Abstract
Background
Upper limb activity following stroke is low, which may limit recovery. We investigated whether a virtually-delivered upper limb program, that included a wearable device with reach-to-grasp feedback, would increase upper limb activity after stroke.
Methods
This was a parallel-group, assessor-blinded, randomized control trial conducted at 6 sites across 5 provinces of the CanStroke Recovery Trials Platform between 2020 to 2022. Participants (n = 73) were community-living, less than 1 year post stroke, and had residual arm movement and upper limb use limitations. Participants were randomized via a central web-based randomization service to receive a virtually delivered program (Virtual Arm Boot Camp [V-ABC], n = 36) or waitlist control (n = 37) receiving usual care. V-ABC consisted of a home exercise program, feedback from a wrist-worn device to monitor reach-to-grasp counts, and 6 virtual sessions with a trained therapist over 3 weeks. The primary outcome was the average daily reach-to-grasp counts over 3 days at 4 weeks post baseline assessment. Secondary outcomes included upper limb function, self-reported use, and quality of life. Within-subject changes between pre, post treatment, and 2 months follow up for all participants were also examined as a tertiary analysis.
Results
The V-ABC group demonstrated greater average daily reach-to-grasp counts (primary outcome) at 4 weeks compared to control (mean difference = 368, 95% confidence interval = 6-730,
Conclusions
This study provided evidence that a virtually delivered upper limb program that consists of exercise, feedback from a wearable device, and therapist support can increase real-world upper limb activity following stroke.
Clinical Trial Registration:
NCT04232163.
Keywords
Introduction
High intensity upper limb practice is associated with greater upper limb recovery after stroke.1-3 While the optimal dose of practice is still unclear,4,5 current rehabilitation practice falls short of achieving practice levels that are associated with greater improvements.6-8 Encouraging greater intensity of upper limb activity, which typically includes strengthening and task-specific training, and/or greater incorporation of the upper limb in daily activities, remain an important goal for optimizing improvement across the recovery timeline.
Feedback from wearable devices has been used to increase physical activity (eg, walking) among older adults 9 but its usefulness in facilitating greater upper limb activity after stroke is lacking. One pilot randomized controlled trial (RCT; n = 33) used a wrist-worn accelerometer that vibrated when activity fell below individually set thresholds. The authors found the protocol to be feasible but did not determine treatment effect estimates due to the small sample size. 10 More recently, another RCT (n = 22) found individuals who received 3 weeks of feedback from a novel magnetic wristwatch/ring system increased their upper limb activity counts over time when feedback was provided. 11 They reported that there was no difference in upper limb activity, compared to the no-feedback group, once feedback was removed but did not provide treatment effect estimates.
The current study investigated whether a virtual upper limb program that incorporates feedback from a wearable device was associated with greater upper limb activity post-stroke compared to usual care. A virtual program was undertaken to reduce barriers to access and to align with restrictions early in the coronavirus disease 2019 pandemic. The trial is innovative in utilizing an objective outcome measure that captures daily functional reach-to-grasp movements in real-time over multiple days.
Methods
Study Design and Setting
We conducted a multicenter, parallel-group, waitlist controlled randomized trial with blinded outcome assessment. 12 A waitlist design was undertaken to support recruitment and retention. Participants were recruited from 6 rehabilitation hospitals across 5 provinces in Canada across the CanStroke Recovery Trials Platform (https://canadianstroke.ca/). As participants did not need to come to a specific centre (ie, assessments and treatment were delivered remotely), each site used multiple strategies to recruit participants across a wide area (eg, newsletters and visits to stroke recovery groups, recruitment from different outpatient rehabilitation units in the region). Ethics approval was obtained from the university and hospital review boards from each site and all participants provided electronic informed consent. This study followed the Consolidated Standards of Reporting Trials reporting guideline. A qualitative descriptive study was conducted alongside the RCT to explore patients’ views and experiences of the virtual upper limb program, including the wearable sensor, assigned exercises, therapy sessions, and trying to use their affected arms more during daily activities. The qualitative results are reported elsewhere. 13
Participants
Individuals were screened for eligibility. Inclusion criteria were: (1) less than 12 months post-stroke (classified as infarct or intracerebral hemorrhage); (2) unilateral upper limb impairment and less self-reported affected upper limb arm use compared to before the stroke (Rating of Everyday Arm-use in the Community and Home scale [REACH] <5) 14 ; (3) living in the community; (4) some ability to move the arm and hand (at least 30° shoulder flexion or abduction and extend at least 1 finger and thumb from a flexed position); (5) completed formal physical and occupational therapy rehabilitation for the upper limb (or no greater than 1 session/week); (6) access to a tablet, computer, laptop, or phone with internet and email access; and (7) willing and able to wear an activity monitor safely for waking hours. Caregivers were invited to assist in the study but were not necessary to participate. Individuals were excluded if they had: (1) musculoskeletal/other neurological conditions that limit movement in their arm; (2) inability to provide informed consent; (3) another medical condition that would affect their ability to participate in the treatment protocol (eg, active cancer); (4) persistent pain in their affected upper limb that affected their ability to use the limb; and (5) inability to speak, understand, or read English, unless another person could be present to translate during assessment, treatment sessions, and home activities.
