Abstract
Background. The cortical plastic changes in response to median nerve electrical stimulation (MNES) in stroke patients have not been entirely illustrated. Objective. This study aimed to investigate MNES-related changes in effective connectivity (EC) within a cortical network after stroke by using functional near-infrared spectroscopy (fNIRS). Methods. The cerebral oxygenation signals in the bilateral prefrontal cortex (LPFC/RPFC), motor cortex (LMC/RMC), and occipital lobe (LOL/ROL) of 20 stroke patients with right hemiplegia were measured by fNIRS in 2 conditions: (1) resting state and (2) MNES applied to the right wrist. Coupling function together with dynamical Bayesian inference was used to assess MNES-related changes in EC among the cerebral low-frequency fluctuations. Results. Compared with the resting state, EC from LPFC and RPFC to LOL was significantly increased during the MNES state in stroke patients. Additionally, MNES triggered significantly higher coupling strengths from LMC and LOL to RPFC. The interregional main coupling direction was observed from LPFC to bilateral motor and occipital areas in responding to MNES, suggesting that MNES could promote the regulation function of ipsilesional prefrontal areas in the functional network. MNES can induce muscle twitch of the stroke-affected hand involving a decreased neural coupling of the contralesional motor area on the ipsilesional MC. Conclusions. MNES can trigger sensorimotor stimulations of the affected hand that sequentially involved functional reorganization of distant cortical areas after stroke. Investigating MNES-related changes in EC after stroke may help further our understanding of the neural mechanisms underlying MNES.
Keywords
Introduction
Stroke is one of the most common neurological disorders and remains a leading cause of disability, with many survivors experiencing residual functional deficits. 1 Understanding and enhancing effective rehabilitation treatment after stroke is likely to play an important role for functional recovery. 2 Median nerve electrical stimulation (MNES) is a noninvasive method of neuromodulation in which electrical pulses from 2 transcutaneous electrodes (cathode and anode) are transmitted along the wrist to the median nerve. MNES as a form of peripheral nerve stimulation targets the sensory system and can produce strong sensory afferent input. 3 MNES has been shown to improve learning as well as retention of learning in healthy volunteers3,4 and stroke patients.5-7 Previous studies about the effect of MNES mostly were based on behavioral measurements. However, to date, the physiological mechanism of MNES-related changes in behaviors is not clear. The research based on neurophysiological measurements is needed to improve our understanding of the underlying mechanisms of MNES on cortical plasticity in stroke patients.
Functional near-infrared spectroscopy (fNIRS), as a novel and promising functional neuroimaging technique, enables the noninvasive detection of local hemodynamics changes in the cortical surfaces. 8 fNIRS is relatively low cost and easy to set up, with fewer physical/environmental constraints and contraindications. 8 Thus, it is readily applicable in clinical settings to detect hemodynamic fluctuation for brain functional architecture analysis. The brain is organized into a set of widely distributed networks that play a fundamental role in controlling behavior. The integration within a distributed system can be better assessed over entire cerebral networks by effective connectivity (EC), which refers explicitly to the influence that one neural system exerts over another. 9 EC can provide crucial knowledge about the direction of information flow and facilitate description of the causality of interactions among brain regions.9,10
The human brain comprises multiple regions with divergent functions. These regions work together to perform, coordinate, and mediate many higher-order processes by sharing information with each other. Previous studies have demonstrated that MNES have a significant impact on the plasticity of the motor cortex (MC).11-13 Performing more complex tasks may require a higher level of attention and sensorimotor processing to integrate visual and proprioceptive information. 14 The prefrontal cortex (PFC) plays an important role in cognitive control, working memory, and synthesis of diverse information needed for complex behavior. Additionally, it exerts control over a wide range of processes in the service of goal-directed behavior.15,16 The occipital lobe (OL) plays a role in perception of the body parts and can be modulated by retinal stimulation attention, working memory, and integration of stimuli from different sensory modalities.17-19
Stroke causes complex changes in the brain, including altered cortical excitability and cortical connectivity.20-22 The functional recovery after stroke is thought to be a result of the relocation and compensation of functional neural units in the brain.23-25 However, the plastic changes among the associated cortex in stroke patients in response to MNES have not been entirely illustrated. This study aimed to investigate MNES-induced changes in brain functional reorganization in stroke patients by using the fNIRS method. We hypothesized that MNES has the potential to induce plastic reconfiguration in the cortical network in stroke patients.
Methods
Participants
A total of 23 ischemic stroke patients with right hemiplegia were recruited from Qilu Hospital of Shandong University to participate in this study (for participant characteristics see Table 1). Inclusion criteria were as follows: (1) imaging (computed tomography or magnetic resonance imaging of the brain) confirmed an ischemic stroke (details on the imaging can be found in Supplement 1), (2) 2 weeks from the onset of stroke, (3) unilateral lesions, (4) age >35 years, and (5) primary school education or above. Exclusion criteria were as follows: (1) any clinically significant or unstable medical disorder; (2) any neuropsychiatric comorbidity other than stroke; (3) schizophrenia, bipolar, obsessive compulsive, or personality disorders and major depression; (4) skin lesions, infections, hyperalgesia, and intolerance in stimulating areas; and (5) patients or their family members not signing informed consent. Experiments were conducted with the understanding and written consent of each participant. The experimental procedure was approved by the Human Ethics Committee of National Research Center for Rehabilitation Technical Aids and was in accordance with the ethical standards specified by the Helsinki Declaration of 1975 (revised in 2008).
Patient Characteristics.
