Abstract
The second Stroke Recovery and Rehabilitation Roundtable “metrics” task force developed consensus around the recognized need to add kinematic and kinetic movement quantification to its core recommendations for standardized measurements of sensorimotor recovery in stroke trials. Specifically, we focused on measurement of the quality of upper limb movement. We agreed that the recommended protocols for measurement should be conceptually rigorous, reliable, valid and responsive to change. The recommended measurement protocols include four performance assays (i.e. 2D planar reaching, finger individuation, grip strength, and precision grip at body function level) and one functional task (3D drinking task at activity level) that address body function and activity respectively. This document describes the criteria for assessment and makes recommendations about the type of technology that should be used for reliable and valid movement capture. Standardization of kinematic measurement protocols will allow pooling of participant data across sites, thereby increasing sample size aiding meta-analyses of published trials, more detailed exploration of recovery profiles, the generation of new research questions with testable hypotheses, and development of new treatment approaches focused on impairment. We urge the clinical and research community to consider adopting these recommendations.
Introduction
The worldwide prevalence of stroke in 2010 was 33 million, with 16.9 million people having a first stroke. 1 Approximately two-thirds of patients have upper limb limitations, with only 5 to 20% demonstrating full recovery at six months post stroke. 2 For individuals and society at large, this has important implications as reduced upper limb capacity is associated with dependence in activities of daily living (ADL) and poor quality of life for both patients and their carers. 3
Upper limb recovery probably occurs through a combination of spontaneous and learning-dependent processes, including both behavioral restitution (i.e. restoring the quality of movement), and behavioral compensation in which patients learn ways to use their residual capacity in the most functional way to accomplish a task.4 –7 Spontaneous neurological recovery is by far the greatest contributor to behavioral restitution and mainly occurs over the first eight weeks post-stroke, 8 and in most patients much earlier. 9
Clinical trials and observational studies have failed to effectively distinguish behavioral restitution from behavioral compensation, leaving the association between recovery of movement quality and recovery of upper limb capacity underexplored. Therefore, it is of great scientific and clinical interest to determine how behavioral restitution might be augmented by pharmacological and behavioral interventions in the first months after stroke to optimize poststroke rehabilitation. Critically, here we argue that accurate and responsive measurement of movement quality is needed to discover such interventions and assess them in subsequent trials.
In a first consensus meeting of the Stroke Recovery and Rehabilitation Roundtable (SRRR) group, we agreed on a number of definitions with respect to stroke recovery. 6 In addition, we achieved consensus about standardized clinical measurements with COSMIN statements provided (https://www.cosmin.nl/) following the International Classification of Functioning and Disability model (ICF) and the time points for their collection. We argued that these recommendations be included in sensorimotor recovery and rehabilitation trials.6,10 However, we also agreed that standard clinical measures do not adequately capture movement quality and therefore are insensitive to detecting whether changes are due to behavioral restitution or compensation. 10 Technologies allowing objective measurement of movement kinematics and kinetics were suggested as the best way to tackle this problem. 10 More importantly, fine-grained measurement of motor control is vital to understand neural repair processes and their interaction with behavioral training during stroke recovery. 11 In particular, understanding how heightened levels of neuroplasticity augment the response to rehabilitation training early post stroke is crucial for trials seeking to test interventions that target brain repair.4,12
Currently, consensus is lacking on the use of kinetic and kinematic measures (metrics) for motor recovery. After reviewing 225 studies, Schwartz et al.
13
found 151 different metrics used for measuring upper limb movements. In most kinematic studies, the functional task was not standardized nor was the device or equipment validated for its psychometric properties.
13
In addition, as Krakauer and Carmichael
14
recently emphasized, the choice of kinematic measures is critical; some are easily contaminated by compensation. To date, research on how detailed kinematic measures reflect behavioral restitution and neuronal reorganization processes early post stroke is limited. For example, there have been few attempts to track motor recovery longitudinally using kinematics.15–18 These studies can be considered kinematic updates of Twitchell’s classic 1951 paper,
19
which presented meticulous clinical observations of a series of patients that he followed from hospital admission to when they reached a stable condition.
19
Instead, most trials have proceeded using relatively crude clinical measures that do not provide detailed analysis of the recovery of movement quality.