Intervention
The intervention protocol for this study is described in detail elsewhere. 12 In summary, the intervention, named Virtual Arm Boot Camp (V-ABC), was delivered virtually by trained physical or occupational therapists. The V-ABC is an intense program aimed at increasing affected upper limb activity. Throughout this manuscript we use “upper limb activity” to refer to any movement of the arm and hand complex that typically results in higher counts of reaching and grasping.
The active ingredients of V-ABC consisted of three major components: (1) home exercise program using an adapted version of the Home Graded Repetitive Arm Supplementary Program (H-GRASP) to increase functional capacity of the affected upper limb; (2) feedback from a wrist-worn device (called the TENZR [explained below]); and (3) 6 virtual treatment sessions over 3 weeks. Three weeks has been shown to be a feasible period for daily wearing of a wrist-worn device and obtaining functional improvements with intense practice.10,11,15 The H-GRASP is a self-administered program that consists of active range of motion, strengthening, task-based, and fine-motor activities 16 and is detailed at neurorehab.med.ubc.ca/GRASP. We adapted the H-GRASP program by changing the following: (1) added more information about posture when doing exercises, (2) added exercises (ie, breathing, seated hip abduction, wall push ups, shoulder external rotation, finger extension, arm abduction, wrist extension), and (3) removed exercises (ie, 1 arm table push up and finger strength with putty). During the treatment sessions, the therapists taught the participants how to perform the GRASP exercises, discussed the importance of practice intensity, provided training on the TENZR app, monitored, and advanced GRASP exercises as needed, set daily TENZR count targets, and problem solved adherence issues. Participants were provided with a binder that contained verbal and pictorial instructions for each GRASP exercise and a kit that included necessary exercise equipment. The participant binder also included a checklist of possible daily tasks in which to incorporate the affected upper limb and information about the TENZR device. Participants were asked to perform the GRASP program twice daily (or at least 2 hours/weekday) and to wear the TENZR during waking hours. Starting at session 2, the treating therapist set daily reach-to-grasp targets, in collaboration with the participant, based on the average daily TENZR counts obtained in between sessions. The guidance on how to set the targets was as follows: (1) if the average of the previous daily counts obtained by the TENZR were <1000, set the target to 1000 counts or (2) if the average of the previous daily counts is greater than 1000 then add an additional 20% to the average. The cut-off of 1000 was based on reach-to-grasp counts obtained while piloting 14 individuals with mild/moderate upper limb stroke impairment when they completed the H-GRASP program (mean = 760 counts). Participants were guided to meet targets by completing more exercises and/or using their affected hand in daily tasks.
The V-ABC was informed by the application of the Capability, Opportunity, Motivation, Behaviour Model (COM-B) 17 to upper limb use after stroke. The COM-B components of the intervention are detailed in the trial protocol, 12 for example, an individual’s psychological ability to participate in upper limb activity (Psychological Capability) was addressed by goal setting, barrier identification, and problem-solving.
Control Group
While on the waitlist period, control participants could be receiving formal physical and occupational therapy rehabilitation for the upper limb (eg, strengthening exercises and functional electrical stimulation) for up to 1 session per week. Rehabilitation services are typically provided in the first 2 to 3 months post-stroke in Canada, so it was anticipated that our participants (average 7 months post-stroke) would not be receiving formal therapy. After that, people often seek community programs that involve general fitness and mobility.
TENZR Wearable Device
The TENZR Neurotracker device (version 4.0.2) is an investigational medical device manufactured by BioInteractive Technologies. The TENZR uses a multi-sensing approach to detect reach-to-grasp activity. Force myography is a component in the device used to monitor surface forces of the musculoskeletal-tendon complex at the wrist. The device outputs a count of 1 when it detects a functional reach-to-grasp movement, which is defined as any finger/grip and wrist activity accompanied by inertial movements at the arm just before or after the hand/wrist activity. 18 Our previous work found that the TENZR can reliably capture upper limb functional activity as expected in both controlled and real-world settings among individuals with mild to moderate upper limb stroke impairment.18,19 For reference, the TENZR averaged 1 to 2 counts when opening a door in a controlled environment (ie, reaching the arm to grasp and turn a lever handle, opening the door slightly and then returning the hand to the side). 18 In the real-world setting, we previously obtained median daily counts of 1480 and 4691 for the affected and unaffected hand among individuals with stroke and 3559 and 5021 for the non-dominant and dominant hand among age-matched controls. 19 The TENZR device does not have a visual display so participants were provided with a tablet (Microsoft Surface Go2) containing a pre-installed app and a Bluetooth dongle. The wearable device synchronizes with the app every 15 seconds for the purpose of displaying data to the participant (Figure 1). Data are stored on the wearable in 15-minute time segments. Participants were asked to remove the TENZR if they were immersing their wrist in water (eg, bathing, swimming, and washing dishes) as the device is not waterproof. The reported battery life on the TENZR is 14 hours when charged for 8 hours. Participants were asked to charge the device overnight. A flashing green status light on the top of the TENZR indicated the device was working properly. The treating therapist/study coordinators downloaded the treatment TENZR count data to the tablet at each training session and transferred the data to the study server via remote connectivity software (TeamViewer, Göppingen, Germany)

TENZR wrist-worn device and app. (A) TENZR wrist-worn sensor. (B) App showing a target of 1000 reach-to-grasp counts with 1029 completed. (C) App showing a target of 1000 reach-to-grasp counts with 408 completed.
