Abstract
Introduction
Spasticity after stroke—just 1 component of the upper–motor neuron syndrome—may contribute to functional impairment as a result of reduced range of motion, dexterity, and pain. Spasticity has been reported in more than one third of patients within 12 months after stroke,1,2 and a proportion of these patients will develop disabling spasticity requiring intervention. 3 During the early phases, about 19% of stroke patients, 4 or possibly more, 5 develop spasticity 3 months poststroke; indeed a recent study suggests that as many as 80% of patients without useful functional arm movement poststroke develop spasticity within 6 weeks of first stroke (measured by muscle activation recording). 6
Botulinum neurotoxin type A (BoNT-A) has been proved to be effective in treating spasticity of various etiologies, as measured by lowering the Modified Ashworth Scale (MAS) score that clinically assesses hypertonicity during passive range of motion across a joint. 7 A recent therapeutics and technology assessment from the American Academy of Neurology supports the use of BoNT-A in the treatment of spasticity. 8 However, in most studies to date, BoNT-A injection has been performed more than 6 months after stroke (average of 2.5 years). 9 Although this late intervention may be effective in treating spasticity per se, because chronic spasticity can lead to changes in the rheological properties of the involved muscles with resultant muscle stiffness and contractures that can affect limb movement, it is possible that earlier intervention may decrease early-onset muscle stiffness and achieve a better long-term outcome. 10 A small Phase II trial examined the effect of early treatment with low-dose BoNT-A (half or quarter the usual dose) 11 but was powered only as a preliminary evaluation to estimate sample and treatment effect sizes. A recent international consensus statement calls for further study into the early injection of BoNT-A as preventive treatment for the development of contracture or unmasking potential for active movement. 12
The Asia Botulinum Toxin-A Clinical Trial Designed for Early Post-stroke Spasticity (ABCDE-S) study was performed to evaluate the efficacy and safety of BoNT-A injection for the reduction of excessive muscle tone of upper-limb muscles in adult patients. The intervention began 2 to 12 weeks after stroke.13,14 Secondary objectives were to evaluate whether BoNT-A can improve neurological outcome by improving mobility and function and whether it can alleviate painful spasm. This is the first fully powered study designed to evaluate the effects of early BoNT-A treatment after a stroke. We hypothesize that BoNT-A is effective in the treatment of early poststroke upper-limb spasticity compared with placebo.
Methods
Participants
Participating centers were 5 neurological and rehabilitation units in Hong Kong, Malaysia, the Philippines, Singapore, and Thailand. Men and women aged 18 to 80 years and of Asian ethnicity were eligible. Participants were required to give written informed consent.
Patients were recruited within 2 to 12 weeks after their first-ever stroke with impairment according to World Health Organization criteria. 15 Ischemic or hemorrhagic stroke was confirmed by CT/MRI. Patients were required to have a MAS score of 1+ (slight increase in muscle tone manifested by a catch, followed by minimal resistance throughout the remainder [less than half] of the range of motion) or higher in the elbow or wrist joint. They also had to have weakness of at least grade 2 according to Medical Research Council 16 criteria in the relevant joint to be eligible. Exclusion criteria included pregnancy/lactation, prestroke Rankin score greater than 1, known hypersensitivity to test materials or related compounds, preexisting neuromuscular junction disease or neurogenic disorder that could induce muscle hypertonus, and previous treatment with botulinum toxin. Patients who were unable (eg, those with dysphasia or cognitive deficit) or unwilling to comply with the protocol were also excluded. Our protocol did not record the number of potential participants screened. We did not try to distinguish between central and peripheral mechanisms of hypertonicity. 17
Procedures
This double-blind, placebo-controlled study evaluated the efficacy and safety of 1 injection of BoNT-A into wrist and elbow mover muscles at the discretion of the investigator. Eligible patients were allocated to receive BoNT-A or placebo in a 1:1 ratio according to a predetermined randomization schedule (block size of 4) generated by the sponsor-assigned biostatistician. The sponsor-assigned randomization manager provided treatment allocation codes to the drug supplier and drug safety officer, with the master listing kept confidential and secured by the sponsor representative. Drug vials were prepared centrally, labeled with the randomization code, and delivered to the investigating centers. Placebo vials were not visually distinguishable from BoNT-A vials. Blinding was maintained until all participants had completed the study and data collection was complete. BoNT-A (Dysport 500 U toxin–hemagglutinin complex, human albumin and lactose, freeze-dried powder for injection, Tredegar, UK) and placebo (same constituents except toxin–hemagglutanin complex) were reconstituted locally with 2.5 mL normal saline.
Before commencement of the study, all investigators participated in an investigators’ meeting, which included training on all the assessment scales used in the study, including use of instruments (eg, joint angle and pain) and adverse event (AE) reporting.