20
In the second SRRR consensus meeting, three key research questions were posed for achieving consensus on the use of metrics for measuring the quality of movement: Which performance assays of the paretic upper limb should be used to address questions about the quality of upper limb movement execution at the ICF level of body function? Which functional task(s) should be recommended to measure quality of upper limb movement execution at the ICF level of activities? Which types of technology equipment (e.g. optoelectronic, electromagnetic movement tracking systems) are recommended for measuring movement during performance assays and functional tasks?
Consensus Building
Similar to the first SRRR meeting, experts were invited to take part in the kinematic consensus process based on their publications on upper limb kinematics after stroke: either peer-reviewed original articles or reviews.
Consensus recommendations were reached via a five-stage process (Figure 1). In stage 1, chair (GK) and co-chairs (JB and EVW) created a survey comprising 13 Yes/No, multiple-choice and open questions seeking expert view on the types of movement and task that should be assessed; choice of motion capture tool, including their psychometric properties, data sampling and feasibility; movement metrics; and timing of assessment. In August 2018, the “CORE” group of experts (N = 5) and an “ADVISORY” group (N = 8) completed the survey. In Stage 2, GK, JB and EVW, who did not take part in the survey, collated survey responses to form the framework for Stage 3 (available from GK); the two-day consensus meeting of the CORE group in Saint-Sauveur in Canada last year. The CORE group first achieved consensus on terminology and definitions, compiling a glossary for terms such as quality of movement, motor control, behavioral restitution, and compensation. Subsequently, we discussed and achieved consensus on a set of performance assays and a functional task, and on their standardized use. Third, the metrics for the performance assays and the functional task, as well as the types of technologies that could be used to implement them, were agreed upon. Finally, we achieved consensus on: (1) When post-stroke to acquire kinematic measurements; (2) the most important measurement properties of assays; (3) upper limb movement types (including trunk movements); (4) the preferred movement-sampling frequency; (5) the minimal number of movement repetitions; (6) the periods of rest between repeated movements, as well as; (7) experts’ opinions about testing the less affected arm early post stroke. Outputs from the consensus meeting were: (a) an audio recording and verbatim transcription of the discussions, (b) a framework on which to build the recommendations and, (c) a recommendations document.

Flow chart of the different stages for achieving consensus between 16 experts involved in the process.
Between October 2018 and January 2019, the CORE group members synthesized the first draft of the consensus paper. In February 2019, the second draft was distributed to the CORE group for review, resulting in a prefinal draft. Stage 5 was held in March 2019 during which the ADVISORY group was asked to review and provide their feedback to the pre-final version of the manuscript.
Recommended Biomechanical Measures
Panel 1 (Supplemental Material) shows the consensus-based definitions for terms that we used in our discussions with respect answering how to test quality of movement.
How to Test Quality of Movement?
The metric task force agreed that quality of movement should be assessed in two ways using:
(A)
We recommend at least 15 trials per target for the 2D performance assays and 3D functional tasks. There are three main reasons for this: (1) We are often interested in the mean and variance of movements; the latter requires more movements than the former for a good estimate, (2) In the absence of knowing the test–retest reliability of a measure, it is better to err on a higher number of repetitions and, (3) underpowered comparisons can over-estimate effect sizes.
Participants need to receive sufficient rest between the movements to minimize the risk of fatigue that might affect the quality of movement. In case of fatigue, the requested movement can be executed in blocks of three or five trials with rest periods between trials. A minimal change (improvement or decline) of approximately 15% in performance based on 2D- or 3D metrics will be considered as a clinically important difference.21,22 However, an absolute value is preferred to signify a clinically important change for any given metric and task.
Acknowledging that spontaneous neurological recovery is a dynamic process that occurs within the first 12 weeks post stroke,2,8 we recommend that fixed time points for kinematic and kinetic assessments be obtained. As a minimum, at: 1, 12, and 26 weeks after stroke onset6,10; consistent with the first SRRR consensus statements. 10 In addition, to better identify and quantify the logistic time course of underlying mechanisms of spontaneous neurological recovery, additional assessments at four and eight weeks are strongly recommended.
We recommend four performance assays and one functional task: (1) a planar (2D) reaching task; (2) finger individuation; (3) grip strength (force) and (4) precision grip strength. The functional task is a standardized reach and prehension task ‘drinking’ (3D).
