Abstract
Introduction
Spasticity is defined as abnormal excessive muscle tone resulting from hyperexcitability of the stretch reflexes. 1 It is a major symptom of spinal cord injury (SCI). 1 Spasticity causes many serious problems including pain, contractures, and movement limitations, all of which may create persistent obstacles in the rehabilitation of patients with SCI.2,3 Despite efforts to manage spasticity, clinical trials with antispastic drugs including baclofen and tizanidine have shown limited success because of side effects such as sedation, vertigo, and depression. 4 A poor understanding of the underlying mechanisms of spasticity further complicates its management. Thus, the discovery of novel therapies for spasticity remains of strong interest to clinicians and scientists.
Generally, spasticity is thought to result from an imbalance between excitatory and inhibitory inputs to α-motor neurons that subsequently increase excitability. 5 Although several theories have been proposed to explain spasticity after SCI,6-11 the underlying mechanisms of spasticity remain unclear. Recently, it has been suggested that changes in neuroimmune responses could contribute to excitability of spinal neurons. In particular, activation of microglia after nervous system injury contributes to neuronal excitability by releasing inflammatory mediators, which promote neuronal hypersensitivity in the spinal cord.12-16 Thus, a hyperexcitable network of spinal neurons mediated by activated microglia can be a potential mechanism that underlies spasticity after SCI. Nevertheless, no studies to date have specifically examined whether microglia play a role in spasticity after SCI.
Previous studies have demonstrated the effects of transcutaneous electrical nerve stimulation (TENS) on spasticity in several neurological conditions including SCI. The majority of these studies have suggested that TENS, particularly high-frequency (HF) TENS, can alleviate spasticity.17-22 However, others have showed that TENS is ineffective in reducing spasticity in patients with SCI, 23 stroke, 24 and multiple sclerosis. 25 To address these conflicting results, we aimed to determine whether TENS reduces spasticity in an animal model of SCI.
Several animal models of SCI have been used to examine spasticity.21,26-31 Among these models, a contusive SCI model, which closely mimics the pathology in human SCI, produces particularly uniform injuries. 28 Although spasticity commonly manifests itself after SCI, sustained spasticity has been difficult to reproduce in animal models without complete spinal transection.26,29,30 We previously reported that, following a moderate contusive SCI, spasticity was attenuated with the recovery of motor function. 31 This suggests that severe SCI produces sustained spasticity, which is the requisite condition for experimental study. We have thus chosen a 50-mm contusive SCI induced with the New York University (NYU) impactor to produce an animal model of severe SCI.
In the present study, we examined (
Methods
Experimental Animals
All animal protocols were approved by the Korea University Institutional Animal Care and Use Committee (KUIACUC-2012-207). Adult male Sprague-Dawley rats (200-220 g, n = 108; Orient Bio Inc, Seoul, Korea) were used in this study. Animals were kept under a 12 hour/12 hour light–dark cycle (08:00-20:00) with free access to water and food.
Contusive SCI
Under isoflurane anesthesia (3% isoflurane and 97% O2), contusive SCI was made at the T12 spinal segment using the NYU impactor. In brief, a laminectomy was performed and the spinal column was stabilized by clamping the T10 and T12 spinous processes with tissue forceps. A 10 g weight was dropped onto the exposed spinal cord (directly on the T12 spinal segment) from a 50-mm height. The musculature was then sutured, the skin was auto-clipped, and the animals were allowed to recover from anesthesia. Bladders were manually expressed twice a day until spontaneous urination returned. Unasyn (ampicillin/sulbactam, 100 mg/kg, Pfizer, Seoul, Korea) was administered intraperitoneally twice a day to prevent urinary tract infection.
Intrathecal Catheterization and Drug Delivery
To determine the role of microglia on spasticity, minocycline (Sigma Aldrich, St Louis, MO) in saline (100 µg/5 µL), a microglia inhibitor, was intrathecally (IT) injected 5 weeks after SCI.14,32 A sterilized PE 10 tubing was inserted through an incision of the atlanto-occipital membrane and threaded down to the lumbar segments of the spinal cord, under isoflurane anesthesia (3% isoflurane and 97% O2). In order to test effects of minocycline on spasticity in rat’s hindlimbs, based on a previous report that showed an increase in activated microglia in lumbar segments after thoracic SCI, 14 delivery of minocycline was targeted to the L5-6 spinal segments where spinal neurons are known innervate the triceps surae muscles. As a vehicle control, saline was injected 5 weeks after SCI. The location of the IT catheters was confirmed with an Evans Blue injection at the end of the experiment.
TENS Application
A single 30-minute TENS (Intelect Portable Electrotherapy, Chattanooga Medical Supply Inc, Chattanooga, TN) treatment was performed to both hindlimbs under light isoflurane anesthesia (2% isoflurane and 98% O2) 5 weeks after SCI. Rats were randomly assigned to HF- (100 Hz), LF- (4 Hz), or sham-TENS using a 250 µs pulse width. Two circular electrodes (1 cm in diameter) were attached to a skin region where the rats’ hair had been removed in each hindlimb; 5-mm lateral and inferior to the anterior tubercle of the tibia and the ankle joint in front of the Achilles tendon at the level of the superior border of the medial malleolus. 21 The motor threshold (MT) was then determined by gradually increasing the stimulation intensity of TENS until a slight muscle contraction was observed. The stimulus intensity at which the contraction was first observed was the MT. HF- and LF-TENS were applied at the MT, 90% MT, and 50% MT. In the sham-TENS group, electrodes were attached but no stimulation was applied.