As doses of different commercially available BoNT-A preparations are not interchangeable, the doses used in this study are specific to Dysport. Significant reductions in spasticity (MAS scores) in the hemiparetic arm have been reported with 500 to 1500 U doses of Dysport. 18 Because of the relatively small body size of Asian patients, a dose of Dysport 500 U was selected. Also, because of the muscle weakness present in early stroke, administering a low dose of 500 U was thought to be an appropriate approach. The recommended dose distribution was 2 injections of 200 U in a 1-mL volume for the biceps brachii, 1 injection of 100 U in a 0.5-mL volume in the brachioradialis, 1 injection of 100 U in a 0.5-mL volume in the flexor carpi ulnaris, and 1 injection of 100 U in a 0.5-mL volume in the flexor carpi radialis. Optional muscles were the flexor digitorum superficialis, the flexor digitorum profundus, and the flexor pollicis longus. Investigators were permitted to adjust the dose per targeted muscle, depending on the level of hypertonicity, as long as the total dosage per patient was 500 U. Such adjustments were recorded on the case report form.
No additional antispasticity medication was permitted after entry. Patients were permitted to continue any antispasticity medication already in place, although dose adjustment was not permitted. All patients continued with their standard rehabilitation programs throughout the study, as deemed suitable by the attending physician. These generally consisted of a 30- to 60-minute program of range of motion plus stretching exercises, strengthening and endurance exercises, and electrical stimulation in some cases.
Baseline assessments were made at the first injection visit, and all assessments were made by the same team of 3 assessors within a study site, one of whom was a physiotherapist. The primary outcome measure was MAS score 4 weeks from baseline for the most affected joint (wrist or elbow flexors) in the supine anatomic position for joints with a MAS score of 1+ or above at baseline. If both joints were equally affected, the wrist joint was retained in the analysis as the most functionally important joint. The measurement of muscle tone using the MAS has demonstrated inter- and intrarater reliability, particularly in the upper-limb joints but has some limitations.4,19-21 The timing of the primary end point measure was to coincide with the expected peak effect of BoNT-A.18,22,23
Secondary outcome measures were performed at 2, 4, 8, 12, and 24 weeks. These were changes from baseline in MAS score, Barthel index, Modified Rankin Scale scores, and Functional Motor Assessment Scale24,25 scores for upper-arm function, hand movements, and advanced hand activities. Spasticity-related pain was assessed with a visual analog scale (VAS; 0–100). Voluntary and passive joint range of motion goniometry was performed on the elbow (patients lying down with gravity eliminated), wrist (side to side with gravity eliminated), and active range of motion fingers based on hand closed, one quarter open, half open, three quarters open, and fully open.
AEs were assessed by questioning the patient at each study visit. The relationship of an AE to the study drug was classified as probable, possible, not related, or not assessable by the investigator.
The study was conducted in compliance with the Declaration of Helsinki and International Conference on Harmonisation Good Clinical Practice guidelines. Ethical approval was obtained from each of the participating centers. This study is registered at ClinicalTrials.gov (registration number NCT00234546).
Statistical Analysis
Based on previous experience, 22 the sample size calculation assumed a reduction in MAS score of 1 and a standard deviation (SD) of 2. For 86% power to detect an effect size of 0.5 between treatment groups (based on the change in MAS score) at the 2-sided, 5% significance level, a total of 162 patients were required (nonparametric Wilcoxon test).
The efficacy population was intent-to-treat (ITT), with last observation carried forward (LOCF) to account for missing values. The per-protocol (PP) analysis was conducted as a sensitivity analysis. The safety population included all patients who received at least 1 dose of study medication. Primary efficacy was assessed for the ITT and PP populations using nonparametric analysis of covariance (ANCOVA), where pooled country and baseline MAS scores were treated as covariates. Interactions between treatment and covariates were assessed using a parametric ANCOVA model. In the case of a significant treatment by baseline score interaction, treatment effect estimates would be presented by mean, first quartile (Q1), and third quartile (Q3) baseline MAS scores. As a MAS score of 1+ was arbitrarily coded as 1.5, a robustness analysis to determine the effect of coding as 1.1, 1.5, or 1.9 on treatment effect size was planned.
Secondary efficacy end points were assessed in the ITT population (LOCF) by repeated-measures ANCOVA with adjustment for baseline score. In cases of nonnormality or nonhomogeneity of variance, nonparametric ANCOVA was used. Subgroup analyses of time from stroke onset (<7 weeks vs ≥7 weeks) and stroke type (bleed vs infarct) were performed as post hoc analyses, as was an analysis of the subgroup of patients experiencing troublesome pain (≥30 mm on the VAS) at baseline. No adjustments were made for multiplicity of testing as there was only 1 primary efficacy comparison. No statistical testing was performed for AEs.