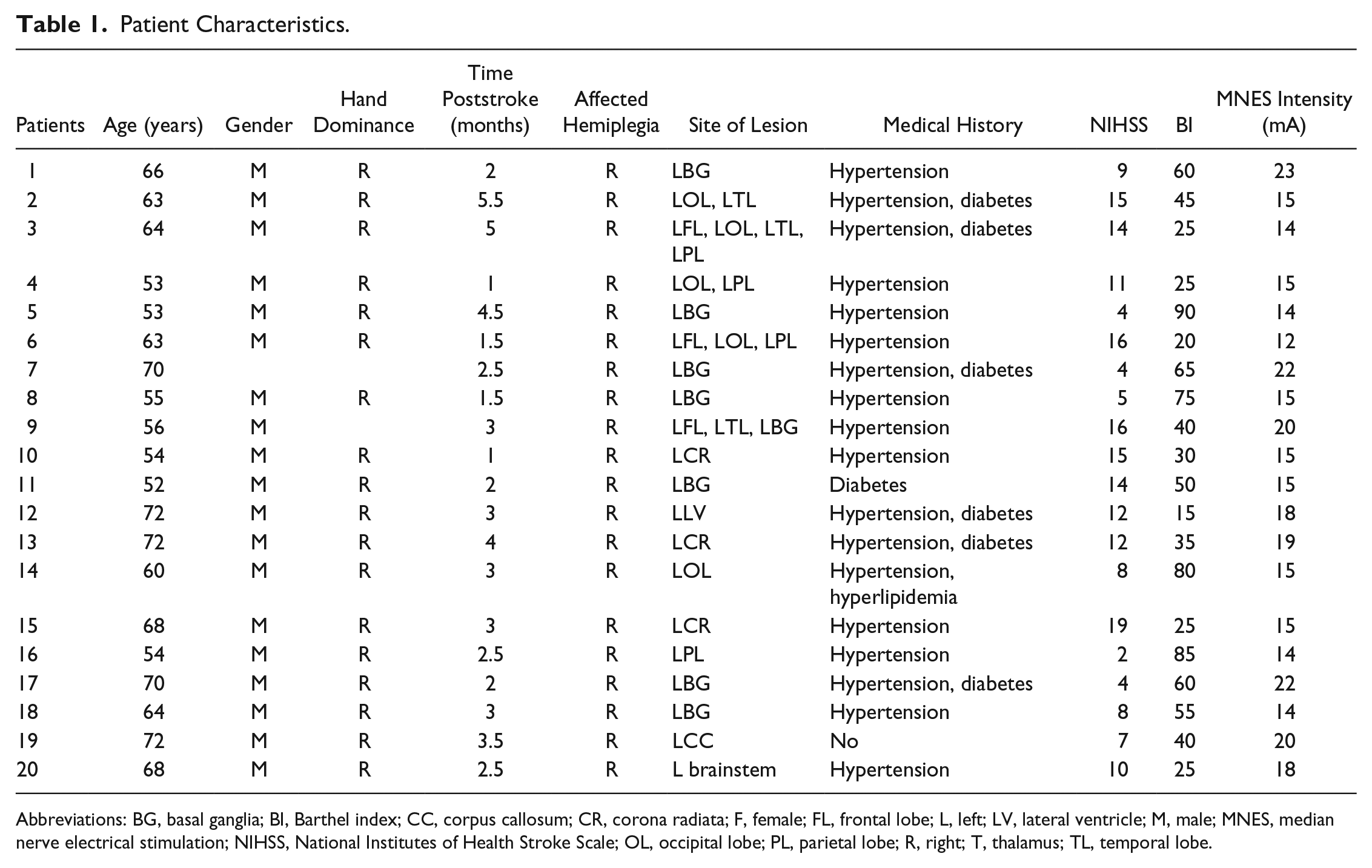
Abbreviations: BG, basal ganglia; BI, Barthel index; CC, corpus callosum; CR, corona radiata; F, female; FL, frontal lobe; L, left; LV, lateral ventricle; M, male; MNES, median nerve electrical stimulation; NIHSS, National Institutes of Health Stroke Scale; OL, occipital lobe; PL, parietal lobe; R, right; T, thalamus; TL, temporal lobe.
Functional Near-Infrared Spectroscopy
A continuous-wave fNIRS system (Nirsmart, Danyang Huichuang Medical Equipment Co, Ltd, China) using light of 760 and 850 nm wavelength was utilized to measure the changes in concentrations of oxygenated hemoglobin (Δ[oxy-Hb]) with a sampling rate of 10 Hz. A total of 32 channels set up by 20 source optodes and 10 detector optodes were symmetrically positioned over the regions of the left and right PFCs (LPFC/RPFC), MCs (LMC/RMC), and OLs (LOL/ROL). The calibration function of the instrument and corresponding template were used to ascertain the channels to fill exactly in correspondence of the 10/10 electrode positions with different head sizes of the participants, as shown in Figure 1A. To prevent interference of ambient light on optical signals, the participants wore a black hood after the probes were attached to the head.