Behavioral Testing
To determine whether the 50-mm contusive SCI model produced adequate and sufficient spasticity, behavioral tests for spasticity and motor function were performed preoperatively and postoperatively. These tests were performed at 1 day prior to SCI surgery and then at 1, 3, 5, 7, 14, 21, 28, 35, 42, and 49 days after SCI. The test for spasticity was also performed before and 20, 30, 40, 50, and 60 minutes after TENS or minocycline treatment at the 5 weeks after SCI. Rats that showed slight spasticity (below grade 2 on the modified Ashworth scale [MAS]) were excluded. All examinations were performed by an experimenter who was blinded to the treatment groups.
After SCI, rats showed an increased resistance to passive movements of the hindlimbs. This was interpreted as spasticity and assessed using the MAS.21,31,33 In brief, the MAS is composed of 6 grades, where grade 0 means no increase in muscle tone and grade 5 indicates limb rigidity either for flexion or for extension. To assess spasticity, the rater covered the eyes of rats and waited until it was relaxed before performing a passive ankle dorsiflexion. 21 All rats were trained for 1 week before the experiments to acclimate them to the environment and study procedures. We measured spasticity in each rat 3 times and then averaged the scores. The average value of both left and right hindlimbs was used as the MAS score. Spasticity was measured before and after TENS or minocycline treatment every 10 minutes over a 60-minute time course.
Motor function was assessed 20 minutes after TENS application to test motor recovery after SCI and to determine whether TENS affected motor function. The Basso, Beattie, and Bresnahan (BBB) locomotor rating scale 34 and a modified combined behavioral score (CBS) 35 were used. The CBS assigns a weighted score to particular tests, which are then combined into one total score. Individual tests comprising the CBS are motor score, toe spread, right reflex, extension withdrawal reflex, and placing reflex.
Immunohistochemistry
Lumbar spinal segments (L5-6) were removed 5 weeks after SCI from the normal (n = 4), SCI (n = 4), SCI + sham-TENS (n = 4), SCI + HF-TENS (n = 4), SCI + LF-TENS (n = 4), and minocycline (n = 3) groups. Rats were deeply anesthetized with sodium pentobarbital (50 mg/kg, IP) and perfused with 4% paraformaldehyde (pH 7.4). Tissue was cryopreserved with 30% sucrose at 4°C overnight. Tissues were sectioned at 14 µm in a cryostat.
Sections were immunostained for OX-42 positive microglia. They were first blocked with avidin and biotin for 15 minutes each and then incubated overnight with the primary antibody: mouse anti-CD11b (1:2000; AbD Serotec, Oxford, UK). Subsequently, sections were incubated with the secondary antibody, biotinylated goat anti-mouse IgG (1:1000, Vector labs, Burlingame, CA), and strep-488 (1:1000, Invitrogen, Carlsbad, CA) for 1 hour each. Control experiments were performed without primary or secondary antibodies to identify the background signal levels.
The stained sections were examined with a Leica DM 2500 (Leica Microsystems GmbH, Wetzlar, Germany) fluorescent microscope, and images were captured with a Leica Camera DFC 450C (Leica Microsystems GmbH). The number of positively labeled cells was counted in the dorsal and ventral regions of the spinal gray matter. Resting microglia have a small cell body with long and thin processes (soma diameter < processes length) whereas activated microglia show marked cellular hypertrophy (soma diameter > processes length). 14 The proportion of activated microglia (number of activated microglia/number of total microglia) was calculated.
Statistics
All values were expressed as the mean ± standard error of the mean (SEM). The percent maximal possible effect (% MPE) was calculated from the MAS using the following formula: 100 × (post-TENS response − baseline)/(maximal response − baseline), where the maximal response was grade 5. The overall antispastic efficacy of TENS was determined for each condition using the area under a curve (AUC) for the time course of % MPE. Changes in spasticity after TENS, minocycline, or saline treatment were analyzed with the Friedman test followed by multiple comparisons. The BBB and CBS data were analyzed with the Kruskal–Wallis test. A one-way ANOVA followed by a post hoc analysis (Tukey’s HSD) was used to analyze the proportion of activated microglia and to compare differences in the AUC for % MPE among the groups. A
Results
Changes in Motor Function and Spasticity in Rats After SCI
We investigated changes in motor function for both hindlimbs in rats with a 50-mm contusive SCI (Figure 1A and B). Prior to the injury, rats showed normal locomotion with a BBB score of 21. However, 1 day after SCI, the rats showed paralysis in both hindlimbs and their BBB scores decreased to 0. Slight joint movements in the hindlimbs were observed beginning at 7 days postoperatively (PO). Locomotion gradually recovered by 35 days PO and there were no further improvements in locomotor function after 35 days PO (4.22 ± 0.37). The CBS for all rats was 0 before the injury but increased after SCI to 68.9 ± 1.4 at 1 day PO and 66.23 ± 1.10 at 7 days PO. Recovery of combined motor function reached a plateau 28 days after SCI (50.83 ± 1.10).