Study Procedures
Participant characteristics including age, sex, date, and type of stroke, pre-stroke handedness (indicated by their writing hand), living situation, cognitive status, 20 and depressive symptoms 21 were collected by site coordinators following consent.
All outcome measures were collected at baseline, 4 weeks post baseline and 2 months after the intervention was completed (Figure 2). The control group was offered the intervention after the 4-week evaluation and also had an evaluation immediately after the intervention (Figure 2). All outcome measures except the primary outcome were administered via a videoconference session with a blinded assessor. At each assessment time point, participants were asked to wear the TENZR during their waking hours for 3 days to capture average daily reach-to-grasp counts. Feedback on the app was disabled when used for outcome assessment. A valid day was defined as a minimum of 6 hours of wear. Wear time was determined as the number of time segments in between the first non-zero segment and the last non-zero segment of the day and converted to hours. Attempts were made to schedule the clinical assessment and TENZR assessments on different days. However, when this was not possible, the TENZR counts associated with the time of the clinical assessments were removed (10 participants at baseline and 10 participants at post-evaluation). TENZR assessment data was downloaded from the wearable and transferred to the study server via remote connectivity software.
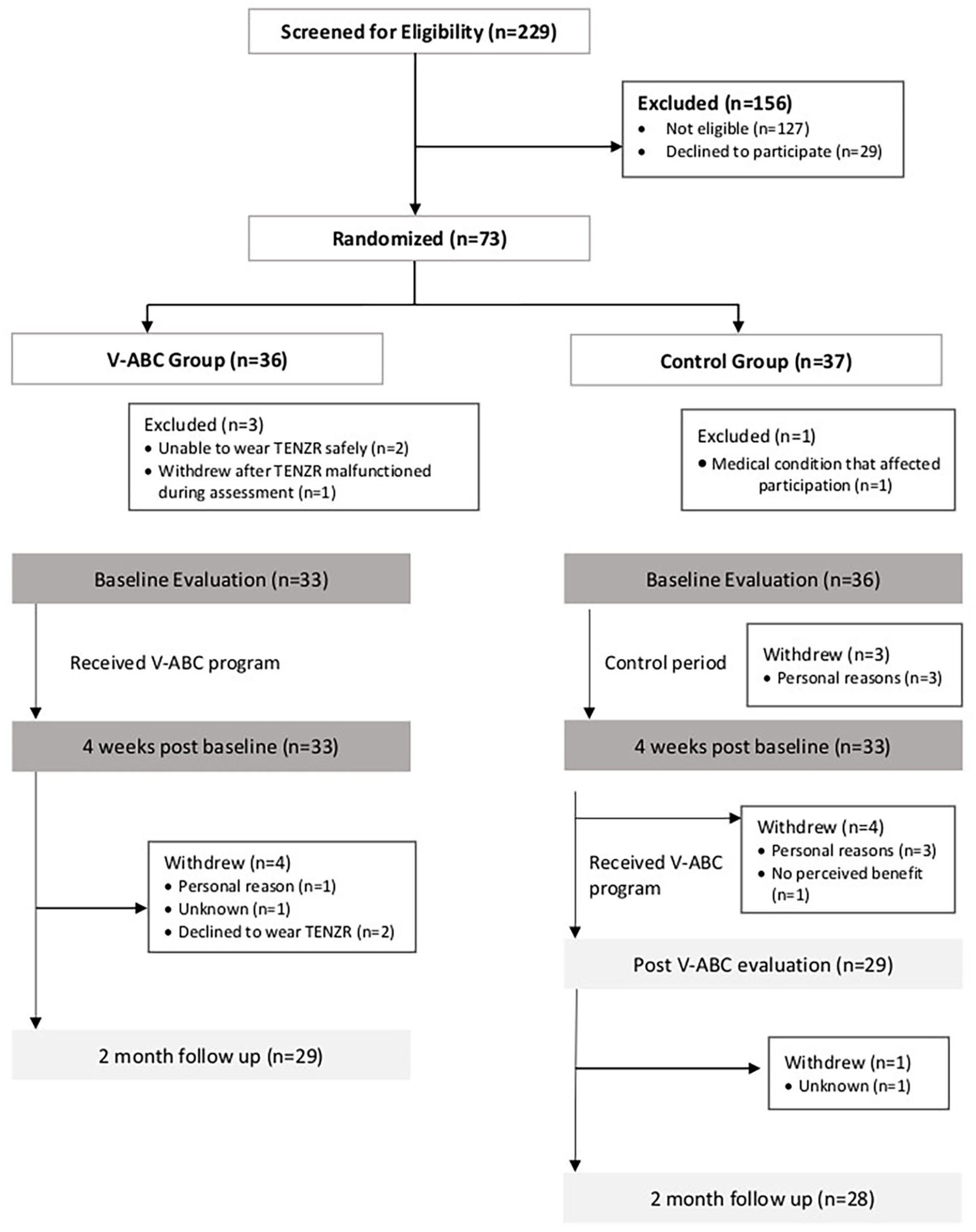
Consolidated Standards of Reporting Trials flow diagram.
We allocated participants in a 1:1 ratio using central web-based randomization (randomize.net) employing random permuted blocks of varying size (2 and 4). The randomization was stratified by site and self-report upper limb use level (ie, REACH score <3 and ≥3). A score of 3 distinguishes between individuals with mild versus moderately affected upper limbs. Assessors were blinded to group assignment. The local research coordinators informed the participant of their assigned grouping following the baseline assessment.
