacunar stroke patients observed that increased resting state FC between the left and right superior temporal gyri was correlated with poorer language function. 124
In the early subacute phase, there are relationships between lesion location and aphasia symptoms that suggest potential biomarkers. Kreisler et al. 125 correctly classified 67% to 94% of patients based on analysis of lesion location and symptoms. Forkel et al., 126 demonstrated prediction of recovery at six months was improved by adding volume of the left long segment of the arcuate fasciculus to a regression model including age, sex, and lesion size; including volume of the right long segment of the arcuate fasciculus further improved recovery prediction. Recent work by Geranmayeh et al., 127 showed that propositional language production is predicted by interactions between brain networks (DMN, fronto-temporo-parietal, and cingulo-opercular networks) rather than by activity within a single individual network highlighting the distributed nature of language operations. Functional MRI activity in the early subacute phase shows promise as a predictor of long-term recovery when analyzed using a multivariate machine learning technique. Saur et al., 128 employed this method with a mask of task-induced fMRI activity in bilateral frontal and temporal regions in combination with behavioural language performance and age. This approach correctly predicted good versus poor language recovery in 86% of individuals with stroke who had aphasia at two weeks. In the largest case-control study of subacute stroke patients with aphasia to date, Yang et al. 124 found that in patients with lacunar stroke, interhemispheric rsFC was increased in the superior temporal gyrus, the inferior frontal gyrus and the lingual gyrus. Of note is the observation that the interhemispheric hyperconnectivity of the superior temporal gyrus was inversely correlated with the aphasia quotient, indicating that greater connectivity was associated with worse aphasia.
Voxel-based analyses in the chronic phase has established utility in multiple brain systems and specifically have identified structural damage associated with particular aphasic symptoms, distinguishing between semantic and phonological processes and recognition versus production.129–131 Arcuate fasciculus lesion load negatively influences speech production 132 and classifies severe and non-severe outcomes with 90% accuracy for naming and 96% accuracy for speech fluency. 133 The PLORAS (Predicting Language Outcome and Recovery After Stroke) system 134 uses a Gaussian process model regression with a large database of stroke patients (from one month post, therefore covering early and late subacute, and chronic phases) with structural MRI, demographic, and language performance to provide predictions of aphasia recovery at the individual level. Using this approach and covariate factors of time of stroke, volume, and 35 different brain regions, predictions of language outcome, and within subject changes in speech production, have been identified. 135 This method has high potential to provide measures that can serve as biomarkers to predict recovery.
Posterior middle temporal lobe damage can negatively affect aphasia therapy outcome in the chronic phase. 122 Meinzer et al. 136 observed a negative relationship between the proximity of the lesion to the hippocampus and response to a naming treatment. Bonilha et al. 137 showed that measures of neural network connectivity combined with initial behavioral deficit severity accounted for 78% of variance in response to anomia treatment. Several small studies identified a relationship between therapy success and integrity of the left arcuate fasciculus, 138 right arcuate fasciculus 139 and white matter in proximity to the hippocampus. 136 Further, several fMRI studies have investigated treatment-induced aphasia recovery, predominantly in the chronic stage. Fridriksson 122 identified a significant relationship between treatment-induced naming improvements and fMRI activity in the both a posterior cluster (including parietal lobe and precuneus) and an anterior cluster (including middle frontal gyrus and pars opercularis). Subsequent analyses 140 showed that altered activity in perilesional areas was associated with increased naming accuracy, but measures of pre-treatment brain activity (as opposed to changes in activity) predicted improvement in semantic errors, suggesting additional factors contribute to treatment outcome. Resting state FC has also shown potential for understanding and predicting aphasia recovery in the subacute and chronic phases (including treatment induced improvements); however, larger studies are required to validate this approach. 141
In summary, in the acute and early subacute stages, the use of structural MRI provides insights into the neural basis of language deficits, but there are not sufficient large studies demonstrating that these methods clearly improve prediction of recovery or treatment response. Functional brain assessments such as via fMRI show potential at the early subacute stage for significantly improving prediction of outcome; 127 however, this approach needs validation. Structural MRI and DTI may forecast recovery at the late subacute and chronic stage, suggesting the possible use of these techniques to stratify patients for clinical trials, understand therapy mechanism and predict outcome. It should be noted that: (1) there is still considerable variability in outcome that is not accounted for by these methods, (2) each method uses a unique and complex analysis technique, (3) different aphasia treatments may engage unique networks (Table 4d), and (4) detailed studies examining the combined utility of anatomical and functional brain measures for predicting language recovery are warranted.