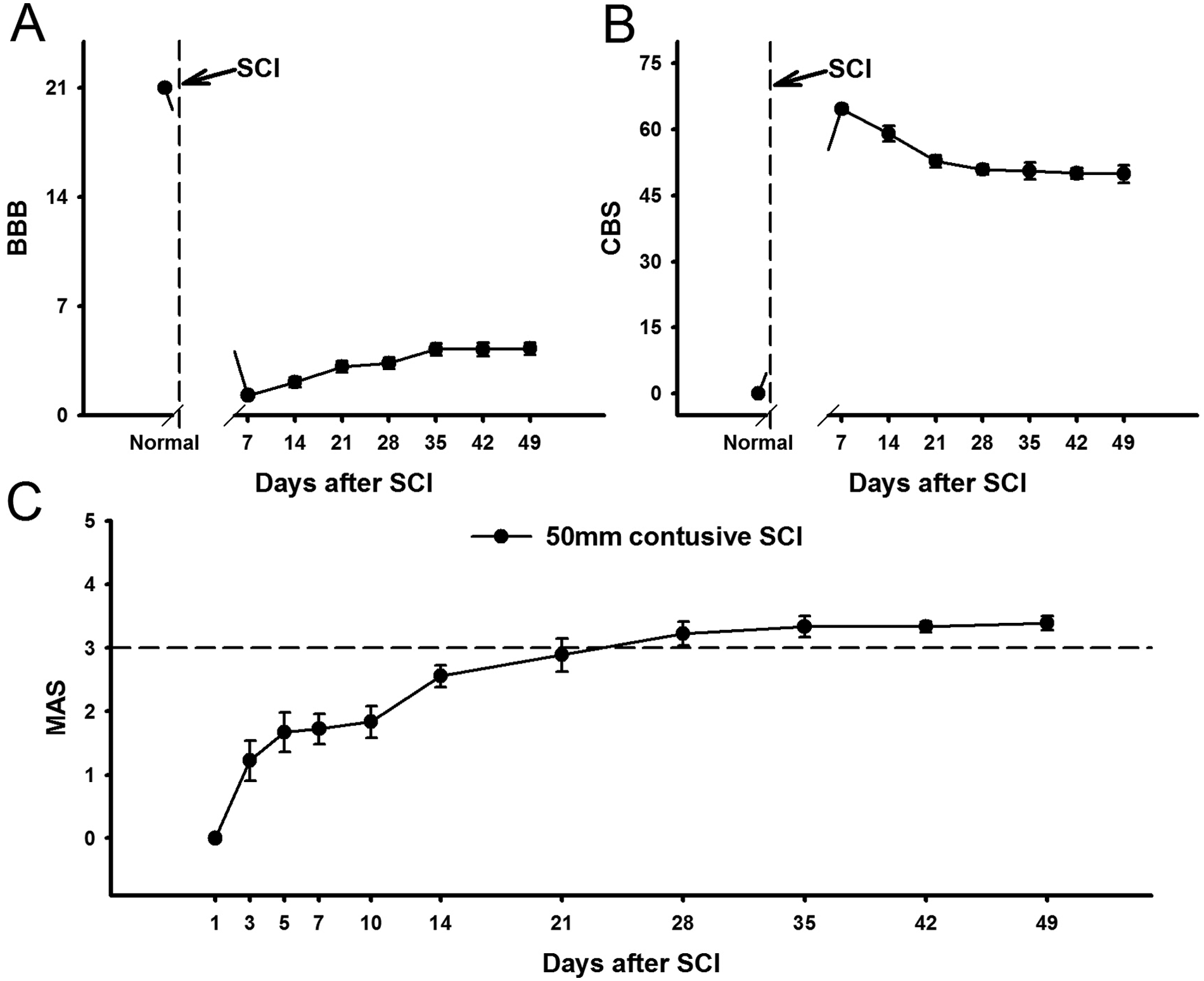
Time course for changes in spasticity and motor function in rats after spinal cord injury (SCI).
Spasticity in the hindlimbs was measured before and after SCI using the MAS, which has a good interrater reliability 33 (Figure 1C). MAS scores began to increase 3 days after SCI, and gradually increased until 28 days PO. Thereafter, spasticity remained well developed and was sustained at grade 3 or higher until 49 days after SCI.
Comparison of Antispastic Effects by LF- or HF-TENS
To test the effects of TENS on spasticity following SCI, MAS scores in the hindlimbs were measured after TENS treatment. Only rats with SCI showing MAS scores above 3 were used in this experiment. To exclude anesthetic effects on spasticity, MAS scores were measured 20 minutes after TENS application. There were no significant differences among the groups in the pretreatment MAS scores before TENS application. HF-TENS at MT and 90% MT significantly reduced spasticity, whereas HF-TENS at 50% MT did not decrease MAS scores (Figure 2A). The antispastic effect peaked 20 minutes after TENS treatment and persisted for 40 minutes in the HF-TENS at 90% MT group and for 50 minutes in the HF-TENS at MT group. Thereafter, it gradually diminished. In the sham-TENS group, there was no reduction in spasticity. We also assessed whether LF-TENS affected spasticity (Figure 2B). LF-TENS at 50% and 90% MT did not appear to change the MAS scores. Only LF-TENS at MT significantly reduced spasticity 20 minutes after application. As shown in Figure 2C, HF-TENS at MT and 90% MT exhibited a prominent antispastic effect compared to LF-TENS at MT and 90% MT. However, there were no significant differences in the AUC for % MPE between HF- and LF-TENS groups at 50% MT.
