Abstract
Keywords
Introduction
Electrical spinal stimulation, via electrodes implanted in the epidural space, has been used for the treatment of complex chronic pain by virtue of its purported neuromodulatory effect on the nociceptive afferents that enter the dorsal horn of the spinal cord.1,2 However, the effect of spinal stimulation on motor disorders that develop with pathologies and injuries, such as multiple sclerosis 3 and spinal cord injury,4,5 has also been studied. Both preclinical and human stud-ies have observed that spinal epidural stimulation trig-gers alternating rhythmic muscle activity patterns.1,3-11 Furthermore, these studies reported an increase in amplitude of voluntary movements in paralyzed limbs and a reduction in spasticity among patients with incomplete spinal cord injury.4-7 Activation of motor activity below the complete or incomplete motor spinal cord injury may be related to increased excitability of the motor pool mediated by several neuromodulatory mechanisms,12,13 including enhanced spinal and residual supraspinal input to the motoneuronal pools. 14
At the spinal level, transcutaneous spinal stimulation increases spinal reflex activity similar to that obtained with epidural stimulation. 15 Evoked Hoffmann-like reflex (H-reflex) activity may result from activation of proprioceptive afferents within the dorsal roots.16,17 The theory that noninvasive spinal stimulation may also evoke motor outcomes by facilitating both spinal and residual supraspinal motor pathways has also been advanced.16,17 Taking these studies into account, activation of the motor pools via activation of dorsal roots or interneurons, in people with spinal cord injury, could lower their activation threshold by residual supraspinal input. 14 The possibility of activating neural networks within the spinal cord by means of a noninvasive stimulation method opens a therapeutic window for the treatment of motor disorders following spinal cord injury.
Transcutaneous spinal stimulation applied alone or in combination with activity-based rehabilitation programs to improve motor function for individuals with chronic spinal cord injury has been studied during the previous 10 years and has recently received considerable scientific, clinical, and media attention. This review is timely because it provides detailed information regarding the published studies of transcutaneous spinal stimulation by assessing the methodological quality of the selected studies. Furthermore, and despite the number of studies that have examined the motor response and the therapeutic potential of the stimulus, the feasibility and efficacy of transcutaneous stimulation to evoke motor outcomes has not been characterized. This review, therefore, is aimed at analyzing the feasibility and efficacy of noninvasive spinal cord electrical stimulation to promote motor activity and function in patients with spinal cord injury. In addition, the range of stimulation parameters and spinal site of stimulation are also reviewed to understand the optimal protocol required to promote motor activity.
Methods
Search Strategy
To undertake this systematic review, we followed a protocol drawn up in accordance with the PRISMA guidelines.
18
A search was made of the following databases: PubMed, Cochrane Registry, and Physiotherapy Evidence Database (PEDro). In addition, we also carried out an inverse manual search of the references cited by the articles located. For search purposes we used the keywords
Selection Criteria
Using the PECOS structure (P-patients, E-exposure, C-comparison, O-outcome, and S-study design), we established the following inclusion criteria, requiring (1) patients to have spinal cord injury, (2) tonic electrical spinal stimulation to be applied transcutaneously, and (3) outcomes to include some type of assessment of motor response. We excluded articles that (1) failed to specify the stimulation parameters, (2) were based on the direct current stimulation modality, (3) used spinal stimulation for evaluation or neurophysiological and nontherapeutic purposes, and (4) reported narrative reviews. The selection of articles was decided by 2 independent researchers (AMG and DS-M) working in parallel, with any points of disagreement (2 in total) being settled by a third researcher (JG-S).
Study Selection
The process for selecting articles was as follows: first, any duplicates of studies found in the various databases were eliminated; second, after a perusal of the titles and an initial screening, the abstracts were read to identify those articles that fulfilled the preestablished inclusion criteria; and finally, the full text of the remaining studies was read, with any deemed to meet the exclusion criteria being ruled out (Supplementary Figure 1).
Assessment of Methodological Quality
The reviewed studies were principally composed of case reports and case series. Therefore, GRADE (Grading of Recommendations Assessment, Development and Eval-uation) and CARE (Case Report Guidelines) scores were calculated to estimate the methodological quality of these studies. Case reports and case series are uncontrolled study designs that are known to increase the risk of bias. The GRADEpro application, whose utility has been approved, 19 was used. The GRADE Scale rates the level of evidence of results, taking into account aspects relating to study methodology and results, such as (1) risk of bias, (2) inconsistency, (3) indirect evidence, (4) imprecision, and (5) other considerations (dose-response and reported biases). Moreover, GRADE can be used for both clinical trials and case reports. In addition, we used the CARE tool. This guideline provides information about the accuracy and transparency of case reports and case series. Finally, the PEDro Scale was used to assess clinical trials. 20
Data Analysis
Data were extracted for the following independent variables: number of patients included in the study, sociodemographic and clinical variables of the participants, and characteristics of the intervention applied. We analyzed the motor response as the principal dependent variable regardless of the tool used for its measurement (eg, electromyography [EMG]) and spasticity, reflexes, and perceived clinical improvement as secondary variables. In addition, we specifically took note of any possible adverse effects recorded. Because of the high degree of heterogeneity among studies and the lack of randomized clinical trials, a meta-analysis was ruled out, and a descriptive qualitative analysis of the data was performed instead.
