Abstract
Background. OnabotulinumtoxinA injections improve upper-limb spasticity after stroke, but their effect on arm function remains uncertain. Objective. To determine whether a single treatment with onabotulinumtoxinA injections combined with upper-limb physiotherapy improves grasp release compared with physiotherapy alone after stroke. Methods. A total of 28 patients, at least 1 month poststroke, were randomized to receive either onabotulinumtoxinA or placebo injections to the affected upper limb followed by standardized upper-limb physiotherapy (10 sessions over 4 weeks). The primary outcome was time to release grasp during a functionally relevant standardized task. Secondary outcomes included measures of wrist and finger spasticity and strength using a customized servomotor, clinical assessments of stiffness (modified Ashworth Scale), arm function (Action Research Arm Test [ARAT], Nine Hole Peg Test), arm use (Arm Measure of Activity), Goal Attainment Scale, and quality of life (EQ5D). Results. There was no significant difference between treatment groups in grasp release time 5 weeks post injection (placebo median = 3.0 s, treatment median = 2.0 s; t(24) = 1.20; P = .24; treatment effect = −0.44, 95% CI = −1.19 to 0.31). None of the secondary measures passed significance after correcting for multiple comparisons. Both groups achieved their treatment goals (placebo = 65%; treatment = 71%), and made improvements on the ARAT (placebo +3, treatment +5) and in active wrist extension (placebo +9°, treatment +11°). Conclusions. In this group of stroke patients with mild to moderate spastic hemiparesis, a single treatment with onabotulinumtoxinA did not augment the improvements seen in grasp release time after a standardized upper-limb physiotherapy program.
Keywords
Introduction
Upper-limb disability is problematic for many stroke survivors. Clinical and experimental observation suggest that disordered grasp release is a significant contributory factor. Although difficulties with grasp release have been shown to be primarily a result of extensor weakness and impaired motor control,1,2 there is some evidence that persistent flexor activation may also contribute. 2 In the clinical setting, rehabilitation specialists increasingly postulate that temporarily reducing finger flexor overactivation could enhance interventions to improve hand function after stroke.
Sustained or intermittent involuntary muscle activation after stroke is a manifestation of spasticity, as recently defined by Pandyan et al. 3 Spasticity is common after stroke,4,5 and its treatment is increasingly integrated in stroke rehabilitation and care. Systematic review provides strong evidence that botulinum neurotoxin type A (onabotulinumtoxinA) reduces spasticity and improves passive function—for example, easing opening of the hand for cleaning and skin care. 6 Observational studies without controls or blinding have shown some promising results in improving active function of the upper limb.7-9 However, effects have been variable, and significant changes are yet to be shown in placebo randomized controlled trials.10-12 According to previous reviews, the lack of effect could reflect study limitations, and recommendations were made that future research should measure change in specific active functional tasks or outcomes and that an objective quantification of spasticity should be used.12-15
PROMBIS (Predicting Outcomes and Measuring benefit of Botulinum therapy in Stroke) is an exploratory randomized, double-blind, placebo-controlled, parallel-group trial to investigate the effect of onabotulinumtoxinA on one such specific outcome: grasp-release time. We hypothesized that the active treatment group would demonstrate a statistically significant improvement in grasp release time compared to the control group.
Primary Objective
To determine whether targeted onabotulinumtoxinA injections (BOTOX, Allergan Limited, Marlow, UK) combined with standardized physiotherapy treatment of the upper limb after stroke will reduce grasp release time, a quantitative measure of active upper-limb function.
Methods
Study Design
This was a parallel-group, double-blind, randomized controlled trial (RCT) with participants randomly allocated in a 1:1 ratio between treatment and placebo groups. Participants performed baseline assessment and were then given injections of either placebo or onabotulinumtoxinA in week 0, followed by 10 sessions of intensive standardized physiotherapy over 4 weeks. 16 Outcomes were then reassessed at weeks 5, 9, and 13.
Standard Protocol Approvals, Registrations, and Patient Consent
The trial was undertaken at The National Hospital for Neurology and Neurosurgery from 2009 to 2014 and the protocol and all amendments approved by the local review board. Written informed consent was obtained from all participants at the screening assessment. The study is registered on the EU Clinical Trial Register (EudraCT: 2009-009357-22) and is reported here in accordance with the CONSORT guidelines for reporting of RCTs. 17
Participants
Patients presenting to focal spasticity clinics at the National Hospital for Neurology and Neurosurgery were screened for eligibility by the multidisciplinary team, including members of the independent research team. Inclusion criteria were the following: (1) confirmed diagnosis of stroke more than 1 month previously; (2) established focal finger or wrist spasticity that the multidisciplinary team felt could be interfering with active grasp and release function and had the potential to benefit from treatment with onabotulinumtoxinA (this included an assessment on whether the potential participant presented with sufficient residual strength and motor control for rehabilitation to be effective); (3) score of 2 or more in the modified Ashworth Scale (MAS) in the joints of interest; and (4) ability to transport the assessment cup to at least 1 of the target positions and release it at baseline. Exclusion criteria were the following: onabotulinumtoxinA injections to any site within the previous 3 months; contraindications to onabotulinumtoxinA; fixed contracture in the upper limb; additional neurological impairment not related to stroke; uncontrolled upper-limb pain; cognitive impairment preventing informed consent or the ability to follow task instructions.
Participants were not required to be naïve to onabotulinumtoxinA treatment.