Results
Patient Characteristics and Populations
Between February 2003 and April 2007, 163 patients were recruited and randomized to receive placebo (n = 83) or BoNT-A (n = 80). Demographic and baseline characteristics are shown in Table 1; patient flow through the study is shown in Figure 1. The ITT population comprised all patients who were randomized, had received the study drug, and had completed baseline and at least 1 postbaseline efficacy assessment (163 patients: 83 in the placebo group and 80 in the BoNT-A group). The PP population excluded 10 patients identified during the blind data review as having major protocol deviations (Figure 1). The safety population comprised all patients who were randomized and received the study drug and was identical to the ITT population. Groups were well matched demographically at baseline, including clinical measures, except for a longer lead time to recruitment in the placebo (7.7 weeks) than the BoNT-A (7 weeks) group and a higher incidence of cortical strokes in the placebo (26/83) than the BoNT-A (16/80) group. The muscles injected, frequency, and dose ranges are shown in Table 2. All results presented are from the ITT population and are fully supported by the PP analysis.
Demographic and Other Baseline Characteristics
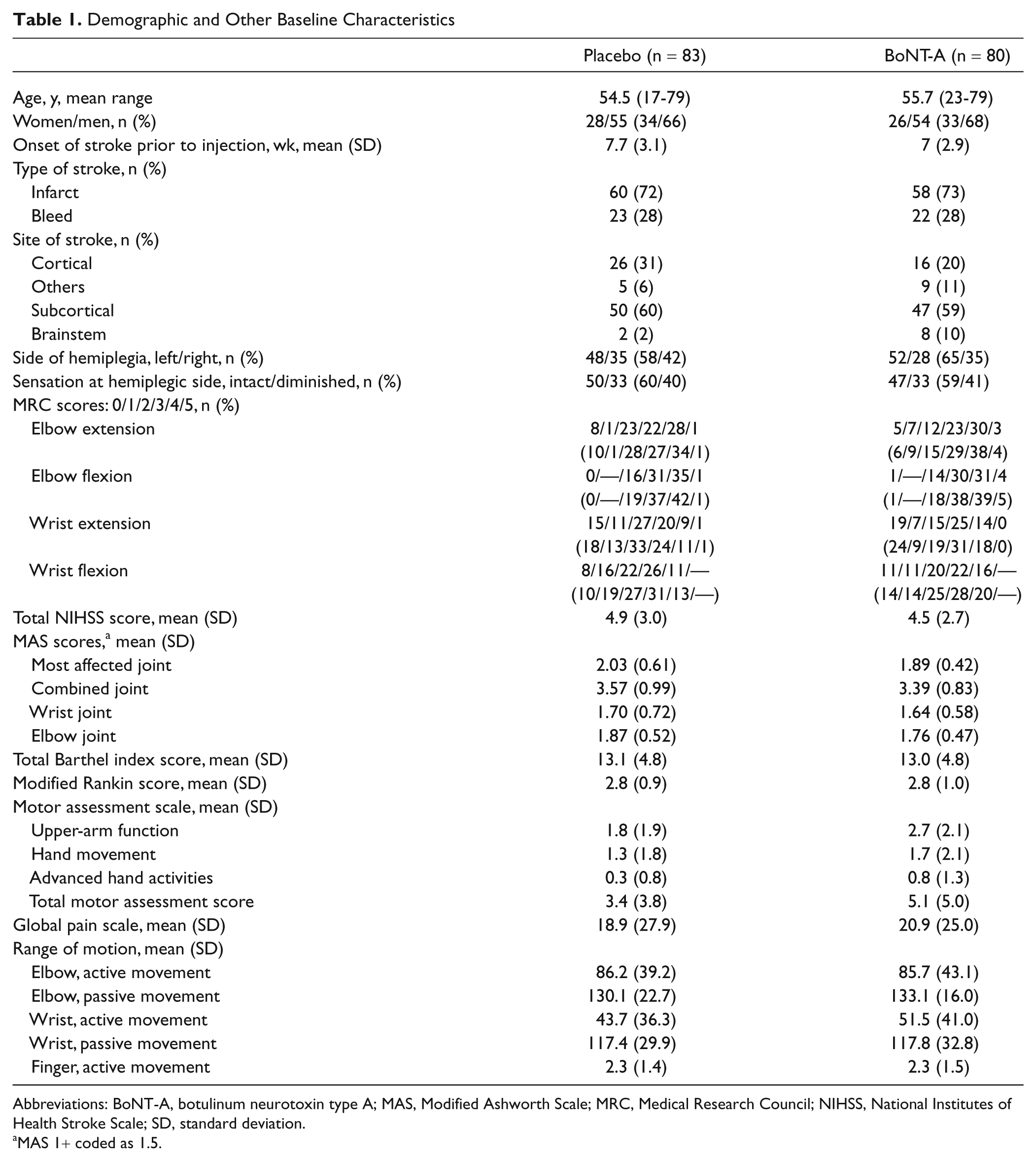
Abbreviations: BoNT-A, botulinum neurotoxin type A; MAS, Modified Ashworth Scale; MRC, Medical Research Council; NIHSS, National Institutes of Health Stroke Scale; SD, standard deviation.
MAS 1+ coded as 1.5.