Performance Assays
2D-Reaching Assay
The 2D-reaching assay is used to identify the capacity of the participant to coordinate planar movements around more than one joint to produce smooth and accurate reaching trajectories and to maintain a stable endpoint position at the end of the movement (Figure 2). The participant performs a horizontal reaching movement in a transverse plane, requiring coordinated movements of the shoulder and elbow. In the initial position, the participant is seated in a standard chair with back support with feet flat on the floor. The trunk is supported by and strapped to a backrest to constrain sagittal trunk displacement, while allowing for scapular protraction and elevation.
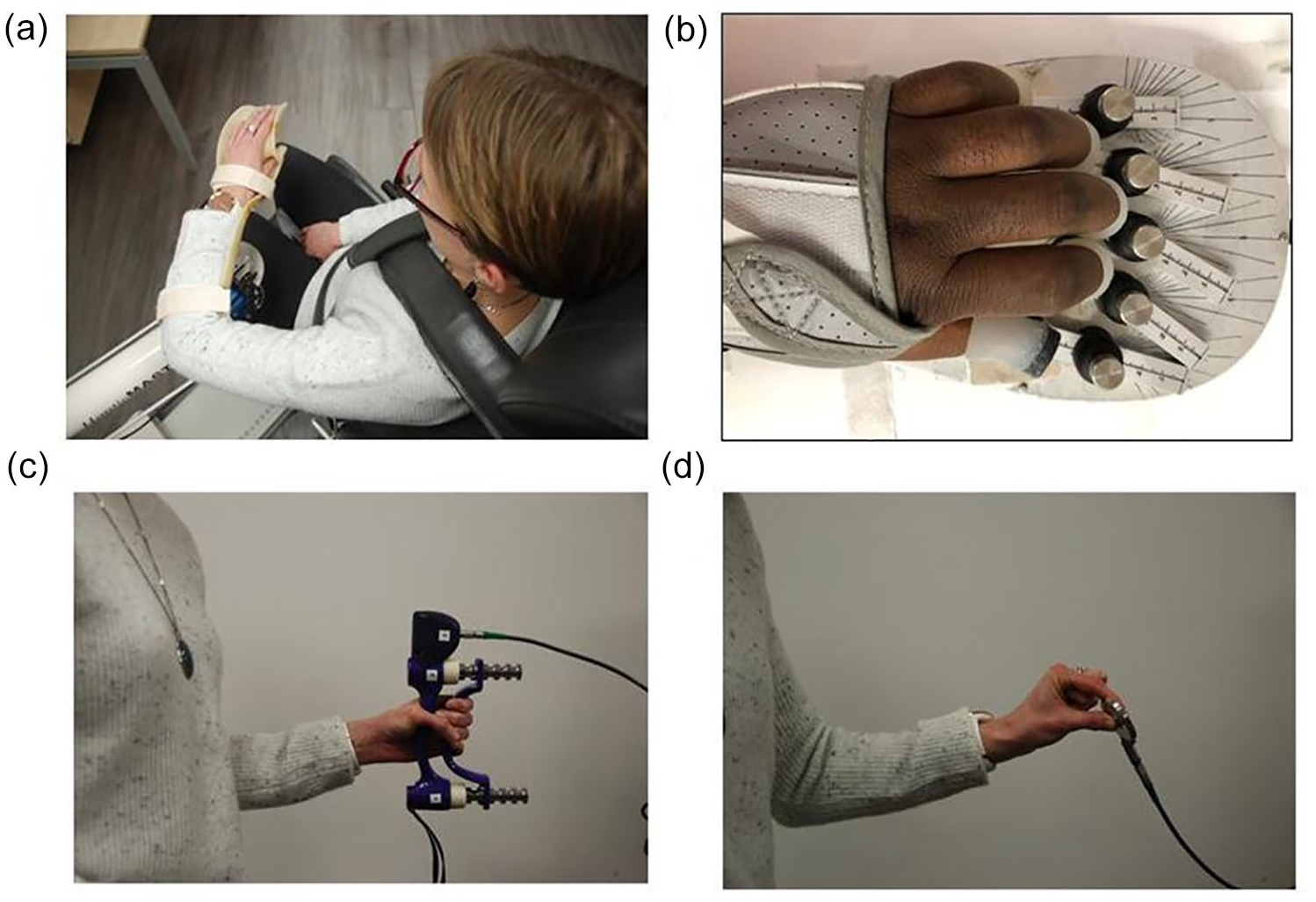
(a–d) Recommended 2D-performance assays. Panel a: The 2D-planar reaching task performed in a Haptic Robot. The participant performs a horizontal, transverse plane movement, requiring co-ordination of the elbow and shoulder. (The nine targets placed in front of the patient at distances of 15–20 cm are not shown.) Panel b. Finger individuation device; The hand is strapped onto an ergonomic hand device capable of measuring isometric forces generated at the fingertips. Force transducers beneath each key dynamic range 0–25 N, allowed for a responsive measurement of the individual finger forces of the instructed hand (see also for details literature 18, 30). Panel c. Testing grip strength (Electronic Hand Grip Dynamometer, Biometrics Ltd, UK) and Panel d. Testing precision grip (Electronic Pinch Dynamometer, Biometrics Ltd, UK).