Randomization and Blinding
Randomization was performed by an additional statistician prior to trial commencement using a block randomization process to ensure equal numbers in each treatment arm (the trial statistician was blind to group allocation). Details of the blinding procedures, including allocation concealment are provided in Appendix e-1 (supplementary material).
Intervention
Drug Intervention
Injection sites were identified using standard neurophysiological technique (electromyography [EMG] and electrical stimulation) using a portable handheld device (Clavis; Medtronic, Minneapolis, MN). 18 The doses and distribution of the injections were guided by the clinical and neurophysiological evaluation (including the magnitude of the audible stretch response and degree of resting muscle overactivity) per standard clinical practice.
Allergan Botox, diluted as 100 units in 2 mL of saline, or a saline placebo was injected through a fine-bore EMG needle electrode into the muscles identified by the multidisciplinary assessment as likely to be hindering function. Treatment and placebo solutions looked identical and were reconstituted out of sight of the injecting doctor, treating physiotherapist, and the participant.
Physiotherapy Intervention
The standardized physiotherapy intervention has previously been described in detail. 16 The original protocol consisted of daily sessions over 10 consecutive working days. For this study, it was modified to occur over 4 weeks to focus training during the peak action of the drug and reflect current clinical practice of outpatient therapy provision. The total session time ranged from 45 minutes up to 1.5 hours to accommodate each patient’s need to complete the tasks, rest, and stretch without affecting the overall intensity (repetitions) of the therapy.
In summary, the protocol included both strength training (3 different muscle groups) and functional task practice (3 different tasks). Strength training consisted of 3 sets of 10 repetitions of wrist extension, finger extension, and grip strength at 60% to 80% of maximal isometric voluntary contraction measured in midrange and was recalibrated every 3 training days.
Functional training tasks were chosen by the participant relevant to their personal treatment goals. The intervention was tailored to the individual’s impairment level, so that the intensity of intervention was standardized despite differing impairment levels at enrolment.
Participants were encouraged to stretch whenever needed throughout the strength and functional training.
Primary Outcome Variable
We used a functionally relevant grasp-release task that measured the time taken to release grasp after moving an instrumented cup to a target. Grasp release timing data were calculated from first contact of the cup on the target until all digits were released from the cup. All electronic data were collected in a custom software program (LabVIEW, National Instruments Corporation, Austin, TX), then exported and further analyzed with MATLAB scripts (The MathWorks Inc, MA).
The targets were spread over 4 positions (2 in near space at 50% of arm length and 2 in far space at 90% of arm length) and 2 heights (low, at 2 cm above the table surface, and high, which was individually adjusted, so that the participant required 90° of shoulder flexion to place the ulnar border of their hand on the target surface). Test-retest variability varied from excellent (in the near and low target, Intraclass Correlation Coefficient (ICC) = 0.8) to moderate (in the low far target, ICC = 0.54). 19 The participants could have assistance to place the cup into the hand but were required to be able to transport the cup to at least 1 target and release it unassisted. The cup weighed 300 g and measured 124 mm in height, with a base diameter of 58 mm and top diameter of 88 mm. For more details see Appendix e-1 (supplementary material).
Secondary Outcome Variables
Reliable measurement of spasticity remains controversial.3,20 Spasticity is defined as “disordered sensori-motor control, resulting from an upper motor neurone lesion, presenting as intermittent or sustained involuntary activation of muscles” (p. 5). 3 We used the MAS 21 because it has been widely used in previous studies and would allow comparisons. The MAS was converted to a 6-point score from 0 to 5. However, because the MAS assesses overall passive stiffness 22 and has been shown to be a subjective measure,21,22 a servomotor (Kollmorgen D062M Direct Drive) was used to measure spasticity objectively. The motor was programmed to measure torque in response to slow and fast passive movements. In healthy controls, there is no significant difference between these measurements. 19 In people with upper motor neuron lesions such as stroke, involuntary activation of the flexor muscles in response to the fast stretch of the motor (spasticity) can be objectively quantified as an increase in torque. The technique provides quantification of stretch reflex–mediated activity, which is proportional to and correlates with the size of the EMG response in people with Upper Motor Neuron Syndrome. 23
The servomotor held the participants’ arm firmly in position with the axis of either the metacarpophalangeal joints or the wrist joint aligned to the axis of the motor. Wrist stretches started at 10° of flexion moving through 20° to 10° extension. Finger stretches started at 45° flexion, moving back 20°. The fingers were measured together, because the apparatus was not capable of measuring individual fingers. Participants were instructed to relax and let the motor move their arm without resistance. Passive stiffness of the flexor muscles was measured by recording mean torque over 5 slow stretches (20° at 6°/s) while EMG recordings confirmed that the muscle was not activated. There was no reflex activity at this slow speed, and therefore, the measure represents soft-tissue stiffness without the reflex (spasticity) component. Total stiffness, which includes passive and reflex components, was measured by recording mean torque over 5 fast stretches (20° at 300°/s) while EMG recordings identified and quantified the presence and duration of reflex response (spasticity). For more details of the methodology see Appendix e-1 (supplementary material).
Isometric wrist and finger flexion and extension strengths were measured against the stationary servomotor with joints at 0°. Grip strength was measured using a dynamometer. Wrist and finger range of movement was measured using a standard goniometer and positioning.
Functional tests comprised the Nine Hole Peg Test 24 and the ARAT. 25 The Arm Measure of Activity (ArMA) 26 and EQ5D 27 quality-of-life measure were completed. The ArMA is a self-reported functional scale with passive and active subscales that use a Likert scoring system between 0 (no difficulty) and 4 (unable to do task).