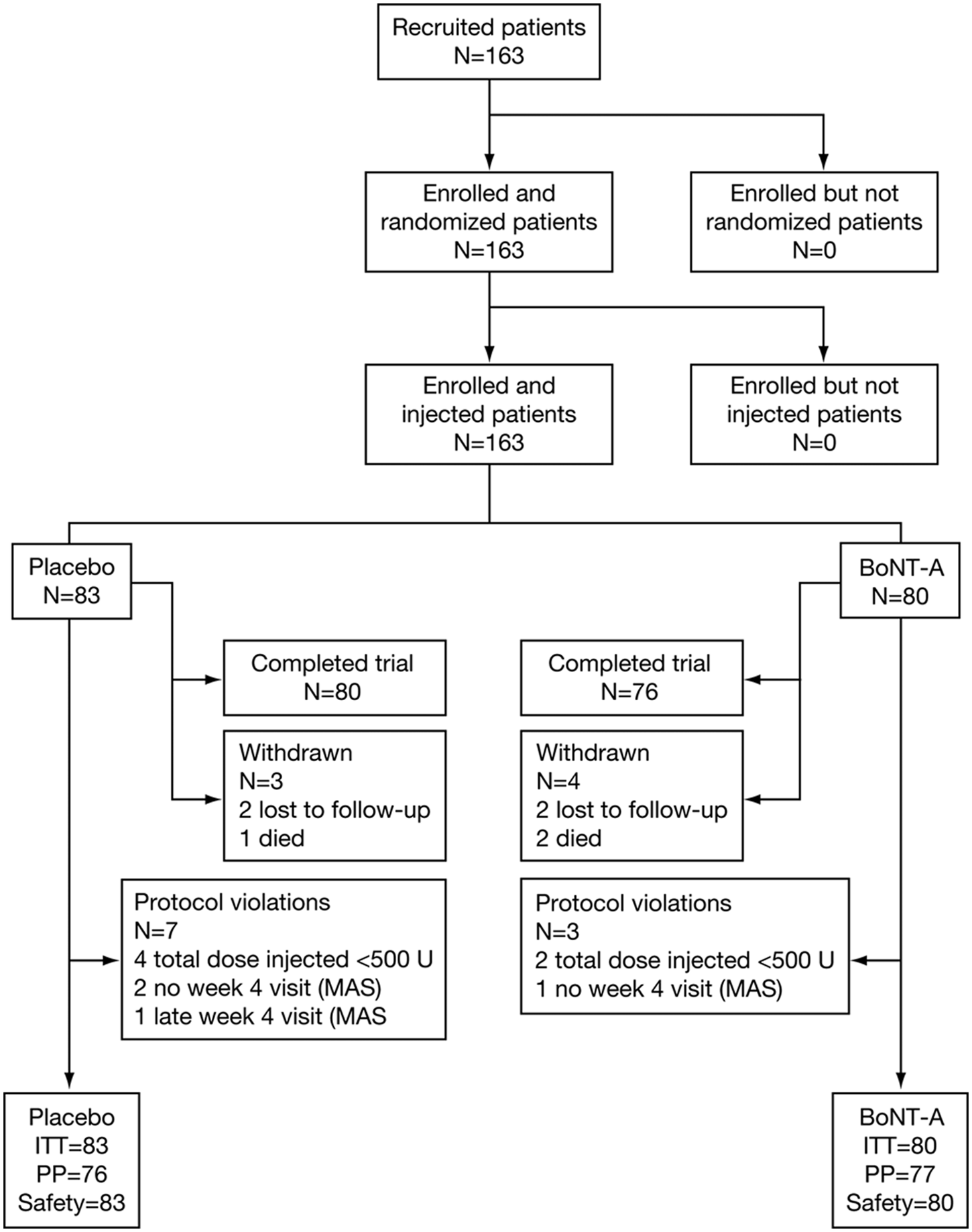
Disposition of patients. Abbreviations: BoNT-A, botulinum neurotoxin type A; ITT, intent to treat; MAS, Modified Ashworth Scale; PP, per protocol.