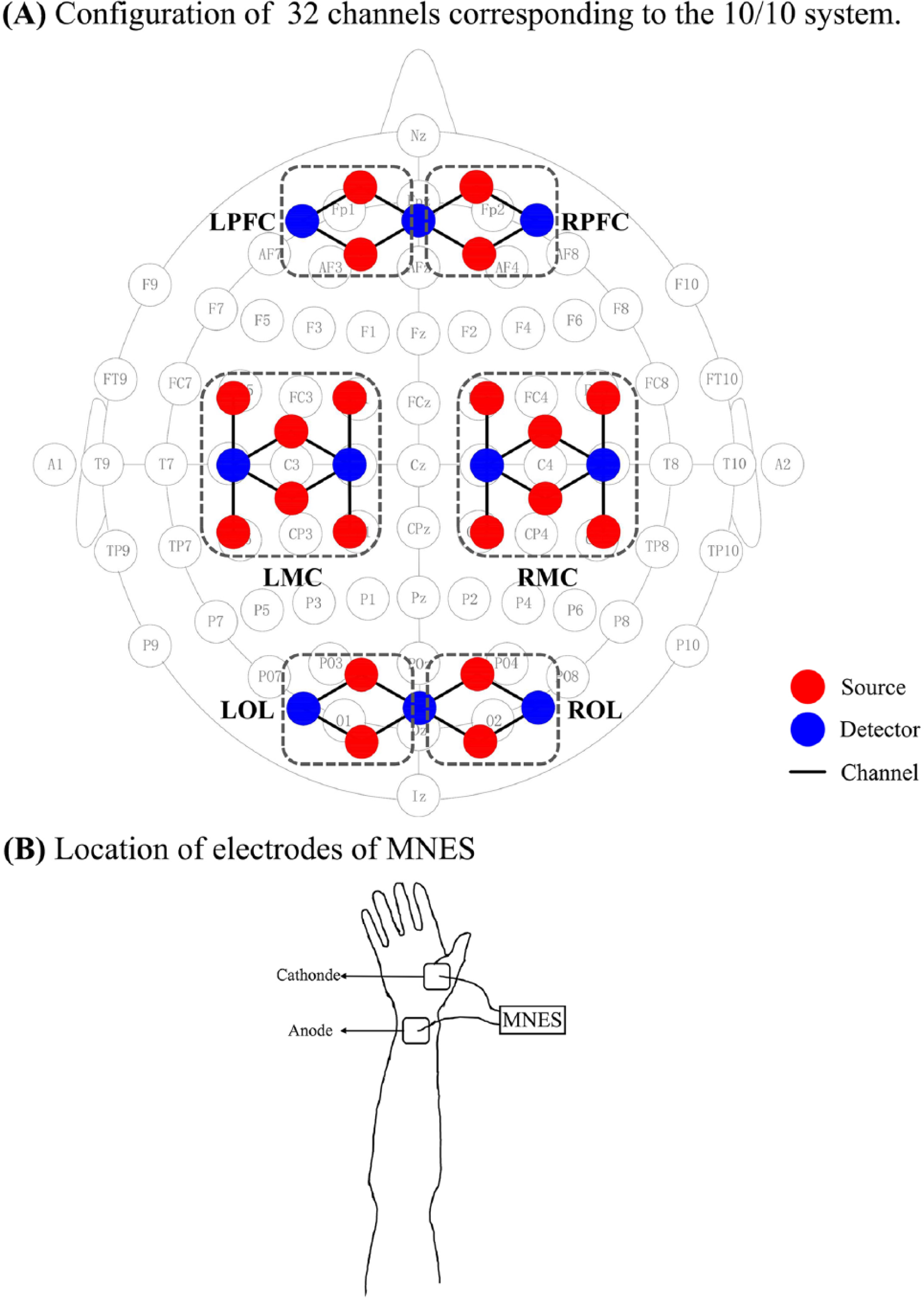
Schematic diagram of the experimental setup: A. Configuration of source optodes, detector optodes, and channels: 32 channels corresponding to the 10/10 system. Six cerebral areas are separated by the rectangular frame as LPFC, RPFC, LMC, RMC, LOL, and ROL. B. Location of electrodes of MNES: the low-frequency electrical pulses from 2 transcutaneous electrodes are transmitted along the wrist to the median nerve.
Experiment
The fNIRS measurement was conducted under 2 conditions: the resting state (10 minutes) and the MNES state (10 minutes). In both the resting state and MNES states, the participants were instructed to keep still with their eyes closed, relax their mind, and remain as motionless as possible. The participants were seated in a silent room with dim lighting. In the MNES state, weak electric shocks were applied to the right wrist above the median nerve, as shown in Figure 1B. The stimulation (rectangular pulse, 50 Hz) was delivered with a 40-s on 20-s off cycle for 10 minutes, and the intensity was set at the highest tolerable level for the patients (mean 16.8 ± 3.1 mA) and at a level sufficient to create a visible muscle twitch or contraction without pain. The whole hand and forearm were relaxed throughout the MNES period.