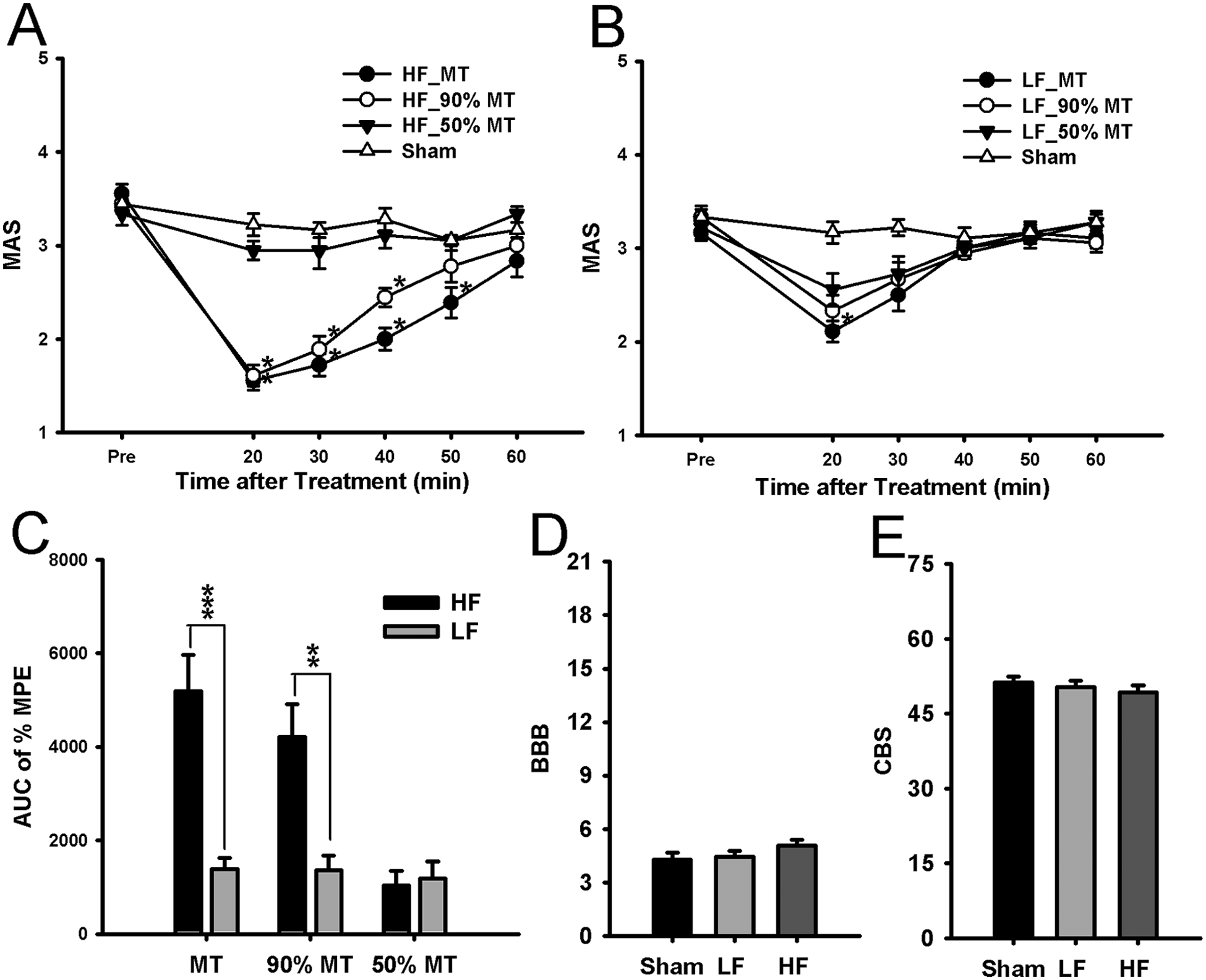
Behavioral analysis of the antispastic effects by high-frequency (HF) or low-frequency (LF) transcutaneous electrical nerve stimulation (TENS) application.
To observe the effects of TENS on motor function, the BBB and CBS were measured 20 minutes after TENS application in the sham, HF-, and LF-TENS groups. There were no significant differences in the BBB scores (Figure 2D) or CBS (Figure 2E) among the 3 groups.
Microglia Expression in the Spinal Cord of Normal and SCI Rats
To determine whether microglia activation is involved in spasticity following SCI, we performed immunohistochemistry to identify resting and activated microglia in the spinal segments that innervate the gastrocnemius and soleus muscles. In normal rats, most resident spinal microglia exhibited the resting phenotype (Figure 3A and A1), whereas spinal microglia with an activated phenotype markedly increased 5 weeks after SCI (Figure 3B and B1). As shown in Figures 3C and D, the resting microglia had small cell bodies with long and thin processes that distinguish them from the activated microglia, which had marked cellular hypertrophy and retracted processes. The microglia expression was examined in both the dorsal and ventral regions of the spinal gray matter. We observed no significant differences between the right and left sides of the spinal cord in any group. A significant increase in the proportion of activated microglia was observed in both the dorsal (0.53 ± 0.01) and ventral (0.58 ± 0.02) gray matter of SCI rats compared to normal rats (dorsal, 0.12 ± 0.01; ventral, 0.13 ± 0.01; Figure 3E).