Distribution of Placebo or BoNT-A Doses
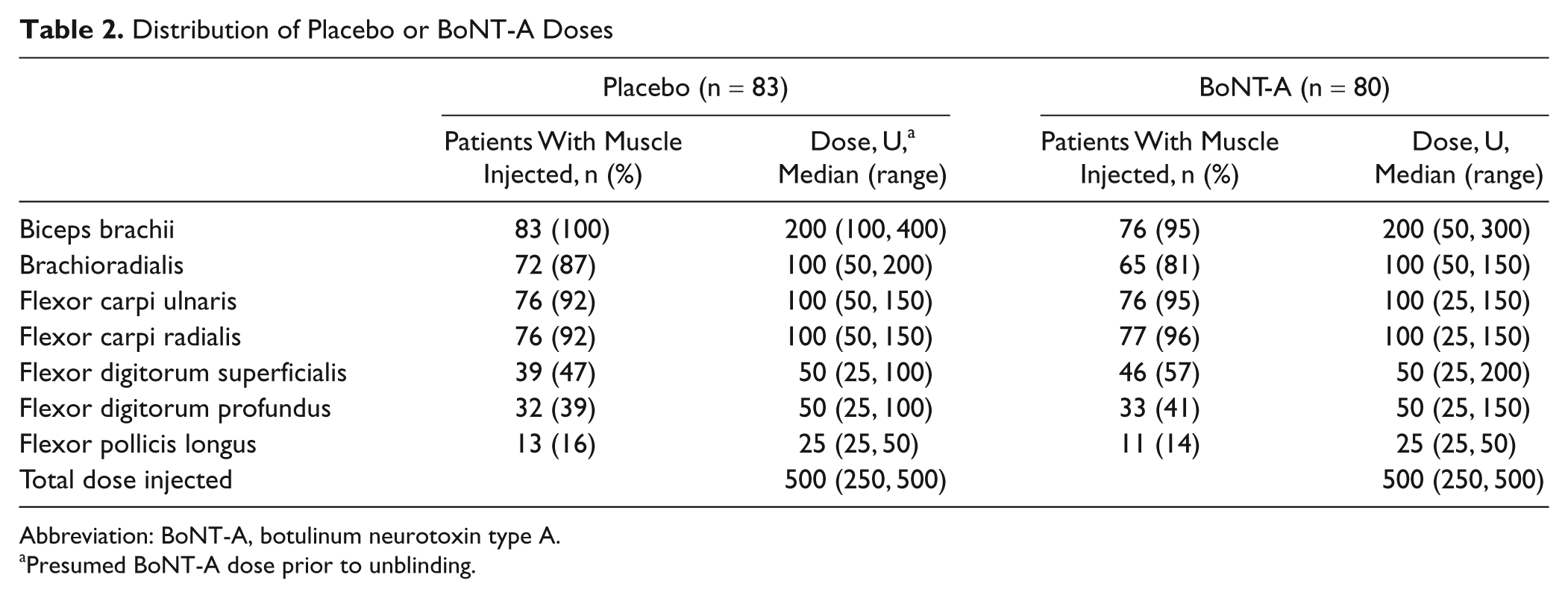
Abbreviation: BoNT-A, botulinum neurotoxin type A.
Presumed BoNT-A dose prior to unblinding.
Primary Efficacy Analysis
Mean (SD) MAS scores 4 weeks after injection were 1.73 (0.77) and 0.96 (0.77) for the placebo and BoNT-A groups, respectively. Based on the primary efficacy parameter, ANCOVA showed no significant interaction of treatment by country, so the model was simplified to include only treatment, baseline MAS score, and treatment by baseline score interaction. The model showed a significant interaction of treatment by baseline MAS score (
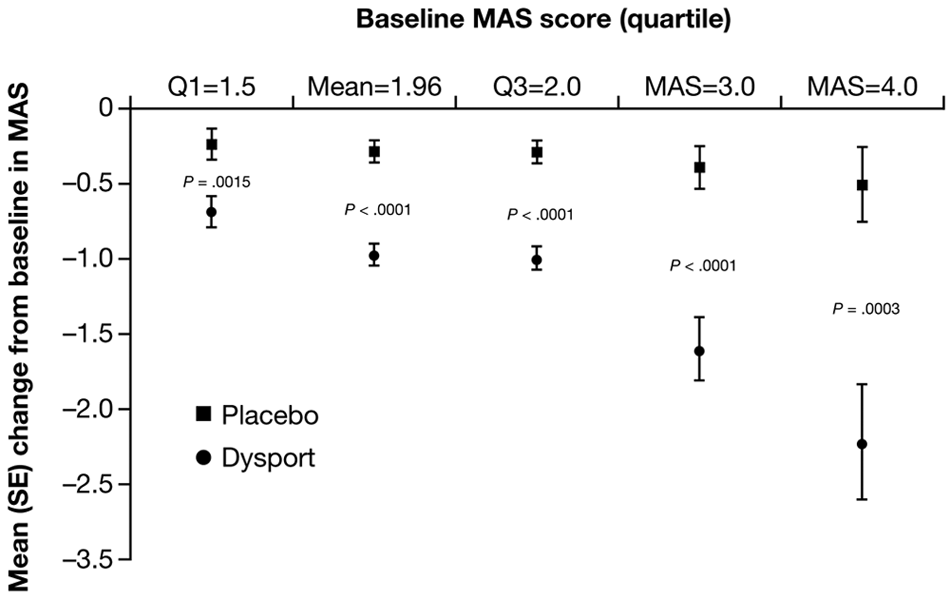
Predicted mean (standard error [SE]) change from baseline in Modified Ashworth Scale (MAS) score (most affected joint) according to baseline MAS score at week 4, intent-to-treat population (primary end point; MAS score 1+ coded as 1.5).