The arm is raised to a level that allows shoulder and elbow movement in a horizontal (i.e. transverse) plane. In cases of shoulder pain or limited range of motion, a lower arm elevation is advised along with sufficient documentation and performed consistently thereafter, i.e. at each assessment time-point. The forearm and hand are fully supported by a two-joint manipulandum (or haptic robot device).
The initial position is located in the trunk midline at a distance equal to 2/3rd the participants arm length (measured from the superior-lateral border of the acromion to the center of the distal wrist crease (Figure 2, panel a). The shoulder is at ∼70° elevation and adduction, and the elbow is flexed to ∼90°. Different targets can be located 15–20 cm from the center target around the circle. The task is to move the hand from the starting position to the final target upon hearing a ‘go’ signal, within a fixed speed and time window (to allow direct comparison with age-matched non-disabled controls) and to hold the hand in the final position for 1–2 s (to assess the ability to hold still). A minimum of 15 complete reaches from starting position to each of the final targets is recorded with the sequence of reaches randomized. In terms of target number, we recommend a minimum of four to eight reach directions that are equally spaced and arrayed over 360°. This ensures capturing both single and multi-joint movements. 9 Alternatively, the size of the available workspace can be probed without specific individual targets. 23 In this case, however, movement quality per se is not being measured. Kinematics is recorded from the trunk, arm, and endpoint from active or passive markers placed on bony landmarks, with a high-resolution optical tracking system and a minimum sampling frequency of 60 Hz. If inertial measurement units (IMUs) are used, they should only be in conjunction with a camera-based system because there is currently insufficient evidence for their validity, responsiveness, and reliability. In our view, the number of metrics (e.g. peak velocity, endpoint accuracy, curvature, and smoothness) acquired should be limited and carefully selected to avoid a multiple comparisons problem and problems in interpretation (e.g. what does it mean if kinematic variable A is abnormal but B is not?). Instead, an examination of movement trajectories at a global level with a method that is responsive to changes in overall movement quality would be preferable (see recommended list of performance assays in Appendix A online).
Recently, a novel method of trajectory analysis, based on functional principal component analysis (FPCA) was devised to characterize reaching kinematics.16–24 This approach extends principal component analysis to time-series data.16,25–28 The two main benefits are: (1) It examines the entire trajectory, and shows responsiveness for change undetected by conventional analyses, and does not focus on preselected measures such as end-point accuracy or peak velocity. (2) A direct comparison to controls can be made by computing a difference score of each movement trajectory relative to a non-disabled reference population.27,28
Finger Individuation