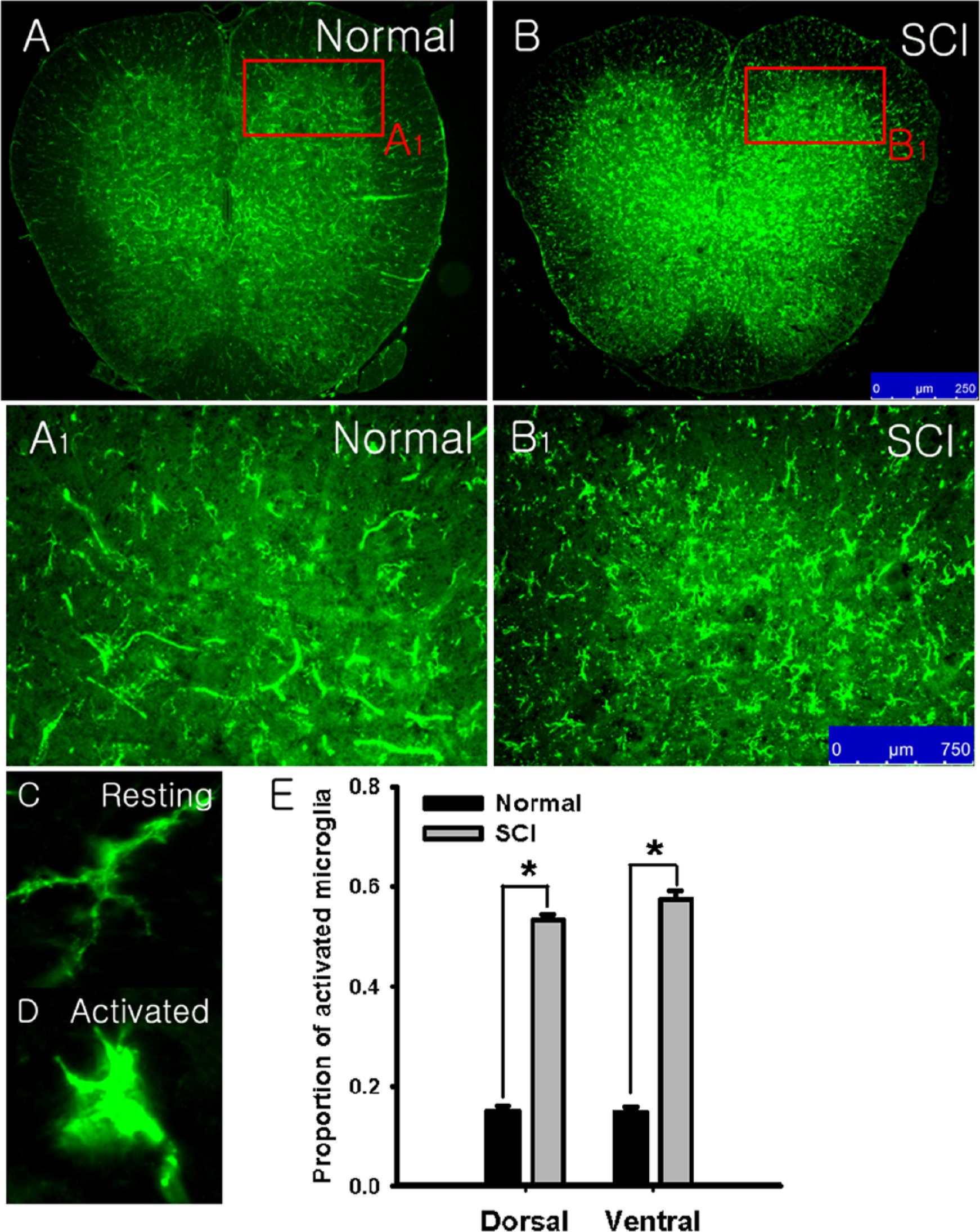
Immunohistochemistry for OX-42 positive microglia in the spinal cord of normal and spinal cord injured (SCI) rats.