Outcomes
The primary outcome was real-world upper limb activity measured by the average daily reach-to-grasp counts over 3 days at 4 weeks post-baseline. The average was calculated as the total counts during valid days of wear divided by valid days of wear. There were 4 secondary outcomes: (1) self-reported affected arm use using the REACH scale.
14
The REACH is a 6-level classification scale that captures how the upper limb is used in everyday life with a 1-level change on the scale considered a clinically important difference (CID).
22
The REACH consists of separate scales for dominant and non-dominant affected upper limbs with a similar progression of levels that range from a score of 0 (no use) to 5 (full use).
14
Higher levels represent greater complexity of use. (2) upper limb function using the Arm Capacity and Movement Test (ArmCAM)
23
; The ArmCAM was designed by our research team to capture upper limb function remotely (eg, slide a towel, pour a cup, stack coins). Scores range from 0 to 30. Clinically important differences in the ArmCAM have not been investigated. A minimal detectable change of 2.05 points has been reported.
23
(3) self-reported upper limb ability in one’s own environment using the Stroke Impact Scale (SIS) hand subscale. (4) Self-reported upper limb strength using the SIS strength subscale (upper limb portion only).
24
Domain scores for the hand and upper limb strength scales were transformed to be a score ranging from 0 to 100 using the following formula: (raw score − minimum possible score)/possible raw score range
Adverse Events and Adherence
Study staff reported any serious adverse events (eg, hospitalizations and life-threatening injuries/illness) that occurred during the V-ABC intervention. In addition, pain (measured on a 0-10 numerical scale) 27 was captured at each assessment time point. Participants were asked to record the start and end time of their exercise sessions on an exercise log and therapists recorded this information during each virtual treatment session. Logs were considered valid if they were at least 75% complete. Incomplete entries on valid logs were counted as zero exercise performed. Adherence was defined as completing a mean of 75% of 2-hours/day of GRASP exercises for the monitored period (ie, period of time in between the first and last virtual treatment sessions).
Statistical Analysis
We assumed there would be a large effect size (Cohen’s
Constrained linear mixed effect models fitted using restricted maximum likelihood were used to estimate between group differences in the primary and secondary outcomes at 4 weeks. Constrained linear mixed effect models, which constrains the mean of the baseline outcomes to be equal between groups, allows the use of all data points under the missing at random assumption and provides an unbiased estimate of the treatment effect. 28 All models for these group comparisons included participant as a random effect while time point (ie, baseline and 4 weeks) and time point by group interaction were set as fixed effects. If there is no missing data, this analysis is equivalent to an analysis of covariance analysis. 28 Participants were analyzed in the group to which they were randomized regardless of adherence. Site was not a variable in the models as our participants did not need to come to a specific geographical centre (ie, assessments and treatment were delivered remotely); sites were used to coordinate recruitment over large and sometimes overlapping regions.
An additional tertiary analysis was conducted given that the control group was offered the V-ABC intervention after the waitlist control period. This analysis examined the within person changes in all outcomes across the pre-treatment, post-treatment, and follow up time points by combining the data for all participants, whether they were in the immediate group of the RCT or waitlist group who were offered V-ABC after the control period. Linear mixed effects models were used to conduct this tertiary analysis. All models for the tertiary analysis included participant as a random effect and time point and treatment group (ie, immediate vs waitlist) as fixed effects. Treatment group was included to account for any difference in average level between groups. The assessment undertaken immediately prior to starting the V-ABC intervention (ie, at 4 weeks post-baseline) was selected as the pre-treatment value for the waitlist group participants. The baseline value was used as the pre-treatment value for the three participants in the waitlist group who dropped out before the 4-week time point. All analyses were conducted with R v4.1.1 and the
Results
A total of 73 individuals were randomized to the V-ABC or waitlist groups between September 2020 and July 2022 (Figure 2) with 69 completing the baseline evaluation and 66 (33 in each group) completing the evaluation at 4 weeks. Three individuals were found to be ineligible while completing the TENZR portion of the baseline assessment and 1 individual dropped out while completing the TENZR portion of the baseline assessment when it malfunctioned (Figure 2). Table 1 displays the descriptive characteristics of the 69 participants. On average, participants were 7 months post-stroke (ie, early chronic phase). At least 10 participants lived in cities with populations less than 100 000, with 5 of these from rural cities of less than 5000.
Participant Characteristics.