Finally, 2 personalized functional goals, including a grasp release goal, were also agreed with each participant.
Statistical Analysis
Primary outcome analysis was performed using analysis of covariance (ANCOVA). Week 5 measurements were used as the outcome measure, with baseline score as the covariate, and a complete case analysis performed. All participants completed baseline and immediate outcome measurement sessions. Data required log transformation to achieve normal distribution prior to analysis. Missing values, where participants were unable to complete the grasp release task at all target positions, were imputed per trial protocol using the maximum grasp release time recorded in the data set at that given time point. The baseline adjusted mean difference between groups was reported along with the corresponding 95% CI.
Secondary analyses analyzed combined data from all time periods. Generalized estimating equation models were fitted to account for clustering of patients over time. Baseline values were included as one of the covariates in the model.
Assumptions were checked, and where they were not met, data transformation was performed prior to analysis. Where data transformation was not possible, a nonparametric test was used.
Results
Participants
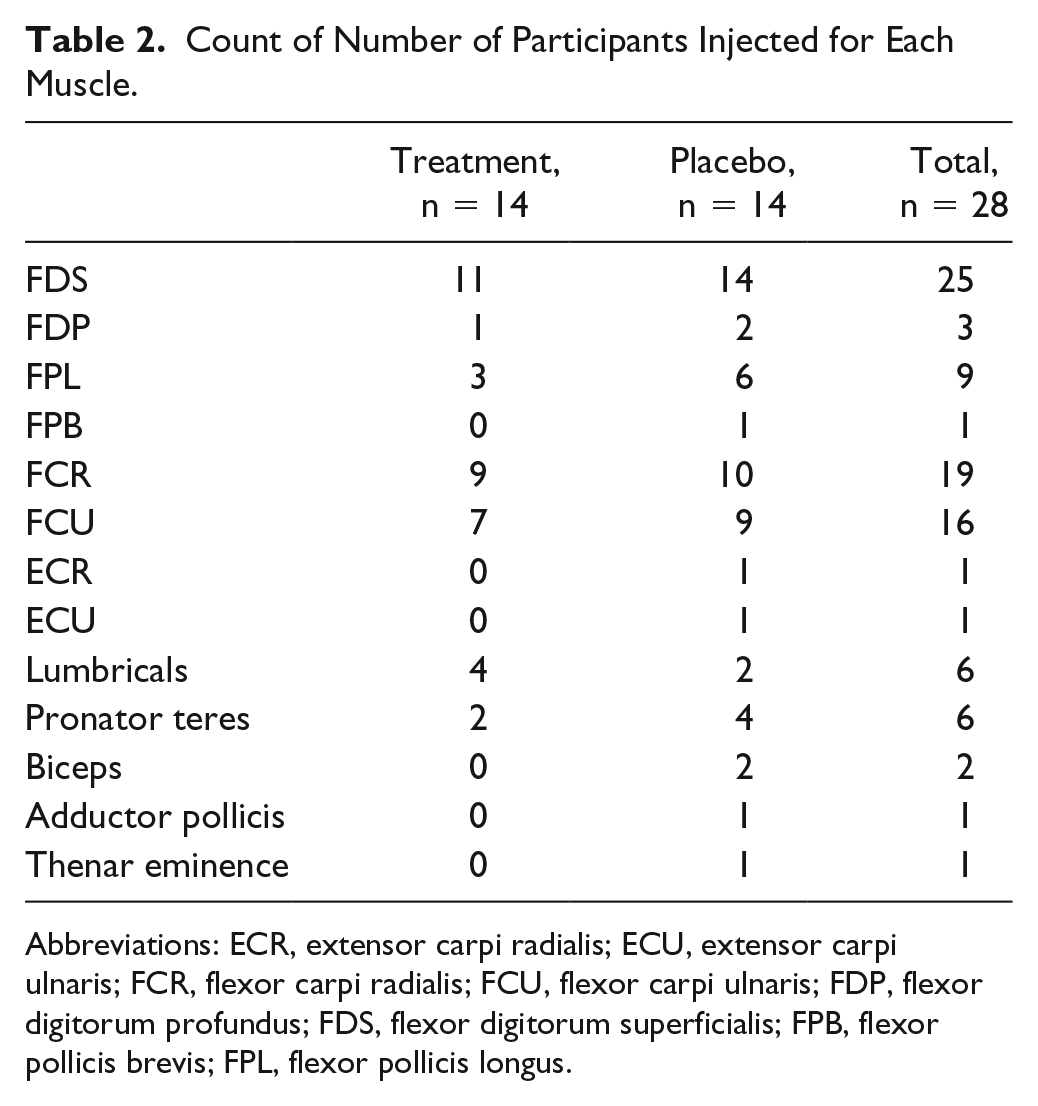
A total of 28 participants were recruited in total and randomized, 14 to each arm of the trial. The flow of participants is shown in Figure 1 (CONSORT diagram). Baseline characteristics are presented in Table 1. There were no significant differences between the groups at baseline. Most participants had experience of onabotulinumtoxinA treatment (not within the preceding 3 months), but none had previously received an intensive upper-limb–specific intervention. The treatment group received a total of 115 units (range 40-190), and a median of 5 muscles were injected per participant (range 1-8). The placebo group received 151 units (range 55-290) with a median of 7 muscles injected per participant (range 4-12). Table 2 gives a summary of the number of participants injected broken down by muscle. Please see e-Table 1 in the supplementary information for a more detailed breakdown of dose and muscles injected per participant.