Effects of TENS on Microglia Expression
To determine whether the TENS-induced reduction in spasticity following SCI was associated with a change in microglia phenotype, the expression of spinal microglia in response to different TENS treatments was examined. SCI rats with sham-TENS showed an increased expression of activated microglia phenotype with a marked cellular hypertrophic appearance that was similar to that seen in rats with SCI only (Figure 4A and A1). However, in the HF-TENS group (Figure 4C and C1), SCI rats showed a significant shift from the activated to the resting microglia phenotype. LF-TENS produced only a slight change from the activated to the resting microglia phenotype (Figure 4B and B1). As shown in Figure 4D, HF-TENS significantly reduced the proportion of activated microglia to total microglia in both the dorsal (0.28 ± 0.02) and ventral (0.28 ± 0.01) spinal gray matter compared to sham-TENS (dorsal, 0.51 ± 0.01; ventral, 0.57 ± 0.02;
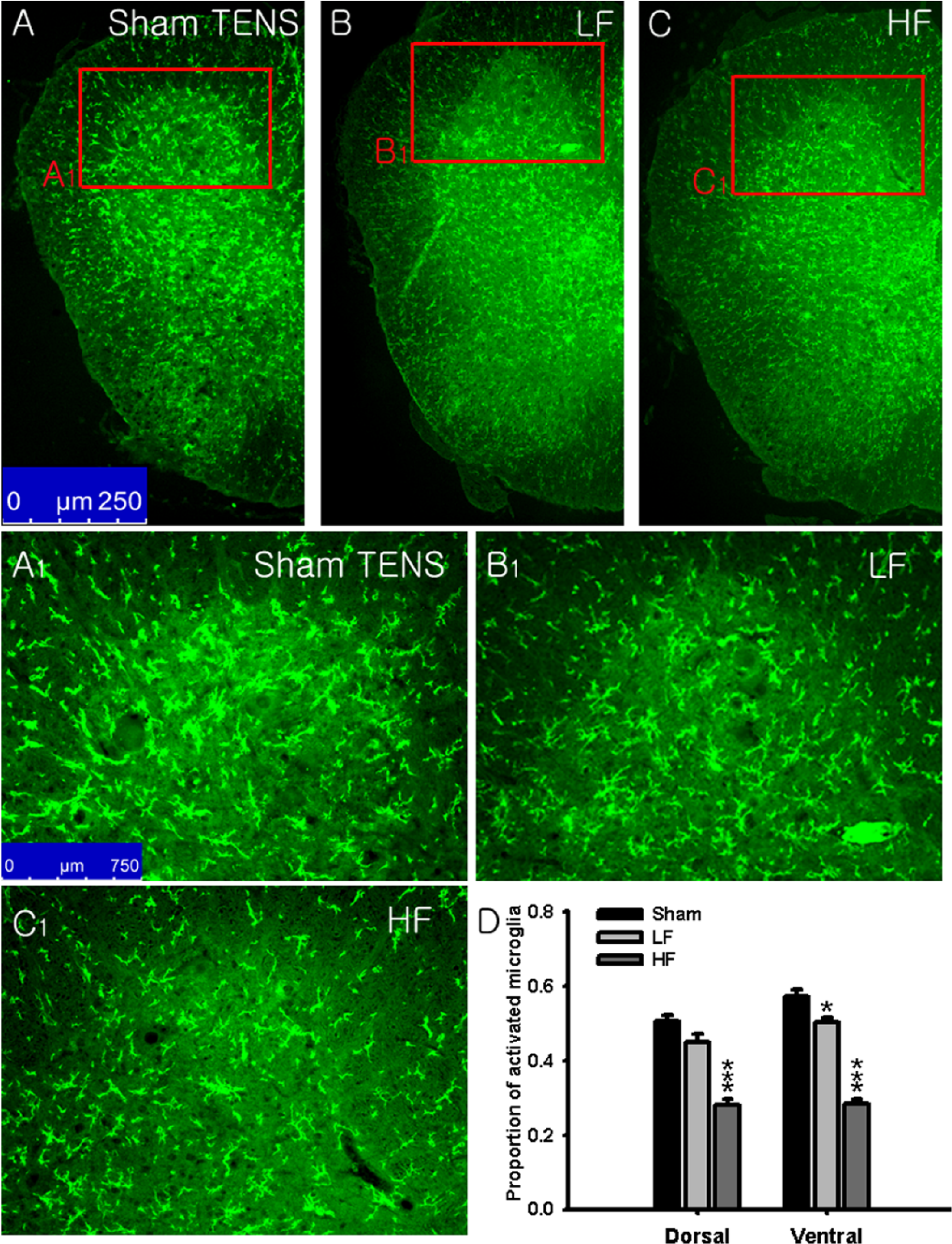
The expression of microglia in the spinal cord of spinal cord injured (SCI) rats after transcutaneous electrical nerve stimulation (TENS) application.
Changes in Spasticity and Microglia Expression by HF-TENS or Minocycline
The effects of minocycline on spasticity and spinal microglia expression were examined to determine whether the TENS-induced reduction in spasticity was related to microglia activation. IT minocycline reduced spasticity for up to 30 minutes after the injection, after which the spasticity returned to the pretreatment levels. HF-TENS also reduced spasticity significantly for up to 40 minutes after application (Figure 5A). Saline had no effect on spasticity. For overall antispastic efficacy (Figure 5B), minocycline (2242.58 ± 271.60) and HF-TENS (3540.72 ± 217.43) significantly increased the AUC of % MPE compared to the saline group (229.17 ± 375.12). The overall antispastic efficacy of HF-TENS was higher than that of minocycline.