Data Preprocessing and Wavelet Transform
The absorbance signals recorded by fNIRS were first low-pass filtered at 2 Hz (zero-phase, sixth-order Butterworth filter) to reduce the high-frequency interference, such as instrument noise. Then, Δ[oxy-Hb] signals of channels were calculated from changes in detected light intensity according to the modified Beer-Lambert law. 26 The first 2 minutes of data were excluded for the converted Δ[oxy-Hb] signal to reach a steady state, with 4800 remaining time points for each patient. Data preprocessing was performed on Δ[oxy-Hb] signals using the following procedures: (1) The Δ[oxy-Hb] signals were visually inspected to detect motional artefacts. Three participants were excluded from subsequent analysis because of excessive head artifact interference in their signals. (2) The moving average method was adopted to eliminate the obvious abnormal points in the signal. 27 A method based on moving SD and cubic spline interpolation was then applied to remove motion artifacts. 28 (3) Independent component analysis (ICA) analysis was then performed on Δ[oxy-Hb] signals of each channel to reduce physiological interference in fNIRS measurements, including cardiac pulsations, respiratory signals, and blood pressure changes. 29 All the ICA-derived components were visually inspected to determine the components that might be related to noise and artifacts. The components of interest were visually identified according to the criteria that the associated time course should have a prominent low-frequency (around the band of 0.01-0.08 Hz) spectrum, indicating the functional hemodynamic response in the brain. 30
Continuous wavelet transform (WT) can project signals from the time to the time-frequency domain and enable us to continuously derive the frequency content in time by adjusting the length of wavelet windows. 31 The WT of a signal g(u) is defined as
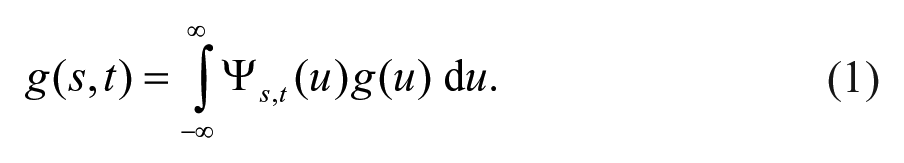
For our analysis, the Morlet wavelet was used as the mother wavelet for WT because its Gaussian envelope provides good localization of events in both time and frequency. 32 “s” Is the scaling factor. In this work, oscillators with phase dynamic information of a specific frequency band (0.01-0.08 Hz) have been distinguished by WT.
Effective Connectivity and Surrogate Test
The dynamical interactions between a pair of oscillators can be described by the corresponding coupling function.33,34 A widely used approach for studying the coupling functions between interacting oscillators is based on their phase dynamics. 35 Based on the phase dynamics of oscillators derived from the chosen frequency band, a coupled-phase-oscillator model between each channel pair was established. Dynamic Bayesian inference (DBI) was applied to extract the optimal set of parameters for describing the model. 36 The directed coupling strength σ i,j (the influence of oscillation i on oscillator j) and σ j,i (the influence of oscillation j on oscillator i) of all possible pairs of 32 channels were determined by DBI. A surrogate test was then applied to ascertain whether the detected coupling parameters are genuine or spurious. In this study, the significant channel-wise ECs were calculated in each participant and condition to describe the MNES-related changes in cortical directed interactions. (Details on the EC method and surrogate test can be found in Supplement 1.)
Region-Wise EC and Main Coupling Direction
To more clearly characterize EC among the 6 brain regions, the significant coupling parameters derived from all possible pairs of 32 channels were averaged as more than 30 directed interregional connections, thus corresponding to mutual interactions among the 6 regions: LPFC→RPFC, LPFC→LMC, LPFC→RMC, LPFC→LOL, LPFC→ROL, RPFC→LPFC, RPFC→LMC, RPFC→RMC, RPFC→LOL, RPFC→ROL, LMC→LPFC, LMC→RPFC, LMC→RMC, LMC→LOL, LMC→ROL, RMC→LPFC, RMC→RPFC, RMC→LMC, RMC→LOL, RMC→ROL, LOL→LPFC, LOL→RPFC, LOL→LMC, LOL→RMC, LOL→ROL, ROL→LPFC, ROL→RPFC, ROL→LMC, ROL→RMC, ROL→LOL (region 1→region 2, region 1 was the source of directed influence, and region 2 was the target of the directed influence).
In the coupling parameters of each channel pair, when the value of σ i,j exceeded σ j,i , we defined i→j as the main coupling direction (mCD) of the interaction between oscillator φ i and oscillator φ j . There exists a mCD for every significant interaction of all possible channel pairs between 2 brain regions, either from region 1 to region 2 or from region 2 to region 1. The distribution of mCD of all channel pairs between the 2 brain regions was collected and analyzed at the group level. Binomial tests were performed on the distribution of mCDs between each of the 2 brain regions to determine whether there is a significant difference in the interregional coupling direction. A significant difference suggests the presence of interregional mCD, which indicates that there is a dominant coupling direction between the 2 brain regions; otherwise, it was considered bidirectional coupling between the 2 brain regions. Bidirectional connection between the 2 brain regions illustrates absence of dominant functional source of coupling. Thus, the 2 brain regions regulate each other. In contrast, unidirectional connection indicates that there is a dominant role in the coupling function between the 2 brain regions. One of the 2 brain regions serves as the predominant functional source of coupling and plays a major regulatory role, whereas the other region acts as the target of regulation.
Statistical Analysis
The Kolmogorov-Smirnov and Levene tests were applied to test the variance normality and homogeneity of the data at the group level. Significant differences for the interregional coupling strength between the resting state and MNES state were assessed by 1-way ANOVA. Differences were considered significant if their P value was less than .05. χ2 Tests were calculated to identify statistically significant differences for the interregional coupling direction between the resting state and MNES state. A difference of P <.05 was considered statistically significant.
Result
MNES-Related Changes in EC Among Regions
Figure 2 shows the MNES-related changes in region-wise EC in stroke patients. Compared with the resting state, the EC values from LPFC, RPFC, LMC, and RMC to LOL show an increasing trend in the MNES state. The significantly increased EC values were found in connectivity of LPFC→LOL (P = .048) and RPFC→LOL (P = .002). The EC values from LMC, RMC, LOL, and ROL to RPFC were increased during the MNES state compared with the resting state, which were significantly increased in connectivity of LMC→RPFC (P = .037) and LOL→RPFC (P = .032). Compared with the resting state, the EC from the contralesional MC to ipsilesional MC (P = .027) was significantly decreased during the MNES state compared with the resting state.