Conclusions
How might biomarker data be incorporated into future stroke recovery research? As a first point, the term “stroke” is inadequate, as it describes a very heterogeneous group of disorders that are unified by a vascular injury, but not by size, location, or impact of injury. Biomarkers present a way forward to subgroup or stratify patients in order to reduce variance and increase power, allowing for smaller sample sizes. 7 Moreover, the final behavioral phenotype after stroke can arise from many different biological states, which could result in differential therapeutic responses; functional measures are complementary to anatomical/injury measures. Thus, a patient exploiting all possible compensatory brain mechanisms might have little room to improve, while a similar patient who uses no compensatory mechanisms might achieve benefit. 142 Furthermore, inclusion of appropriate biomarkers may improve the ability to disentangle treatment responders from non-responders.
Clinical trials therefore need to base participant eligibility on more than presence of a stroke, or behavioural status. Instead, patient selection should include appropriate biomarkers; ideally, these will be linked with preclinical methods as well as the biological mechanism of the therapy or treatment under investigation. For example, recently, a threshold was defined whereby no patient in the early and late subacute stage with >63% injury to the CST achieved clinically important gains associated with a robotic therapy. 27 This result highlights the ascendant role that neuroimaging measures need to play in clinical-decision making for post-stroke rehabilitation. 143
A useful example comes from the recent phase III Everest trial, 144 which relied on behavioural assessments to determine participant eligibility, and ultimately found that patients randomized to epidural motor cortex stimulation did not reach the primary efficacy endpoint more often than patients in the control group. However, a post hoc analysis of patients randomized to epidural stimulation found that the primary efficacy endpoint was reached more often (67%) by those with preserved motor evoked responses upon cortical stimulation compared to those lacking a response (27%). 26 Thus, had confirmation of physiological integrity of the biological target been an eligibility criterion (as was the case in all preclinical studies that were translated to generate this trial), the effect size would have been substantially higher and the trial results quite different. We believe that this example is highly useful in illustrating the utility of biomarkers in recovery and rehabilitation research and expect that the inclusion of biomarkers will enhance future clinical trials.
Footnotes
Acknowledgements
We would like to acknowledge Professor Julie Bernhardt for convening this first Stroke Recovery and Rehabilitation Roundtable (SRRR), Ms Katie Lafferty for moderating discussions at SRRR and Dr Karen Borschmann for project management. We acknowledge the following organisations for their financial support of the meeting: National Health and Medical Research Centre (NHMRC) Centre of Research Excellence in Stroke Rehabilitation & Brain Recovery (Australia), Heart and Stroke Foundation (HSF) of Canada, Heart and Stroke Foundation Canadian Partnership for Stroke Recovery (CPSR). An unrestricted educational grant was provided by Ipsen Pharma.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
LAB was supported by the Canada Research Chairs and MSFHR (CI-SCH-01796); KSH was supported by NHMRC (1088449) and Micheal Smith Foundation for Health Research (MSFHR) (15980); SCC was supported by a grant from NIH (K24 HD074722). DAC was supported by a University of Queensland Vice Chancellor’s Fellowship; LMC acknowledges support from National Health and Medical Research Council (NHMRC) grants (#1022694; #307905), James S. McDonnell Foundation 21st Century Science Initiative in Cognitive Rehabilitation -Collaborative Award (# 220020413), NHMRC CRE in Stroke Rehabilitation and Brain Injury (#1077898) and an Australian Research Council Future Fellowship (#FT0992299).