Results
A total of 356 research reports were located in the above databases, with 25 studies identified through the review of bibliographic references cited by the studies. After elimination of duplicate reports and perusal of the titles and abstracts, 36 reports were selected for a reading of the full text. Finally, 13 studies were included in the systematic review (see Supplementary Figure 1). The results of the assessment of methodological quality are shown in Supplementary Table 1. The results of assessment of the strength of the evidence in references are shown in the appendix.
Of the reports included in the review, 10 were case or case-series studies21-31 and 2 were clinical trials32,33 with crossover designs; only 1 study compared the results of treatment with those of placebo stimulation. 33 Of these studies, 9 analyzed the lower extremities,21-27,29,33 3 analyzed the upper extremities,28,31,32 and 1 assessed motor response in the trunk. 30 The main characteristics of the studies are shown in Table 1.
Study Design Parameters of the Selected Studies for Transcutaneous Spinal Stimulation Facilitation of Motor Outcomes Following Spinal Cord Injury.
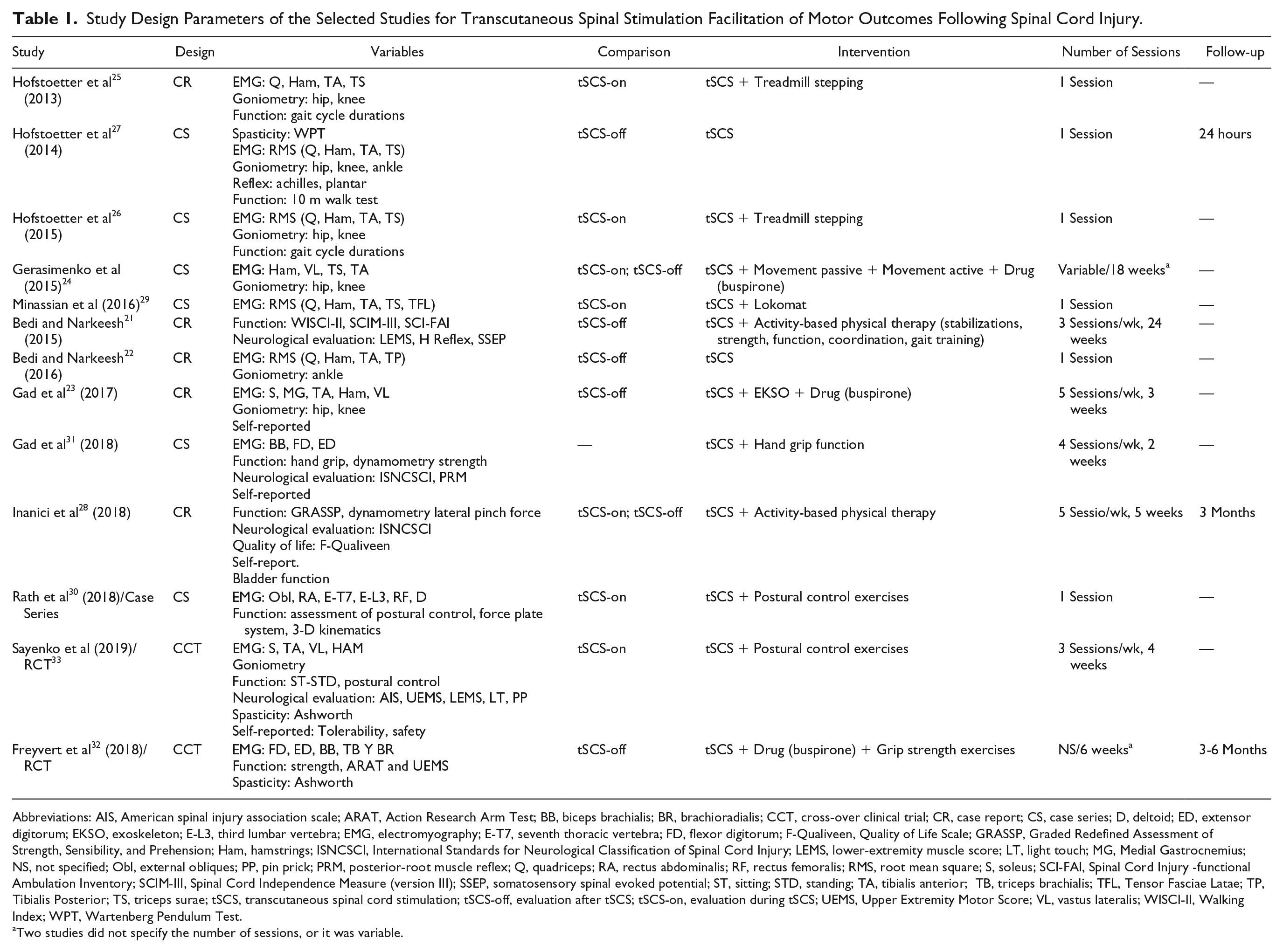
Abbreviations: AIS, American spinal injury association scale; ARAT, Action Research Arm Test; BB, biceps brachialis; BR, brachioradialis; CCT, cross-over clinical trial; CR, case report; CS, case series; D, deltoid; ED, extensor digitorum; EKSO, exoskeleton; E-L3, third lumbar vertebra; EMG, electromyography; E-T7, seventh thoracic vertebra; FD, flexor digitorum; F-Qualiveen, Quality of Life Scale; GRASSP, Graded Redefined Assessment of Strength, Sensibility, and Prehension; Ham, hamstrings; ISNCSCI, International Standards for Neurological Classification of Spinal Cord Injury; LEMS, lower-extremity muscle score; LT, light touch; MG, Medial Gastrocnemius; NS, not specified; Obl, external obliques; PP, pin prick; PRM, posterior-root muscle reflex; Q, quadriceps; RA, rectus abdominalis; RF, rectus femoralis; RMS, root mean square; S, soleus; SCI-FAI, Spinal Cord Injury -functional Ambulation Inventory; SCIM-III, Spinal Cord