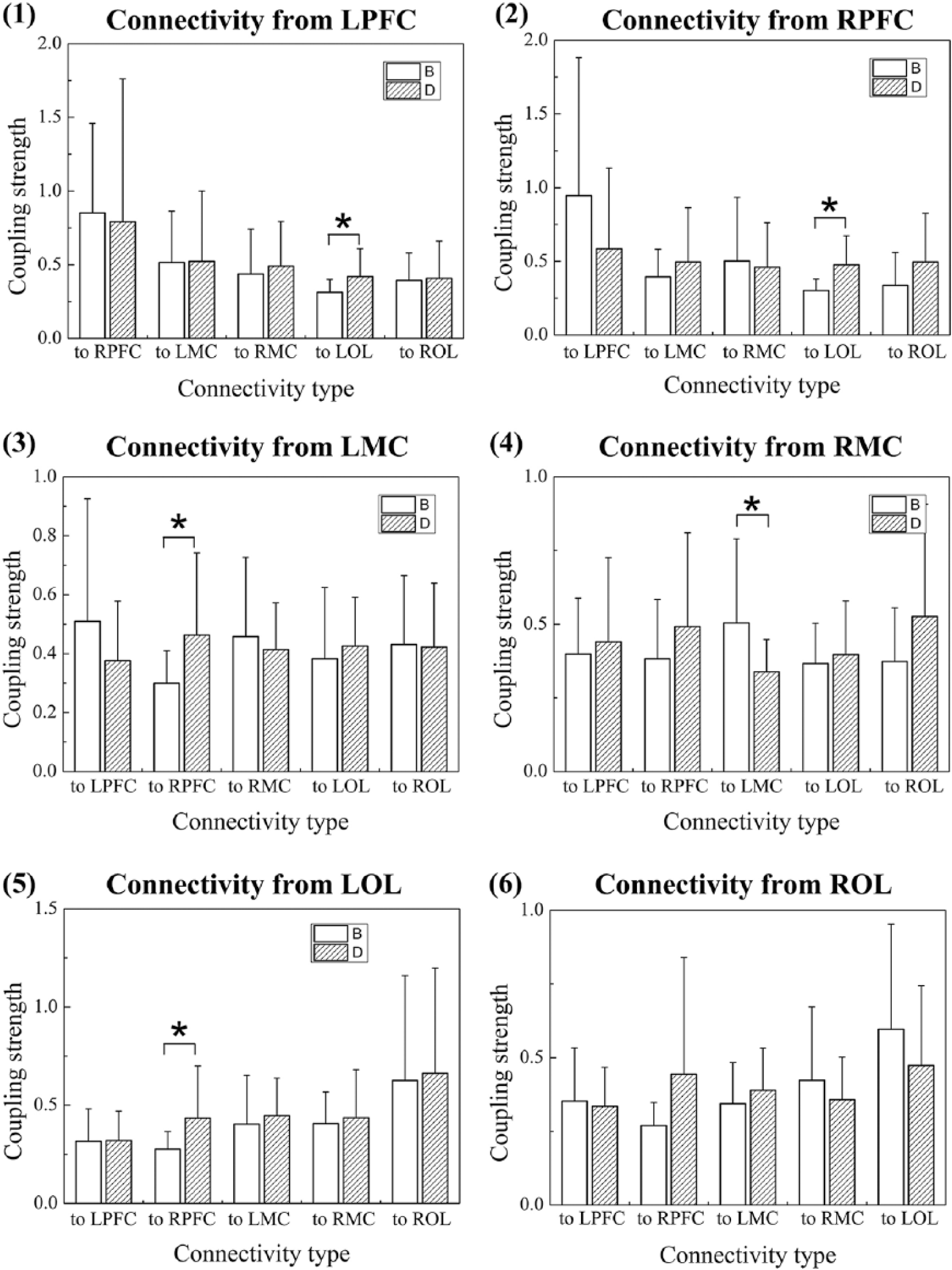
Comparison of region-wise EC between the resting state and the MNES state: (1) MNES-related changes in EC from LPFC to RPFC, LMC, RMC, LOL, ROL, respectively; (2) MNES-related changes in EC from RPFC to LPFC, LMC, RMC, LOL, ROL, respectively; (3) MNES-related changes in EC from LMC to LPFC, RPFC, RMC, LOL, ROL, respectively; (4) MNES-related changes in EC from RMC to LPFC, RPFC, LMC, LOL, ROL, respectively; (5) MNES-related changes in EC from LOL to LPFC, RPFC, LMC, RMC, ROL, respectively; (6) MNES-related changes in EC from ROL to LPFC, RPFC, LMC, RMC, LOL, respectively.
MNES-Related Changes in the Interregional mCD
Figure 3 illustrates the interregional coupling direction among the 6 brain regions in the resting state and the MNES state. The mCD in the resting state was mostly observed from RPFC to LPFC, LMC, and RMC and from LMC and RMC to LPFC. These results indicate that the contralesional PFC plays a main regulatory role and the ipsilesional PFC acts as the target of regulation of motor areas in the brain network in the resting state. In responding to MNES, the coupling directions between RPFC and LPFC, between RPFC and LMC, and between RPFC and RMC were transformed from unidirectional to bidirectional connections. In the MNES state, the interregional mCD was mostly observed from LPFC to LMC, RMC, LOL, and ROL. The interregional coupling direction between PFC and OL was mostly shifted from bidirectional to unidirectional connection in responding to MNES. In the MNES state, the interregional mCD was mostly observed from the prefrontal to occipital areas. In addition, stroke patients exhibited a mCD from LOL to LMC in the MNES state.