The robustness analysis showed that the treatment effect estimates were not sensitive to the coded value of MAS 1+ (whether coded as 1.1, 1.5, or 1.9). Subgroup analyses showed that type of stroke (bleed vs infarct) and time to treatment after stroke onset (<7 vs ≥7 weeks), did not interact with treatment (
Secondary Efficacy Analyses
Secondary efficacy analyses are presented in Table 3. MAS scores for most-affected joint significantly improved with BoNT-A versus placebo at all time points throughout the 24-week study (
Secondary Efficacy Outcomes a
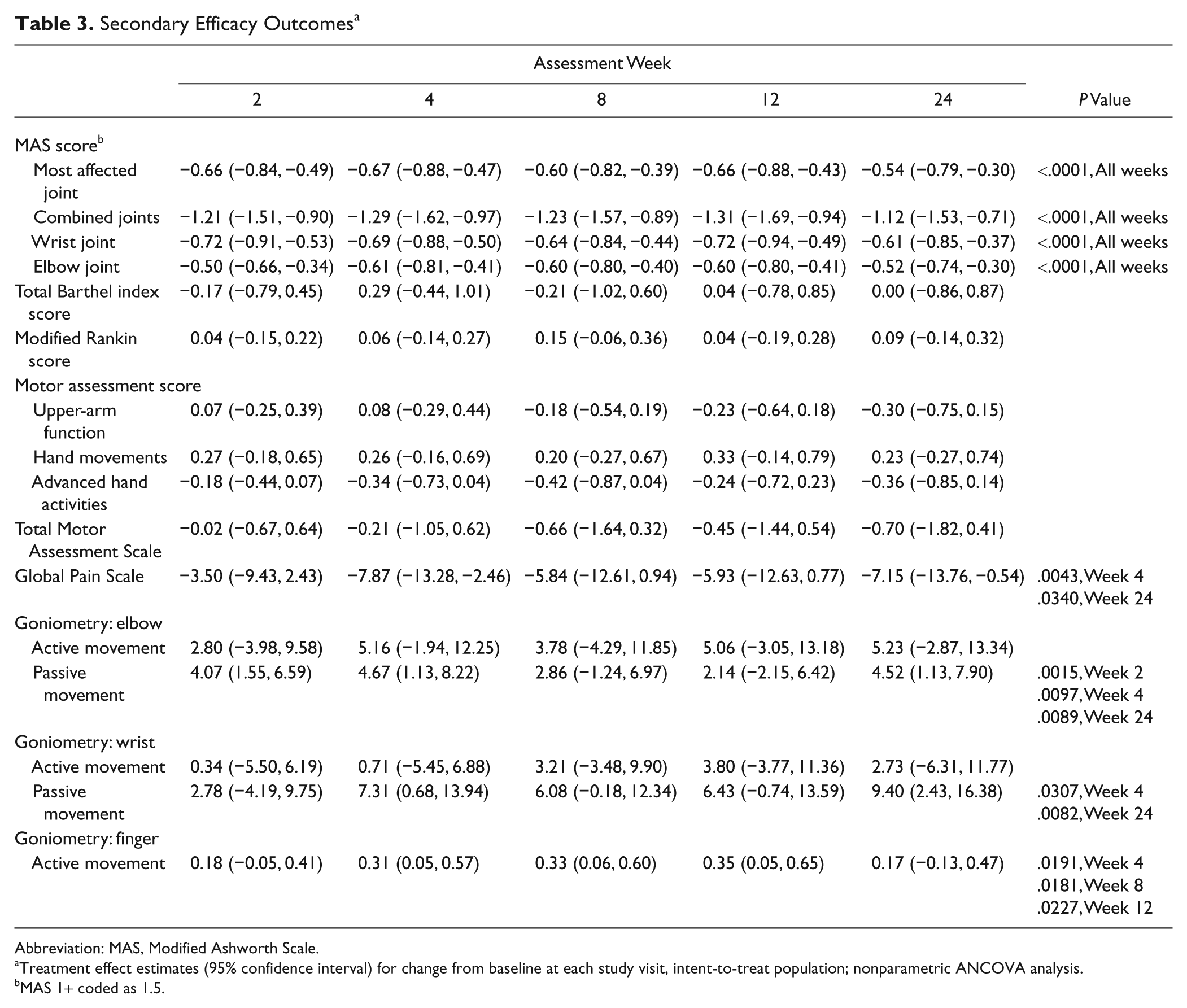
Abbreviation: MAS, Modified Ashworth Scale.
Treatment effect estimates (95% confidence interval) for change from baseline at each study visit, intent-to-treat population; nonparametric ANCOVA analysis.
MAS 1+ coded as 1.5.
No significant changes were seen in the Barthel index, modified Rankin scores, or Functional Motor Assessment Scale scores. Passive but not active elbow and wrist range of motion showed significant improvement at several time points (Table 3), and active finger movements (hand closed) were improved at weeks 4, 8, and 12.