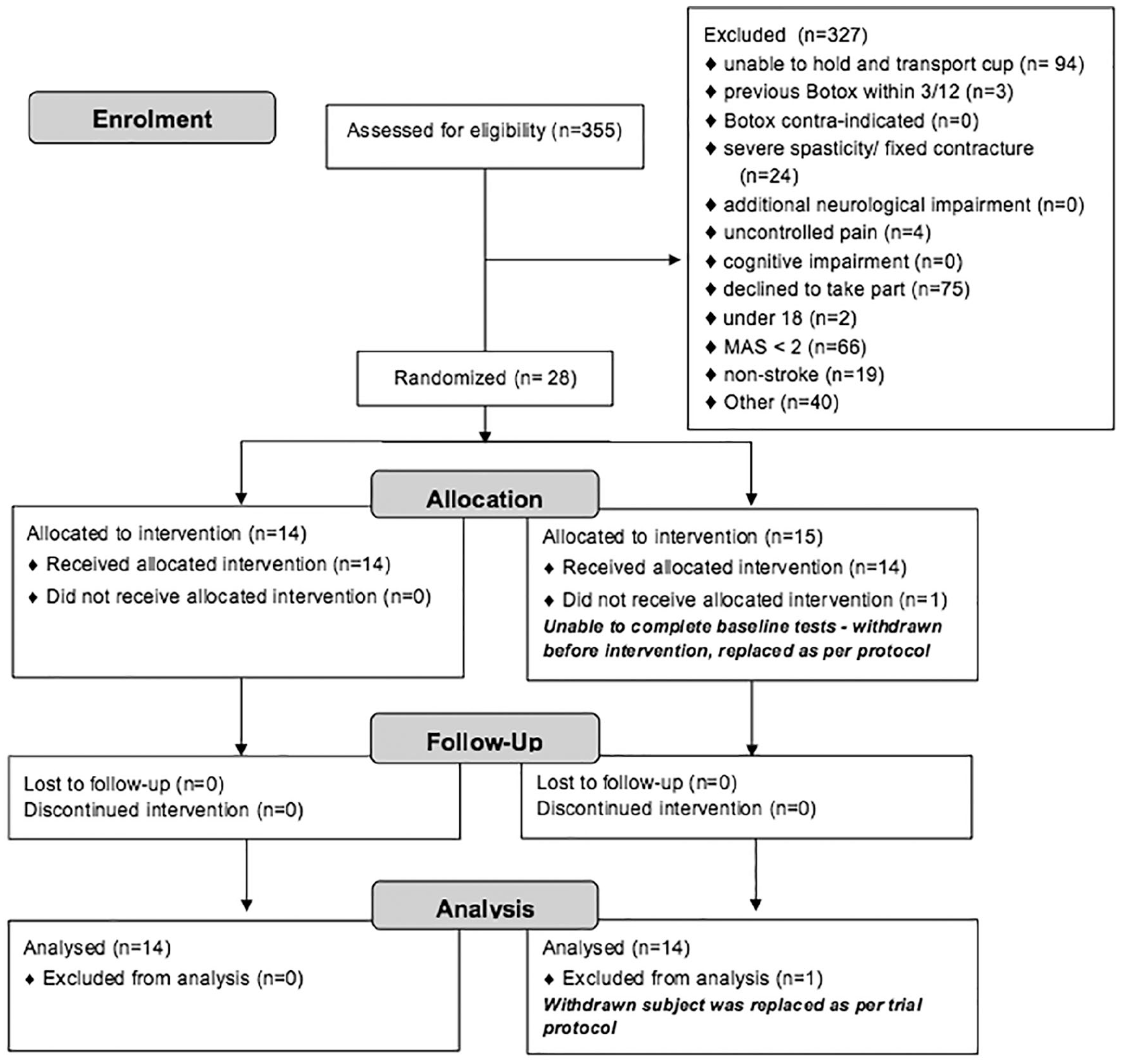
Consort diagram.
Baseline Patient Demographics.