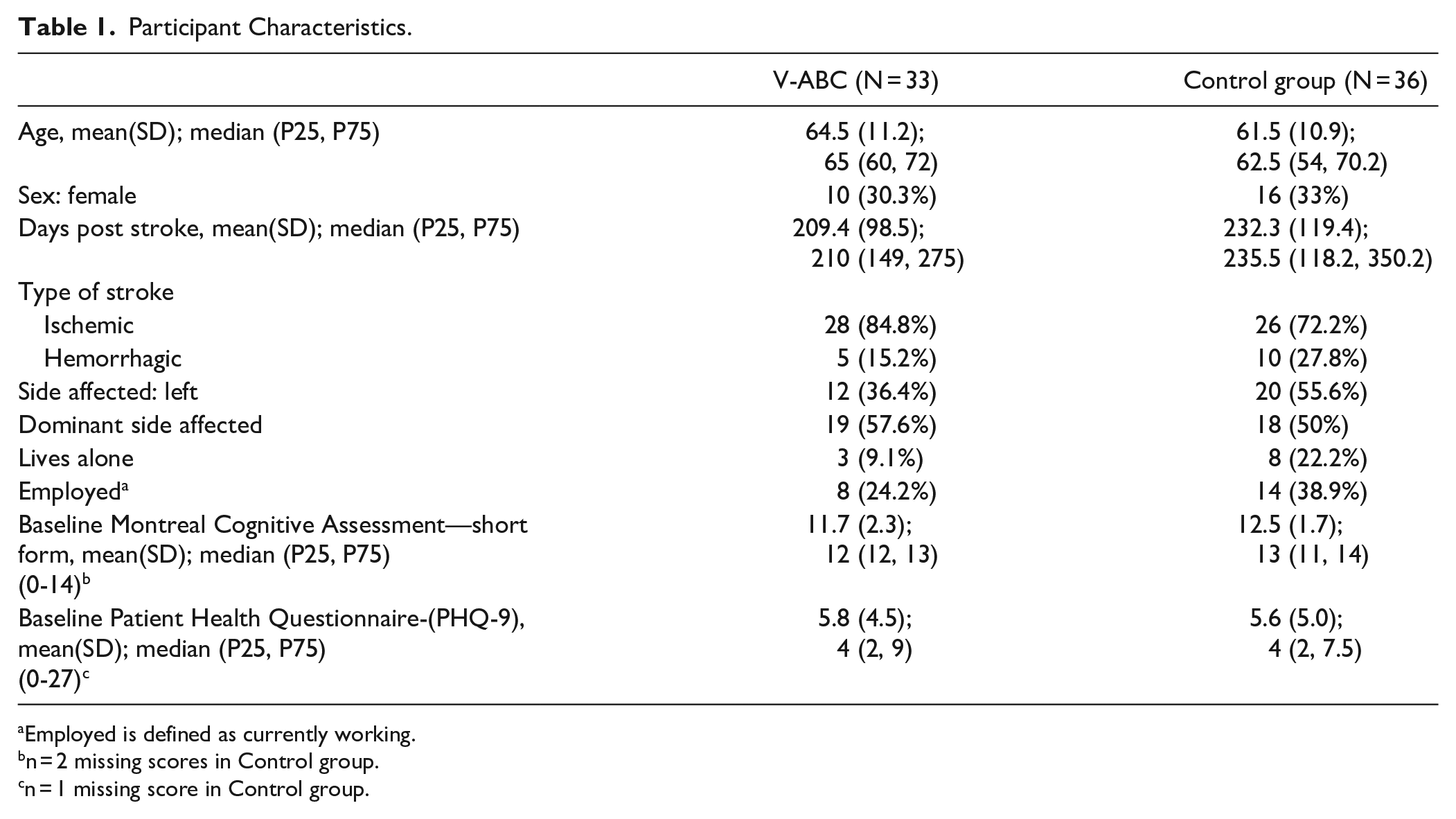
Employed is defined as currently working.
n = 2 missing scores in Control group.
n = 1 missing score in Control group.
Primary Outcome at 4 Weeks (RCT Analysis)
Participants wore the TENZR for an average (standard deviation [SD]) of 12.8 (2.3) hours/day at baseline and 12.5 (1.5) hours/day at 4 weeks. A mean of 3.0 (0.7) valid days was obtained at baseline and 3.0 (0.5) valid days at 4 weeks. While we did not collect information from the control group on what activities they might be participating in, their baseline upper limb activities over a three-day period (1218 counts) were similar to the intervention group (1239), suggesting they were undertaking similar intensities of upper limb activity. At the 4-week evaluation, the V-ABC group had higher average daily reach-to-grasp counts than the control group (mean difference = 368, 95% confidence interval = 6-730
Primary and Secondary Outcomes.