The finger individuation assay is used to determine the ability of participants to isolate movement of a single finger whilst keeping the others still. Two indices quantify two different aspects of digit independence: individuation index and the stationarity index.29,30 One way to assess finger individuation is using a “data glove” or “cyber glove” with sensors that capture bending or angular displacement of pre-specified finger joints (Figure 2, panel b). A drawback of the glove approach is that it relies on forces that are not directly measured or controlled for. 31 To address this problem, isometric devices have been designed that capture similar indices as those above, while a participant exerts differing degrees of isometric force with their fingers. An advantage of being able to measure force is that weakness can be factored out as a confounding contributor to an individuation deficit. 18
Grip Strength Using Calibrated Dynamometer (Kinetic)
Grip strength should be measured in a seated position with a hand-held dynamometer (Figure 2 panel c). The participant is asked to hold the dynamometer with the elbow flexed at 90° by the side of the body. If needed, the elbow can be supported on a table. The forearm is in a neutral position, wrist between 0° and 30° dorsiflexion and between 0° and 15° ulnar deviation. The handle of the dynamometer is adjusted to an appropriate grip width if required – the base should rest on the first metacarpal (heel of palm), while the handle should rest on middle of the four fingers. When ready, the participant squeezes the dynamometer with maximum isometric effort for about 5 seconds. The participant should be strongly encouraged to give a maximum effort. A mean of three successive trials with 30 seconds rest in between the attempts is recommended. 32 Normative values are available. 33
Precision Grip Between Thumb and Index Finger Using a Calibrated Dynamometer (Kinetic)
The participant is seated (see A3) and instructed to pinch the force transducer between the thumb and index finger (Figure 2 panel d). If needed, the elbow can be supported on a table. The pinching force should be tested three times with appropriate rest periods (e.g. 30 seconds) to avoid fatigue. Support or aid from the other fingers invalidates the trial. Similar standardization as for grip strength should be used for the precision grip between thumb and index finger force. 33 Normative values are available stratified by the age, sex and hand dominance. 33
Functional Task
The goal is to assess the ability of the participant to generate the requisite trajectory and joint kinematics of the upper limb to accurately perform a functional task. Figure 3 (panels a–e) and Appendix B (online) demonstrate the test protocol, equipment and metrics recommended for the 3D reach-to-grasp task performed within a natural context. Although the goal is meant to be functional (i.e. picking up a cup to take a drink), the required movement depends on the participant’s capacity at the body function level of the ICF. While recognizing the effect of goal and context on task performance,34,35 e.g. reaching with versus without grasping, the question of whether, and to what degree, this impacts motor performance changes over time remains to be determined. The emphasis here is on the quality of movement within a functional task and not ICF activity per se.
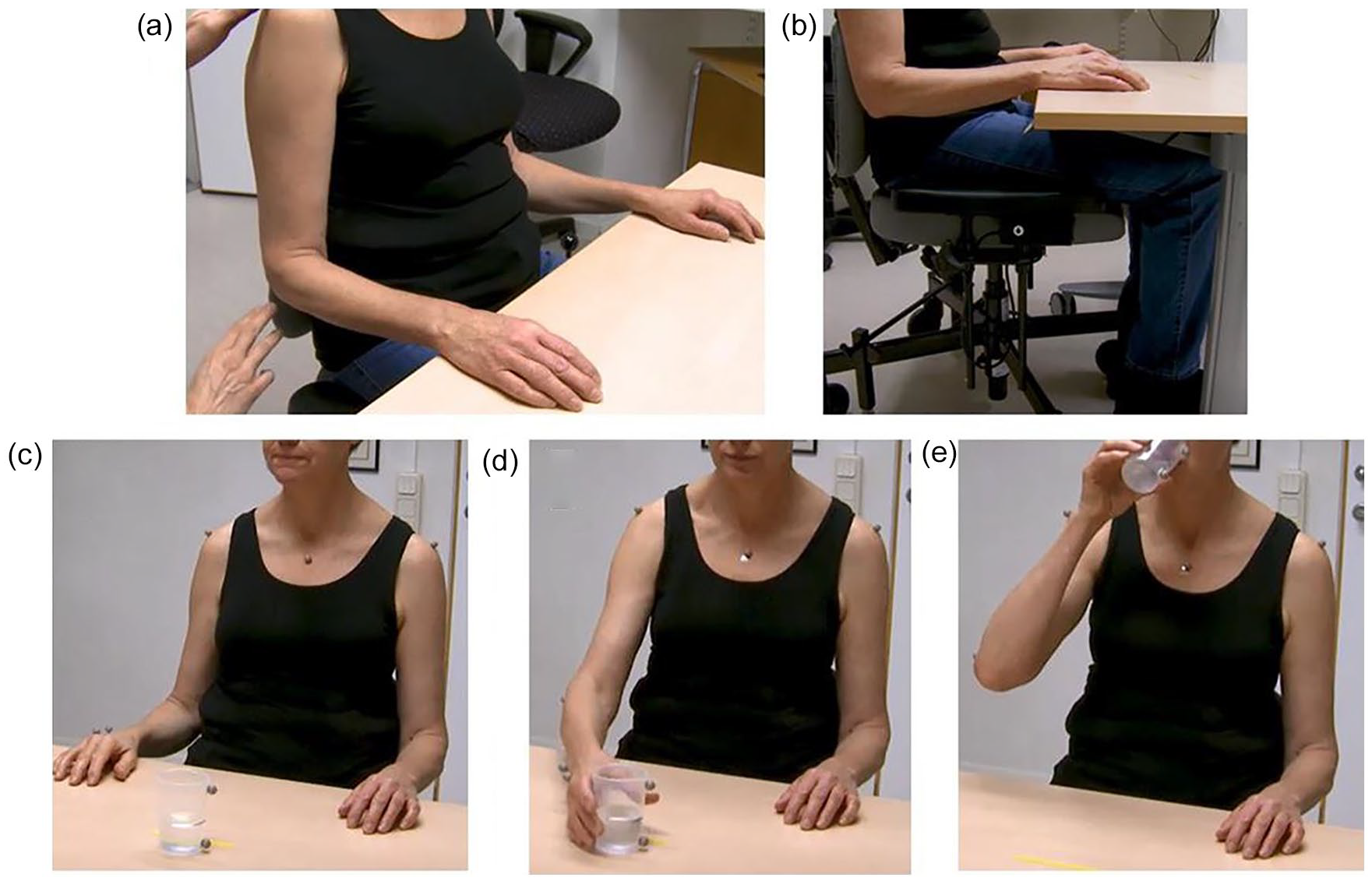
Recommended 3D-functional task. Panels a and b: Sitting position and Panel c start and end position. Panel d: Illustration of the end of the reach phase, while panels d and e present grasping the cup (d) and drinking phase, respectively.
While obtaining reliable and valid 3D movement-quality kinematics during a functional task can be challenging, the drinking task was chosen because it is already learned, it can be easily standardized, and is accomplished with minimal investment in motion tracking equipment. The post-stroke properties of the kinematics of drinking task are well established. 22