Independence Measure (version III); SSEP, somatosensory spinal evoked potential; ST, sitting; STD, standing; TA, tibialis anterior; TB, triceps brachialis; TFL, Tensor Fasciae Latae; TP, Tibialis Posterior; TS, triceps surae; tSCS, transcutaneous spinal cord stimulation; tSCS-off, evaluation after tSCS; tSCS-on, evaluation during tSCS; UEMS, Upper Extremity Motor Score; VL, vastus lateralis; WISCI-II, Walking Index; WPT, Wartenberg Pendulum Test.
Two studies did not specify the number of sessions, or it was variable.
Characteristics of Participants
The total study sample comprised 55 persons with spinal cord injury, 31 men, 9 women, and 15 participants where gender was not specified. Table 2 shows the demographic and clinical characteristics of the participants.
Demographic and Clinical Characteristics of Patients With SCI.
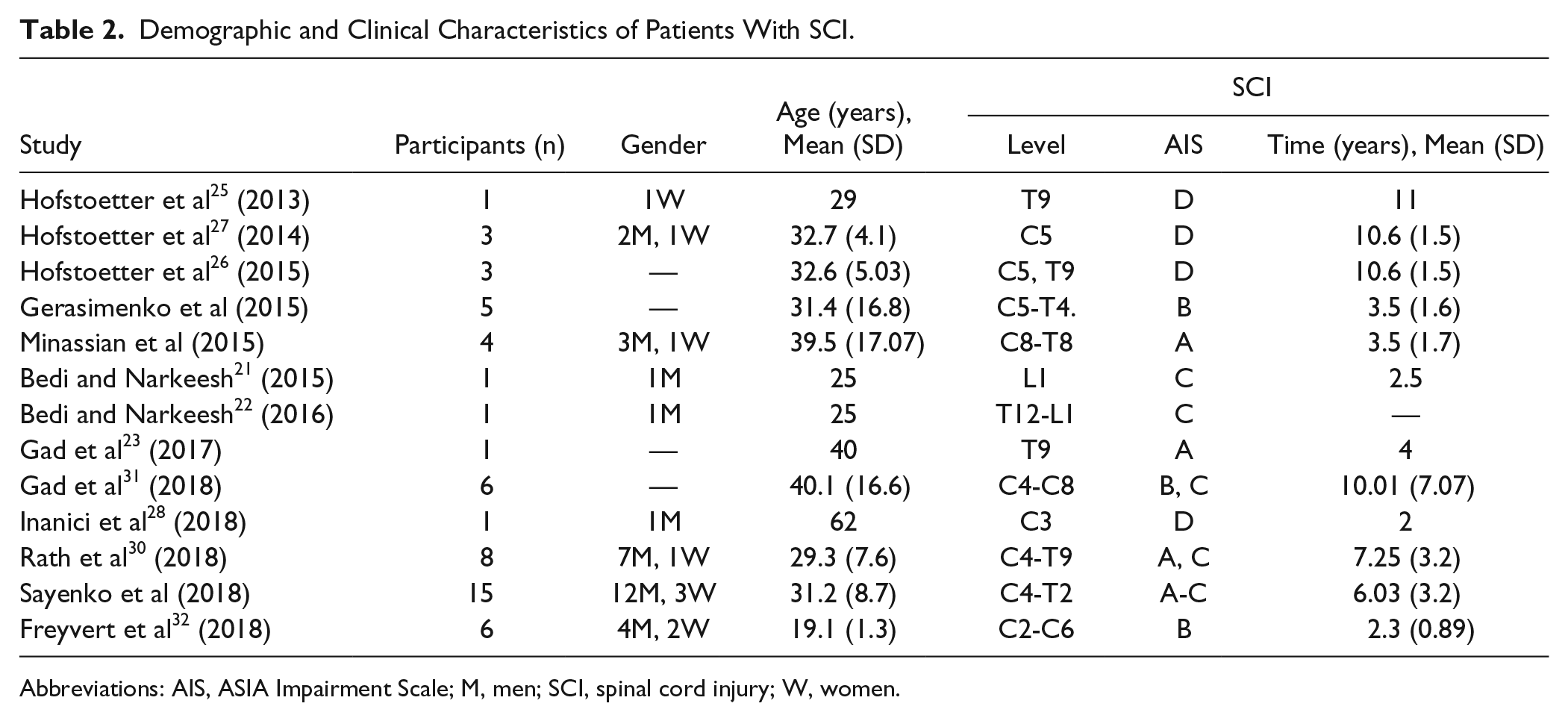
Abbreviations: AIS, ASIA Impairment Scale; M, men; SCI, spinal cord injury; W, women.
Stimulation Parameters
All studies that sought to induce muscle activation patterns in the lower extremities applied stimulation at the level of the T11-T12 interspinous space. Of these, 6 applied stimulation simultaneously at adjacent levels, such as L1-L221,22,30,33 or the first coccygeal vertebra.23,24 Three studies28,31,33 applied current to the cervical spine at the level of the C3-C4 and C5-C6 spinal segments (interspinous space) to assess the effects on the upper extremities.
In terms of the type of current, all studies used a rectangular wave, with the waveform being reported as biphasic in 5 studies,25-28,31 monophasic in another 5,24,29-31,33 and without specification in the remaining reports.21,23,32 Eight studies21,22,24,28,30,31,33 applied stimulation currents applied at a carrier frequency of between 2.5 and 10 kHz with a burst frequency of 30 Hz. The remaining studies used isolated pulse protocol, with frequencies of bursts or pulses applied between 5 and 90 Hz. All studies applied the stimulus with a pulse width of between 0.5 and 2.0 ms.