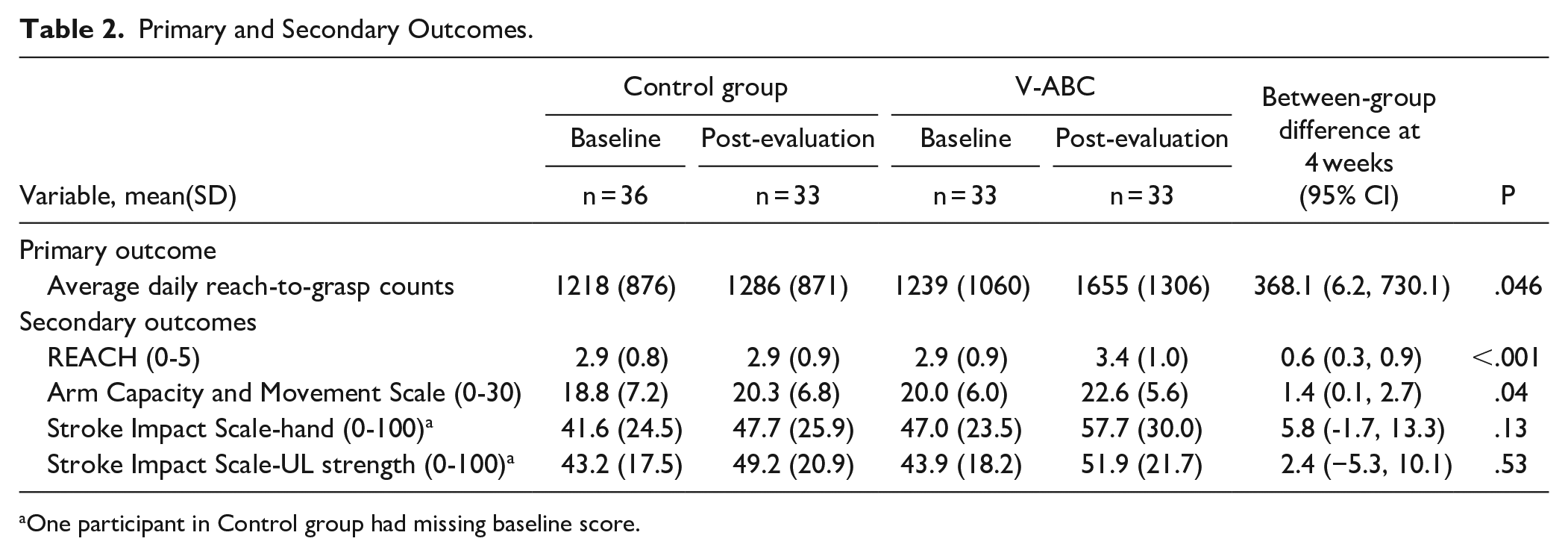
One participant in Control group had missing baseline score.
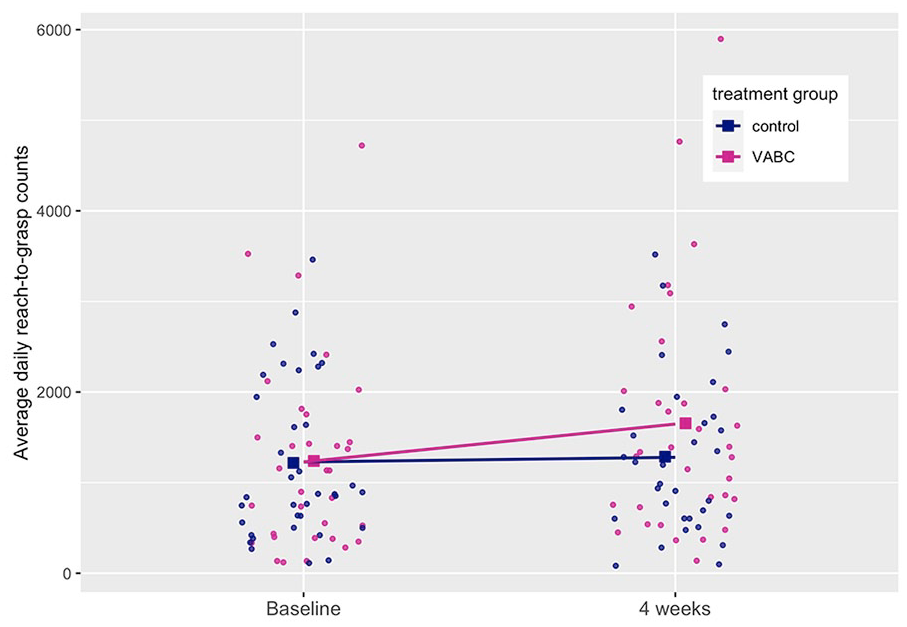
Plot of average daily reach-to-grasp counts at baseline and 4 weeks.
Secondary Outcomes at 4 Weeks (RCT Analysis)
There were significant group differences at 4 weeks in how the affected upper limb was used (mean difference in REACH score = 0.57, 95% confidence interval [CI] = 0.26-0.87,
Longitudinal Models Examining Pre, Post, and Follow up V-ABC Program (N = 69).