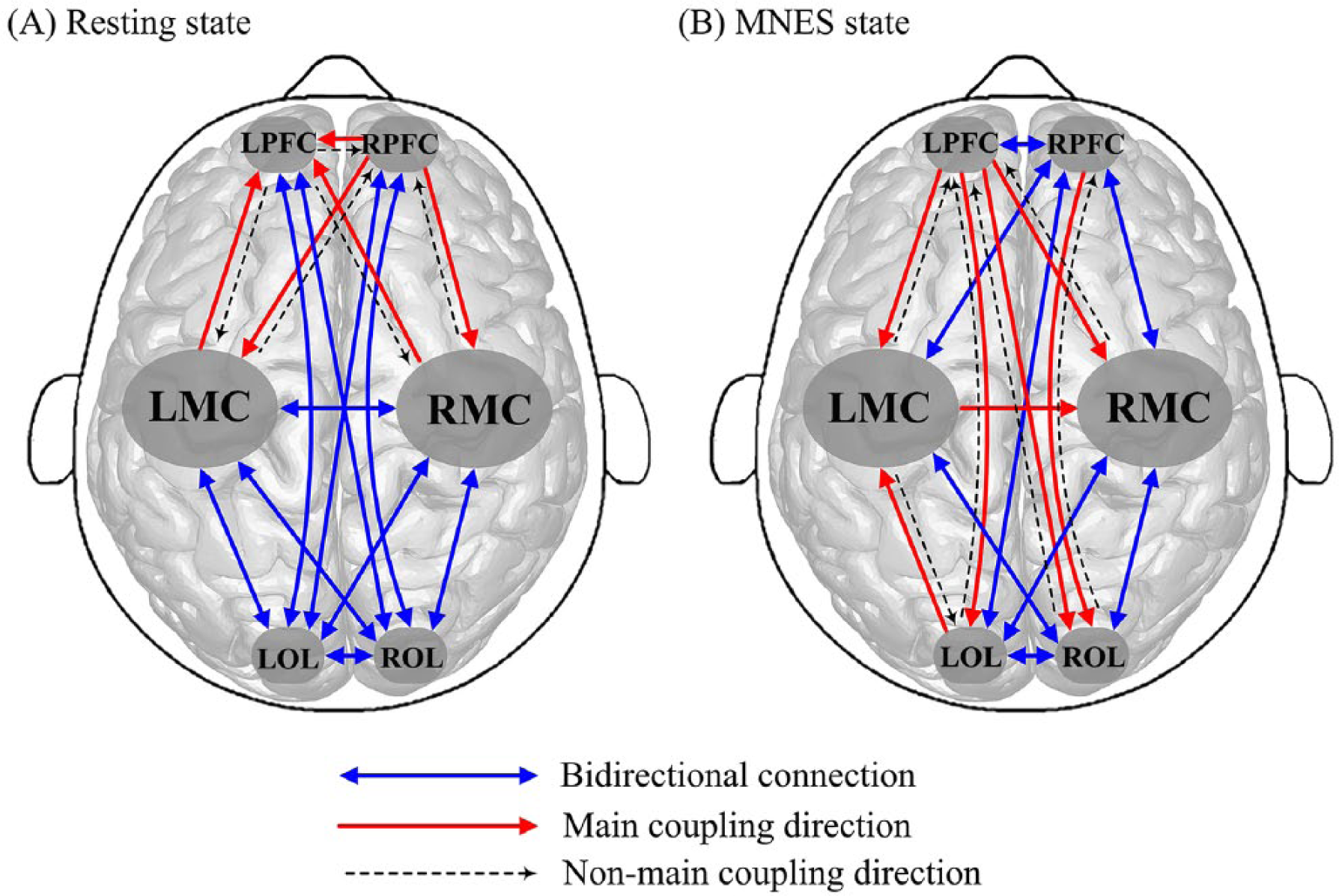
mCDs among the brain regions in the resting state (A) and the MNES state (B).
Table 2 shows the results of MNES-related changes in the distribution of mCDs among the 6 brain regions. In each condition, the distribution statistics of coupling direction of all possible channel pairs between 2 brain regions and its corresponding significance P calculated using the χ2 test are listed to quantitatively analyze MNES-related changes in the interregional coupling direction. Results show that there is significant difference in the distribution of coupling direction between the resting state and the MNES state in the following connections: LPFC-RPFC (P = .014), LPFC-LMC (P < .001), LPFC-RMC (P < .001), LPFC-LOL (P = .008), LPFC-ROL (P = .006), LMC-LOL (P = .01), and LMC-ROL (P = .027). The number of interchannel mCDs significantly increased in connectivity directed from the LPFC to RPFC, LMC, RMC, LOL, ROL and directed from the LOL and ROL to LMC in responding to MNES in stroke patients.