With respect to current intensity, there was great variability when it came to defining the stimulation thresholds, though most of the studies used high intensities close to the participants’ tolerance threshold. The definitions used to establish intensity were “necessary to induce paraesthesia in lower extremity,” 25 “high intensity with observable muscle response,”24,29 “sensory stimulation,”21,22 “high with minimal discomfort,” 23 “necessary to increase handgrip strength,” 31 and “necessary to induce postural correction.”30,33 Current was, thus, applied with an intensity that ranged from 10 to 250 mA. The stimulation parameters are shown in Supplementary Table 2.
Observed Effects
Motor Response During Transcutaneous Electrical Spinal Cord Stimulation
All studies reported an increase in motor response as measured using EMG activity and other outcome variables, such as movement, force, and function, and in all cases, the reports conducted an intrapatient or intragroup comparison (before/during transcutaneous electrical spinal cord stimulation). In addition, 2 studies compared the stimulation outcomes with those of a control 32 or placebo 33 group (Table 3).
Motor Outcomes Reported by the Selected Studies That Met the Inclusion Criteria for Transcutaneous Spinal Stimulation Following Spinal Cord Injury.

Abbreviations: AIS, ASIA Impairment Scale; ARAT, Action Research Arm Test; BB, biceps brachialis; BF, biceps femoris; BR, brachioradialis; Co1, coccyx 1; COP, Centre of Pressure; ED, extensor digitorum; E-L3, third lumbar vertebra; EMG, electromyography; E-T7, seventh thoracic vertebra; FD, flexor digitorum; GRASSP, Graded Redefined Assessment of Strength, Sensibility, and Prehension; Ham, hamstrings; ISNCSCI, International Standards for Neurological Classification of Spinal Cord Injury; LEMS, lower-extremity muscle score; Obl, external obliques; Q, quadriceps; ROM, range of motion; S, soleus; SCI-FAI, Spinal Cord Injury -functional Ambulation Inventory; SCIM-III, spinal cord independence measure (version III); STD, standing; TA, tibialis anterior; TS, triceps surae; tSCS, transcutaneous spinal cord stimulation; UEMS, Upper Extremity Motor Score; VL, vastus lateralis; VS, Versus; WISCI II, Walking Index Spinal Cord Injury; WT, Walking Test.