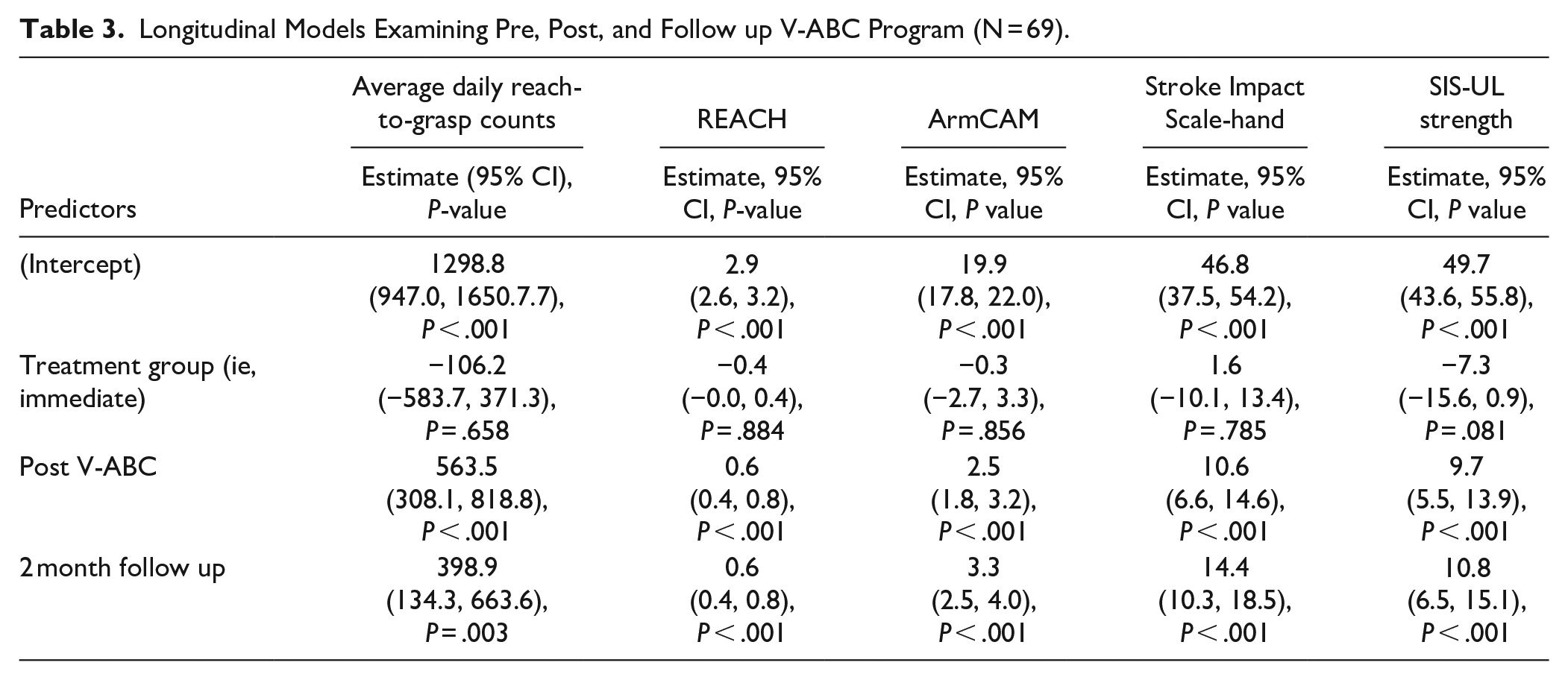
Tertiary Analysis of Longitudinal Changes for All Participants
A total of 62 participants completed the V-ABC program and post-treatment evaluation (33 from the Immediate Group of the RCT and 29 from the Waitlist Group who completed V-ABC after the control period). A total of 57 participants completed the 2-month follow up evaluation (Figure 2). We observed pre-post V-ABC program improvements in all outcomes (Table 3).
Treatment Adherence and Adverse Events
Exercise logs were obtained from 22/33 (67%) of the V-ABC participants and their monitoring period lasted a mean of 16.3 (SD = 2.0) days. The mean duration of exercise performed per day during the monitoring period for this group was 2.3 (SD = 0.9) hours. A total of 16/22 (73%) of the treatment group with valid logs were adherent to the V-ABC program.
There were 2 serious adverse events that occurred during the trial. One person was hospitalized with an unrelated illness and another person was hospitalized with a new-onset stroke that was deemed not related to the intervention. The between-group difference in pain scores at 4 weeks was small and not compatible with a clinically important difference (ie, >2 out of 10; mean difference = −0.3 (95% CI = −1.2 to 0.7;
Discussion
This novel multicomponent program utilized feedback from wearable devices to increase daily arm and hand activity. Our upper limb program resulted in greater daily reach and grasp activity compared to usual care for individuals with some residual movement in the first-year post-stroke. Importantly, as a result of V-ABC, participants were using their arm and hand for 368 more reach-to-grasp counts over the day after the program ended and this represents a 30% increase from the participant’s baseline activity. Moreover, our study observed increases in daily arm and hand activity when participants were no longer receiving feedback from the device. This is in contrast to the study by Schwertz et al 11 which found that feedback from a device was associated with increases in upper limb activity only when feedback was provided.
These results are encouraging given the observed plateaus in upper limb activity at this point in stroke recovery22,29 and because this is one of the first trials to use an objective real-time continuous measure of upper limb activity as the primary outcome. Multicomponent programs, such as constraint-induced movement therapy (CIMT), which have been shown to increase upper limb activity, used self-report measures of use as their activity outcome. 30 Our findings are promising as they suggest a multicomponent intervention, such as V-ABC, can increase objective real-time upper limb activity. Moreover, this study demonstrated the feasibility of utilizing sensors for feedback through a virtually delivered intervention which facilitated the reach of the program, including some participants from small cities, as well as rural regions. Such a virtual program could improve the known inequities in accessing health care services 31 and help to improve positive stroke outcomes with rural patients. 32
Encouraging activity via sensors alone without improving an individual’s upper limb function is unlikely to result in activity changes once feedback is removed. 11 Conversely, it is not likely that changes in upper limb function will automatically translate into greater upper limb activity at home. 33 For these reasons, the V-ABC program coupled daily intensive practice to improve function, together with components to facilitate transfer to home activity (support on how to incorporate the upper limb in daily activities, feedback from wearable sensors).
We did observe small statistically significant group differences in the ArmCAM and REACH scale. It also should be noted that 16 (48%) people in the treatment group improved by at least 1 level in the REACH scale (ie, CID) versus 5 (15%) in the control group. The REACH scale asks participants specific questions about how they are using their limbs in daily life (eg, “in the last 3 days did you use your affected limb to reach for a light item in the fridge, cupboard, drawer, or from a table”). Small group differences in the REACH scale at 4 weeks suggests that more individuals in the V-ABC group were using their arms differently, and in a more complex way, than the control group.