Safety
A total of 84 AEs were reported: 31 serious and 53 nonserious. There was no clinically relevant difference in the distribution of AEs (36 [43%] in the placebo group and 48 [57%] in the BoNT-A group) or of serious AEs (13 [15%] in the placebo group and 18 [21%] in the BoNT-A group). None of these serious AEs was considered to be related to treatment. The most frequent AEs for the placebo and BoNT-A groups, respectively, were dizziness (4 events in 4/83 patients; 4 events in 3/80 patients), arthralgia (2 events in 2/83 patients; 3 events in 3/80 patients), shoulder pain (2 events in 2/83 patients; 2 events in 2/80 patients), recurrent stroke (1 event in 1/83 patients; 2 events in 2/80 patients), and urinary tract infection (1 event in 1/83 patients; 2 events in 2/80 patients). There were 6 AEs (6%) that were considered related to treatment: 2 in the placebo group (dysuria and complex regional pain syndrome) and 4 in the BoNT-A group (fatigue [2 events], pyrexia, and muscular weakness). There were 3 deaths: 1 in the placebo group (lower-respiratory tract infection) and 2 in the BoNT-A group (cerebral hemorrhage/coma and multiple infections/cerebrovascular accident). None was considered related to treatment.
Discussion
This is the first trial showing that BoNT-A with physiotherapy is effective and safe in the treatment of early poststroke spasticity of the upper extremity.
BoNT-A was more effective than placebo in improving MAS scores at 4 weeks after injection, but no change in functional use of the upper extremity, which was a secondary outcome, was found. Even though patients entered the study with moderate spasticity (median MAS score of 2), a significant treatment effect was shown across the whole spectrum of baseline MAS scores. Efficacy was greatest in those with a higher MAS score at baseline, as seen by the treatment effect size in patients with a baseline MAS score of 4. Based on a MAS score of 2, our observations of treatment effect sizes are in accordance with a meta-analysis on botulinum toxin for chronic poststroke spasticity.
26
The efficacy of BoNT-A was independent of both stroke type (bleed or infarct) and time of treatment (<7 or ≥7 weeks poststroke). Secondary efficacy outcomes on combined joints and individual joint MAS scores show that this improvement is sustained up to
We also found that BoNT-A is effective in reducing spasticity-related pain at weeks 4 and 24. In terms of its prevalence as well as its intensity, spasticity-related pain was generally not a major problem in our patient cohort, hence the small treatment effects of BoNT-A. The long-term effect of pain reduction in spasticity may be a result of the avoidance of complications of spasticity, such as spasm and contracture, or perhaps as an independent pain mechanism. 30
As anticipated, improvement in range of motion reflects improvements in MAS score and spasticity-related pain throughout the 24 weeks of the study. Although the changes in passive goniometry for elbow, wrist, and fingers were statistically significant at several time points, the changes in active movement were not. No functional gains were seen despite a reduction in muscle tone; other components of the upper–motor neuron syndrome, such as weakness and altered motor control, are probably more important in determining upper-limb function.
Pharmacodynamically, one would expect to see effects of BoNT-A lasting for 3 to 4 months. 31 Although our trial did not find improvements in motor control following BoNT-A, the unexpectedly long duration of effect on muscle tone in our study may be a result of early modification of changes in the rheological properties of spastic muscles. Further potential implications of early treatment may be related to an alteration in the natural history of the disorder, possibly taking advantage of BoNT-A effects beyond the known peripheral mechanisms,32-36 or by facilitating an improved response to concomitant acute physiotherapy.11,37 Systematic reporting of the number and type of physiotherapy sessions for the affected limb received by each patient was not performed in our study. So, as with many other similar studies, confounding effects associated with nonspecific rehabilitation therapy cannot be ruled out. There is always a possibility that patients who responded to BoNT-A therapy may have received a different physiotherapy regimen to those who did not. However, because this was a double-blind trial, a systematic difference in concomitant physiotherapy is unlikely.
Two of the measurements of function used in this study—the Barthel Index and Rankin scale—did not show any significant improvements in patients treated with BoNT-A, almost certainly because these scales are not sensitive enough to detect affected upper-extremity functional improvement. 18 The failure to demonstrate significant improvement in the Motor Assessment Scale suggests that a reduction in muscle tone does not necessarily lead to a change in functional outcome, as observed in other studies. For example, a small pilot study of very early intervention (3 weeks poststroke) in 30 patients who were unable to score the maximum on the easiest test in the grasp subsection of the Action Research Arm Test found that low (half or quarter)-dose BoNT-A gave no overall benefit to arm function at the 20-week end point compared with placebo. 11 It is interesting to note that in a subanalysis of patients with no arm function at baseline (ie, 3 weeks poststroke), there was some functional recovery at 20 weeks in the groups that received BoNT-A. The authors of this study speculated that altering the time course of spasticity development may influence functional outcomes, and that to test their theory fully, a trial including 101 patients in each arm would be necessary to detect an effect size of 0.5 on the Action Research Arm Test. 11
The lack of improvement in active function of the upper limb despite reduction in spasticity was also observed in the Botulinum Toxin for the Upper Limb after Stroke (BoTULS) trial 38 for predominantly chronic stroke patients. Despite an improvement in basic arm tasks (ie, hand hygiene and dressing), the BoTULS investigators concluded that BoNT-A applied more than 1 month after onset of upper-limb spasticity is unlikely to be useful for improving active upper-limb function (eg, reaching and grasping). 30 Shaw and associates 30 argue that although spasticity is an important component of reduced upper-limb function, motor weakness is the most important factor.