Statistical significance (
All studies that recorded EMG activity reported changes in some outcome parameters. Hence, increases in measures were observed in the following: the amplitude of spinal reflexes 31 ; EMG during active movement of the quadriceps (Q), hamstring, triceps surae (TS), tibialis anterior (TA), and extensor digitorum (ED) and flexor digitorum (FD) muscles22-27,29,33; and coactivation of the trunk (erector spinae and external oblique) muscles. 30 This increase was superior to that of placebo in the only study that used this passive comparator intervention as control. 33 When stimulation was applied during gait, the increase in EMG activity was seen to be dependent on the phase of the cycle.23,25,26,29 Furthermore, those studies that studied stimulation at several spinal levels observed a response dependent on the site of application of the current23,33 and a summation effect when the stimulus was simultaneously applied at various spinal levels.23,24 Thus, whereas spinal stimulation at T11-T12 evoked an increase in muscle response in the Q and hamstring, at L1-L2 it evoked an increase in the TS and TA. Finally, one study reported induced muscle activation patterns in the absence of voluntary effort, inducing coordinated movement of the lower extremities similar to that of locomotion, in a gravity-neutral position (lateral decubitus position with support of the lower extremities). 24
Eight studies analyzed motor response by means of assessment of active movement, observing an increase in hip and knee flexion during gait,23-26 an increase in ankle dorsiflexion, 27 and an improvement in the ability to open/close the hand. 31 Furthermore, 3 studies achieved an increase in handgrip strength as measured by dynamometry.28,31,33
With respect to function, 8 studies used functional variables to assess evoked motor responses. Four studies assessed gait and found improvements in quantitative and qualitative aspects, reporting an increase in the length of the complete cycle, 25 a decrease in the time needed to cover 10 m,23,27 an improvement in coordination, 23 and a reduction in the need for external assistance (manual or instrumental).23,26 Similarly, this reduction, along with an improvement in stability and an increase in resistance, was observed by a further 2 studies that assessed control of the trunk in seated and standing positions,30,33 with this outcome being reported by one of the studies as being better than that of placebo. 33
Retained Motor Response After Stimulation
Six studies assessed retained motor response after the electrical stimulation. In terms of the immediate effects of a single session, 2 studies reported an increase in the amount of active movement, an increase in EMG activity from the Q and TA muscles,24,27 and a decrease in the time required to move 10 m. 27 Application of current during repeated sessions led to an increase in the capacity to generate voluntary movement, 24 a decrease in the need for external assistance in standing upright and gait, 33 an increase in manual dexterity and grip strength,28,31,32 and an increase in functional scale scores.21,32 Furthermore, the 2 studies that conducted a medium-term follow-up (3-6 months) after 5 to 6 weeks of stimulation observed a statistically significant increase in handgrip strength, changes in EMG activity and an improvement in functionality (Upper Extremity Motor scores and Action Research Arm Test) with respect to baseline, similar to the immediate postintervention effects.28,32
Following the spinal stimulus intervention, 5 studies carried out a neurological evaluation covering motor and sensory aspects. Accordingly, two-third of the studies24,29 registered an improvement in the American Spinal Injury Association Injury Score, which in 1 case amounted to reclassification of ASIA B to ASIA C. 24 Using the International Standards for Neurological Classification of Spinal Cord Injury, 2 studies28,31 registered an increase in the neurological level: in 1 patient, this improvement amounted to a reclassification from C3 to C4. 28
Other Effects
Three studies27,32 examined the effects of stimulation on the spinal spasticity syndrome. Whereas 1 report showed no difference in the Wartenberg Pendulum Test after a single stimulation session, both reported a decrease in clonus, a reduction in cutaneous plantar reflex, and improved smoothness of the lower extremity during gait. 27 Furthermore, there was evidence of a reduction in spasticity of the upper extremity, as measured on the Ashworth Scale, after termination of the stimulus intervention based on noninvasive spinal stimulation, rehabilitation, and a neuromodulatory drug (buspirone). 32 However, one study 27 reported a modest increase of muscle tone on the Ashworth Scale (<1 point) in the first 24 hours after stimulation and training.