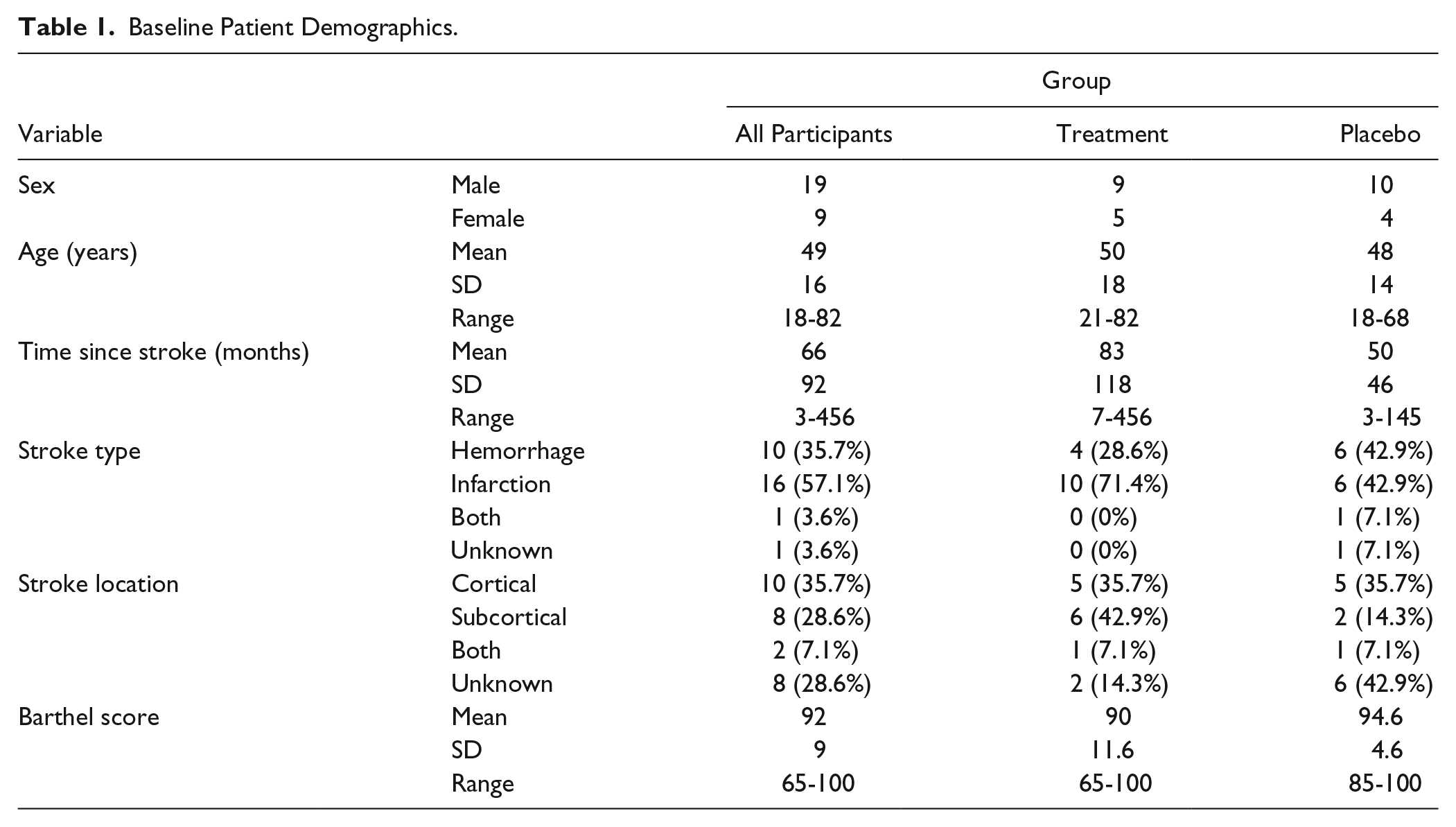
Count of Number of Participants Injected for Each Muscle.
Abbreviations: ECR, extensor carpi radialis; ECU, extensor carpi ulnaris; FCR, flexor carpi radialis; FCU, flexor carpi ulnaris; FDP, flexor digitorum profundus; FDS, flexor digitorum superficialis; FPB, flexor pollicis brevis; FPL, flexor pollicis longus.
Safety
There were 2 unrelated serious adverse events during the trial. One occurred after consent but prior to administration of the trial drug (ocular migraine, treated initially as a possible ocular transient ischemic attack), and the other occurred 4 months after injection (kidney vasculitis). Neither participant was withdrawn. The emergency unblinding procedure was not used. Minor adverse reactions were reported as expected (detail in supplementary material Table e-2).
Primary Outcome
There was no statistically significant difference in grasp release time in all 4 positions between treatment groups at follow-up at week 5 (placebo median = 3.0 s, treatment median = 2.0 s; t(24) = 1.20, P = .24). Treatment effect was −0.44, and the 95% CI was −1.19 to 0.31. Within-group median change for placebo was −606 ms and treatment, −1200 ms. This finding remained true even when the low-near position (easiest target to reach) was considered in isolation (placebo median = 4.0 s, treatment median = 2.0 s; t(24) = 0.65, P = .52; treatment effect = −0.30, CI = −1.27 to 0.67). Within-group median change for placebo was −423 ms and treatment, −2364 ms. See Table 3 and Figure 2.
Treatment Effects on Primary Outcomes. a
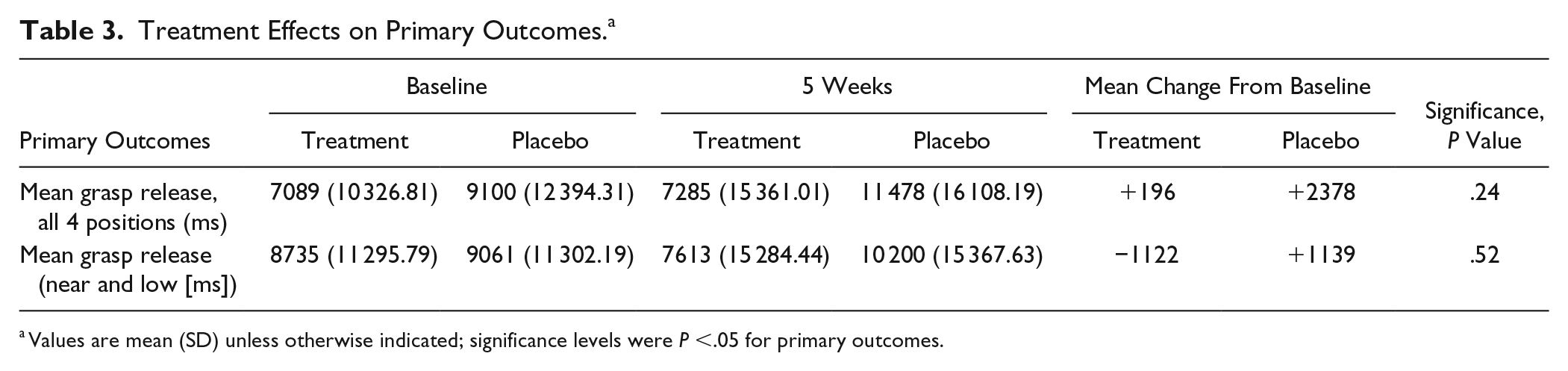
Values are mean (SD) unless otherwise indicated; significance levels were P <.05 for primary outcomes.