Pre-post study designs have made important contributions to the field of upper limb rehabilitation following stroke.2,15 Our tertiary analysis of all participants, included the immediate treatment group, as well as the original control group who were offered V-ABC after the control period. This analysis provides support for the ability of the V-ABC program to induce changes in activity when these patients would typically have a period of observed plateau in recovery.22,29 Improvements in reach to grasp counts represented a 45% increase over pre-program levels (ie, 1241). It should be noted that 7 individuals from the waitlist group dropped out of the study before the post-intervention evaluation. These individuals may have responded differently to the V-ABC program than those that stayed in the study.
Most participants who completed the V-ABC program adhered to the exercise minimum with an overall average of 2.3 hours/day. Interestingly, a large home-CIMT trial, which asked participants to complete 2 hours of weekday practice over 4 weeks, only saw participants completing an average of 1.4 hours per day. 34 Feedback from the TENZR may have contributed to greater adherence in our study. Our qualitative investigation that was embedded within this RCT showed that the participants found the V-ABC protocol to be motivating for practice; they found that the TENZR sensor provided a useful mechanism for goal setting and accountability, and they found the therapists provided helpful support with encouragement and progression of the GRASP exercises. 13 The V-ABC program encouraged participants to continue exercising and using their upper limb in daily activities beyond program completion. It is thus possible that some of the between group difference in activity at post-evaluation is due to participants continuing to perform upper limb exercise.
This trial has several limitations. Our sample size was small and limited to those with some residual upper limb movement. In addition, our study included individuals with a large range of impairment and activity levels. A larger sample would enable the analyses of factors that might influence the outcome such as sex, severity of arm impairment and adherence to the protocol. A recent study suggests that among individuals with mild/moderate upper limb impairment, the dose response varied widely between individuals. 35 Thus some participants in our study may have required greater than 2 hours of exercises per day and/or a longer duration of treatment to experience improvements while others may not have required this much. Individuals currently receive little to no care after discharge from outpatient therapy, but we cannot isolate the effect of attention with our waitlist design. We also acknowledge that a within-subject design was used for the 2-month follow-up analysis and did not account for changes due to natural recovery or usual care. Finally, we had a low completion rate of exercise logs to measure adherence among the immediate V-ABC group participants. This was due to participants having difficulty filling them out and therapists initially forgetting to transcribe this information at each session.
Footnotes
Acknowledgements
The authors are indebted to the following for their support of the study: (1) the study sites (GF Strong Rehabilitation Centre, Foothills Medical Centre, Riverview Health Centre, Parkwood Institute, Sunnybrook Health Sciences, UHN-Toronto Rehabilitation Institute, Nova Scotia Rehabilitation and Arthritis Centre); (2) central assistance from Chihya Hung, Farrell Leibovitch and Josie Chundamala; and (3) the study participants.
Author Contributions
Lisa A. Simpson: Conceptualization; Data curation; Formal analysis; Methodology; Project administration; Supervision; Writing—original draft; and Writing—review & editing. Ruth Barclay: Conceptualization; Funding acquisition; Methodology; Project administration; Resources; Supervision; and Writing—review & editing. Mark T Bayley: Conceptualization; Funding acquisition; Methodology; Project administration; Resources; Supervision; and Writing—review & editing. Penelope M. A. Brasher: Formal analysis and Writing—review & editing. Sean P. Dukelow: Conceptualization; Funding acquisition; Methodology; Project administration; Resources; Supervision; and Writing—review & editing. Bradley J. MacIntosh: Conceptualization; Funding acquisition; Methodology; Project administration; Resources; Supervision; and Writing—review & editing. Marilyn MacKay-Lyons: Conceptualization; Funding acquisition; Methodology; Project administration; Resources; Supervision; and Writing—review & editing. Carlo Menon: Conceptualization; Methodology; and Writing—review & editing. W Ben Mortenson: Conceptualization; Methodology; and Writing—review & editing. Tzu-Hsuan Peng: Data curation; Investigation; and Writing—review & editing. Courtney L. Pollock: Conceptualization; Funding acquisition; Methodology; Project administration; Resources; Supervision; and Writing—review & editing. Sepideh Pooyania: Conceptualization; Funding acquisition; Methodology; Project administration; Resources; Supervision; and Writing—review & editing. Noah D. Silverberg: Conceptualization; Methodology; and Writing—review & editing. Robert Teasell: Conceptualization; Funding acquisition; Project administration; Resources; Supervision; and Writing—review & editing. Jennifer Yao: Methodology; Project administration; Resources; Supervision; and Writing—review & editing. Janice J. Eng: Conceptualization; Formal analysis; Funding acquisition; Methodology; Project administration; Resources; Supervision; and Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Menon has a vested interest in commercializing the technology and may benefit financially from its potential commercialization through BioInteractive Technologies. The other authors declare that they have no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by: the Canadian Institutes of Health Research (MOP-137053; CPG 134766; FND-143340; JJE); Canada Research Chairs Program (JJE); Canadian Institutes for Health Research Fellowship (LAS); and Canada Brain Research Fund (CanStroke Recovery Trials Platform). The funders of the study had no role in study design, data collection, data analysis, data interpretation or writing of the manuscript.