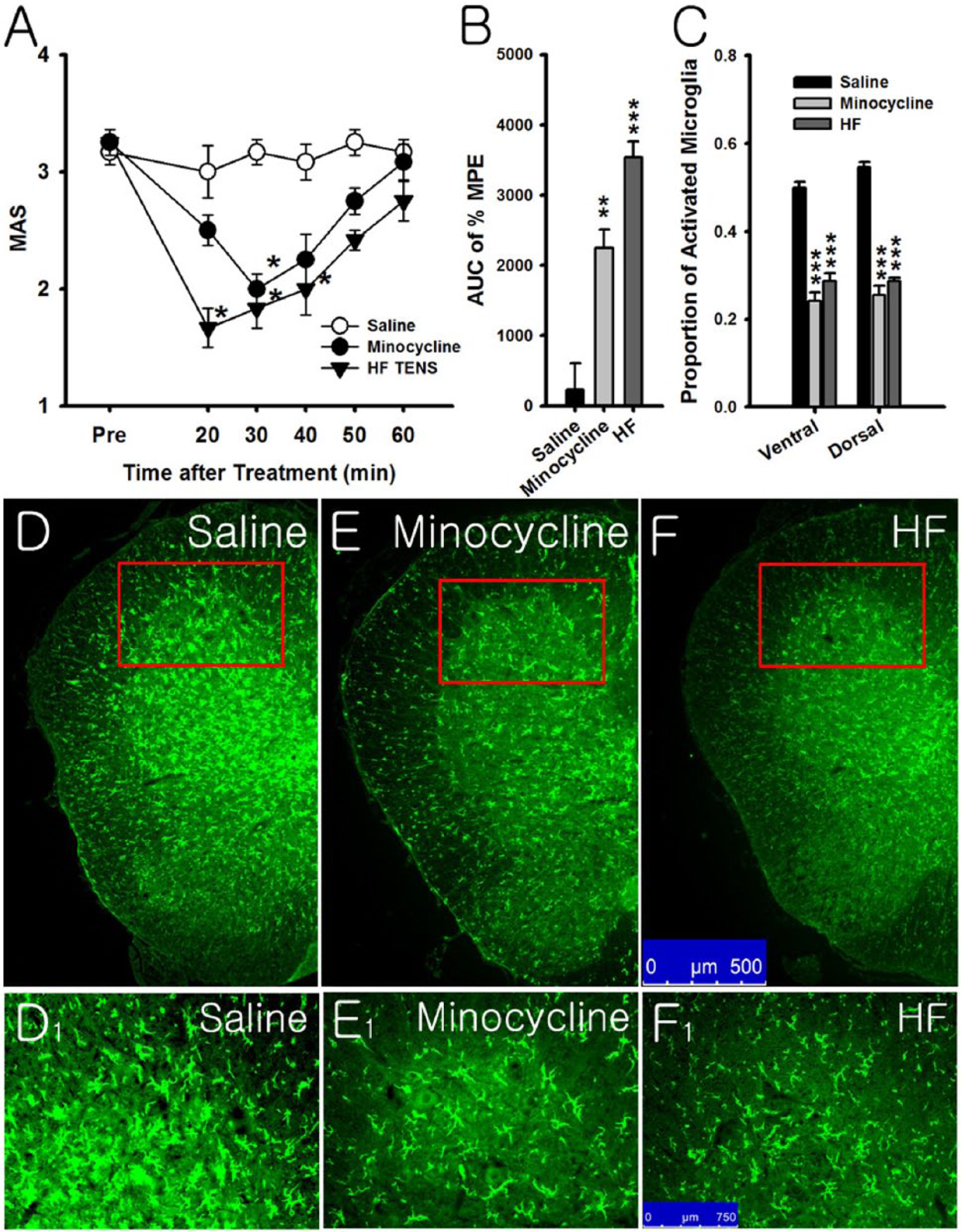
Comparison of the antispastic effects and changes in microglia expression by high-frequency transcutaneous electrical nerve stimulation (HF-TENS) or minocycline.
Immunostaining for microglia showed that both HF-TENS and minocycline significantly reduced the number of activated microglia (Figure 5F, F1, E, and E1), but saline did not reduce the number of activated microglia (Figure 5D and D1). As shown in Figure 5C, both HF-TENS (dorsal, 0.28 ± 0.02; ventral, 0.28 ± 0.01) and minocycline (dorsal, 0.24 ± 0.02; ventral, 0.25 ± 0.02) groups significantly reduced the proportion of activated microglia in both the dorsal and ventral spinal gray matter compared to saline group (dorsal, 0.55 ± 0.01; ventral, 0.50 ± 0.01). There were no significant differences in the proportion of activated microglia between the HF-TENS and minocycline groups.
Discussion
The present study demonstrated that spasticity was well developed and maintained in rats with a 50-mm contusive SCI as well as a significant change in the phenotype of spinal microglia from the resting to the activated type. HF-TENS at 90% MT was able to reduce spasticity and inhibit the activated microglia. Minocycline, a microglia inhibitor, also decreased spasticity in SCI rats. Taken together, these results suggest that activated microglia are associated with the development and maintenance of spasticity following SCI and that HF-TENS at 90% MT can control spasticity by inhibiting activated microglia. Our findings suggest a strong therapeutic potential of HF-TENS to control spasticity after SCI.
Spasticity is characterized by abnormal and excessive muscle tone, which often causes problems in SCI patients’ activities of daily living due to impaired voluntary movements. 36 Much research has been directed to uncover the mechanisms underlying spasticity.6-11 However, the conclusions have remained unclear partly due to the difficulty in obtaining homogenous groups of SCI patients as well as the paucity of research in animal models of spasticity. Attempts to develop experimental animal models for SCI spasticity26,29,30,37,38 have shown limited success in mimicking the characteristics and pathology of spasticity of human SCI. In the spinal transection model, spasticity is sustained following SCI but is accompanied by postsurgical complications and required bowel and bladder expression several times a day.27,39,40 The sacral transection model is limited to spasticity in the tail musculature, which is not representative of spasticity seen in SCI patients.37,38
In the current study, we used a 50-mm contusive SCI animal model produced by the NYU impactor to determine the TENS effects and the role of microglia on spasticity following SCI. To our knowledge, this is the first study that examines spasticity using a severe (50-mm) contusive SCI. The contusive SCI model is considered similar to human clinical SCI cases, 28 and the 50-mm contusive SCI model used in this study is easier to be managed than the spinal transection model. In this study, we observed a change in spasticity up to 49 days after SCI. Spasticity was well developed from 4 weeks after injury with a MAS score above 3 and was maintained for up to 7 weeks after SCI, which was the end-point in this study. These findings suggest that the 50-mm spinal contusive injury model is compatible with spasticity research.