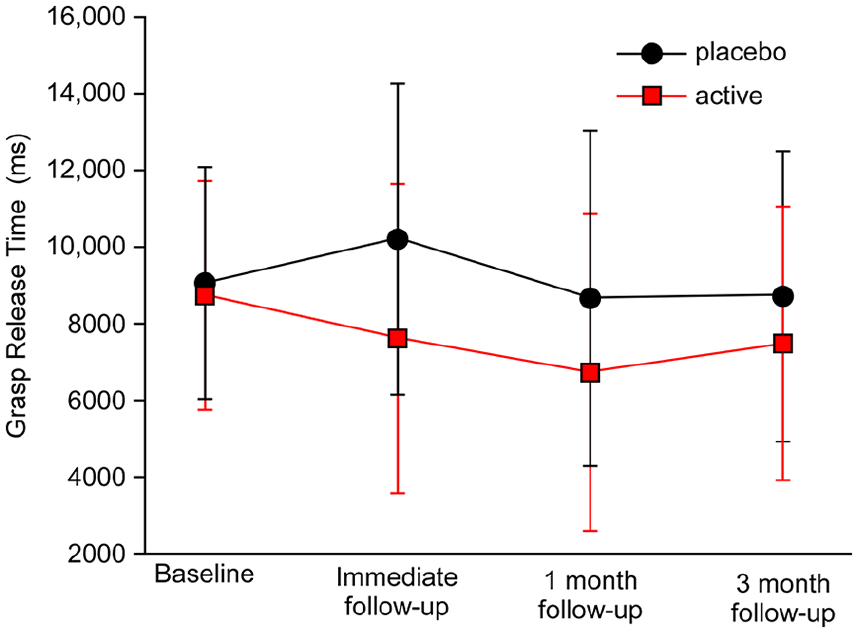
Time taken to release grasp (mean ± standard error of the mean).
Secondary Outcomes
There was no significant difference between groups on any of the secondary outcomes after correction for multiple comparisons (see Table 4).
Treatment Effects on Secondary Outcomes. a
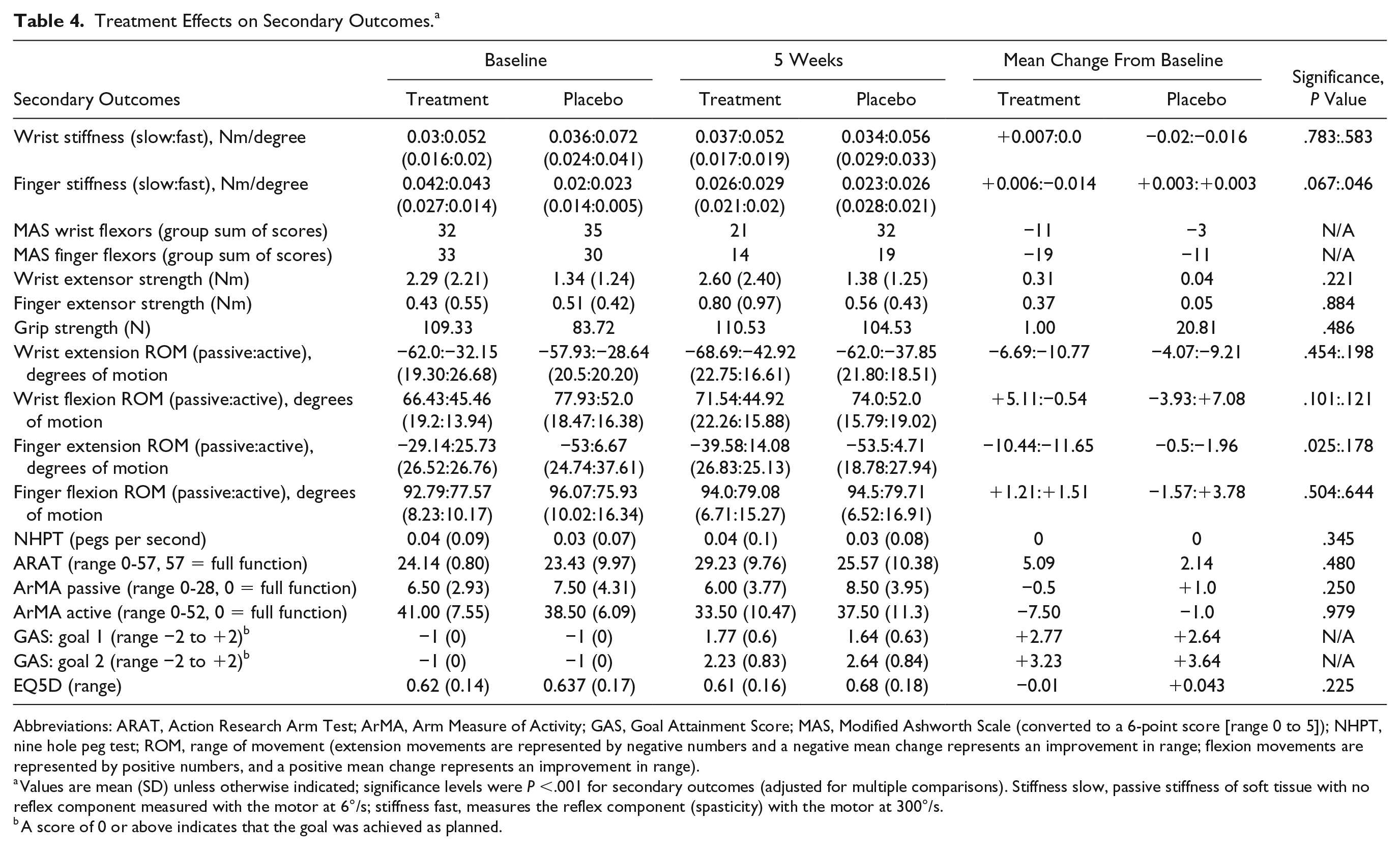
Abbreviations: ARAT, Action Research Arm Test; ArMA, Arm Measure of Activity; GAS, Goal Attainment Score; MAS, Modified Ashworth Scale (converted to a 6-point score [range 0 to 5]); NHPT, nine hole peg test; ROM, range of movement (extension movements are represented by negative numbers and a negative mean change represents an improvement in range; flexion movements are represented by positive numbers, and a positive mean change represents an improvement in range).