Appendix B (online) describes the test protocol, equipment and metrics 22 for the drinking task. The execution of a task including grasping and drinking is recommended for people with moderate to mild hemiparesis (i.e. Fugl-Meyer Assessment scores of the Upper Extremity (FM-UE) ≥32 points out of 66). 21 For individuals with a lower level of body function, the task can be modified (e.g. stabilizing the cup with the other hand while grasping, or using a different cup design that is easier to grasp). The protocol should always be calibrated for each individual to correct for different body sizes. Appendix C (online) provides the reference metrics in non-disabled controls.
Recommended Motion Capture Equipment
The metric task force agreed that, given the current maturation of the technology, only high speed and high-resolution digital optoelectronic systems should be used to measure kinematics during the performance assays and the functional task. In contrast, wireless wearables including 2D and 3D-IMU’s, as well as Kinect or other optical systems are deemed currently inadequate for measuring the quality of movement. We agreed that more evidence for their reliability, responsiveness, and validity is needed.
Discussion
The key message of the metric group is that brain repair maps best onto fine-grained movement quality measures that are sensitive and specific, i.e. able to capture small but true behavioral changes. Until now, there has been no consensus on how to use kinematics and kinetics to achieve this goal. 10 The development of recommendations to help standardize measurement of movement quality will improve the design and interpretation of future stroke recovery trials.
Consensus on measuring the quality of movement is imperative not only for stroke recovery and rehabilitation trials but also for allowing proper interpretation of neuroimaging studies (e.g. fMRI, DTI). Specifically, only by measuring the quality of movement is it possible to distinguish neural changes associated with behavioral restitution from compensatory strategies. This granularity of behavioral measurement is the only way that neuroimaging can potentially make a useful contribution to neurorehabilitation.4,5
We emphasize that these recommendations first need to be implemented in observational studies and Phase II trials, before being applied in Phase III trials. Indeed, the less than impressive results of Phase III trials to date may be attributed in part to the lack of preliminary mechanistic studies that generated kinematic and kinetic data about the post-stroke recovery trajectory. In addition, the proper investigation of changes in quality of movement both in natural history and interventional studies requires a translational perspective, executed by a multidisciplinary team of biomedical engineers, movement scientists, neuroscientists, computer scientists, and statisticians working with clinicians in the field of stroke rehabilitation and recovery. Further, we are of the opinion that this type of complex research should be restricted only to those centers that have access to the requisite expertise. Therefore, in order for neurorehabilitation research to reach scientific maturity, specialized centers are required as in other medical disciplines. Neurorehabilitation should no longer be seen as a pragmatic minimalist specialty.