Assisted hygiene and dressing were end points in a study by Brashear et al 39 but were not specifically sought in our trial. In the Brashear et al study, patients with symptomatic upper-limb spasticity were asked to define upper-limb goals on the Disability Assessment Scale before BoNT-A or placebo injection, as was done in 2 other studies.40,41 The results were positive for BoNT-A groups; predefined subjective rather than objective functional outcome measures may reveal benefits associated with patient satisfaction and perceived functional improvement.
BoNT-A was well tolerated at the doses used in this study and did not induce any undue muscle paralysis, which is consistent with the doses applied in previous studies of upper-limb spasticity with Asians.42,43 There was only 1 report of muscle weakness among our BoNT-A-treated patients, though because weakness was common, it may not have been recognized as an AE. Study limitations include the choice of the Barthel index, the Modified Rankin Scale, and the Functional Motor Assessment Scale as functional assessment scales. The trial was not specifically powered to compare the effects on clinically meaningful outcomes. Also, the nonstandardized rehabilitation therapy across sites allows the possibility of a differential effect of these programs.
Although most prior trials did not include physiotherapy, 38 guidelines now consider that BoNT-A plus physiotherapy is first-line treatment for poststroke spasticity, 8 and BoNT-A in combination with constraint-induced movement therapy was effective in patients with chronic stroke. 44 Based on our study, patients with MAS 1+ seem unlikely to benefit from prophylactic BoNT-A to improve arm and hand function. A longer follow-up may have yielded further information on trends for spasticity over time.
This is the first well-designed, placebo-controlled study of the use of BoNT-A in stroke patients within 3 months of first stroke and is the first study to show that BoNT-A (Dysport) 500 U can significantly reduce poststroke upper-limb spasticity in Asian patients treated within 2 to 12 weeks of first stroke. As in most prior trials, we could not show any effects on voluntary upper-extremity functioning. The effects on tone were sustained for up to 24 weeks, which suggests the possibility that early treatment may prevent the development of more severe spasticity.
Footnotes
Acknowledgements
This was an Ipsen-sponsored study (Ipsen Pharma, Boulogne Billancourt, France). The sponsor provided logistical support, study drugs, support for investigator meetings, central statistical support, and data analysis. The investigators had full access to the data, and the corresponding author had final responsibility for the decision to submit the manuscript for publication. The data analysis was performed by the Clinical Research Center of Ministry of Health of Malaysia under supervision from the sponsor. Peter Moore, The Walton Centre NHS Foundation Trust, Liverpool, UK, and Andrew Hughes, Austin Hospital, Melbourne, Australia, are thanked for their valuable comments on data interpretation and useful discussions. The authors take full responsibility for the content of the article but thank Jackie Marchington, PhD, Caudex Medical, Oxford, UK (supported by Ipsen Pharma), for assistance in preparing the initial draft of the manuscript, collating the comments of authors and other named contributors, assembling tables and figures, and coordinating submission requirements.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RLR received payments as principal investigator of this study, honoraria for consultancy, and travel support for congress attendance from Ipsen Pharma; KHK received institutional funding in support of this study, travel support to present findings of this study, and honoraria for advisory boards for this study from Ipsen Pharma; KJG received institutional funding in support of this study, travel support fees for investigator meetings and data monitoring boards for this study, honoraria for lectures, and travel support for congress attendance from Ipsen Pharma; WK received institutional funding and travel support for investigator meetings for this study and honoraria for lectures and travel support for congress attendance from Ipsen Pharma; VCTM received institutional funding for a research assistant and honoraria for investigator meetings related to this study from Ipsen Pharma; MMD-DLS received travel support for congress attendance from Ipsen Pharma; KSGC has nothing to disclose; SJbFA received institutional funding in support of this study, travel support for investigator meetings, and fees for data monitoring boards for this study and travel support for congress attendance from Ipsen Pharma; BZ, PM, and AM are employees of Ipsen Pharma; KSLW received travel support to attend investigator meetings from Ipsen Pharma.
Funding
The author(s) received financial support for the research from Ipsen Pharma, as detailed in the Declaration of Conflicting Interests. The authors received no financial support for authorship, and/or publication of this article. Medical writing support as described in the Acknowledgements was funded by Ipsen Pharma.