Values are mean (SD) unless otherwise indicated; significance levels were P <.001 for secondary outcomes (adjusted for multiple comparisons). Stiffness slow, passive stiffness of soft tissue with no reflex component measured with the motor at 6°/s; stiffness fast, measures the reflex component (spasticity) with the motor at 300°/s.
A score of 0 or above indicates that the goal was achieved as planned.
The MAS group sum of scores decreased a total of 3 and 11 levels, respectively, for wrist and finger flexors in the placebo group, versus 11 and 19 in the active treatment group. Physiological measures of stiffness (slow motor) were not significantly different between groups in the wrist or fingers (for detailed values see Table 4); there was similarly no group difference on change in spasticity measures (fast motor) for either of the joints tested.
Wrist and finger extension strength improved with no significant difference between groups (placebo, +0.04 Nm and +0.05 Nm, and treatment, +0.31 Nm and +0.37 Nm, respectively). Extrinsic grip strength was maintained in the treatment group at 5 weeks despite the weakening effect of onabotulinumtoxinA injections (see Table 4).
Active wrist extension range of movement improved in both groups (placebo, +9°; treatment, +11°) as did active finger extension range of movement (placebo, +1.96°; treatment, +11.65°). Both groups also achieved treatment goals (placebo, 65%; active, 71%) and improved on the ARAT (placebo, +3; treatment, +5) and the ArMA active subsection (−1.0 in the placebo group versus -7.5 in the active treatment group). There was no change in either group for the 9HPT or the EQ5D.
Discussion
In this randomized placebo-controlled proof-of-concept trial, clinically and physiologically measured hypertonia decreased and function improved regardless of whether participants received active treatment with onabotulinumtoxinA or placebo. There was no statistically significant difference between the onabotulinumtoxinA and placebo groups on any measure of impairment, functional outcome, or goal attainment. Although all measures were in favor of the onabotulinumtoxinA group, this was not statistically significant.
Although previous botulinum toxin studies have shown a significant reduction in spasticity measures,12,13 thus far, translation to improvement in active function has not been clearly shown.12,13,28 Despite that, clinicians often propose onabotulinumtoxinA injections to temporarily weaken overactive flexors and give a “window of opportunity” for active physiotherapy to strengthen extensor muscles and improve hand function. This treatment approach requires residual extensor strength and some level of voluntary control over the extensors in order to strengthen and train grasp release function. In this trial, we wanted to test this physiologically plausible principle using a scientifically robust but also pragmatic design. We opted for a relevant quantitative variable (grasp release time) as our primary outcome and we included physiological measures of spasticity, as recently recommended for new trials in this area.14,15 Moreover, this is the first study to use a standardized physiotherapy program. Our results did not support our hypothesis; in other words, a single treatment with onabotulinumtoxinA group targeting the wrist and finger flexors does not seem to offer additional improvement in grasp release after 4 weeks of a targeted physiotherapy intervention when compared with a placebo group.
We acknowledge that the current study with its relatively small sample size does not provide a definitive answer on the usefulness of onabotulinumtoxinA as an adjunct to improve active function in stroke patients with upper-limb spasticity. A recent review has also shown no effect on active function at immediate outcome. 28 However, the authors acknowledge several limitations, including the fact that adjuvant treatment may help optimize voluntary control, which was not controlled for in the included studies (16 of 35 studies had no adjuvant treatment, and quality and standardization of adjuvant therapy was unclear in the others). They also acknowledge that there was insufficient evidence for effects at a functional level and that the diversity of clinical measures used may have contributed to this. Our study has attempted to address some of these issues with standardized adjuvant therapy and the attempt to target and accurately measure 1 important functional activity (grasp release) that might be expected to benefit.
A few other points merit discussion. In contrast to previous studies,12,13 clinical spasticity measures (MAS) did not differ between the groups. The same was true for the servomotor measures that are more likely to measure spasticity rather than overall stiffness. This lack of difference may, in part, be related to the adjuvant standardized physiotherapy regime used in this study because more than half of the participants in Gracies et al 12 were not receiving any therapy; in others, patients received unspecified and nonstandardized interventions, introducing a potential confounder into that data.13,27 It is also possible that the population investigated here, who were required to be able to complete a grasp release task for inclusion, represent a less severely impaired sample than those in the Gracies et al study. Patients with more severe spasticity—that is, unable to extend their fingers enough at baseline—may have shown better spasticity outcomes after the injections, but whether that would have translated to functional gains is debatable. As shown for other interventions, 29 response is more likely when patients have baseline voluntary activity, particularly active finger extension. Indeed, both groups did improve in strength and arm function measures, arguably because of the physiotherapy intervention. Whether this represents a ceiling effect or simply highlights that strength and motor control are stronger modifiers of outcome than spasticity in this group of patients cannot be answered from our current study. A different study design that includes patients with variable impairments as well as a considerably longer intervention could address that in the future.