Statistical Result of the Distribution of mCDs Among the 6 Brain Regions. a
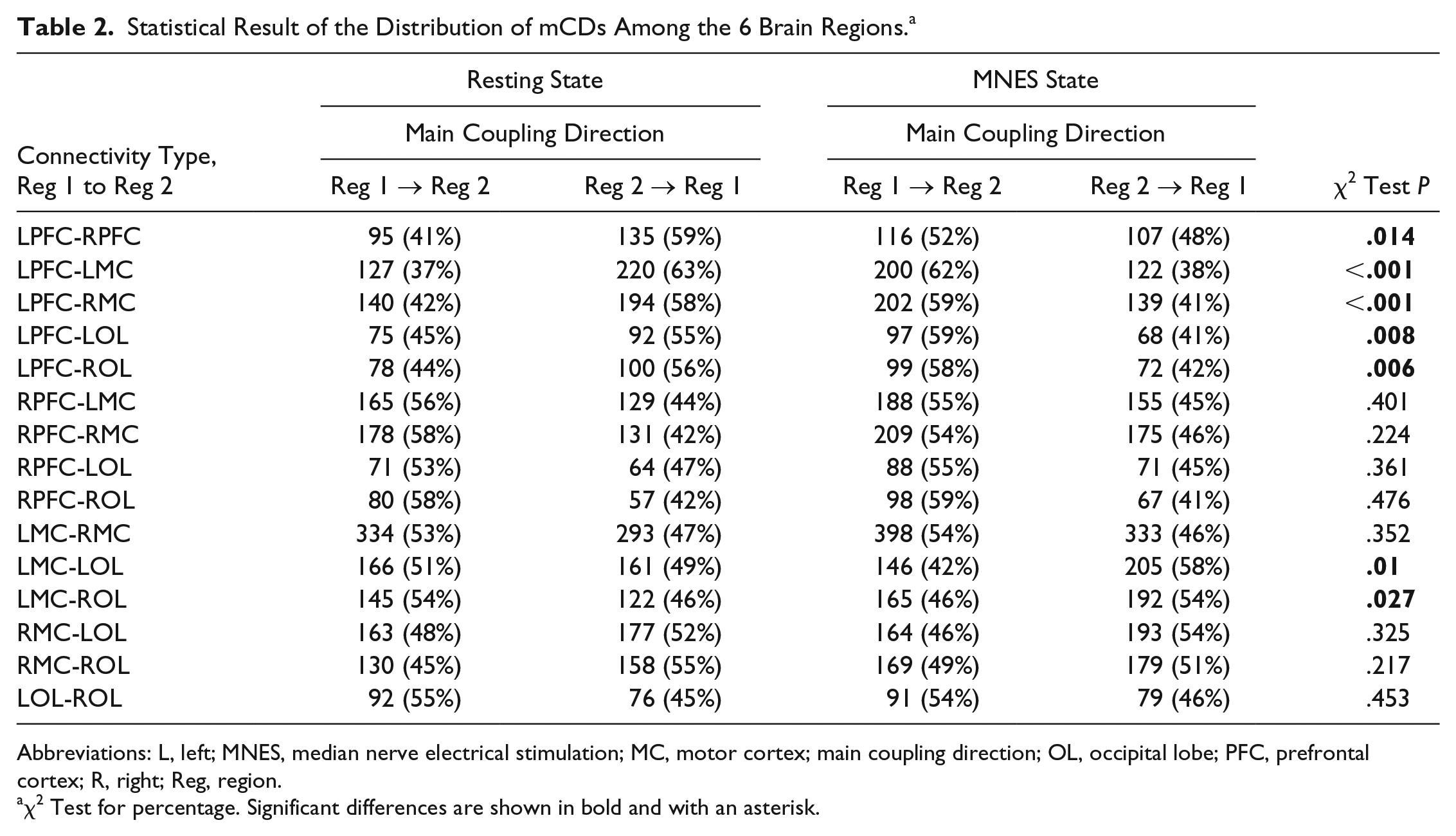
Abbreviations: L, left; MNES, median nerve electrical stimulation; MC, motor cortex; main coupling direction; OL, occipital lobe; PFC, prefrontal cortex; R, right; Reg, region.
χ2 Test for percentage. Significant differences are shown in bold and with an asterisk.
Discussion
The present study based on fNIRS measurements mainly investigated the effects of MNES on the cortical EC network calculated by coupling function and DBI. Significant MNES-related changes in coupling strength and mCD among the cortical areas were observed in stroke patients. The main findings of this study were as follows: (1) MNES triggered significantly increased coupling strength from prefrontal areas to LOL and from the LMC and LOL to RPFC in stroke patients compared with the resting state, whereas significantly decreased coupling strength was observed in the MNES state from the contralesional MC to ipsilesional MC. (2) The interchannels mCD directed from the ipsilesional PFC to contralesional PFC and motor and occipital areas and directed from bilateral occipital areas to the ipsilesional MC significantly increased in responding to MNES in stroke patients. In the MNES state, the interregional mCD was mostly observed from LPFC to LMC, RMC, LOL, and ROL and from the LMC to RMC and LOL. (3) fNIRS-based EC can detect the MNES-related changes in brain reorganization in patients with stroke.
Recent advances in modeling EC enabled inferring causality of interactions among cortical areas and can provide crucial knowledge about the direction of information flow. 9 In particular, the method based on coupling function and DBI has recently attracted considerable attention to calculate dynamical interactions between oscillators. It can describe the physical rule specifying how the functionality of the interactions occur, more than being just a way of investigating correlations and statistical effects.33,34 In this study, oscillations with phase dynamic information of the characteristic frequency band (0.01-0.08 Hz) have been identified in [oxy-Hb] signals by WT, which might reflect the functional hemodynamic response in the brain. A coupled-phase-oscillator model between each channel pair was established according to the phase dynamics. DBI is a promising approach to exploit EC to determine the influence of one oscillator over another under a particular model of causal dynamics.36,37 The information provided by EC allows us to investigate the specific role of a cortical region during a given condition and understand the neural underpinnings of cerebrovascular disorders.