As suggested by previous studies, spasticity can result from hyperexcitability in spinal neurons following central nervous system injury such as a loss or reduction of spinal inhibition,8-11 increased descending facilitation,41,42 and an increase of excitatory as well as a decrease of inhibitory spinal neurotransmission. 43 Recent evidence also suggest that changes of neuroimmune function can contribute to the hyperexcitability of spinal neurons.12-15 Activated microglia release pro-inflammatory cytokines such as interleukin (IL)-1β, IL-6, and tumor necrosis factor-α.12,13,16 These have been implicated in the central sensitization of spinal neurons14,44,45 by enhancing synaptic transmission. In the current study, SCI induced an increase in activated microglia in both the dorsal and ventral spinal gray matter after SCI. In particular, increased activated microglia in L5-6 spinal segments, a remote site from the lesion area, was observed. Our findings are consistent with previous reports that the number of activated microglia increased in the spinal cord caudally as far as the lumbar segments after contusive SCI even though the injury is in the thoracic segments.14,46 Underlying mechanism for the development of spasticity may hence involve microglia activation in caudal site from lesion area such as L5-6 spinal segments, which may therefore increase the excitability of spinal neurons and contribute to the development of hindlimb spasticity. This is strongly supported by our finding in the present study that minocycline applied to the lumbar segments effectively decreased the hindlimb spasticity.
Previous reports have proposed that TENS can be effectively used as a treatment for spasticity.17-25 However, some studies have demonstrated negative results,23-25 and adequate parameters for TENS application to manage spasticity have not yet been established. In this study, we investigated the antispastic effects of HF- and LF-TENS at MT, 90% MT, and 50% MT. Both HF-TENS at MT and 90% MT effectively reduced spasticity, but LF-TENS did not. In addition, the antispastic effects of HF-TENS at MT lasted longer than those for HF-TENS at 90% MT. However, we used HF-TENS at 90% MT in rats with SCI to investigate the TENS effects on motor function (Figure 2D and E) and to compare the effects of TENS with effects of minocycline on the inhibition of activated microglia and spasticity (Figures 4 and 5). The reason for this is that HF-TENS above MT may have potentially harmful effects by decreasing regional blood flow and inducing vascular insufficiency. 47
In the present study, it should be noted that inhibition of activated microglia by TENS can reduce spasticity. In fact, it has been suggested that the chronic neuropathic pain in SCI rat is associated with enhanced microglial activation14,48 and minocycline reduces pain by inhibiting activated microglia.14,32,49 However, the role of activated microglia in spasticity has not yet been studied. If the hyperexcitation of microglia in the spinal cord is one of the factors sustaining spasticity, administration of microglia inhibitor might alleviate it. In the present data, minocycline significantly reduced spasticity after SCI and also changed the microglia phenotype from activated to resting. This result strongly suggests that inhibition of activated microglia can reduce spasticity and may be responsible for the antispastic effects of TENS.
It has been difficult to explain the mechanisms of TENS in reducing spasticity largely due to lack of sufficient experimental data. However, these may be speculated based on studies for the mechanisms of TENS-induced analgesia and inflammatory mediators modulated by microglia. Previous studies have demonstrated that HF-TENS, but not LF-TENS, increases γ-amino-butyric-acid (GABA) and reduces glutamate and aspartate in the cerebrospinal fluid (CSF) in rats with joint inflammation.50,51 Increases in dynorphin-A, a kappa opioid receptor agonist by HF-TENS, has also been observed in the lumbar CSF of patients with neurological disorders. 52 We show that HF-TENS is more effective than LF-TENS for alleviating spasticity and inhibiting activated microglia. Considering that microglia have several receptors including GABA, glutamate and kappa opioid receptors, 53 changes in GABA, glutamate, and dynorphin-A concentrations in the CSF by HF-TENS may inhibit spinal neurons and activated microglia simultaneously. In addition, HF-TENS may affect the inflammatory response via activated microglia thereby contributing to the reduction in spasticity. Decreased glutamate by HF-TENS may reduce tumor necrosis factor-α released by activation of mGluR2 in microglia and increased GABA by HF-TENS may attenuate lipopolysaccharide-induced interleukin release by GABAB in microglia.54,55 Taken together, inhibition of activated microglia and reduction in inflammatory mediators released from microglia by TENS may hence decrease excitability of spinal neurons, which may alleviate spasticity. Thus, these changes by TENS may be the key mechanisms underlying the antispastic effects of TENS.
In the present study, we have demonstrated therapeutic effects of TENS with a single application on spasticity in SCI. Our results also suggest that a single TENS application is not effective for recovery of motor function. However, positive effects of repetitive HF-TENS on reducing spasticity and the recovery of motor function have been reported.21,31 Thus, further studies are needed to demonstrate the effects of repetitive TENS on alleviation of spasticity and recovery of motor function following SCI and establish the adequate parameters of repetitive TENS application.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (MEST), 2012-0003901. It was also partially supported by a grant of the Korea University College of Health Science (K1031192). Suk-Chan Hahm was supported by a scholarship from the BK21+ Program in Embodiment: Health-Society Interaction in the Department of Public Health Science of Korea University Graduate School.