In terms of quality of life, 1 study 28 used questionnaires to evaluate this variable, with increases of 1 point (Spinal Cord Independence Measure [SCIM] II questionnaire) being obtained in relation to self-care and 7 points in relation to physical function (World Health Organization-Quality of Life- Brief [WHO-QoL-BREF] questionnaire). However, no differences were found in the SCIM-III questionnaire scores pertaining to mobility, sphincter control, or environmental and psychosocial aspects. Finally, 4 studies23,27,28,31 used self-reports to record the effects of stimulation: some of the results included improvements in coordination, 23 trunk control, 33 gait, 28 manual function, and autonomic changes (gastric motility, bladder filling, and sexual function). 31
Safety and Adverse Effects
Three studies27,28 assessed the safety of spinal stimulation by systematically recording adverse effects: in general, there was good tolerability of the intervention by patients, without any apparent adverse effects other than cutaneous irritation after repeated stimulation. 33
Discussion
The results of this review indicate that noninvasive spinal stimulation is a viable option for increasing voluntary motor response of the upper and lower limbs, trunk stability, and improvement of function and quality of life of patients with spinal cord injury. Even so, most of the results come from case or case-series studies with a very low level of evidence according to the GRADE scale criteria. Moreover, the lack of clinical trials with appropriate control groups makes it impossible to know whether the motor response obtained with spinal stimulation is better than that of the placebo effect or of other interventions used in the clinic.
Site of Application of Current and Specificity of Motor Response
The results of this review, taken together with findings observed in healthy individuals, appear to indicate that the optimal site for spinal stimulation for enhancing voluntary control of the lower extremities is T11-T12. This spinal level corresponds to the L1-L2 metamere, a level at which propriospinal control of the central pattern generator is partly located.12,13 Although stimulation at this spinal level evokes a multisegmental response, a number of studies on healthy individuals34,35 have observed that the excitation threshold and motor response magnitude differ according to the positioning of the stimulus. Hence, observation shows greater activation of the anterior rectus femoris and vastus lateralis during T10-T11 stimulation and an increased motor response in the hamstring, TS, and TA during T12-L1 stimulation. The optimal stimulation site, thus, differs according to which muscle group is sought to be stimulated. Furthermore, a phenomenon of spatial summation has been reported, with greater motor response being obtained during electrical stimulation at various spinal levels.24,36,37
In the case of the upper extremities, the effects of stimulation at different metameric levels have not been compared. Similarly, it is not known whether application at various spinal levels generates a superior response. However, a study conducted on healthy individuals reported finding multisegmental motor responses during stimulation at the C7 spinous process level,38,39 which would mean that, as occurs in the case of the lower extremities, the electric current activates a propriospinal network organized over several metameric levels. Two studies applied current to the level of the C3-C4 and C6-C7 interspinous spaces simultaneously28,31 and another to the C5 level. 32 Both methodologies were observed to result in an increase in manual dexterity and grip strength and an increase in FD and ED muscle activity. In addition, Gad et al 31 reported a decrease in uncoordinated “aberrant” EMG activity of the biceps brachii and an increase in FD and ED activity during handgrip, using a multisegmental application (C3-C4 + C6-C7). This finding could be a result of the activation of the modulatory propriospinal network simultaneously and rostral to the level of upper limb and hand muscle motoneurons.
Electrical Stimulation Frequency and Waveform
The majority of studies agree that the stimulation frequency required to boost the motor response is approximately 30 Hz. This frequency has been previously used in studies conducted with epidural stimulation.4,5,7 Nonetheless, Bedi and Narkeesh 22 reported a greater EMG muscle response with frequencies of up to 70 to 90 Hz, compared with 30 to 50 Hz. Similarly, Gorodnichev et al 40 reported that in healthy individuals there was a tendency for muscle activity to increase as the stimulation frequency rose from 5 to 40 Hz. Sayenko et al 32 likewise observed great variability among individuals when it came to establishing the optimal frequency for obtaining the greatest muscle response with the least intensity. However, little is known about the effects of stimulation with frequencies higher than 90 Hz. Thus, more studies are needed to analyze the relationship that exists between stimulation frequency, response threshold, and patient tolerance to the spinal stimulus.
In terms of waveform, the studies included in the review used rectangular current, which was monophasic in some cases and biphasic in others. The difference between the 2 reports resides, essentially, in the fact that biphasic current has no electrochemical polarization effect, thereby improving its tolerance and allowing for its application at a higher level of intensity than monophasic current, given that it reduces the risk of burns. In contrast, depolarization of neuronal membranes is evoked by the negative stimulus emitted by the cathode. 41 According to computational models, however, the sharp change in polarity caused by biphasic current might favor neurostimulation.17,42 It, therefore, seems that biphasic current is the ideal waveform, not only to evoke neuromuscular activity with transcutaneous spinal stimulation, but also to improve tolerance to the intervention. Despite these findings, to date, no study has compared the effects of biphasic and monophasic stimulus waveforms in terms of efficacy and safety.