Functional recovery after stroke is now widely considered to be a consequence of central nervous system reorganization. 38 Reorganization of activity in existing cortical/subcortical networks in a way can largely promote functional improvement after stroke. 39 Previous studies have demonstrated that peripheral neuromuscular electrical stimulation increased corticospinal excitability.11,12,40 In the present study, significantly higher EC values were found from the LPFC and RPFC to LOL during the MNES state compared with the resting state. Furthermore, the neural coupling from the LOL to RPFC was significantly higher in the MNES state than in the resting state. These results indicate that MNES can facilitate functional interactions between prefrontal and ipsilesional occipital areas. Combining the results of interregional mCD, we found that the coupling direction between bilateral prefrontal and occipital areas shifts from bidirectional to unidirectional in response to MNES. In the MNES state, interregional mCD was mostly observed from bilateral prefrontal areas to occipital areas. These results suggest that application of MNES could promote the regulation function of prefrontal for occipital areas in the cortical network. Prefrontal areas play a central role in the synthesis of diverse information needed for complex behavior, 16 and the OL is related to the perception of body parts during sensory stimulation. 18 MNES as a form of peripheral nerve stimulation targets the sensory system and can produce strong sensory afferent input. The MNES-related increased interaction between prefrontal areas and the OL in stroke patients might play an important role in integrating the spatial attention and sensory-motor signals involved in the representation of the observer’s body. 19 Performing more complex motor tasks may require a higher level of attention and sensorimotor processing to integrate visual, proprioceptive, and somatosensory feedback information with motor output.
In the resting state, the interregional mCD mostly observed from the RPFC to LPFC, LMC, and RMC suggests that the contralesional PFC plays a crucial regulatory role in the cortical network in stroke patients. Furthermore, the prefrontal area in the affected side was mainly modulated by bilateral motor areas after stroke in the resting state. Significantly increased neural coupling from the LMC to RPFC observed in stroke patients in responding to MNES indicates an increased influence from the LMC to RPFC following unilateral MNES stimulation utilizing EC analysis. The increased influence from motor areas to RPFC induced by MNES results in the absence of mCD between the contralesional PFC and motor areas during the MNES state. In responding to MNES, interregional coupling direction related to ipsilesional prefrontal areas was mostly transformed into the mCD from the ipsilesional prefrontal to the bilateral motor and occipital areas. These results suggest that the MNES could promote the regulation function of ipsilesional prefrontal areas in the cortical network. A previous neuroimaging study has demonstrated that the movements of the stroke-affected hand were associated with more widespread activation clusters in the lesioned hemispheres, extending into the frontal and parietal areas. 41 In summary, our data showed that there was MNES-related increased modulation of the ipsilesional prefrontal area in the cortical network, which has been shown to play a key role in cognitive control and in the synthesis of diverse information for goal-directed behavior. 16
Stroke may alter interhemispheric balance such that there is an abnormally high interhemispheric inhibitory drive from contralesional to ipsilesional motor areas, thereby contributing to the impaired motor function of the paretic hand.38,42 Additional inhibitory influences from the contralesional to ipsilesional M1 correlated with the degree of motor impairment when there were movements of the stroke-affected hand. 41 In the present study, the affected hand of stroke patients produced a visible muscle twitch or contraction according to the stimulation cycle during the MNES state. In the analysis of cerebral EC network, we found a significantly lower EC from contralesional to ipsilesional motor areas in the MNES state than in the resting state. Compared with the resting state, interhemispheric influence driving from contralesional to ipsilesional MC significantly decreased during the MNES state with movement of the affected hand. MNES can trigger sensorimotor stimulations of the stroke-affected hand involving a decreased neural coupling of the contralesional motor area on the ipsilesional MC. In addition, the interregional modulation of connectivity between LMC and LOL shifts from bidirectional to unidirectional regulation following unilateral MNES. Our results suggest that MNES can facilitate the driving input exerted by occipital on the ipsilesional motor area involved in the movement of the stroke-affected hand during MNES. The occipital-MC interaction might be crucial for the integration of spatial attention and sensory-motor signals involved in the stimulation of the affected hand of stroke patients.18,43
Conclusions
The present study demonstrated that fNIRS-based EC allows the assessment of MNES-related changes in functional network among the cortical areas in stroke patients. Results revealed that MNES can trigger sensorimotor stimulations of the affected hand that sequentially involved distant cortical areas and ipsilesional and contralesional functional reorganization. The neural coupling was significantly increased in stroke patients from the prefrontal areas to occipital areas following unilateral MNES. Interregional mCD was mostly observed from the ipsilesional prefrontal area to bilateral motor and occipital areas in responding to MNES, suggesting that MNES could promote the regulation function of ipsilesional prefrontal areas in the cortical network. Additionally, significantly decreased neural coupling from the contralesional motor area on the ipsilesional MC was found in the MNES state following movement of the affected hand induced by MNES. Collectively, MNES has the potential to redistribute brain resources and induce cortical plastic changes in stroke patients. Investigating changes in cortical connectivity caused by MNES in stroke patients may, therefore, help further our understanding of the neural mechanisms underlying MNES.
Supplemental Material
Supplement – Supplemental material for Median Nerve Electrical Stimulation–Induced Changes in Effective Connectivity in Patients With Stroke as Assessed With Functional Near-Infrared Spectroscopy
Supplemental material, Supplement for Median Nerve Electrical Stimulation–Induced Changes in Effective Connectivity in Patients With Stroke as Assessed With Functional Near-Infrared Spectroscopy by Congcong Huo, Xinglou Li, Jing Jing, Yanping Ma, Wenhao Li, Yanqin Wang, Wanlin Liu, Yubo Fan, Shouwei Yue, Yonghui Wang and Zengyong Li in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by the National Natural Science Foundation of China (NSFC Nos. 61761166007, 31771071, 11732015, and 81672249), Beijing Municipal Science and Technology Project (No. 161100001016013), and Fundamental Research Funds for Central Public Welfare Research Institutes (118009001000160001).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