The metric task force limited its current recommendations for measuring quality of movement to the paretic upper limb. This manuscript may, however, serve as a blue print for capturing recovery of the lower limb using kinetics and kinematic measures.
Recommendations
By lack of current consensus, there is an urgent need to measure quality of movement in stroke recovery and rehabilitation trials to understand what patients learn and how they improve their upper limb capacity early post stroke.
We recommend to use the principles derived from motor control as a framework for measuring quality of movement.
We recommend to measure the standardized 2D-reaching assay, finger individuation, pinch- and grip strength for assessment of behavioral restitution.
We recommend to use the standardized 3D-reach-to-grasp drinking task for measuring recovery of upper limb capacity.
The recommended 2D-reaching assays and 3D-drinking task should be measured repeatedly at fixed times post stroke concomitant with the recommended clinical measurements of outcome.
The metric task force strongly recommends that only high-resolution digital optoelectronic systems be used to measure both performance assays and functional tasks. Only people who have the expertise and access to these technologies should therefore conduct quality of movement assessment.
Supplemental Material
sj-pdf-1-nnr-10.1177_1545968319886477 – Supplemental material for Standardized Measurement of Quality of Upper Limb Movement After Stroke: Consensus-Based Core Recommendations From the Second Stroke Recovery and Rehabilitation Roundtable
Supplemental material, sj-pdf-1-nnr-10.1177_1545968319886477 for Standardized Measurement of Quality of Upper Limb Movement After Stroke: Consensus-Based Core Recommendations From the Second Stroke Recovery and Rehabilitation Roundtable by G. Kwakkel, E. E. H. van Wegen, J. H. Burridge, C. J. Winstein, L. E. H. van Dokkum, M. Alt Murphy, M. F. Levin, J. W. Krakauer, Catherine E Lang, Thierry Keller, Tomoko Kitago, Nurdiana Nordin, Valery Pomeroy, Janne M. Veerbeek and Frederike van Wijck in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank the ADVISORY group [Catherine E Lang (Physical Therapy, Occupational Therapy, Neurology Washington University School of Medicine, St. Louis, MO USA), Thierry Keller (Neurorehabilitation Department, Health Division, Tecnalia Research & Innovation, San Sebastian, Spain), Tomoko Kitago (Burke Neurological Institute and Department of Neurology, Weill Cornell Medicine, USA), Nurdiana Nordin (Centre for Robotics and Industrial Automation, Fakulti Kejuruteraan Elektrik, Universiti Teknikal Malaysia Melaka, Malaysia), Valery Pomeroy (School of Health Sciences, University of East Anglia, Norwich, United Kingdom), Janne M. Veerbeek (Division of Vascular Neurology and Neurorehabilitation, Department of Neurology, University Hospital Zurich, University of Zurich, Switzerland; Cereneo, Center for Neurology and Rehabilitation, Vitznau, Switzerland) and Frederike van Wijck (School of Health and Life Sciences, Glasgow Caledonian University Cowcaddens Road, United Kingdom)] for being part of the external advisory group for this working group. We would like to acknowledge Julie Bernhardt for convening the second Stroke Rehabilitation and Recovery Roundtable (SRRR II), Dale Corbett and Karen Borschmann for organizing the meeting and Farrell Leibovitch for moderating the overall discussions within the different SRRR groups. We like to thank Alejandro Melendez-Calderon for his suggestions.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclose receipt of the following financial support to conduct this meeting: Canadian Institutes of Health Research (CIHR) CaSTOR (Canadian Stroke Trials for Optimized Results) Group (note that CasTOR is a joint initiative of the Canadian Stroke Consortium and the Canadian Partnership for Stroke Recovery), Heart and Stroke Canadian Partnership for Stroke Recovery, and NHMRC Centre of Research Excellence in Stroke Rehabilitation and Brain Recovery. An unrestricted educational grant was provided by Ipsen Pharma.
Note
This contribution, first published in International Journal of Stroke, is being co-published in the following journals: Neurorehabilitation and Neural Repair.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