Stimulus Intensity and Intervention Time
The electrical stimulation intensity as measured by current density (mA/cm2) and length of stimulus intervention are also key parameters to evoke an effective neuromuscular effect. Rather than setting the stimulation intensity objectively, most of the studies that met the inclusion criteria for review instead expressed it using subjective and ambiguous terms of perception of the current by participants or observations by researchers. Recently, Serrano et al 43 showed that, when therapists set the intensity of the current on the basis of subjective terms of perception of the stimulus, the defined stimulus intensity is highly variable, which in turn has an impact on the therapeutic effect. Despite the lack of sufficient data to perform a correlational analysis between the stimulus intensity and motor effect, computational models17,42 indicate that the intensity needed to depolarize the dorsal root afferent fibers, at the dorsal root entry zone, should be ≥17.6 V because it is estimated that the amount of current that penetrates this region through the intervertebral tissues is only 9%.
Insofar as the length of intervention time is concerned, although most studies observed an immediate increase in motor response (EMG activity, active movement, or function) when the current was switched on, the studies that assessed postintervention effects nevertheless used application times of 20 minutes or more. Even so, the durability of the effect after a session of spinal stimulation has not been objectively assessed. The only information available regarding postintervention effects is from the perception of motor effects by the participants themselves, who reported a duration of around 6 hours. 25 In addition, studies that conducted a medium-term follow-up (3 months) found that the results in terms of motor response and function, albeit with variabilities between individuals, were superior to patients’ baseline situation and, in some cases, equal to those achieved after termination of the treatment protocol.28,32
Facilitation of Voluntary Motor Drive by Increased Spinal Excitability Following Afferent Stimulation
According to the literature consulted, it seems that to evoke adaptive neuroplasticity following spinal stimulation, the intervention must be combined with activity-based rehabilitation (movement, strength, coordination, assisted gait, transfer training, or manual dexterity and grip strength exercises). 44 Similarly, the patient’s position during stimulation has been observed to modify the threshold and magnitude of the motor response mainly because of better stimulation access of the proprioceptive afferents. 45 Gerasimenko et al 46 also found a reduction in motor threshold during imagination of the movement. Furthermore, severity of the spinal injury seems to be another key factor for achieving results in function. However, even in patients lacking all residual motor activity, solicitation by the investigator for the patient to perform a voluntary movement boosted the motor response evoked by stimulation. 19 It, therefore, seems that electrical stimulation at the spinal level may modulate the functional status of the spinal network below the injury, thereby improving the interaction between the motor drive from the cortex by an increase in spinal excitability mediated by stimulation of sensory afferents. 14 Hence, spinal stimulation is not only able to evoke reflex muscle activity, but can also improve voluntary motor control by patients.
Limitations
The principal limitation of the review is attributable to the methodological quality of the studies analyzed because these are feasibility studies rather than studies of real efficacy. The total study sample is thus small, and it is not known whether the results are superior to the effect of placebo or other already existing treatments. In addition, there may be a publication bias, which implies that studies with positive results are easier to publish than are case studies in which no significant changes are observed.
Conclusions
The results of this review indicate that, at present, no clear statement on the efficacy or effectiveness of noninvasive spinal stimulation can be concluded. Nevertheless, the selected studies support the feasibility of the transcutaneous spinal stimulation technique as a potential strategy to enhance motor function following spinal cord injuries with the absence of adverse effects. Electrode placement and current intensity appear to be the key parameters for achieving a selective response. Other methodological issues that need to be addressed in the future are the small study sample size and the paucity of controlled trials. There is, therefore, a need for the design of randomized controlled clinical trials to study the efficacy of this intervention for motor rehabilitation following spinal cord injury.
Supplemental Material
SupplementaryTable_2._Stimulation_parameters_FINAL – Supplemental material for Transcutaneous Spinal Cord Stimulation and Motor Rehabilitation in Spinal Cord Injury: A Systematic Review
Supplemental material, SupplementaryTable_2._Stimulation_parameters_FINAL for Transcutaneous Spinal Cord Stimulation and Motor Rehabilitation in Spinal Cord Injury: A Systematic Review by Alvaro Megía García, Diego Serrano-Muñoz, Julian Taylor, Juan Avendaño-Coy and Julio Gómez-Soriano in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Table_1._Methodological_quality_FINAL – Supplemental material for Transcutaneous Spinal Cord Stimulation and Motor Rehabilitation in Spinal Cord Injury: A Systematic Review
Supplemental material, Supplementary_Table_1._Methodological_quality_FINAL for Transcutaneous Spinal Cord Stimulation and Motor Rehabilitation in Spinal Cord Injury: A Systematic Review by Alvaro Megía García, Diego Serrano-Muñoz, Julian Taylor, Juan Avendaño-Coy and Julio Gómez-Soriano in Neurorehabilitation and Neural Repair
Footnotes
Appendix
Summary of Findings and Quality of Evidence Assessment.