Another potential problem is the fact that patients were invited to enter if they were at least 1 month poststroke, with no upper limit; this was again a pragmatic representation of the population referred to spasticity services. The vast majority of the patients were at least 6 months poststroke at enrollment (only 1 patient in the placebo group was enrolled at 3 months), which is the accepted cutoff point for chronic stroke, and thus, heterogeneity associated with plasticity-related recovery should be minimal.
In this study, we have implemented a single treatment with onabotulinumtoxinA and 4 weeks of standardized therapy, and all our patients improved. It is possible that 2 or more cycles of onabotulinumtoxinA treatment could be more effective, as some recent observational studies have suggested,30,31 by consolidating the effects on spasticity and/or allowing more opportunity for retraining. The history of previous onabotulinumtoxinA injections was not addressed in this study but may also have had potential to influence the outcome.
The intensity of physiotherapy is another important factor. We have opted for a pragmatic physiotherapy intervention, but longer and more intensive therapy may have yielded better results. Looking from the opposite angle, one wonders if onabotulinumtoxinA treatment without the same level of physiotherapy intervention (as often happens in clinical practice) may have shown stronger effects of the toxin per se and be more relevant to the current reality in stroke rehabilitation. Finally, we have investigated a specific functional outcome aiming to address a common clinical question for chronic stroke patients with moderate to mild spasticity, and our results were in line with those using more traditional functional outcome measures (eg, ARAT); it is conceivable that another parameter may prove more responsive or sensitive to change. The slightly higher test-retest variability associated with far targets during the grasp release task suggests more cautious interpretation of this specific subset of results.
We selected predominantly distal injection sites. In some cases, proximal spasticity might have contributed to difficulty in the grasp release task. If participants were struggling with the reach part of the task, as many did, the additional effort could have made grasp release even more challenging. Targeting additional, including more proximal, targets thought clinically to be interfering with function may prove a better model for future trials.12,31,32 Likewise, the measures of impairment gathered here (stiffness, spasticity, and muscle strength) have all been recorded in nonfunctional laboratory conditions, different to those encountered during activities of daily living. Functional use of the arm and hand often involves reaching out and requires proximal muscle activity to take the weight of the arm or stabilize it in space. This effort could alter the impact of the impairment.33,34
Muscle stiffness and spasticity in the fingers were measured in midrange to prevent contractures or tightness at end of range from interfering with measurement. Other studies though have tested throughout the available range, and this could represent a more accurate measurement of stiffness within the functional range of movement35,36; this becomes even more important as the interval from the stroke increases. 37 Accurate measurement of spasticity remains a challenge3,20 and requires careful consideration when used for patient selection or interpretation of functional change in research studies. 3
Conclusion
In this group of stroke patients, addition of a single treatment with onabotulinumtoxinA injection(s) aiming to reduce spasticity in the forearm flexors was not significantly better than physiotherapy alone in improving grasp release time or other active functional outcomes.
Supplemental Material
PrOMBiS_supplementary_information_V4 – Supplemental material for Exploratory Randomized Double-Blind Placebo-Controlled Trial of Botulinum Therapy on Grasp Release After Stroke (PrOMBiS)
Supplemental material, PrOMBiS_supplementary_information_V4 for Exploratory Randomized Double-Blind Placebo-Controlled Trial of Botulinum Therapy on Grasp Release After Stroke (PrOMBiS) by Amanda Claire Wallace, Penelope Talelli, Lucinda Crook, Duncan Austin, Rachel Farrell, Damon Hoad, Aidan G. O’Keeffe, Jonathan F. Marsden, Richard Fitzpatrick, Richard Greenwood, John C. Rothwell and David J. Werring in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would also like to acknowledge the assistance afforded by the following groups in the performance of the trial: Allergan Inc. for providing the IMP. The following groups helped with patient recruitment: The North Thames Clinical Research Network, Clinical Staff at NHNN, Different Strokes Group, the ARNI Institute. We thank all patients for their time and commitment to the trial. Dr Rachel Farrell was supported by the UCLH NIHR Biomedical Research Centre.
Authors’ Note
Clinical trial registration: EudraCT 2009-009357-22.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RF has received honoraria or consultancy fees from GW Pharma, Canbex Pharmaceuticals Ltd, Biogen Idec, Merck, and Allergan PLC. PT has received a portable EMG/stimulation machine from Allergan to facilitate the operation of the spasticity service. DW reports the following disclosures in the preceding 5 years: Honoraria (speaking) from Bayer 2016, 2017. Consultancy fees from Amgen (2016), Bayer (2013), Allergan (2013), Daiichi-Sankyo (2012), and Bayer (2017). UK Chief investigator for A9951024 (Pfizer). UCLH PI for NAVIGATE-ESUS (Bayer, 2016), B2341002 (Pfizer 2014-2016), Action-2 (2016-Biogen). The remaining authors report no disclosures.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Study funding: Supported by UK Stroke Association (TSA 2008/01).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