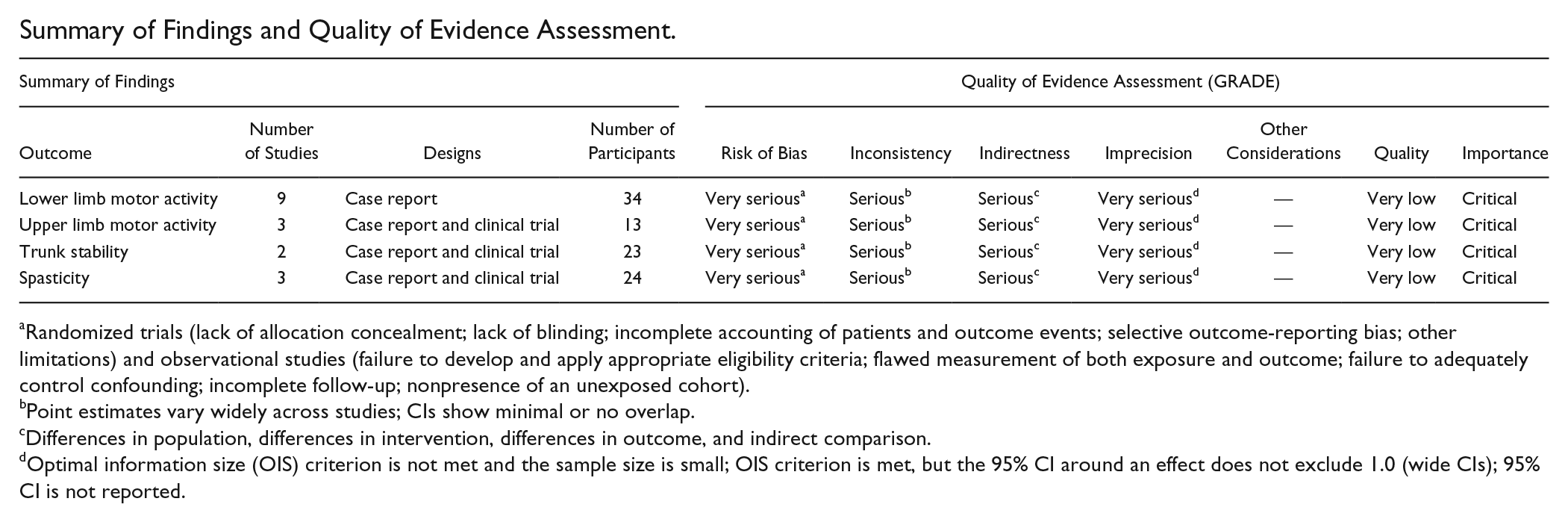
| Summary of Findings |
Quality of Evidence Assessment (GRADE) |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | Number of Studies | Designs | Number of Participants | Risk of Bias | Inconsistency | Indirectness | Imprecision | Other Considerations | Quality | Importance |
| Lower limb motor activity | 9 | Case report | 34 | Very serious a | Serious b | Serious c | Very serious d | — | Very low | Critical |
| Upper limb motor activity | 3 | Case report and clinical trial | 13 | Very serious a | Serious b | Serious c | Very serious d | — | Very low | Critical |
| Trunk stability | 2 | Case report and clinical trial | 23 | Very serious a | Serious b | Serious c | Very serious d | — | Very low | Critical |
| Spasticity | 3 | Case report and clinical trial | 24 | Very serious a | Serious b | Serious c | Very serious d | — | Very low | Critical |
Randomized trials (lack of allocation concealment; lack of blinding; incomplete accounting of patients and outcome events; selective outcome-reporting bias; other limitations) and observational studies (failure to develop and apply appropriate eligibility criteria; flawed measurement of both exposure and outcome; failure to adequately control confounding; incomplete follow-up; nonpresence of an unexposed cohort).
Point estimates vary widely across studies; CIs show minimal or no overlap.
Differences in population, differences in intervention, differences in outcome, and indirect comparison.
Optimal information size (OIS) criterion is not met and the sample size is small; OIS criterion is met, but the 95% CI around an effect does not exclude 1.0 (wide CIs); 95% CI is not reported.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been supported by the “Instituto de Salud Carlos III”, NEUROTRAIN project (PI17/00581), and by “Explora Ciencia/Tecnología 2017” RECODE project (DPI2017-9111-EXP).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
